Abstract
The current study was designed to investigate possible protective effect of allyl isothiocyanate (AITC) in nitrite- and nitrosamine-treated human hepatoma cells (HepG2) with the evaluation by cytotoxic effects and genotoxic effects determined by the single-cell gel electrophoresis (SCGE). Allyl isothiocyanate treatment enhanced cell viability and reduced intracellular reactive oxygen species (ROS) production in both nitrite- and nitrosamine-treated cells significantly. In SCGE, when compared to untreated control cells, all of the treated groups caused increases in the tail intensity (%) such as nitrite at 17%, N-nitrosodimethylamine (NDMA) at 279%, N-nitrosodiethylamine (NDEA) at 324%, and N-nitrosomorpholine (NMOR) at 288%. Allyl isothiocyanate reduced the tail intensity caused by nitrite 36%, by NDMA 36%, by NDEA 49%, and by NMOR 32%, respectively, when compared to each individual toxic compound–treated group. In conclusion, AITC protected HepG2 cells against cytotoxic and genotoxic effects caused by nitrite and the nitrosamines.
Introduction
Diet may contain many cancer-causing substances as well as many cancer-preventive agents and is the major source for N-nitrosamines and their precursors nitrates and nitrites. Nitrites and nitrosamines are a large group of substances that are thought to be potent carcinogens and mutagens. 1 –5 Other than diet, human exposure to preformed N-nitrosamines occurs in certain occupational settings and through the use of tobacco products, cosmetics, pharmaceutical products, and agricultural chemicals. 6 –11
Nitrite exposure is suspected to be the cause of several cancer types including childhood brain cancers. 12,13 However, at least 30 animal species are responsive to the carcinogenic effect of N-nitrosamines. 14 Several studies showed that N-nitrosodimethylamine (NDMA) produced tumors in experimental animals through different exposure routes and might cause tumors of bile duct, kidney, lung, and nasal cavity. 15 N-nitrosodiethylamine (NDEA) was shown to induce liver tumors in mouse and N-nitrosobenzylmethylamine (NBMA)-induced esophageal tumors in rat. 16,17 N-nitrosamines require metabolic activation to exert their carcinogenic effects. The key metabolic activation pathway is through the cytochrome P450 (CYP450) enzymes. 18
Three important nitrosamines, namely NDMA, NDEA, and N-nitrosomorpholine (NMOR), are classified as probably carcinogenic to humans (group 2B) by International Agency for Research on Cancer (IARC). 19 The increasing evidence that nitrosamines are potential human carcinogens encouraged researchers to study the protective dietary factors in chemical carcinogenesis. 18 Vegetables and fruits are very rich in dietary agents that retard or prevent carcinogenesis. Some of these agents are vitamin C, vitamin E, vitamin A, and naturally occurring (or synthetic) isothiocyanates (ITCs). 4,20 –23
There is powerful evidence in literature for a cancer-protective effect of the vegetables of the family Cruciferae that includes broccoli, watercress, cabbage, kale, horseradish, radish, turnip, and garden cress. 24,25 This effect is attributed to ITCs, which occur naturally as thioglucoside conjugates (glucosinolates). They are the hydrolysis products of glucosinolates and are generated through catalytic mediation of myrosinase, which is released upon processing (cutting or chewing) of cruciferous vegetables from a compartment separated from glucosinolates. Evidence exists for conversion of glucosinolates to ITCs in the gut. At least 120 different glucosinolates have been identified. Isothiocyanates have a common basic skeleton but differ in their terminal R group, which can be an alkyl, an alkenyl, an alkylthioalkyl, an aryl, a β-hydroxyalkyl, or an indolylmethyl group. The widely studied ITCs include phenylethyl isothiocyanate (PEITC), benzyl isothiocyanate (BITC), indole-3-carbinol (I3C), and allyl isothiocyanate (AITC). 26,27
The most important biological property discovered about ITCs is their ability to inhibit carcinogenesis, induced by several chemicals including nitrosamines in the lung, stomach, colon, liver, esophagus, bladder, and mammary glands in animal models. 25,27 –31 The underlying mechanism is the ability of ITCs to inhibit specific CYP450 enzymes involved in the activation of carcinogens, including nitrosamines. 32 Several recent epidemiological studies have suggested that humans who consumed higher levels of ITCs might be less likely to develop lung and colon cancers. 33 –35
Different cell types have been used for assessing genotoxicity/antigenotoxicity of some natural compounds. 36 Because many carcinogens require metabolic activation to react with DNA, the use of cells that have endogenous biotransforming activity can reduce some of the problems associated with the use of exogenous activation mixtures such as S9 mix. 37 Human hepatoma (HepG2) cell line contains several enzymes responsible for the activation of various xenobiotics and appears to be an alternative for assessing genotoxicity or antigenotoxicity. 38 –40
One of the mechanisms underlying the toxicity of nitrite and nitrosamines might be through their oxidative stress-causing effect. We have shown the oxidant effects of the nitrite and nitrosamines in a previous study and their DNA-damaging effect through such mechanism. Besides, we have determined the protective effect of ascorbic acid against cytotoxicity and genotoxicity induced by nitrite and nitrosamines. 41
The main goal of this study is to provide information for the possible protection by AITC against cytotoxicity and oxidative stress caused by nitrite and nitrosamines and whether genotoxicity of these compounds may also be prevented by this particular ITC in HepG2 cells.
Material and Methods
Chemicals
All chemicals used in the study, including single-cell gel electrophoresis (SCGE, Comet assay) chemicals and nitrite (Chemical Abstracts Service [CAS] number: 7632-00-0), NDMA (CAS number: 62-75-9), NDEA (CAS number: 55-18-5), NMOR (CAS number: 59-89-2), and AITC (CAS number: 57-06-7) were purchased from Sigma-Aldrich (St Louis, Missouri). All chemicals and solvents were of the highest grade available. 5-(and 6-)Chloromethyl-2′,7′-dichlorodihydrofluorescein diacetate (CM-H2DCFA) was purchased from (Invitrogen Molecular Probes, Eugene, OR, USA).
Preparation of AITC, Nitrite, and Nitrosamines
Allyl isothiocyanate stock solution (0.5 mmol/L) was prepared in sterile dimethyl sulfoxide (DMSO; 0.1%) and was divided into portions. Aliquots were kept at −80°C at dark. The aliquots were dissolved immediately before use and diluted to 0.5 µmol/L with sterile deionized water.
Stock nitrite solution (2 mmol/L) was prepared in sterile deionized water and was divided into portions. Aliquots were kept at −80°C at dark. The aliquots were dissolved immediately before use and diluted to 20 µmol/L with sterile deionized water. N-nitrosamines were dissolved in sterile DMSO (0.1%). N-nitrosodimethylamine, NDEA, and NMOR stocks were prepared at 100, 100, and 30 mmol/L, respectively. The stock solutions were stored in deep freezer (−80°C).
Cell Treatment
The human hepatoma line (HepG2) was purchased from American Type Culture Collection (ATCC, HB-8065). The cells were grown as monolayer cultures in Dulbecco’s modified Eagle medium (DMEM) supplemented with 15% fetal calf serum (FCS) and antibiotics (1% penicillin/streptomycin) in culture flasks. Cells were incubated at 37°C in a humidified atmosphere containing 5% CO2. Confluent cells were trypsinized for 10 minutes and washed with sterile phosphate-buffered saline (PBS) and centrifuged at 1500g for 5 minutes at 20°C. Cells only with 15 to 17 passages were used during the experiments. Cell suspensions of 3000 cell/Petri dish were used for SCGE assay.
Cell Viability Assays
HepG2 cells were trypsinized, stained with the trypan blue, and the number of viable (uncolored) and dead (colored) cells was counted by using light microscope and Countess Cell Counter (Invitrogen) to double check. The ratio of number of viable cells to all cells gives the percentage of viable cells.
3-(4,5-Dimethyl-thiazol-2-yl)-2,5-diphenyl-tetrazolium bromide (MTT) assay was used to detect cell viability. HepG2 cells of each group in 200 µL of medium with or without AITC (0.5 µmol/L) in the presence of nitrite and nitrosamines (NDMA, NDEA, and NMOR) were seeded in 8 wells of the 96-well culture plates. After 24 hours of culture, growth rates were detected as described before. 41 After cell viability assays, treatment doses of nitrite and nitrosamine were chosen according to the 50% cell viability (IC50) value and the cells were grouped as follows:
Group 1: untreated control cells (C)
Group 2: AITC-treated cells (AITC): cells were treated with AITC (0.5 µmol/L) for 24 hours.
Group 3: nitrite-treated cells (nitrite): cells were treated with nitrite (20 µmol/L) for 30 minutes.
Group 4: nitrite- and AITC-treated cells (nitrite + AITC): cells were treated with AITC (0.5 µmol/L) for 24 hours and then treated with nitrite (20 µmol/L) for 30 minutes in the presence of AITC.
Group 5: NDMA-treated cells (NDMA): cells were treated with NDMA (10 mmol/L) for 30 minutes.
Group 6: NDMA- and AITC-treated cells (NDMA + AITC): cells were treated with AITC (10 µmol/L) for 24 hours and then treated with NDMA (10 mmol/L) for 30 minutes in the presence of AITC (0.5 µmol/L).
Group 7: NDEA-treated cells (NDEA): Cells were treated with NDEA (10 mmol/L) for 30 minutes.
Group 8: NDEA- and AITC-treated cells (NDEA + AITC): cells were treated with AITC (0.5 µmol/L) for 24 hours and then treated with NDEA (10 mmol/L) for 30 minutes in the presence of AITC (0.5 µmol/L).
Group 9: NMOR-treated cells (NMOR): cells were treated with NMOR (3 mmol/L) for 30 minutes.
Group 10: NMOR- and AITC-treated cells (NMOR + AITC): cells were treated with AITC (0.5 µmol/L) for 24 hours and then treated with NMOR (3 mmol/L) for 30 minutes in the presence of AITC (0.5 µmol/L).
Detection of Cell Death
Cell death was assessed by measuring the activity of lactate dehydrogenase (LDH) released into the culture medium. Cells (1.5 × 105) were treated as described before and were seeded into 24-well plates. The decrease in absorbance of nicotinamide-adenine dinucleotide hydrogenase at 340 nm was measured using a spectrophotometer. Lactate dehydrogenase activity was normalized to control values (0% cell death).
Measurement of Reactive Oxygen Species Production
HepG2 cells (70%-80% confluent) were plated in 24-well multiwells at a number of 2 × 105 cells per well. The cells were seeded at 96-well plate with or without AITC as identified above. The generation of reactive oxygen species (ROS) was evaluated by fluorometric assay using intracellular oxidation of 2′,7′-dichlorofluorescin (DCFH) as described before. 42
Testing Genotoxicity by SCGE (Comet Assay)
HepG2 cells (10 000 cells/Petri dish) were treated with or without AITC (0.5 µmol/L) in the presence of nitrite and nitrosamines for 24 hours. No cytotoxicity has been found in AITC concentrations between 0.1 and 1.0 µmol/L. 0.5 µmol/L was chosen as 1 µmol/L did not supply more protection than 0.5 µmol/L in cytotoxicity assays. Treatment doses of sodium nitrite and nitrosamines were chosen in consideration to the IC50. Nitrite was added as 20 µmol/L, NDMA as 10 mmol/L, NDEA as 10 mmol/L, and NMOR as 3 mmol/L to the medium.
The SCGE was performed according to Hininger et al, with minor modifications. 43 For quantification, a fluorescence that is connected to a charge-coupled device (CDC) was used. A computer-based analysis system (Comet IV) was used to determine the extent of DNA damage after electrophoresis migration of DNA fragments in the agarose gel. For each condition, 50 randomly selected comets on each slide were scored and tail intensity (%; length of the tail) and the tail moment were determined using 3 slides prepared as described above.
Statistical Analysis
All of the results were expressed as mean ± standard error of mean (SEM). The differences among the groups were evaluated with Kruskal-Wallis 1-way analysis of variance, followed by Mann-Whitney U test. P values <.05 were considered statistically significant.
Results
Cell Viability Assays
Results of MTT assay for nitrite and nitrosamines are shown in Figure 1 . The IC50 values for nitrite, NDMA, NDEA, and NMOR were found to be 20 µmol/L, 10 mmol/L, 10 mmol/L, and 3 mmol/L, respectively. For AITC, 0.5 µmol/L supplementation was chosen as it was the modest dose that protected the cells from nitrite- and nitrosamine-induced cytotoxicity. Further supplementation (upto 1 µmol/L) did not supply further protection.
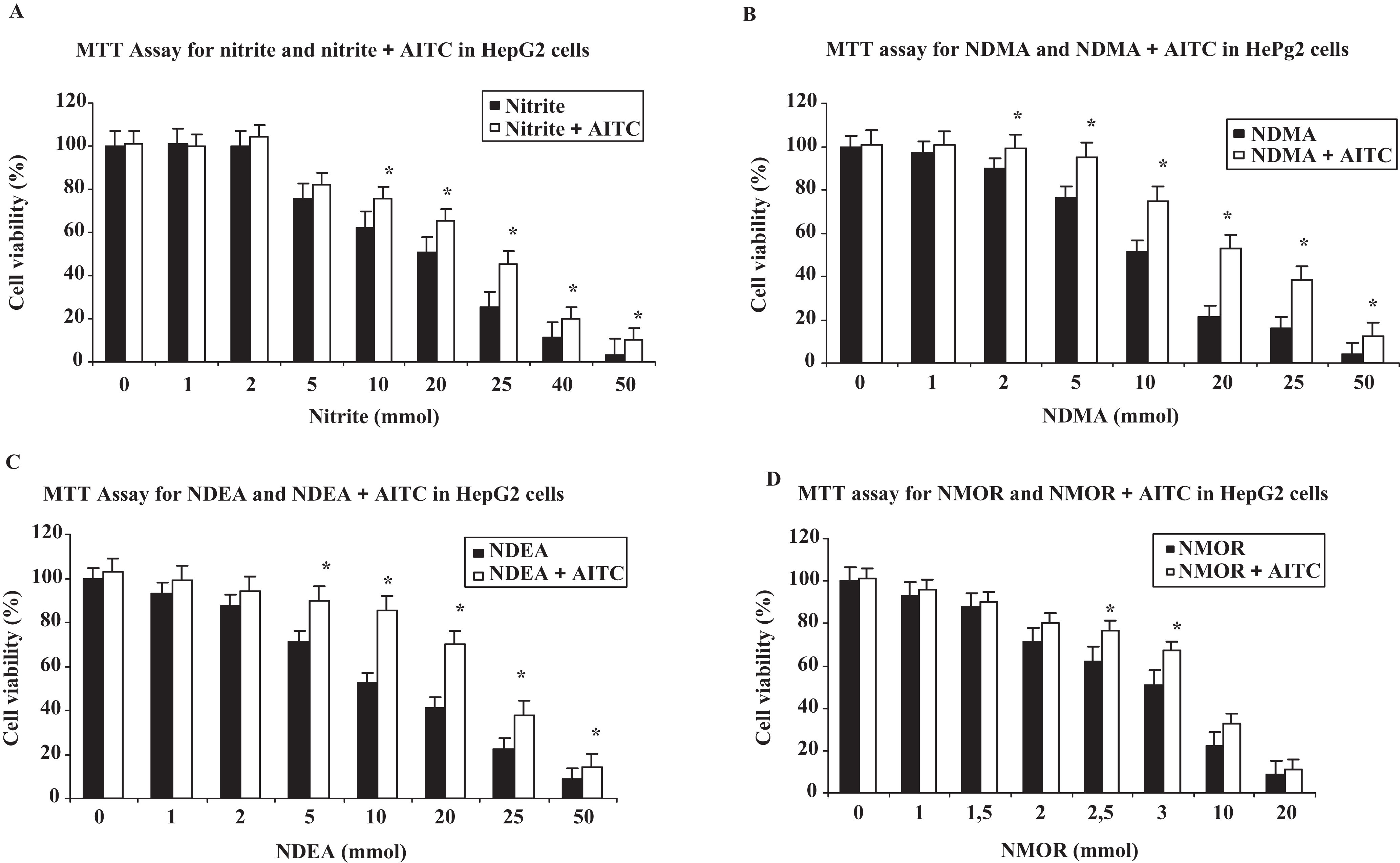
Effects of nitrite at various micromolar concentrations and nitrosamines at various millimolar concentrations on cell viability and evaluation of prevention of cell death by allyl isothiocyanate (AITC) at 0.5 µmol/L by using 3-(4,5-dimethyl-thiazol-2-yl)-2,5-diphenyl-tetrazolium bromide (MTT) assay. *indicates that AITC treatment in combination with nitrite or nitrosamines protects significantly from cell death when compared to each individual cytotoxic compound (P < .05); AITC (0.5 µmol/L), nitrite (20 µmol/L), N-nitrosodimethylamine (NDMA, 10 mmol/L), N-nitrosodiethylamine (NDEA, 10 mmol/L), and N-nitrosomorpholine (NMOR, 3 mmol/L).
In MTT assay, AITC was found to be protective against nitrite- and nitrosamine-induced cytotoxicity. Allyl isothiocyanate supplementation in the HepG2 cells treated with nitrite, NDMA, NDEA, or NMOR enhances cell viabilities and this protection was found to be statistically significant at IC50 value in nitrite- and nitrosamine-treated cells (all, P < .05).
Detection of Cell Death
The LDH leakage was estimated from the ratio between the LDH activity in the culture medium and that of the whole cell content. As seen in Table 1 , nitrite and nitrosamine treatment caused increases in the proportion of total LDH activity present in the culture medium. Allyl isothiocyanate treatment with nitrite and nitrosamine in HepG2 cells greatly decreased LDH activity when compared to each individual nitrite and nitrosamine treatment.
Effects of Nitrite and Nitrosamines on Cell Death and Evaluation of Protection of Cell Death by AITC Using LDH Levels in the Medium (%) After 24 Hours and 24 Hours + 30 Minutes a
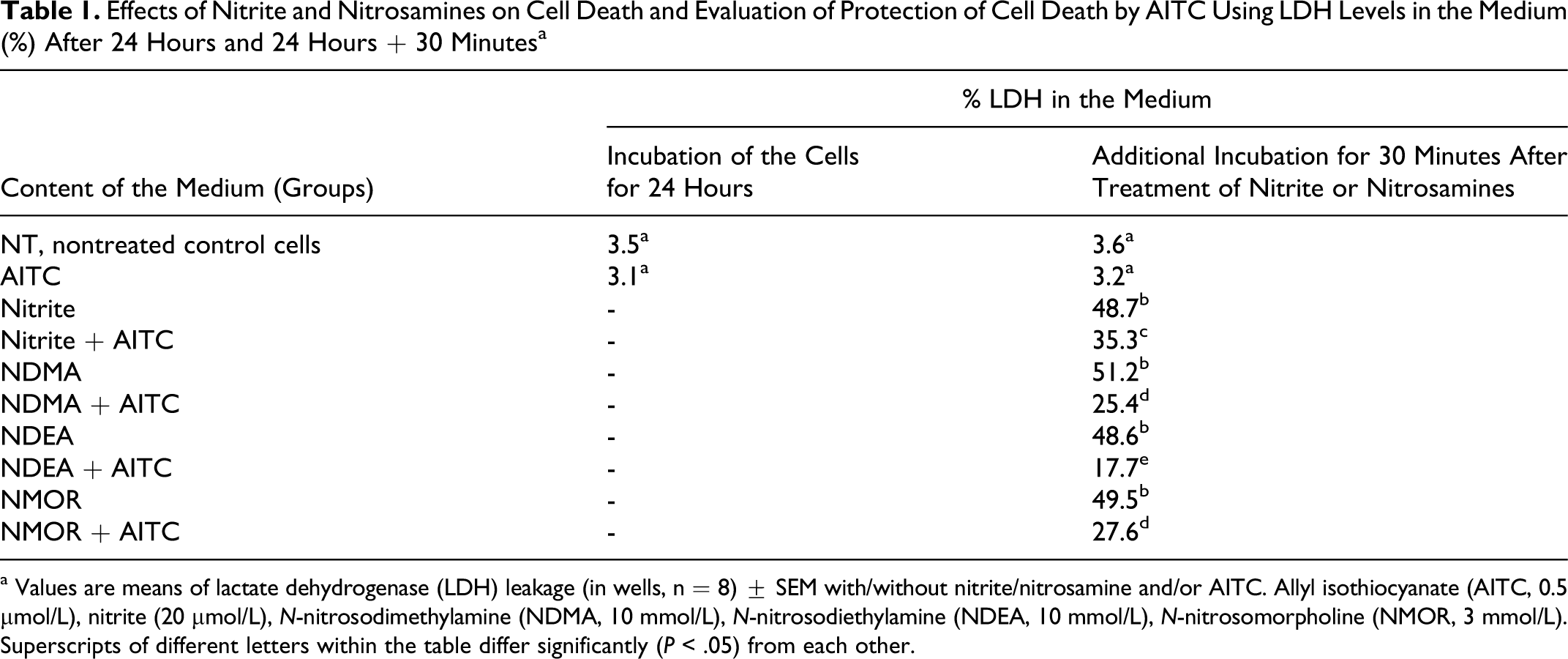
a Values are means of lactate dehydrogenase (LDH) leakage (in wells, n = 8) ± SEM with/without nitrite/nitrosamine and/or AITC. Allyl isothiocyanate (AITC, 0.5 µmol/L), nitrite (20 µmol/L), N-nitrosodimethylamine (NDMA, 10 mmol/L), N-nitrosodiethylamine (NDEA, 10 mmol/L), N-nitrosomorpholine (NMOR, 3 mmol/L). Superscripts of different letters within the table differ significantly (P < .05) from each other.
Reactive Oxygen Species Production
In this study, we used DCFH to measure intracellular generation of ROS. 2′,7′-Dichlorofluorescin is rapidly oxidized in the presence of ROS to a highly fluorescent 2′,7′-dichlorofluorescein (DCF). The effects of nitrite and nitrosamines on ROS production and evaluation of protection by AITC at 30, 60, and 90 minutes are shown in Figure 2 . The DCF fluorescence intensity of nitrite increased ~1-fold at 30 minutes, ~2-fold at 60 minutes, and ~3-fold at 90 minutes. For nitrosamine-treated cells, the same pattern in ROS increase was also observed. Reactive oxygen species production was found to be significantly lower at 30, 60, and 90 minutes in AITC-treated cells (all, P < .05).
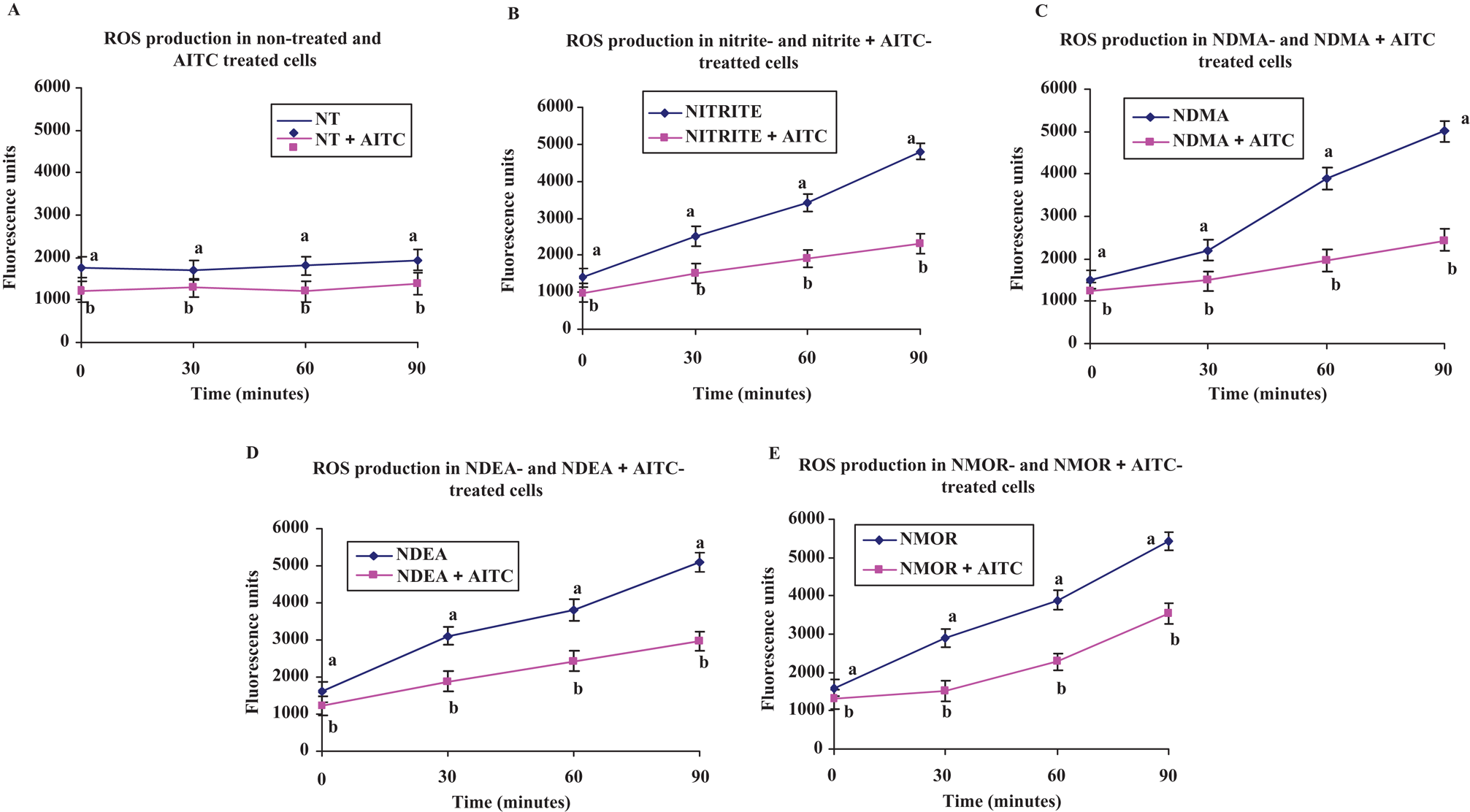
Reactive oxygen species (ROS) formation measured by the oxidation of 2′,7′-dichlorofluorescin (DCFH) to 2′,7′-dichlorofluorescein (DCF) in HepG2 cells incubated with or without nitrite and nitrosamines and protection by allyl isothiocyanate (AITC) at 30, 60, and 90 minutes. Values are mean fluorescent units of wells (n = 8) ± SEM with/without nitrite/nitrosamine and/or AITC calculated from 3 independent experiments. Superscripts of different letters differ significantly (P < .05) from each other. NT, nontreated control cells, allyl isothiocyanate (AITC, 0.5 mol/L), nitrite (20 mol/L), N-nitrosodimethylamine (NDMA, 10 mmol/L), N-nitrosodiethylamine (NDEA, 10 mmol/L), and N-nitrosomorpholine (NMOR, 3 mmol/L).
Genotoxicity Assay
The comet assay results are given in Figures 3 and 4 . When compared to untreated cells, nitrite, NDMA, NDEA, and NMOR raised the tail intensity (%) up to 17%, 279%, 324%, and 288%, respectively (all, P < .05). Allyl isothiocyanate was able to reduce the tail intensity caused by nitrite 36%, by NDMA 36%, by NDEA 49%, and by NMOR 32%, respectively. These reductions were statistically significant when compared to each individual toxic compound applied group (all, P < .05). Besides, when compared to untreated cells, nitrite, NDMA, NDEA, and NMOR raised the tail intensity up to 94%, 126%, 157%, and 207%, respectively (all, P < .05). Allyl isothiocyanate was able to reduce the tail moment caused by nitrite 16%, by NDMA 32%, by NDEA 41%, and by NMOR 19%, respectively, and these reductions were statistically significant when compared to each individual toxic compound applied group (all, P < .05).
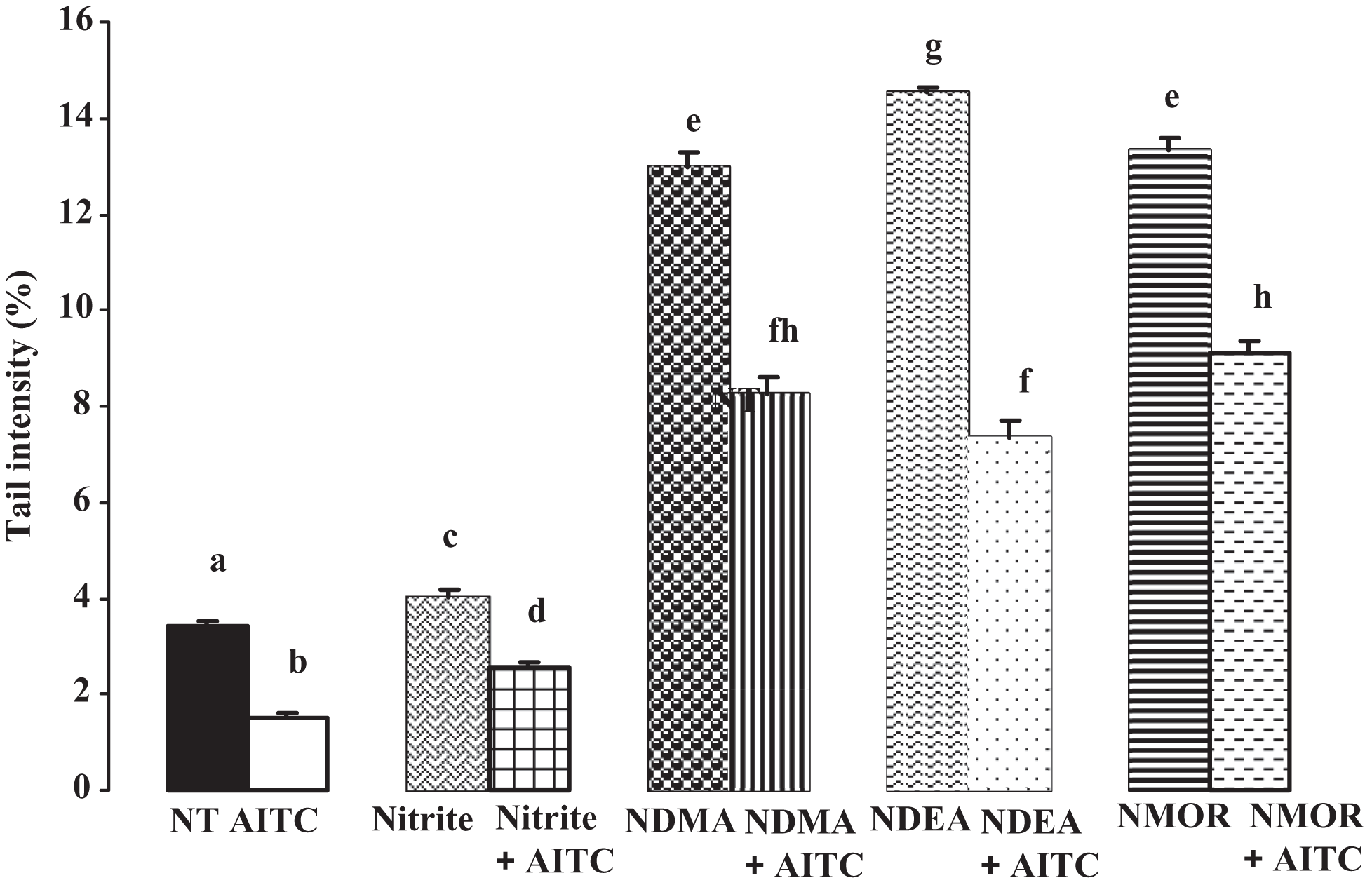
Evaluation of tail intensities obtained from comet assay by nitrite and nitrosamines and protection by allyl isothiocyanate (AITC). Superscripts of different letters differ significantly (p < 0.05) from each other. NT, nontreated control cells, allyl isothiocyanate (AITC, 0.5 µmol/L), nitrite (20 µmol/L), N-nitrosodimethylamine (NDMA 10 mmol/L), N-nitrosodiethylamine (NDEA, 10 mmol/L), and N-nitrosomorpholine (NMOR, 3 mmol/L).
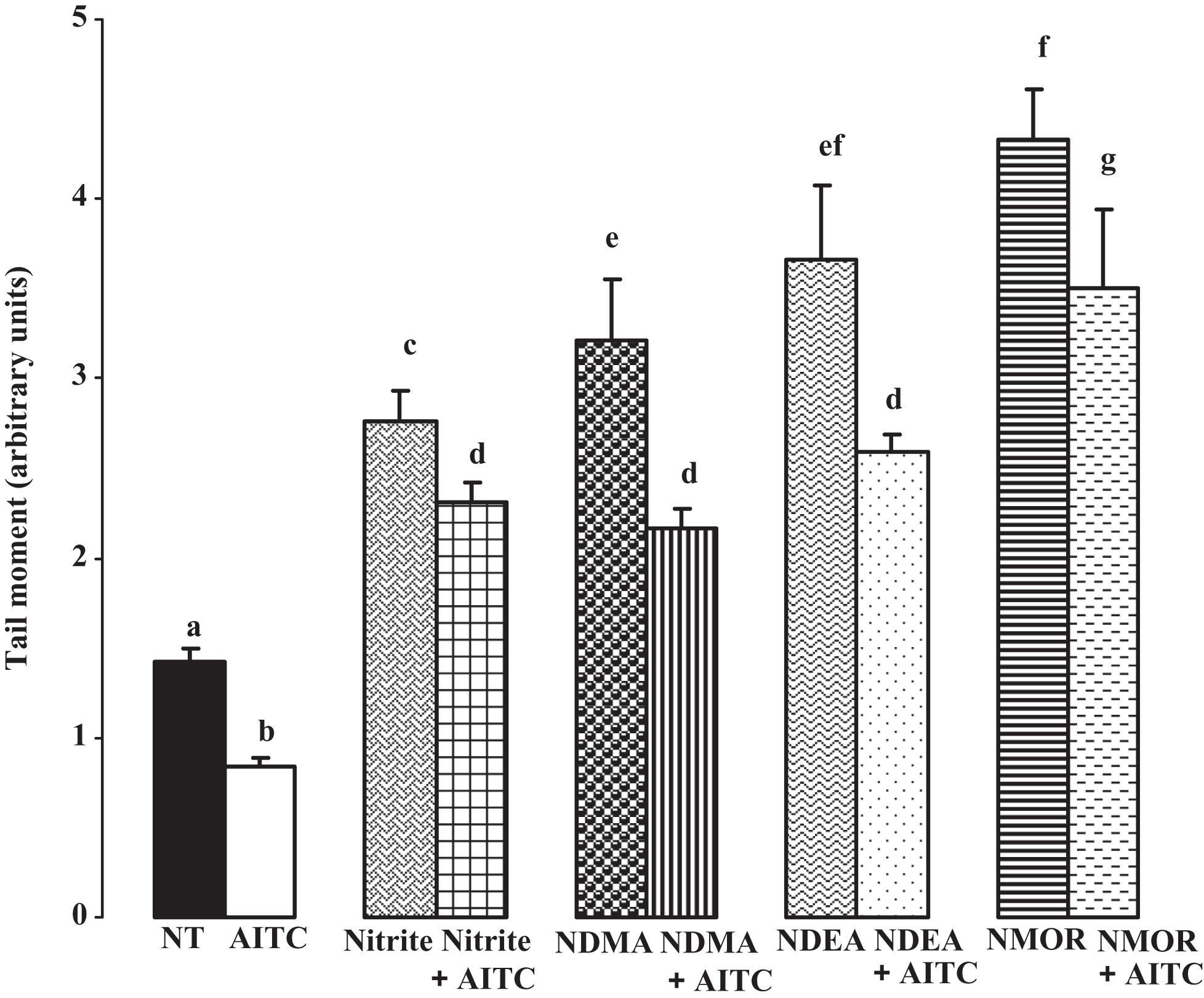
Evaluation of tail moments obtained from comet assay by nitrite and nitrosamines and protection by allyl isothiocyanate (AITC). Superscripts of different letters differ significantly (P < .05) from each other. NT, nontreated control cells, allyl isothiocyanate (AITC, 0.5 µmol/L), nitrite (20 µmol/L), N-nitrosodimethylamine (NDMA, 10 mmol/L), N-nitrosodiethylamine (NDEA, 10 mmol/L), and N-nitrosomorpholine (NMOR, 3 mmol/L).
Dimethyl sulfoxide (0.1%) was also tested for DNA damage by Comet assay; however, it did not induce any damage; tail intensity (%) was 3.45 and tail moment was 1.40.
Discussion
Superoxide anion, hydroxyl radicals and hydrogen peroxide (H2O2) are generated as byproducts of the normal metabolism of oxygen and can be formed by several environmental chemicals. They are named as ROS. These species are capable of reacting with and damaging DNA, proteins, and lipids. They also serve as very important signaling molecules regulating numerous physiological and pathological pathways. In particular, there is growing evidence suggesting that ROS plays a key role in tumorigenesis. Reactive oxygen species causes DNA damage, promotes cell proliferation and survival, and activates several tumor-promoting signaling pathways. 44 Nitrosamines, one of the most significant environmental carcinogens, have been suggested to cause the generation of ROS resulting in oxidative stress and cellular injury. 45,46 N-nitrosopiperidine (NPIP) and NDBA were shown to induce apoptosis through their oxidative stress-producing effect. 46 However, metabolic activation of nitrosamines is through CYP450-mediated pathways and each CYP form shows substrate specificity for activating promutagens to their mutagenic intermediates. 47
Carcinogenesis is a multistep process, involving a series of epigenetic and genetic alterations. The long and complex process begins with genomic instability and ends with the development of cancer. This complexity gives opportunity for the development of interventions in preventing the initiation, promotion, and progression. Failure and high systemic toxicity of conventional cancer therapies have forced the researchers for newer agents, which could prevent and/or slow down cancer growth and have more human acceptability by being less or nontoxic. 48 There is a strong interest in isolating and characterizing the nutritive and nonnutritive components of fruits and vegetables as potential chemopreventive agents. The most important biological property discovered about various phytochemicals including ITCs is their ability to inhibit chemical carcinogenesis. Several epidemiological studies also indicated that the dietary consumption of ITCs or ITC-containing foods inversely correlates with the risk of developing lung, breast, and colon cancers, providing evidence that they have a potential to prevent cancer in humans. 49 Two mechanisms can be suggested for the protective effect of ITCs against nitrosamine-induced DNA damage: either blocking the production of genotoxic intermediates by inhibiting phase I enzymes (ie, CYP2E1, CYP1A1, CYP2A6) activities and/or enhancement of detoxification pathways through the induction of phase II enzymes. 27,50
The aim of our study was to evaluate the protective effect of AITC in nitrite- and nitrosamine-induced cell death, ROS production, and oxidative DNA damage. Allyl isothiocyanate showed a strong inhibition against cell death and oxidative DNA damage and increased cell viability. One feasible mechanism by which AITC exerts their protective effects toward N-nitrosamine-induced oxidative DNA damage could be by the inhibition of CYP450-dependent bioactivation. There are studies demonstrating the protective effect of ITCs against DNA damage induced by other chemicals. A study performed by Kassie et al determined that PEITC exerted a protection against benzo(a)pyrene-induced DNA damage in HepG2 cells using SCGE assay. 50 However, some studies concluded that the vegetable sprout (eg, Brussels sprout) or juice (garden and water cress) itself was protective against benzo(a)pyrene though this protection could not be attributed to the ITC content as ITCs themselves were also genotoxic at high concentrations. 52,53
The antimutagenic properties of ITCs have been reported toward NDMA and N-nitrosopyyrolidine (NPYR)-induced oxidative stress before. In studies performed by Knasmüller et al 53,54 using PEITC as a chemopreventive agent, they observed a reduction in NDMA- and NPYR-induced DNA damage in Escherichia coli K-12 and a considerable reduction in NDMA-induced micronuclei in HepG2 cells. The results of other studies demonstrated that ITCs exhibited strong antimutagenic effects against NDMA and NPYR in a dose-dependent manner. The protection was 67% against NPYR and 60% against NDMA-induced DNA oxidation by PEITC at 0.1 µmol/L and further supplementation did not enable more protection in the presence and absence of formamidopyrimidine DNA glycosylase (FpG). 27 Another study performed by Garcia et al showed that at 0.1 µmol/L concentration, PEITC exerted a weak protective effect of 19% to 26% toward NPIP-induced oxidative damage. 48 We observed a protection of 36%, 36%, and 55% against nitrite, NDMA-, NDEA-, and NMOR-induced tail intensity in SCGE, respectively, at 0.5 µmol/L of AITC. Our results are in accordance with the study of Arranz et al. Combined treatments of HepG2 cells with PEITC (1 µmol/L), AITC (1 µmol/L), or I3C (1 µmol/L) and NPYR or NDMA reduced the genotoxic effects of the nitrosamines in a dose-dependent manner. The protection was stronger toward NDMA and nitrite than NDEA. The results demonstrated that protected effect of ITCs in human-derived cells against the DNA damaging effect of NPYR and NDMA, 2 carcinogenic compounds which occur in the environment. 27 The results demonstrated the protective effect of ITCs on human-derived cells against the DNA damaging effect of NPYR and NDMA, two carcinogenic compounds which occur in the environment. Moreover, it is an interesting finding that nitrite-induced DNA damage can also prevented by AITC.
To summarize, modest antioxidant supplementation might help prevent chemical-induced carcinogenesis in healthy individuals. Plant foods including several vitamins and ITCs appear to exert a general risk-lowering effect against cancer initiators and promoters. Although genetic susceptibility may determine the variations in the site-specific risks of cancer seen across populations, modest DNA-damaging effects of several environmental chemicals could be functionally rescued by dietary antioxidants. The future interest of our studies is to understand the pathways that are affected by antioxidants, particularly ITCs, in protection against nitrite- and nitrosamine-induced toxicity and the outcomes of this protection in other cell types.
Footnotes
Acknowledgement
The authors would like to thank Dr. Walid Rachidi (CEA/Grenoble/France), for instructing the methods used in the study and for his generous assistance of knowledge.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The authors received no financial support for the research and/or authorship of this article.
