Abstract
The chemical warfare agents sulfur mustard (SM) and nitrogen mustards (HN-1, HN-2, and HN-3) are highly reactive vesicants. The study was planned to investigate the protective efficacy of amifostine, DRDE-07 and their analogues, and few conventional antidotes (30 minutes pretreatment) against dermally applied SM and nitrogen mustards in preventing hematological and biochemical changes in mice. Mustard agents (1.0 median lethal dose [LD50]) induced a significant decrease in the body weight and spleen weight. A significant decrease in the white blood cell (WBC) count and an increase in serum transaminases and alkaline phosphatases (ALPs) were observed. A significant decrease in reduced (GSH) and oxidized glutathione (GSSG) and an increase in thiobarbituric acid reactive substances were also observed. All the mustard agents increased DNA fragmentation. The effects of SM were significantly ameliorated by DRDE-07 analogues, and with nitrogen mustards the protection was partial. Overall, DRDE-30 (propyl analogue) followed by DRDE-35 (butyl analogue) are favored as safer and better compounds.
Introduction
The chemical warfare agents sulfur mustard (SM) and its related compounds nitrogen mustards (HN-1, HN-2, and HN-3) are highly reactive blistering agents. 1 Sulfur and nitrogen mustards are also potential threat by their use by terrorists, as they are cheap, easy to manufacture and store, but sometimes difficult to verify. Although nitrogen mustards are listed as chemical warfare agents (schedule 1A, chemical weapons convention), little attention has been focused for the development of an antidote and to date no really effective antidote against nitrogen mustards has evolved. An effective antidote for the nitrogen mustards will also have beneficial effect as an adjuvant to cancer chemotherapy as a cytoprotectant. Although various prophylactic agents have been suggested to counteract its toxic effects, none of them have been found to protect the major consequences of nitrogen mustards toxicity. All efforts of drug development are targeted toward attenuation of secondary biochemical or pathophysiological consequences of alkylation. Several antidotes have been reported for the systemic toxicity of sulfur as well as nitrogen mustards in experimental animals. 2 –4 All the antidotes screened so far have given only limited protection.
The synthetic aminothiol amifostine (S-2[3-aminopropylamino]ethyl phosphorothioate), earlier known as WR-2721, has been extensively used as a cytoprotector for normal tissues in cancer radiotherapy and chemotherapy. This stimulated us to use amifostine and some of its analogues with better lipophilicity against SM. Amifostine and DRDE-07 (S-2[2-aminoethylamino]ethyl phenyl sulfide) gave very good protection as a prophylactic agent against SM. 5,6 When given orally, DRDE-07 was more effective than orally administered amifostine as a prophylactic agent against percutaneously administered SM. 7,8 Later, several DRDE-07 analogues were synthesized and many of them were found to be effective against SM in mice, that is, DRDE-10, DRDE-30, and DRDE-35. 6,9 These analogues were found to be partially effective against mechlorethamine (HN-2) in correcting the biochemical alterations. 10 Because mustard agents also cause oxidative stress and free radical generation, it may be worthwhile to administer the drugs repeatedly for a better efficacy. In continuation of this in the current study, we evaluated these analogues by repeated administration against dermally applied nitrogen mustards (HN-1, HN-2, and HN-3) and SM. Some of the recommended drugs for the treatment of SM and nitrogen mustard, such as N-acetyl cysteine (NAC), melatonin, and sodium thiosulfate (STS), were included in this study for comparison.
Materials and Method
Chemicals
Nitrogen mustards (HN-1, bis-(2-chloroethyl)ethylamine; HN-2, bis-(2-chloroethyl)methylamine; and HN-3, Tris-(2-chloroethyl)amine) and SM (SM, 2,2-dichloroethyl sulfide) were synthesized in the Synthetic Chemistry Division and was found to be above 99% pure by gas chromatographic analysis. Utmost care was taken during the synthesis of these scheduled chemicals, per the guidelines of the institute. S-2[3-aminopropylamino]ethyl phosphorothioate (amifostine), S-2-(2-aminoethylamino) ethyl phenyl sulfide (DRDE-07), S-2-(2-aminoethylamino) ethyl tolyl sulfide (DRDE-10), S-2-(2-aminoethylamino) ethyl propyl sulfide (DRDE-30), and S-2-(2-aminoethylamino) ethyl butyl sulfide (DRDE-35) were synthesized in the Synthetic Chemistry Division of Defence Research and Development Establishment (DRDE) and were characterized by elemental analysis, infrared (IR), 1 H nuclear magnetic resonance (NMR), and mass spectrometry (MS) analysis, and the purity was checked by thin-layer chromatography (TLC). O-pthalaldehyde, glutathione, and 4′6-diamidino-2-phenylindole were purchased from Sigma Chemical Company (St. Louis, MO, USA). Other chemicals of high purity were from Qualigens (Mumbai, India) or E-Merck (Mumbai, India).
Animals
Randomly bred Swiss female mice (25-30 g) from the Institute’s Animal Facility were used for the study. The animals were kept in polypropylene cages with sterilized and dry paddy husk as a bedding material. Free access to food (Ashirwad Ltd, Chandigarh, India) and water were allowed until 2 hours before the experiment. The care and maintenance of animals were per the approved guidelines of the Committee for the Purpose of Control and Supervision of Experiments on Animals (CPCSEA, New Delhi, India). A day before percutaneous exposure, hair on the back of the animals was closely clipped using a pair of scissors. Food and water were allowed 2 hours after the experiment. All animal procedures were approved by the Institutional Animal Ethical Committee.
Protective Efficacy of Different Antidotes Against Nitrogen Mustards and SM Toxicity
For amifostine, NAC, melatonin, and sodium thiosulphate, a dose of 185, 250, 250, and 1000 mg/kg, respectively, was used. The dose of DRDE-07 was 249 mg/kg and for other analogues, equimolar dose of DRDE-07 was selected for this study. All antidotes were dissolved in water except melatonin that was dissolved in dimethyl sulphoxide (DMSO). All the antidotes were administered orally, one dose 30 minutes before the exposure of HN-1, HN-2, HN-3, and SM and remaining doses on the next 3 or 7 successive days (a total of 4 and 8 doses). Six animals per group were used. Animals in the mustard only (toxicant) group received distilled water as a pretreatment. Dimethyl sulphoxide or PEG-300 was applied on the back of animals in the control group. HN-1, HN-2, and HN-3 (dissolved in DMSO) and SM (dissolved in PEG-300) were applied dermally on the animals at a dose of 1.0 median lethal dose (LD50; HN-1 = 11.9, HN-2 = 20.0, HN-3 = 7.1, and SM = 8.1 mg/kg). The dilutions were made in such a manner that the quantity applied was between 30 and 50 µL. The diluted solution was smeared uniformly on the back of the animals on a circular area of 1.5-cm diameter, using a gas tight syringe (Harvard Apparatus, HSE-Harvard, March-Hugstetten, Germany).
After exposure of nitrogen mustards and SM, the animals were kept in a well-ventilated room for 24 hours and then shifted to the experimental animal room for further monitoring. The body weight was recorded daily and the animals were observed for mortality and general health.
Hematological and Biochemical Evaluation
Twenty-four hours after the last antidote administration, the animals were anaesthetized with ether for collection of blood from orbital sinus and then sacrificed by cervical dislocation for removal of liver and spleen, and they were weighed. Part of the liver tissue was used for biochemical estimations. Hepatic GSH and GSSG contents were estimated fluorimetrically per the method of Hisin and Hilf. 11 For this, 150 mg of liver tissue was homogenized in 4 mL of phosphate EDTA buffer and 25% metaphosphoric acid. The content of the tube was centrifuged and the supernatant was used for the estimation of GSH and GSSG. Hepatic lipid peroxidation was determined by measuring the level of malondialdehyde (MDA) according to the method of Buege and Aust. 12 One hundred milligrams of liver tissue were directly homogenized in 5 mL of thiobarbituric acid reagent and boiled for 30 minutes. The contents of the tubes were cooled, centrifuged, and absorbance of the clear supernatant was measured at 535 nm. The amount of MDA formed was calculated using a molar extinction coefficient of 1.56 × 105/M per cm. DNA fragmentation assay was carried out per the method of Rao et al. 13 Briefly, the liver from control and treated animals was quickly excised and frozen. The frozen tissue was homogenized in ice-cold lysis buffer (10 mmol/L Tris, 20 mmol/L EDTA, 0.5% triton X-100, pH 8.0) and then centrifuged at 20 000g for 30 minutes. Both pellet (intact chromatin) and supernatant (DNA fragments) were assayed for DNA content fluorimetrically by using 4,6-diamidino-2-phenyl indol (DAPI). To 2 mL of the reagent, 20 µL of the sample was added, and then fluorescence intensity was measured at 450 nm with excitation at 362 nm. The percentage of fragmented DNA was defined as the ratio of the DNA content of the supernatant at 27 000g to the total DNA in lysate. 14 The hematological variables, such as WBC, red blood cell (RBC), and hemoglobin (Hb), were analyzed using Bechman Coulter Analyser (Brea, CA, USA). The plasma enzymes such as alanine transaminase (ALT), aspartate transaminase (AST), and ALP were analyzed by Alfa Wassermann Clinical Analyser (Milan, Italy), per the manufacturer’s protocol.
Statistical Analysis
The data were analyzed using 1-way analysis of variance with Dunnet comparisons test. A probability of less than 0.05 was taken as statistically significant. The analyses were carried out using SigmaStat for Windows version 2.03 (SPSS Inc, Richmond, CA, USA).
Results
Effect of Various Treatments on Mortality, Body, and Spleen Weight
Following repeated administration of the antidotes alone for 4 or 8 days in mice, the body weight or organs weights did not show any significant change compared to control group. There was also no significant change in the various hematological and biochemical variables following repeated administration of the antidotes only. No death occurred in HN-1, HN-3, and SM group on fourth day in presence of the antidotes. However, mortality occurred with the antidotes in HN-2 after 4 days and HN-1, HN-2, HN-3, and SM on eighth day. There was a significant weight loss on fourth and eighth day, with a pronounced effect on eighth day following administration of HN-1 alone. Weight loss was also observed with all antidotes, except DRDE-10, DRDE-30, and melatonin showing partial recovery on fourth day. Significant decrease in spleen weight was observed on fourth day and on eighth day following percutaneous administration of HN-1. None of the antidotes protected the decrease in spleen weight on fourth day, but on eighth day, significant protection was observed with DRDE-30, amifostine, and melatonin groups. Drastic loss in body weight was observed on fourth day in HN-2 group, which was further decreased on eighth day. There was drastic fall in spleen weight on fourth day, but the same was normalized on eighth day compared to control group. No recovery in body weight and spleen weight loss was observed in any of the antidotes on fourth day as well as eighth day (Tables 1 and 2).
Body Weight (Percentage) of Mice With and Without the Antidotes (Oral) Against Percutaneously Administered Mustard Agents a
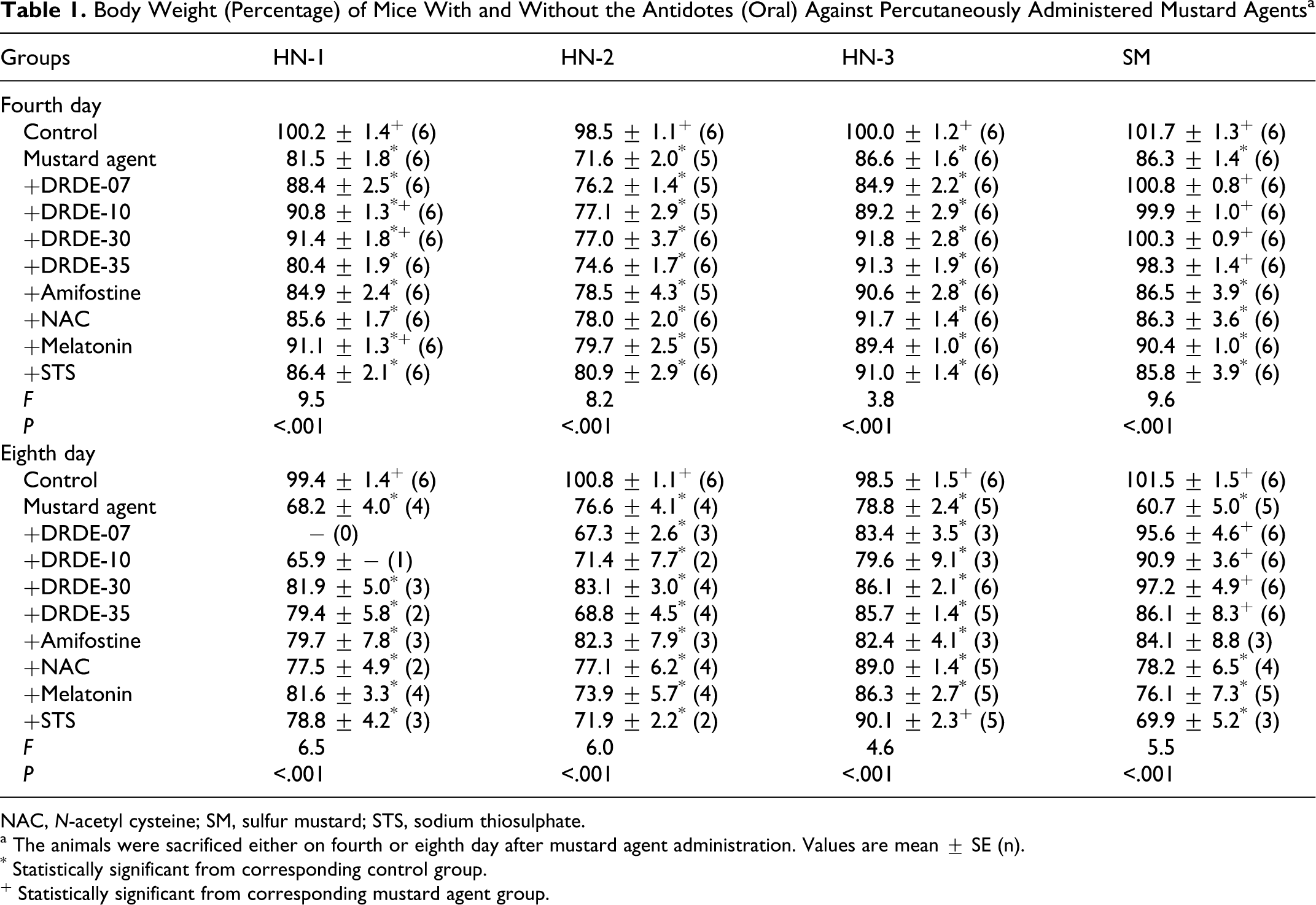
NAC, N-acetyl cysteine; SM, sulfur mustard; STS, sodium thiosulphate.
a The animals were sacrificed either on fourth or eighth day after mustard agent administration. Values are mean ± SE (n).
* Statistically significant from corresponding control group.
+ Statistically significant from corresponding mustard agent group.
Spleen Weight (Percentage) of Mice With and Without the Antidotes (Oral) Against Percutaneously Administered Mustard Agents a
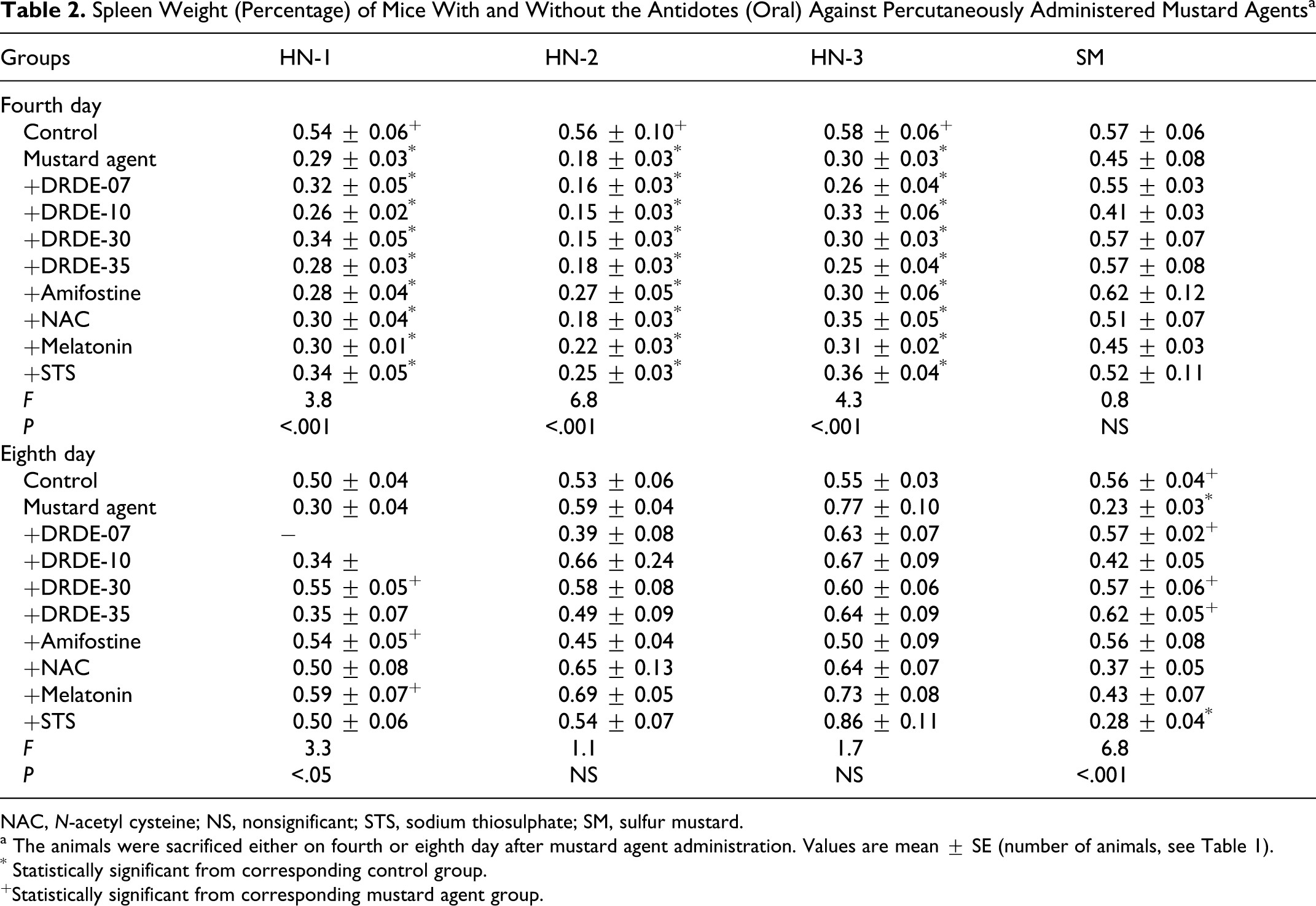
NAC, N-acetyl cysteine; NS, nonsignificant; STS, sodium thiosulphate; SM, sulfur mustard.
a The animals were sacrificed either on fourth or eighth day after mustard agent administration. Values are mean ± SE (number of animals, see Table 1).
* Statistically significant from corresponding control group.
+Statistically significant from corresponding mustard agent group.
HN-3 also showed significant body weight loss on fourth day in all groups except in STS. The body weight further declined on eighth day in all groups except in STS. There was a significant fall in the spleen weight on fourth day, which was not significant on eighth day when compared to control group. Like all nitrogen mustards, SM also showed significant body weight loss on fourth day. All analogues of DRDE-07 gave a significant protection. On eighth day, the body weight loss was more than fourth day. There was significant recovery in body weight loss in amifostine, DRDE-07, and its analogous. In the spleen weight, there was no significant change on fourth day, but on eighth day significant fall was observed in SM-treated group. Amifostine, DRDE-07, and its analogous showed significant protection.
Effect of Various Antidotes on Hematological Parameters of Mice After Mustard Agent’s Systemic Toxicity
Table 3 summarizes WBC count, RBC count, and Hb level of 4 and 8 days treatment of antidotes against percutaneously administered HN-1, HN-2, HN-3, and SM. There was a significant decrease in the level of WBC count on fourth day as well as on eighth day in HN-1 group when compared to control group. Only DRDE-30 showed partial protection on fourth day and on eighth day, and other antidotes did not protect the decrease in WBC count. No significant difference was observed in RBC count and Hb level when compared to control group, even in HN-1 alone group. The WBC count was significantly decreased in all groups on fourth day in HN-2 group, but on eighth day no significant change was observed. None of the antidotes significantly altered the fall in the WBC count on eighth day. There was no significant difference observed between the groups in case of RBC count and Hb level on fourth day as well as eighth day. In HN-3-exposed group, there was a significant decrease in the level of WBC count on fourth day and on eighth day when compared to control group. No protection was observed on fourth day in any of the groups when compared to HN-3-treated group. However, on eighth day, significant protection was observed in DRDE-07, DRDE-30, melatonin, and STS groups. No significant difference was observed between the groups in case of RBC count and Hb level on fourth day as well as eighth day. Sulfur mustard induced a significant fall in the level of WBC count on fourth and eighth day. Significant protection in WBC count was observed in DRDE-07, DRDE-10, DRDE-30, and DRDE-35 on fourth day and DRDE-07, DRDE-30, and DRDE-35 on eighth day. Red blood cell count and Hb level were slightly increased in SM-treated group on fourth and eighth day but that was not statistically significant.
Hematological Variables of Mice on Fourth and Eighth Day With and Without the Antidotes (Oral) Against Percutaneously Administered Mustard Agents a
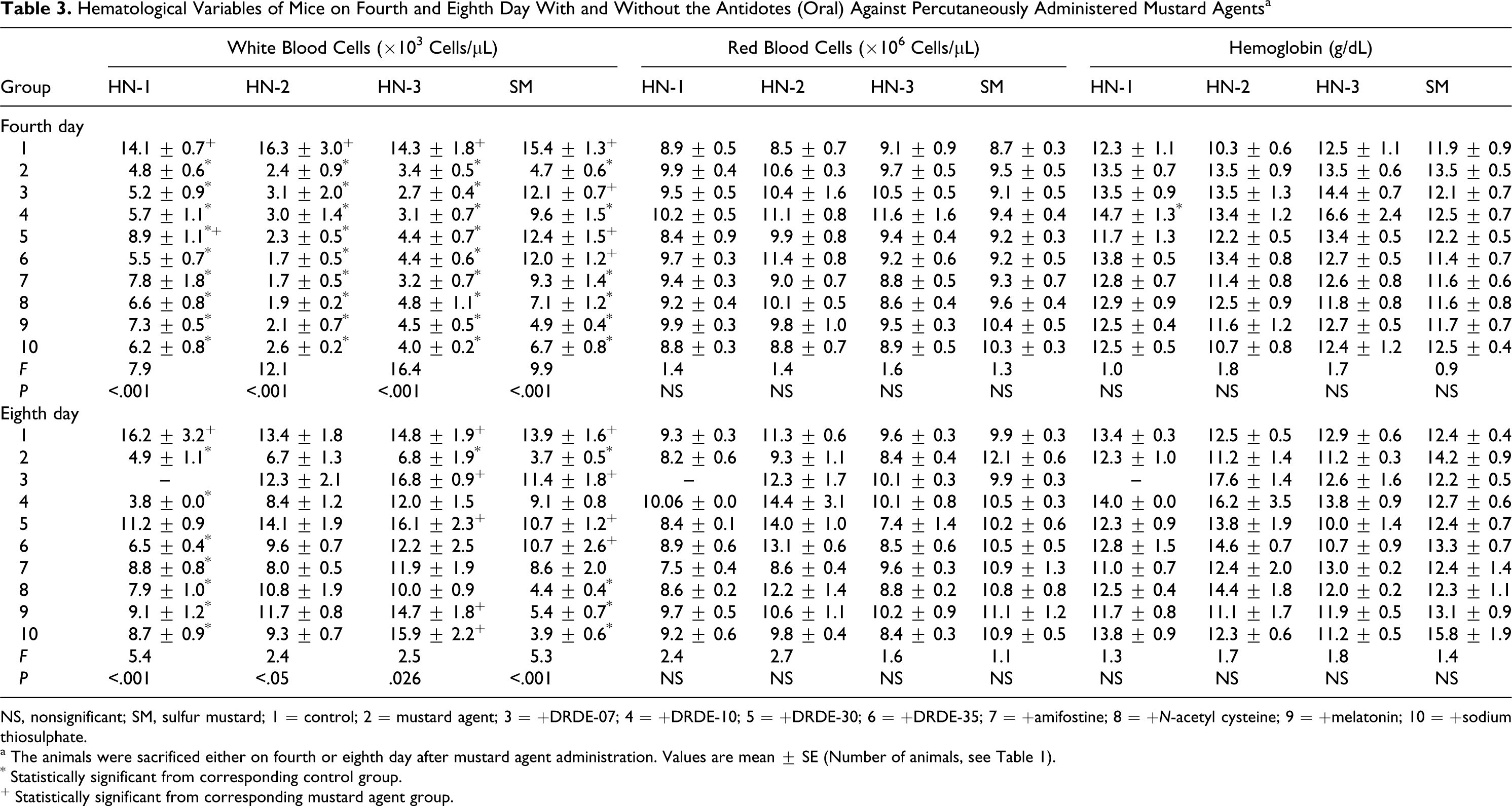
NS, nonsignificant; SM, sulfur mustard; 1 = control; 2 = mustard agent; 3 = +DRDE-07; 4 = +DRDE-10; 5 = +DRDE-30; 6 = +DRDE-35; 7 = +amifostine; 8 = +N-acetyl cysteine; 9 = +melatonin; 10 = +sodium thiosulphate.
a The animals were sacrificed either on fourth or eighth day after mustard agent administration. Values are mean ± SE (Number of animals, see Table 1).
* Statistically significant from corresponding control group.
+ Statistically significant from corresponding mustard agent group.
Effect of the Antidotes on Selected Enzymes
Table 4 summarizes AST, ALT, and ALP levels after repeated dosing for 4 and 8 days with various antidotes against percutaneously administered HN-1, HN-2, HN-3, and SM, respectively. HN-1, HN-2, HN-3, and SM showed an increase in the level of AST, ALT, and ALP on fourth as well as eighth day but the effect on 8 days was more prominent. All the antidotes showed either no protection or partial protection against HN-1, HN-2, and HN-3. In case of SM, DRDE-07, DRDE-10, DRDE-30, and DRDE-35 on fourth day and DRDE-07 and DRDE-30 on seventh day significantly protected the AST level. The ALT level was protected by DRDE-07, DRDE-10, DRDE-30, and DRDE-35. The level of ALP was not changed significantly with SM and there was no change in the presence of the antidotes.
Few Liver Enzymes Variables of Mice on Fourth and Eighth Day With and Without the Antidotes (Oral) Against Percutaneously Administered Mustard Agents a
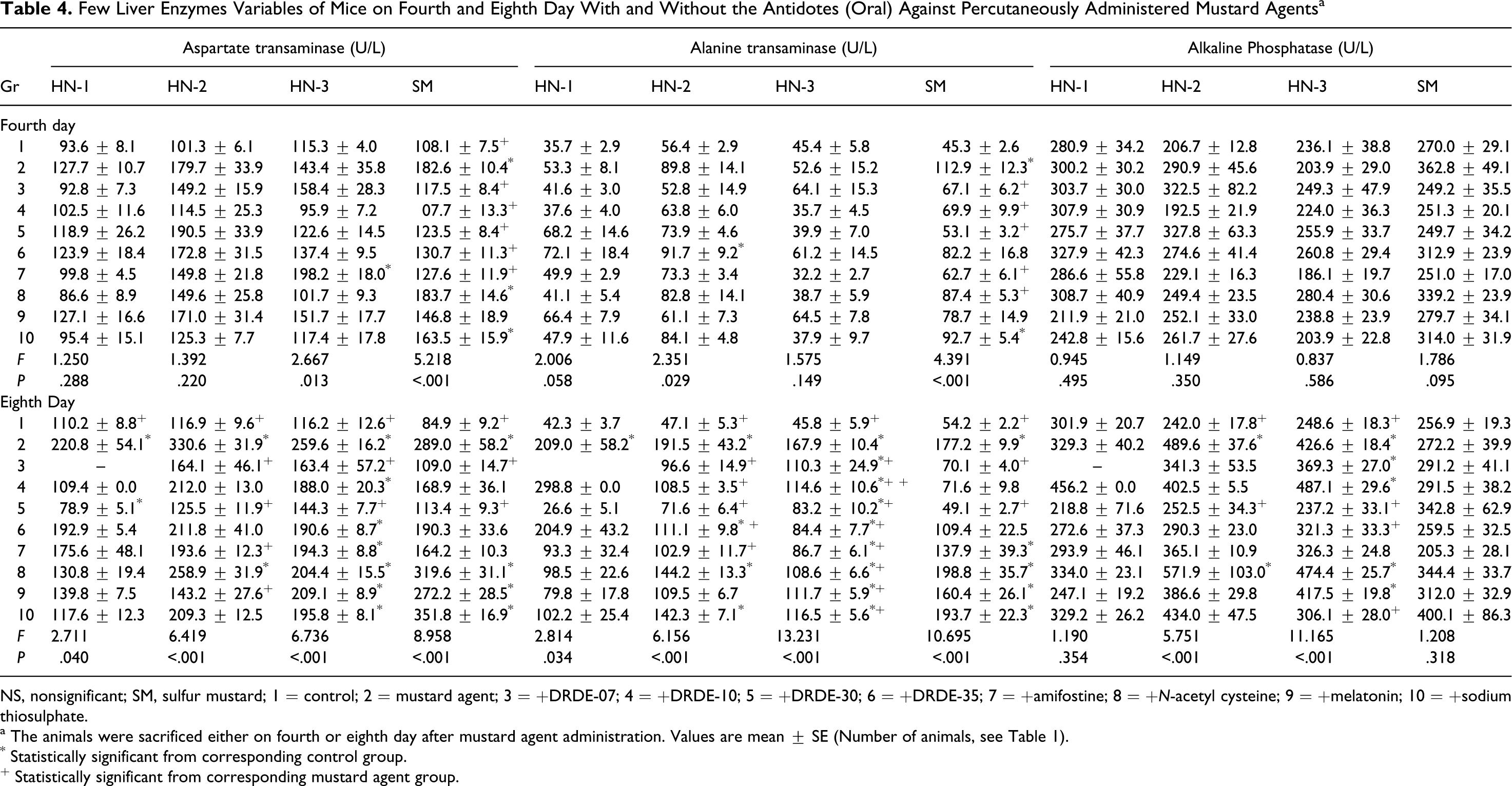
NS, nonsignificant; SM, sulfur mustard; 1 = control; 2 = mustard agent; 3 = +DRDE-07; 4 = +DRDE-10; 5 = +DRDE-30; 6 = +DRDE-35; 7 = +amifostine; 8 = +N-acetyl cysteine; 9 = +melatonin; 10 = +sodium thiosulphate.
a The animals were sacrificed either on fourth or eighth day after mustard agent administration. Values are mean ± SE (Number of animals, see Table 1).
* Statistically significant from corresponding control group.
+ Statistically significant from corresponding mustard agent group.
Effect of Various Antidotes on Biochemical Parameters of Mice After Mustard Agent’s Intoxication
Figures 1, 2, 3, and 4 summarizes the effect of percutaneously administered HN-1, HN-2, HN-3, and SM with various antidote treatments for 4 and 8 days on the levels of GSH, GSSG, lipid peroxidation, and percentage DNA damage, respectively. Following HN-1 administration, there was a marked decrease in GSH level on fourth day. Except DRDE-35 and amifostine, the other antidotes significantly protected it. On eighth day also, the level of GSH was significantly lower and DRDE-30 and DRDE-35 significantly protected. Following HN-2 administration, there was a significant decrease in the level of GSH on fourth and eighth day. None of the antidotes protected the level of GSH on fourth day but all analogues of DRDE-07 and other antidotes gave partial to significant protection on eighth day except DRDE-10 and STS. Exposure to HN-3 also showed a significant fall in GSH level in all groups on fourth day, which remained decreased up to eighth day. DRDE-07, DRDE-30, DRDE-35, amifostine, and melatonin showed significant protection on eighth day. Sulfur mustard exposure also showed a decrease in GSH on fourth day, which was further decreased on eighth day. Amifostine, DRDE-07, and its analogues gave significant protection on fourth day as well as on eighth day (Figure 1).
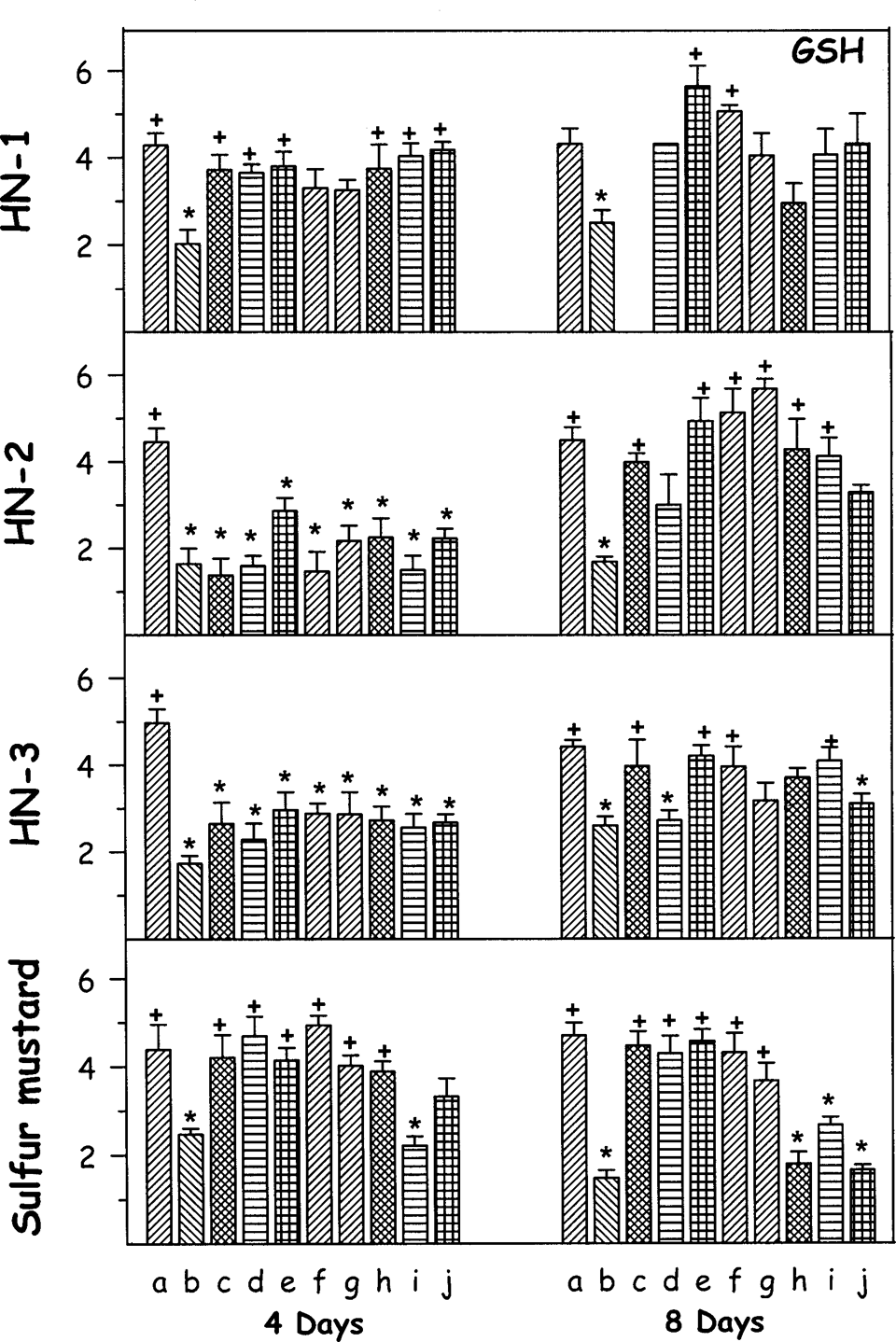
Effect of oral treatment of various antidotes (repeated dosing) on reduced glutathione (GSH) against percutaneously administered 1.0 median lethal dose (LD50) of various mustard agents on female mice after third and seventh day. The animals were sacrificed on fourth or eighth day after administration of mustard agents. Values are in mean ± SE. a = control; b = toxicant; c = +DRDE-07; d = +DRDE-10; e = +DRDE-30; f = +DRDE-35; g = +amifostine; h = +N-acetyl cysteine; i = +melatonin; j = +sodium thiosulphate. *Statistically significant compared to control. +Statistically significant compared to toxicant group. GSH = ×10−3 µmols/g of tissue.
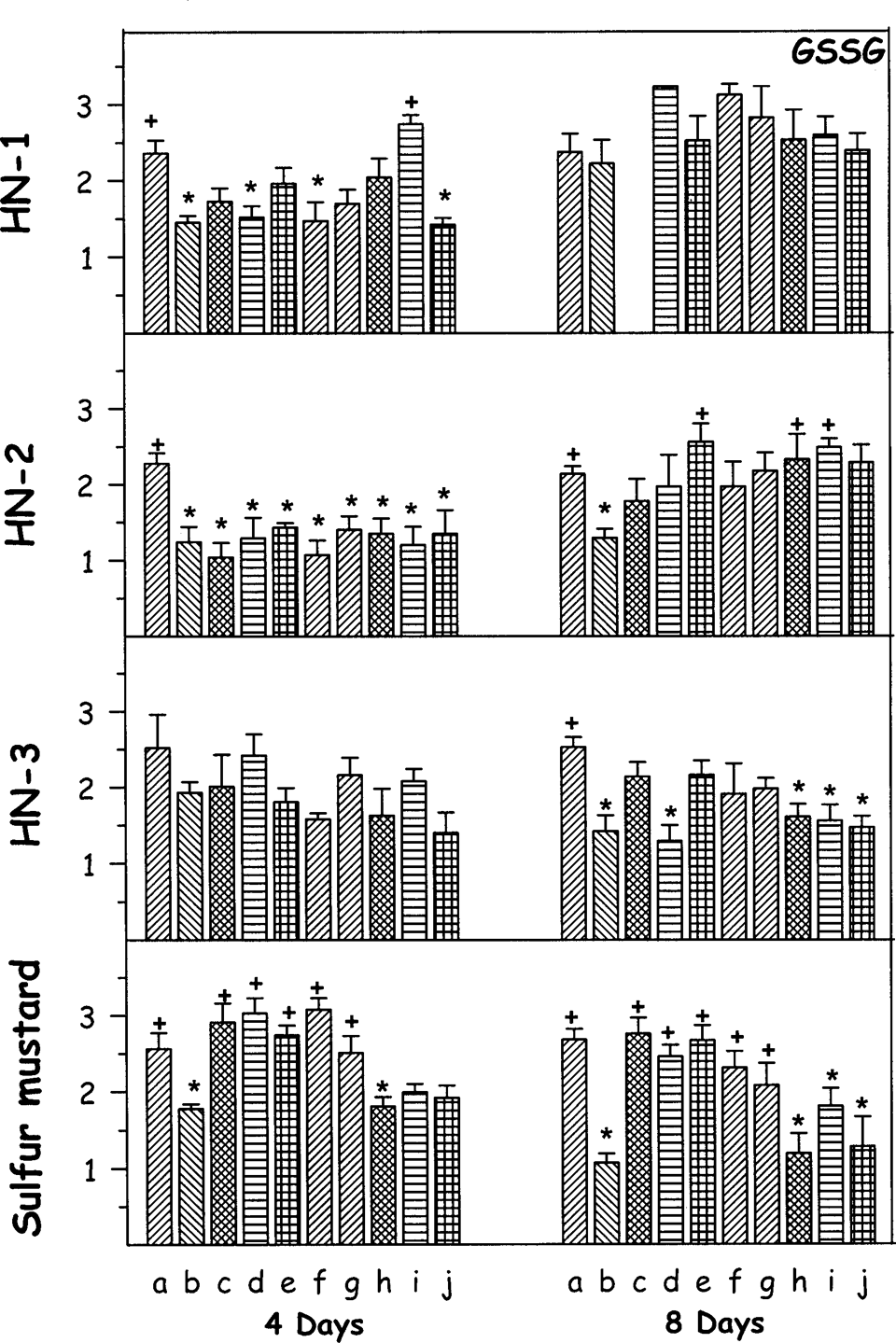
Effect of oral treatment of various antidotes (repeated dosing) on oxidized glutathione (GSSG) against percutaneously administered 1.0 median lethal dose (LD50) of various mustard agents on female mice after third and seventh day. The animals were sacrificed on fourth or eighth day after administration of mustard agents. Values are in mean ± SE. a = control; b = toxicant; c = +DRDE-07; d = +DRDE-10; e = +DRDE-30; f = +DRDE-35; g = +amifostine; h = +N-acetyl cysteine; i = +melatonin; j = +sodium thiosulphate. *Statistically significant compared to control. +Statistically significant compared to toxicant group. GSSG = ×10−3 µmol/g of tissue.
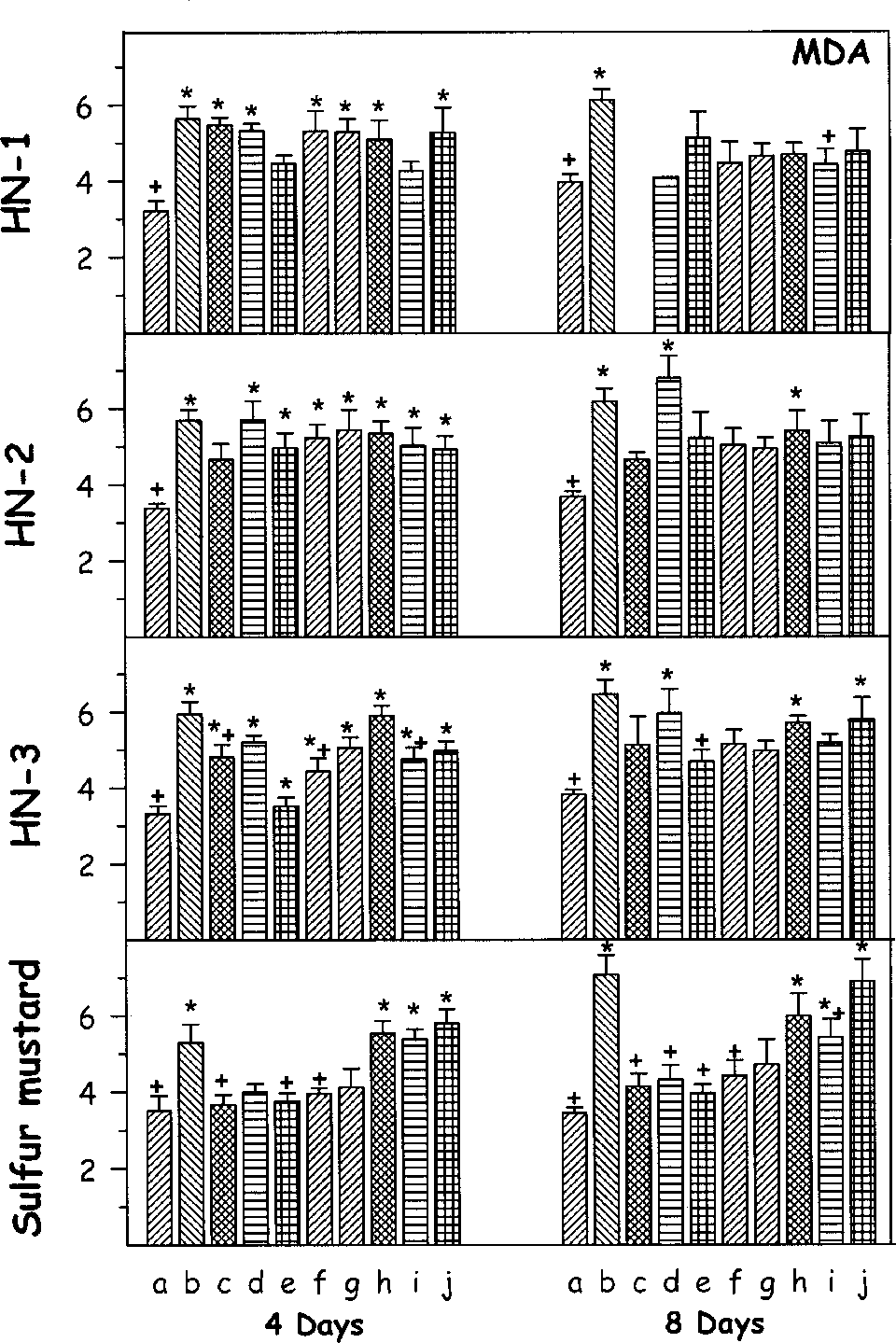
Effect of oral treatment of various antidotes (repeated dosing) on lipid peroxidation (malondialdehyde [MDA]) against percutaneously administered 1.0 median lethal dose (LD50) of various mustard agents on female mice after third and seventh day. The animals were sacrificed on fourth or eighth day after administration of mustard agents. Values are in mean ± SE. a = control; b = toxicant; c = +DRDE-07; d = +DRDE-10; e = +DRDE-30; f = +DRDE-35; g = +amifostine; h = +N-acetyl cysteine; i = +melatonin; j = +sodium thiosulphate. *Statistically significant compared to control. +Statistically significant compared to toxicant group. MDA = ×10−4 nmols/g of tissue.
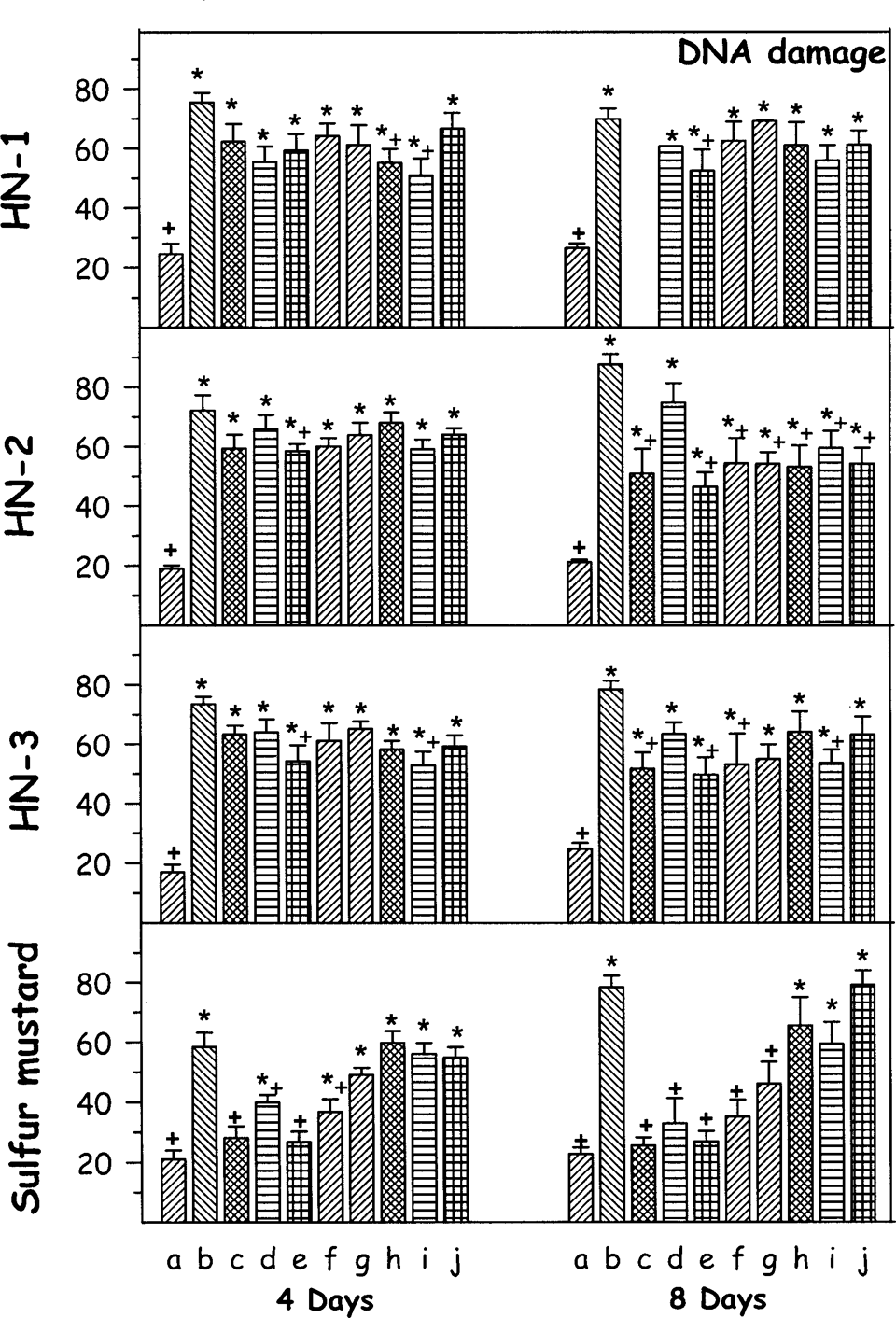
Effect of oral treatment of various antidotes (repeated dosing) on percentage DNA damage against percutaneously administered 1.0 median lethal dose (LD50) dose of various mustard agents on female mice after third and seventh day. The animals were sacrificed on fourth or eighth day after administration of mustard agents. Values are in mean ± SE. a = control; b = toxicant; c = +DRDE-07; d = +DRDE-10; e = +DRDE-30; f = +DRDE-35; g = +amifostine; h = +N-acetyl cysteine; i = +melatonin; j = +sodium thiosulphate. *Statistically significant compared to control. +Statistically significant compared to toxicant group. DNA = percentage DNA damage.
Following HN-1 administration, the GSSG level decreased significantly on fourth day and was near to normal values on eighth day. Melatonin treatment significantly protected it on fourth day. There was a significant decrease in the level of GSSG on fourth day and on eighth day following exposure to HN-2. None of the antidotes protected the level of GSSG on fourth day. DRDE-30, NAC, and melatonin gave significant protection on eighth day and the other antidotes gave partial protection. There was no significant change in the level of GSSG following percutaneous administration of HN-3 on fourth day, but on eighth day significant decrease was observed. DRDE-07, DRDE-30, DRDE-35, and amifostine gave partial protection. Sulfur mustard decreased the GSSG level on fourth day, which was further decreased on eighth day. Amifostine, DRDE-07, and its analogues gave significant protection on fourth day as well as on eighth day (Figure 2).
There was an increase in the level of lipid peroxidation as shown by the significant increase in MDA on fourth day following percutaneous administration of HN-1. DRDE-30 and melatonin significantly protected it. On eighth day also, the MDA level was significantly higher in HN-1 group, and DRDE-30, DRDE-35, amifostine, NAC, melatonin, and STS protected it. Percutaneous administration of HN-2 significantly increased the MDA level on fourth day and DRDE-07, DRDE-30, melatonin, and STS significantly protected it. On eighth day also, the MDA level was significantly increased, and DRDE-07, DRDE-30, DRDE-35, amifostine, melatonin, and STS showed partial protection. Exposure to HN-3 also showed an increased lipid peroxidation in all groups on fourth day, except DRDE-30 and DRDE-35 groups. On eighth day, the MDA level was increased significantly and DRDE-30 gave significant protection. Sulfur mustard increased the level of MDA on fourth and eighth day. Amifostine, DRDE-07, and its analogues on fourth day as well as on eighth day significantly protected it (Figure 3).
The DNA damage following percutaneous administration of HN-1 was drastic on fourth as well as on eighth day. The antidote treatment did not show any appreciable change on fourth as well as eighth day. However, DRDE-30 and melatonin gave partial protection. HN-2 administration also showed significant increase in percentage DNA damage on fourth day and eighth day. DRDE-07, DRDE-30, DRDE-35, amifostine, NAC, melatonin, and STS showed partial protection on eighth day. There was a significant increase in the percentage DNA damage on fourth day and eighth day following HN-3 administration. However, DRDE-30, NAC, and melatonin gave partial protection on fourth day, and DRDE-07, DRDE-30, DRDE-35, and melatonin significantly protected it on eighth day. Sulfur mustard exposure also showed DNA damage on fourth as well as on eighth day. DRDE-07 and its analogues on fourth day and amifostine, DRDE-07, and its analogues on eighth day significantly protected it (Figure 4).
Discussion
The available evidences show that DNA is one of the major targets of sulfur and nitrogen mustards, but other factors may also modulate the final effect. Hypotheses vary from the release of proteases 15 to the ill-defined concept of “unbalanced growth” or “out of phase synthesis.” The consequences of many of the effects, for example, RNA alkylation and the function of DNA protein and RNA protein cross-links remain to be elucidated. Mustard agents are alkylating agents and their vesicant properties have been well documented, but the mechanism of systemic toxicity is not clearly defined. One of the documented mechanisms is alkylation of DNA by SM and nitrogen mustards, subsequently leading to DNA damage and cell death. 16 As suggested by Halliwell, 17 DNA strand breaks may be associated with oxidation of GSH and marked rise in intracellular calcium. In this study, we found significant DNA fragmentation in the mustard agent (HN-1, HN-2, HN-3, and SM) exposed animals, which were variably protected by DRDE-07 analogues. Thus, these analogues may be able to attenuate the deleterious consequences of alkylation.
A dose of 1.0 LD50 of each mustard agent (HN-1, HN-2, HN-3, and SM) was selected for the biochemical and histological studies. Generally, most animals survive until 7 days in case of SM and until 3 days in case of HN-1 and HN-2, and mortality occurs only thereafter. The decrease in the body weight is one good indication of the toxic effects of the mustards. Although not specific, there are few vital parameters that significantly change after the nitrogen mustard and SM toxicity. 7,10 In the current study, the body weight change, spleen weight, and hematological and biochemical variables such as WBC and RBC count, Hb level, reduced and GSSG, MDA level, and DNA fragmentation were taken as markers for evaluating the antidotal efficacy.
Reduced glutathione exerts cytoprotection by reacting with reactive oxygen species, restoration of damaged molecules via hydrogen donation, reduction of peroxidases, and maintaining protein thiols in their reduced state. 18 Oxidative stress is a well-known proposed mechanism of mustard agent–induced cytotoxicity, with a decrease in the level of GSH and an increase in MDA. 19 –21 Being electrophilic in nature, sulfur and nitrogen mustards have high affinity toward sulfydryl groups and deplete GSH in the body. Direct interaction of GSH with mustard agents is also a possibility. 16 The in vitro and in vivo studies show that glutathione level is increased in response to administration of amifostine, DRDE-07, and their analogues. 7,9,22 In the current study, DRDE-07 analogues gave partial protection on GSH against nitrogen mustards.
Lipophilicity is of prime importance for orally effective compounds. To be effective against mustard agents, compounds having optimum lipophilicity can penetrate the cell and protect it from mustard agent–induced alkylation. Here the prophylactic benefits of the analogues were related to their improved lipophilicity, which increased the penetration of analogues within the cell. To increase the lipophilicity, the phosphorothioate moiety of the amifostine has been replaced by S-aryl or S-alkyl. Amifostine is converted to its free thiol metabolite by the membrane-bound ALP for its cytoprotective action. 23 –25 DRDE-07, DRDE-30, and DRDE-35 do not have a phosphate group but still they are better than amifostine against mustard agent’s toxicity, probably by a different mechanism.
The best strategy to decrease the toxicity of the mustard agents is to reduce the available amount of mustard agents. For this purpose, scavengers such as the sulphydryl-containing molecules, NAC, amifostine, 7,26,27 DRDE-07 and its analogues, 7 and melatonin were used to trap mustards. DRDE-07 and its analogues were found to reduce both nitrogen mustards and SM toxicity.
The organic thiophosphate and its analogues, which are chemo- and radioprotective, 28 –30 were investigated as cytoprotective agents. In the current study, we found that amifostine, DRDE-07, and its analogues are mainly effective against SM but only partially effective against nitrogen mustards. Reports are available of the protection afforded by several classes of molecules against HN-2 also. 26 Sulfhydryl-containing molecule such as NAC, melatonin, and the radioprotector, WR-1065, reduce HN-2 toxicity mainly by direct extracellular interaction with HN-2. 29,31 These agents prevented metabolic disruption and sulfhydryl group depletion in both immediate and delayed cotreatment.
The antidotal activity of STS has also been shown in other animal models. Hatiboglu et al 32 have shown that intravenous (IV) infusion of STS in dogs has efficiently antagonized nitrogen mustard. These investigators have suggested that STS could directly neutralize the nitrogen mustard and thereby prevent systemic toxicity. Bonadonna and Karnofsky 33 have also described extensive animal and clinical studies using STS as a systemic antidote to HN-2 or its chlorimine picryl sulfonate derivative. They found that in rodents and rabbits 1 g/kg STS given IV afforded minimal protection. Our result shows that orally administered STS is less effective than orally administered DRDE-07 analogues. Sodium thiosulfate is the only recognized pretreatment for mustard agents but the dose required is too large and has to be administered by IV route. 3,34 When compared with DRDE-07 and its analogues, the protection offered by STS is much less. The beneficial effect of STS is expected to be due to its scavenging property, but the major problem is that it cannot enter the cell and thus cannot reverse the toxicity in the case of massive systemic intoxication. 16 However, DRDE-07 and its analogues are orally effective compounds and offer greater protection compared to STS. Moreover, these analogues have better lipophilicity and can enter the cell when given as pretreatment. Systemic toxicity studies in mice have also shown that STS was ineffective when the interval between HN-2 and STS was more than 15 minutes. 32 It appears from the current study that the pretreatment alone is very important for protection and repeated treatment may not have any effect.
Requirement of a common antidote for all the mustard agents is an ambition. DRDE-07 and few of its analogues such as DRDE-30 and DRDE-35 may prove as prototype compounds for the development of better oral prophylactic or therapeutic drug for mustard agents either alone or in combination. Overall, DRDE-30 (propyl analogue) followed by DRDE-35 (butyl analogue) are favored as safer and better compounds as their acute LD50 values are more than DRDE-07 and amifostine. 9
Footnotes
Acknowledgments
The authors are thankful to Dr S. J. S. Flora, Head of Pharmacology and Toxicology, DRDE, Gwalior, for providing all necessary facilities. Thanks are also due to Dr K. Ganesan and Dr Uma Pathak for providing nitrogen mustard, sulfur mustard, and all antidotes of DRDE-07 series. The technical help provided by Mr Satish is highly appreciated.
The authors declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.
