Abstract
Insect repellent N,N-diethyl-m-toluamide (DEET) and sunscreen oxybenzone are capable of enhancing skin permeation of each other when applied simultaneously. We carried out a cellular study in rat astrocytes and neurons to assess cell toxicity of DEET and oxybenzone and a 30-day study in Sprague-Dawley rats to characterize skin permeation and tissue disposition of the compounds. Cellular toxicity occurred at 1 µg/mL for neurons and 7-day treatment for astrocytes and neurons. DEET and oxybenzone permeated across the skin to accumulate in blood, liver, and brain after repeated topical applications. DEET disappeared from the application site faster than oxybenzone. Combined application enhanced the disposition of DEET in liver. No overt sign of behavioral toxicity was observed from several behavioral testing protocols. It was concluded that despite measurable disposition of the study compounds in vivo, there was no evidence of neurotoxicological deficits from repeated topical applications of DEET, oxybenzone, or both.
Keywords
Introduction
Application of insect repellents and sunscreens has been integrated into summer daily life for the general public in the developed countries, due primarily to the conscious awareness of the health threats from vector-borne diseases and skin cancers induced by sunlight radiation. Repellent and sunscreen products are the most practical, cost-effective, and well-accepted choice of defense, and a variety of preparations including sprays, lotions, aerosols, and cloth wipes have been commercially available as specialty products for decades. Concurrent application of repellent and sunscreen preparations has become prevalent in North America since 1999 when mosquito-transmitted West Nile virus first arrived in the continent.
Numerous active repellent and sunscreen ingredients are used for commercial civil use. DEET (N,N-diethyl-m-toluamide) and oxybenzone are 2 principal repellent and sunscreen components, respectively. Their repellency and UV-blockage efficacy have been adequately investigated and documented. 1,2 With the approach to concurrent application, the latest investigations have been focused on the percutaneous interactions between active repellent and sunscreen compounds. 3 –5
As topically applied preparations, protection efficacy of the repellent and sunscreen products relies solely on their presence and retention on the skin surface after each application. Transdermal permeation and systemic absorption of the active ingredients are considered neither desirable nor productive. Nevertheless, both DEET and oxybenzone are known to be capable of permeating across stratum corneum and reaching the general circulation after topical skin applications. 6,7 Moreover, laboratory tests demonstrated permeation synergy between DEET and oxybenzone, when both compounds were applied simultaneously. 4,5 The rate and extent of permeation varies, depending on application dose, preparation type, and application method.
Previous studies were carried out primarily to assess pharmacokinetics and toxicology of DEET and oxybenzone from individual applications, 8 –10 as concurrent application of repellents and sunscreens was not common. There were studies evaluating concurrent use of DEET or oxybenzone in association with several chemicals that were specifically designed for military or farming purposes. 11,12 Loss of sun protection factor (SPF) has also been reported from mixing DEET-based repellents and sunscreens. 13,14
Both repellents and sunscreens are designed for regular daily application over the summer months; it is not uncommon for military personnel, field workers, and farmers to apply both products for an extended period of time while working or training outdoors. Inappropriate use of these chemicals could potentiate unwanted adverse effects in susceptible individuals. In particular, enhanced systemic permeation and absorption of DEET and oxybenzone from repeated concurrent applications may be of health concerns and potential adverse effects should be subsequently elucidated and eliminated. Therefore, the primary objective of this study was to assess the disposition of DEET and oxybenzone from prolonged exposure at cellular levels and repeated skin applications in an animal model, respectively. Since DEET had demonstrated potential neurological toxicity in several earlier studies, 11,15 –17 we focused our experiments on cellular viability of rat astrocytes and neurons in vitro and tissue retention of the test compounds in vivo. Various behavioral testing protocols were also used to assess whether noticeable toxicological evidence would be observed from concurrent use of DEET and oxybenzone.
Materials and Methods
Materials and Reagents
Pure DEET and oxybenzone standards were purchased from Fluka Chemika GmbH (Buchs, Switzerland) and Riedel-de Haën GmbH (Seelze, Germany), respectively. They were accurately weighed and dissolved in 70% ethanol at a concentration of 100 mg/mL of DEET and 12.5 mg/mL of oxybenzone, either individually or in combination. The prepared test solutions were tightly packed in amber glass bottles and stored in a refrigerator for the duration of the study.
To carry out cellular studies, Neurobasal media, Dulbecco’s modified Eagle medium F12 (DMEM-F12), B-27 supplement, fetal bovine serum (FBS),
To carry out drug extraction and analysis, acetonitrile, methanol, and sodium hydroxide were purchased from Fisher Scientific (Fair Lawn, New Jersey). Glacial acetic acid was obtained from Mallinckrodt Specialty Chemical Company (Paris, Kentucky). Ammonium acetate was obtained from Aldrich Chemical Co, Inc (Milwaukee, Wisconsin). Nitric acid was purchased from LabChem Inc (Pittsburgh, Pennsylvania). All solvents were HPLC-grade and other chemicals were AC-grade. Deionized water was obtained from a Milli-Q Pure Water System (Nepean, Ontario, Canada) in the laboratory.
For negative and positive control studies in rats, 0.9% sodium chloride injection USP was obtained from Astra Zeneca Inc (Mississauga, Ontario, Canada), and acrylamide standard was purchased from Sigma-Aldrich Corporation (St Louis, Missouri). The saline solution was used directly; sterile acrylamide solution (6.25 mg/mL) was prepared using deionized water and sealed in ampoules prior to the study.
Cellular Study
Rat primary cortical neurons and primary cortical astrocytes were cultured from E17 and E19 rat fetuses, respectively, according to the procedures described previously. 18 The neurons and astrocytes were cultured on 24 well plates and were generally ≥95% and ≥98% pure, respectively.
The cultures were treated with DEET, oxybenzone, or DEET/oxybenzone combination at various concentrations (0.1 µg/mL, 1 µg/mL, and 10 µg/mL) for different time durations (24 hours, 48 hours, and 7 days). After the treatment, cells were washed and incubated with respective media for 24 hours. The cell viability was then analyzed using standard MTT (3-(4,5-dimethylthiazolyl-2)-2,5-diphenyltetrazolium bromide) assay by adding MTT at a final concentration of 0.25 mg/mL into wells containing media and cells. With this assay, yellow MTT is metabolized by mitochondria of living cells, and membrane impermeable purple formazan crystals accumulate. After 4 hours of treatment, the media/MTT supernatant was aspirated followed by solubilization of the crystals with a mixture of 2-propanol and 0.2 mol/L HCl (7.3:1). Absorbance, which was proportional to the number of surviving cells, was read at 570 nm in triplicates in a 96-well scanning spectrophotometer. The experiments were performed in triplicates on a minimum of 3 independent cultures.
Animal Study
The animal use protocol was approved by the University of Manitoba Animal Use Protocol Management and Review Committee and conducted according to the current guidelines published by the Canadian Council for Animal Care (CCAC).
A total of 60 Sprague-Dawley rats were used in the study, which were randomly divided into 6 groups of 10 animals (5 males and 5 females in each group). The 8-week-old rats (mean weight for males 268 g and mean weight for females 256 g) were obtained from the Central Animal Care Services, University of Manitoba. They were housed individually in holding cages and provided food and water ad libitum. The holding room was programmed to a 12-hour reversed light cycle to facilitate behavioral experiments that were performed on the animals during the study period.
Prior to topical skin application of the study preparations, an area of approximately 4 cm2 (2 × 2 cm) on the back of the study animals was shaven using an electric clipper. This area remained relatively hair free by regular shaving to facilitate topical applications and skin observations over the 30-day study period.
The 3 study doses were group 1, 40 mg/kg (2500 µg/cm2) of DEET; group 2, 5 mg/kg (312.5 µg/cm2) of oxybenzone; and group 3, 40 mg/kg (2500 µg/cm2) of DEET and 5 mg/kg (312.5 µg/cm2) of oxybenzone. The weight of the study animals was recorded periodically over the study duration and doses were adjusted accordingly. Briefly, approximately 100 µL of the test solution was measured and applied onto the skin surface using a pipette. The solution was carefully spread over the shaven skin area with a disposable pipette tip and allowed for ambient evaporation before the study rats were returned to their holding cages. The upper dorsal end was selected for dose application because it was extremely difficult for the study animals to reach this area of the body. This application was repeated daily for a 30-day period. The applications were performed by the animal facility staff and blinded to the investigators involved in all other testing. Use of animal care staff ensured the consistency of the topical applications.
Two negative control study groups received topical applications of 0.9% saline (group 4) and 70% ethanol solution (testing vehicle, group 5) for 30 days. The positive control study group received acrylamide at a dose of 25 mg/kg, by intraperitoneal injection, for 10 days (group 6). Behavioral testing was performed in all study groups to examine potential toxicity from topical application of DEET and oxybenzone.
Sample Collection
After the last topical application of the study dosing on day 30, 300 µL of blood samples were collected from the saphenous vein of the rats using Microvette capillary collection tubes (Sarstedt AG & Co, Nümbrecht, Germany) at time 2, 6, 8 and 24 hours. The plasma was separated by centrifugation of the samples at 13 000g for 30 minutes and stored in labeled polypropylene tubes at −20°C until drug analysis.
Skin tape stripping was also performed to evaluate retention and penetration of DEET and oxybenzone after euthanasia of the animals. Before skin stripping, the application surface was swabbed using 400 µL of acetonitrile to collect the extra dose that was still left behind. On complete drying of the skin, 12 pieces of D-Squame stripping disks (CuDerm Corporation, Dallas, Texas) were consecutively applied to the site, gently pressed for 10 seconds, and then peeled off. The collected tape strips were placed individually in labeled polypropylene tubes and stored at −20°C until drug analysis.
The liver and brain of the study animals were also collected after the euthanasia. They were first perfused with saline to remove blood, then harvested, dried, weighed, and stored in labeled polypropylene tubes at −80°C until drug analysis.
Concentrations of DEET and oxybenzone in all collected samples were measured using an HPLC assay developed and validated in our laboratory. 19 The method was able to simultaneously quantify the compounds using photodiode array detection. For drug extraction, an automatic solid-phase extraction method was developed using a Zymark Rapidtrace SPE Workstation (Caliper Life Sciences, Hopkinton, Massachusetts). Briefly, the separation was completed on a Waters Oasis MAX 3cc (60 mg) extraction cartridge, by using acetonitrile, 0.03 mol/L ammonium acetate (pH 4.5) and water as preconditioning and washing solvents. Methanol, 300 µL, was used as the final solvent to elute the cartridge; the eluent was vortexed for 15 seconds, transferred to an HPLC vial, and 50 µL of the sample injected. Plasma samples, 50 µL, were used for drug extraction. For skin swipes and tape strips, they were dissolved in 1.5 mL of acetonitrile and extracted. Liver and brain samples were first homogenized in acetonitrile using an electric homogenizer (Biospec Products, Bartlesville, Oklahoma). A portion of the homogenate was further subjected to solid-phase extraction, and 50 µL of the eluent was injected to the HPLC system for drug measurement.
Behavioral Testing
Various behavioral testing protocols were used to assess the arousal (open field test), locomotion (open field and ladder test), habituation (open field test), and motor coordination (open field test and ladder test) of the animals over the study duration. 20 It was hypothesized that systemic exposure of the study substances, particularly repellent DEET, could induce behavioral changes due to its reported effect on the neurological system. It was anticipated that the behavioral testing could identify changes among the different study groups.
The ladder rung walking test evaluated the ability of study animals crossing a narrow passage of ladder rungs. 21 The device was composed of a 1m-long plexiglass alley equipped with irregularly spaced stainless steel rungs. The ladder was elevated 100 cm above the ground, with a goal box located at one end of the track. The progress of the animals was videotaped, so that the time used by the animals to cross the entire ladder as well as the miss or slip of the rungs by the animals could be accurately scored and compared.
The open field testing assessed the mobility and agility of the study animals over the course of the experiment. 22 The apparatus was composed of a wooden square box (1 × 1 m) with walls of 30 cm; the whole floor area was further divided into 16 equally sized squares. Each animal was placed in the center of the apparatus to begin the experiment, and the movement was videotaped for 4 minutes. The study recording was also scored and analyzed afterward.
All behavioral testing was performed in a quiet dark room under red lights, separated from the animal holding room. Each rat was tested individually for these experiments. The study animals were trained in the ladder test prior to each study, and baseline was recorded. The positive control study group was tested on day 10, when clear signs of adverse effects were observed. All other study groups were tested 4 hours after the dosing on day 29 to assess behavioral changes from the topical applications.
Data Analysis
Amounts of DEET and oxybenzone were calculated from the average HPLC calibration curve. Total recovery of the compounds in collected samples was obtained where applicable. Drug concentrations among biological samples and between study groups were correlated. In addition, results from behavioral testing were also scored and compiled for data analysis.
Plasma concentrations of DEET and oxybenzone were subjected to noncompartmental pharmacokinetic simulation using WinNonlin software (Version 5.0.1, Pharsight Corporation, Mountain View, California) to calculate apparent elimination half-life. The following statistical analyses of the data were also conducted: (a) a 1-way analysis of variance (ANOVA) followed by the Tukey test for all behavioral results among the 6 study groups and all cellular results among the study groups (PC-SAS 8.02, SAS Institute Inc, Cary, North Carolina); (b) skin distribution parameters, liver and brain concentrations, and total recoveries were calculated as a percentage of the final dose, and then compared between single and combined applications using the Mann-Whitney U Test (PC-SAS 8.02, SAS Institute Inc). Normally distributed data were expressed in Mean ± SEM whereas the nonparametric data were represented as Median ± SD. Differences were considered statistically significant at P < .05.
Results
Cell Viability
Figure 1 shows the cellular viability results from rat astrocytes and neurons after the cultures were exposed to DEET, oxybenzone, or a combination of the 2 compounds at 3 concentration levels for 3 time intervals. No significant differences in astrocyte viability were observed for 24-hour or 48-hour exposure to the test substances in comparison to the control group. However, astrocyte viability was significantly reduced by 25% after the cultures were exposed to both DEET and oxybenzone at 10 µg/mL for 7 days.
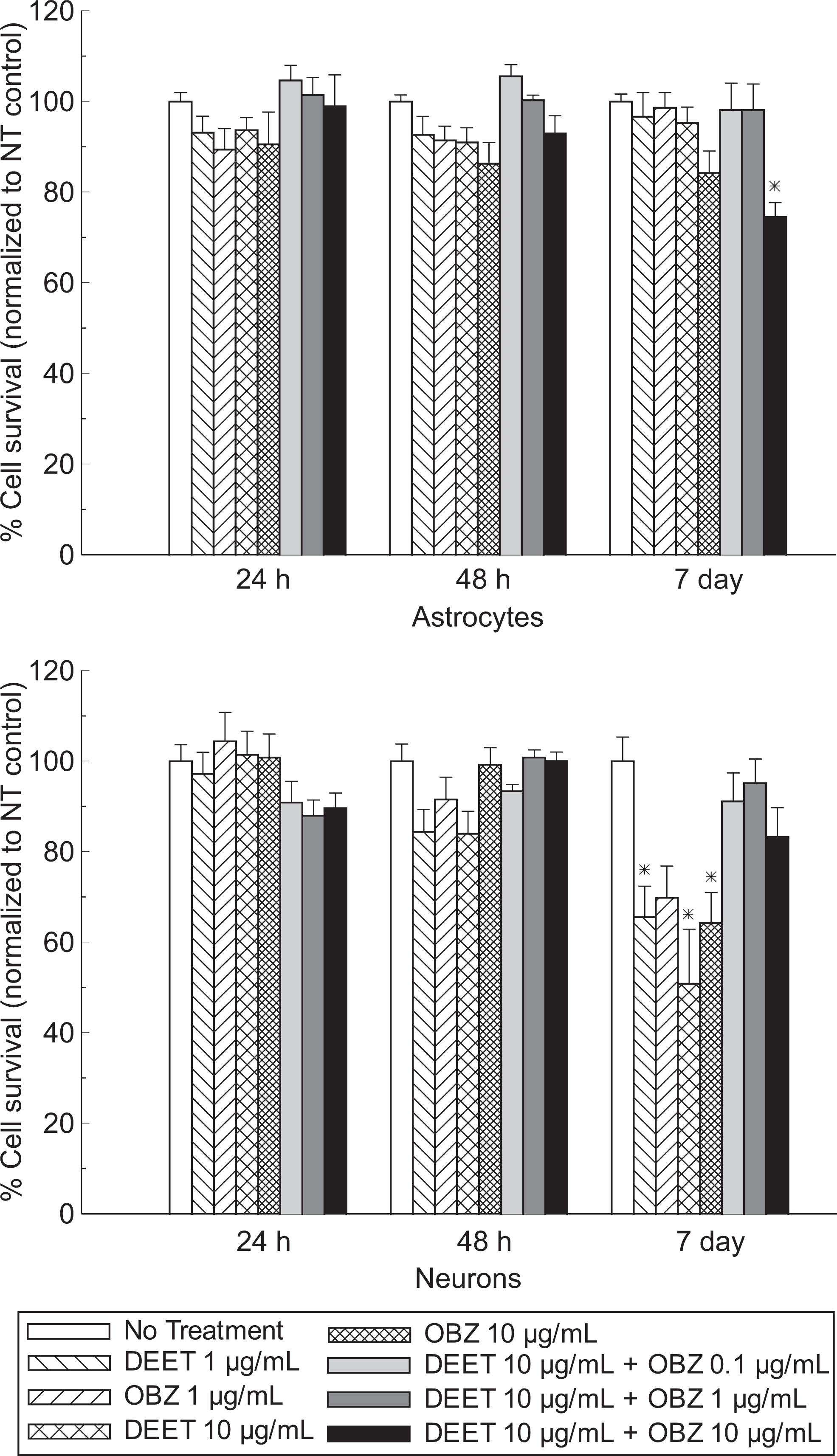
Results of cellular viability of astrocytes and neurons exposed to DEET and oxybenzone (*significant difference from no treatment (NT) control, P < .05, n = 4-8, mean ± SEM). DEET indicates N,N-diethyl-m-toluamide; SEM, standard error of mean.
Similarly, no significant differences in neuron viability were observed for 24-hour or 48-hour exposure to the test substances in comparison to the control group. However, treating the neurons for 7 days with 1 and 10 µg/mL of DEET decreased the cellular survival by 34% and 49%, respectively. A 36% decrease in neuron viability was also noted when the cultures were exposed to 10 µg/mL of oxybenzone for 7 days. Combined use of DEET and oxybenzone did not affect the viability of neurons.
Recovery of DEET and Oxybenzone from the Skin
DEET and oxybenzone were recovered from the site of skin application in 2 ways. Skin swipes were taken 24 hours after the final application to collect the extra dosing that was still left after a 30-day topical use, while skin tape strips recovered the application dose that permeated into skin layers. Table 1 lists the concentration and last dosing recovery of DEET and oxybenzone from the topical application site.
Concentration (µg/cm2) and Total Recovery (%) of DEET and Oxybenzone From Skin Application Site a
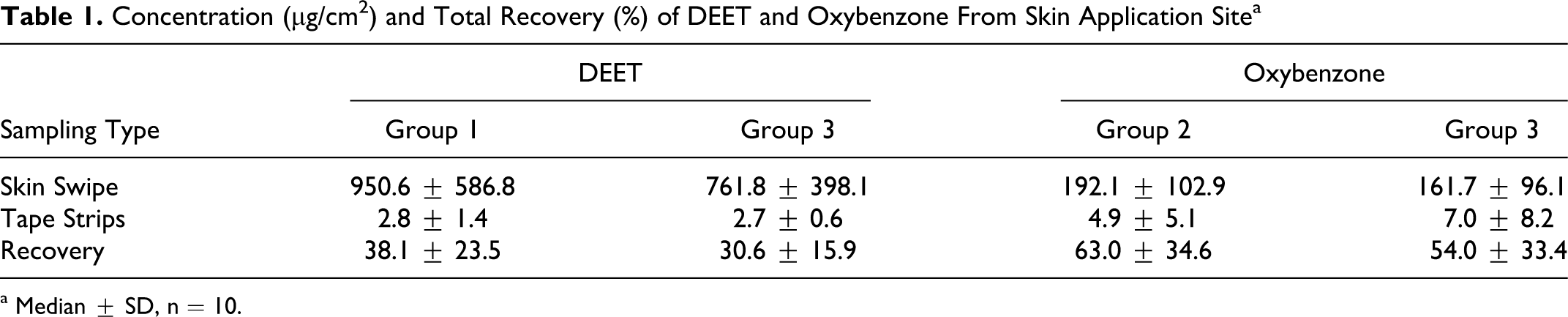
a Median ± SD, n = 10.
Total recovery of DEET was 25% lower in group 3 (combined DEET and oxybenzone) than group 1 (DEET), and that of oxybenzone was 17% lower in group 3 than group 2 (oxybenzone). Although no statistically significant difference was observed between the 2 application groups for either DEET or oxybenzone, recovery data indicated that the combined application disappeared more quickly from the surface of the skin than the single application. This skin disposition profile might also have been expedited by the solvent, as ethanol is a quickly dispatching vehicle that facilitates permeation and evaporation of DEET after topical skin application. Oxybenzone tends to form a skin deposition due to its higher lipophilicity; this property had been observed in our previous studies. 5
Concentrations of DEET and Oxybenzone in Plasma and Tissues
Both DEET and oxybenzone were detected in plasma. Figure 2 shows the concentrations of DEET and oxybenzone over a period of 24 hours after the last skin application. Concurrent application of DEET and oxybenzone appeared to elevate the levels of DEET (20%-66%) but not oxybenzone. Concentration of oxybenzone at 2 hours was approximately twice that of DEET. DEET and oxybenzone were still detectable in plasma 24 hours after the skin application.
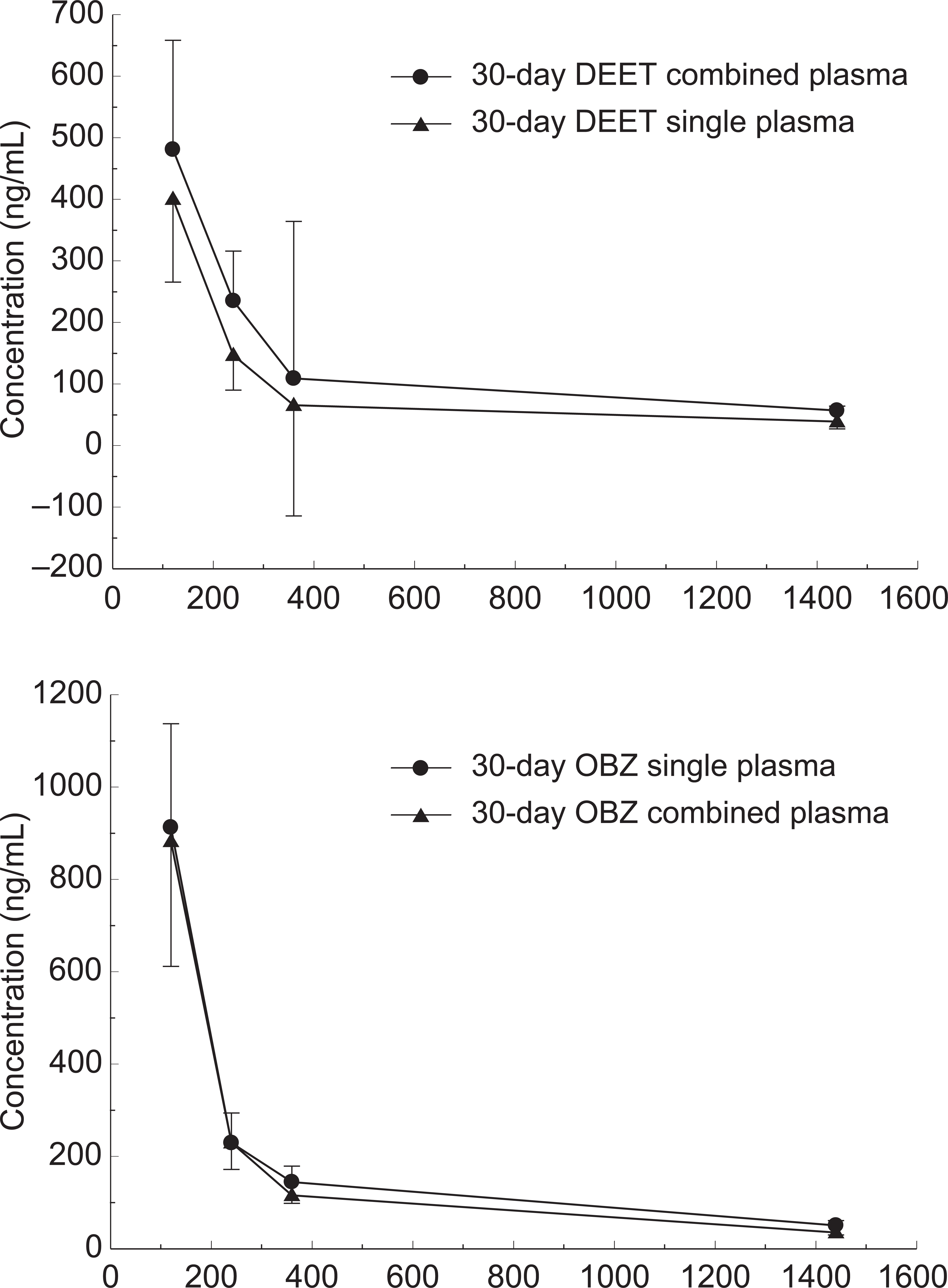
Plasma concentrations of DEET and oxybenzone after last topical skin application on day 30 (n = 10, mean ± SEM). DEET indicates N,N-diethyl-m-toluamide; SEM, standard error of mean.
DEET and oxybenzone were also detected in both liver and brain samples. Table 2 lists the concentration and recovery of the compounds. Concurrent application of DEET and oxybenzone produced significantly higher deposition of DEET in liver than application of DEET alone, although disposition of DEET in brain was much lower than that in liver, the prime metabolism site of the body. Concurrent application of DEET and oxybenzone did not produce enhanced disposition of oxybenzone in liver and brain; the only difference found was lower oxybenzone concentration in brain (121% lower than single application).
Concentration (ng/g) and Total Recovery (%) of DEET and Oxybenzone in the Liver and Brain a
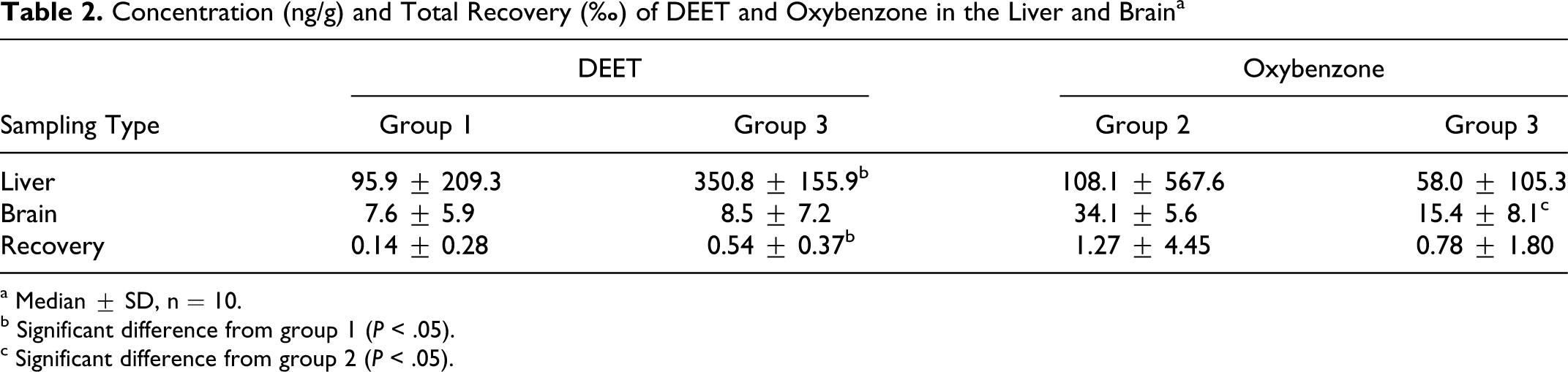
a Median ± SD, n = 10.
b Significant difference from group 1 (P < .05).
c Significant difference from group 2 (P < .05).
Behavioral Testing Results
As expected, the positive control of acrylamide resulted in visible impairment of the study animals after a 10-day injection period. There was a significant difference detected between this positive control group and negative control group (saline solution, topical application) or vehicle control group (70% ethanol solution, topical application) for all study groups. No significant differences were observed among rats when DEET, oxybenzone, and combined DEET/oxybenzone were topically applied to the animals for a 30-day period. Figures 3 and 4 show the results of ladder test and open field test for all study groups, respectively.
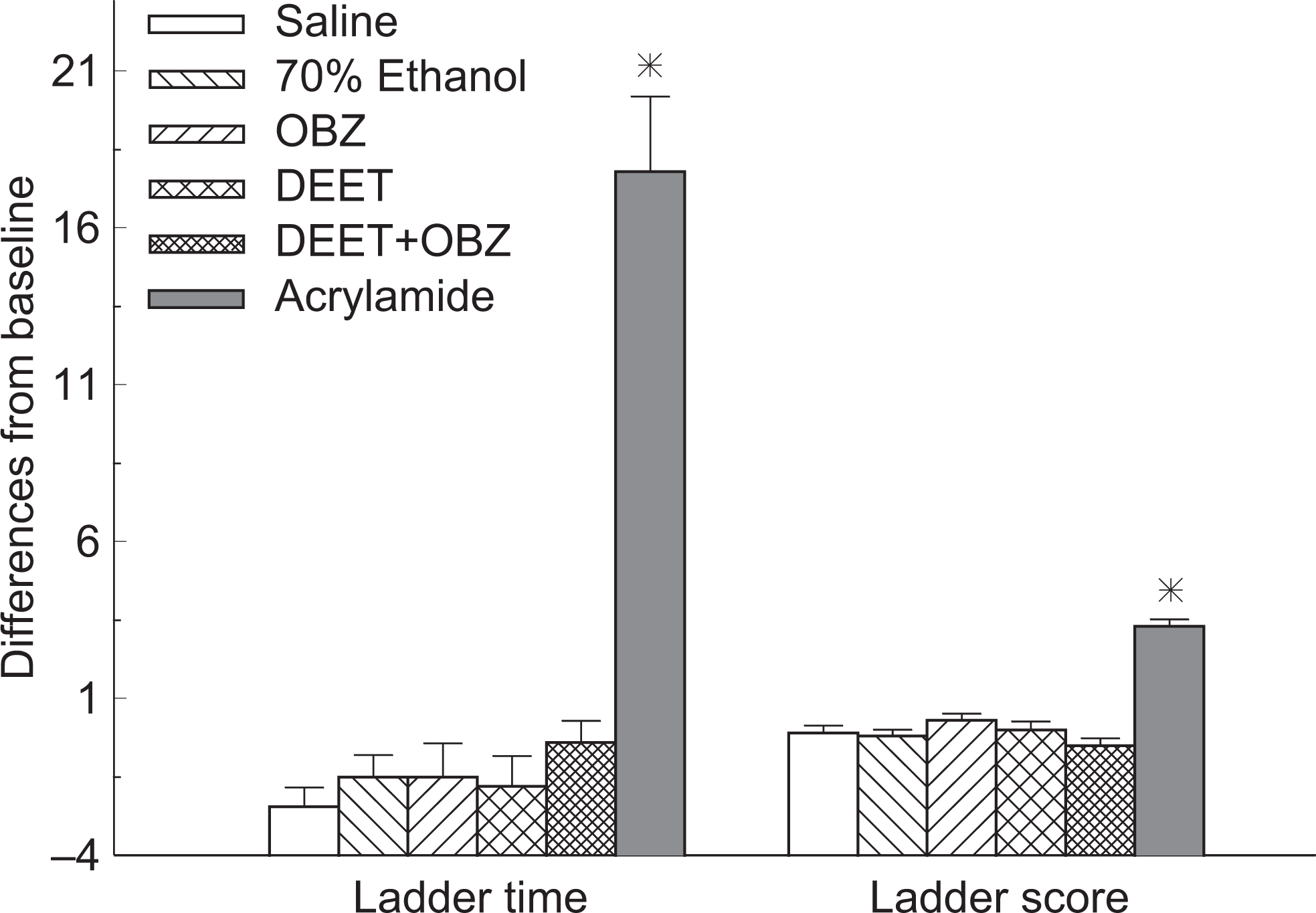
Results of ladder test from 6 study groups on day 29 (*significant difference from saline, vehicle, OBZ, DEET, and combined groups, P < .05, n = 10, mean ± SEM). DEET indicates N,N-diethyl-m-toluamide; SEM, standard error of mean.
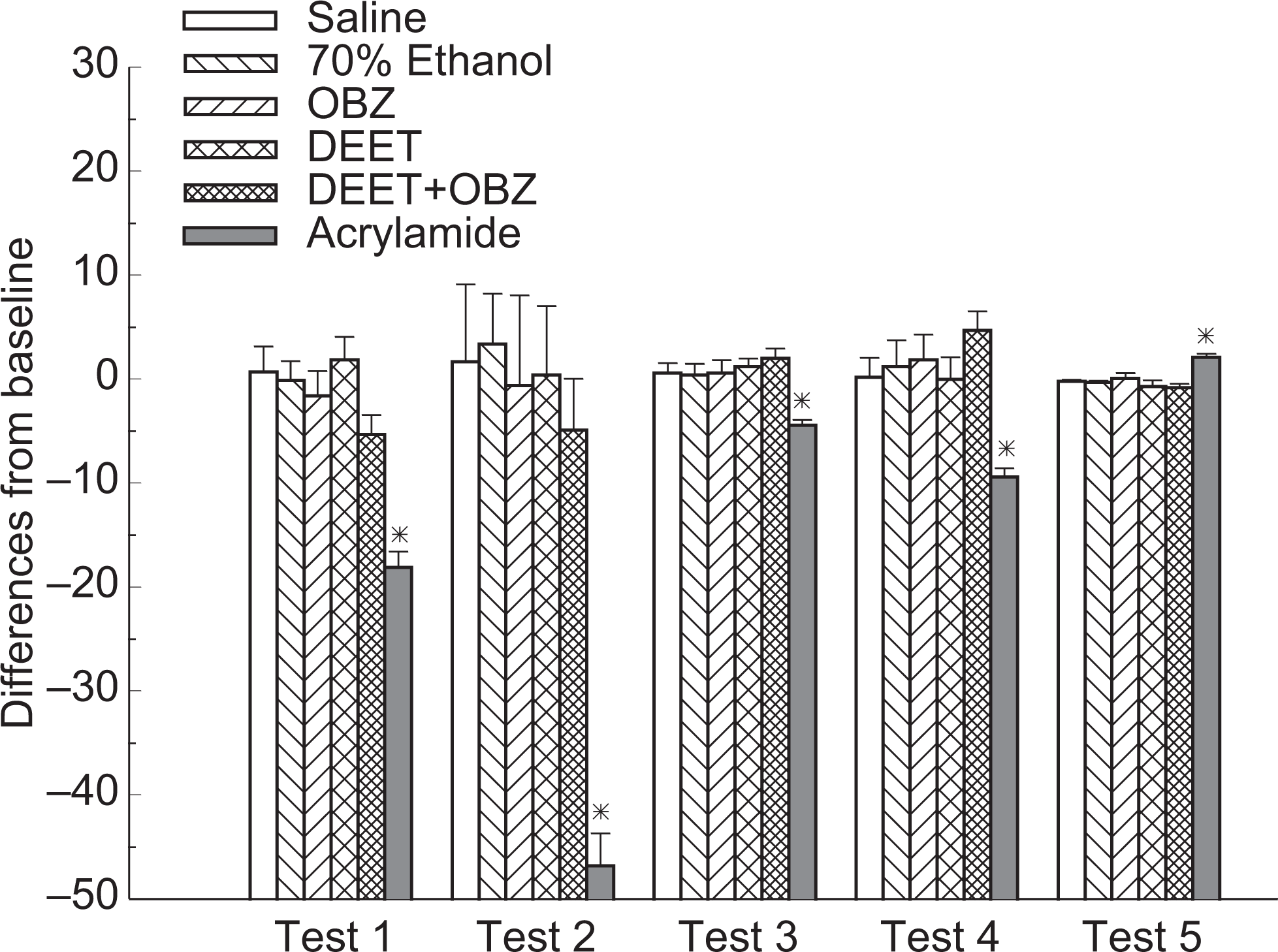
Results of open field test from 6 study groups on day 29. Test 1: number of rearings; test 2: total number of squares entered; test 3: number of times entered center squares; test 4: number of center squares entered; test 5: number of fecal boli (*significant difference from saline, vehicle, OBZ, DEET, and combined groups, P < .05, n = 10, mean ± SEM). DEET indicates N,N-diethyl-m-toluamide; SEM, standard error of mean.
In the ladder test, slips were defined as a total miss or a deep slip of the paws of the study animal while passing the ladder corridor. 21 Rats that did not finish (DNF) the ladder test were automatically assigned a slip number 4 and a time of 30 seconds, and differences between study day 29 and baseline (day 0) were calculated. A negative score indicated an increase in moving ability and a positive score, a reduction in moving ability.
In the open field test, total and center squares entered were measured, as well as the number of times the animals entered the central 4 squares, number of fecal boli and rearings. Habituation is defined as a form of simple, nonassociative learning by a subject in which the magnitude of the response to a specific stimulus decreases with repeated exposure to that stimulus; a reduction in the number of rearings may indicate either a weakness in hind limbs of the subjects or habituation of the testing environment. 23,24
No significant changes were recorded from the study groups on the testing day. Study animals from the 3 treatment groups as well as the negative/vehicle control groups passed the 29-day study period without significant adverse effects. Further systemic studies are, therefore, needed to refine behavioral testing criteria and observed for subtle changes possibly associated with the study compounds.
Discussion
In general, preparations intended for topical skin application should exert optimal efficacy either on the skin surface or within the top skin layers, without substantial percutaneous systemic permeation and absorption. This characteristic is of particular importance to specialty products like repellents and sunscreens, because they are designed to protect the skin from externally damaging factors such as biting insects and UV radiation. Systemic exposure of the body to DEET and oxybenzone is detrimental and counterproductive from clinical perspectives. Repellents and sunscreens are largely applied at the discretion of individual users, without medically recommended or accepted doses. Moreover, repeated applications of repellents and sunscreens are commonplace during summer months when mosquito infestation and UV radiation are high. Any inadvertent exposure of the body to an increased level of DEET and oxybenzone could lead to potential toxicity of the substances in susceptible individuals.
It is well known that neurons have a remarkable ability to transmit rapid electrical signals in the form of action potentials. 25 However, in the past few years, the emergence of astrocytes as the main neural cell type responsible for the maintenance of brain homeostasis has become evident due to their cooperation with neurons on several levels, including neurotransmitter trafficking and recycling, ion homeostasis, energy metabolism, and defense against oxidative stress. 26 Disposition of DEET and oxybenzone in astrocytes and neurons could result in severe neurological effects by disrupting this brain homeostasis. Thus, neurocellular experiments were an essential component of this study, as it could provide a better understanding of the underlying mechanisms causing potential behavioral deficits in the study animals from repeated skin applications.
Although the mammalian brain has a very high metabolic rate, neurons are by nature extremely sensitive to small modifications to their microenvironment. 26 However, the viability of rat astrocytes and neurons was affected only after a prolonged exposure to the test compounds in this study. Astrocytes were sensitive to high concentrations of DEET and oxybenzone in combination, whereas neurons were more sensitive to individual applications. Past studies looking at DEET alone reported significant neuronal degeneration in the motor cerebral cortex, the dentate gyrus, hippocampal cells, and the Purkinje cell layer of the cerebellum, but those studies did not find any significant behavioral differences between DEET and control animals. 15 Based on results obtained from our study, further cellular studies might be beneficial to clarify the inconsistencies in the literature regarding the toxic effects of insect repellent DEET. 27
As one of the largest organs in surface area and body weight, skin has long been viewed as a passive physical barrier between human body and external environment. Stratum corneum is the outermost skin barrier that restricts the penetration of a majority of foreign substances. Systemic percutaneous absorption will take place only when the barrier function of stratum corneum is compromised. There are various mechanisms by which chemical substances permeate across stratum corneum. Passive diffusion driven by a high-concentration gradient is one of the primary permeation mechanisms for drug compounds to overcome the skin barrier and reach the systemic circulation. For lipophilic molecules like DEET and oxybenzone, direct penetration through the intercellular laminae of stratum corneum should also allow for ready availability of the compounds for systemic absorption within the skin. 28
Both DEET and oxybenzone were recovered in appreciable amounts from the application site in the study animals. Recovery of oxybenzone was higher than that of DEET, attributing partially to ready absorption/evaporation of DEET and prolonged retention of oxybenzone at ambient temperature. In this study 70% ethanol was used as a vehicle to dissolve both test compounds and to facilitate easy skin applications. Recent studies have demonstrated that evaporation of DEET was 47% to 49% and 33% to 40% for microcapsules and ethanol solution, respectively. 29 In addition, DEET evaporates from the skin surface at a rate proportional to its localized concentration in the skin. 30 Results obtained from this study were consistent with those previously reported. In view of the significant recovery of DEET and oxybenzone by skin swabs, it is, therefore, important for individuals to clean skin thoroughly of repellent and sunscreen products once indoors. This will significantly minimize prolonged transdermal exposure of the active ingredients, particularly beneficial to frequent and heavy users of the preparations.
Skin tape stripping was used to assess the movement of DEET/oxybenzone within the skin. Other studies have indicated no DEET accumulation in the skin, 10,31 which was similar to what was found in this study. In comparison to a previous study using piglets, recovery of DEET and oxybenzone by tape stripping was much lower from this study. 5 This discrepancy might be partially attributed to the lotion products used in piglets where preparations prolonged the retention of test compounds in the skin. In addition, anatomical differences between rat skin and piglet skin might have also played a role in skin retention of DEET and oxybenzone. Sprague-Dawley rats have a thickness of 14.7 ± 2.2 and 4.3 ± 1.2 µm for the epidermis and stratum corneum, respectively, whereas piglets have a thickness of 36.9 ± 7.4 and 3.5 ± 0.9 µm for the epidermis and stratum corneum, respectively. 32
Quantifiable amounts of DEET and oxybenzone were detected from plasma samples in the study. This indicated ready systemic absorption of the compounds from the skin applications. Both DEET and oxybenzone primarily undergo metabolism in the liver and are subsequently excreted in urine. This metabolism is relatively quick as previous studies have shown; elimination half-life of DEET was reported to be approximately 2.5 hours in beagle dogs and cattle. 7,33 In our previous study with piglets, the half-life of DEET and oxybenzone was 7.3 and 8.0 hours, respectively. 5 Apparent elimination half-life from this study was estimated to be 9.1 ± 2.1 hours for DEET and 7.9 ± 1.7 hours for oxybenzone. Both DEET and oxybenzone were measurable 24 hours after the skin application. This might have resulted from 2 factors, that is, topical skin applications and a 30-day study period. Dermal application tends to prolong the absorption phase of applied chemicals across the skin, which subsequently influenced the disposition of the compounds in vivo. Higher amounts of oxybenzone were detected in the tape strips; therefore, as has been reported, it is probable that oxybenzone was forming a skin depot that continued to supply the compound into the general circulation. 5,9,28 This characteristic should be further assessed to provide guidelines for the safe and effective use of repellents and sunscreens for long-term skin applications.
Similarly, both DEET and oxybenzone were detected in the 2 vital organs from the study. Tissue levels of DEET and oxybenzone tend to be higher in the liver, the prime metabolism site of the body, which had also been proved in previous studies. 8,9,34 The differences observed between combined application of DEET with oxybenzone and application of DEET alone might be detrimental from a clinical viewpoint, as oxybenzone appeared to slightly enhance systemic accumulation and disposition of DEET. This could result in undesirable adverse effects of the repellents in susceptible individuals, from prolonged application of the 2 preparations. Disposition of oxybenzone in liver and brain was quite different from that of DEET, in which concurrent application with DEET did not apparently enhance its percutaneous absorption. In our previous studies with the compounds, it was found that concurrent application affected DEET more than oxybenzone. 5 In this study, concentration of oxybenzone present in brain was higher than DEET. This might be attributed to different lipophilicity attributes of the 2 compounds. Partition coefficients of DEET and oxybenzone in octanol and water (log P octanol/water) are 2.0 and 3.8, respectively. 35,36 High concentrations of oxybenzone had also been detected from fat tissues after topical administration. 37 Based on disposition results obtained from this study, further studies should be carried out to investigate the distribution of the compounds in other tissues or organs.
The primary objective of the animal study was to evaluate whether concurrent application of DEET and oxybenzone would induce observable adverse effects in the study animals. Neurological toxicity of DEET in combination with permethrin and/or malathion/pyridostigmine bromide from repeated applications had been documented in previous studies. 11,15 –17,38 Oxybenzone, however, was only related to minor side effects such as skin allergy and contact dermatitis. 39 However, oxybenzone possesses structural resemblance to steroid hormones, and it demonstrated disruptive potential to the general endocrine system. 40 Carcinogenicity of oxybenzone was also studied but results were inconclusive. 41 In this study, experiments were focused on behavioral abnormalities, which were considered directly correlative of neurological toxicity from DEET and potentially oxybenzone, as shown by neurocellular data.
Animal behavioral responses have been shown to provide integrative markers of neurotoxicity reflecting biochemical, physiological, and neuropathological reactions to toxicants. 42 Previous research has indicated that animals treated with DEET 40 mg/kg alone exhibited significant sensorimotor impairment compared to control, which was reflected in inclined plane performance, forepaw grip time, beam-walk scores, and beam-walk time when assessed after 30 days of daily exposure. 11 However, no significant behavioral changes were observed in this study when DEET and/or oxybenzone were applied topically for 30 days. Any small differences seen among the 6 treatment groups were likely attributed to acute increases in emotionality and/or unfamiliarity with the testing protocols. In addition, to minimize the risk of type II statistical error, male and female rats were grouped together to create a sufficient power index. Furthermore, this study demonstrated that the chemical concentrations found in the biological samples did not produce any changes in animal behavior thus indicating that concurrent use of DEET and oxybenzone at 40 and 5 mg/kg did not produce any overt signs of toxicity. Behavioral testing is one of the assessment methods for neurological toxicity, but it does sometimes yield variable results when replicated in different laboratories; the degree of variability may also increase with experimental parameters such as laboratory environment, apparatus setting, testing time, and evaluation criteria. 43 Studies at higher doses or in susceptible animal models might, therefore, be needed in future experiments.
Conclusion
In conclusion, exposure of rat astrocytes and neurons to the insect repellent DEET and the sunscreen oxybenzone led to reduced astrocyte viability from combined application, whereas neurons were affected only by DEET or oxybenzone used alone. DEET and oxybenzone were detected to penetrate across the skin after a 30-day topical application in a rat animal model. Systemic absorption of the compounds was correlative among skin, plasma, and tissue samples. Both DEET and oxybenzone demonstrated a fast transdermal penetration, but no significant differences were observed between single and combined applications. Concurrent application of DEET and oxybenzone enhanced the concentration of DEET in the liver, but it reduced the brain disposition of oxybenzone. Behavioral testing did not find overt signs of toxicity that had been reported from previous studies, from either single or combined application of the study substances. Insect repellents and sunscreens are specialty consumer care products widely used by the general public for summer outdoor activities. Future studies will evaluate kinetic profiles of concurrent application as well as the potential for hepatic toxicity due to the accumulation of the parent compounds and/or primary metabolites. Formulation development will also be optimized to minimize overall skin permeation of the active ingredients; this will be particularly beneficial to those special workers and outdoor enthusiasts that use repellents and sunscreens regularly for extended periods of time.
Footnotes
Acknowledgment
The authors acknowledge research support from Canadian Institutes of Health Research (CIHR)/Manitoba Health Research Council (MHRC, 2005ROP-148547) and Canada Foundation for Innovation (CFI). DF and TW received graduate studentship from MHRC and the University of Manitoba. Generous supply of DEET metabolites and the internal standard from Dr WG Taylor of Agriculture and Agri-Food Canada is also acknowledged.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: Canadian Institutes of Health Research (CIHR)/Manitoba Health Research Council (MHRC, 2005ROP-148547): operating funding; Canada Foundation for Innovation (CFI): instrument support.
