Abstract
Polycyclic aromatic hydrocarbons (PAH) have been demonstrated to affect immune system modulation. The freshwater species of fish, Lepomis macrochirus (bluegill), was employed to investigate the effects of a 14-day dietary exposure to PAH including 2-aminoanthracene (2-AA), 2-methylnaphthalene (2-MN), and 9,10-dimethylanthracene (9,10-DMA) and a mixture of these 3 compounds at a total dose of 3.1 ± 0.01 mg on lymphocyte proliferation stimulated with 3 mitogens (concanavalin A [Con A], phorbol ester, and calcium ionophore). 2-Aminoanthracene was mitogenic itself and with added mitogens. 2-Methylnaphthalene induced some stimulatory and some inhibitory effects upon cell proliferation by Con A. 9,10-DMA and the mixture each suppressed cell proliferation. The mixture was highly suppressive to lymphocytes. Intracellular baseline calcium levels were reduced, possibly as a step prior to cell death. All PAH compounds tested were immunomodulatory to bluegill lymphocytes. Bluegill were demonstrated to have utility as a biomarker species for investigation of immunotoxicity.
Introduction
Exposure to carcinogenic polycyclic aromatic hydrocarbons (PAH), such as those found environmentally in energy-related waste, can have immunomodulatory effects on human, murine, and teleost lymphocytes. 1 –3 A strong correlation between PAH carcinogenic potency and immunosuppression has been demonstrated in human and murine lymphocytes. 4,5 Strong PAH 5- and 4-ring carcinogens such as benzo[a]pyrene (BaP) and 7,12-dimethylbenzanthracene (DMBA) are also strongly immunosuppressive. 1,2 Far less is known about the action of 2- and 3-ring aromatic petroleum-associated PAHs such as alkylated and amine-substituted naphthalenes, anthracenes, and phenanthrenes. In fish, exposure to individual PAH as well as mixtures of PAH can be immunomodulatory. Carcinogenic 4- and 5-ring PAH such as DMBA and BaP have immunomodulatory effects on lymphocyte proliferation in fish. 6 –9 Polycyclic aromatic hydrocarbons−mediated immunosuppression may be due to alteration of cellular signal transduction pathways via alteration of intracellular Ca2+ homeostasis and/or cellular protein phosphorylation and activation. 4,10 –12 Polycyclic aromatic hydrocarbons have been shown to cause an upset in cellular calcium homeostasis in a structure-related fashion. The more carcinogenic and immunosuppressive PAH have been shown to stimulate a sustained increase in intracellular Ca2+ levels. 7,12-Dimethylbenzanthracene also produces a sustained rise in intracellular calcium in murine B cells. 13,14 Minimally immunosuppressive PAH such as benzo[e]pyrene (BeP) and anthracene cause only a transient (3 minute) rise in intracellular calcium in lymphocytes. 11
Immunomodulatory effects on lymphocytes may be demonstrated by their effects on mitogen-stimulated cell proliferation. Benzo[a]pyrene and DMBA suppress mitogen-stimulated cell proliferation in human T and B lymphocytes. 8,15 7,12-Dimethylbenzanthracene, 3-methylcholanthrene (3-MC), and BaP suppress mitogen-stimulated proliferation in T and B murine lymphocytes both in vivo and in vitro. 16 –20 However, BaP has also been demonstrated to have either no effect or a proliferative effect on both T and B lymphocytes. 21 –23
Mitogen-stimulated cell proliferation is commonly used as a measure of immunocompetence. Different mitogenic agents stimulate different receptors in several signal transduction pathways. The mitogenic lectin from Canavalia ensiformis, concanavalin A (Con A), and the pharmacologic agents phorbol myristate acetate (PMA) and A23187 each interact with different proteins in the lymphocyte and stimulate different signal transduction pathways (Figure 1). Concanavalin A (denoted as #arrow1) interacts with cell surface receptors including the T-cell receptor and/or CD3 receptor. Phorbol esters, such as PMA (denoted as #arrow3) mimic di-acyl glycerol (DAG), and activate Protein Kinase C (PKC), ras, Raf-1, Mitogen-activated protein, external-signal regulated Kinase (MEK-1), Extracellularly-regulated Kinase (ERK), and ERK-2 signaling proteins in humans. 24 –26 A23187 is a calcium ionophore (denoted as #arrow2) whose actions mimic (inorganic triphosphate) IP3 by transporting Ca2+ across biologic membranes, increasing intracellular calcium levels. 27 In mammalian and teleost models, lymphocyte proliferation can be stimulated by either a combination of A23187 and PMA or by the mitogen Con A. 28 –30
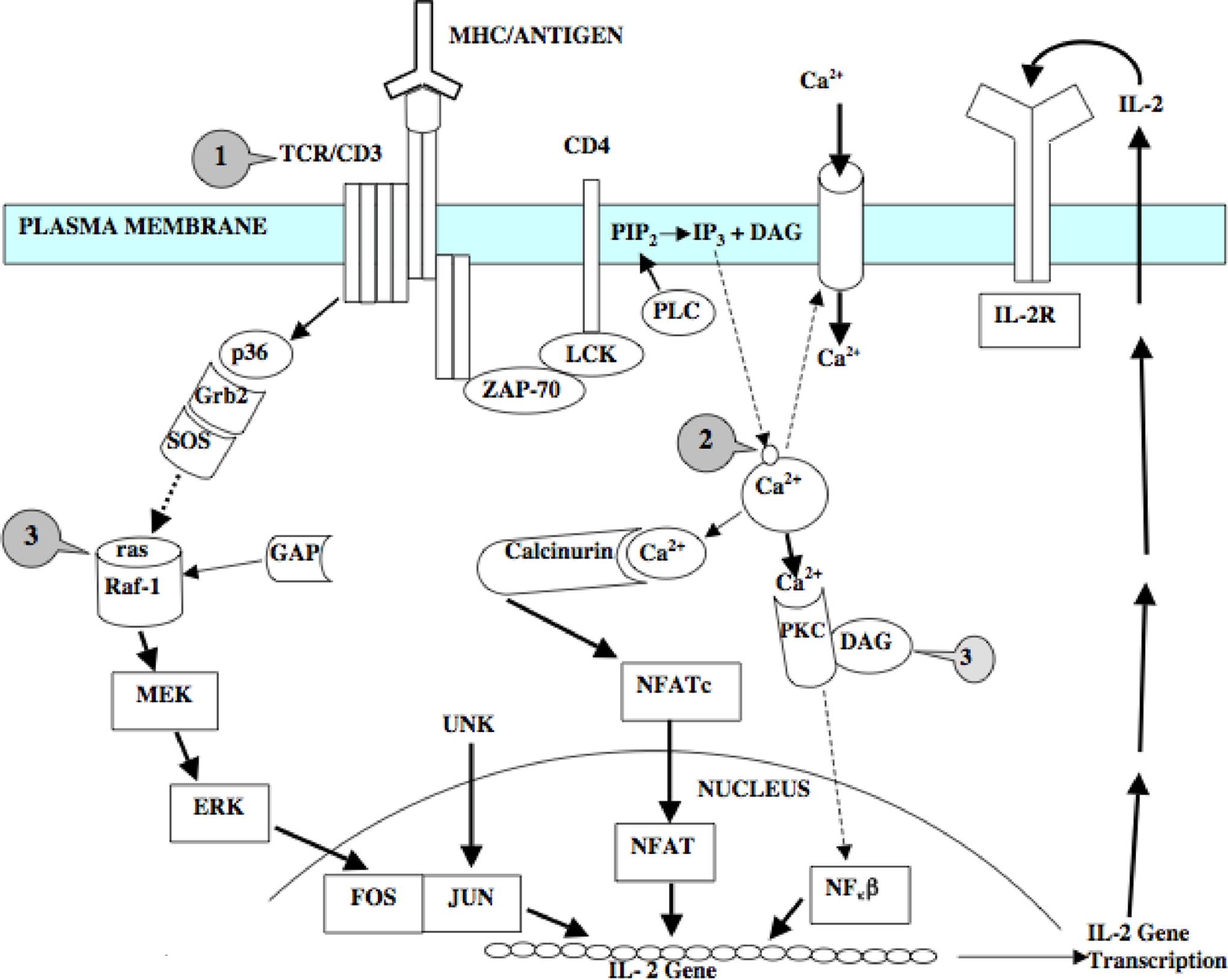
Cell-signaling pathways involved in lymphocyte activation in rodent models. Label arrow 1 = site of Con A activity; Label arrow 2 = site of calcium ionophore activity and Label arrow 3 = site of PMA activity. Signaling pathways are thought to be highly conserved in vertebrates and are presented as putative pathways for investigation in fish.
In this study, a 14-day diet of the mutagenic 3-ring compounds 2-AA (CAS#613-13-8) and DMA (CAS#781-43-1) as well as the 2-ring compound 2-2-MN (CAS#91-57-6) and a mixture of these 3 compounds were evaluated for immunomodulatory effects by mitogen-stimulation assays in fish (Lepomis macrochirus) lymphocytes. The 14-day diet of these 3 carcinogens individually and as a mixture were also tested for effects on baseline or resting intracellular calcium levels in isolated fish lymphocytes. An additional objective of this study is to validate the use of this teleost fish model for use with these methods to probe the cellular immune system and advance our understanding of the immune system as well as biomarker endpoints in this fish model.
Methods and Materials
Reagents
All chemicals and reagents used in these studies were reagent grade unless otherwise specified. All kits were used as is from the manufacturer according to their instructions.
Fish
Lepomis macrochirus, weighing 120 to 156 g and measuring 13 to 18 cm, were collected from the inbred hatchery stock population in Louisiana State University Lakes by electro-shocking. Fish tissue analyses showed that the fish were not environmentally contaminated with PAHs. Fish were laboratory-acclimated for 2 weeks in 10 L glass aquaria, 1 fish per tank, in dechlorinated (DCl) water. During the acclimation period, fish were fed trout chow floating pellets (Ziegler, Gardeners, Pennsylvania). The water was changed daily, and was maintained at 21 ± 1°C. A 12 hour/12 hour light/dark cycle was maintained in the exposure room at all times. Animals were maintained in accordance with the Association for Assessment and Acceditation of Laboratory Animal Care (AAALAC) regulations and experiments were performed under an approved Animal Care and Use protocol.
Diets
All PAHs (PAH > 98% purity as assessed by the manufacturer), including 9,10-DMA, 2-MN, and 2-AA, were purchased from Sigma Chemical Company (St Louis, Missouri). Pellets were prepared by dissolving 2-AA, 2-MN, and 9,10-DMA in corn oil (Sigma) and mixing with pulverized trout chow (Ziegler). The diets were homogenized by mixing on a roller mill for 3 days. Pellets were then reformed by extruding the mixture through a 60-mL catheter tip syringe into pellet forms (Fisher, Houston, Texas). Pellet diets of individual compounds were tested at a dose of 3.1 ± 0.01 mg/g-diet over a 14-day period. The MIX diet was tested at a total PAH dose of 0.13 ± 0.01 mg/g-diet 2-AA, 0.19 ± 0.01 mg/g-diet 9,10-DMA, and 0.22 ± 0.02 mg/g-diet 2-MN over a 14-day period. 2-AA, 2-MN, and 9,10-DMA were made at 220 mg/pellet for a total 14-day dose of 3.08 ± 0.01 mg. A vehicle control diet was made up of trout chow and corn oil only. Diets were stored in the dark at 0°C until just before distribution to the fish. Fish were fed 1 pellet per day for 14 days. Fish had been previously trained to eat a floating pellet diet and consistently ate their pellet immediately upon being fed. Fish were fed at 9
Dosing Regimes
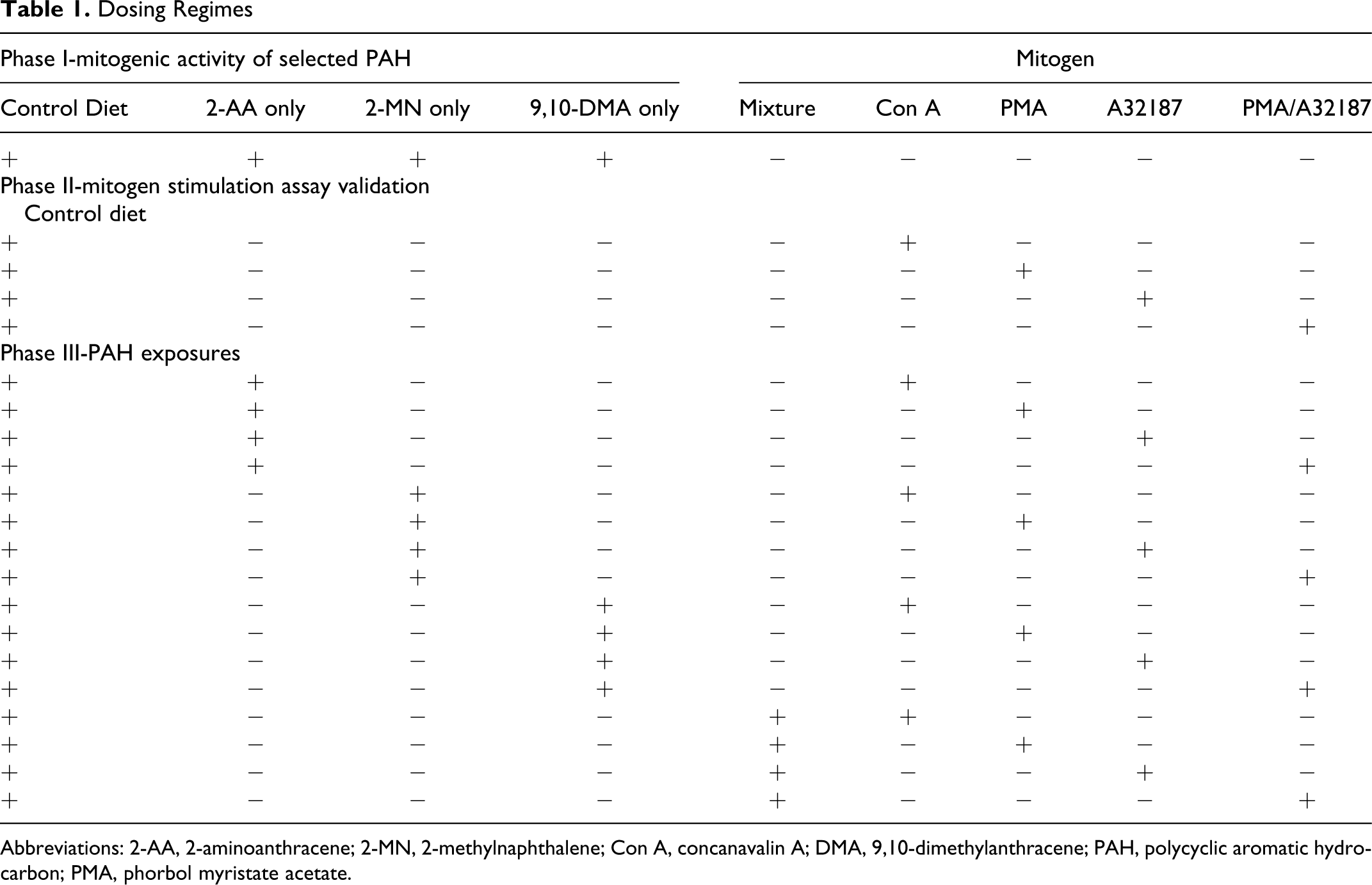
Abbreviations: 2-AA, 2-aminoanthracene; 2-MN, 2-methylnaphthalene; Con A, concanavalin A; DMA, 9,10-dimethylanthracene; PAH, polycyclic aromatic hydrocarbon; PMA, phorbol myristate acetate.
Dose Verification
Verification of pellet PAH content was performed in triplicate on each diet. 31 Briefly, food pellets containing either 2-MN or 9,10-DMA were sonicated and extracted with pesticide grade dichloromethane (Mallinckrodt, Paris, Kentucky). Pellets containing 2-AA were extracted with 80% dichloromethane and 20% pesticide grade methanol (Mallinckrodt). Excess lipids were removed by passing them through an activated fluorosil column (1 cm × 10 cm; Sigma). Five deuterated PAH were used as surrogate standards for the samples, and 2-fluorobiphenyl was used as an instrument internal standard. Samples of 2 μL were analyzed using a Hewlett-Packard (Hewlett-Packard, San Jose, California) Model 5980 gas chromatograph interfaced to an HP model 5970 quadrupole mass spectrometer.
Cell Separation
Lymphocytes were collected from peripheral blood as described in Miller and Clem.
29
Approximately 0.5 mL blood was collected from bluegill anesthetized in dechlorinated water containing 0.25 g/L tricaine methanesulfonate (Sigma). Blood was collected from the caudal sinus at the base of the anal fin using a 1-cc tuberculin syringe and 22-gauge needle containing 100 units heparin sodium (Elkins-Sinn, Cherryhill, New Jersey). The collected blood was immediately suspended in 2 mL of incomplete media
29
(AIM V; Lebovitz
Cell Proliferation
Cell proliferation assays were done in 96-well plates at 500 000 cells per well, 200 mL final volume. Cells were cultured in osmotically adjusted (310 mOsm) L-15/AIM V (Gibco) media supplemented with 5% bluegill serum. 29 Mitogens were added to the cell culture at the time of incubation. Con A was used at a final concentration of 25 μg/mL, PMA was used at a final concentration of 2 ng/mL, and calcium ionophore (A23187) was used at a final concentration of 200 ng/mL. All mitogens were sterile tissue culture grade and purchased from Sigma. Mitogens were dissolved in sterile tissue culture grade anhydrous dimethyl sulfoxide (DMSO; Sigma) just prior to use. The final concentration of DMSO per well was 45 parts per million (ppm). Cells were incubated at 25°C in a 5% CO2 humidified environment (Forma Scientific, Marietta, Ohio). The complete media containing PMA and A23187 was changed after 18 hours initial incubation and replaced with fresh complete media, due to observed cytotoxicity after 20 hours. Cells were pulsed after 40 hours of incubation with 0.5 μCi [3H] thymidine per well and harvested 20 hours later. The radioactivity was counted in a liquid scintillation counter (LKB Wallac 1205 Betaplate Liquid Scintillation Counter). Cell proliferation is measured as the disintegrations per minute (DPM) of [3H] thymidine incorporation into DNA during the incubation period. Disintegrations per minute were corrected for quenching using a method of standard addition.
Flow Cytometry Measurement of Calcium
Bluegill lymphocytes from PAH-fed fish and control fish were analyzed for differences in resting or baseline intracellular Ca2+. Cell samples were prepared in triplicate for each treatment and control group. Baseline intracellular Ca2+ levels were analyzed using a FACScan Flow Cytometer (Beckton Dickinson, Bedford, Massachusetts) equipped with an argon ion (488 nm emission) laser. The Ca2+-chelating dyes, Fluo-3 AM and Fura-Red AM (Molecular Probes, Eugene, Oregon) were used as ratiometric indicators of Ca2+. Fura-Red fluoresces most intensely in the red region when not bound to Ca2+, and Fluo-3 fluoresces with greatest intensity in the green region when bound to Ca2+. By loading the 2 dyes simultaneously, and using a ratiometric approach, heterogeneity in dye loading becomes less important, and the magnitude of response is greater than that achieved using only 1 fluorophore. Simultaneous use of these dyes yielded a low-noise signal in the flow cytometer, which gave a highly sensitive measurement of free intracellular Ca2+. 32 Lymphocyte samples were suspended at 500 000 cells/100 mL media containing 6 mmol/L Fluo-3 and 13.8 mmol/L Fura-Red. Cell viability was assessed as 95% to 98% by trypan blue (Sigma). Cells were incubated for 1.5 hours at 25°C in a 5% CO2 environment, with cells being shaken at 45 minutes to ensure even loading. Just prior to analysis, media volume was brought up to 800 mL. Lymphocyte populations were gated on the flow cytometer from monocytes and debris on the basis of forward versus right angle scatter. The dyes were excited at 488 nm with Fluo-3 emission detected at 515 to 535 nm and Fura Red emission detected at 665- to 685 nm. Ten thousand events were counted for each sample. Baseline cellular response was determined using DMSO control-treated cells. Addition of 1 mmol/L thapsigargin (Sigma), a specific sarcoplasmic/endoplasmic reticulum calcium ATPase inhibitor, served as a positive control for Ca2+ mobilization. Results were generated as a ratio of mean channel fluorescence Fluo-3/Fura-Red × 500 using Verity Winlist software. The means of these ratios are reported for each fish.
Statistics
Data were analyzed for normality, significant differences, and interactions. A mixed functional model that incorporated the variables of diet, mitogen, and mitogen × diet interactions was used (SAS Institute, Cary, North Carolina). A significance threshold of P < .02 was used throughout.
Results
Lymphocyte Proliferation in the Absence of Added Mitogen
Peripheral blood lymphocytes (PBL) from fish fed the diet of 2-AA showed a significant increase (P < .02) in proliferation in the absence of any mitogen to approximately 5 times that of the control (Figure 2). The proliferative responses of cells in the 2-MN- and 9,10-DMA-treated fish were reduced slightly as compared with control cells and were significant at the P < .02 level, but the differences between 2-MN and 9,10-DMA were not significant. Mean DPM for the control (representing basal cell replication) were 638 ± 115 DPM.
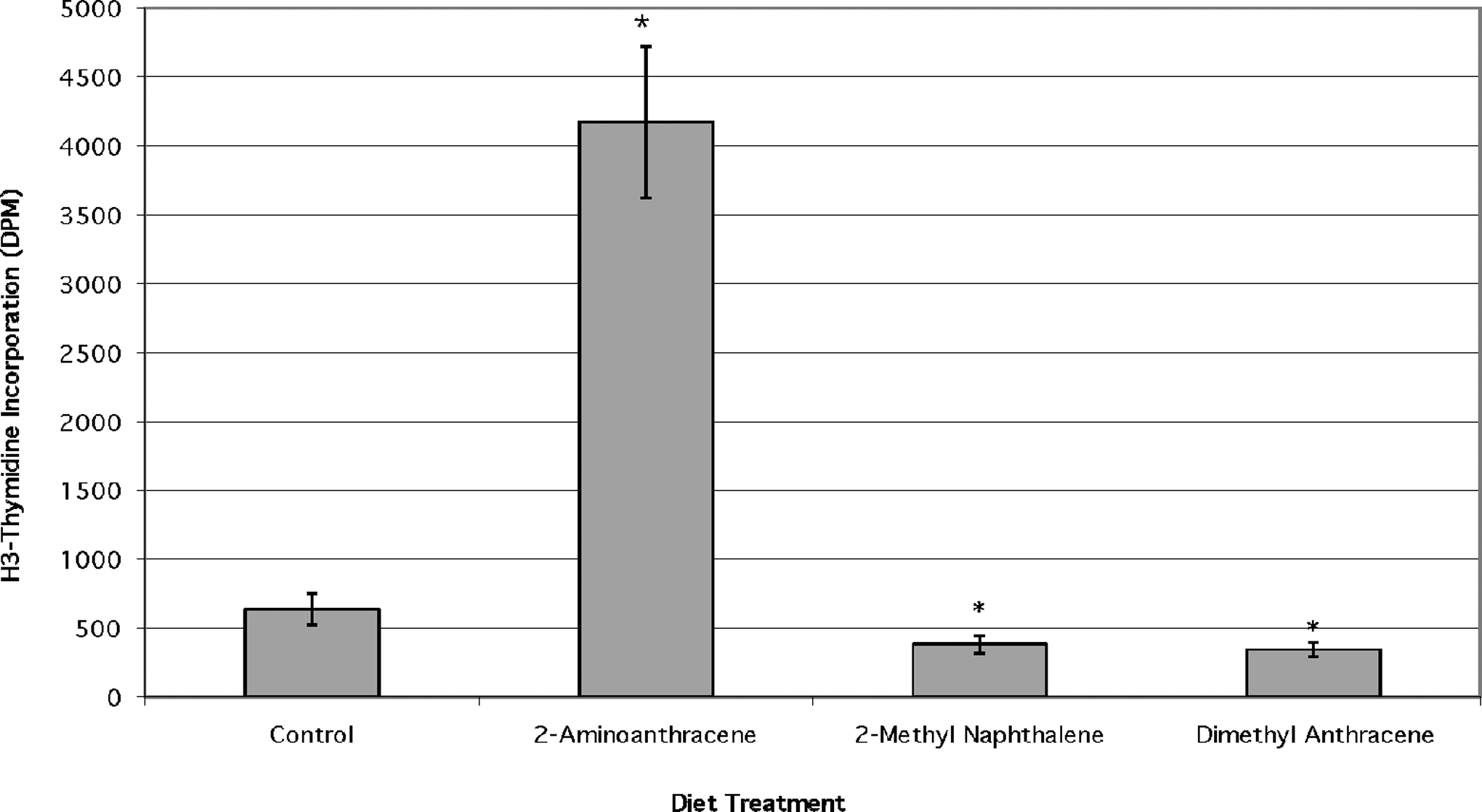
Direct mitogenic activity of dietary exposure to 3 aromatic compounds; n = 6 unless indicated, (*) = significant at P < .02 from no polycyclic aromatic hydrocarbon (PAH) control.
Con A-induced proliferative responses
Peripheral blood lymphocytes treated with Con A only (no PAH controls) were stimulated to significantly higher proliferation levels than the no mitogen controls (P < .02). Lymphocytes from Con A-treated fish fed 2-AA demonstrated proliferation slightly greater than the no PAH control group. However, this difference was not significant (Figure 3). Proliferative response to the T-cell mitogen Con A (optimized at 25 μg/mL) was significantly reduced (P < .02) in lymphocytes from fish fed 2-MN or 9,10-DMA relative to the Con A-only controls. The mean DPM values for the no PAH control Con A-stimulated fish were 11410 ± 2479 DPM as compared with the no mitogen controls (~640 DPM). Although the proliferation response to Con A treatment was suppressed by 2-MN and 9,10-DMA, the responses in these were still greater than the no mitogen controls (baseline cell replication).
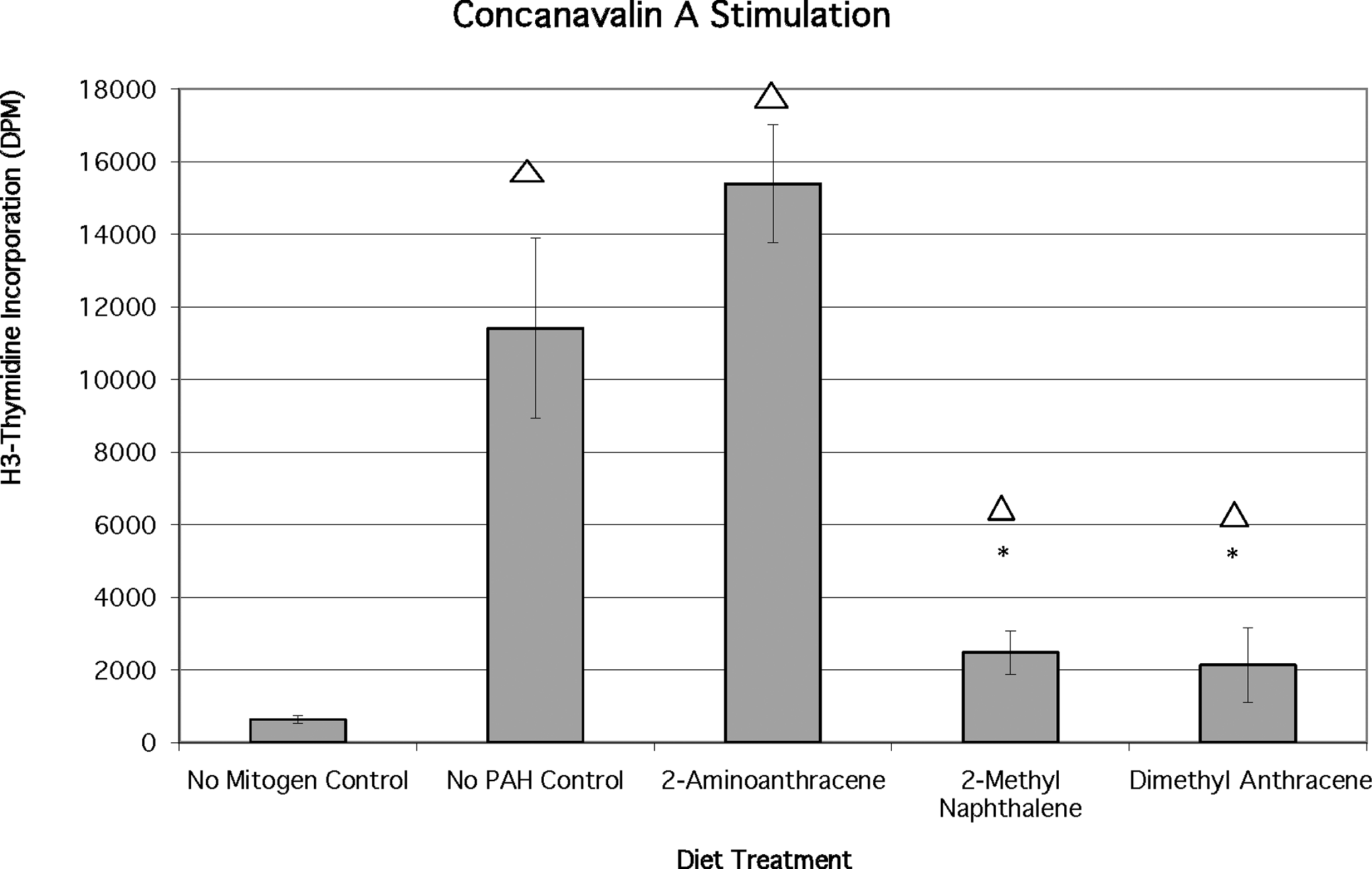
Effects of chronic dietary exposure to selected aromatic compounds on mitogen stimulation by concanavalin A (n = 6 unless indicated), (*) = significant at P < .02 from no polycyclic aromatic hydrocarbon (PAH) control; Δ = significant at P < .02 different from no mitogen control.
Phorbol ester (PMA) stimulated lymphocyte proliferation
Phorbol myristate acetate is not thought to stimulate cell proliferation in the absence of monocytes or exogenous addition of interleukin 2 (IL-2). 33 Bluegill control lymphocytes did not respond to PMA administration (761 ± 305 DPM) because this value is not significantly different from the no mitogen control value (~640 DPM; Figure 4). However, the combination of dietary 2-AA and PMA resulted in a proliferation response that was significantly greater than either the no mitogen controls or the mitogen only controls (P < .02). PBL from 2-MN-exposed fish and PMA-treated cells (Figure 4) demonstrated significant proliferation increases (P < .02) relative to controls. The mean H3-thymidine incorporation for lymphocytes from fish fed 2-AA was 8807 ± 914 DPM, and the mean incorporation for lymphocytes from fish fed 2-MN was 6269 ± 508 DPM. Lymphocytes from fish fed the 9,10-DMA diet and treated with PMA demonstrated proliferation in the same range as the mitogen control group and the no PAH control group.
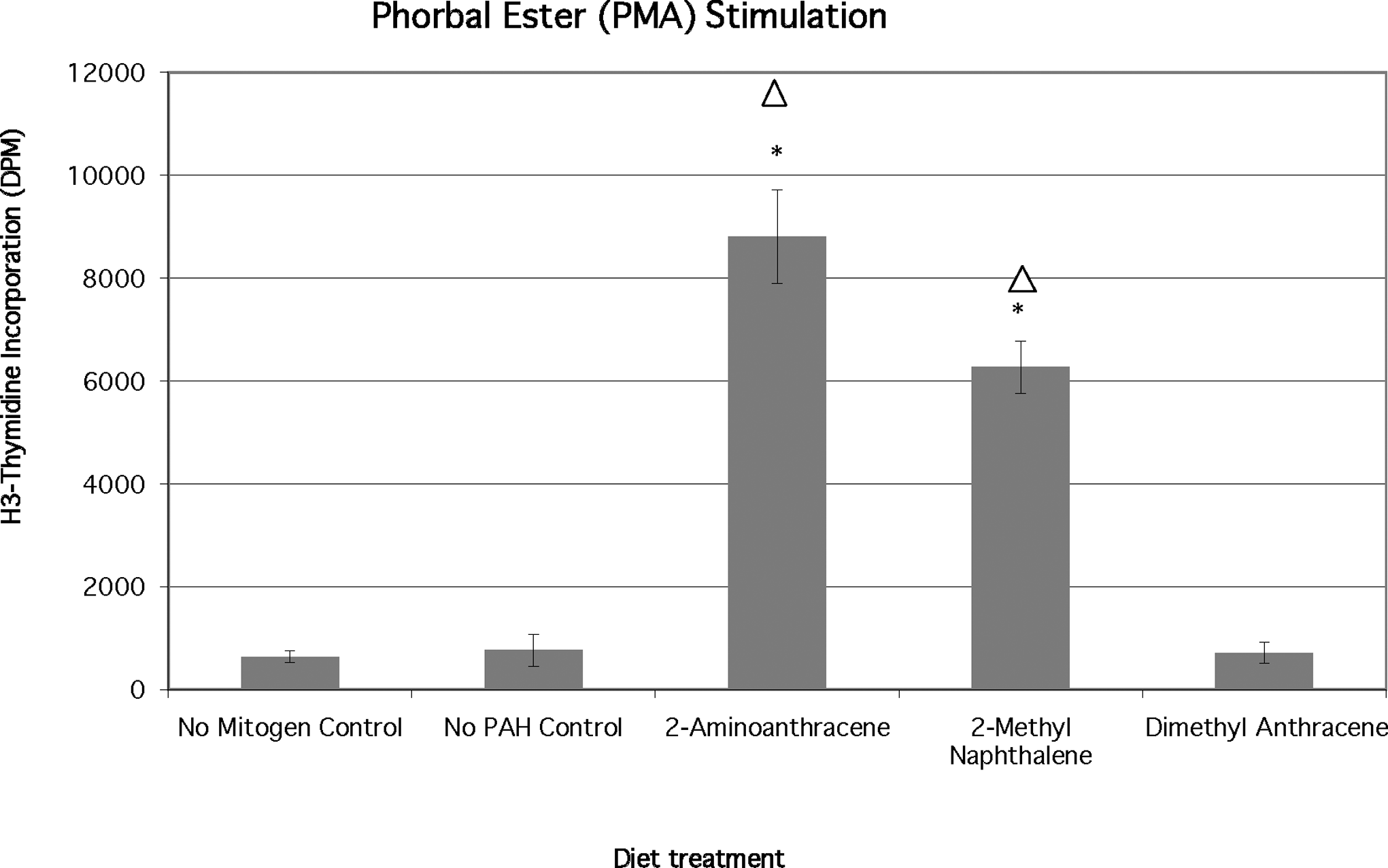
Effects of chronic dietary exposure to selected aromatic compounds on mitogen stimulation by phorbal ester phorbol myristate acetate (PMA; n = 6 unless indicated), (*) = significant at P < .02 from no polycyclic aromatic hydrocarbon (PAH) control; Δ = significant at P < .02 different from no mitogen control.
Calcium ionophore−stimulated lymphocyte proliferation
Calcium ionophore is thought not to stimulate cell proliferation in the absence of monocytes or addition of exogenous IL-2. 33 However, bluegill A32187 only-treated control (no PAH) cells responded significantly (P < .02) to calcium ionophore alone (Figure 5) approximately tripling the response (averaged 2129 ± 645 DPM) as compared with unstimulated (no mitogen) control cells. The proliferation due to calcium ionophore addition was significantly higher (P < .02) in cells from 2-AA-fed fish (10581 ± 1129 DPM) and in cells from 2-MN-fed fish (7419 ± 1548 DPM). The responses of these PAH and mitogen-stimulated cells were also significantly (P < .02) greater than the PAH only stimulated cells (no mitogen controls). Lymphocytes from 9,10-DMA-fed fish incorporated significantly less H3-thymidine than the control (mitogen only) group but were not significantly different from control (PAH only) levels.
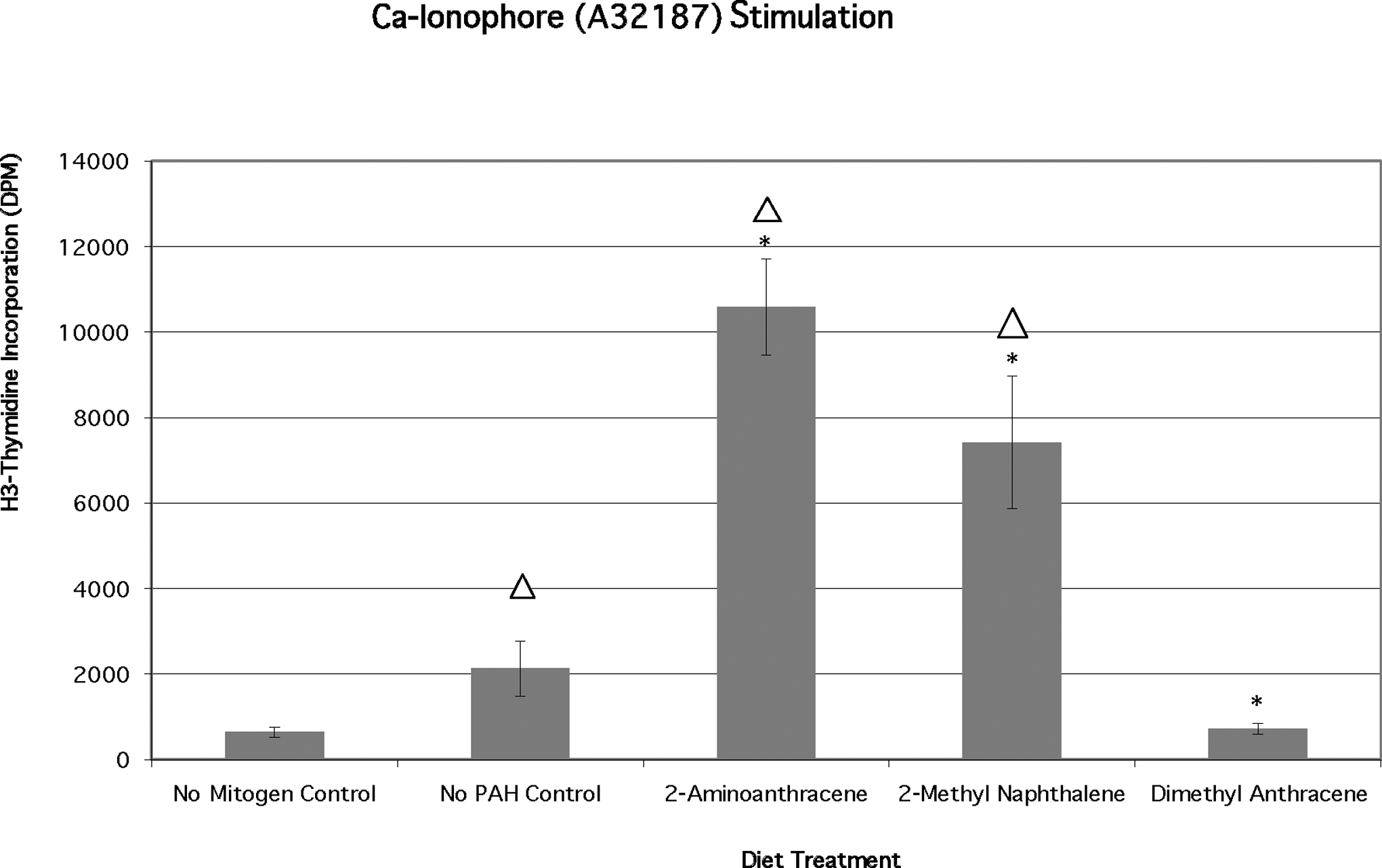
Effects of chronic dietary exposure to selected aromatic compounds on mitogen stimulation by calcium ionophore A32187 (n = 6 unless indicated), (*) = significant at P < .02 from no polycyclic aromatic hydrocarbon (PAH) control; Δ = significant at P < .02 different from no mitogen control
Calcium ionophore/phorbol ester mixture−stimulated lymphocyte proliferation
The largest control (mitogen only) proliferation response was observed in bluegill lymphocytes treated with a combination of calcium ionophore and phorbol ester (13304 ± 2232 DPM; Figure 6). Phorbol myristate acetate/A23187 stimulated a greater proliferative activity than either Con A only or PMA alone. Con A is a T-cell mitogen, while calcium ionophore/phorbol ester can stimulate both T and B cells. 34 Cells from 2-AA-fed fish incorporated significantly less H3-thymidine than the mitogen-only control (7366 ± 848 DPM vs 13304 ± 2232 DPM) group. Cells from 2-MN-fed fish proliferated at a rate approximately equal to the control (mitogen only) group but significantly greater than the PAH only (no mitogen) controls. Significantly less (P < .02) lymphocyte proliferation was observed in 9,10-DMA-fed fish cells (938 ± 357 DPM) treated with the PMA/A23187 combination. The responses of these mitogen- and DMA-treated cells were equivalent to the no mitogen control levels for DMA-only fed lymphocytes. The H3-thymidine incorporation response to PMA/A23187 in 2-AA-treated fish cells was suppressed as compared with 2-AA only controls. The response was also less than that elicited in cells from 2-AA-treated fish by either phorbol ester (Figure 4) or calcium ionophore (Figure 5) alone.
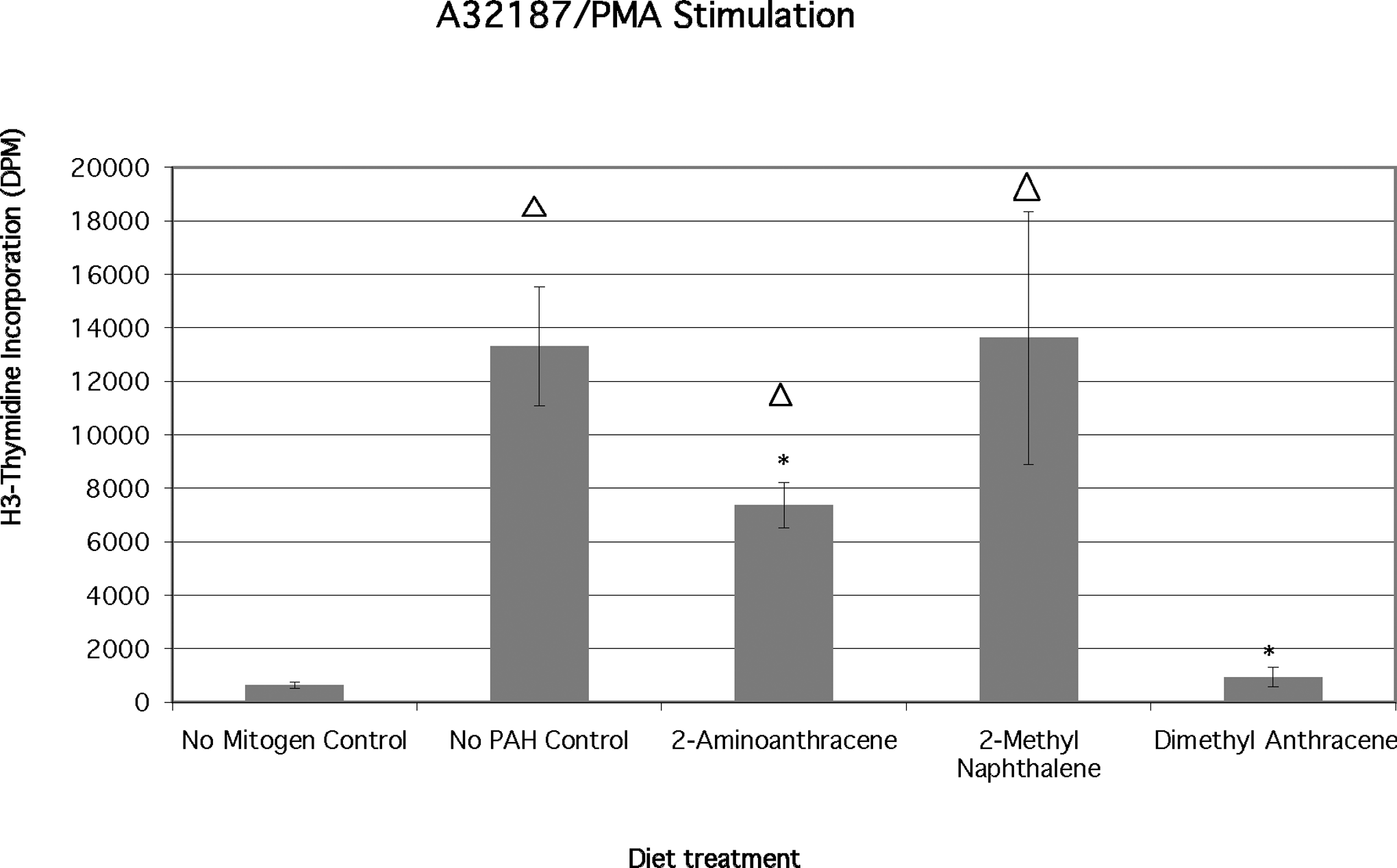
Effects of chronic dietary exposure to selected aromatic compounds on mitogen stimulation by a mixture of calcium ionophore and phorbal ester (n = 6 unless indicated), (*) = significant at P < .02 from no polycyclic aromatic hydrocarbon (PAH) control; Δ = significant at P < .02 different from no mitogen control.
Responses of Fish Exposed to a Mixture of PAHs to Mitogen Stimulation
The proliferation responses of fish lymphocytes to mitogen stimulation after chronic dietary exposure to a mixed diet of all 3 aromatic compounds studied in this investigation revealed that responses were suppressed dramatically (P < .02) by the PAH mixture (Figure 7). The presence of the PAH mixture in the diet suppresses the baseline lymphocyte replacement in fish not treated with any mitogen (note that all values for the PAH mixture are 10× to be visible). Furthermore, there is an almost complete suppression of a stimulatory effect by any of the 3 mitogens added singly or the combination of PMA and Ca-ionophore. All of the suppression responses observed were significant at the P < .02 level.
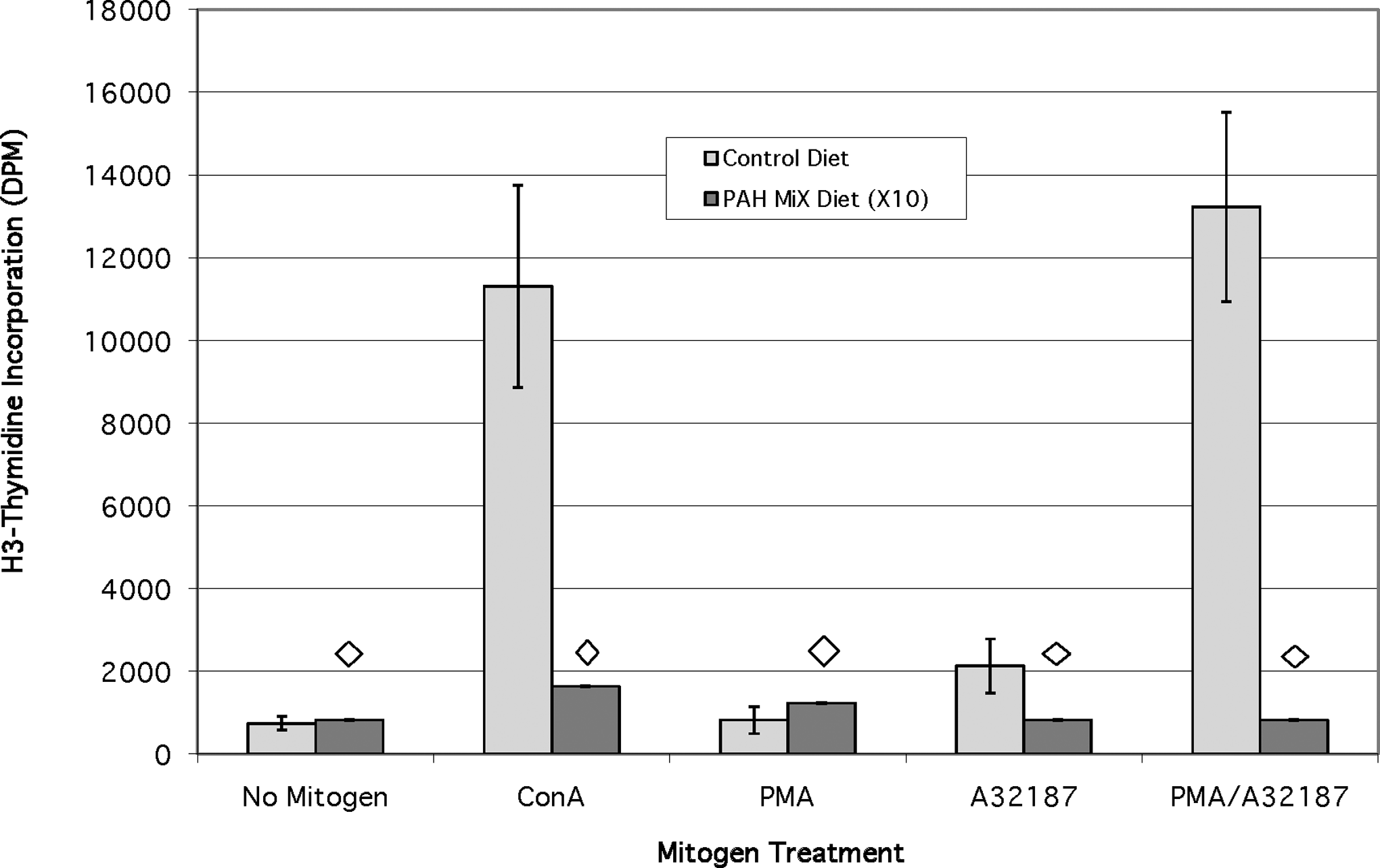
Effects of a chronic dietary exposure to a mixture of 3 PAH on mitogen stimulation response (n = 6). Diamond indicates significant difference (P < .02) from no polycyclic aromatic hydrocarbon (PAH) control diet for each PAH or PAH/mitogen treatment. Values for the PAH mixture diet have been multiplied by 10x to make them visible.
Baseline intracellular calcium in lymphocytes from PAH-fed fish
Resting or baseline intracellular calcium levels in cells from PAH-fed fish were slightly reduced as compared with controls (Figure 8). The lowest intracellular calcium levels were determined in cells from fish fed 2-MN or 2-AA, which correlates with changes in proliferative responses observed with these 2 diets. Calcium levels in cells from fish fed 9,10-DMA were similar to control values. Results are graphed as the value of the ratio of mean channel fluorescence of green/mean channel fluorescence of red multiplied by 500 (Figure 8). The multiplier value of 500 was chosen to yield data ratios between 0 and 100. The mean ratio value for the control group is 70.6 ± 7.1. Mean ratio values for 2-AA- and 2-MN-treated fish were 56.9 ± 1.6 and 56.5 ± 0.4, respectively.
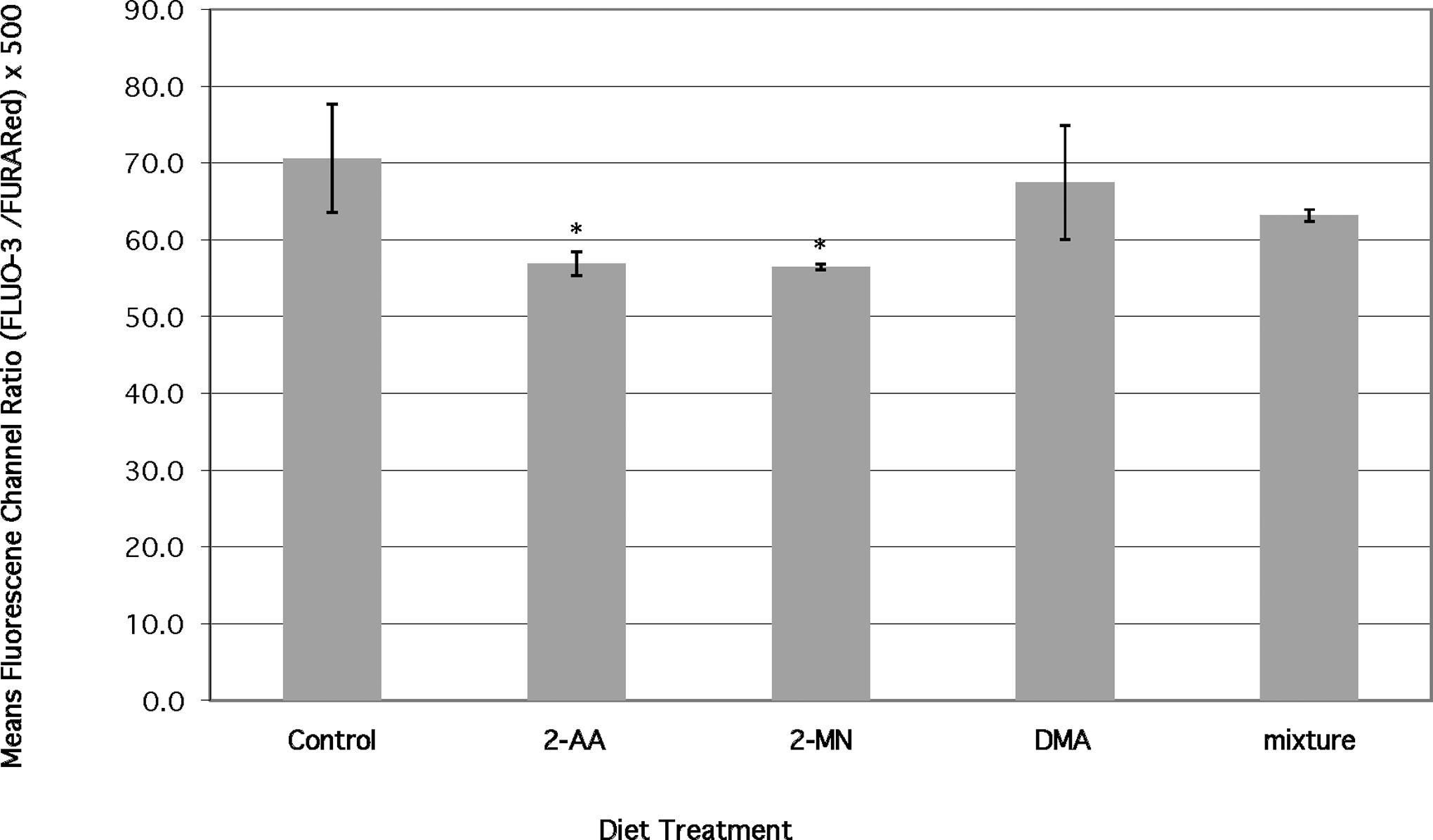
Effects of chronic dietary exposure to selected aromatic compounds on baseline intracellular calcium in lymphocytes (n = 6 unless indicated) (*) = significant at P < .02 from no PAH control.
Discussion
Three carcinogenic PAH compounds were tested for their immunomodulatory effects on bluegill PBL following dietary exposure singly or in a mixture for 14 days and were found to have differential effects. Cell proliferation was measured after treatment with various mitogenic stimulants known to act on lymphocyte-signaling pathways in humans and other mammalian systems but also in some cases in other fish models. Intracellular calcium concentrations were also measured and compared in resting lymphocytes as another measure of potential PAH effects on immune cell function. First, the PAHs were tested for mitogenic activity in the absence of any added mitogen. 2-Aminoanthracene was found to be mitogenic to fish PBL, which is a unique finding. In contrast, 2-MN and 9,10-DMA mildly suppressed baseline PBL replication.
The responses of fish PBL in the absence of dietary PAH exposures when treated with 3 known mitogens in a mitogen stimulation assay revealed that these proliferation responses and their modulation by chemical contaminants have use as potential biomarkers of immunotoxicity in fish models and further that the responses were parallel with responses observed in rodent models such as rat and mouse and also in human PBL. Briefly, Con A was strongly stimulatory, while PMA and A32187 generally yielded little stimulation of PBL. The mixture of PMA and A32187 was however stimulatory to PBL proliferation. In the third phase of the study, PBL isolated from fish fed the 3 carcinogenic PAH individually or as a mixture of the 3 and then treated with the selected mitogens were examined. These results showed a mixture of responses, which demonstrated both stimulatory and suppression effects of the dietary PAH on the mitogen responses on fish PBL in the absence of the PAH as described in the results section.
Bluegill have at least 2 populations of lymphocytes: 1 that responds to phorbol ester-type mitogens (PMA; B-like cells) and 1 that responds to lipopolysaccharide-type (Con A) mitogens (T-like cells). 35,36 Recent chemical and genetic information on fish suggest that there are similar cell-signaling pathways−involved fish lymphocytes as have been documented in rodent models. 37 –39 The mixed T and B lymphocyte populations from bluegill were not separated in this study. However, the responses measured in this study (mitogen-stimulated cell proliferation and intracellular calcium levels) in bluegill should be similar to those observed in mammalian and other teleost T and B lymphocyte populations. Like mammals, 39 –41 fish signal transduction pathways in B cells include: phosphorylation of proteins, second messenger generation, and Ca2+-dependent mechanisms. 39,42 –45 In certain fish models, intracellular calcium levels rise and proteins are phosphorylated upon cross-linking of cell-surface receptors or in response to mitogen stimulation. 39,42 –45
The mitogens used in this study were selected to stimulate different populations of lymphocytes (B-like vs T-like) via different mechanisms. Phorbol myristate acetate−stimulated cell proliferation should activate PKC and components of the ras pathway. Calcium ionophore should increase intracellular calcium levels and activate Ca2+-dependent proteins. The combination of A23187 and PMA should be sufficient to activate cell proliferation by bypassing early protein tyrosine activation steps, mobilizing calcium, and by activating PKC and ras pathways.
A 14-day diet of 9,10-DMA is immunosuppressive in bluegill and exhibited significantly reduced lymphocyte proliferation in response to all mitogens tested: Con A, PMA, A23187, and PMA + A23187 (Figures 3-6). Suppression of immune function after exposure to 9,10-DMA has been demonstrated previously in human T cells 8 but has not been reported in fish. Environmental mixtures of PAH compounds have been demonstrated to have varying effects on lymphocyte proliferation in fish models. Immunosuppression as well as enhanced lymphocyte proliferation has been reported in response to a mixture of PAH. 6,7,9,38,46 –49 Feral fish collected from a contaminated sediment setting showed an 7,46 initial inhibitory effect followed by a proliferative response due to the response of carcinogen-resistant cells proliferating selectively relative to the overall population of cells with chronic exposure (months to years). The later proliferative response observed is considered a clonal adaptation in response to the long-term PAH challenge. 50 The strong suppression of proliferation observed in bluegill cells due to 14-day dietary exposure to the 9,10-DMA in this study (Figures 3-6) is in contrast to proliferation as reported in the environmentally contaminated settings in feral fish. 6 This may be due to various factors. It is possible that in addition to clonal adaptation of feral fish lymphocytes, the mechanism or route of exposure may affect the action of PAH on the fish immune system. The primary route of exposure for environmentally exposed fish is likely via the gills, while in dietary exposures, it is via the gut and liver. These 2 routes of exposure may proceed at different rates, and the extent and mechanisms of metabolism of the PAH may differ, resulting in different effects on lymphocyte proliferation.
The effect of a 2-MN diet on bluegill lymphocyte proliferation is variable depending upon the mitogen used. Proliferative cell response from fish fed 2-MN is depressed in the absence of any mitogen (Figure 2) as well as in the presence of Con A (Figure 3). This response is consistent with previous reports of PAH depression of Con A-stimulated cell proliferation in rodents. 20,51 The effect of 2-MN on cell proliferation stimulated by the calcium ionophore, the phorbol ester, and a combination of the 2 is a novel response not previously observed. With phorbol ester, a diet of 2-MN enhances the proliferative response. This is in contrast to the overall suppressive effect seen with Con A. 2-Methylnaphthalene may mediate alteration of signal transduction in such a way that the overall effect is suppression. Because 2-MN-treated cells respond to calcium ionophore and phorbol ester, it is possible that the 2-MN effect on parts of signal transduction is actually stimulatory. These stimulatory effects may not be apparent until the addition of phorbol ester that activates the ras/PKC pathway followed by the addition of calcium ionophore to stimulate the calcium-dependent pathway. 2-Methylnaphthalene may trigger a small amount of calcium mobilization and PKC/ras pathway signaling, which alone is not sufficient to generate proliferation. However, the additional signal input from phorbol ester or calcium ionophore, is sufficient to stimulate proliferation.
Glutathione, which acts to protect cells from the effects of reactive oxidants, is reduced in DBA/2J mice as a result of 2-MN exposure. 52 It is possible that reactive metabolites of 2-MN bind and activate sensitive proteins in the PKC, ras, or calcium pathways. This type of reactive metabolite-mediated signaling has been demonstrated with other compounds such as nitric oxide, HgCl2, and H2O2. 53,54
Another possible explanation for the suppressive effects of 2-MN on Con A-induced proliferation, in the context of the stimulatory effects in response to A23187 and PMA, may be inhibition of early tyrosine phosphorylation events. By inhibiting early tyrosine phosphorylation events that are essential to all other cell activation events, 2-MN could elicit a reduced response to Con A stimulation. 55
The promutagenic compound 2-AA investigated here does not exhibit a suppressive effect on lymphocyte proliferation when administered by itself; however, when administered in the mixture of immunosuppressive PAH, the overall effect is suppression. By itself, 2-AA has a direct mitogenic or stimulatory effect on cell proliferation (Figure 2). Aromatic amines have been shown to stimulate cell proliferation. Lee 56 reported enhanced proliferative response in murine splenocytes to N-hydroxy-2-aminofluorene, a primary metabolic product of 2-aminofluorene.
2-Aminoanthracene undergoes P450 activation, like 2-aminofluorene, to an N-hydroxyamine. The hydroxyamine can be O-acetylated and subsequently converted to a reactive electrophile (nitrenium ion) that covalently binds DNA, ultimately leading to mutational events. One possible mechanism of action is that a reactive metabolite of 2-AA could bind DNA, mimicking transcription factors, and thus stimulate cytokine transcription.
Another possible explanation for the proliferative response seen with 2-AA in the absence of any other mitogen is that 2-AA or a metabolite may serve to activate or stimulate a cell-surface receptor. Aromatic amines like 2-acetylaminofluorene (2-AAF) have been reported to bind surface receptors such as the estrogen receptor. It may be possible that the effect of 2-AA is on a different cell receptor inducing proliferation in the absence of a mitogen.
The results of feeding a mixture of PAHs (Figure 7) leads to the conclusion that exposure to one or more of these alkylated PAHs may have a cumulative effect on the functioning of the fish cellular immune system. The almost complete suppression observed in response to feeding the mixture seen in Figure 7 was greater than the suppression observed with any single compound. Furthermore, although the mixture contained the mitogenic and stimulatory compound 2-AA, these effects were overwhelmed by the presence of the other 2 alkylated PAH in the diet. It should also be noted that the innate immune cell replacement (mixture diet and no mitogen added) was also suppressed significantly (P < .02) to nearly nondetectable levels.
The baseline intracellular calcium level comparisons in this study are reported as the ratio of the mean channel green fluorescence to red fluorescence chromophores. 57 The differences in mean channel ratios observed in this study were relatively small but represent statistically significant changes in intracellular calcium concentrations as reported in other studies (eg Burchiel 4 ; Figure 8). The differences in the means of the ratio data observed are ~14 units, which represent a decrease in intracellular calcium levels of approximately 22% relative to unexposed controls for both 2-AA- and 2-MN-treated fish.
Alterations in intracellular calcium levels have also been linked to cell viability. The reduction in baseline or resting Ca2+ may be an indication of approaching cell death. Cells from fish fed 2-MN and 2-AA demonstrated the lowest levels of intracellular calcium. These cells were more proliferative in responses to stimulation than were cells from fish fed the 9,10-DMA diet. The exact mechanisms involved in reduction of cytosolic baseline calcium by the PAHs in this study are not clear. One possible mechanism may involve direct damage of the plasma membrane, resulting in reduced transport efficiency for influx of extracellular calcium. 2-Aminoanthracene is known to form protein adducts (McCarrol and Means, unpublished data) that may inactivate or suppress the activity of the protein.
Carcinogenic PAH cause modulations of signal transduction pathways in human and murine lymphocytes. 7,12-Dimethylbenzanthracene stimulates phosphorylation of signaling proteins in both a human T cell line and murine B cells. 10,13 7,12-Dimethylbenzanthracene also stimulates a sustained rise in intracellular Ca2+ in both T and B lymphocytes. 14 Other PAH such as BaP, dibenz[a,h]anthracene and 9,10-DMA also stimulate a sustained increase in intracellular Ca2+ levels in a human T cell line. 58 These compounds also cause highly variable effects on lymphocyte proliferation in fish. 6,7,46,59
Phorbol esters activate multiple signaling proteins in vertebrates. 24 –26,59 The calcium ionophore (A23187) effectively mimics the actions of inositol 1,4,5-trisphosphate (IP3) in the second messenger pathway by mobilizing intracellular and extracellular calcium stores. The phorbol ester, PMA, with calcium ionophore (A23187) used in tandem are pharmacologic agents that stimulate T and B lymphocyte proliferation in murine, human, red drum, and catfish cells. 30,42 Catfish lymphocytes are not stimulated with phorbol ester alone, 30 but in another species of fish, red drum, lymphocytes proliferate in response to PMA by itself. 42 A23187 is a calcium ionophore that increases intracellular calcium levels by mobilizing both intracellular and extracellular calcium stores. Phorbol myristate acetate and A23187 together are mitogenic in lymphocytes and were demonstrated to have strong mitogenic activity in bluegill PBL.
The pathways involved in Con A-stimulated cell activation are not as clear as for PMA and A23187. Con A-stimulation begins at the cell surface and results in tyrosine phosphorylation of proteins in T cells 60 as well as leading to an increase in intracellular calcium in both T and B cells. 61 However, the exact pathway of Con A activation remains to be fully investigated. Lymphocyte proliferation stimulated by Con A is thought to be dependent upon Con A binding to CD3. 60 The CD3 receptor is known to be expressed in fish PBL. However, stimulation by Con A and stimulation by antibodies to CD3 result in differing amounts of cell proliferation and Ca2+ mobilization. 62
In summary, the teleost fish has been demonstrated to be a useful model for testing of immunomodulatory effects of PAH administered through the diet in both mitogen stimulation assays and flow cytometric assays of intracellular calcium levels. Dietary exposure (14 days) of bluegill to 2-MN, 9,10 DMA, and 2-AA has demonstrated the immunosuppressive and immunostimulatory activity of these compounds to the cellular immune system. A mixture of the 3 PAHs administered in the diet was highly immunosuppressive. 2-Aminoanthracene has also been identified as a direct-acting mitogen in this fish model. Further research into the interactions with specific cytokines or cell-surface receptors are required to fully elucidate the mechanisms involved at the molecular or gene level. Also, ongoing genomic expression analyses of the genes involved in regulation of the cellular immune system in these fish can add significantly to our understanding of these effects.
Footnotes
Acknowledgment
The authors wish to thank Dr Burchiel for sharing his laboratory and his insights into this research at a critical stage of this work.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: The US Department of Energy and the Louisiana Educational Quality Support fund for grant DOE/LEQSF 2000-05-01 support to JCM for this research.
