Abstract
This study was conducted to determine the in vitro dermal absorption of ethylene glycol (EG) through dermatomed human abdominal skin (containing epidermis and dermis), obtained from cadavers within 24 hours of death and kept frozen until processed. Three formulations of EG (neat, 50%, and 10% aqueous solutions) were applied in triplicate to skin samples from 6 donors, and placed in Teflon Bronaugh flow-through diffusion cells. Barrier integrity of each sample was evaluated with 3H-H2O prior to applying EG and only data from samples passing the test were used. A physiological receptor fluid was pumped beneath the skin samples and collected in a fraction collector at predetermined time points through 24 hours. Possible volatilized EG was trapped in a charcoal basket located above each skin sample. Each skin sample was treated with an infinite dose of 500 µL of EG formulation/cm2. At the end of 24 hours, volatilized EG trapped in the headspace was collected, the unabsorbed dose was removed from the skin and the skin was rinsed, tape stripped, and solubilized along with a rinse of the flow-through cells, and total radioactivity was determined. Only a small fraction (≤1%) of the applied EG was absorbed in 24 hours, of which <0.7% penetrated through the skin and ~0.4% remained in the skin. Recovery (mass balance) of the applied EG was between 93% and 99%, which further validated the observed low dermal penetration of EG. The net penetration of the applied EG over 24 hours was concentration proportional, comprising 2.97 ± 0.78, 1.75 ± 0.62, and 0.23 ± 0.12 mg/cm2 of the neat, 50%, and 10% formulations, respectively. The steady-state flux of EG was established between 16 and 24 hours. The mean steady-state flux of EG through dermatomed skin was 217, 129, and 15 µg/cm2h for the neat, 50%, and 10% aqueous formulations, respectively, consistent with concentration-proportional penetration of EG. The steady-state permeation coefficient (Kp) for EG was low, between 1.5 × 10−4 and 2.6 × 10−4 cm/h. These findings demonstrate that EG dermal penetration is expected to be very low and to be slow, indicating very limited systemic or internal dose of EG due to dermal exposure.
Keywords
Introduction
Ethylene glycol (EG) is a colorless, odorless, and sweet tasting liquid. Ethylene glycol is a high production volume chemical with wide industrial and commercial applications. 1 The estimated annual world production of EG is about 18 million tons. The largest use of EG (~82% of the total production) is in the plastics industry, where it is used in the production of polyester fibers, resins, and films. The largest commercial uses are as automotive coolants, heat transfer fluids, and aircraft deicers.
Ethylene glycol has been studied extensively in animals for its acute, renal, and developmental toxicity. 2 –13 Toxicity from EG following acute high-dose exposures arises from oxidation to the metabolite glycolic acid (GA). 3,14 Accumulation of high levels of GA results in metabolic acidosis. Ethylene glycol can also induce renal toxicity in animals and humans following chronic exposure. Renal effects arise from metabolism of the test material to GA, and then subsequently to oxalic acid (OX), followed by deposition of calcium oxalate crystals in the proximal tubules of the kidney. This sequence of events is based on the extensive animal toxicity database, several human case reports of intentional or accidental overdoses, and research into the mechanisms of human kidney stone formation. However, there is a distinct difference in the developmental toxicity of EG when administered to different species and by different routes. It is a mouse teratogen when administered through oral route 11 but not via the dermal 12 or inhalation 10 routes. Rat sensitivity to EG developmental toxicity is approximately the same as for the mouse, 3 and rabbit does not manifest any developmental toxicity, 13 although high doses are lethal. For developmental toxicity, human relevance is less clear with no reported cases of human developmental effects induced by EG. The likelihood of developmental toxicity occurring in humans through occupational or consumer exposures is considered negligible, primarily because of the high-dose rates needed to produce this effect in rodents. 15
As stated above, EG is present in commercial products such as paints, automotive coolants, aircraft deicers, and heat transfer fluids. These uses of EG comprise the majority of potential for human exposures, and the most common route of potential human exposure to EG is through skin contact. The data on toxicity of EG from dermal exposure is limited to rabbits whose skin typically is more permeable to chemicals than humans. 16
Data on the permeability of EG through human skin is limited; Loden, 17 Driver et al, 18 and Sun et al 19 have reported dermal penetration of EG through cadaver and fresh human skin. The reported values for the skin penetration of EG vary by up to 1300-fold across studies (Loden 17 : 118 µg/cm2h and Driver et al 18 : 0.02-0.9 µg/cm2h); therefore, it is not clear how reliable these data are for dermal exposure assessment. Sun et al 19 conducted their study using fresh full-thickness skin samples obtained from surgical procedure and obtained lower penetration rates (7-13 µg/cm2h) than Loden 17 who conducted the study using dermatomed skin. Driver et al 18 reported a much lower rate of penetration of EG through dermatomed skin; however, they applied very low (finite) doses of EG to skin samples. The use of dermatomed skin is preferred in the determination of dermal penetration of a chemical as it determines penetration through the keratinized layer of stratum corneum and epidermis and avoids sequestration of the chemical in the dermis, which may occur when full-thickness skin is used. Because of the availability of only limited data on the dermal penetration of EG through human skin, and the highly variable results among studies, regulatory agencies have difficulty in deciding which data to use for human risk assessment. Therefore, the current study was conducted to better define the dermal penetration of EG through human skin. The results of this study will help in validating the results of earlier studies and the refined data will be useful in a proper risk assessment of the dermal exposure of EG to human.
Materials and Methods
Chemicals
14C-labeled EG [(1,2-14C) ethylene glycol] in water (385 µCi/mL) was purchased from Moravek Biochemicals (Brea, California). The radiochemical purity of EG was 99.9%. Nonradiolabeled EG with a chemical purity of 99.9% was obtained from Sigma-Aldrich (Milwaukee, Wisconsin). The received test material was further analyzed by gas chromatography-mass spectrometry (GC-MS) to confirm the identity. The mass spectrum obtained matched a library spectrum (Hewlett Packard NIST/EPA/NIH Mass Spectral Database) for EG. Tritiated water (3H-H2O), used to determine skin membrane integrity, was purchased from PerkinElmer Life Sciences, Inc (Boston, Massachusetts). All other chemicals used were of the purest grade available and obtained from standard sources.
Dose Preparation and Analysis
The dosing solutions were prepared at 3 different concentrations (neat, 50%, and 10% in water). The neat dosing solution was prepared by mixing concentrated 14C-EG and nonradiolabeled EG. The 14C-EG was then concentrated by evaporating water from the stock solution. The other dosing solutions (50% and 10%) were prepared by adding nonradiolabeled EG and MilliQ water to neat 14C-EG. Homogeneity and concentration of the dose solutions were determined by taking 2 weighed aliquots from different regions of the containers and analyzing for radioactivity. Radioactivity was determined using a Packard 2900TR (Packard Instrument, Downers Grove, Illinois) liquid scintillation counter (LSC). Counts per minute were corrected for quench and background and then converted to disintegrations per minute (dpm). The average 14C-EG-derived radioactivity in dose solutions was 0.016, 0.020, and 0.022 mCi/g in the neat, 50%, and 10% formulations, respectively (data not shown). The dose solutions were found to be homogeneous (data not shown). The dose solutions were stored in a refrigerator at 5 ± 4°C and used after bringing it to room temperature at least half an hour before the experiments. On each dosing day, specific activity of each dosing solution was determined in weighed aliquots by LSC, separate aliquots were also analyzed for radiochemical purity and stability by high-performance liquid chromatography with radiochemical detection (HPLC/RAD). The EG in the dose solution preparations was stable during the course of the study, about 1 month (data not shown).
Human Skin Source and Preparation
The dermal penetration of EG was determined in human cadaver skin. Abdominal skin samples were obtained within approximately 24 hours of death as required by the Standards of the American Association of Tissue Banks. 20 Skin samples were obtained from the International Institute for Advancement of Medicine (IIAM, Jessup, Pennsylvania). The skin samples were shipped on dry ice and stored frozen at −20°C until used. Abdominal skin from 6 individuals, 3 males aged 27 to 69 years, and 3 females aged 54 to 73 years, were used in this study.
On the day of experiment, skin samples were cleaned of underlying fat if needed, and thawed at room temperature. Once thawed, each skin sample was sprayed with a mild 1% soap solution and dermatomed with a mini-dermatome (Vincent Willey, Tyne & Wear, England) to obtain a split thickness of 200 to 500 µm containing epidermis and the top layer of dermis. The thickness of the dermatomed skin samples ranged from 260 to 410 µm. Dermatomed skin samples were placed on a gauze saturated with cell culture medium (Minimal Essential Medium [MEM]; Gibco, Carlsbad, California), while other skin sections were processed. Skin samples were visually inspected, and sections free of blemishes and of the appropriate thickness were chosen from each donor. Three disc-shaped pieces, large enough to fit the flow-through diffusion cells, were punched out from each skin sample. Each piece of the skin was rinsed with ultra pure water and its thickness measured with a digimatic micrometer (Mitutoyo Corporation, Japan), before being placed in the Bronaugh flow-through diffusion cell system. 21,22 The surface area of the skin available for dosing after mounting in the diffusion cell was 0.64 cm2. The receptor fluid was pumped beneath the skin samples at a flow rate of 1.9 to 2.1 mL/h.
The receptor fluid was a physiological buffer (Hank Balanced Salt Solution with 0.6% HEPES, 0.005% Gentamicin, and 4% bovine serum albumin, pH 7.4; Gibco). The pH of the receptor fluid was adjusted to 7.4 with NaOH and filtered sterilized through a 0.22 µm filter before use. The receptor fluid was pumped beneath the skin samples at a constant rate during the course of the exposure (24 h). The receptor fluid was maintained at 37°C to keep temperature on the skin surface at 32 ± 1°C. Experiments were conducted under semioccluded conditions.
Evaluation of Barrier Integrity of Skin
Barrier integrity of each piece of skin was determined before initiation of the penetration experiments. Each piece of skin was applied with an infinite dose (200 µL containing 0.75 µCi/g radioactivity) of 3H-H2O for 20 minutes; the applied 3H-H2O was removed after 20 minutes of exposure by blotting the skin surface and the skin was rinsed with Millipore reverse osmosis water and dried by gentle blotting. Aliquots of receptor fluid were collected in preweighted vials during the exposure and collection continued after removal of the applied 3H-H2O for an additional 60 ± 10 minutes. After the completion of barrier integrity test but before its results were determined, all skin samples were applied with appropriate 14C-EG dose solutions. Receptor fluid aliquots were analyzed for radioactivity by LSC and the barrier integrity of the skin samples was assessed as soon as the data become available. Three skin samples from 6 donors were used and at least 2 of the 3 pieces from each individual donor had to meet the barrier integrity criteria to include data from that individual in the final analysis. Skin integrity acceptance criteria were established previously in our laboratory and reviewed by Meidana and Roperb. 23 Acceptable barrier integrity results were that at least 2 of the 3 skin samples from each individual donor must have ≤0.21% absorption of 3H-H2O, and the coefficient of variation for a minimum of 2 replicate skin samples from each donor must be less than 60%.
Application of 14C-EG Onto Skin Samples
Weighed infinite doses (484-588 mg/cm2) of 14C-EG (neat, or formulated [50% or 10% aqueous solution]) were drawn into a 1-mL glass syringe and applied to the skin samples mounted in the diffusion cells of the Bronaugh flow-through apparatus. 21 The actual amount of dosing solution applied to each skin sample was determined by weighing the syringe prior to and after dosing. Skin samples were exposed to 14C-EG for 24 hours. The volume of 14C-EG applied to each skin sample was enough to provide an infinite dose for 24 hours, without depleting the dose reservoir at the apical site of the skin or affecting the rate of penetration.
Sample Collection and Processing
After the application of 14C-EG, the receptor fluid, which was pumped through the individual flow-through chambers, was collected directly into preweighed scintillation vials at 1, 2, 4, 6, 8, 10, 12, 14, 16, 18, 20, 22, and 24 hours post-dosing using a fraction collector. Immediately after the application of 14C-EG, a charcoal basket was placed at the top of the flow-through cell in a specially designed holder to collect any volatilized radioactivity from the surface of the skin. At the end of 24-hour exposure period, air was gently flushed through the headspace above the skin for approximately 2 minutes to force the vapors into the charcoal basket. This method has proven to help account for all the applied test material (mass balance ≥95%; unpublished data). The charcoal baskets were then removed, extracted in acetone, and radioactivity determined by LSC. The unabsorbed 14C-EG was removed from the application site skin by gently blotting with cotton-tipped applicators and by rinsing several times with 0.7 mL of 1% aqueous mild soap solution, which was then blotted using dry-cotton-tipped applicators. The rinse and swabs were combined, extracted in acetone, and radioactivity determined.
After washing the application site, the skin sample was removed from the flow-through cell, placed on Saran wrap, and tape stripped for a maximum of 20 times with D-Squame adhesive tape to remove the stratum corneum and any radioactivity remaining on the skin surface. Tape stripping was discontinued if the epidermis ruptured. All tape strips used for each individual cell were combined and extracted in acetone. The used Saran wrap was extracted in acetone separately. Skin discs were digested in a 35% aqueous solution of tetraethylammonium hydroxide (TEAH) and then neutralized with 6N HCl. The holders, which were used to hold charcoal baskets over the skin, were placed in ethanol for extraction. The diffusion cells were rinsed with ethanol. The rinses and extracts were analyzed for radioactivity by LSC.
Dermal penetration of the applied 14C-EG dose was measured by summing radioactivity in 24-hour receptor fluid, and the remaining radioactivity in the skin after tape stripping. The unabsorbed dose was determined by summing radioactivity in the dose removed from the skin surface, and radioactivity found in skin washes, swabs, and tape strips. The volatilized portion of the applied dose was measured by analyzing charcoal baskets and holders for radioactivity. The overall recovery was calculated as the sum of radioactivity recovered from charcoal baskets, holders, skin surface, diffusion cell rinses, skin, Saran wrap, and receptor fluid.
The study followed Good Laboratory Practice (GLP) guidelines. 24 The study generally followed Organization for Economic Co-operation and Development (OECD) 428 guideline for in vitro dermal penetration, 25 with appropriate modifications to address chemical-specific issues.
Data Analysis
The percentage of absorbed and volatilized dose remaining on the skin surface after 24 hours was determined relative to applied dose (applied radioactivity). The percentage absorbed dose may not be of much value when infinite dose is used; however, it was determined for the completeness of the study and mass balance. Data are presented as mean ± standard deviation of 2 or 3 replicates for each of 3 (50%) or 4 (neat and 10% formulations in water) donors. Three replicates of each donor skin were applied with each formulation; 1 donor in each formulation had only 2 replicates that passed the skin integrity test. Parameters associated with the dermal penetration of 14C-EG over 24 hours were calculated using following formula:
Percutaneous penetration of 14C-EG of the applied dose through the skin:
A flux curve was created by plotting penetration of 14C-EG (µg equivalent per cm2 of skin) that passed through the skin into receptor fluid over time, based on the radioactivity associated with the test article. Steady-state flux is achieved when the radioactivity content in the receptor fluid becomes steady from 1 hour to the next and the concentration of test article in the donor compartment is not diminished (infinite dose). The flux curves were visually inspected and analyzed to determine the best estimate for the linear portion of the curve. The slope for the linear portion of the curve was estimated by linear regression and represents the steady-state rate of penetration (steady-state flux).
Permeability coefficient (Kp):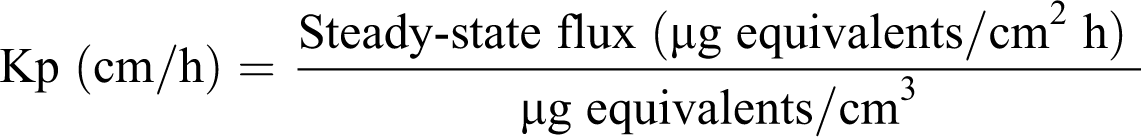
14C-EG remaining in the skin after 24 hours:
Absorbed 14C-EG:
Unabsorbed 14C-EG:
Volatilized 14C-EG: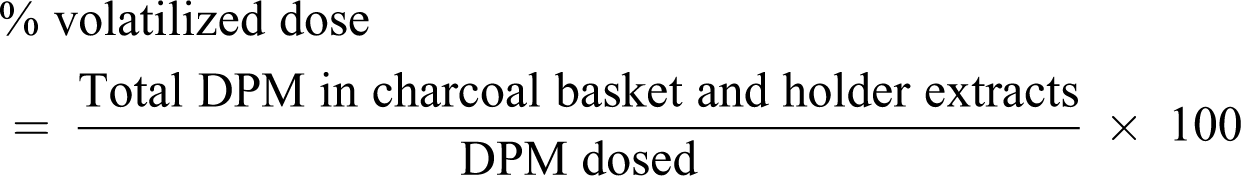
Results
At least two skin samples from 5 of the 6 donors met all the established criteria of barrier integrity for usable data (20 minutes absorption of 3H-H2O <0.21%; coefficient of variation [CV] <60%); a total of 6 skin samples mounted in Bronaugh flow-through cells did not meet the established criteria. At least 2 of the 3 samples of all donors passed the barrier integrity test, with the exception of 1; none of the samples of that donor passed the barrier integrity test. The samples that did not pass the barrier integrity test were excluded from the analysis of the dermal penetration of EG.
A total of 543 ± 18, 270 ± 9, or 51 ± 1 mg EG per cm2 containing 7.3 to 11.1 µCi/cm2 radioactivity was applied to each skin sample of neat, 50%, or 10% aqueous formulations, respectively (Tables 1). The applied dose roughly represented the expected dose proportionality. Skin samples in each donor cell of the Bronaugh flow-through diffusion cell system were treated with 324 to 348 mg of the dose formulations, which provided enough EG on the apical side of the skin to last 24 hours without depletion, a requirement for infinite dosing conditions. This was also confirmed by the amount of the dose (92%-97%) recovered from the site of application 24 hours after dosing (skin rinses [40%-98%], swabs [1%-44%], tape strips [<0.4%]; separate component data are not shown; Tables 2 ). The applied dose was not sequestered in the skin as most of the remaining dose formulations were easily aspirated with a micropipette (data not shown).
Dosing of Ethylene Glycol-Containing Formulations and Radioactivity to Skin Samples

a mg solution/cm2 = mg dose solution per cell ÷0.64 cm2 (dosing area of skin).
b mg EG/cm2 = mg dose solution × formulation (1, 0.5 or 0.1) ÷ 0.64 cm2 (dosing area of skin).
c μCi/cm2 = mg dose solution/cm2 of skin × Dose solution specific activity (μCi/mg).
Disposition of Radioactivity Following Application of 14C-Ethylene Glycol to Human Skin In Vitro
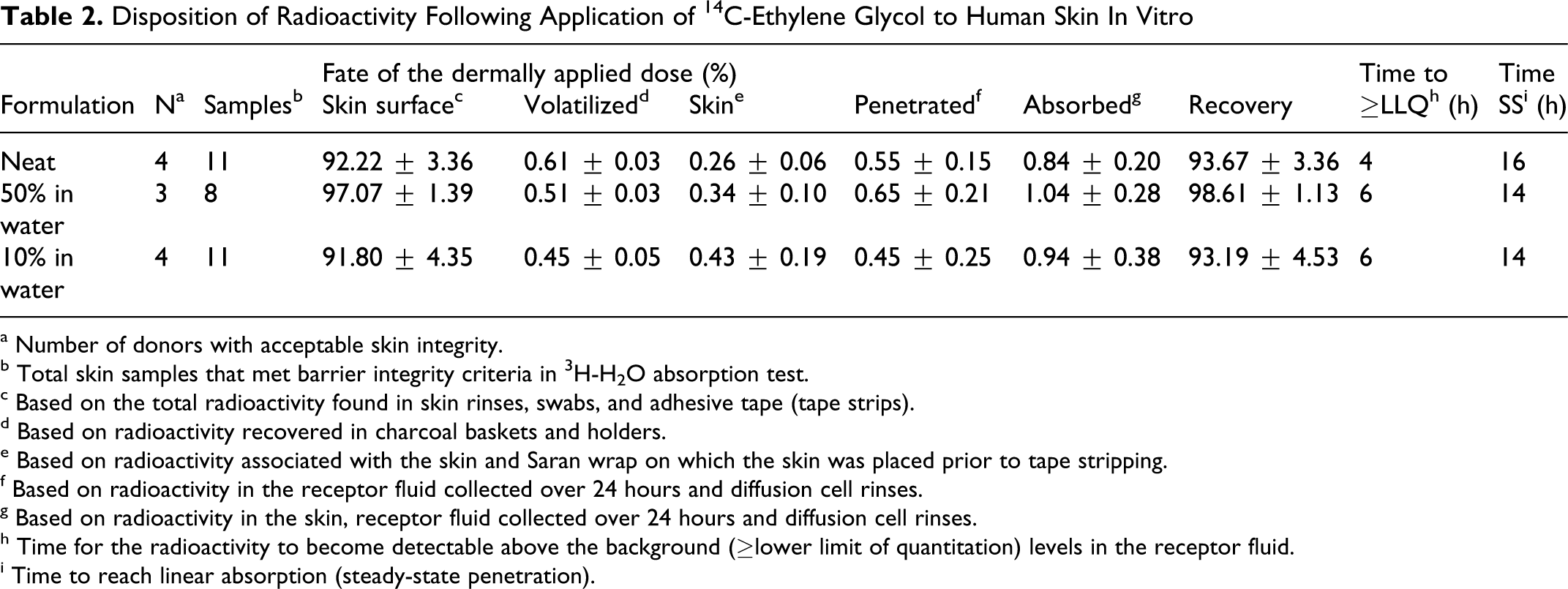
a Number of donors with acceptable skin integrity.
b Total skin samples that met barrier integrity criteria in 3H-H2O absorption test.
c Based on the total radioactivity found in skin rinses, swabs, and adhesive tape (tape strips).
d Based on radioactivity recovered in charcoal baskets and holders.
e Based on radioactivity associated with the skin and Saran wrap on which the skin was placed prior to tape stripping.
f Based on radioactivity in the receptor fluid collected over 24 hours and diffusion cell rinses.
g Based on radioactivity in the skin, receptor fluid collected over 24 hours and diffusion cell rinses.
h Time for the radioactivity to become detectable above the background (≥lower limit of quantitation) levels in the receptor fluid.
i Time to reach linear absorption (steady-state penetration).
Based on the radioactivity recovered from the charcoal baskets, volatilization of the applied EG during the exposure period was negligible (<0.7% of applied dose; Table 2), as was expected given the low vapor pressure of EG (0.092 mm Hg at 25°C). 15 The total absorption (sum of penetrated and skin-bound radioactivity [after washing and tape stripping] at the end of the experiment) of the dermally applied infinite 14C-EG dose after 24 hours of exposure was ≤1% (Table 2). Recovery of the applied 14C-EG-derived radioactivity from the system was between 93% and 99% of applied dose, which further validated the low dermal penetration of EG observed in this study. At the end of the study (after 24 hours of exposure), only 0.26% to 0.43% of the applied dose remained associated with the skin tissues, demonstrating a low capacity of the dermatomed skin to retain EG during the steady-state penetration phase (Table 2).
The net penetration of the applied EG through the skin samples over 24 hours was 2.97 ± 0.78, 1.75 ± 0.62, and 0.23 ± 0.12 mg/cm2 for the neat, 50%, and 10% formulations, respectively (Figure 1 ). Penetration of the total amount of 14C-EG-derived radioactivity through dermatomed skin over 24 hours was essentially concentration proportionate. The total dermal penetration of the 50% and 10% aqueous EG formulations, were ~59% and ~8% that penetrated for the neat EG, respectively. The concentration-proportional change is also depicted in Figure 1, with the expected decreases in total penetration of EG at the 50% and 10% formulations compared to the neat EG, correlating with the respective dilution factors. There was approximately 4 to 6 hours of lag time for the radioactivity to become detectable above the background levels in the receptor fluid, representing delay due to diffusion through the skin; it took about 14 to 16 hours, depending on concentration, to reach linear absorption (Tables 2, Figure 2 ).
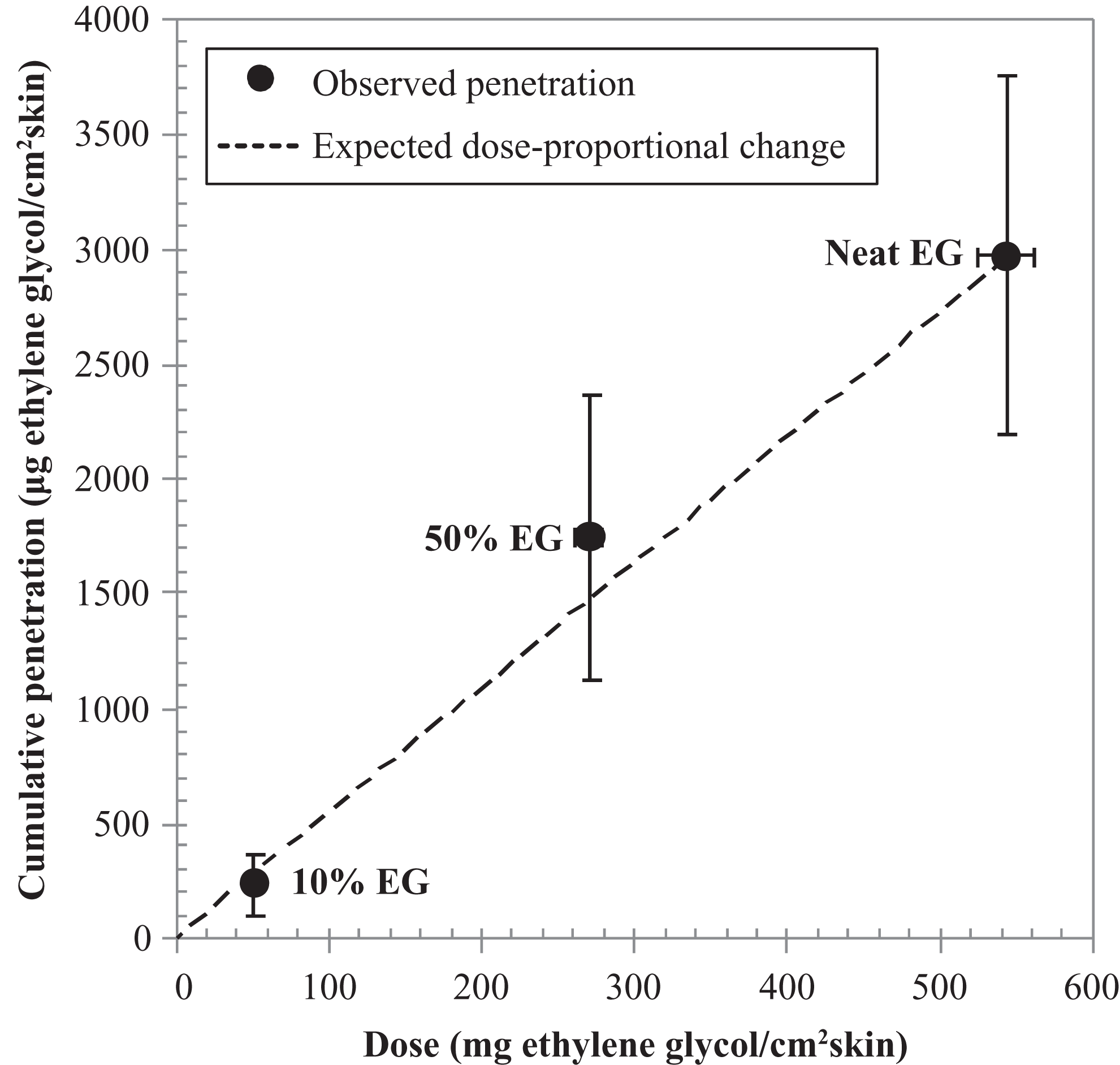
Correlation between dose of 14C-ethylene glycol (EG) applied to human skin and cumulative penetration through human skin into receptor fluid over 24 hours. Symbols represent measured mean ± standard deviation (n = 8-11 samples from 3 to 4 donors), and the dotted line represents the expected dose-proportional increase based on the actual applied EG to skin samples at 3 different formulations.
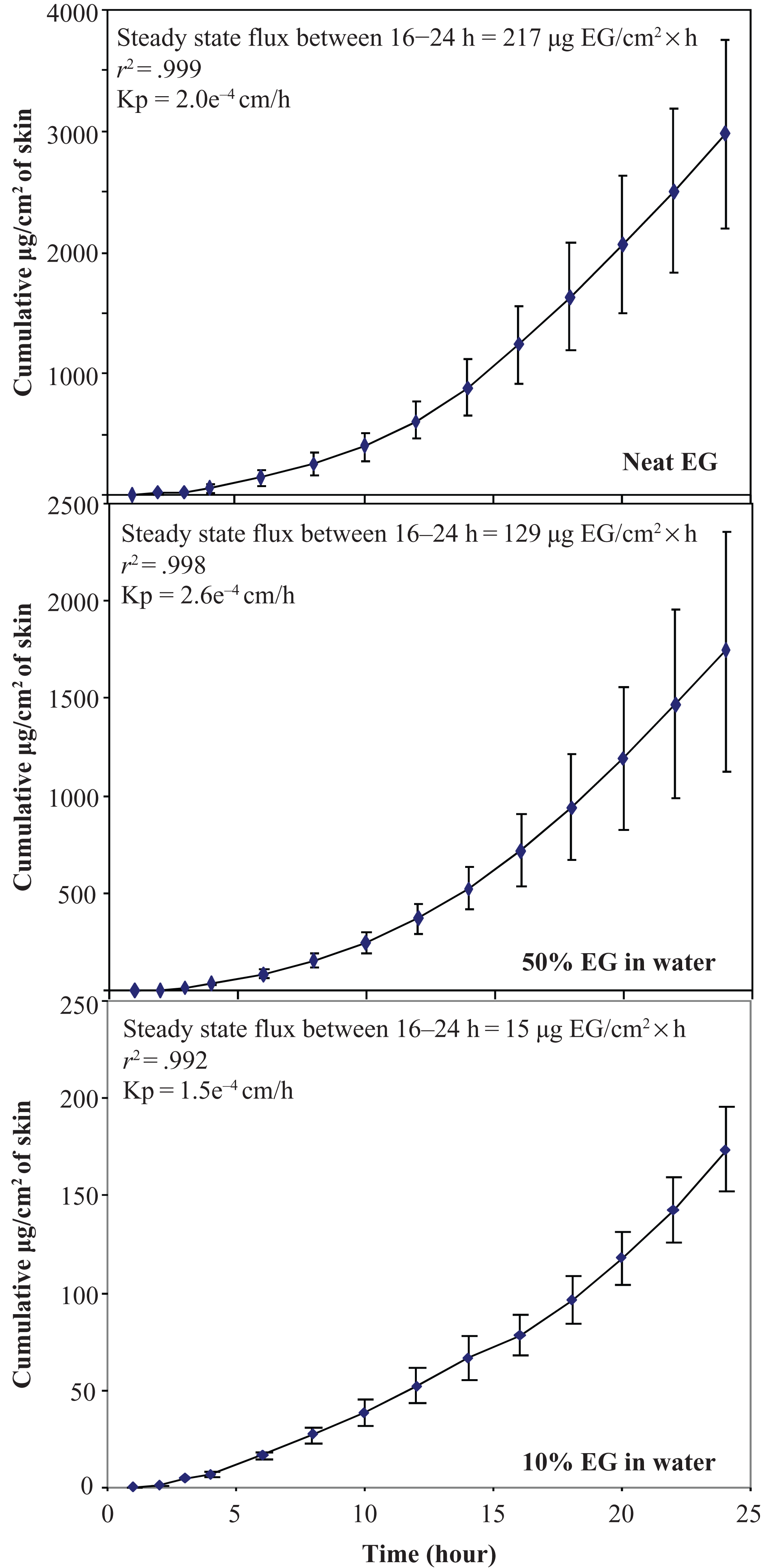
Time course of cumulative mean radioactivity values ± standard deviations (n = 8-11 samples from 3 to 4 donors) of in vitro penetration for 3 formulations of 14C-ethylene glycol (EG) through dermatomed human skin into flow-through receptor fluid.
The permeability coefficients (Kp) of EG were estimated from the linear portion of the dermal penetration curves (steady-state flux; Figure 2), under conditions of continuing infinite dose of the test material to the skin (92%-97% of test material remaining on skin surface after 24-hour exposure; see Table 2). The steady-state flux of EG from all 3 formulations was established from data collected between 16 and 24 hours of exposure, and proportionally corresponded to the concentration of EG in the formulations and to the net penetration of EG (Figure 1). The mean steady-state flux of EG through dermatomed skin was 217, 129, and 15 µg EG/cm2h for the neat, 50%, and 10% aqueous formulations, respectively (Figure 2). Because of the concentration-dependent influx of EG through the dermatomed skin after reaching steady-state rate of penetration, the total amount of each formulation penetrated remained fairly constant across all the time intervals. This resulted in very comparable estimates of Kp for all 3 formulations, ranging from 1.5 × 10−4 to 2.6 × 10−4 cm/h (Figure 2). The individual variability (determined as %CV) of the steady-state flux of EG among donors (between 16 and 24 hours after the application of the dose) was 30%, 44% and 16% percent for the neat, 50%, and 10% aqueous formulations, respectively (data not shown).
Discussion
Systemic toxicity of a chemical is best correlated with the actual internal concentration of the toxicant (parent and/or metabolite/metabolite[s]) as measured in the blood or plasma (systemically available dose), rather than the administered dose. 26 –28 Not all of an administered dose is necessarily systemically bioavailable, which is especially true for the cutaneous route of exposure where keratinized stratum corneum greatly limits penetration of a dermally administered dose of most chemicals. The internal dose resulting from dermal exposure to a chemical is the amount that penetrates the dermal barrier and becomes systemically available. Internal dose from the dermal route of exposure is most commonly determined as systemic plasma or blood levels but can often be predicted from the concentration-normalized permeability rates (or flux) experimentally determined under steady-state conditions. 29,30 This approach is based on Fick law relating constant flux (mg/cm2h) of a chemical across a membrane or skin to the difference in concentration between the inner and outer surfaces of the membrane. The dermal permeability coefficient (Kp) is a function of several factors, that is, path length of chemical diffusion, the membrane:vehicle partition coefficient of the chemical, and the diffusion coefficient of the chemical in the membrane.
As mentioned, EG has wide industrial and commercial use with potential for human exposure, especially from its use in water-based paints (normally <2% EG), automotive coolants (up to 50% EG, if EG based), and aircraft deicers (30%-70% EG, if EG based). The most common route of potential exposure to EG from these applications is through dermal contact. The previously available data on dermal penetration of EG through human skin under in vitro conditions are limited and of high variability. 17 –19 The reported rates of penetration for EG were 0.02 to 0.9 µg/cm2h, 18 118 µg/cm2h, 17 and 7 to 13 µg/cm2h. 19 Others have reported even lower permeability constant of EG (2.2 × 10−5 to 3.2 × 10−5 cm/h) following dermal application of 13C-labeled EG to the forearm of human volunteers, which would correspond with full-thickness skin and intact stratum corneum. 31 The objective of this study was to refine the dermal penetration information predicted for humans to assess realistic internal exposure in humans from dermal EG exposure to the most common formulation concentrations.
In the current data set, the steady-state flux of EG through the skin was fairly linear with increasing concentration (Figure 1), as expected from Fick law stating that the rate of chemical penetration through skin is directly related to its concentration. This resulted in fairly constant Kp values across formulations (Figure 2), as Kp is the measure of skin permeability of a chemical for a given species and is independent of chemical concentration. Confidence in the present data set is much higher than the earlier studies, 17 –19 in large part due to the following aspects: use of human skin; the reliable demonstration of good recoveries and mass balance (93%-99% of applied dose) of the 14C-EG-derived applied dose; GLP audited data set; and use and confirmation of an infinite dose (Table 2), which further validated the steady-state flux and 24-hour cumulative absorption. The mass balance of the applied dose was less reliable in some of the earlier studies, typically accounting for less than 90% of radiolabelled test material (Sun et al19 [76%-89% in humans and 75%-92% in mouse]; Frantz et al 32 [85%-100% in mouse and 42%-85% in rat]); whereas, Driver et al 18 used a finite dose of 8 µg EG/cm2.
Several studies have described dermal penetration of EG in rat and mouse using both in vivo and in vitro systems (eg, refs 19, 32). Frantz et al 32 have reported 26% to 60% absorption of 50% aqueous formulation of EG through the skin of rat and mouse over 96 hours (Figure 3A), under occluded conditions. Occluded exposure conditions are used to maximize dermal penetration by preventing volatilization and increasing hydration status and are considered “worst case” scenarios as described in the comprehensive review of the principles and application of dermal exposure. 29 The actual penetration of EG through human skin would be much lower than what has been reported in rodents by Frantz et al, 32 given they used the worst case exposure conditions with rodent skin, and human skin is significantly less permeable to chemicals than rodent skin. 16 Due to these factors, data from Frantz et al 32 should not be used to estimate dermal penetration of EG through human skin, especially with reliable data now available from human skin.
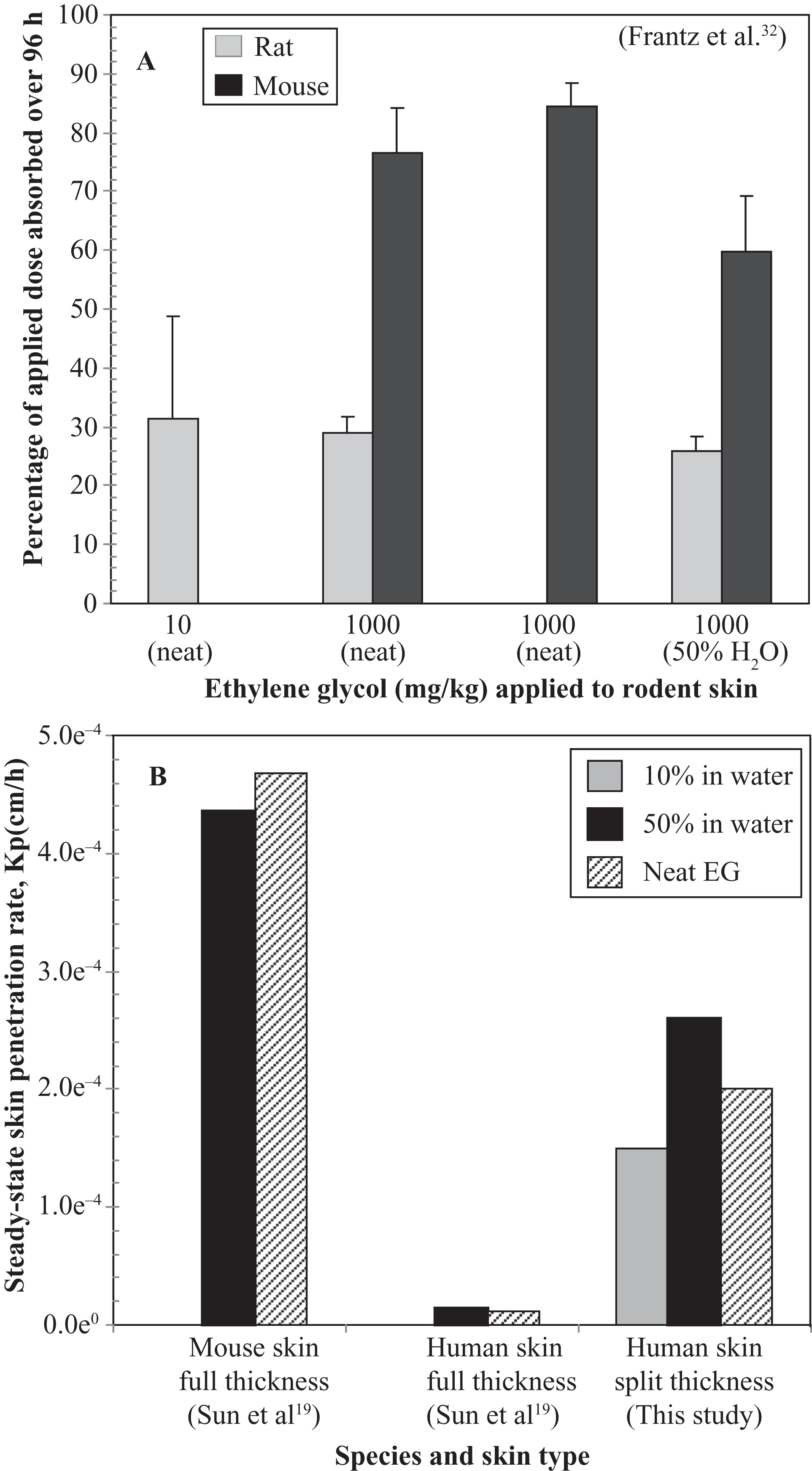
Comparison of dermal penetration data for ethylene glycol (EG) from previously published and current study results using in vivo (A) or in vitro (B) study designs.
As mentioned, the reported in vitro penetration rates for EG through human skin have been quite variable, with dermal penetration rates of 0.02 to 0.9 µg/cm2 h 18 , 118 µg/cm2h, 17 and 7 to 13 µg/cm2h. 19 The low penetration rate reported by Driver et al 18 was due to the use of a small finite dose, which apparently depleted from the application site during the exposure, resulting in calculation of a slow penetration rate. This explanation is further supported by the fact that a steady-state rate of penetration was never achieved in the Driver et al 18 study. The steady-state dermal penetration rate reported by Loden 17 was roughly comparable to that of the 50% aqueous formulation results from the current study (129 µg/cm2h). In contrast, Sun et al 19 reported 17- and 18-fold lower steady-state dermal penetration rates than were obtained in this study, for the neat and 50% aqueous formulations, respectively (Figure 3B). The low rates of penetration described by Sun et al 19 were probably due to the use of full-thickness skin rather than excised (dermatomed) skin, as used by Loden 17 and the current study.
A recent study of EG dermal penetration in human volunteers has been reported. Upadhyay et al 31 concluded that continuous wetting of both hands with neat liquid EG for 8 hours would result in only slight increases in plasma and urinary levels of EG and its metabolites, when compared to the endogenous background levels. Their conclusions were based on a mean permeability coefficient of 2.1 × 10−5 cm/h in human volunteers, which was about 10-fold lower than that found in the current study, and was similar to the reported Kp in excised full-thickness cadaver skin of Sun et al 19 As a result, the Kp values obtained from dermatomed skin samples in the current study likely provide a conservative, worst case assessment of dermal penetration of EG through human skin, at formulations of 10% to 100%.
The data from the current in vitro study can be used to estimate absorption of EG in humans following a variety of dermal exposure scenarios, with specified exposure period and surface area of skin involved. For example, a worst case (neither practical nor recommended) scenario of continuous dermal exposure of 10 µL formulation/cm2 of latex paint 33 containing ~2% EG to both hands and forearms of a person (area = 1980 cm2) 34 would lead to a total dermal exposure dose of ~0.4 g EG. At a dermal penetration rate of 3 µg EG/cm2h (extrapolated to one fifth the steady-state flux of 15 µg EG/cm2 h of the 10% aqueous formulation from the current study, as the flux rates were fairly linear across concentrations; Figure 2), the total penetration during 8 hours continuous exposure would be ~24 µg EG/cm2. Extrapolation to the total exposure area of both hands and forearms combined would yield a total systemic dose of ~48 mg EG, 11% of the total dermal exposure. This is more than 2300-fold lower than the estimated minimum lethal dose to human after bolus oral ingestion (~100 mL). 35 In the case of exposure to higher volumes of the same formulation of a product containing EG, the total systemic dose for a given exposure period would remain same, as the available surface area of the skin to come in contact with the product would not change. Accidental exposure of the whole body to neat EG for shorter period of time (minutes to hour) would yield lower total systemic dose than that determined for the scenario described above.
The above estimate should serve as a short-term exposure guidance for the dermal penetration of EG at workplace and in accidental exposure scenarios. The dermal penetration of water and simple polar nonelectrolytes at steady state has been reported to be in the order of palm > back of the hand (~2-fold less permeable than palm) > forearm ≈ abdomen (1.6- and 1.8-fold less permeable than the back of the hand, respectively). 36 Regional variation in permeability is due to the pattern of arrangement of cells and the amount of lipid; however, differences in thickness and diffusivity of the stratum corneum compensate for each other to provide the skin with relatively uniform steady-state permeability. 36 Therefore, the overall penetration of EG from both hands and forearms of exposed workers would be similar to that estimated using the rate of penetration obtained from abdominal skin. These calculations used the surface area of both palms and the back of both hands as equivalent to 150 cm2 and 108 cm2, respectively, as reported by Liao et al. 37
On the basis of the results of this study, it is concluded that exposure of EG (neat or formulations) to intact skin, either through dermal routes that may occur during normal working hours or by accidental exposure of the whole body for a short duration, will not elevate the systemic levels of toxic metabolites (glycolate and/or oxalate). In all cases, the systemic levels will remain around the endogenous background levels. This is not only due to the slow and low dermal penetration of EG but also to its continuous elimination via metabolism and excretion following absorption. 31 Likely these kinetic explanations account for the lack of any developmental toxicity in mice after dermal exposure, even though total penetration of the dermally applied dose is much higher in rodents than humans (Figures 3A and B). Overall, these findings demonstrate that EG dermal penetration in humans is expected to be very low and to be slow, indicating very limited systemic or internal dose of EG due to dermal exposure.
Footnotes
Acknowledgment
Authors acknowledge Marina Jovanovic and her team at Dow Corning, Midland, for conducting this study. The study was partly funded by the American Chemistry Council.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.
