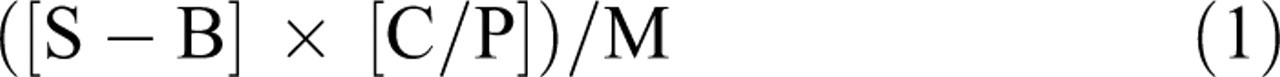Abstract
The goal of our study was to determine whether maternal exposure to red raspberry leaf (RRL) and its constituents can permanently alter biotransformation of fluorogenic substrates by cytochrome P450 (CYP) in the livers of male and female offspring. Nulliparous female rats received vehicle, raspberry leaf, kaempferol, quercetin, or ellagic acid orally once breeding had been confirmed until parturition. Hepatic microsomes were prepared from animals at birth (postnatal day 1 [PND1]), weaning (PND21), PND65, and PND120 to determine the biotransformation of 8 fluorogenic substrates. The pattern of biotransformation of all but 2 of the substrates was gender specific. Maternal consumption of RRL increased biotransformation of 3 substrates by female offspring at PND120 resulting in a more masculine profile. Kaempferol and quercetin had a similar effect to RRL. These results suggest that maternal consumption of either RRL or some of its constituents leads to long-term alterations of CYP activity in female offspring.
Introduction
Fetal and/or neonatal exposure to xenobiotic compounds can permanently alter the activity of the cytochrome P450 (CYP) drug-metabolizing enzymes in postnatal life, thus producing permanent changes in the kinetics of drug metabolism of exposed individuals. For instance, in utero exposure to barbiturates induces irreversible elevation of CYP2C6 and 2C7 messenger RNA (mRNA) and protein expression. 1 To date, much of the work on this phenomenon of “imprinting” has focused on the consequences of maternal exposure to pharmaceuticals and chemicals present in the environment including pesticides and air pollution. 2 –4 However, fetuses and neonates are exposed to other xenobiotic compounds as a result of maternal diet and lifestyle choices such as smoking and herbal medicine use.
In recent years, the use of herbal medicines has increased in North America, Europe, and Australasia. As many as 60% of women use natural health products during pregnancy. 5 –8 Examples of products often used during pregnancy are ginger for treatment of morning sickness and white or black cohosh for uterine and labor stimulation. 9 However, the most commonly used herbal during pregnancy is red raspberry leaf (RRL). 10 Red raspberry leaf can be taken as a tea, capsule, or leaf extract for use during all stages of pregnancy and lactation. Widely disseminated information in the lay press recommends the use of RRL to prevent morning sickness and miscarriages, induce labor, and treat menstrual pains 9 but most commonly for its ability to shorten and ease labor. 11 Red raspberry leaf is a rich source of polyphenols including ellagic acid and flavonoids such as quercetin and kaempferol, 12 all of which have been shown to alter the activity of CYP enzymes responsible for drug metabolism and steroid hormone biotransformation. 4,13,14 However, although RRL is commonly used during pregnancy, there is no information available regarding the long-term consequences of fetal exposure to this herbal preparation on postnatal drug-metabolizing enzymes. Therefore, the goal of this study was to determine the effects of maternal RRL consumption on the CYP drug-metabolizing enzymes of the offspring during postnatal development. In addition to examining the whole RRL herbal preparation, we examined whether the constituent polyphenols kaempferol, quercetin, and ellagic acid could also affect these outcomes independently.
Materials and Methods
Materials
All compounds and reagents were purchased from Sigma-Aldrich (Oakville, Ontario, Canada) except 3-[2-(diethylamino)ethyl]-7-hydroxy-4-methylcoumarin hydrochloride (AHMC), 3-[2-(N,N-diethyl-N-methylammonium)ethyl]-7-methoxy-4-methylcoumarin (AMMC), 7-benzyloxy-4-(trifluoromethyl)-coumarin (BFC), 7-benzyloxyquinoline (BQ), 3-cyano-7-ethoxycoumarin (CEC), 3-cyano-7-hydroxycoumarin (CHC), 7-hydroxy-4-(trifluoromethyl)-coumarin (HFC), 7-hydroxyquinoline (HQ), and 7-methoxy-4-(trifuoromethyl)-coumarin (MFC), which were purchased from BD Gentest (Woburn, Massachusetts). Methoxyresorufin (MRES), ethoxyresorufin, (ERES) and 7-ethoxy-4-trifloromethyl-coumarin (EFC) were purchased from Invitrogen (Burlington, Ontario, Canada); NUNC clear 96-well plates and Costar black 96-well plates were purchased from Fisher Scientific (Ottawa, Ontario, Canada). Bio-Rad protein assay reagent was obtained from Bio-Rad Laboratories (Mississauga, Ontario, Canada); NADP from Roche Diagnostics (Laval, Quebec, Canada); RRL (Nature’s Way, Canmore, Alberta, Canada); kaempferol (TCI America, Portland, Oregon); and acetonitrile (AcN), dimethylsulfoxide (DMSO), potassium phosphate, sucrose, and sodium hydroxide, which were of the highest purity available from various suppliers of general reagents located geographically close to McMaster University.
Liver Microsome Preparation
All animal experiments were approved by the McMaster University Animal Research Ethics Board in accordance with the guidelines of the Canadian Council for Animal Care. Nulliparous 200-250 g female Wistar rats (Harlan, Indianapolis, Indiana) were maintained under controlled lighting (12:12 L:D) and temperature (22°C) with ad libitum access to food and water. Dams were randomly assigned to receive vehicle, 10 mg/kg per d RRL (Rubus idaeus), quercetin monohydrate, kaempferol, or ellagic acid orally in a flavored gelatin base, 15 once breeding had been confirmed by the presence of sperm in the vaginal swab (n = 10 per group). Dams were allowed to deliver normally and treatments were terminated on the day of parturition. The recommended human dose for the RRL formulation used in the present study is 1 to 2 capsules of 480 mg/d which would give 7 to 14 mg/kg per day in a 70 kg individual. The content of quercetin, kaempferol, and ellagic acid in RRL is 0.2% to 0.4%, 0.2% to 0.4%, and 2% to 7% of dry weight, respectively. 12 At birth (postnatal day 1 [PND1]), pups were weighed, sexed, and the litters culled to 8 with approximately equal numbers of male and female offspring. At weaning, the males and females were housed separately as sibling pairs.
At PND1 livers from approximately 8 animals were pooled together and at all other ages randomly selected male and female pups (only 1 male and 1 female pup per litter were sampled at each time point to reduce litter effects) were sacrificed at PND21, PND65, and PND120. Liver microsomal membrane suspensions were prepared, protein concentrations determined, and samples were stored at −20°C until used as previously described. 16
Cytochrome P450 Activity in Rat Liver Microsomes
The velocity of biotransformation of the various fluorogenic substrates was determined by kinetic fluorescence intensity assays. Reactions were carried out at 37°C in black, 96-well plates (Costar) in a final volume of 200 μL containing (final concentrations): potassium phosphate buffer pH 7.4 (0.15 mol/L), NADP (330 μmol/L), glucose-6-phosphate (830 μmol/L), MgCl2 (850 μmol/L), glucose-6-phosphate dehydrogenase (70-75 mU/well), substrate (variable), and were started by the addition of microsomal suspension. In all cases, the duration of the incubation and the liver microsomal protein concentrations were in the linear range of metabolite formation. Working conditions for AMMC, BFC, BQ, CEC, EFC, and MFC were as we have described previously. 17 With ERES and MRES, the product formation was monitored every minute for 10 minutes by measuring emission at 590 nm after excitation at 535 nm using the HTS 7000 bioassay reader (PerkinElmer, Waltham, Massachusetts). The liver microsomal protein concentration was 100 μg/well. Reaction velocities (relative fluorescence units [RFU]/min) were calculated using the instrument’s software (SoftMax Pro or HTSoft). Fluorescence intensity was calibrated using authentic AHMC, CHC, HFC, HQ, and resorufin as appropriate.
Data Analysis
The ability of rat liver microsmes to biotransform the 8 fluorogenic substrates was tested in triplicate. The mean fluorescence value of blank was subtracted from the value of each sample using Microsoft Excel. Data were then converted from RFU/min to pmol/min per mg using the following equation:
Statistical Analysis
All statistical analyses were performed using SigmaStat, v2.03 (Systat Software, Chicago, Illinois). Data were tested for normality and equal variance and subsequently analyzed by analysis of variance (ANOVA) followed by a Bonferroni post hoc test. Treatment groups were compared with control groups within each gender and age, and males were compared with females in the control group within each age. The post hoc test corrects for errors resulting from multiple comparisons. Differences were considered significant when P < .05 before correction. None of the data sets failed the normality or equal variance tests. Values are presented as mean ± SEM.
Results
Age- and Gender-Dependent Changes in CYP Activity
Biotransformation of all substrates by liver microsomes from control animals was undetectable at birth. In most cases, it then rose steadily to a peak between 65 and 120 days of age (see Figure 1 for examples). The biotransformation of AMMC, BFC, BQ, EFC, MFC, and MRES exhibited gender-specific patterns of activity (Figure 1). Males biotransformed AMMC at a significantly higher rate than females at PND65 only, thereafter activity declined in both to a common value at PND120. BFC and MFC had very similar patterns of activity, higher activity in males at PND65 and 120 and no significant decline in either gender with aging. BQ was also biotransformed at a significantly greater rate by males on PND65 and 120, but in this case activity in females began to decline after PND21 whereas activity in males rose to a peak at PND65. Biotransformation of EFC was significantly higher in males than in females only at PND120. The resulting pattern for males was for increasing activity right up to PND120 whereas activity in females declined after PND65. Biotransformation of MRES was unique among the substrates tested in that activity was significantly higher in females than males at PND65 and 120. In males demethylation of MRES reached a peak at PND21 and declined to undetectable levels by PND120, whereas the peak in females occurred at PND65 and there was still significant activity at PND120. There was no effect of gender on the biotransformation of CEC or ERES at any age examined (data not shown).
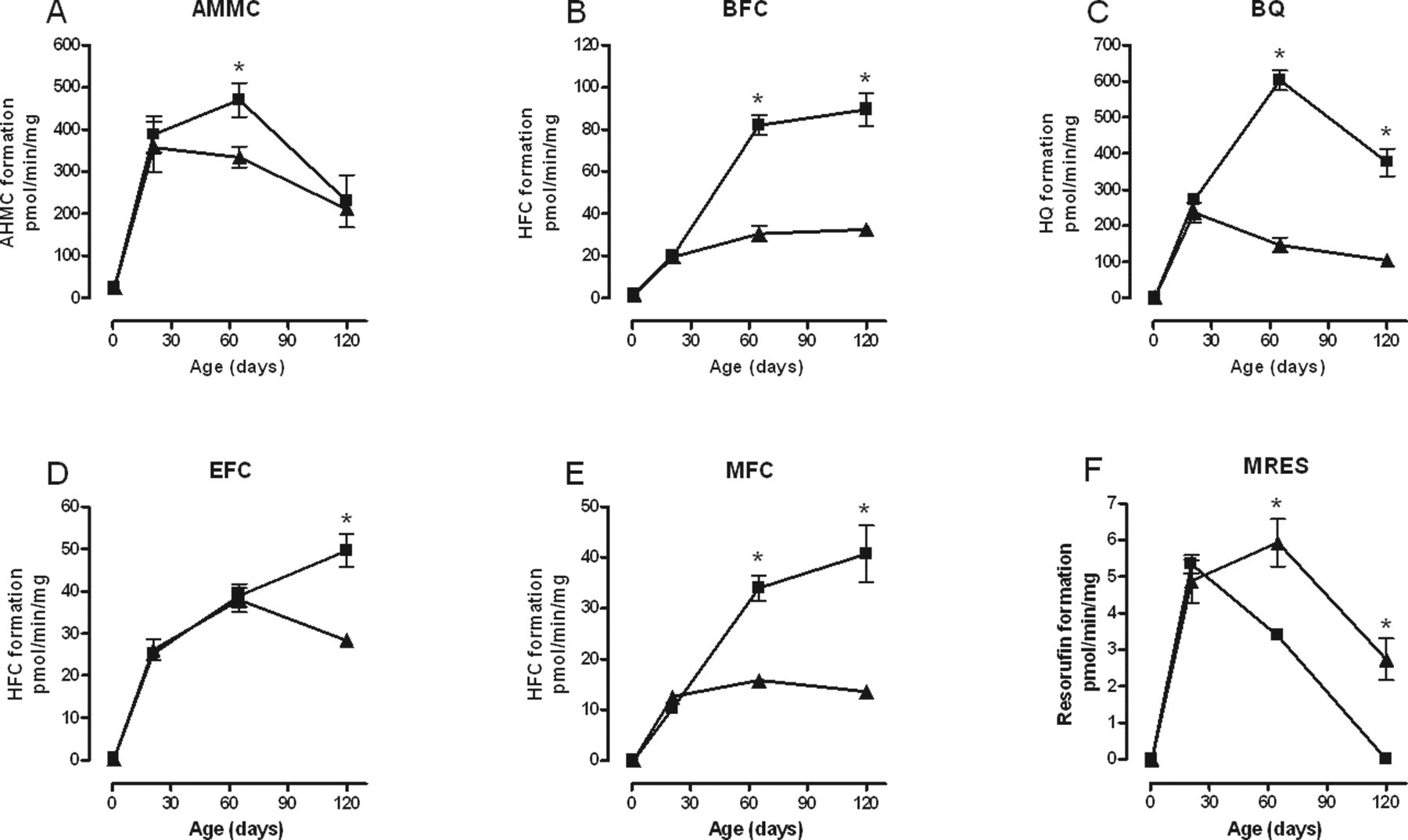
Effect of age on the biotransformation of (A) AMMC; (B) BFC; (C) BQ; (D) EFC; (E) MFC; and (F) MRES by liver microsomes taken from male (▪) and female (▲) control groups. Each point represents the mean ± standard error of measurement from 5 animals, except at PND1 where a maximum of 9 livers were pooled from males and females. Each measurement was performed in triplicate. Values with an asterisk are significantly different (P < .05) between males and females. AMMC indicates 3-[2-(N,N-diethyl-N-methylammonium)ethyl]-7-methoxy-4-methylcoumarin; BFC, 7-benzyloxy-4-(trifluoromethyl)-coumarin; BQ, 7-benzyloxyquinoline; EFC, 7-ethoxy-4-trifluoromethyl-coumarin; MFC, 7-methoxy-4-(trifuoromethyl)-coumarin; MRES, methoxyresorufin; PND, postnatal day.
Effect of Maternal Consumption of RRL, Quercetin, Kaempferol, and Ellagic Acid on CYP Activity
Maternal administration of RRL significantly increased the biotransformation of BFC, EFC, and MFC by liver microsomes of female offspring but not males. These RRL-induced increases in biotransformation of BFC, EFC, and MFC occurred only at PND120 such that rather than reaching a plateau at or declining after PND65, as is seen in control animals, activity in the RRL-exposed animals was still increasing at the last time point measured (Figure 2 ). There was no difference in the biotransformation of AMMC, BQ, CEC, ERES, or MRES in either male or female RRL-exposed offspring (data not shown).
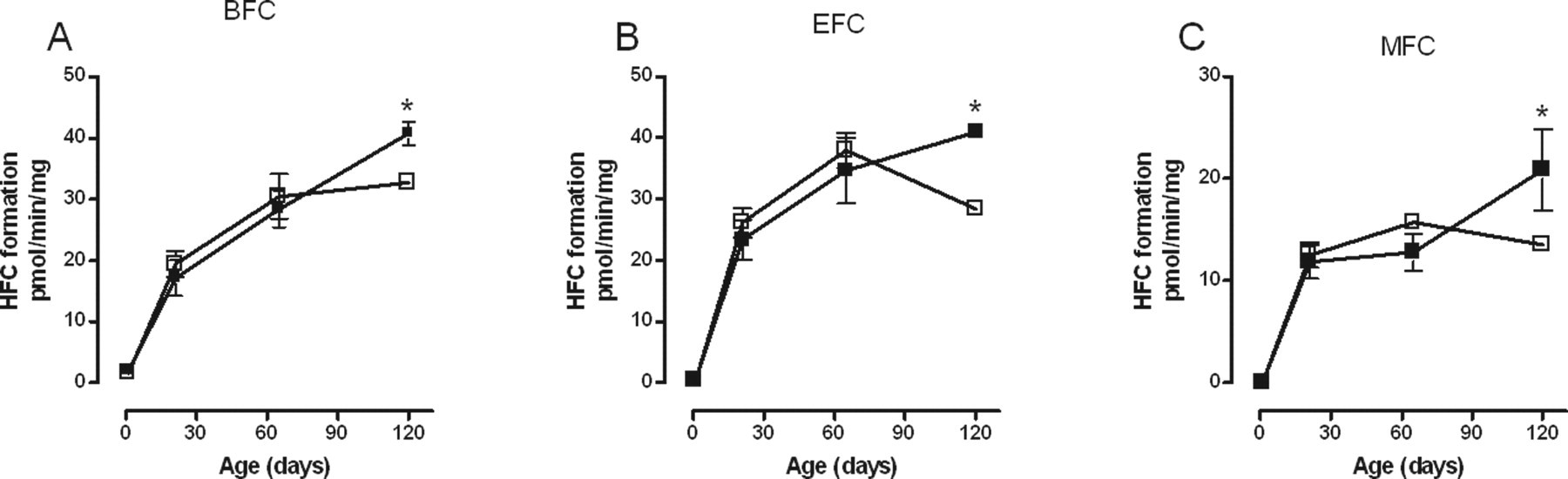
Effect of red raspberry leaf (▪) on the biotransformation of (A) BFC; (B) EFC; and (C) MFC over time by rat liver microsomes from female offspring. Each point represents the mean ± standard error of measurement from 5 animals except at PND1 where a maximum of 9 livers were pooled from males and females. Each measurement was performed in triplicate. Values with an asterisk are significantly different (P < .05) from the control (□) at the same age. BFC indicates, 7-benzyloxy-4-(trifluoromethyl)-coumarin; EFC, 7-ethoxy-4-trifluoromethyl-coumarin; MFC, 7-methoxy-4-(trifuoromethyl)-coumarin; PND, postnatal day.
Maternal quercetin administration produced the same pattern of changes in the biotransformation of fluorogenic substrates by liver microsomes of female offspring as did exposure to RRL, namely increased biotransformation of BFC, EFC, and MFC at PND120. Furthermore, quercetin also affected biotransformation of EFC by male offspring, but in this case activity was significantly increased earlier, at PND65, and not at any other time point (Figure 3 ).
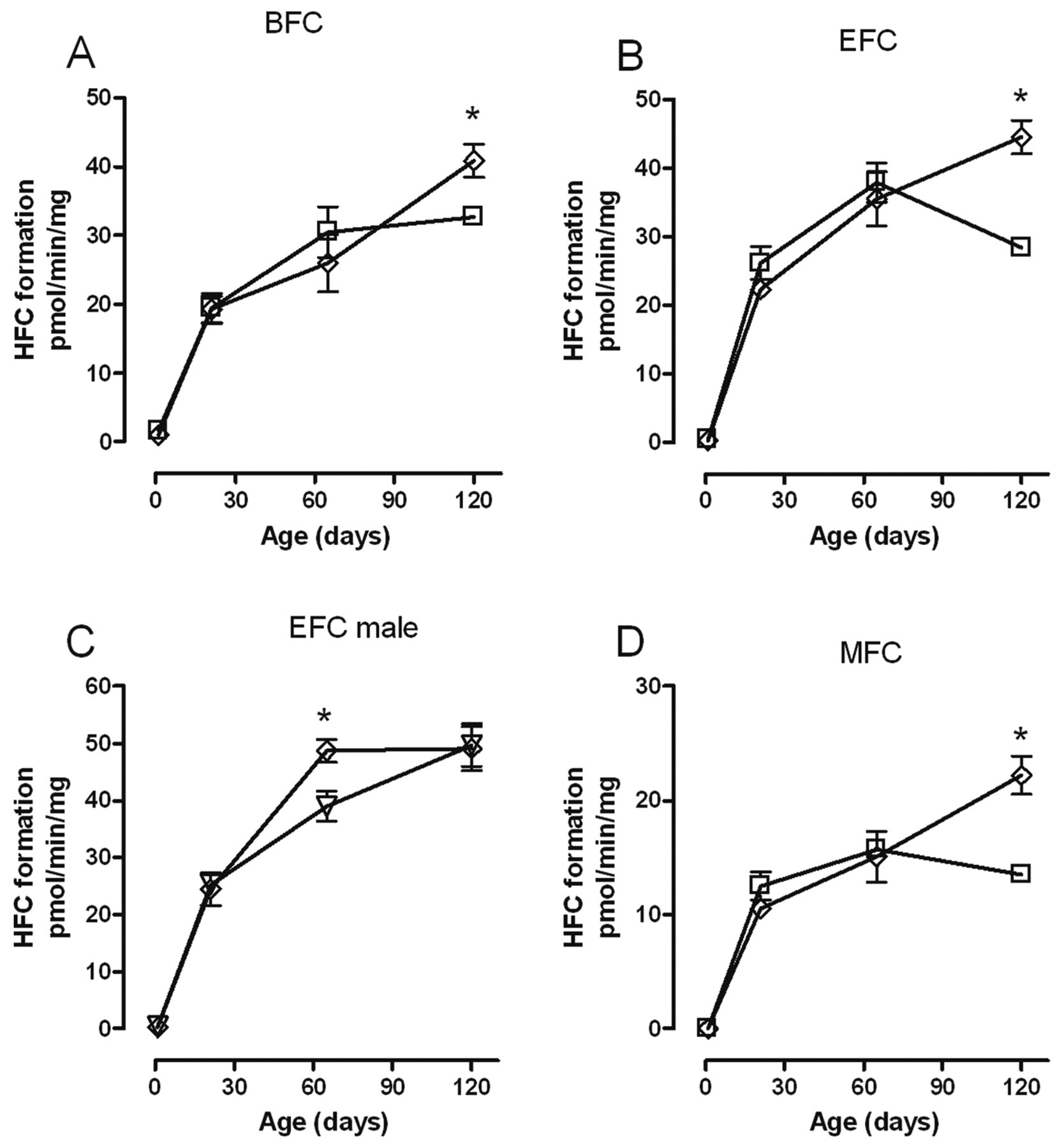
Effect of quercetin (⋄) on the biotransformation of (A) BFC; (B) EFC; (C) EFC; and (D) MFC over time by rat liver microsomes from female (A, B, and D) and male (C) offspring. Each point represents the mean ± standard error of measurement from 5 animals except at PND1 where a maximum of 9 livers were pooled from males and females. Each measurement was performed in triplicate. Values with an asterisk are significantly different (P < .05) from the control females (□) and males (∇) at the same age. BFC indicates 7-benzyloxy-4-(trifluoromethyl)-coumarin; EFC, 7-ethoxy-4-trifluoromethyl-coumarin; MFC, 7-methoxy-4-(trifuoromethyl)-coumarin; PND, postnatal day.
Only female offspring were affected by maternal exposure to kaempferol, and the significant changes were limited to the biotransformation of the substrates EFC and MFC. In both cases, activity was increased at PND120 only, and as with RRL exposure, rather than reaching a plateau at or declining after PND65 as was the case in controls, activity was still increasing at PND120 (Figure 4 ).
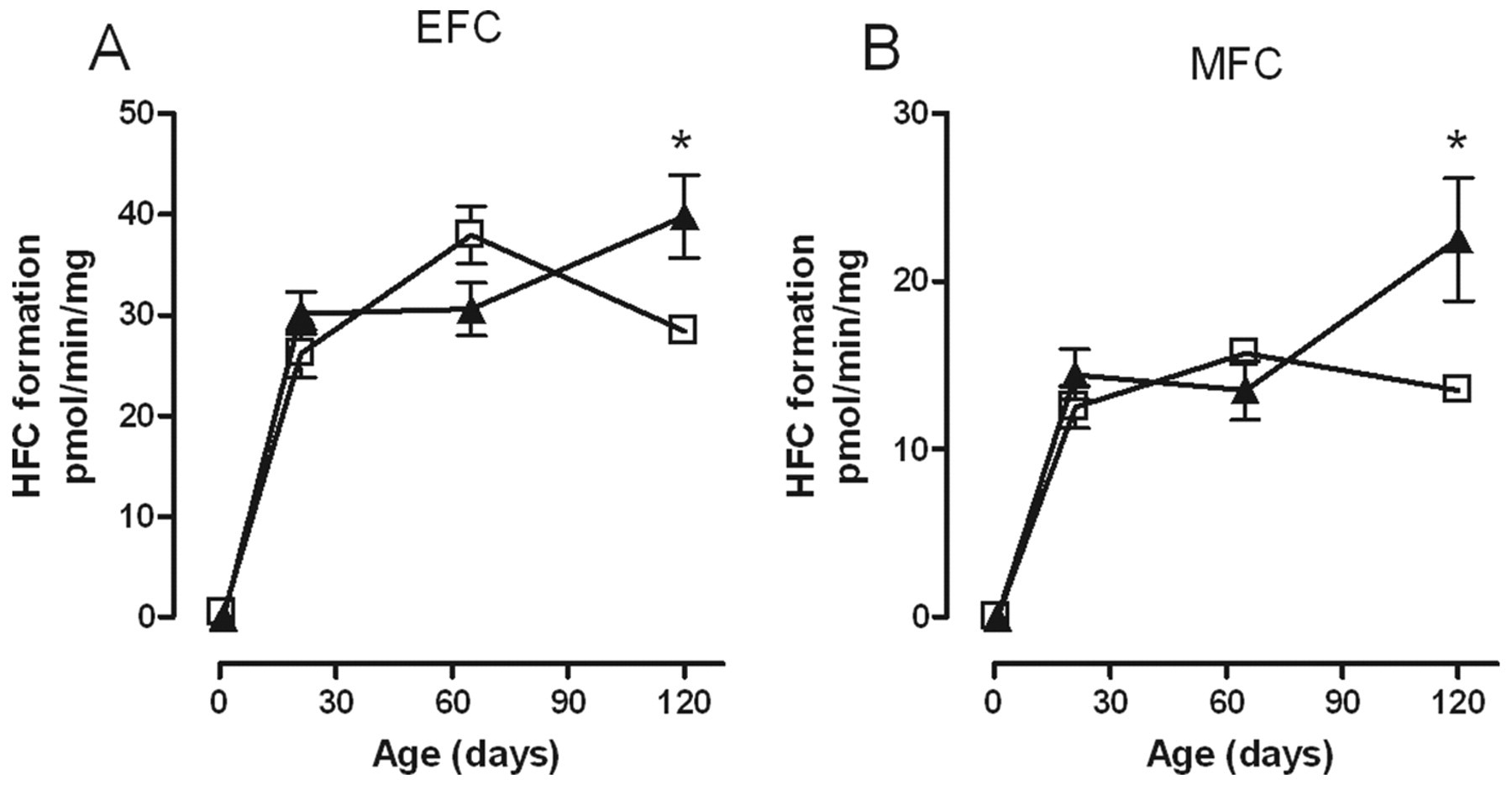
Effect of kaempferol (▲) on the biotransformation of (A) EFC and (B) MFC over time by rat liver microsomes from female offspring. Each point represents the mean ± standard error of measurement from 5 animals except at PND1 where a maximum of 9 livers were pooled from males and females. Each measurement was performed in triplicate. Values with an asterisk are significantly different (P < .05) from the control (□) at the same age. EFC indicates 7-ethoxy-4-trifluoromethyl-coumarin; MFC, 7-methoxy-4-(trifuoromethyl)-coumarin; PND, postnatal day.
Only 2 substrates, BQ and ERES, both of which were insensitive to RRL, quercetin, and kaempferol, were affected by maternal ellagic acid exposure. In female but not male offspring, maternal ellagic acid exposure significantly decreased the biotransformation of ERES in PND21 offspring, the age at which the activity of this enzyme peaks in control animals. In male but not female offspring, ellagic acid significantly increased the biotransformation of BQ at PND65 with a trend to higher activity at PND120 that did not reach statistical significance (Figure 5 ).
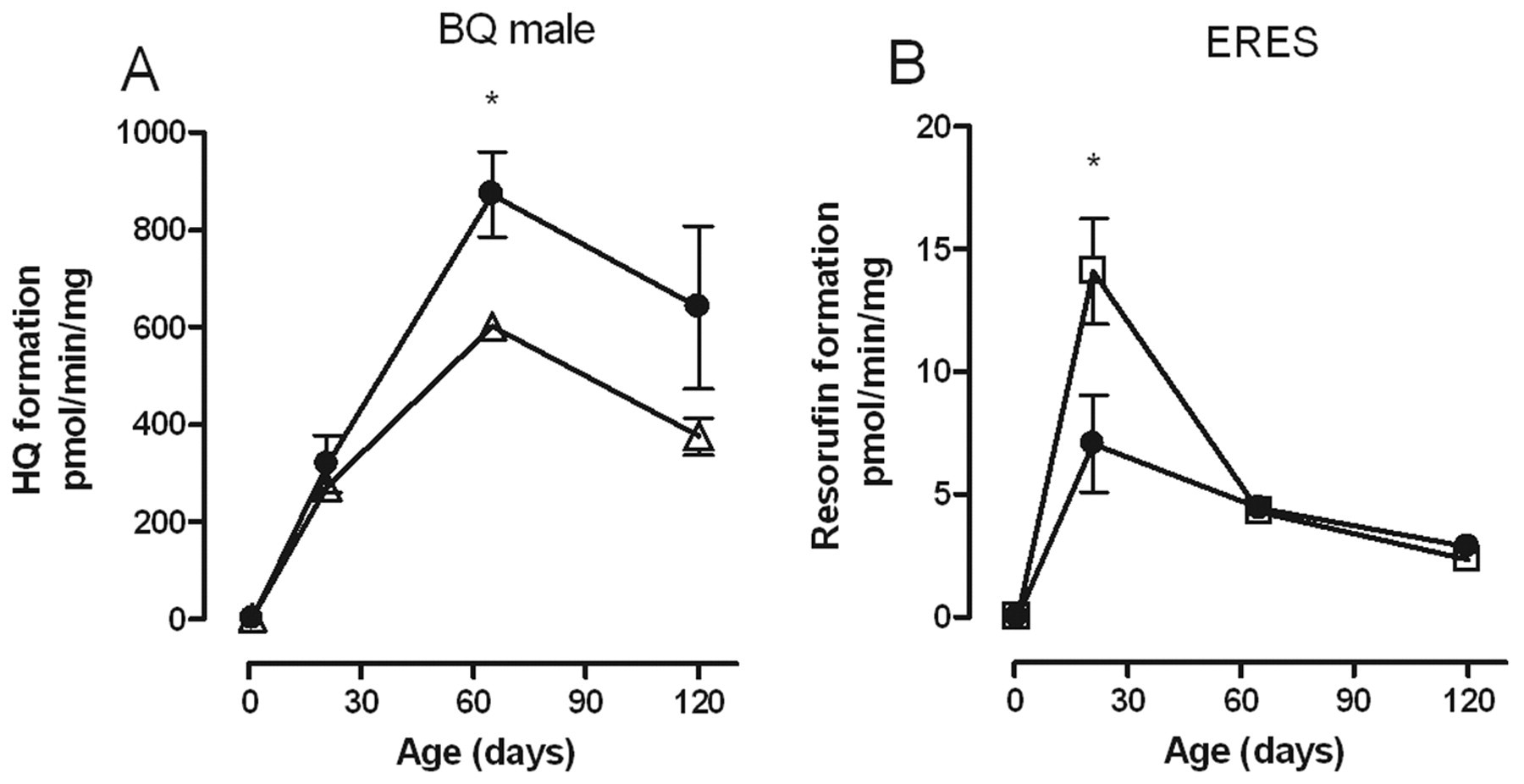
Effect of ellagic acid (•) on the biotransformation of (A) BQ and (B) ERES over time by rat liver microsomes from male (A) and female (B) offspring. Each point represents the mean standard error of measurement from 5 animals except at PND1 where a maximum of 9 livers were pooled from males and females. Each measurement was performed in triplicate. Values with an asterisk are significantly different (P < .05) from control males (Δ) or females (□) at the same age. BQ indicates 7-benzyloxyquinoline; ERES, ethoxyresorufin PND, postnatal day.
These effects of maternal consumption of RRL and some of its constituents on the biotransformation of fluorogenic substrates by liver microsomes from male and female offspring are summarized in Table 1 .
Summary of the Effects of Maternal Consumption of RRL and Some of Its Constituents on the Biotransformation of Fluorogenic Substrates by Liver Microsomes From Male and Female Offspring a

Abbreviations: AMMC, 3-[2-(N,N-diethyl-N-methylammonium)ethyl]-7-methoxy-4-methylcoumarin; BFC, 7-benzyloxy-4-(trifluoromethyl)-coumarin; BQ, 7-benzyloxyquinoline; CEC, 3-cyano-7-ethoxycoumarin; EFC, 7-ethoxy-4-trifluoromethyl-coumarin; ERES, ethoxyresorufin; MFC, 7-methoxy-4-(trifuoromethyl)-coumarin; MRES, methoxyresorufin; RRL, red raspberry leaf; PND, postnatal day.
a Experiments were performed as outlined in the “Materials and Methods” section. Changes (↑, increase; ↓, decrease) indicate significant differences (P < .05) between treated animals and controls (n = 5). None of the treatments affected biotransformation of AMMC, CEC, or MRES by males or females at any of the time points studied.
Discussion
Age-Dependent Changes in CYP Activity
Results from the present study are consistent with numerous other reports in the literature identifying age-dependent differences in the activity of CYP. 18 –20 The diversity in the relationships between CYP activity and age that we have identified (Figure 1) may be due, in part, to the fact that the activity of different isoenzymes are differentially regulated. 18,19 The biotransformation of BFC, EFC, and MFC by male rat liver microsomes increases with age (Figure 1). Similarly, de Zwart et al, 19 found a gradual increase in the N-demethylation of N-ethylmorpine characteristic of CYP 3A1/2 activity from birth until PND42. Overall, the patterns of biotransformation of BFC, EFC, and MFC throughout the present study are very similar (see Figures 1-4), suggesting that these substrates are metabolized by the same enzyme. However, data from adult male rat liver microsomes demonstrate that the 3 substrates have different, though sometimes overlapping, sensitivities to a panel of inhibitors, which imply different substrate selectivities. 17 The capacities to biotransform AMMC, BQ, and MRES decline with advancing age in both males and females (Figure 1). These compounds are substrates for CYPs 2D2, 3A, and 1A2, respectively, 17,21,22 thus these results imply that the activities of those isoforms decline with advancing age.
Although we failed to detect significant biotransformation of any of the substrates by rat hepatic microsomes at PND1, others have reported the presence of either specific isoform protein or mRNA 23 or significant activity of some CYPs 19 during the perinatal period. However, enzyme activity did not necessarily correlate with mRNA expression and, similar to the present findings, was always lower at PND1 than at any subsequent age. 19 Some investigators suggest that hormones in the maternal circulation, such as progesterone, are responsible for the low activity of phase I drug-metabolizing enzymes during the perinatal period, 24,25 while the rapid postnatal increases in enzyme activity are driven by the onset of pituitary released growth hormone. 23,26
Gender-Dependent Changes in CYP Activity
We found significant differences in the biotransformation of AMMC, BFC, BQ, EFC, MFC, and MRES by liver microsomes of male versus female rats (Figure 1). Except for MRES, biotransformation was higher in males than females. Similarly, Shapiro and colleagues found rat hepatic enzyme activity to be higher in males than females. 26 Gender dimorphism in biotransformation of any of the substrates used in this study by neonatal (ie, PND1) rat liver microsomes is unknown as we pooled male and female livers to obtain sufficient tissue for microsomal preparations. We speculate that it would be highly unlikely that there would be any observable differences in CYP activity at PND1, as gender differences in mRNA expression of phase I drug-metabolizing enzymes are absent in the sexually immature animals. 26 However, the presence (or absence) of increased androgen levels at puberty leads to imprinting of CYP enzymes and to gender-dependent differences in CYP activity that are observable in the peripubertal period. 27 Furthermore, the age where gender dimorphism is first observed, PND65, is also the age when CYP enzyme activity reaches maximum levels. These differences might be attributed to the sex steroid levels which are regulated by the pituitary hormones as has been shown by others. 26,28 –30 However, for the biotransformation of CEC and ERES which lack gender differences, the maximum enzyme activity precedes puberty and peaks at PND21 (data not shown). 3-Cyano-7-ethoxycou-marin is likely biotransformed by CYP2C6 17 and ERES by CYPs 2B1 and 2C6. 22
Effect of Maternal Consumption of RRL, Quercetin, Kaempferol, and Ellagic Acid on CYP Activity
Although there is little information in the literature about the effects of RRL on CYP enzyme activity, this herb contains various polyphenols, including ellagic acid and quercetin, known to alter the biotransformation activity of CYP enzymes. 13,14 In addition, it has been shown that in utero exposure to some xenobiotics alters androgen levels during the critical period in utero, leading to permanent changes in gender-specific P450 isoforms in the adult animals. 3,31,32 Maternal consumption of RRL altered and masculinized the biotransformation of BFC, EFC, and MFC. Furthermore, fetal exposure to RRL activated CYP enzyme activity of only female offspring at PND120. The mechanism through which RRL only affects female and not male offspring is unknown. However, the effect of RRL on steroidogenesis of female offspring may impair their fertility and reproductive health. For instance, female rats exposed to RRL during pregnancy had a 22% reduction in pregnancy success (ie, reduced number of deliveries in animals with a confirmed mating), an effect which may be attributed to impaired fertilization and/or reduced implantation. In addition, females exposed to RRL in utero had an increased probability of producing growth-restricted offspring when they themselves became pregnant. 15 Quercetin and kaempferol mimic the effects of RRL and therefore may be the active ingredients in RRL that are responsible for altered CYP activity in the offspring postnatally—although the doses used in the present study are in excess of those achieved by administration of RRL alone. Flavonoids interact with a number of ligand-activated transcription factors that regulate the expression and activity of CYP enzymes. They directly activate the constitutive androstene receptor (CAR) 33 and are agonists and antagonists at the aryl hydrocarbon receptor. 34 Although flavonoids are poor ligands for the pregnane X receptor, 35 they activate it indirectly through action on cyclin-dependent kinases. 36 Similarly, they activate the peroxisome proliferator-activated receptor α (PPARα) 37 which in turn induces CAR and CYP2B expression. 38 Since, across species, many CYP isoforms participate in sex steroid metabolism, 39,40 exposure to kaempferol, quercetin, and RRL may result in an altered sex steroid environment.
The activity of ERES, which is biotransformed by CYP 2B1/2C6 in naive rats, 22,41 was decreased by ellagic acid, a polyphenolic constituent of RRL, in female offspring at PND21. Although other studies show that ellagic acid can inhibit CYP enzyme activity, 42 –44 the mechanism through which ellagic acid can increase the biotransformation of BQ by male offspring at PND65 is unknown.
In summary, we have demonstrated for the first time that in Wistar rats, maternal consumption of RRL or its constituents alters biotransformation activity of some substrates in the offspring of either gender. We have also demonstrated that although these changes are not evident at birth, they are imprinted and become evident over time. Data from this study suggest that maternal consumption of either RRL or any of its constituents may cause transient changes during critical developmental windows and/or persistent changes in drug-metabolizing enzymes. If these same effects are manifested in humans, then the long-term consequences of enzyme imprinting due to maternal consumption of RRL include, but are not limited to, inappropriately rapid biotransformation of pharmaceuticals leading to treatment failures, increased activation of xenobiotics leading to a heightened probability of tumor formation, and altered steroid hormone biotransformation leading to adverse reproductive health/fertility.
Footnotes
Acknowledgments
We thank Laura Paduch and the staff of the Central Animal Facility at McMaster University for assistance with the animal work.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: the SickKids Foundation and the Canadian Institutes for Health Research.