Abstract
Embryonic stem (ES) cells are considered an important alternative to develop in vitro screening methods for embryotoxicity. Mouse ES cells can be cultured as cell suspension aggregates termed “embryoid bodies” (EBs) in which cells start to differentiate. We have studied the expression of several genes in the presence of a wide range of concentrations of 5-fluorouracil (5-FU). This well-established embryotoxic compound completely inhibited cell viability at 200 nmol/L in monolayer cultures. At lower concentrations, 5-FU led to decrease in the expression of the α-fetoprotein gene, a marker of the visceral endoderm, in the EBs. However, the expression of several mesodermal gene markers was not significantly affected at these concentrations. These results suggest a high sensitivity of the visceral endoderm differentiation to 5-FU. Therefore, the quantification of the α-fetoprotein gene after exposure to potential embryotoxicants should be considered an additional end point in future embryotoxicity assays in vitro with ES cells.
Keywords
Introduction
Different approaches to study the possible use of differentiating mouse embryonic stem (ES) cells in vitro to evaluate the potential embryotoxic effect of different chemicals are underway. 1 Such chemicals may hinder in vitro differentiation processes at different levels and they might be considered possible end points to deduce the classification of such chemicals regarding their potential embryotoxicity using this alternative instead of animal-based methods or in vitro whole embryo cultures. 2 Different procedures that use mouse ES cells to predict embryotoxicity have been developed. 3 The potential use of mouse ES cells to differentiate into cardiomyocytes was one initial approach considered. The embryotoxic potential of tested compounds was microscopically evaluated by not only counting the beating areas of differentiating mouse ES cell cultures but also using direct cytotoxicity assays on mouse ES cells and 3T3 fibroblasts. 4,5 This approach has led to the implementation of a variety of in vitro tests to study embryotoxicity. 6 In this vein, the study of the potential embryotoxic effect of chemicals on mouse ES differentiation to cardiomyocytes has improved by analyzing specific protein expression by flow cytometry. 7 More recently, the disturbance of potential embryotoxic compounds on the differentiation of murine ES cells into endothelial cells has been studied by including the analysis of the expression of 2 specific genes of this cell lineage. 8 Additionally, the possibility of including a specific gene expression analysis in further embryotoxicity tests with human ES cells is currently being considered. 9 However, the specific genes that could be analyzed in future tests should be investigated first in murine models in which ES cell culture conditions have so far been standardized. 10
Mouse ES cells can be cultured and expanded in an undifferentiated state in the presence of the leukemia inhibitory factor (LIF). On cytokine withdrawal, differentiation can be stimulated by the formation of cell aggregates cultured in suspension. In these, so-called embryoid bodies (EBs), the differentiation program resembles certain steps of early mouse embryogenesis that includes an initial primitive endoderm expression as well as the primitive ectoderm or epiblast. 11 Pluripotency markers such as Oct4 are downregulated after a few days upon differentiation. 12 Additional specific gene markers can be studied as indicators of the ongoing differentiation process. One such gene is α-fetoprotein (Afp), the expression of which has been used as a representative endoderm specific marker. The expression of Afp was not detected in the first days of EB cultures and appeared in standard conditions only after 9 to 10 days, in parallel with the reduction of the Oct4 gene expression. The expression of Afp correlates the differentiation of the visceral endoderm, having been demonstrated, both in the early embryo development and in vitro EB differentiation. 13,14 During in vitro differentiation of mouse ES cells, the increase in the expression of the other genes, which were representative of different cell lineages, could also be observed. The mesoderm gene marker, β-myosin heavy chain (Mhc), displayed a high level of expression after 14 days of EB culture. 15 However, neuroectoderm gene markers, such as neurofilament 200 (Nf200), were already seen to be high since the beginning, and in the undifferentiated state, as an indication of a default neuroectodermal commitment of mouse ES cells in the monolayer cultures, even in the presence of LIF. 16 Embryoid bodies can be further plated in adherent dishes in which they spread to show areas of heterogeneous cell lineages.
The analysis of the expression of extraembryonic genes, including the visceral endoderm gene markers, could prove very interesting for the study of potential embryotoxicity as these tissues are of primary importance in the first phase of embryo development. 17,18 In this context, an unbalanced gene expression in extraembryonic tissues has been recently correlated with defective development in porcine-cloned embryos. 19 A previous study of the quantitative image analysis of the Afp expression using green fluorescent protein (GFP), as a reporter gene, was also proposed for testing embryotoxicity, 20 although transfected constructs were not necessarily regulated in the same way as endogenous genes.
The aim of the current study was to analyze and to compare the effect of the strong embryotoxic drug 5-fluorouracil (5-FU), 21 also used as a chemotherapeutic agent, 22 on the genes of the extraembryonic endoderm. We also evaluated the representative genes of the mesodermal lineage by real-time polymerase chain reaction (PCR) in mouse ES line D3-derived EBs. Although the toxic and the therapeutic effects of the 5-FU have been previously observed in the micromolar range, we decided to explore lower concentrations because the high proliferation rate of ES cells and also the longer exposure to 5-FU could cause greater sensitivity to 5-FU alterations in gene expression. The inhibitory effect of 5-FU on the extraembryonic endodermal marker Afp was observed at concentrations from 0.01 to 10 nmol/L, with negligible effect on the viability of monolayer ES line D3 cultures. However, 5-FU did not inhibit the gene expression of 2 characteristic mesodermal genes in the studied range of concentrations. This suggests a striking effect of low concentrations of this embryotoxic drug on the visceral endoderm differentiation. Therefore, this result demonstrates a high sensitivity of the cells of extraembryonic tissues to potential embryotoxicants, which may prove useful in the design of future quantitative methods for testing the embryotoxicity of chemicals using ES cells.
Methods
Cell Culture
Embryonic stem line D3 cells (American Type Culture Collection, Rockville, Maryland) were grown in an undifferentiated state on 75 mm gelatine (0.1% in phosphate-buffered saline [PBS], Sigma Chemical Co, St Louis, Missouri) treated surfaces in DMEM medium (Invitrogen, Karlsruhe, Germany) supplemented with 15% heat-inactivated fetal bovine serum (FBS, Cultex, Spain), 1% nonessential amino acids, 50 U/mL penicillin, 100 μg/mL streptomycin, 0.1 mmol/L β-mercaptoethanol (all from Invitrogen), and 103 U/mL LIF (Chemicon, Temecula, California). 10 For viability tests, 103 cells were seeded on 60-mm gelatine-coated dishes containing 4 mL of ES medium without LIF. On LIF removal, EBs were generated in 4 mL of ES medium by suspension culture (2 × 105 cells) in 60-mm bacterial culture plates.
Test Compounds
5-Fluorouracil (Sigma Chemical Co) was selected as the test compound in this assay. 5-Fluorouracil was dissolved in dimethylsulfoxide (DMSO, Sigma Chemical Co) and diluted with PBS to the working concentration.
Reverse Transcriptase PCR and Real-time Quantitative-PCR Assays
For RNA isolation, EB cells were grown for 23 days in the presence of 5-FU at different concentrations of 5-FU, ranging from 10−6 to 10 nmol/L. Cells growing in the absence of 5-FU were considered a control condition. Total RNA was extracted using Tripure (Roche, Basel, Switzerland) as it was previously reported. 23 RNA was quantified and purity determined according to the 260/280 nm optical density ratio. Then 1 μg RNA was reverse transcribed using expand reverse transcriptase and oligo-dT primers (Roche) according to the manufacturer’s recommendations.
Primer sequences and conditions (annealing temperature, cycle number, and fragment size) used in conventional reverse transcriptase (RT)-PCR were the following: β-actin: Forward (Fw): 5′-TGGGAATGGGTCAGAAGGAC-3′ Reverse (Rv): 5′ -CCAGGGCCATCTTGACGTTA-3′ (60°C, 25 cycles, 468 bp) α-Fetoprotein (Afp): Fw: 5′-CCTTGGCTGCTCAGTACGACAAGG-3′ Rv: 5′-CCTGCAGACACTCCAGCGAGTTTC-3′ (67°C, 26 cycles, 301 bp)
Flk1: Fw: 5′-GCGGTACATCCAGTCCCTGAAGAGG-3′ Rv: 5′-CCGAGGCCACAGACTCCCTGCTT-3′ (63°C, 30 cycles, 124 bp) Amnionless (Amn): Fw: 5′-ACTGCCTCCAACTGGAACCAGAAC-3′ Rv: 5′-CGCAGAGGTCACAGCATTGTCCTT-3′ (62°C, 30 cycles, 667 bp) β-Myosin heavy chain (Mhc): Fw: 5′-CCAAACAGTGTCTGCTCTCCACCGG-3′ Rv: 5′-TTTCGCTCGTTGGGAATGATGCA-3′ (66°C, 30 cycles, 104 bp) Afp: Fw: 5′-GCTGCAAAGCTGACAACAAG-3′ Rv: 5′-GGTTGTTGCCTGGAGGTTTC-3′ (62°C, 142 bp) Flk1: Fw: 5′-GCGGTACATCCAGTCCCTGAAGAGG-3′ Rv: 5′-CCGAGGCCACAGACTCCCTGCTT-3′ (63°C, 124 bp) Amn: Fw: 5′-AAGGACAATGCTGTGACCTCTGCGGA-3′ Rv: 5′-TCAGGAAGAGGTCCAGCAGCCG-3′ (66°C, 101 bp) Mhc: Fw: 5′-CCAAACAGTGTCTGCTCTCCACCGG-3′ Rv: 5′-TTTCGCTCGTTGGGAATGATGCA-3′ (66°C, 104 bp) β-catenin (Cbnnt1): Fw: 5′-GGGTGCTATTCCACGACTAGTTCA-3′ Rv: 5′-ACGCCCTCCACAAACTGCTG-3′ (61°C, 105 bp)
For the quantitative real-time PCR analysis (qPCR), 1 μL of complementary DNA (cDNA) was amplified using the LightCycler FastStart DNA MasterPLUS SYBR Green I kit (Roche). Quantification was performed by standard calculations considering 2(−ΔΔCt).
24
β-Actin was used as unvariant internal control. The PCR program consisted of an initial step at 95°C for 10 minutes followed by 40 cycles of 10 seconds denaturing at 95°C, 7 seconds at the annealing temperature, and 12 seconds at 72°C, plus a final melting curve step. The primer sequences and the annealing temperatures for qPCR were as follows:
Viability Tests
Viability was assessed in ES cell monolayer cultures after 10 days of exposure to 5-FU in ES medium without LIF, using the trypan blue exclusion test. Alternatively, cell suspensions (106 cells/mL) were incubated with 5 μg/mL propidium iodide, and fluorescent cells were analyzed in a Beckman Coulter Epics XL flow cytometer according to manufacturer instructions.
Determination of Cytotoxicity Into EBs
Cytotoxic effects of the 5-FU on EBs were determined with the MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazoliumbromide) assay as previously described. 4 Briefly, EBs were exposed in Petri dishes during 20 days to a range of concentrations of 5-FU, from 1 to 500 nmol/L. At the end of this period, EBs were washed twice with 4 mL PBS and incubated at 37°C in 5% CO2 atmosphere in the presence of 1 mg/mL MTT for 3 hours. Finally, MTT was removed by washing EBs with PBS and EBs were treated with 100 μL of DMSO. Absorbance of the formazan produced by viable cells was measured by recording the absorbance at 590 nm in a Biomek 1000 workstation (Beckman Instruments, Madrid, Spain).
Results
Cell Viability in Monolayer Mouse D3-ES Cell Cultures in the Presence of 5-FU
To establish the range of concentrations at which 5-FU could potentially induce embryotoxic effects in cultured mouse ES cells, a previous study on cell viability was initially carried out using a wide range of concentrations of 5-FU, between 0.00001 and 200 nmol/L. Cell viability was studied, as described in the Methods, by a cell proliferation assay using both trypan blue exclusion and flow cytometry with similar results. As expected, this assay demonstrated (Figure 1) how concentrations above 100 nmol/L 5-FU could have evident cytotoxic effects on mouse D3-ES cells after 10 days in monolayer cultures. Cells were unable to survive after exposure to 200 nmol/L of the drug. At 100 nmol/L 5-FU, 20% of cell death was observed and practically 100% survival was evident at concentrations lower than 10 nmol/L. An IC50 (concentration that reduces viability to 50%) between 100 and 200 nmol/L could be extrapolated for the mouse D3-ES cells from these experiments which is in good agreement with previously published studies of 5-FU cytotoxicity in mouse ES cells. 8,25
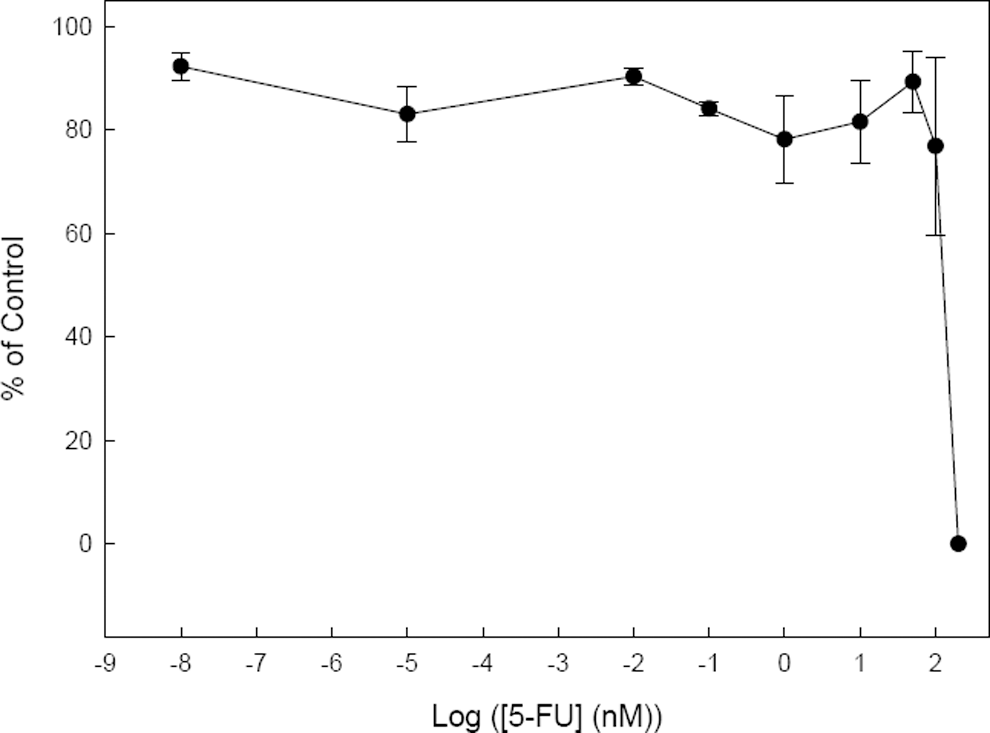
Viability of mouse D3-ES cells cultured in monolayer for 10 days in the presence of increasing concentrations of 5- fluorouracil (5-FU). The percentage of living cells was determined by flow cytometry.
Expression of Different Gene Markers on EBs Development
The mouse ES differentiation process was controlled in the EBs after LIF withdrawal by means of the expression of characteristic genes of the visceral endoderm and mesoderm. The expression of Afp, used as a representative endodermal specific marker, gradually increased throughout the culture and acted as an indicator of the ongoing differentiation processes to reach a high expression after 15 to 20 days, as shown in Figure 2. A similar pattern was observed for the expression of amnionless (Amn), another characteristic gene marker of the primitive endoderm. 26

Gene expression of different gene markers: α-fetoprotein (Afp), Flk1, amnionless (Amn), β-myosin-heavy chain (Mhc), and actin after embryoid body (EB) culture for the indicated days. Genes were analyzed by reverse transcription polymerase chain reaction (RT-PCR).
The expression of Mhc, an established mesodermal marker, displayed a high level after 15 to 20 days in culture and in parallel with Afp and Amn reached a highest expression after 28 days. However, Flk1, another mesodermal gene that was previously expressed in the EBs, displayed higher levels after 15 to 20 days decreasing slightly after 28 days (Figure 2). Neuroectodermal gene markers are constitutively expressed in ES cells (data not shown). 16 In the current study, we have focused on the possible effect of 5-FU on genes which expression arises in the EBs along the differentiation process. Consequently, gene markers characteristic of extraembryonic tissues as well as mesodermal lineages were considered.
Effect of 5-FU on the Different Gene Markers During EBs Development
To establish the possible embryotoxicological potential of 5-FU in EBs derived from mouse D3-ES cells, we studied the effect of the drug on the expression of the Afp, Amn, Mhc, and Flk1 genes by real-time PCR. As observed in Figure 3, the Afp expression decreased significantly over the control, 70% at 0.01 nmol/L 5-FU in the medium and this inhibition was practically sustained at higher concentrations. A concentration which reduces gene expression to 50% (ID50) of 10−3 nmol/L (10−7 μg/mL) could be estimated for this inhibitory effect of 5-FU on the expression of Afp. Interestingly, the cytotoxic effect of 5-FU was barely observed in the monolayer culture (Figure 1) at concentrations ranging between 0.00001 and 10 nmol/L of the drug, at which a significant sustained reduction of the Afp gene expression was observed in EB cultures, which suggests the possible effects of 5-FU in further mouse ES development. The absence of specific markers made it difficult to distinguish between visceral and definitive endoderm lineages during the in vitro differentiation of mouse ES cells. 27 Apparently in the absence of specific conditions that promote a mesendodermal lineage, that is the precursors in the diverging point to endoderm and mesoderm, mouse ES cells spontaneously differentiate toward the extraembryonic endoderm, which suggests that the Afp expression observed in the EB cultures studied in this work could mainly correspond to the visceral endoderm. 28 For that reason, we also studied the effect of 5-FU on the Amn expression, a more specific marker of this lineage. 26 Although Amn expression seems to increase at low concentrations of 5-FU (Figure 3), this effect was not statistically significant. Additionally, we also decided to study the possible effect of the 5-FU on the β-catenin gene (Cbnnt1) expression. Although β-catenin is ubiquitously expressed, the Wnt/β-catenin signaling pathway displays an important role during early embryonic development being first detectable in the extraembryonic visceral endoderm. 29 We did not observe any significant effect of the 5-FU on β-catenin gene expression (Figure 3). These results suggest a specific effect of the 5-FU on the expression of Afp. Because cardiomyocyte differentiation in mouse ES cells has been previously referred to for embryotoxicity studies, we also conducted a parallel investigation on the possible effect of 5-FU on the expression of the gene markers, which were representative of the mesodermal lineage. The expression of Mhc and Flk1, which corresponds to a vascular endothelial growth factor (VEGF) receptor, were considered in this respect. No significant effect of 5-FU on gene expression was observed for Mhc and Flk1 in the studied range of concentrations (data not shown).
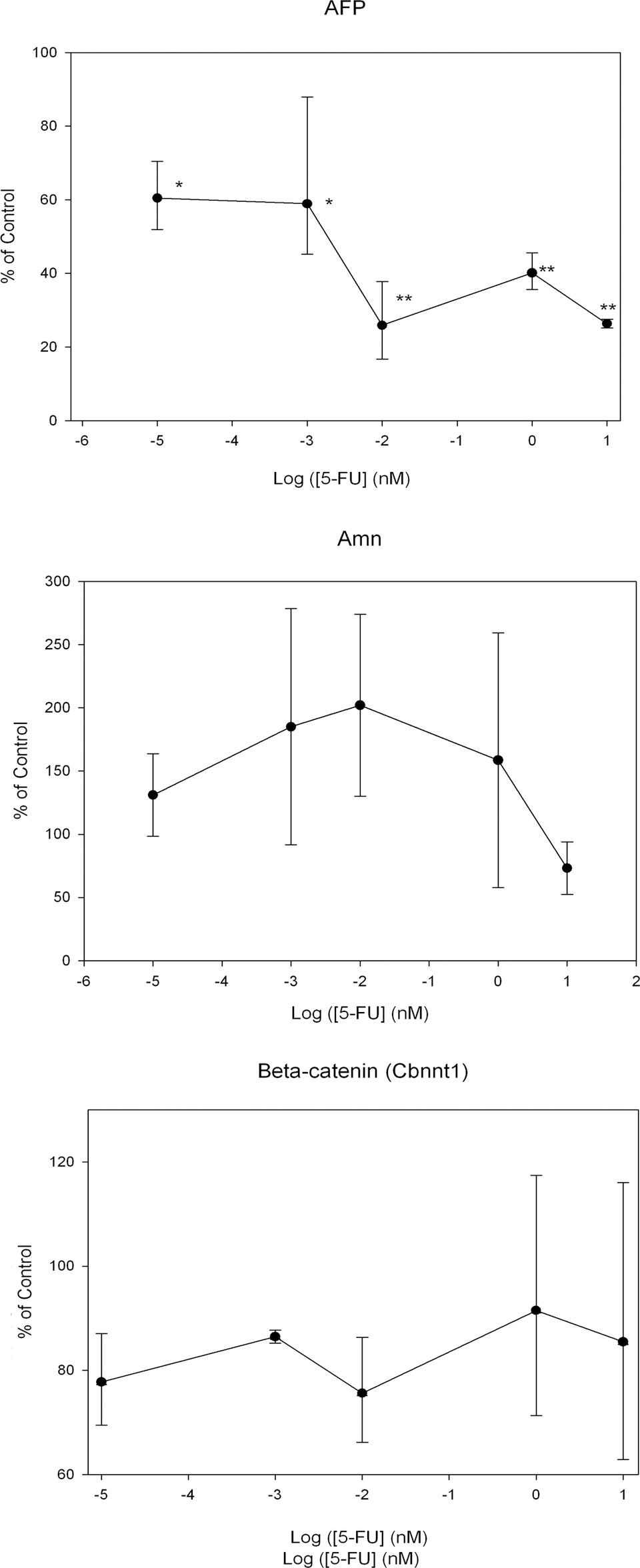
Effect of 5-fluorouracil (5-FU) on the expression of different endodermal gene markers: α-fetoprotein (Afp), amnionless (Amn), and β-catenin (Cbnnt1). Gene expression was quantified after 20 days of EB culture by real-time polymerase chain reaction (PCR). In all cases, the gene expression relative to the control samples is displayed (embryoid bodies [EBs] grown in absence of 5-FU). Asterisks indicate statistically significant differences over the control in the absence of 5-FU (100%). *P < .005, **P < .0005.
Cell Viability Into EB Cultures in the Presence of 5-FU
The toxicological analysis of 5-FU on mouse ES cells has always been studied in monolayer cultures. However, to extend the previous experiments on the effect of 5-FU on the gene expression carried in EBs, we further studied cell viability under the same condition. Embryoid bodies derived from mouse D3-ES cells were cultured in the presence of increasing concentrations of 5-FU and a viability test was carried out as described in Materials and Methods. The cytotoxicity of 5-FU in EBs slightly differed from that in monolayer cultures, and a higher resistance was observed in these cell aggregates. At 300 nmol/L of 5-FU at which cytotoxicity was complete in monolayer cultures, approximately 50% survival was still evident in the EBs (Figure 4) and an extensive cell death was evident in the EBs above 1 μmol/L of 5-FU. Additionally, morphological studies of EBs cultured at different 5-FU concentrations for 20 days confirmed that EBs showed no clear signs of toxicity even at 200 nmol/L of 5-FU (Figure 5) and also that a lethal effect of 5-FU on EB cultures was evident at 1 μmol/L.
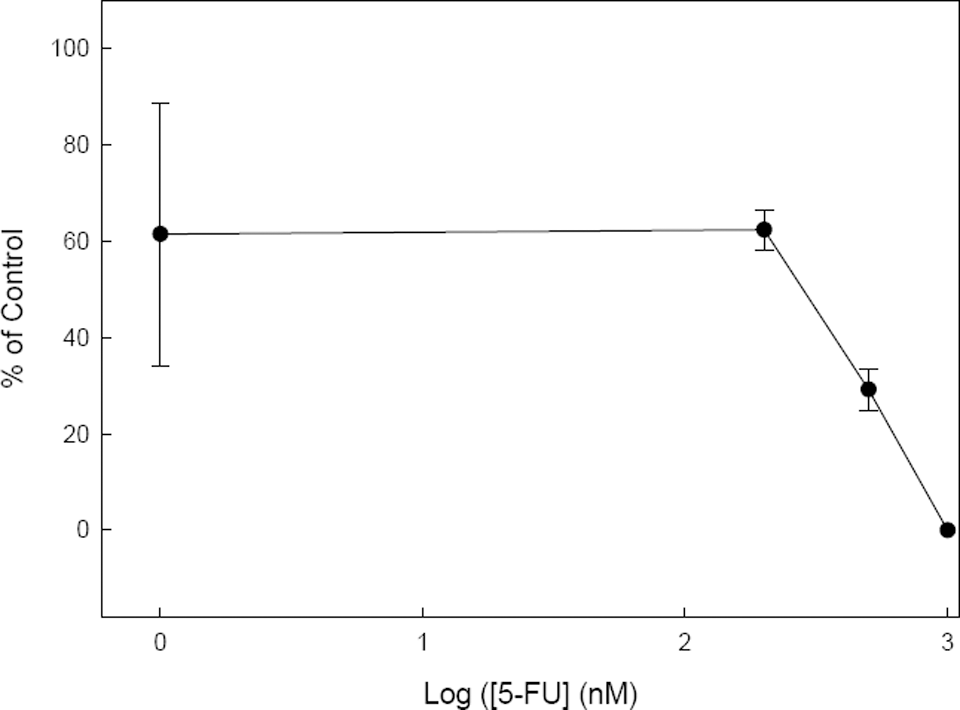
Viability of mouse D3-ES cells cultured as embryoid bodies (EBs) for 20 days in the presence of increasing concentrations of 5-fluorouracil (5-FU). The percentage of living cells was determined by the MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazoliumbromide) cytotoxicity assay.
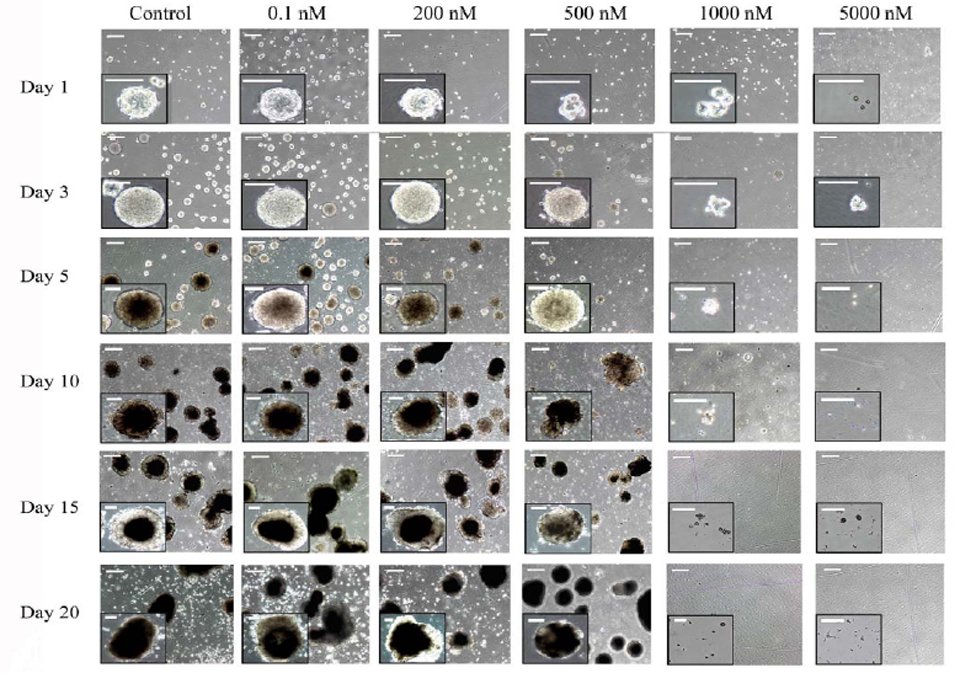
Phase-contrast micrographs of embryoid bodies (EBs) cultured for the indicated days under control conditions or in the presence of the indicated nanomolar concentrations of 5-fluorouracil. Insets are magnifications of EBs in each condition. Bars correspond to 500 μm in low magnification micrographs and to 400 μm in the insets.
Discussion
The correlation between the effects of established embryotoxicants on ES cell differentiation in vitro may allow the design of rapid and feasible assays to improve end points of differentiation for predictive screens of potential teratogenic substances. These effects were initially focused on the maturation of cardiomyocytes from mouse ES cells based on the morphological analysis of the beating areas in EB outgrowths. More recently, the expression of specific genes and proteins from different lineages has also been considered end points of differentiation to test embryotoxicity in vitro to improve an ES cell test for future analysis. 30
Extraembryonic tissues appearing earlier during in vitro ES cell differentiation and in vivo are essential to support of fetal development, in part, by the production of factors that regulate embryonic development. For that reason, a quantitative in vitro test using real-time PCR for the expression of 2 markers of this lineage, such as Afp and Amn, has been studied in EB cultures and in parallel with mesodermal genes in the presence of 5-FU at a wide range of concentrations. The results of the current work have shown a remarkable sensitivity of Afp gene expression to 5-FU. At concentrations as low as 0.01 to 10 nmol/L 5-FU, a decreased expression of this gene was observed. However, the gene expression of Amn, β-catenin, and the mesodermal gene markers was not affected at the same range of concentrations used. It can be assumed that the toxic effect of 5-FU relates with their known potential to inhibit DNA synthesis after conversion to 5-fluoro-2′-deoxyuridine-5′-monophosphate (F-dUMP), which irreversibly binds to thymidylate synthetase. 31 Additionally, 5-FU can be transformed into fluorouridine-5′-triphosphate (F-UTP) that incorporates into RNA and may disrupt further RNA functions as demonstrated in human cell carcinoma. 22 The effects of 5-FU on cell viability in the current study are in good agreement with previous studies. Reported effects of 5-FU on cell toxicity have been considered both time- and concentration-dependent, and the survival of 90% of cells was observed in a clonogenic assay after incubation with 1 μmol/L of 5-FU for 12 hours. 22 Another study on the effects of 5-FU on p53 activation, which could be related with their chemotherapeutic action, was reported at 8 μmol/L after 12 hours of treatment. 32 However, we observed a significant inhibitory effect of 5-FU on Afp gene expression at concentrations at which viability in monolayer cultures was not affected. This effect may be caused by a high rate of proliferation of the entire visceral endoderm in the EBs. However, the expression of Amn and β-catenin was not affected by 5-FU probably because of the more restricted expression of these genes in the visceral endoderm. 26 These results seem to support the fact that the cell differentiation of the bulk of the visceral endoderm in vitro is more sensitive to the effect of 5-FU than differentiation of the mesodermal lineages and that the Afp expression is more sensitive to the toxic effect of 5-FU than other genes expressed in the visceral endoderm, probably because of a higher expression of the Afp gene during the early development of EBs, as it has been previously demonstrated. 33 This effect does not correlate with cell viability in EBs which, in fact, displays a higher resistance to the drug in comparison with monolayer cultures. Therefore, these marker genes should be considered for future developmental toxicity assays.
Because conventional viability tests and differentiation studies were carried out in different cell systems, such as monolayer cultures and EBs, we could partially explain these results by invoking to different accessibilities of 5-FU to the cells under each culture condition. On one hand, we did not observe a similar inhibitory effect in all the tested genes that might correspond to an unspecific effect. Additionally, our results support the hypothesis that cells from the visceral endoderm in EBs that express Afp are more sensitive to the cytotoxic effect of 5-FU than the undifferentiated ES cells in either monolayer cultures or cells from the mesoderm in EBs. Previous studies with different test substances, including 5-FU, have shown that the concentration differences between the cytotoxicity values of 5-FU on mouse ES cells (IC50) and the effect on different end points of differentiation determined from the corresponding concentration-response curves (ID50) are not great. An approximate value of 0.5 μmol/L for the expression of both sarcomeric marker proteins and specific gene markers of endothelial cells has been reported to be in good agreement with the cell viability of mouse ES cells in monolayer cultures. 8,25 Conversely, the current study demonstrates that some precursors from the visceral endoderm lineage in EBs are not only affected at lower concentrations than mesodermal genes but also at lower concentrations that affect cell viability in either monolayer cultures or EBs.
The validated in vitro method for testing embryotoxicity is based on the inhibition of ES cells differentiation to cardiomyocytes. This method has been developed in validation studies displaying sufficient capability to discriminate between strong embryotoxic compounds and weak or nonembryotoxic compounds.31 However, the capability to discriminate between weak and nonembryotoxic compounds needs to be improved and some of the suggestions of the European Centre for Validation of Alternative Methods (ECVAM) to reach this goal include the use of more quantitative end points, such as RT-PCR of specific genes. 34 This work meets this requirement because it has been demonstrated that the expression of the specific gene marker of the endoderm Afp is inhibited after exposure to 5-FU. In addition, the end point used in this work seems to be of several orders of magnitude more sensitive than the differentiation to cardiomyocytes. Indeed, the differentiation to cardiomyocytes is inhibited by 5-FU with an ID50 of 0.05 μg/mL while we observed that the expression of the Afp gene is inhibited with an ID50 of 10−3 nmol/L (10−7 μg/mL). 8 Thus, this higher sensitivity would strongly contribute to improve the capability of the method to discriminate between weak and nonembryotoxic chemicals.
The predictability of the validated in vitro method for testing embryotoxicity is supported in a statistical prediction model based on the correlation of the ID50 for differentiation to cardiomyocytes and the inhibition of the cell viability of D3 (IC50 D3) and 3T3 fibroblasts (IC50 3T3). 8 In this way, function I has been defined as 5.916 log (IC50 3T3) + 3.500 log (IC50 D3) − 5.307 ([IC50 3T3-ID50]/ID50 3T3) − 15.27; function II was defined as 3.651 log (IC50 3T3) + 2.394 log (IC50 D3) − 2.033 ([IC50 3T3-ID50]/IC50 3T3) − 6.85; while function III was defined as −0.125 log (IC50 3T3) − 1.917 log (IC50 D3) +1.500 ([IC50 3T3-ID50]/IC50 3T3) − 2.67. According to this model, the tested compound must be classified as strongly embryotoxic when III > I and III > II. As an initial approach for validating the inhibition of the Afp gene expression as an end point for testing embryotoxicity, we applied this statistical model to our results. A previous report indicated the cytotoxic effect of 5-FU on 3T3 cells resulting in an IC50 3T3 of 0.21 μg/mL. 8 In the current study, we have shown with mouse ES line D3 cells an ID50 of 10−7 μg/mL (10−3 nmol/L) for Afp expression and an IC50 D3 of 300 nmol/L (0.041 μg/mL) for the cytotoxic effects of 5-FU in the monolayer cultures. By applying these values to the described functions for the prediction model, we obtained the following values: function I = −29, function II = −15, and function III = 2, which would also correspond to a strongly embryotoxic compounds, as was clearly established for both the in vivo and in vitro tests.
Therefore, this study demonstrates that visceral endoderm differentiation studied by real-time PCR should be included in future simple and reliable embryotoxicity assays for different potential teratogenic substances. The development of directed differentiation protocols in monolayer cultures could complement the results based on spontaneous differentiation into EB structures.
Footnotes
Acknowledgments
The authors thank Encarna Fuster for her technical assistance.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research and/or authorship of this article: the “Fundación Médica Mutua Madrileña” to JAR and ER, the “Generalitat Valenciana” (AP-089/08 and AP-100/09) and the “Instituto de Salud Carlos III” (PS09/01093) to ER, and “Ministerio de Medio Ambiente y Medio Rural” (A051/2007/3-14.4) to MAS.
