Abstract
This study investigated epigenetic (specifically, DNA methylation) changes and their impact on gene expression in testes induced by maternal exposure to Di-2-(ethylhexyl) phthalate (DEHP) in mice. Testicular dysgenesis syndrome was induced in fetuses and pups by maternal exposure to DEHP at 500 mg/kg/d, and testes were excised for analysis on gestation day (GD) 19 and postnatal days (PNDs) 3, 21, 56, and 90. High-performance liquid chromatography (HPLC) was performed to analyze DNA methylation status, and expression levels of the DNA methyltransferases were examined by quantitative real-time polymerase chain reaction (qPCR). Testis-specific gene, insulin-like hormone 3 (Insl3), and testosterone production were also detected. DEHP significantly increased DNA methylation levels on GD 19 and PND 3 (P < .05 and P < .05) but not on PNDs 21, 56, and 90. DEHP also significantly increased the expression of DNA methyltransferases. For DNA methyltransferase 1, the difference was not significant on PND 21, and DNA methyltransferase 3a and 3b returned to normal levels on PND 56. Fetal testes were a main target for DEHP as evidenced by a reduction in Insl3 expression and testosterone production. Effects of DEHP on Insl3 expression continued until PND 21. The DEHP-induced suppression of testosterone had not recovered on PND 56. Changes in DNA methylation may play an important role in abnormal testicular function caused by environmental factors such as maternal exposure to DEHP, which may be a mechanism of DEHP-mediated testicular toxicity.
Keywords
Adverse trends in male human reproductive health have been observed in many countries in the last few decades. Reproductive disorders like cryptorchidism, hypospadias, low sperm counts, and testicular cancer may be increasing in incidence. 1-3 According to Skakkebaek et al, 4 these interrelated disorders may be symptoms of a common condition, human testicular dysgenesis syndrome (TDS), which originates before birth. The increasing release of synthetic chemicals in the environment may underlie human TDS. Particularly phthalate esters, which are the most abundant produced plasticizers, are known to leach out from polyvinyl chloride (PVC) and disrupt androgen action. 5,6 Di-2-(ethylhexyl) phthalate (DEHP) is the most common phthalate for producing flexible PVC and is present in a wide variety of consumer products. 7 According to recent biomonitoring studies, exposure of the general population to DEHP is widespread and higher than previously assumed. 8-10 Interestingly, reproductive toxicity induced by perinatal exposure of experimental animals to DEHP has remarkable similarities with the symptoms of human TDS. 5,6,11 Like human TDS, the disturbances caused by DEHP are downstream consequences of altered Sertoli and Leydig cell functions during development and inhibit testosterone and insulin-like growth factor 3 (Insl3) production. 5,11,12 The mechanism of DEHP-mediated toxicity on the testis, however, is unclear. One possible mechanism is that DEHP metabolites bind to peroxisome proliferator-activated receptors (PPARs). 13 The action of DEHP on testis cannot be explained entirely by a PPAR-mediated pathway, because PPAR-knockout mice remain sensitive to phthalate-mediated reproductive toxicity. 14
Mammals contain numerous cell types, with the majority having identical genomic sequences. Genetic information is the sequence of DNA bases itself, whereas epigenetics encodes information without changing the DNA sequence. 15 Epigenetics involves multiple mechanisms such as DNA methylation, histone acetylation, and chromatin modification to control cell development and differentiation. Methylated CpGs in CpG islands of promoter areas repress gene expression, and methylation of genomic DNA is catalyzed by DNA methyltransferases (Dnmts) including Dnmt1, Dnmt3a, and Dnmt3b. 16-21 Epigenetic alterations can be caused by chemical substances, food, and environmental factors, particularly maternal environmental exposure to compounds during the reprogramming period. 22-24 DEHP can cause epigenetic changes, 25,26 which may be a mechanism of DEHP-mediated testicular toxicity.
Here, we analyzed DNA methylation density of testes by high-performance liquid chromatography (HPLC) 27 and determined the expression of Dnmts by quantitative real-time polymerase chain reaction (qPCR) in fetal testes (gestation day [GD] 19) and pup testes (postnatal days [PNDs] 3, 21, 56, and 90) after maternal exposure to DEHP. We also examined Insl3 and testosterone production.
Methods
Animals
Kunming mice purchased from Chongqing Medical University (Chongqing, China) were housed under controlled conditions at 24°C ± 1°C at 50% to 70% humidity and 12/12-hour light/dark cycles, with access to food and water ad libitum. 28 Female mice were housed overnight with male mice and were checked for vaginal plugs the following morning. After confirming the existence of plugs, noon that day was designated as GD 0.5. DEHP (Sigma-Aldrich, St. Louis, MO) was administered in corn oil on GD 12.5 with gastric tubes at 500 mg/kg/d until GD 18.5, as described previously. 29 Our previous studies show that Kunming mice exposed to DEHP had remarkable similarities with the symptoms of TDS, like cryptorchidism, hypospadias, and testicular cancer. Corn oil alone was administered to the control mice. On GD 19, half of the mice was sacrificed with CO2 prior to decapitation. Fetuses were rapidly removed from the uterus, and their sex was determined on the base of gonad morphology. Testes were excised under a microscope. The other half of the group of pregnant mice was retained as a nature delivery group. Male offspring were sacrificed with CO2 prior to decapitation on PNDs 3, 21, 56, and 90, and testes were excised as soon as possible. All testes were stored at –80°C until analysis. All studies were performed with the approval of the Experimental Animal Committee at Chongqing Medical University.
HPLC Screening for Genomic Methylation of Cytosines
High-molecular-weight genomic DNA was extracted using the DNeasy system (Qiagen, Valencia, Calif). Standard samples of 2′-deoxycytidine (dC), 5-methyl-2′-deoxycytidine (5mdC), 2'-deoxycytidine (dG), 2′-deoxyadenosine (dA), and 2′-Deoxythymidine (dT) were prepared at 100 μM. DNA or 5mdC-containing oligonucleotide digestion was performed as described. 30 Residual RNA was removed by alkaline hydrolysis with 0.5M NaOH at 37°C for 1 hour. After neutralization with HCl and DNA precipitation, 10 mg of each DNA sample was digested with 2 U of micrococcal nuclease (United States Biochemical, Cleveland, Ohio) and 1 mg of phosphodiesterase II (Roche, Basel, Switzerland) at 37°C for 3 hours in 10 mM CaCl2 and 20 mM succinate at pH 6.0 according to the protocol of Young and Smith. 31 For complete digestion to nucleosides, phosphate groups were removed by digestion with 20 U of alkaline phosphatase (Roche) containing 1 mM MgCl2, 1 mM ZnCl2, and 20 mM Tris (pH 8.0) for 16 hours at 37°C. Samples were lyophilized and stored at –20°C. 5mdC content was measured with a Beckmann Ultrasphere ODS column, 4.6 × 250 mm with 5-mm pore size (Beckmann Coulter, Krefeld, Germany) on a MerckeHitachi (Amsterdam, The Netherlands) HPLC system (L-6200 gradient pump, L-7250 autosampler, and L-7450 DA-detection system) at room temperature. After injection of the samples, a programmed linear gradient was applied from 100% solution A (5% acetonitrile with 0.04% acidic acid in H2O) to 100% solution B (100% methanol) over 35 minutes at 1 mL/min. The concentration of deoxyribonucleotides was assessed by ultraviolet (UV) spectrophotometry at 280 nm. 27 The ratio between 5mdC and dC was determined by the ratio between the elution peak areas. All samples were measured in triplicate.
RNA Isolation and First-Strand cDNA Synthesis
Total RNA was extracted separately from the testes using Trizol reagent (Bioteke, Beijing, China) according to the manufacturer’s instructions. The total RNA was checked for DNA contamination and integrity by UV spectrophotometer and 1.5% agarose gel electrophoresis. The total RNA was reverse-transcribed using a KeyGen reverse transcription kit (KeyGen, China). Reverse transcription was performed for 1 hour at 42°C in a final reaction volume of 20 μL containing 3 μg of total purified RNA, 2 μL of a 10× AMV reaction buffer, 1 μL of dNTPs (each 10 mM), 1 μL of 10 μM Oligo dT(18), 1 μL of DTT (200 mM), 0.5 μL of RNase inhibitor (40 U/μL), and 1 μL of AMV-reverse transcriptase (200 U/μl). The first-strand cDNA was stored at –20°C until use.
Quantitative Real-Time Polymerase Chain Reaction
qPCR was performed in a Bio-Rad CFX96 system using Platinum SYBR Green qPCR SuperMix UDG (Invitrogen, Beijing, China). Gene-specific primers were used to determine the relative expression levels of Dnmt1, Dnmt3a, and Dnmt3b. The primers for the Dnmts, Insl3, and β-actin used in this study were designated elsewhere (Table 1). All qPCR reactions were performed in a final volume of 25 μL containing 2.5 μL of 10× SYBR Green PCR Master, 300 nM of each gene specific primer, 50 ng of cDNA, and sterile deionized water. The standard cycling condition was 95°C for 10 minutes, followed by 40 cycles of 95 °C for 10 s and 60 °C for 1 minute. The comparative threshold cycles (Ct) values for the Dnmts were normalized to β-actin and compared with a calibrator using the 2-ΔΔCt method. 32 qPCR was performed in triplicate to ensure quantitative accuracy. Final results are expressed in relation to the reference gene (β-actin). In all cases, the reactions were performed 3 times using the same 3 independent samples. Data are presented as the mean ± standard error of measurement.
Quantitative Real-Time PCR Primers for DNA Methyltransferases and β-Actin
Testicular and Serum Testosterone Analysis
Fetal (GD 19) and pup (PND 3) testes were placed in a 12×75-mm glass test tube with 100 µL of distilled water. One milliliter of ethyl ether (99% pure) was added and the testis was crushed using a plastic pestle. Testicular steroids were extracted from the testes of control and DEHP-exposed pups (GD 19, n = 10 control and n = 10 DEHP; PND 3, n = 10 control and n = 10 DEHP), and testosterone concentrations were measured by a tritium-based radioimmunoassay validated for use with murine antiserum, as described. 33
Serum testosterone (PNDs 21, 56, and 90) was measured by a previously described tritium-based radioimmunoassay validated for use with murine antiserum. 34 The covariance of intra-assay and interassay for testosterone was less than 15%.
Statistical Analysis
Data are expressed as the mean ± standard error of measurement. Statistical analysis was performed with Student’s t test when the distribution was normal or the Mann-Whitney U test when the distribution was not normal. In serial analyses, significance was determined by 1-way analysis of variance followed by the Scheffé test. A P value of < .05 was considered statistically significant.
Results
HPLC
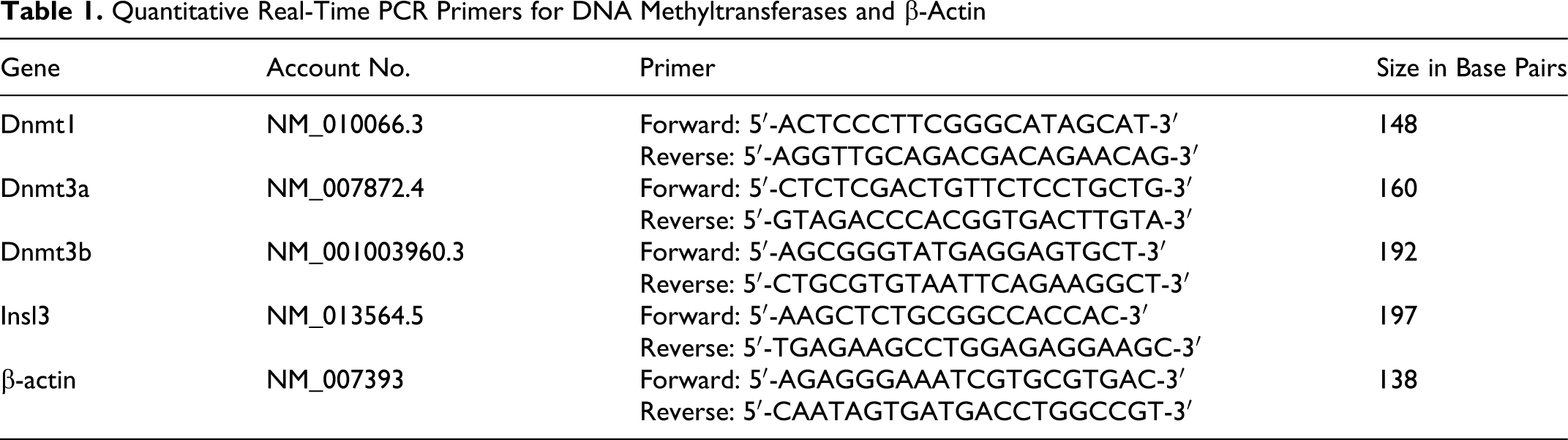
Measurement of the content of 5mdC is a useful parameter for the understanding of not only cellular homeostasis but also the interplay between genome-wide alterations in DNA methylation and their effect on genomic stability and gene-specific alterations in epigenetic regulation. 35 Concentrations of 2′-deoxycytidine (dC) and 5-methyl-2′-deoxycytidine (5mdC) were detected by HPLC and used to evaluate methylation levels (C5mdC / [C5mdC + CdC]) (Figure 1). The level of methylation in control mice was about 3.35% but in DEHP-exposed mice was 3.82% on GD 19. Compared with control, DEHP significantly increased methylation level of testes (P < .05). On PND 3, the methylation level of DEHP-exposed mice was still significantly higher than that in control (P < .05). But on PNDs 21, 56, and 90, there was no significant difference between control and DEHP-exposed mice.
(A) HPLC results of standard samples consisting of 2′-deoxycytidine (dC), 5-methyl-2′-deoxycytidine (5mdC), 2′-deoxyguanosine (dG), 2′-deoxyadenosine (dA), and 2′-Deoxythymidine (dT) at 100 M over 35 minutes at 1 m/min. (B) HPLC analysis of a representative testicular DNA sample. (C) HPLC analysis of changes in DNA methylation for DNA extracted from testes on gestation day (GD) 19 and postnatal days (PNDs) 3, 21, 56, and 90. DEHP treatment significantly increased DNA methylation. n = 30, *P < .05.
Quantitative Real-Time PCR
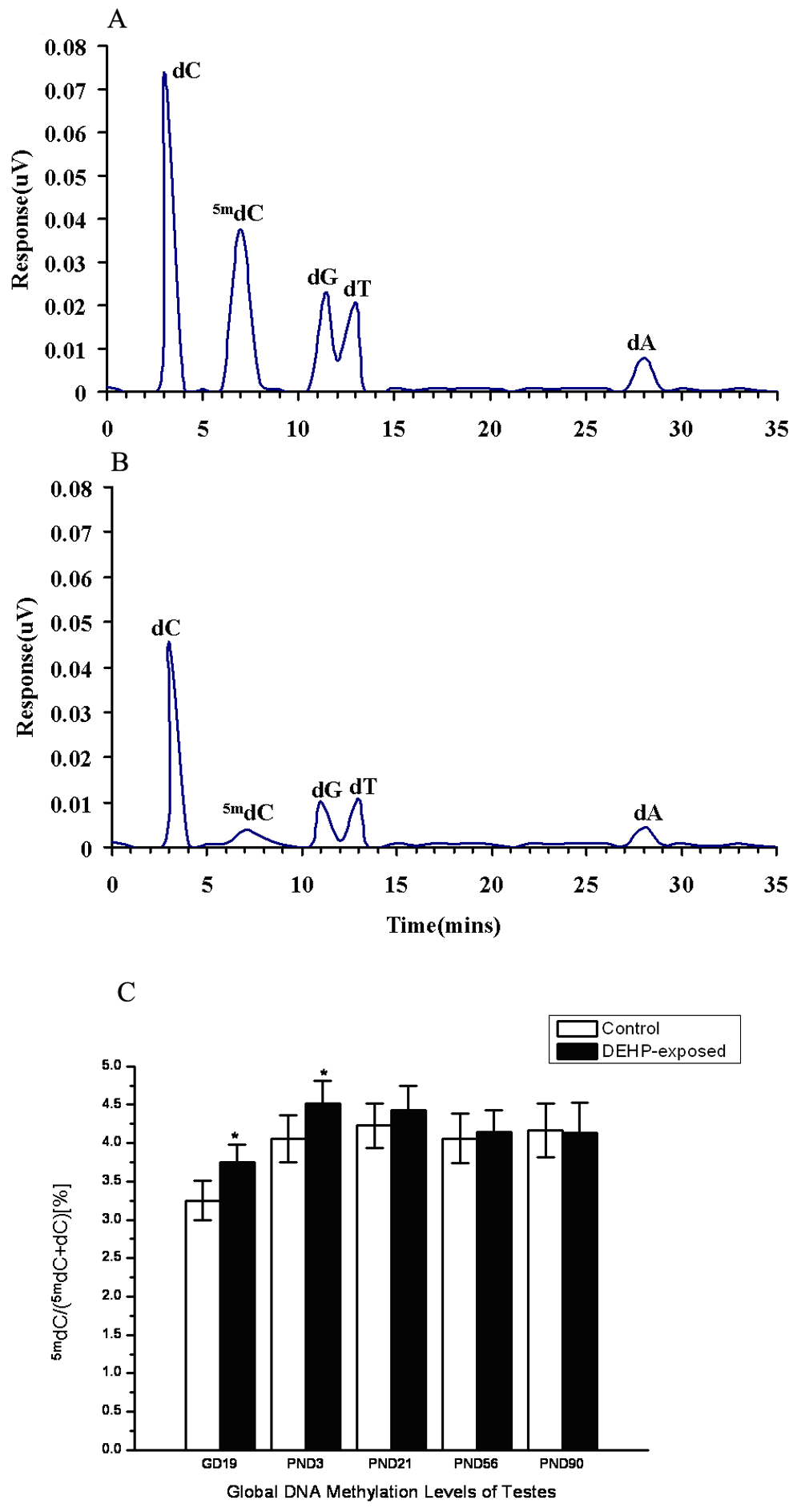
We then measured levels of the maintenance DNA methyltransferase, Dnmt1, and the de novo DNA methyltransferases, Dnmt3a and Dnmt3b (Figure 2). Dnmt1 is the main cellular enzyme responsible for the maintenance of DNA methylation patterns in somatic mammalian cells, and disruption in its activity and/or expression may lead to alterations in DNA methylation. 36 Therefore, we assessed the effect of exposure to DEHP on the level of Dnmt1 in mice testes. Figure 2 shows that the mRNA expression level of Dnmt1 was about 5-fold greater in DEHP-exposed mice than that in controls, which is a significant difference (GD 19, P < .05; PND 3, P < .05). It is known that Dnmt3a creates hemimethylated target sites during DNA methylation. 37 In the control group, the expression of Dnmt3a was not detected. However, DEHP treatment significantly increased Dnmt3a (GD 19, P < .01; PND 3, P < .01; PND 21, P < .01). Dnmt3b in particular is known to be required for de novo methylation of specific genomic regions. Although Dnmt3b was detected in both groups, the expression in DEHP-exposed mice was higher. These differences are significant (GD 19, P < .05; PND 3, P < .01; PND 21, P < .01).
Effect of DEHP on the expression DNA methyltransferases (Dnmts). Quantitative real-time PCR analyses Dnmts mRNA of from control to DEHP-exposed fetuses on gestation day (GD) 19 and postnatal days (PNDs) 3, 21, 56, and 90. Results are expressed in relation to the reference gene (β-actin). (A) Dnmt1, (B) Dnmt3a, (C) Dnmt3b. Error bars represent the mean ± standard error of measurement from 3 separate experiments. *P < .05 and ** P < .01, statistically significant differences from control.
Insl3 and Testicular Testosterone Production
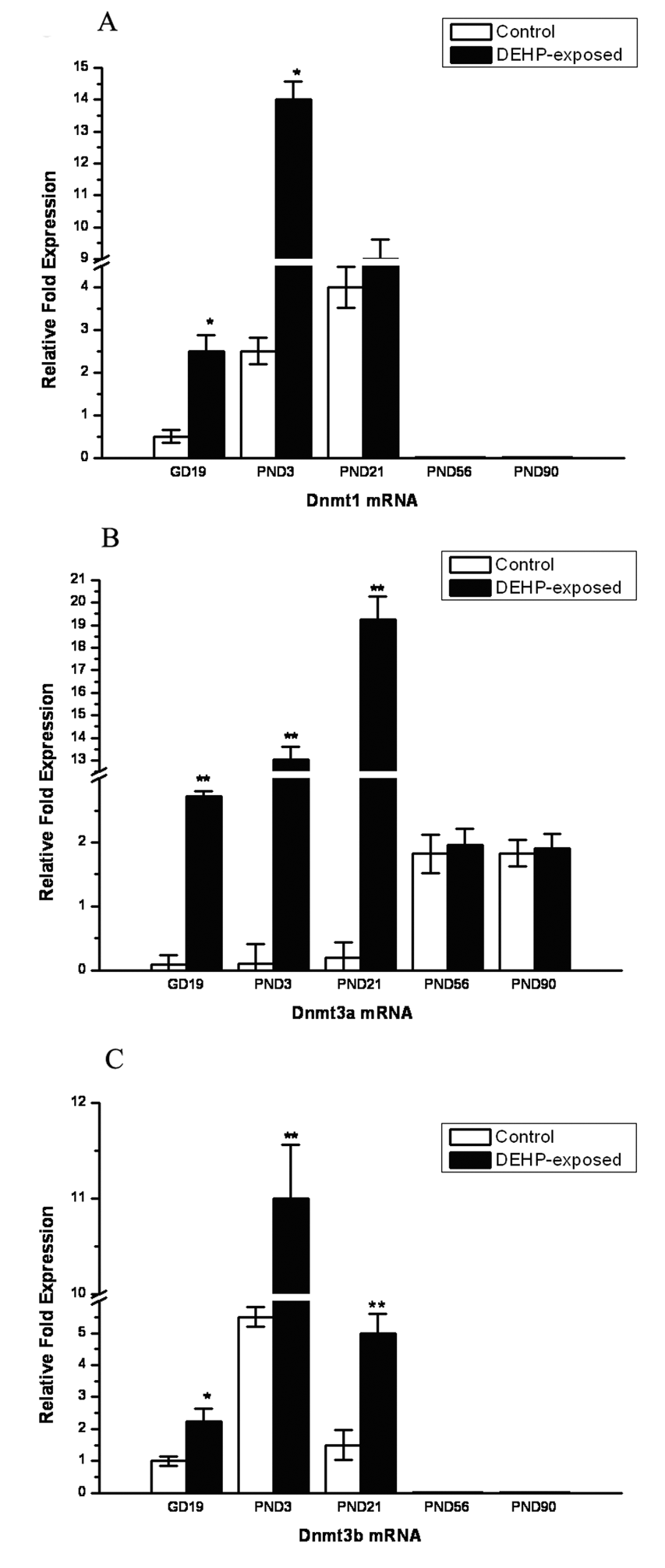
Fetal and postnatal testes from male offspring were examined for gene expression of Insl3. As shown in Figure 3A, Insl3 mRNA levels were significantly decreased in DEHP-exposed mice as determined by qPCR (GD 19, P < .05; PND 21, P < .01). The levels of testosterone in the extract of testicular tissue and serum were also detected. As shown in Figure 3B, testicular testosterone production was significantly lower in the DEHP-exposed mice compared with control (GD 19, P < .05; PND 3, P < .05; PND 21, P < .05; PND 56, P < .05).
Effect of DEHP on the expression levels of Insl3 and testicular testosterone (T) production. (A) Insl3 mRNA levels were determined by quantitative real-time polymerase chain reaction on gestation day (GD) 19 and postnatal days (PNDs) 3, 21, 56, and 90. In contrast to control, the expression levels of Insl3 in DEHP-exposed mice were reduced significantly on GD 19 and PND 21. (B) Levels of testicular T production (ng/testis) between control and DEHP-exposed male fetuses, which were significantly lower in the DEHP-exposed fetuses compared with control on GD 19 and PND 3. Serum T level was also significantly reduced by DEHP on PND 21 and PND 56. *P < .05, statistically significant differences from control.
Discussion
After the complete sequencing of the human genome in the 1990s, the regulatory mechanisms of epigenetics became an area of interest. Epigenetics is now recognized as a major mechanism involved in the development and progression of human diseases such as cancer. One mechanism of epigenetics is DNA methylation, a process by which methyl groups are added to the base of cytosine in DNA, usually suppressing gene expression. In most studies, increased DNA methylation is associated with gene silencing, and decreased methylation is associated with gene activation. In mammals, DNA methylation plays an important role in genomic imprinting and X-chromosome inactivation 38-40 and is involved in the expression of tissue-specific genes. In recent years, the importance of DNA methylation in normal development has become evident. Studies based on knockout mouse models for the 3 DNA methyltransferases (Dnmt1, Dnmt3a, and Dnmt3b) have demonstrated that the lack of any of these 3 enzyme activities in the mouse embryo results in embryonic or perinatal lethality, underscoring the essential role of DNA methylation in normal developmental processes. 16,41 What’s more, epigenetic alterations can be caused by chemical substances, food, and environmental factors, 22-24 particularly maternal environmental exposure to compounds such as environmental endocrine disruptors during the reprogramming period. Reprogramming refers to erasure and remodeling of epigenetic marks, such as DNA methylation, during mammalian development and is likely to have a crucial role in establishing nuclear totipotency in normal development. 42 A handful of reports have implicated environmental endocrine disruptors in epigenetic programming and DNA methylation. 43-46
Levels of human exposure to phthalates were estimated from the metabolite. Silva et al 47 measured 10 phthalate metabolites in 54 amniotic fluid samples collected during routine amniocentesis. The investigators found measurable phthalate metabolites in human amniotic fluid samples, suggesting that in utero exposure to phthalates occurs (ie, the detection of phthalate metabolites in human amniotic fluid demonstrates the presence of phthalates in the human fetal environment). In another recent study, phthalate monoester levels were measured in amniotic fluid (taken during amniocentesis) and urine samples of 65 pregnant women from Taiwan. 48 Their data clearly showed that in utero exposure to phthalates in general has adverse effects on the fetus. Wittassek et al 49 collected 11 amniotic fluid samples of pregnant women from Germany. The results from this study suggested that several phthalates (including DEHP) or their metabolites reach the human fetus and might be able to affect fetal health. In a recent case-control study, an increase in the risks of hypospadias was found among offspring of mothers occupationally exposed during pregnancy to hairsprays and, presumably, to phthalates in the exposure matrix. 50 Studies in human populations reviewed by Matsumoto et al 51 are useful for showing the association between phthalates exposure and health effects in humans. Although the current method of estimating phthalates exposure is inconsistent and studies on health effects of phthalates in humans have been controversial, animal studies remain necessary for risk assessment of chemicals including phthalates. Our previous studies show that Kunming mice exposed to 500 mg/kg/d DEHP had remarkable similarities with the symptoms of TDS, like cryptorchidism, hypospadias, and testicular cancer. 52
DEHP, as an environmental endocrine disruptor, impairs testicular function and induces reproductive tract abnormalities in male offspring. 12,29,53 Analysis of gene expression in testes following in utero exposure to phthalate esters has identified a number of potential mechanisms for phthalate-acting testicular toxicity. Phthalate toxicity may involve the peroxisome proliferator. 54,55 Screening assays exist for environmental endocrine disruptors such as DEHP. 56-58 However, these assays cannot completely explain how phthalate decreases gene expression globally, including signal transduction (Insl3 and Insig-1), transcription factors (Nr4a1, Dax-1, and Tcf1), lipid transport (Scarb1 and Star), cytoskeleton (Prkcbp1), and the steroidogenic pathway (Cyp11a1, Hsd3b1, and Cyp17a1). 59 Phthalate toxicity may also involve androgen antagonist, which blocks the ability of testosterone to bind to its receptor, resulting in alterations in androgen-dependent tissues. Mixtures of phthalate esters with one another and with other anti-androgenic compounds exhibit cumulative, largely dose-additive effects on male reproductive tract development when administered during sexual differentiation in utero, 60 although androgen antagonists and/or inhibitors of testicular testosterone production did not influence insl3 mRNA levels. 61 Testosterone and Insl3 are produced by the Leydig cells of the testis, which have an important role in testicular descent. It is generally accepted that testosterone acts on both the gubernaculum and the cranial suspensory ligament, which are the major mediators of the inguinoscrotal phase of testis descent. Insl3 is highly expressed in the fetal testis, which is the critical hormone responsible for early-stage descent of the testis from the abdominal to inguinal position. According to Howdeshell et al, 60 exposure to reproductive toxicant phthalates in utero alters the testicular hormones involved in Leydig cell migration and maturation, resulting less testosterone and insl3. Here, maternal DEHP treatment at 500 mg/kg/d from GD 12.5 to GD 18.5 caused a reduction in testosterone production and reduced the expression of Insl3. DEHP, as an environmental endocrine disruptor, can act on gene expression, but the mechanism of DEHP-mediated toxicity is still unclear. In fact, DEHP may cause epigenetic changes via DNA methylation and alter gene expression. 25,26 However, these changes are certainly complex and probably affect several candidate genes.
Here, we examined the relationship between epigenetics and DEHP-induced testicular toxicity by determining (1) changes in DNA methylation, (2) Dnmts expression levels, and (3) the possible role of DNA methylation and demethylation in murine TDS.
The pattern of DNA methylation serves as an epigenetic mark in general leading to a repression of gene expression. 62,63 The pattern of DNA methylation is used to memorize developmental decisions of the cell 64 and to control monoallelic expression of genes. De novo methylation of DNA is most prevalent during embryogenesis, where the methylation is restored after an almost complete demethylation of the genome that takes place during the first cleavage divisions. 65 In addition, de novo methylation can also occur later in development and even in adult cells to silence acquired proviral DNA or to alter the developmental program of the cell. In contrast to a study by Pogribny et al, 25 we observed that the extent of 5mdC in DEHP-exposed mice was higher than in controls. Whether the level of methylation is higher or lower, the resultant alteration of methylation patterns can have profound influence on cellular function and gene expression, which may be a mechanistic link to testicular toxicity.
The level of DNA methylation and expression of DNA methyltransferases were recovered on PND 21. We propose 3 possible explanations: the first is that DEHP has a short half-life and is excreted quickly from the body. 66,67 After birth, offspring were not exposed to DEHP, and the toxicity of DEHP in testes became less and less. Second, the possibility exists that the recovery process is associated with germ cell. In rodents, there are 2 populations of Leydig cells in the fetal testis: fetal Leydig cells, which involute after birth, with some persisting in the adult testis; and stem Leydig cells, which can become adult Leydig cells and are present during the entire life span. 68 Third, unidentified enzymes of DNA demethylation may exist. Although Dnmts catalyze DNA methylation, the mechanism of DNA demethylation is still unclear. The DNA demethylation can be active or passive or a combination of both. Some candidate enzymes for active DNA demethylation have been reported, none of which has gained wide approval. Meanwhile, passive DNA demethylation occurs by inhibition or lack of maintenance of Dnmts throughout cycles of replication.
Conclusion
DEHP-induced changes in DNA methylation, especially within CpG islands, may influence the development of the male reproductive tract. Epigenetic change may be one possible mechanism of DEHP-mediated testicular toxicity. Determining the role of epigenetic effects in TDS, such as cryptorchidism, requires identification of tissue-specific genes with changes in DNA methylation. This report suggests an epigenetic contribution to the male reproductive tract. Future studies on the cause of congenital anomalies such as TDS will involve genetic and epigenetic components.
