Abstract
Moringaceae, which belongs to the Moringa oleifera Lam. family, is a well-known herb used in Asian medicine as an antiallergic drug. In the present study, the efficacy of the n-butanol extract of the seeds of the plant (MONB) is examined against ovalbumin-induced airway inflammation in guinea pigs. The test drugs (MONB or dexamethasone) are administered orally prior to challenge with aerosolized 0.5% ovalbumin. During the experimental period, bronchoconstriction tests are performed, and lung function parameters are measured. The blood and bronchoalveolar lavage fluid are collected to assess cellular content, and serum is used for cytokine (tumor necrosis factor-α, interleukin-4, and interleukin-6) assays. Histamine assays of lung tissue are performed using lung tissue homogenate. The results suggest that in ovalbumin-sensitized model control animals, tidal volume is decreased, respiration rate is increased, and both the total and differential cell counts in blood and bronchoalveolar lavage fluid are increased significantly compared with nonsensitized controls. MONB treatment shows improvement in all parameters except bronchoalveolar lavage tumor necrosis factor-α and interleukin-4. Moreover, MONB treatment demonstrates protection against acetylcholine-induced bronchoconstriction and airway inflammation. These results indicate that MONB has an inhibitory effect on airway inflammation. Thus, MONB possesses an antiasthmatic property through modulation of the relationship between Th1/Th2 cytokine imbalances.
Acute or chronic asthma is usually associated with hypersensitivity to specific allergens in individuals who have elevated serum immunoglobulin levels. 1 This “atopic” or “allergic” asthma accounts for approximately 90% of cases in most surveys worldwide. 2 Asthma is characterized by airway inflammation, excess reversible constriction of airway smooth muscles, and airway hyperresponsiveness to a wide variety of spasmogens. 3 Pathophysiological features of asthma show infiltration of inflammatory cells like eosinophils, 4 neutrophils, 5 lymphocytes, 6 and macrophages/monocytes. 6 Many mediators derived from these inflammatory cells have been implicated in asthma pathophysiology viz histamine, 7 cytokines, 8 leukotrienes, prostaglandins, and thromboxanes. 9
Several studies have demonstrated elevated levels of histamine in the plasma of asthma patients 10 ; similar effects have been noted in the plasma 11 and lung tissues 12 of guinea pigs. Many cytokines have been detected in increased quantity in bronchial biopsies from asthmatic subjects. 13 Specifically, elevated levels of tumor necrosis factor (TNF)-α, 14 interleukin (IL)-4, 15 and IL-6 16 have been noted in the airways of asthmatic patients. Furthermore, recent studies have shown the presence of these mediators in bronchoalveolar (BAL) lavage fluid and urine of asthmatic patients after allergen challenge. 17
Moringa oleifera Lam. belongs to the family Moringaceae (genus Moringa). It is a small, deciduous tree of 2.5 to 10 m in height. The plant has been well positioned in Ayurvedic, Unani, and even allopathic systems of medicine. 18 Numerous varieties of M oleifera have been developed to meet the tastes of local populations. 19 The medicinal values of the seeds and the different parts of the plant have long been recognized in folklore medicine. 20 The seeds of the plant have been used as a purgative, antipyretic, and anti-inflammatory agent. 21 We have reported on the antiarthritic 22 activity of the ethanolic extract of these seeds in rats. Guevara et al 23 noted the antigenotoxic activity of these extracts. Ayurvedic practitioners have administered the seed extract intranasally in diseases like rhinitis, and the dried seeds were used as an antiallergic medication. 24 Treatment with the plant is also reported to elicit good clinical responses in children suffering from upper respiratory tract infections as well as skin infections. It has been reported that one of the alkaloids, Moringine, which relaxes bronchioles from the plant closely, resembles ephedrine in action and has been useful in the treatment of asthma. 25
Our previous preliminary clinical studies on dried seed powder of M oleifera showed a decrease in severity of asthma symptoms and simultaneous improvement in peak expiratory flow rate. 26,27 In preclinical studies, we also investigated the activity of M oleifera ethanolic extract in chemical (ie, toluene diisocyanate)–induced immune-mediated inflammatory responses in rats 28 and ovalbumin-induced chronic airway inflammation in guinea pigs. 29 We established that the extract inhibited the activity of cytokines and subsequently prevented eosinophilia and neutrophilia. Furthermore, to determine the constituents responsible for antiasthmatic activity of the extract, we fractionated the ethanolic extract using n-butanol solvent. To elucidate the mechanism of action of antiasthmatic drugs on human allergic asthma, ovalbumin-sensitized guinea pigs have been preferred as an animal model. 30 In this context, in the present study we have selected this model to reveal the potential treatment of immune-mediated airway inflammation in guinea pigs with an herbal extract.
Materials and Methods
Plant Material and Extract Preparation
Seeds of M oleifera were obtained from a commercial supplier of Ahmedabad and were identified and authenticated by Department of Pharmacognosy, L. M. College of Pharmacy, Ahmedabad, India. Voucher specimens were deposited at the Department of Pharmacognosy, Ahmedabad. The coarse powder (500 g) of the dried seeds was defatted using petroleum ether (60°C-80°C) and then exhaustively extracted using 95% ethanol (2 L) in a Soxhlet extractor at 55°C for 6 hours. The obtained ethanolic extract was subjected to fractionation using 300 mL (150×2) of n-butanol for 48 hours (by intermittent shaking) in a separatory funnel. The fraction obtained was concentrated under reduced pressure, and residual solvent in the fraction was evaporated in a fume cupboard at controlled temperature (40°C). The freshly prepared solution of n-butanol fraction (in 0.2% agar solution) was used for the further treatment of animals.
Phytochemical Analysis
Preliminary phytochemical studies of the n-butanol fraction was performed for alkaloids, flavanoids, saponins, glycosides, phenols, steroids, tannins, and terpenoids according to published standard methods. 31 Furthermore, the fraction was subjected for quantitative estimation of known marker compounds present in the plant M oleifera viz ascorbic, quercetin, benzylisothiocyanate, glycerol-1-(9-octadecanoate), and β-sitosterol by high-performance thin layer chromatography (HPTLC) and high-performance liquid chromatography (HPLC) analysis.
Drugs and Chemicals
All the solvents and chemicals were obtained from S.D. Fine Chemicals Private Limited (Mumbai, India) and were of analytical grade. O-phthalaldehyde (OPT), acetylcholine (ACh), histamine, ovalbumin (OVA), and dexamethasone were obtained from Sigma-Aldrich (St. Louis, Mo). Enzyme-linked immunosorbent assay (ELISA) kits for TNF-α (BD Biosciences, San Diego, Calif), IL-4 (Immunotech, Marseille Cedex, France), and IL-6 (R & D Systems, Minneapolis, Minn) were purchased for cytokine assay.
Animal Husbandry
Specific pathogen-free Dunkin-Hartley guinea pigs (500-700 g) of either sex were housed in a room maintained at 22°C ± 1°C with a relative humidity of 60% ± 5% and a 12-hour light–dark cycle. The animals were housed in stainless steel cages, where they were allowed to acclimatize for a week prior to experiment. They had free access to a standard pellet diet (Maharashtra Chakan Oil Mills Ltd., India), and 0.01% vitamin C solution was provided ad libitum. All experiments were carried out with strict adherence to ethical guidelines and were conducted as per approved protocol by the Institutional Animal Ethics Committee and as per Indian norms laid down by the Committee for the Purpose of Control and Supervision of Experiments on Animals, New Delhi. Throughout the entire study period, the animals were monitored for growth, health status, and food intake capacity to be certain that they were healthy.
Sensitization and Treatment of Animals
Guinea pigs used in the study were divided into 4 different groups of 4 animals each: group I, nonsensitized controls received 0.2% agar solution; group II (model control group), OVA sensitized and then received 0.2% agar solution orally (vehicle for dexamethasone [DXM] and MONB treatments); group III, OVA sensitized and then received DXM (2.5 mg/kg body weight orally) as reference standard; group IV, OVA sensitized and then received MONB (n-butanol fraction of ethanolic extract of seeds of M oleifera; 100 mg/kg orally). All animals (except group I) were sensitized by the method of Duan et al 32 using a subcutaneous injection of 100 μg of ovalbumin (which had been adsorbed onto 100 mg of aluminum hydroxide in saline) on day 0 as the first sensitization. Boosting was then carried out using the same dose of antigen 2 weeks later (ie, on day 14). The daily doses of drug or vehicle were initiated on day 18 and continued until day 29 by oral route.
Ovalbumin Exposure
On days 18 through 29, 2.5 hours after receiving their drug or vehicle treatment, the animals were challenged with 0.5% of aerosolized OVA for 10 minutes. For the challenge, conscious animals were placed into a plastic circular chamber (diameter = 70 cm, height = 40 cm) connected to a nebulizer (CX4-Omron Healthcare, Kyoto, Japan). Animals in the nonsensitized group (group I) were exposed to aerosolized saline using the same protocol.
Lung Function and Bronchoconstriction Test
On days 18, 21, 24, and 29, tidal volume and respiration rate of animals were measured after 2 hours of 10 minutes of OVA exposure using a data acquisition system (MP-35, Biopac Systems, Santa Barbara, Calif). Immediately after measurement of lung function parameters, all animals were subjected to a bronchoconstriction test. All OVA-sensitized animals were exposed to 0.25% ACh solution for 30 seconds using a nebulizer (CX4-Omron Healthcare). Guinea pigs in the nonsensitized control group were exposed to normal saline in place of ACh. Tidal volume and respiration rate of each animal were recorded before and after the exposure.
Cellular Count and Serum Preparation
On day 30, blood (3 mL) was collected from each animal under light ether anesthesia. Each sample was then divided into 2 portions. The first aliquot (2.5 mL) was placed in a nonheparinized tube for serum; the separated serum was stored at –80°C for later quantitative determination of cytokines. The second portion (0.5 mL) was placed in a heparinized tube and used for total and differential leukocyte counts. Detailed procedures for total and differential leukocyte counts were the same as those outlined for the BAL fluid (see below).
Bronchoalveolar Lavage
At the end of experiment (ie, day 30) for the collection of BAL fluid, an overdose of ketamine (30 mg/kg) and xylazine (20 mg/kg) was subcutaneously administered to each animal. A polypropylene cannula was then inserted into the trachea, and 0.9% (wt/vol) normal saline solution (10 mL) at 37°C was introduced into lungs via a 10-mL syringe and then recovered 5 minutes later. The recovered lavage fluid (5 mL) was centrifuged at 500 g for 10 minutes at 4°C; the resultant supernatant was collected and stored at –80°C until used in cytokine determinations. The cells in the pellet were washed in 0.5 mL of saline, and total cell counts were then performed in an automated cell counter (Cell Dyne 3500, Abbott Laboratories, New York, NY). To perform differential analyses, cells were identified as eosinophils, lymphocytes, macrophages, or neutrophils using standard morphologic determinants.
Cytokines in Serum and BAL Fluid
The levels of TNF-α, IL-4, and IL-6 in recovered serum and BAL samples were measured using ELISA kits. After following the manufacturer’s protocol, all plates were analyzed on an automated plate reader (Lab System Multiscan 51118220, Thermo Bioanalysis, Helsinki, Finland). The detection limit of the kits for TNF-α, IL-4, and IL-6 were less than 15 pg/mL, less than 5 pg/mL, and less than 10 pg/mL, respectively.
Histamine Assay in Lavaged Lung Tissue
Lung tissue lobe from each animal was dissected out separately immediately following BAL fluid collection. Lung tissue (250 ± 20 mg) was placed in 2.5 mL of normal saline for the preparation of homogenate. The presence of histamine was determined fluorometrically (SL-174, Elico, India) by the modified method of Shore et al. 33
Statistical Analysis
Results were reported as mean ± SEM. Statistical analysis was performed using a 1-way analysis of variance. If the overall F value was found to be statistically significant (ie, P < .05), further comparisons among groups were made using the post hoc Tukey’s test, and differences were considered statistically significant at P < .05. When data were found to be very significant (ie, P < .01) or highly significant (ie, P < .001), this was indicated in the results. All statistical analyses were performed using Graph Pad software (San Diego, Calif).
Results
Phytochemical Study
The preliminary phytochemical screening of the n-butanol fraction of seeds of M oleifera showed the presence of alkaloids, flavanoids, steroids, glycosides, tannins, and terpenoids (Table 1 ). On performing HPTLC analysis of the fraction, we found following percentage weight by weight quantity of ascorbic acid (0.45%), quercetin (2.38%), benzylisothiocyanate (2.72%), and glycerol-1-(9-octadecanoate) (0.29%). The concentration of β-sitosterol was found to be 3.42% by HPLC analysis.
Table 1. Preliminary Phytochemical Study on n-Butanol Fraction of Ethanolic Extract of Seeds of M oleifer a
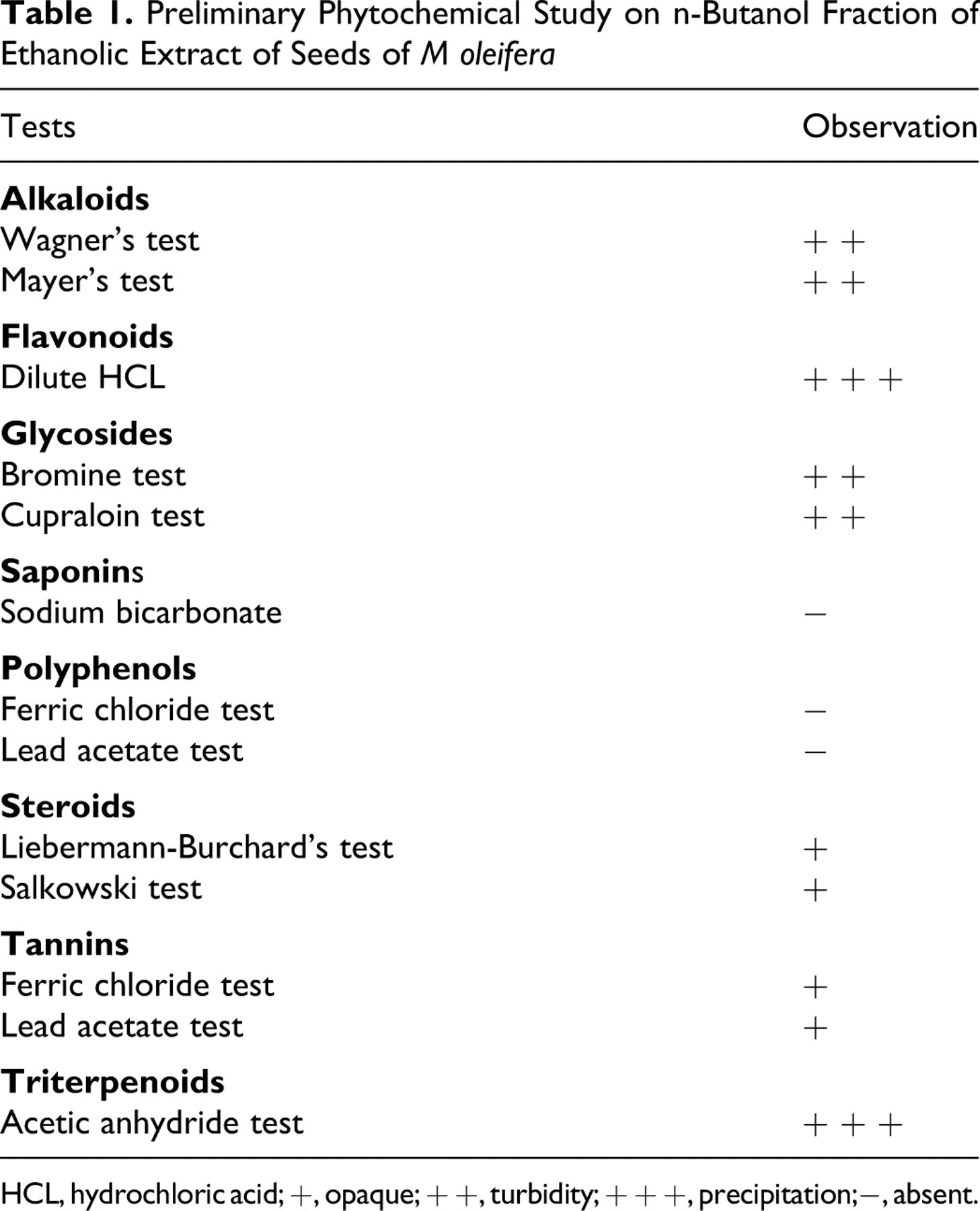
HCL, hydrochloric acid; +, opaque; + +, turbidity; + + +, precipitation;−, absent.
Effect of MONB on Body Weight
No animals in the model control (group II) and drug regimen groups (groups III and IV) exhibited any significant differences in body weight compared with those of the nonsensitized controls during the experimental period (data not shown). Furthermore, there were no apparent effects on appetite/water consumption or outward appearance (ie, fur coat, eyes) of these animals in each treatment group.
Effect of MONB on Lung Function and Acetylcholine-Induced Bronchoconstriction Test
There was no significant effects observed on days 18, 21, or 24; however, on day 29 the tidal volume was decreased significantly (P < .01) and respiration rates increased significantly (P < .001) in model control animals compared with values in nonsensitized controls. DXM- and MONB-treated animals showed significant improvement in each of these parameters (Table 2). When all OVA-sensitized animals were exposed to the aerosol of 0.25% ACh for 30 seconds on days 18, 21, 24, and 29, bronchospasm in the form of preconvulsion dyspnea was commenced. However, significant changes in tidal volume (P < .01) and respiration rate (P < .001) were observed on day 29 only (data not shown). Figure 1 shows the change in the pattern of lung function in OVA-sensitized model control animals.
Table 2. Effect of M oleifer a n-Butanol Fraction on Lung Function Parameters of Guinea Pigs
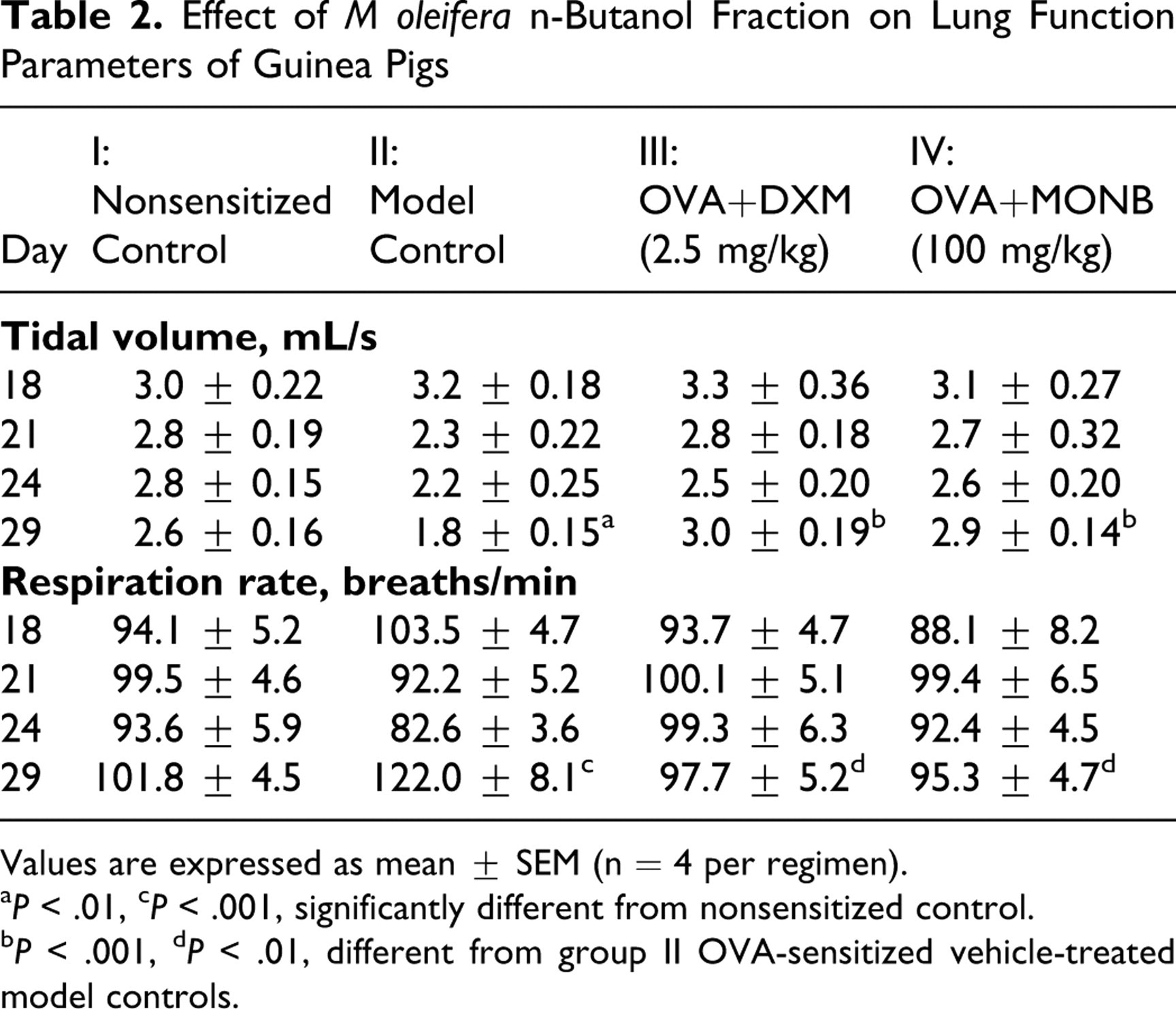
Values are expressed as mean ± SEM (n = 4 per regimen).
a P < .01, c P < .001, significantly different from nonsensitized control.
b P < .001, d P < .01, different from group II OVA-sensitized vehicle-treated model controls.
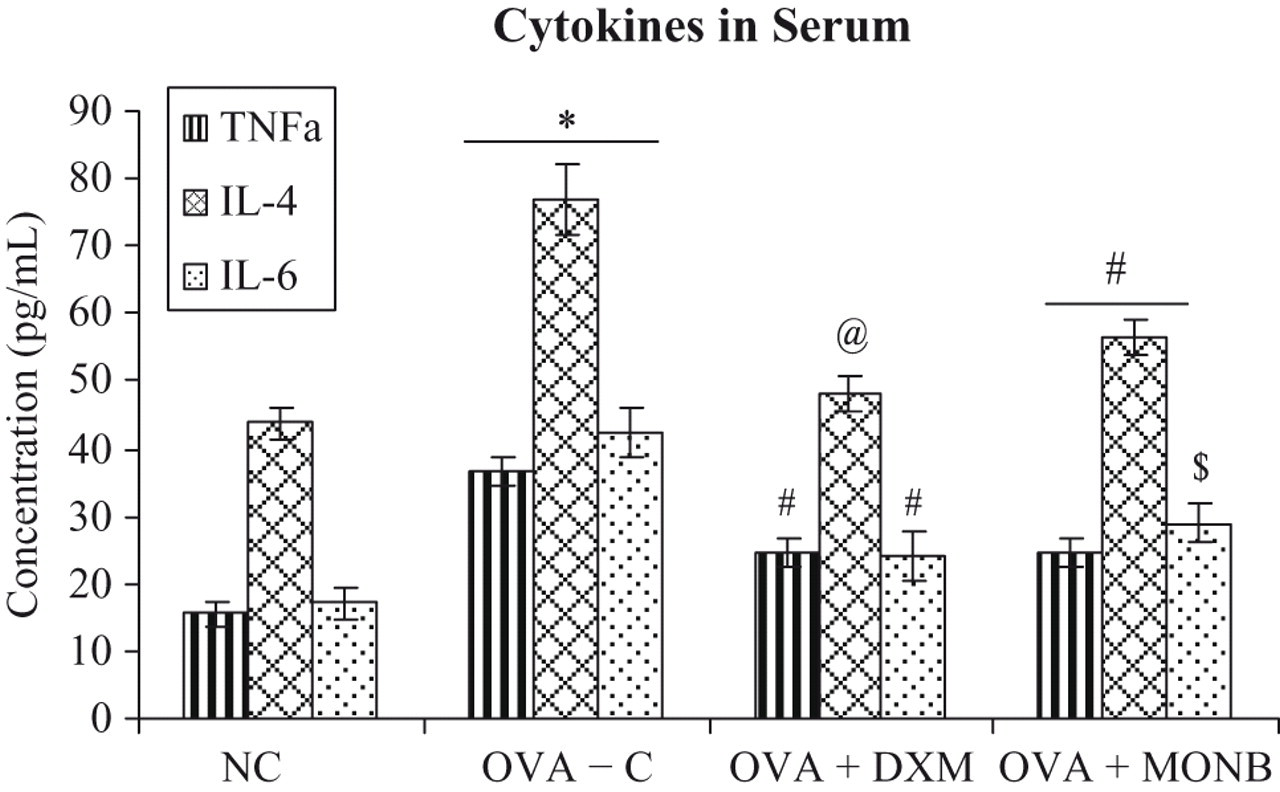
Effect of treatments on serum cytokine levels of guinea pigs. Value is significantly different from non-sensitized control (*P < 0.001). Value different from Group II OVA-sensitized vehicle-treated model controls (@ P < 0.001; # P < 0.01; $ P < 0.05). All bars represent the mean ± SEM from n = 4 guinea pigs/treatment group.
Effect of MONB on Circulating Cellular Count
Analysis of the number of total leukocytes (P < .001) and differential counts revealed an increase in the number of eosinophils (P < .001) and neutrophils (P < .01), whereas lymphocytes (P < .001) and monocytes (P < .01) were decreased significantly in model control animals compared with nonsensitized controls. However, the numbers of circulating eosinophils and neutrophils were decreased significantly in DXM- and MONB-treated animals compared with model control animals (Table 3 ).
Table 3. Effect of M oleifera n-Butanol Fraction on Total Cells and Differential Leukocyte Count in Blood (×103 cells/mL)
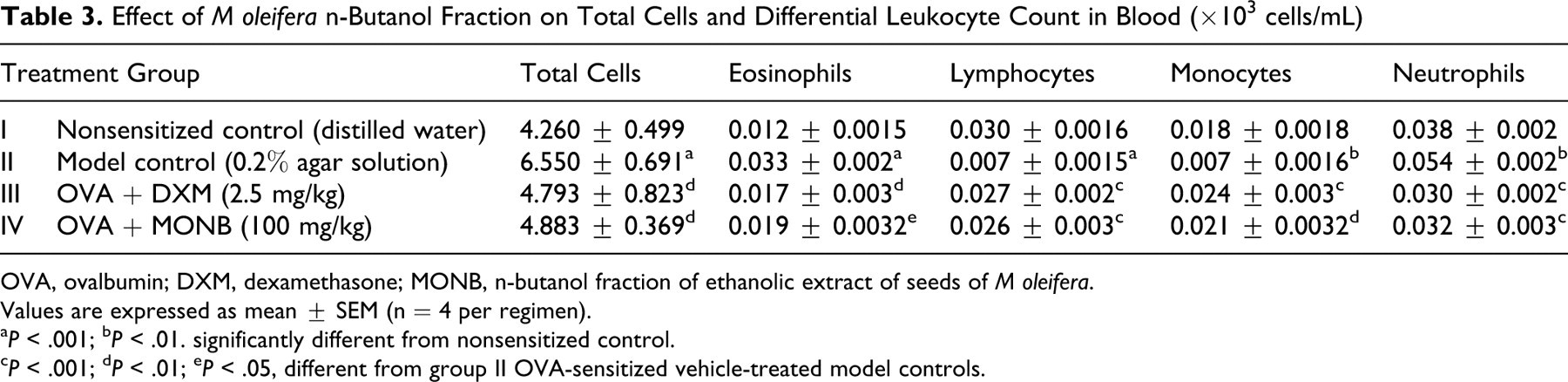
OVA, ovalbumin; DXM, dexamethasone; MONB, n-butanol fraction of ethanolic extract of seeds of M oleifera.
Values are expressed as mean ± SEM (n = 4 per regimen).
a P < .001; b P < .01. significantly different from nonsensitized control.
c P < .001; d P < .01; e P < .05, different from group II OVA-sensitized vehicle-treated model controls.
Effect of MONB on Inflammatory Cellular Counts in BAL Fluid
The analysis of BAL fluid revealed that numbers of total cells as well as each differential cell in model control animals were increased significantly. Treatment with DXM and MONB significantly (P < .01 and P < .05, respectively) decreased total cell counts compared with model control animals. Furthermore, decreases in number of eosinophils, lymphocytes, macrophages, and neutrophils were found with MONB treatment (Table 4 ).
Table 4. M oleifera n-Butanol Fraction on Total Cells and Differential Leukocyte Counts in BAL fluid (×103 cells/mL)
OVA, ovalbumin; DXM, dexamethasone; MONB, n-butanol fraction of ethanolic extract of seeds of M oleifera.
Values are expressed as mean ± SEM (n = 4 per regimen).
a P < .001, b P < .01, c P < .05, significantly different from nonsensitized control.
d P < .01, e P < .05, different from group II OVA-sensitized vehicle-treated model controls.
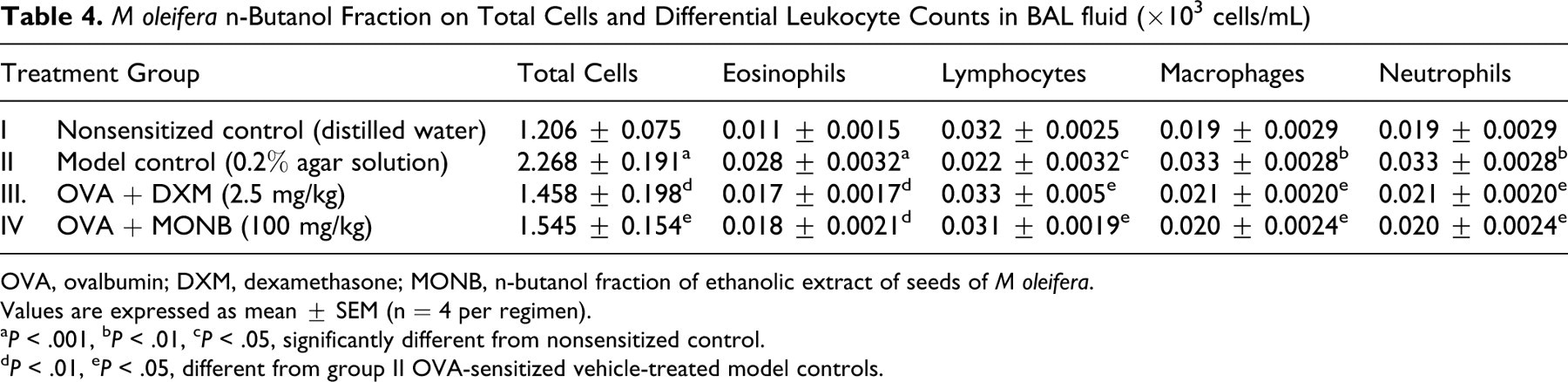
Effect of MONB on Cytokine Production in Serum
TNF-α, IL-4, and IL-6 levels (Figure 2 ) in the serum of OVA-sensitized model controls were significantly (P < .001) increased compared with those in nonsensitized animals. Treatment with DXM and MONB caused significant reductions in the levels of TNF-α (P < .01), IL-4 (P < .001; P < .01), and IL-6 (P < .01; P < .05), respectively, relative to those in the group II animals.
Effect of treatments on BAL fluid cytokine levels of guinea pigs. Value is significantly different from non-sensitized control (*P < 0.001). Value different from Group II OVA-sensitized vehicle-treated model controls (# P < 0.01; $ P < 0.05). All bars represent the mean ± SEM from n = 4 guinea pigs/treatment group.
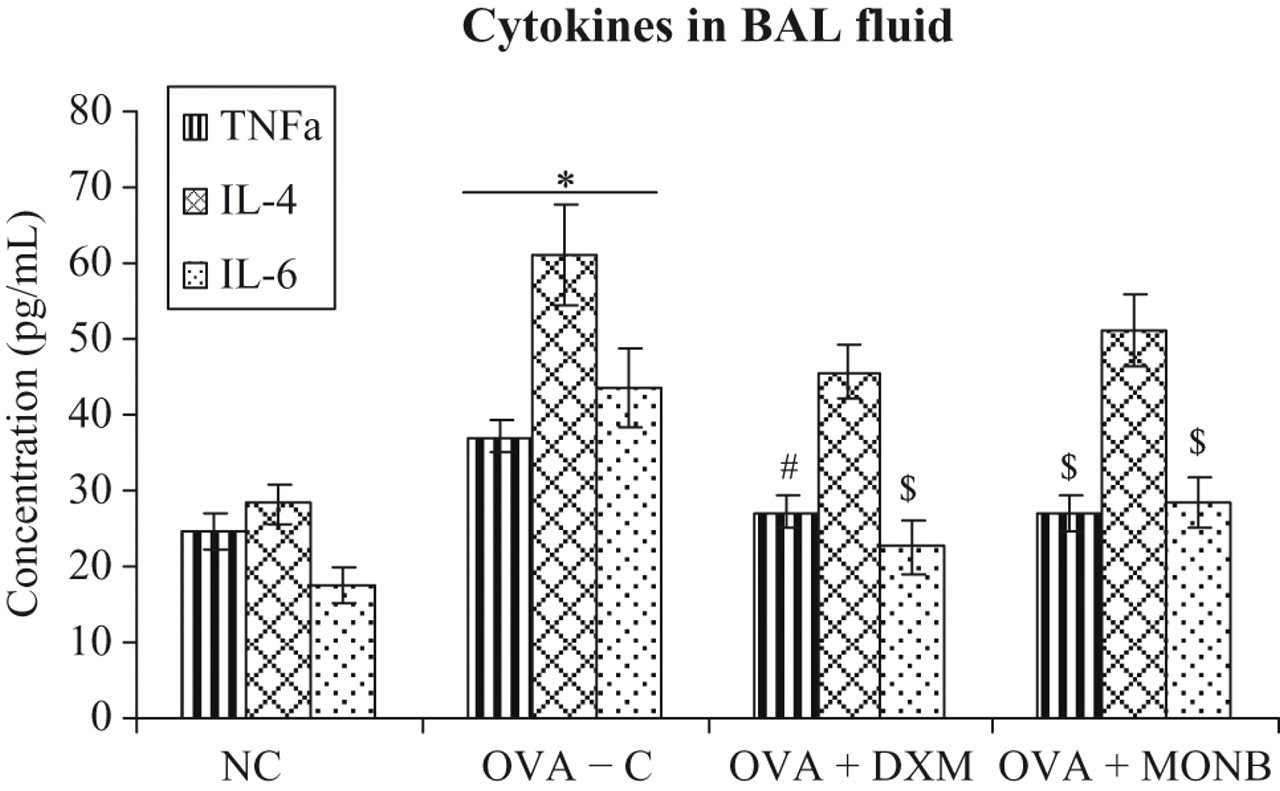
Effect of MONB on Cytokine Production in BAL Fluid
The level of BAL TNF-α< IL-4, and IL-6 (Figure 3 ) in guinea pigs of model control group were significantly (P < .001) greater than those in the nonsensitized controls. The DXM and MONB treatment did cause significant reductions in TNF-α (P < .01, P < .05) and IL-4 (P < .05, P < .05) levels compared with the model controls, but there were no effects from either of these treatments on IL-4 levels.
Effect of treatments on lung tissue histamine levels of guinea pigs. Value is significantly different from non-sensitized control (*P < 0.001). Value different from Group II OVA-sensitized vehicle-treated model controls (@ P < 0.001, # P < 0.05). All bars represent the mean ± SEM from n = 4 guinea pigs/treatment group.
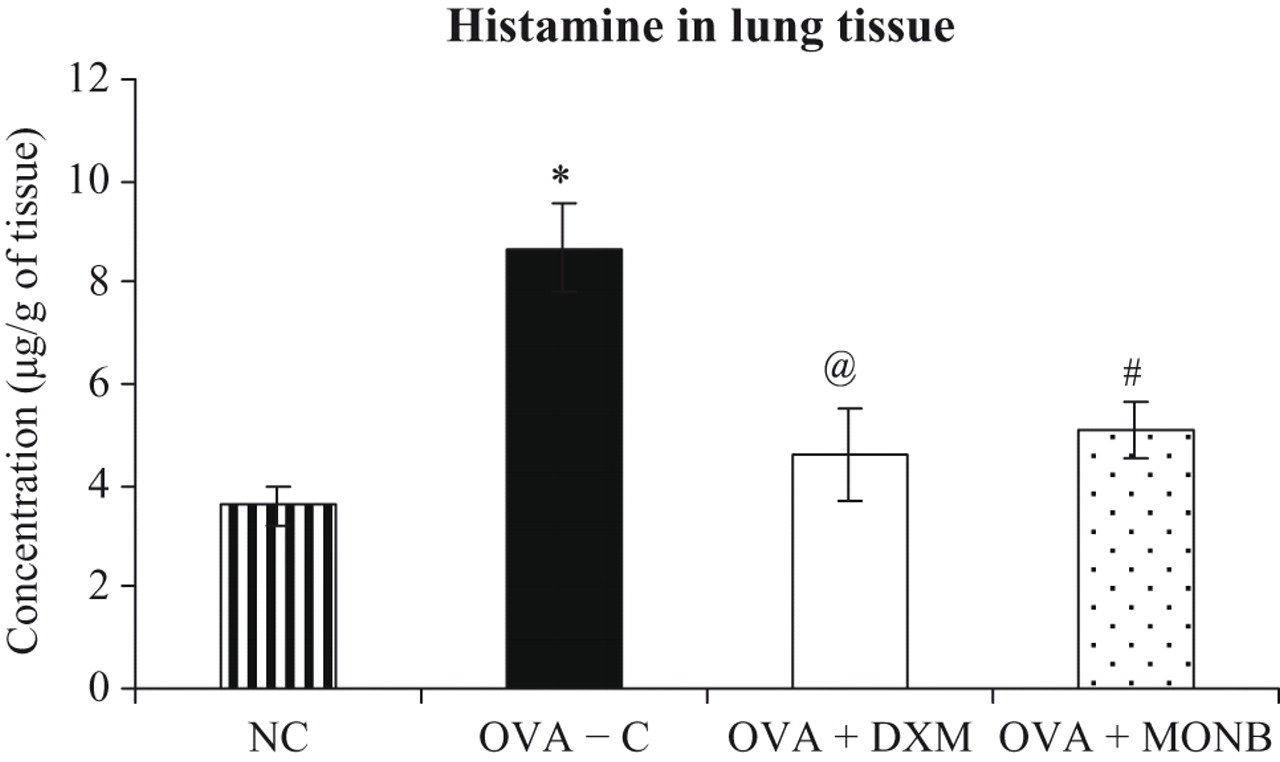
Effect of MONB on Histamine Levels
Histamine levels in lung homogenates of the previously lavaged lungs were assayed (Figure 4 ). The levels of histamine in model control animals were significantly (P < .001) greater than those in the nonsensitized controls. Treatment with MONB showed a significant (P < .05) decrease in the degree of elevated histamine release that was seen in the animals present in group II.
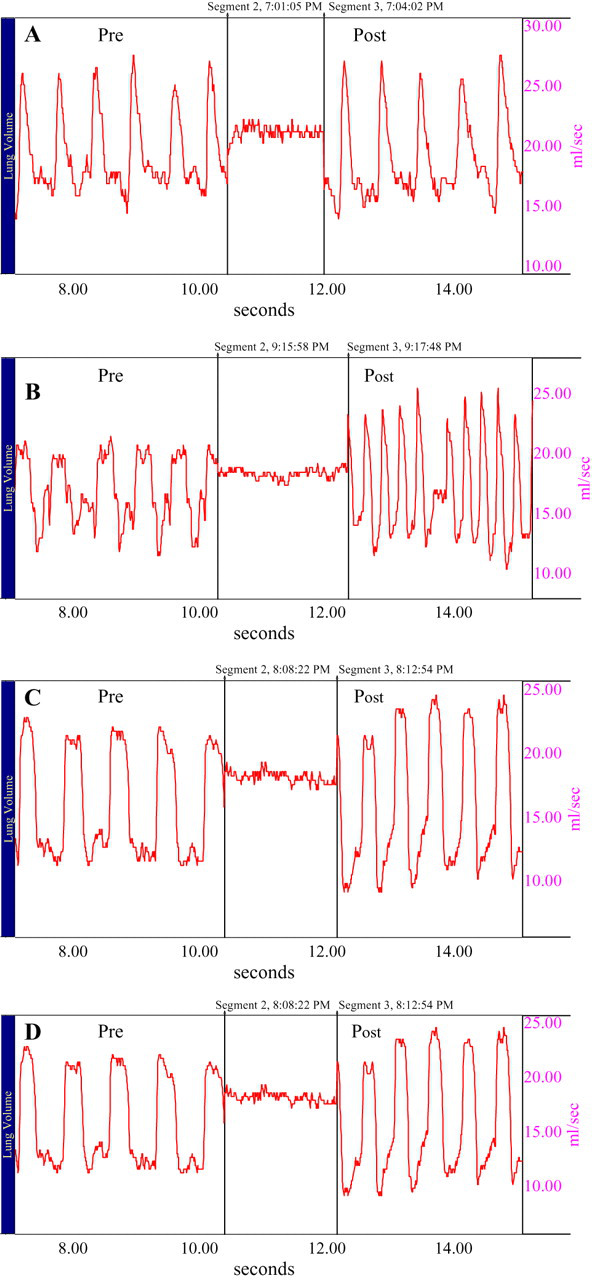
Effect of treatments on acetylcholine-induced bronchoconstriction in guinea pigs. Representative tracings showing the changes in pre and post tidal volume (Vt) and respiration rate (f) on bronchoconstriction induced by acetylcholine in guinea pigs. (A) Nebulisation with saline aerosol to nonsensitized control animals, (B) OVA-sensitized model control animals, (C) DXM-treated animals sensitized with OVA, (D) MONB-treated animals sensitized with OVA. All animals present in above mentioned groups (except group I) were challenged with 0.5% of OVA followed by 0.25% acetylcholine for 30 sec which produces bronchoconstriction. -induced bronchoconstriction in guinea pigs.
Discussion
Complementary and alternative medicine has been used to treat asthma for hundreds of years. 34 Indian systems of traditional medicine are well systematized but are largely unrecognized in the West. Ayurveda is gaining greater visibility; however, related systems, such as Unani-Tibb, Siddha, and Tibetan, are more obscure. 35 Some Ayurvedic drugs of interest for consideration in asthma were subjected for clinical studies based on their effects on experimental studies. These include Datura stramonium; the efficacy of this plant was studied in patients with bronchial asthma and chronic bronchitis. 36 Furthermore, clinical efficacy of Solanum xanthocarpum at a dose of 300 mg 3 times daily for 3 days was investigated in mild to moderate bronchial asthma. 37 One study showed that Coleus forskohlii is superior to placebo and equivalent to fenoterol in protecting against methacholine-induced bronchoconstriction. 38 In light of the above facts, and as part of our continuing search for biologically active antiallergic agents from the medicinal resources, we evaluated the efficacy of n-butanol fraction of seeds of Moringa oleifera Lam. for its activity against allergic guinea pigs.
In the present study, none of the OVA-sensitized animals that were treated with MONB showed any change in body weight (data not shown), suggesting that the extract did not interfere with the normal growth of the animals. In model control animals, a significant decrease in the tidal volume is suggestive of bronchoconstriction due to airway inflammation that resembles an asthmatic condition. All the animals in the model control group exhibited irritability, sneezing, and hyperrhinorrhea. The significant increases in respiration rate observed in these animals were indicative of exertional breathing, a symptom of asthma. Furthermore, protection against acetylcholine-induced bronchoconstriction in drug-treated animals demonstrated the bronchodilatory effect of an extract. Treatment with DXM and MONB showed protective effect by improving tidal volume and respiratory symptoms.
It has been well documented that in guinea pigs, 39 nonhuman primates, 40 and humans 4 there is a correlation between the activation of eosinophils and the development of bronchial hyperresponsiveness. Neutrophilic inflammation in the airway is also increasingly recognized in acute exacerbations of asthma and in status asthmaticus. 4 In our study, we found a significant increase in number of these cells in blood as well as in BAL fluid of model control animals. However, these cell numbers, especially eosinophils and neutrophils, decreased significantly with MONB treatment. Therefore, these results provide evidence that MONB treatment may be useful in controlling the activation of the inflammatory processes underlying the exacerbation of allergic asthma.
Cytokines play a critical role in orchestrating and perpetuating inflammation in asthmatic airways. 8 Among the various cytokines, T-lymphocyte-derived TNF-α, IL-6, and Th2-type cytokine IL-4, in particular, have significant roles in asthma. 41 Elevated levels of TNF-α have been detected in the sputum, bronchoalveolar lavage fluid, and biopsy samples from asthmatic patients. 13 Recent studies confirmed a predominance of Th2 cell-derived cytokines such as IL-4 in the pathogenesis of bronchial asthma. 42 Moreover, IL-6 has been reported to have activities on a wide range of cellular processes, including T-lymphocyte activation and immunoglobulin production by B-lymphocytes. Our study suggests that the numbers of inflammatory cells and levels of cytokines that were increased predominantly in model control animals were suppressed significantly in animals treated with extract. These results validate the popular use of M oleifera for the treatment of asthma and associated allergic diseases. These findings also indicate that the possible mechanism of action may be associated with a reduction in cytokine production/release.
The mast cells are thought to play a major role in the development of many physiologic changes during allergic responses. 43 Bartosch et al 12 were the first to report that histamine is released from guinea pig lung during anaphylaxis. It also has been reported that histamine activates eosinophils. 44 In the present study, increased histamine levels and number of eosinophils due to sensitization with ovalbumin were significantly decreased in the MONB-treated animals. This suggests that the extract might have some efficacy in inhibiting the release/synthesis of histamine.
The results of present study are consistent with the work of several other investigators in determining the effectiveness of selected natural products against inflammation-related pathologies in the lungs. For example Singh et al 45 demonstrated that Elaeocarpus sphaericus fruits possess both mast-cell-stabilizing activity and efficacy against experimental asthma. Recently, Christofidou-Solomidou and Muzykantov 46 demonstrated that ascorbic acid is the most extensively investigated antioxidant for effects on asthma and has been shown in numerous case-control and cross-sectional studies to be associated with a reduced risk of asthma. An experimental study confirmed that quercetin inhibits mast cell degranulation along with subsequent release of histamine. 47 Antiasthmatic activity of benzylisothiocyanate has been reported against the bronchial obstruction induced by ovalbumin in experimental animals. 48 Yuk et al 49 reported the beneficial effects of lactose-β-sitosterol and β-sitosterol on ovalbumin-induced lung inflammation in actively sensitized mice. Our results show that these effective compounds are present in MONB. Furthermore, phytochemical investigations of seeds of M oleifera in the present study and by other investigators also have shown the presence of various active components (Tables 1 and 5 ). These compounds might be responsible for the one or more mechanisms for antiasthmatic activity of n-butanol fraction.
Table 5. Major Phytochemical Constituents of the Seeds of M oleifera

In this study, we highlighted the possible mode of action of n-butanol fraction to bring about anti-inflammatory activity in situ by investigating its efficacy against T-cell-derived cytokine production and subsequent cytokine-mediated cellular infiltration and its bronchodilatory potential and capacity to block mediator release into the local tissues like lung. Such observations support some of the traditional uses of the plant for therapeutic purposes, and thus M oleifera could be useful in developing better therapeutic molecules.
