Abstract
Hypothyroxinemia in rats has been well documented as a result of exposure to polychlorinated biphenyls (PCBs). Hypothetical mechanisms include induction of hepatic catabolic enzymes and cellular hormone transporters, and/or interference with plasma transport proteins. We hypothesized that if thyroxine displacement from transport proteins by PCBs occurs in vivo, it would result in increased free thyroxine (FT4). This study investigates the effects of a single oral dose of 2,2’,4,4’,5,5'-hexachlorobiphenyl (PCB 153 at 60 mg/kg) or 3,3’,4,4’,5,5'-hexachlorobiphenyl (PCB 169 at 1 mg/kg) on rats at 28 or 76 days of age. Total thyroxine (TT4) and FT4 were measured at 0.5, 1, 2, 4, 8, 24, or 48 hours post
Introduction
Polychlorinated biphenyls (PCBs) disrupt thyroid hormone (TH) homeostasis in rats and mice following developmental and adult exposures. 1-7 A number of mechanisms have been postulated to account for this effect. Polychlorinated biphenyls increase biliary elimination of thyroxine resulting from induction of uridine diphosphoglucuronosyl transferase (UGTs) and/or hepatic hormone transporters. 8,9 The induction of UGTs and other xenobiotic metabolizing enzymes by PCBs may occur through activation of either the aryl hydrocarbon (Ah) receptor, pregnane X receptor (PXR), or constitutive androstane receptor (CAR). 10-12 An alternative hypothesis for the effects on serum THs is that PCBs and their hydroxylated metabolites interfere with serum transport of THs. 13-16 Because of structural differences seen among PCBs, it is likely that multiple mechanisms are responsible for the decreases in serum TH concentrations following exposures to PCBs. 4,5
There are 2 main thyroid hormone plasma transport proteins, transthyretin (TTR, also called thyroid binding pre-albumin, or TBPA) and thyroxine binding globulin (TBG). Transthyretin is the predominant binding protein found in most nonprimate mammalian species, including adult rodents, and is highly conserved. 17 Thyroxine binding globulin is the predominant binding protein form in humans. 18-21 However, thyroid binding proteins are developmentally regulated in rats. Thyroxine binding globulin is expressed in rats until approximately postnatal day (PND) 60, with peak expression occurring during weaning. 18-21 Transthyretin is predominant in adult rats, however, low levels of TBG do persist 19 and can be experimentally induced by hypothyroidism, malnutrition, or caloric restriction. 18,22,23 Xenobiotics, including PCBs, have been shown to displace thyroxine from TTR, but not TBG, in vitro. 13-15,24,25 Competition for serum transport proteins as the major site of action of the PCBs has several implications. First, blood is a target tissue, and the peak effects of these chemicals should be related to peak blood concentrations. Since blood concentrations of persistent organic pollutants, such as dioxins and PCBs, peak several hours after the initial exposure and then decrease as the chemicals diffuse into adipose tissue, 26,27 peak effects on serum TH concentrations should occur within hours following chemical exposure. Second, because serum free thyroxine (FT4) concentrations are less than 0.5%-0.1% of the total thyroxine (TT4) concentrations in blood, 28 serum FT4 should be a sensitive marker for the effects of PCBs on thyroid hormones. In addition, compared with adult rats, weanling rats should be less sensitive to the effects of PCBs on THs because of significant concentrations of TBG.
The present study tests the hypothesis that if thyroxine is displaced from transport proteins by PCBs it will result in increased FT4 concentrations in the blood. Further, this study will examine treatment with both PCB 153 and PCB 169 in young weanling and adult rats. The coplanar PCB 3,3′,4,4′,5,5′-hexachlorobiphenyl (PCB 169, dioxin-like) and the ortho-substituted PCB 2,2′,4,4′,5,5′–hexachlorobiphenyl (PCB 153, non-coplanar, phenobarbital-like) were used as examples of the 2 classes of PCBs shown to bind TTR with similar affinity as thyroxine itself in vitro. 14 Young and adult rats were examined to account for differences in the predominant plasma transport protein found in rats of different ages (TBG and TTR 19 ). Serum TT4 and FT4 concentrations, as well as hepatic enzyme activity of CYP1A1 (using ethoxyresorufin as a substrate), CYP2B (using pentoxyresorufin as a substrate), and UGT activity were also measured as biomarkers of PCB actions on hepatic enzymes.
Methods and Materials
Animals
Five groups of 56 nulliparous female Long-Evans rats were obtained from Charles River Laboratory (Raleigh, NC and Portage, MI) at 23 or 52-62 days of age. There was a minimum of 5 days of acclimation in an American Association for Accreditation of Laboratory Animal Care (AAALAC)-approved animal facility prior to treatment. Animals were housed 2 per standard plastic hanging cage (45 cm × 24 cm × 20 cm), with heat-sterilized pine shavings bedding. Colony rooms were maintained at 21°C ± 2°C with 50% ± 10% humidity on a photo-period of 12L:12D (6:00
Chemicals and Treatment
Rats were acutely exposed, via gavage, to a single dose of 60 mg/kg PCB 153 (2,2’,4,4’,5,5'-hexachlorobiphenyl, Radian International, Austin, TX, lot #32466-47, 99% purity) or 1 mg/kg PCB 169 (3,3’,4,4’,5,5'-hexachlorobiphenyl, AccuStandard, New Haven, CT, lot# 101393, 99% purity). Stock solutions were prepared by dissolving the PCB in acetone and then adding an equal amount of corn oil (Sigma Chemical Co., St. Louis, MO). The acetone was evaporated in a speed-vac for approximately 4 hours. Final dosing solutions were obtained by dilution of the stock with additional corn oil. Polychlorinated biphenyls were administered in a dosing volume of 1.0 mL corn oil/kg body weight. Female rats were chosen to be consistent with ongoing research on the effects of short-term exposure to PCBs. 2,29 Dose levels were selected based on previous work from this laboratory from short-term, 4-day dosing studies. The dose levels selected caused drastic reductions in TT4 (about 50%) with no adverse affects on the animals' general health or body weight. 2,29 Rats were randomly assigned to treatment groups by counter-balancing body weights. Separate groups of rats were used for each of the 7 time points (0.5, 1, 2, 4, 8, 24, or 48 hours) in the study for each chemical and rat age. There were 8 rats/time point for a total of 56 rats in each group, with the exception of the group of young rats (23 days of age) treated with PCB 153, where there were 2 replicates of 56 rats each, making a total of 16 rats/group/time point.
At 0.5, 1, 2, 4, 8, 24, or 48 hours posttreatment, rats were weighed, and then trunk blood and liver were collected. Blood was collected in serum separator tubes (Becton-Dickinson, 36-6154). Serum was obtained after clotting whole blood on ice for approximately 0.5 hour, followed by centrifugation at 1278 ×
Thyroid Hormone Assay
Serum TT4 and FT4 were measured in duplicate using solid-phase Human Coat-A-Count radioimmunoassay (RIA) kits (Diagnostic Products Corporation, Los Angeles, CA). These specific TT4 and FT4 kits have been previously used in our lab according to the method of Craft et al. (2002) and Kato et al. (2007), respectively, and are routinely used worldwide for rat serum. Although the kit uses human serum for the calibrators, the antibody is against thyroxine, which is exactly the same molecule in rats and humans. Thus, the use of rat serum may yield a small change in the slope of the calibration curve, but does not invalidate its use. Inaccuracies may occur when reporting absolute serum concentrations, and we report our results and analysis as a percentage of control values. Intra-assay and interassay coefficients of variance for all assays were below 10%.
Hepatic Enzyme Activities Assay
Activity of 2 hepatic enzymes (EROD, PROD) were measured as biomarkers for CYP1A1 and CYP2B activity to confirm biological activity of the treatments. Hepatic microsomal ethoxyresorufin-O-deethylase (EROD) activity (a marker for hepatic aryl-hydrocarbon [Ah] receptor-mediated induction of CYP1A1) was determined in livers from only those rats dosed with PCB 169, using the method of DeVito et al (1993) with ethoxyresorufin as a substrate. Numerous studies have shown that PCB169 primarily induces CYP1A1. 30,31 A similar method was used to measure hepatic microsomal pentoxyresorufin-O-deethylase (PROD) activity (a marker for induction of hepatic CYP2B), 32 in only those rats exposed to PCB 153, using pentoxyresorufin as a substrate. Previous studies have shown that exposure to PCB153 primarily induces CYP2B. 2,33,34 Liver microsomal fractions were prepared as described previously. 32 Microsomal protein concentrations were determined using a microprotein assay kit (Bio-Rad, Richmond, CA) with bovine serum albumin as the standard (Bio-Rad, Richmond, CA). All substrate concentrations were 1.5 nM. Both EROD and PROD values were calculated as pmol resorufin/mg protein/min and graphically presented as percentage of control group mean activity.
Hepatic microsomal thyroxine-UGT activity was determined on 4 randomly chosen samples from each treatment group using the method of Visser et al, 35 as modified by Zhou et al. 36 125I-labeled thyroxine (T4) was purchased from PerkinElmer Life and Analytical Sciences, Inc. (Boston, MA). The calculated UGT activity was expressed as pmol T4-glucuronide (G)/mg protein/min and graphically represented as percentage of control group mean activity. For analysis, the limit of detection for UGT was set at 0.05 pmol T4-G/mg protein.
Data Analysis
All statistical analyses were performed using SAS version 6.12 (SAS Institute Inc., Cary, NC). Analysis of variance (ANOVA) procedures were used to test for treatment-by-time interactions, and main effects of treatment and time. Significant interactions were followed by step-down ANOVA tests for each independent variable. TT4, FT4, EROD, and PROD data were log-transformed because of unequal variances, and UGT data was not. For graphic comparative purposes, data are shown as percentage of control. Data from different age rats were analyzed independently. An α value of .05 was used for all comparisons.
Results
General Health, Body and Liver Weight
There were no treatment-related mortalities in young or adult rats exposed to PCB 153 or PCB 169. Likewise, there were no observable health effects of either treatment at either age. Similarly, there were no effects on body weight, liver weight, or liver-to-body weight ratios in this study (data not shown).
Thyroid Hormone Concentrations
In general, serum TT4 and FT4 were decreased following exposures to PCB 153 and 169 in young and adult rats, except for TT4 in rats given PCB169 and FT4 in adult rats given PCB169 (Figures 1 and 2). Serum %TT4 was affected by treatment with PCB 153 in both young and adult rats, but there were no significant time or time-by-treatment interactions (Figure 1A and 1B, see insets for main effect of treatment). In contrast, %TT4 was not significantly decreased by treatment with PCB 169 in young or adult rats (Figure 1C and 1D). There was a significant time-by-treatment effect on FT4 in young rats, but only a main effect of treatment in adult rats following exposure to PCB 153. There was a significant main effect of PCB 169 treatment on FT4 in young rats, but no effect in adult rats. All the effects of these single doses of PCBs on TT4 and FT4 were moderate (20%-30% decreases).
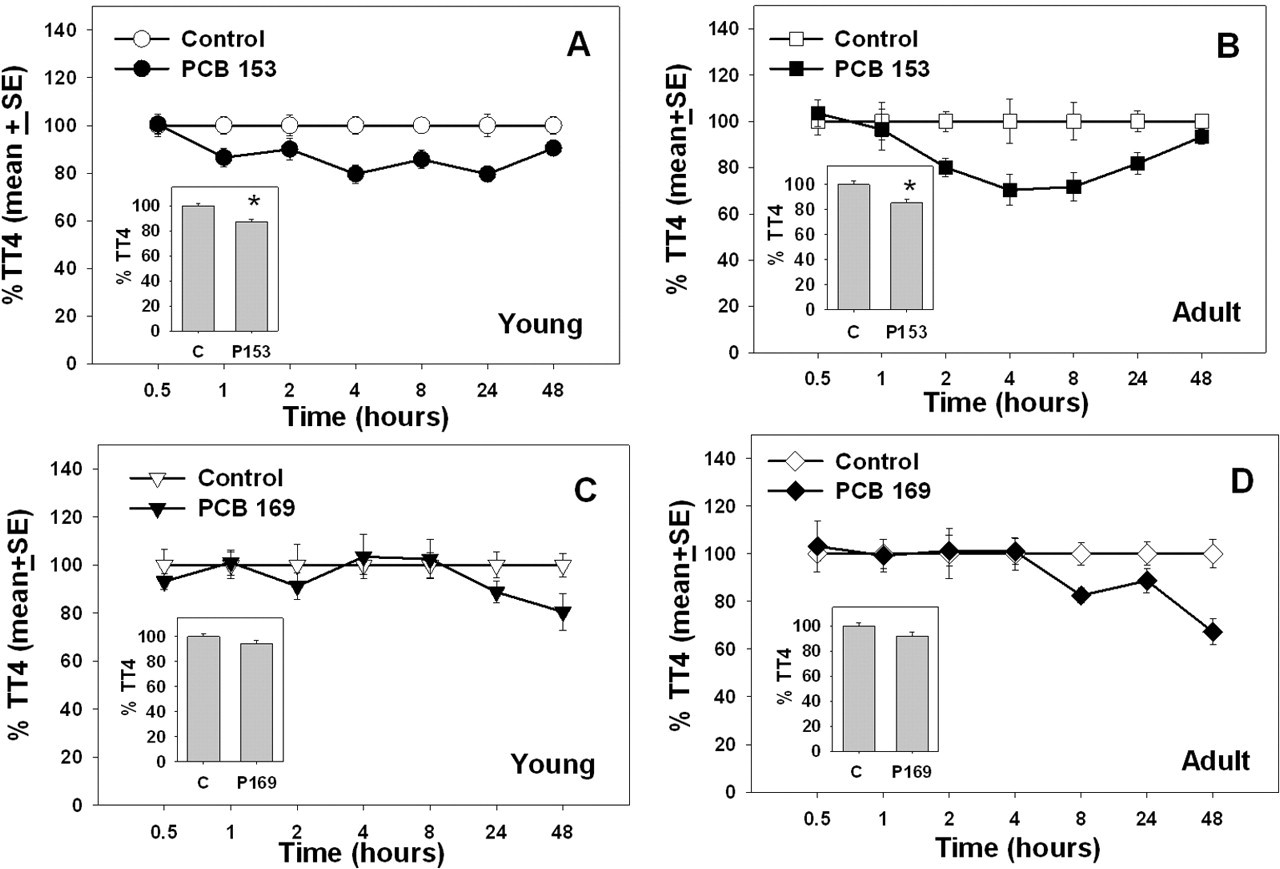
Percentage of control total thyroxine in 28-day-old (young) and 76-day-old (adult) rats (average age) after treatment with PCB 153 or PCB 169. Inset graphs illustrate treatment mean collapsed across all time points. Data points with * are significantly different (
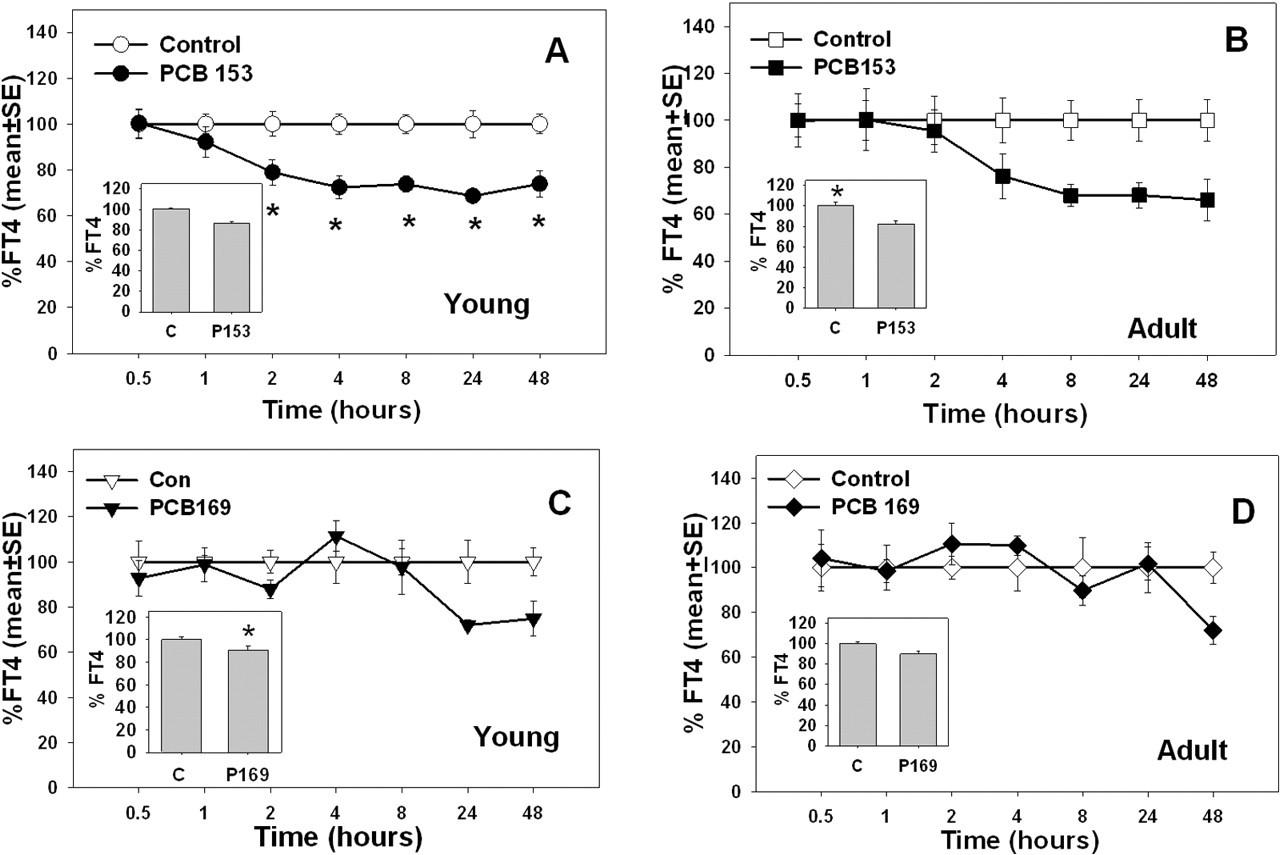
Percentage of control free thyroxine in 28-day-old (young) and 76-day-old (adult) rats (average age) after treatment with PCB 153 or PCB 169. Inset graphs illustrate treatment mean collapsed across all time points. Data points with * are significantly different (
In young rats given PCB 153 there was a significant effect of treatment (
In young and adult rats given PCB 169, there were no significant effects of treatment, time, or time-by-treatment interactions on TT4. There was a main effect of treatment (
UGT Induction
Hepatic UGT activity was not affected by treatment with PCB 153 in rats at either age (Figure 3A and 3B). However, UGT activity was induced following treatment with PCB 169, at both ages (Figure 3C and 3D). In young rats given PCB 169, there was a significant time-by-treatment interaction (
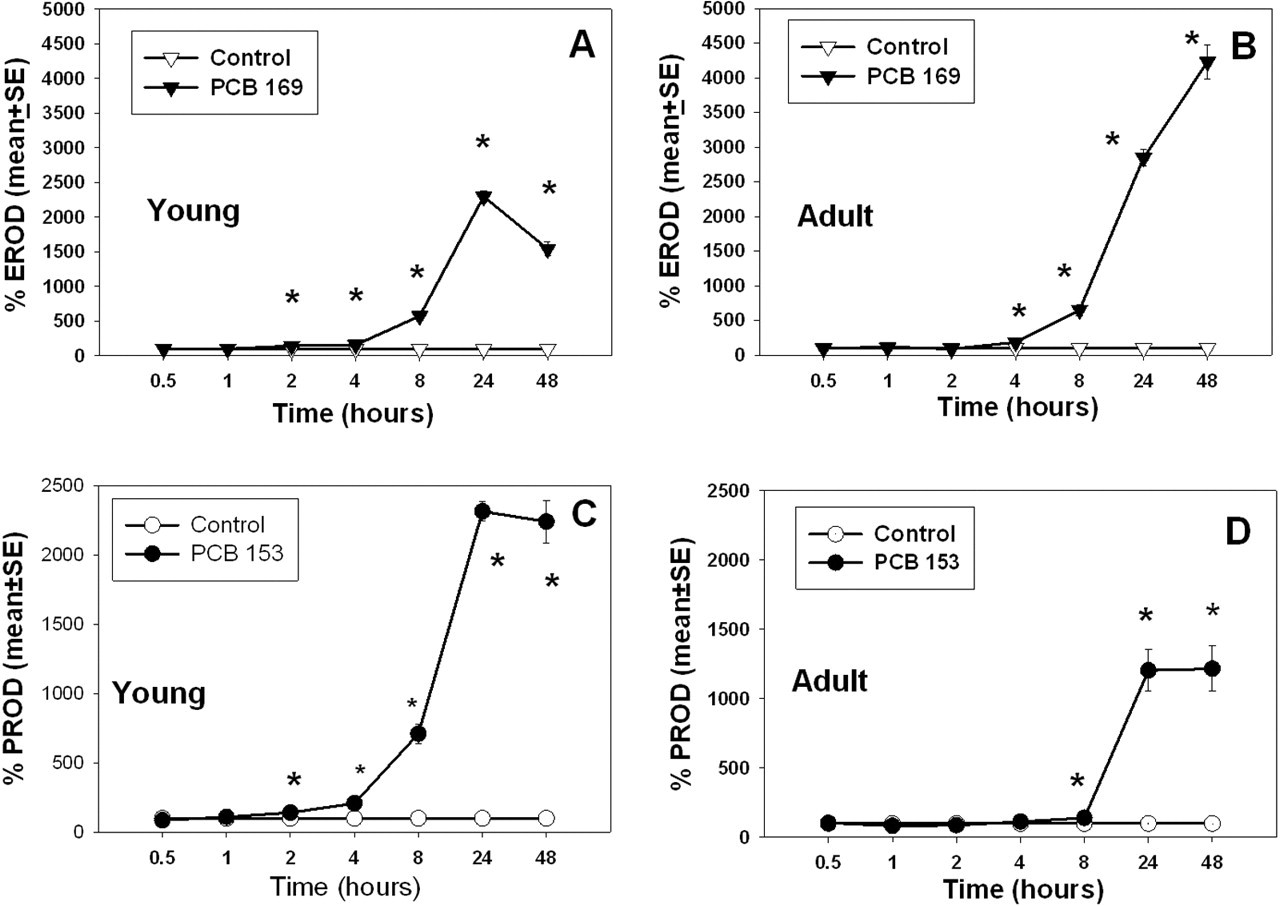
Percentage of control microsomal EROD (A and B) and PROD (C and D) activity in 28-day-old (young) and 76-day-old (adult) rats (average age) after treatment with PCB 153 or PCB 169. Data are represented as a percentage of control values. Data points with * are significantly different (
Cytochrome P450 Activity
Hepatic microsomal EROD activity following PCB 169 treatment increased in rats at both ages (Figure 4A and 4B). Hepatic microsomal PROD activity increased in both young and adult rats following treatment with PCB 153 (Figure 4C and 4D).
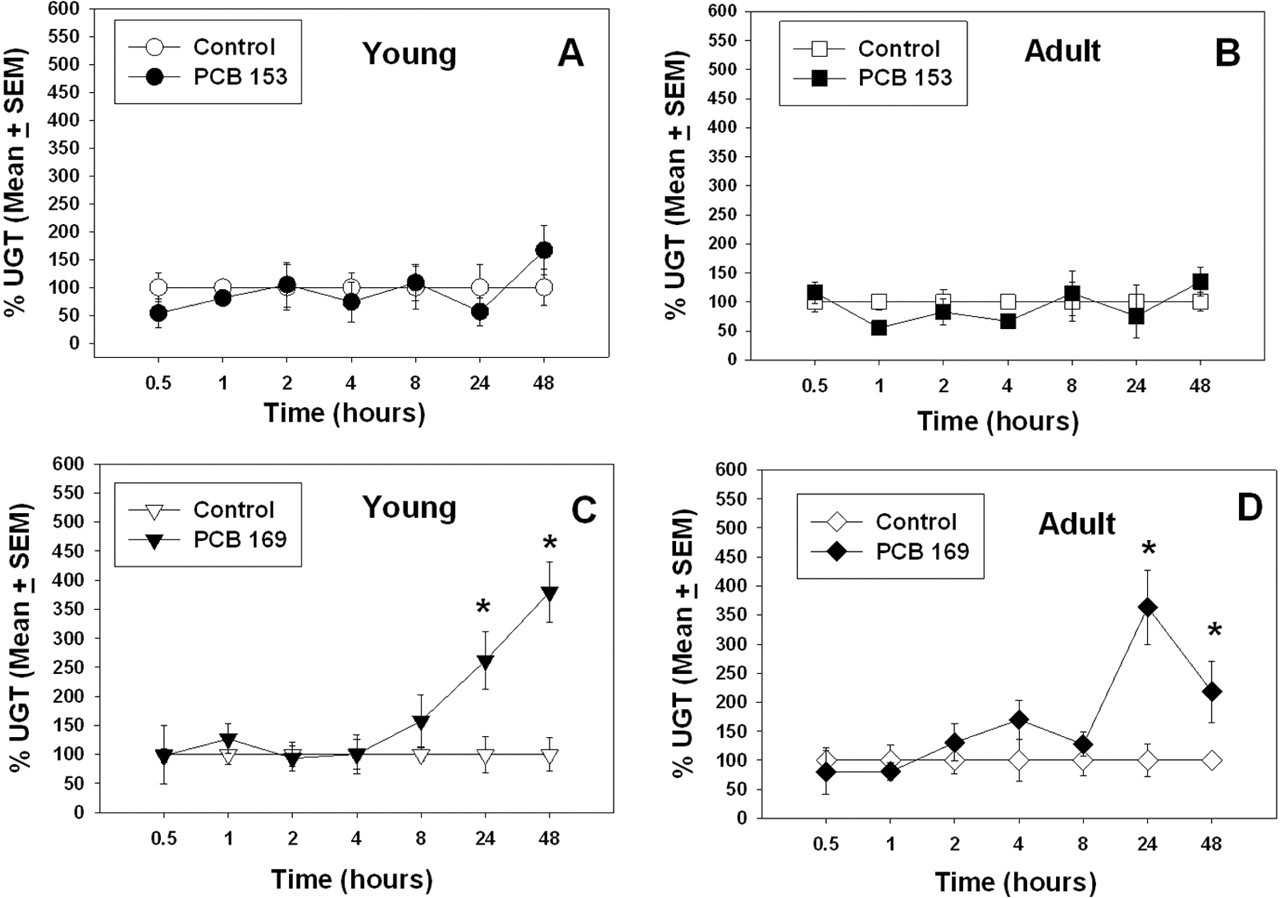
Percentage of control UGT in 28-day-old (young) and 76-day-old (adult) rats (average age) after treatment with PCB 153 or PCB 169. Data points with * are significantly different (
In young rats exposed to PCB 169, there was a significant time-by-treatment interaction (
In young rats given PCB 153, there was a significant time-by-treatment interaction (
Discussion
The present study tested whether acute exposure to PCBs would result in measurable increases in serum FT4 over a 48-hour time course in rats. At no time following treatment with PCB 153 or PCB 169 in young or adult rats were there any significant increases in FT4 in serum. In fact, in both young and adult rats, treatment with PCB 153 led to decreases in both FT4 and TT4, and PCB 169 decreased FT4 in young rats and TT4 in adults. These data demonstrate that acute PCB exposure does not increase serum FT4 concentration in rats in vivo. The decreases in THs seen following exposure to PCB 169 in the current study were associated with increased hepatic microsomal T4 glucuronidation activity, as seen by increased UGT in rats at both ages. However, no effect on UGT was seen following treatment with PCB 153 at either age. The reduction in FT4 and TT4 after PCB 153 and 169 exposure at both ages was coincident with induction of hepatic phase I enzymes (EROD and PROD).
A number of authors have presented evidence that individual PCB congeners and hydroxy-metabolites of PCB will displace TT4 and TT3 from TTR but not TBG. 13-15,24,25 This finding led to the hypothesis that TH displacement in vivo is responsible for decreased serum thyroxine concentrations, 13,37 possibly because of increased hepatic catabolism of FT4. In the present in vivo study, both PCB 169 and PCB 153 failed to increase serum FT4 in either young or adult rats. These 2 age groups were chosen to cover developmental stages where either TBG or TTR is the prominent transport protein of thyroxine. 18,19,21 These results are inconsistent with the hypothesis that displacement of thyroxine from its plasma binding proteins increases FT4 concentrations in serum. These results are more consistent with alternative mechanisms, including induction of hepatic UGTs 2,7,35 and induction of cellular hormone transporters, 11 which may lead to increased biliary excretion of conjugated and unconjugated THs.
In the present study, the most dramatic decreases in both FT4 and TT4 were seen following PCB 153 exposure in rats at both ages, and this finding was directly correlated with significant induction of PROD activity, but surprisingly, no change in UGT. These results are in contrast to other reports that PCB 153 induced UGT and decreased serum TT4. 2,6 Craft et al (2002) pointed out that the magnitude of UGT induction does not correlate well with the magnitude of TT4 decrease in rats or in mice. The lack of effect of PCB 153 in the present study may simply be a result of a short dosing regime (single dose), whereas Craft et al (2002) administered 4 consecutive daily doses. In contrast, PCB 169 decreased FT4 in the young rats and decreased TT4 in the adult rats. The reason for this difference is unknown.
An alternative explanation for the failure to detect increased FT4 concerns the method of measurement. There is debate on the best method to quantitate free concentrations of thyroxine. The present study used a commercial, direct solid-phase FT4 radioimmunoassay (RIA) kit. The ability of this RIA for use with rat serum is evidenced by successful use in detecting decreases in FT4 in many other publications. 38-40 However, equilibrium dialysis is often considered the reference method of choice, even though it may be prone to overestimate FT4 concentrations 41 and has some technical and theoretical problems with separation of free and bound thyroxine. 42 There are no published reports comparing various FT4 measurement methods in rats. Any underestimation of the serum FT4 levels would make no difference in our current data interpretation, because there was no significant increase in FT4 following either treatment at either age of rats tested, and in fact, statistically significant decreases were found. These present results are supported by recent findings that acute PCB 126 exposure decreases FT4 in rat serum, as measured by equilibrium dialysis. 43
The failure to find elevated FT4 concentrations in vivo in the current study is not necessarily inconsistent with displacement of thyroxine from TTR. There are a number of other serum proteins (eg, albumin and lipoproteins) that could act as “sinks” for displaced thyroxine. 44,45 Alternative explanations for the decreases in serum TT4 concentrations in vivo include increased hepatic clearance of glucuronidated thyroxine, 8,9,46 increased hepatic uptake of thyroxine, 11,12 or increased biliary elimination of unconjugated thyroxine. 47,48 Induction of UGTs is likely regulated by the AhR, CAR, or PXR receptors. 10,49,50 It appears that a combination of mechanisms is likely responsible for decreases in serum thyroxine in response to PCB exposure.
In summary, these data show that in both young and adult rats, in vivo exposure to PCB 153 and PCB 169 did not increase serum FT4. Increased UGT and EROD activity following exposure to PCB 169 and increased PROD activity after exposure to PCB 153, indicate that the decreases seen in THs may result from increased metabolism and/or biliary excretion. The lack of serum FT4 increases, even in the presence of up to a 36% decrease in TT4 after acute exposure to PCB 153 and 169, suggest that the in vitro interactions of these compounds with TTR do not predict the in vivo effects on serum FT4 concentrations.
Footnotes
Notes
Acknowledgments
Technical assistance from Michele M. Taylor, Dr Elena S. Craft, and Dr Tong Zhou is greatly appreciated. The authors are grateful to Drs Doug Wolf, Richard Wiggins, and Prasada Kodavanti, for critical reviews of this manuscript.
