Abstract
This investigation was aimed to study the effect of 2,3-dimercapto-1-propanesulfonic acid (DMPS) on mercuric chloride (HgCl2)-induced alterations in urinary excretion of various carnitine fractions including free carnitine (FC), acylcarnitine (AC), and total carnitine (TC). Different groups of Wistar male rats were treated with HgCl2 at the doses of 0.1, 0.5, 1.0, 2.0, and 3.0 mg/kg body weight, and the animals were sacrificed at 24 hours following HgCl2 injection. A separate batch of animals received HgCl2 (2 mg/kg) with or without DMPS (100 mg/kg) and sacrificed at 24 or 48 hours after dosing. Administration of HgCl2 resulted in statistically significant and dose-dependent increase in the urinary excretion of FC, AC, and TC in rats. However, the ratio of urinary AC:FC was significantly decreased by HgCl2. Pretreatment with DMPS offered statistically significant protection against HgCl2-induced alterations in various urinary carnitine fractions in rats.
Introduction
Humans are exposed to various species of mercury including charged inorganic mercurous (Hg1+) and mercuric salts (Hg2+), neutral element (Hg0), and organic molecules (eg, methyl mercury). Exposure to mercury vapor and organic mercurials specifically affects the central nervous system, whereas kidney is the preferential target organ for inorganic mercury compounds such as mercuric chloride (HgCl2). 1,2 In general, mercury toxicity derives from the binding of mercury to sulfide groups that disrupt the proper functioning of sulfhydryl enzymes. 3 A positive relationship has been observed between mercury content and organ injury. 4
The chelating therapy with 2,3-dimercapto-1-propanesulfonic acid (DMPS), a tissue-permeable metal chelator, is commonly used as an antidote to heavy metal poisoning by rapidly increasing the urinary excretion of these toxicants. Torres-Alanís et al 5 have determined the clinical efficacy of DMPS on the urinary excretion of mercury in 10 men occupationally exposed to mercury; they observed statistically significant increase in urine mercury after DMPS treatment. The therapy with DMPS has been effectively used in 95 cases of chronic and acute intoxication with combination of mercury vapor, inorganic, and organic mercury, with rare side effects. 6 Zander et al 7 have observed a 6- to 7-fold increase in mercury excretion after application of DMPS, while the subjects with amalgam fillings excreted significantly more mercury before as well as after application of DMPS than subjects without amalgam fillings. Drasch et al 8 have concluded after urinalysis of 75 mercury-intoxicated volunteers that DMPS increases the renal excretion of both inorganic and organic mercury, the later to a lesser extent. 2,3-Dimercapto-1-propanesulfonic acid has been found to increase urinary mercury excretion and decrease mercury content in the rat brain after methyl mercury exposure. 9
Carnitine (3-hydroxy-4-N-trimethylammonium butyrate) is an essential cofactor in fatty acid metabolism. Carnitine transfers and regulates fatty acid transport across the inner mitochondrial membrane to be oxidized via β-oxidation. Above-normal urinary excretion of carnitine and acylcarnitine (AC) has been recognized as a biomarker of proximal renal tubular damage. 10,11 This study reports the effect of HgCl2 on urinary excretion of carnitine and AC in rats and its modification by DMPS treatment.
Materials and Methods
Animals
Adult Wistar male rats weighting 150 to 200 g were obtained from Animal Breeding Facility, College of Pharmacy, King Saud University, Riyadh, Saudi Arabia. The animals were kept in a temperature- (23 ± 2°C) and humidity- (60 ± 2%) controlled room and maintained on 12-hour light:dark cycle (light period: 7
Experimental Groups and Drugs Dosing
Two batches of animals were used to study (a) the effect of different doses of HgCl2 on urinary excretion of carnitine and AC and (b) the effect of DMPS on HgCl2-induced alterations in urinary carnitine and AC levels. For the first batch, 7 different groups (5 rats in each group) were studied: (1) control group (rats without any treatment), (2) placebo group (rats with vehicle treatment), (3-7) rats intraperitoneally (IP) injected with HgCl2 at the doses of 0.1, 0.5, 1.0, 2.0, and 3.0 mg/kg body weight, respectively. The animals were sacrificed at 24 hours following HgCl2 injection.
For the second batch, rats were divided into 5 groups of 10 animals each. The following groups were studied: (1) control; (2) placebo; (3) rats injected with a single IP dose of 100 mg/kg DMPS; (4) rats injected with a single IP dose of 100 mg/kg DMPS followed by a single dose of 2.0 mg/kg HgCl2, 1 hour after DMPS; this time gap between the 2 injections was intended to ensure the complete dissipation/absorption of DMPS before injecting HgCl2 and therefore avoiding any in situ drug interaction; and (5) rats injected with a single dose of 2.0 mg/kg HgCl2. Five rats from each group were sacrificed after 24 hours of the HgCl2 treatment and the remaining 5 were sacrificed after 48 hours.
All the animal experiments in this study were undertaken according to the guidelines of Canadian Council on Animal Care (CCAC) and approved by the Institutional Research and Ethical Committee of College of Science, King Saud University, Riyadh, Saudi Arabia.
Determination of Carnitine and AC
Urine samples were collected on dry ice, centrifuged, and stored at −10°C until analyzed. Aliquots (0.5 mL) of urine in duplicate were deproteinized with 0.5 mL of ice-cold 1.2 mol/L perchloric acid and allowed to stand at room temperature for 10 minutes. The sample was then centrifuged at 5000 g for 10 minutes. The pellet was washed by suspending it in 0.5 mL of 0.6 mol/L perchloric acid followed by centrifugation. The supernatants were used for estimation of free carnitine (FC) and total carnitine (TC) as described below. The concentration of AC was calculated by subtracting the levels of FC from TC.
An aliquot of supernatant was neutralized with 1 mol/L KOH to bring the pH to 6.5 to 7.0 (pH should never exceed 7.0). The neutralized samples were used to determine the concentration of FC as described in the next paragraph. For the estimation of TC (i.e., FC + AC), 0.5 mL of supernatant was treated with 1.0 mol/L KOH until the pH was alkaline (pH should never be less than 10.0), mixed, and allowed to stand at room temperature for 30 minutes to ensure the release of carnitine from its ester linkage of AC. After incubation, samples were neutralized with ice-cold 1.2 mol/L perchloric acid to bring the pH to 6.5 to 7.0 and analyzed as mentioned below.
The concentration of carnitine in the urine was determined as reported earlier. 12 Briefly, the assay mixture consisted of 10 μL of acetyl CoA (15 mmol/L), 10 μL of 5,5′-dithiobis (2-nitrobenzoic acid) (DTNB), and 975 μL of the neutralized extract. The reaction was initiated by adding 5 μL of carnitine acetyltransferase (0.9 U). The absorbance was measured at 412 nm.
Statistics
The statistical comparisons among various treatment groups were made by Dunnett multiple comparison test using Statistical Package for the Social Sciences (SPSS, Chicago, IL), version 10. P values less than .05 were considered statistically significant.
Results
Figure 1 shows the effect of various doses of HgCl2 on urinary excretion of FC, AC, and TC by rats sacrificed 24 hours after the treatment. Administration of HgCl2 resulted in statistically significant and dose-dependent increase in the excretion of FC, AC, and TC in rats (Figure 1). The medium dose of HgCl2 (2 mg/kg) was selected to study the effect of DMPS on HgCl2-induced alteration in urinary excretion of various carnitine fractions following 24 and 48 hours after drug treatment; these results are shown in Figure 2 . Pretreatment of rats with DMPS significantly reverses HgCl2-induced increases in FC, AC, and TC levels to near-normal levels (Figure 2). A statistically significant decrease in urinary AC:FC ratio by HgCl2 was also normalized by DMPS treatment (Figure 3 ).
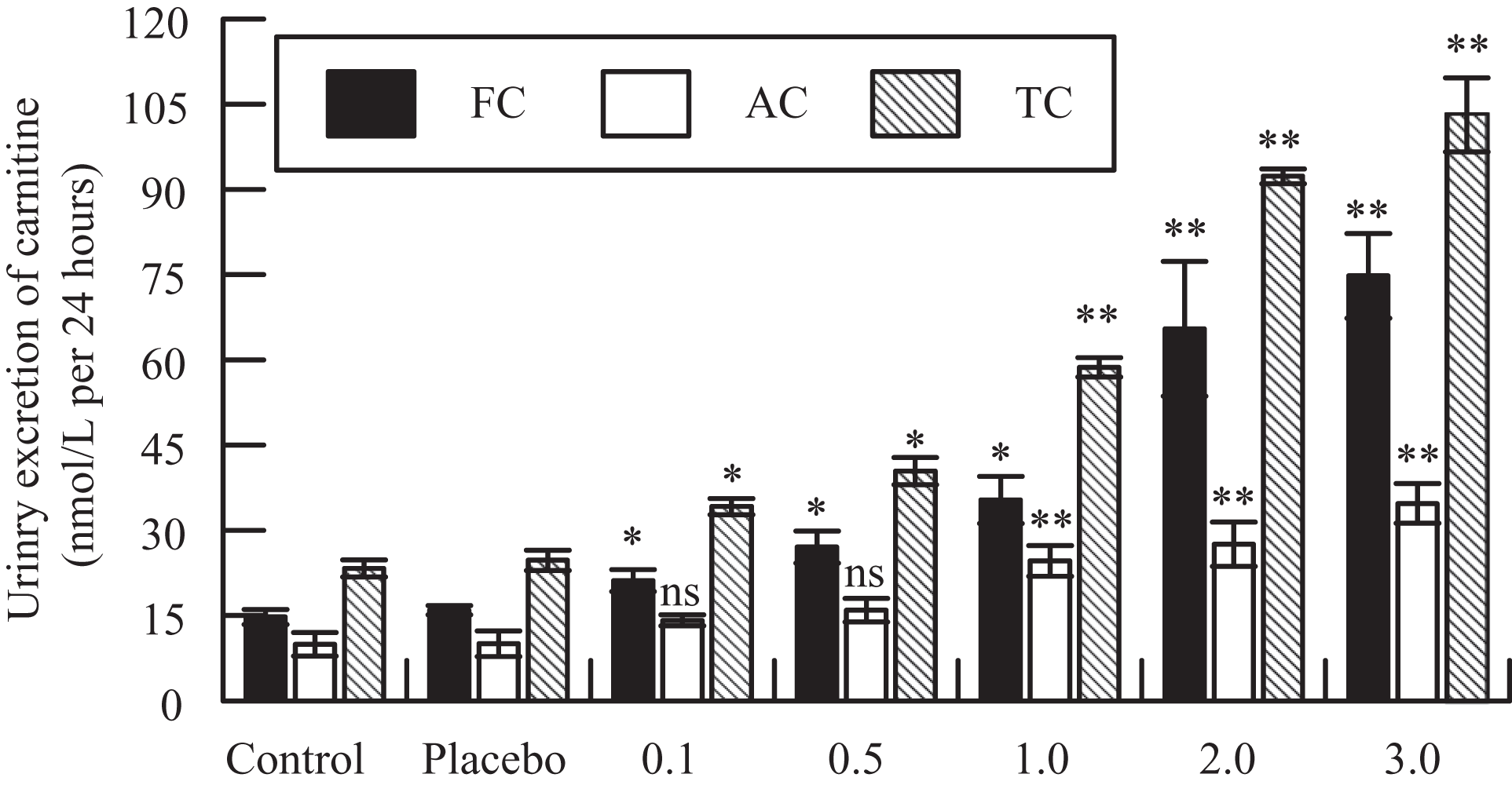
Effect of various doses of mercuric chloride (HgCl2) on urinary excretion of free carnitine (FC), acyl carnitine (AC), and total carnitine (TC). *P < .01 and **P < .001 versus control and placebo groups using Dunnett multiple comparison test. ns, not significantly different.
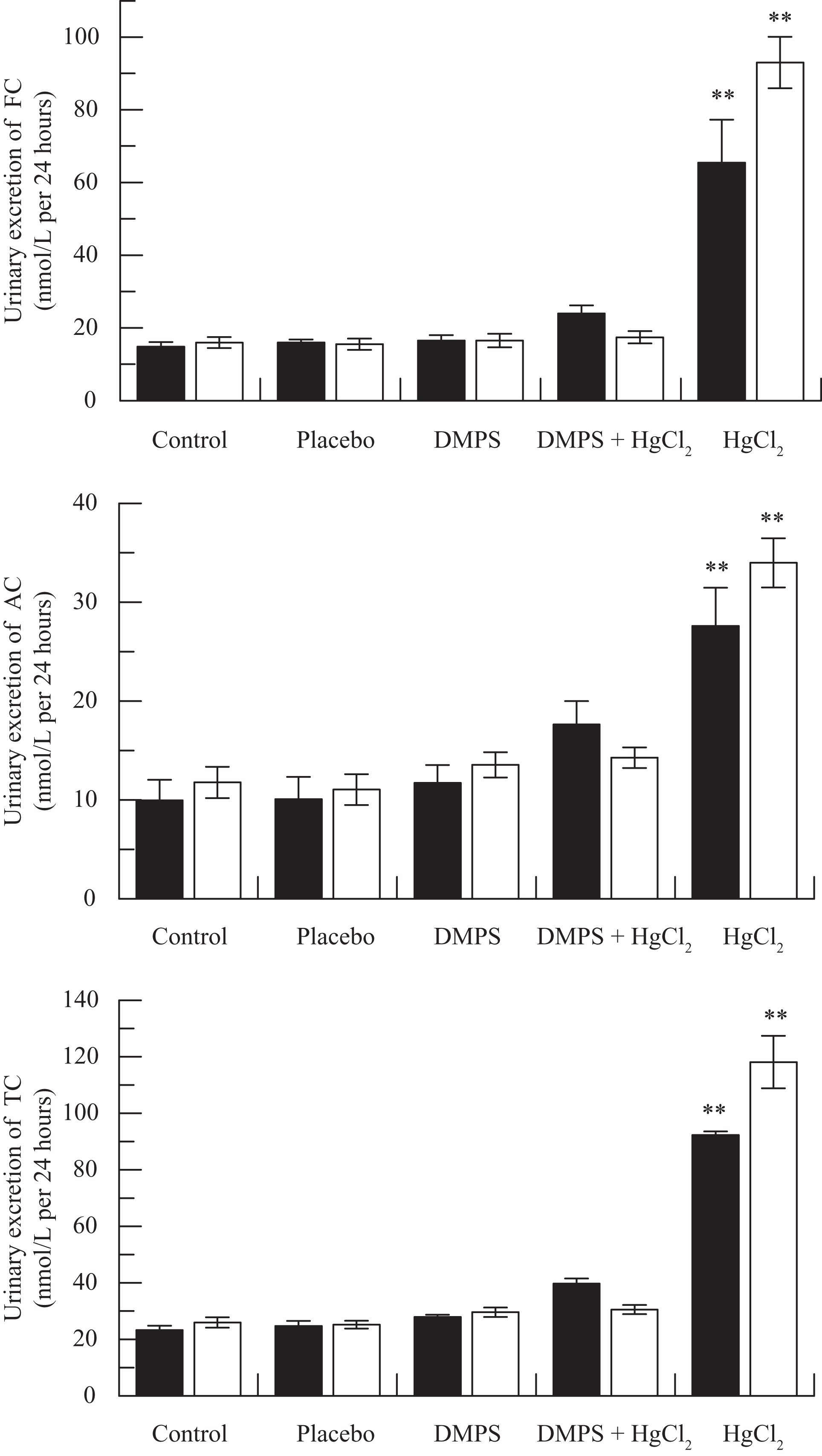
Effect of 2,3-dimercapto-1-propanesulfonic acid (DMPS) on mercuric chloride (HgCl2)-induced increase in urinary excretion of free carnitine (FC; top), acyl carnitine (AC; middle), and total carnitine (TC; bottom) at 24 (▪) and 48 (□) hours posttreatment. **P < .001 versus any of the remaining 4 groups using Dunnett multiple comparison test.
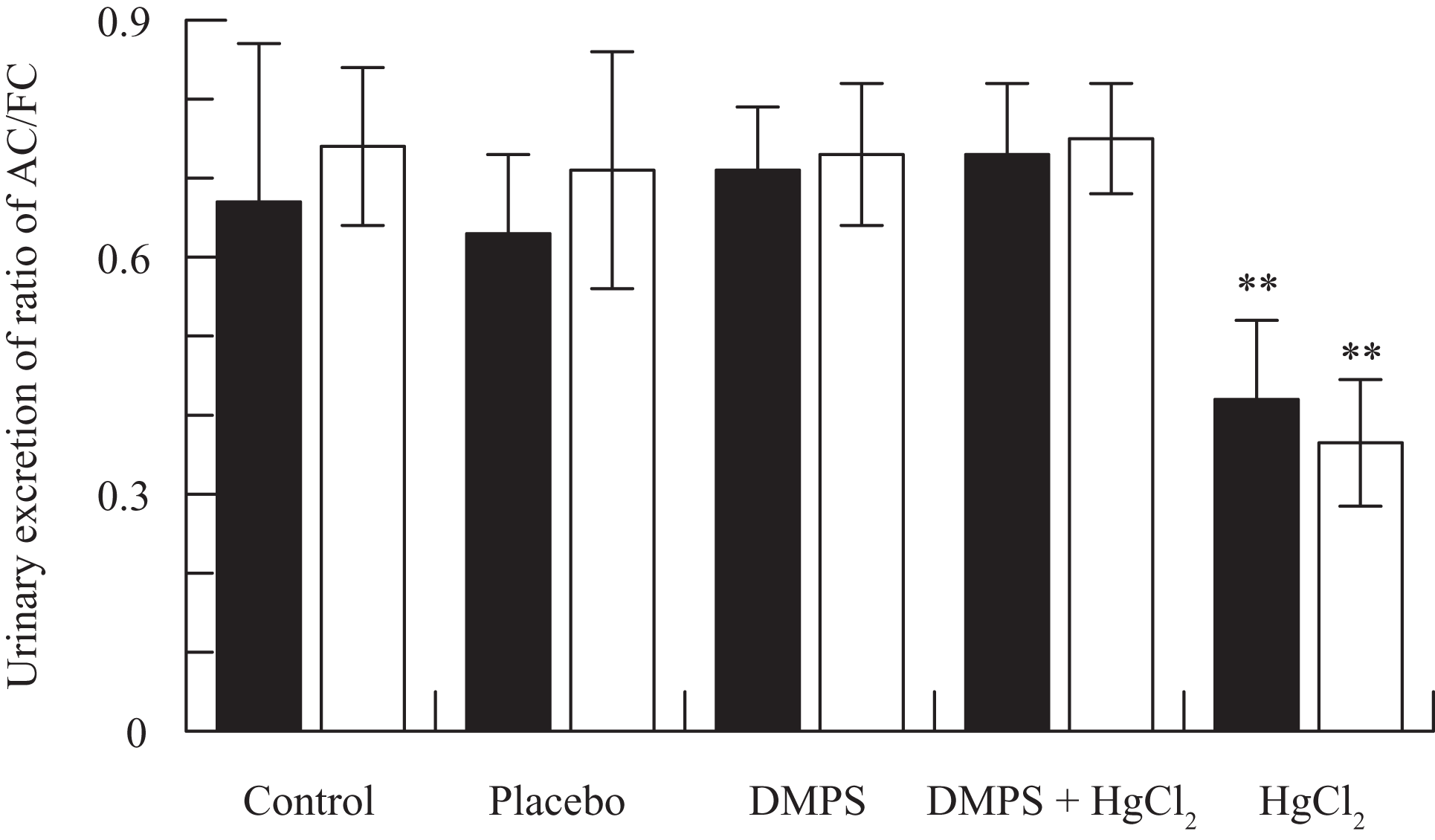
Effect of 2,3-dimercapto-1-propanesulfonic acid (DMPS) on mercuric chloride (HgCl2)-induced decrease in the ratio of acyl carnitine (AC)/free carnitine (FC) in urine at 24 (▪) and 48 (□) hours posttreatment. **P < .001 versus any of the remaining 4 groups using Dunnett multiple comparison test.
Discussion
The results of this study showed a statistically significant and dose-dependent increase in urinary excretion of various carnitine fractions by HgCl2 (Figure 1). The rise in urinary excretion of FC, AC, and TC in HgCl2-treated rats can be attributed to reduced reabsorption of carnitine due to renal proximal tubular damage by HgCl2. Similar doses of HgCl2 have already been shown to produce dose-dependent nephrotoxicity in rats. 13 Mercuric chloride can induce acute renal failure in rats by a single dosage while proximal tubules are the main renal mercury targets. Ultrastructural analysis has confirmed HgCl2-induced dose-dependent changes such as necrosis, apoptosis, nucleolar segregation, swollen mitochondria, vacuolization, and disrupted brush border within straight proximal tubules. 14,15 Although HgCl2 increased the urinary excretion of both FC and AC, the clearance of the former was much greater than the later as evident by a statistically significant decrease in the urinary ratio of AC:FC by HgCl2 administration (Figure 3). Carnitine is important for the transport of acyl groups across the inner mitochondrial membrane. By virtue, carnitine is not degraded in mammalian systems and is eliminated in urine in the forms of FC and AC. 16 Decreased ratio of AC to FC in urine could have resulted by increased use of fatty acids and/or disruption of carnitine transporters by HgCl2. A statistically significant decrease in urinary FC/AC may partly be attributed to increased lipid peroxidation by HgCl2 exposure. 17,18 However, there are some reports indicating that lipid peroxidation is not the primary toxic mechanism in acute nephrotoxicity induced by HgCl2 19 neither the redox enzymes appear to be an important determinant of HgCl2 toxicity. 20
This study demonstrates a near-normal reversal of HgCl2-induced increases in urinary excretion of various carnitine fractions as well as AC:FC ratio by DMPS, suggesting the beneficial effects of chelating therapy for counteracting mercurial toxicity. Several experimental studies have shown the protective effects of DMPS on arsenic-, 21 beryllium-, 22 cadmium-, 23 methyl mercury-, 24 vanadium-, 25 and zinc- 26 induced toxicities in animals. In addition to the metal-removing ability, the antioxidant properties of DMPS may play an important role in manifestation of its beneficial effect in metal intoxications. 27 Administration of DMPS has also been shown to reduce metal-induced excessive generation of free radicals and lipid peroxidation. 9,23 However, it is important to note that not all the antioxidants offer protection against mercury-induced nephrotoxicity. The antioxidant, lycopene, despite preventing lipid peroxidation, failed to counteract plasma creatinine levels and renal tubular necrosis induced by HgCl2 in rats. 28 Similarly, administration of the antioxidant selenium resulted in further enhancement of mercury concentration in the tissues while kidney showed the highest mercury deposits among the organs examined. 29 It has been suggested that besides the antioxidant property, the ability of an agent to reduce the mercury body burden is important to ensure protection against mercury nephrotoxicity. 30 Girardi and Elias 4 have emphasized an important role of sulfhydryls in detoxification and clearance of HgCl2 from various tissues. Treatment of mice with DMPS exerts beneficial effects on reversing methyl mercury–induced motor deficits and cerebellar damage in mice due to its capability of increasing the urinary excretion of mercury and thus removing mercury from brain tissues. 9 Moreover, carnitine supplementation has been shown to enhance the activity of mitochondrial antioxidant system and decrease the free radical-induced lipid peroxidation in rats. 31,32
In conclusion, the findings of this study clearly demonstrated that HgCl2 treatment significantly increased the urinary excretion of FC, AC, and TC in rats. However, the ratio of urinary AC/FC was significantly decreased by HgCl2. The chelating therapy with DMPS significantly protected the animals against HgCl2-induced urinary losses of carnitine and AC.
Footnotes
Acknowledgments
The authors thank King Abdulaziz City for Science and Technology, Riyadh, Saudi Arabia, and Vice Rectorate for Graduate Studies and Research, King Saud University, Riyadh, for financial support. We also acknowledge the support from the British Council, Saudi Arabia, for a Postdoctoral Summer Research Fellowship to one of us (ASA).
The authors declared no conflicts of interest with respect to the authorship and/or publication of this article.
King Abdulaziz City for Science and Technology, Riyadh; King Saud University, Riyadh, Saudi Arabia and British Council, Saudi Arabia.
