Abstract
Streptococcus viridans are commensal bacteria that constitute a significant portion of the resident oral microflora. The objective of the present study is to investigate adverse effects, if any, of a blend of 3 natural strains, Streptococcus uberis KJ2, Streptococcus oralis KJ3, and Streptococcus rattus JH145 (probiotic mouthwash, ProBiora3). The blend is administered to rats orally once daily (5 days per week) at doses of 0, 106, or 109 colony-forming units of each strain for 14 weeks. No treatment-related adverse effects are observed in the physiological parameters during the study or in the evaluation of blood and tissue samples taken from the animals at the end. Results of an in vitro antibiotic susceptibility study demonstrate that all 3 ProBiora3 strains are susceptible to commonly used therapeutic antibiotics. The results of these investigations reveal that the no-observed-adverse-effect level of the probiotic mouthwash is 2.16 × 109 colony-forming units per strain per kilogram of body weight per day, the highest dose used.
Lactic acid–producing bacteria are among the prominent microorganisms inhabiting the gastrointestinal tract, and the importance of these nonpathogenic bacteria to human health has been well documented. 1-3 Many strains of lactic acid bacteria are commonly used for both food production and food preservation. Additionally, lactic acid bacteria have been used extensively as probiotics to promote gastrointestinal and genitourinary health, and these bacteria have been used to improve the immune system. 4-6 In general, naturally occurring and probiotic lactic acid bacteria have an excellent safety profile, and no major problems or health risks to humans have arisen during widespread use. 7-9 Among the lactic acid bacteria, Streptococcus viridans, a pseudo-taxonomic term for a large group of generally nonpathogenic microorganisms, are a commensal streptococcal bacteria that are either α-hemolytic, producing a green coloration on blood agar plates (hence the name viridans), or nonhemolytic. 10
The viridans streptococci constitute a significant portion of the resident oral microflora. Evidence suggests that certain viridans streptococci contribute to the oral well-being of humans. 11 Almost invariably, subgingival dental plaque taken from healthy periodontal sites was found to contain a significant proportion of 2 viridans streptococci species: Streptococcus oralis (previously Streptococcus sanguis type II) and Streptococcus uberis. Plaque taken from diseased sites, however, almost always lacked these species. 12 Hydrogen peroxide production by S oralis and S uberis was shown, in vitro, to inhibit the growth of 9 bacterial species implicated as periodontal pathogens. 13 Hydrogen peroxide production by S oralis strain KJ3sm was also shown to inhibit the growth of Aggregatibacter actinomycetemcomitans, the etiologic agent of localized aggressive periodontitis, in a gnotobiotic rat model. 14
Databases that quantify bacterial species present in healthy and diseased periodontal sites have shown a strong inverse relationship in human dental plaque between S oralis and S uberis and most periodontal pathogens. 11-14 The presence of infectious bacteria Aggregatibacter actinomycetemcomitans in a particular periodontal site was shown to be inversely proportional to the percentage of S oralis in that site, indicating that the negative interaction was “dose dependent.” 11 Haffajee et al 15 reported that successful treatment of active periodontal lesions correlated with recolonization of the treated site with S oralis. Together, these various lines of evidence indicate that certain indigenous viridans streptococci, notably S oralis and S uberis, play an important role in promoting periodontal health by inhibiting the outgrowth of microorganisms capable of causing periodontal disease.
Hillman 16 isolated Streptococcus rattus strain JH145, a spontaneous mutant, from the oral cavity of a human subject. JH145 lacks the enzyme activity L(+)-lactate dehydrogenase and thus the ability to produce lactic acid. Because of the similarity between Streptococcus mutans, the principal etiologic agent of dental caries, 17-19 and S rattus, it was proposed that the 2 species may compete with each other for the same niche in the plaque biofilm. 20 Johnson et al 21 used a Sprague-Dawley rat model to show that daily oral infusion of the spontaneous lactic acid–deficient mutant of S rattus, strain JH145, resulted in a significant decrease in the oral levels of an established wild-type strain of S mutans.
The findings from the above-mentioned in vivo and in vitro studies, including animal and human studies, have shown that the 3 natural human oral microorganisms, S uberis, S oralis, and the lactic acid–deficient S rattus, have beneficial biological activities and could be used to promote periodontal and dental health by maintaining the levels of these beneficial species at levels high enough to inhibit the outgrowth of pathogenic species. The combination of specific strains of these well-studied bacterial species has been formulated into a prototype probiotic called ProBiora3. The objective of the present study was to investigate the adverse effects, if any, of a standardized preparation of ProBiora3 following subchronic oral exposure to rats at daily doses of up to 109 colony-forming units (CFU) per strain per day for 90 days. The effects of the preparation were investigated in a dose-response study. The bacterial strains constituting ProBiora3 were also tested for their sensitivity to a panel of commonly used clinical antibiotics.
Method
Subchronic Toxicity Study
The study was performed according to a well-designed protocol following U.S. FDA Good Laboratory Practices for Nonclinical Laboratory Studies. All animals used in the testing were treated and housed humanely.
Animals
Male and female Sprague-Dawley rats, 24 days old at receipt, were used in the investigation. The animals were obtained from Taconic Laboratories (Hudson, NY). The animals were housed separately under BSL-2 (Bio Safety Level II for microbiological and biomedical laboratories) conditions for the duration of the study. The animals were allowed to acclimatize for 4 days before the initiation of experiments, with food and water available ad libitum. All animals were maintained on TD99366 diet obtained from Harlan Teklad (Madison, Wis).
Treatment
Following acclimatization, the rats were randomly divided into 3 groups (10 rats per sex per group). The selected animals were approximately 4 weeks old at the initiation of the experiment. The animals were treated orally (bucal cavity) with a blend of 3 microorganisms (ProBiora3) once per day, Monday through Friday, as follows. Animals in group 1 received 100 μL of sterile resuspension medium. Animals in group 2 received the probiotic formulation adjusted to deliver 106 CFU each of S uberis strain KJ2sm, S oralis strain KJ3sm, and S rattus strain JH145 per dose. Animals in group 3 received the probiotic formulation adjusted to deliver 109 CFU of each strain per dose. Immediately following the oral administration, the rat mouth was closed to avoid any loss of the blend. During the course of the subchronic study, all animals were provided ad libitum feed, until the day prior to the scheduled euthanasia. At completion of the 90-day treatment period, all animals (groups 1, 2, and 3) were euthanized.
Parameters Investigated
All animals were observed daily for any abnormal physical and behavioral changes. Individual animal body weights, food consumed, and water consumed were recorded at least weekly. Mean body weight and food and water consumption were calculated for the corresponding weekly intervals. Final body weights were recorded prior to the scheduled necropsy. At the conclusion of the treatment, blood samples were collected for clinical evaluation (hematology and serum chemistry) under anesthesia prior to scheduled necropsy. To minimize the killing of animals and because the available evidence did not indicate any potential adverse effects of the microorganisms of ProBiora3, blood and tissue samples from 6 animals, 3 males and 3 females, from each group were collected. The blood was used immediately for hematology, and serum was separated to determine chemistry parameters. Following blood collection, the animals were administered a lethal dose of sodium pentobarbital, and necropsy was conducted on the animals. The necropsies included, but were not limited to, examination of the external surface, all orifices, and the cranial, thoracic, abdominal, and pelvic cavities. More than 40 tissues and organs that were harvested from the rats were weighed and fixed in 10% buffered formalin. Hematological and blood chemistries were performed in a blinded fashion by the Veterinary Medical Teaching Hospital, University of Florida, Gainesville, Florida. The hematological panel of tests included all standard parameters such as lymphocytes, monocytes, eosinophils, red blood cells, segs, hemoglobin, hematocrit, mean corpuscular volume, mean corpuscular hemoglobin, mean corpuscular hemoglobin concentration, and platelets. Blood chemistry tests included alkaline phosphatase, activated alanine aminotransferase with P5P, activated serine aminotransferase with P5P, total bilirubin, total protein, albumin, globulin, albumin/globulin ratio, calcium, phosphorus, creatinine, blood urea nitrogen, glucose, cholesterol, magnesium, sodium, potassium, chloride, carbon dioxide, and anion gap. Twenty-four 1-way analyses of variance (ANOVA) were conducted separately for males and for females to determine whether the low and high doses of the probiotic treatment differed from the control group with regard to any of the blood chemistry and hematology tests conducted.
Necropsy and histopathological evaluation of harvested tissues were performed in a blinded fashion by Gene Logic Laboratories (Gaithersburg, Md). The pathology evaluation of animal tissues was conducted following current US FDA Good Laboratory Practices for Nonclinical Laboratory Studies and in accordance with Gene Logic Laboratories' standard operating procedures. All tissues received by Gene Logic Laboratories were embedded in paraffin, sectioned at nominal 5 μm for slide preparation, and stained with hematoxylin and eosin. The tissues were then evaluated microscopically by a board-certified veterinary pathologist. The organs and tissues examined included brain, spinal cord, thyroid gland, parathyroid gland, esophagus, trachea, aorta, lung, heart, mandibular salivary gland, mandibular lymph node, tongue, kidney, thymus, adrenal gland, liver, spleen, stomach, duodenum, pancreas, small and large intestines, rectum, mesenteric lymph node, uterus/cervix/ovary/vagina (females), epididymides/testes/prostate/seminal vesicle (males), urinary bladder, skin, skeletal muscle, sciatic nerve, eye with optic nerve, Harderian gland, sternum, and femur.
Statistical Analysis
All statistical tests were performed using appropriate computing devices or software. Data were analyzed using statistical methods, and values were presented as mean with the standard deviation and the number of animals used to calculate the mean. Body weight, food and water consumption, hematology, and clinical chemistry test results were analyzed using 1-way ANOVA separately for each sex to determine intergroup differences.
Antimicrobial Susceptibility Study
The 3 strains of oral microorganisms contained in the probiotic formulation were evaluated for antimicrobial susceptibility at Focus Diagnostics (Cypress, Calif) using a standard panel of antibiotics. Briefly, an agar plate with an overnight culture of 1 of the 3 microorganisms was spotted with serial dilutions of 1 of 9 antibiotics: cefepime, ceftriaxone, chloramphenicol, erythromycin, levofloxacin, linezolid, meropenem, penicillin, and vancomycin. Based on these observations, minimum inhibitory concentrations (MIC) and minimum bactericidal concentrations (MBC) were calculated. For the reported results, MIC is defined as the lowest concentration of an antibiotic that inhibited the in vitro growth of a selected organism and is reported as micrograms per milliliter of the antibiotic, and MBC is the lowest concentration of an antimicrobial agent needed to kill 99.9% of the initial inoculum. The strains were scored as susceptible, intermediate, or resistant to each antibiotic based on established Clinical and Laboratory Standards Institute interpretive guidelines.
Results
In the subchronic toxicity study, all animals survived until the scheduled necropsy in all the 90-day study groups. No tolerability problems or treatment-related adverse events were noted by daily in-life physical and behavioral observations of the animals in any of the groups. The effects of ProBiora3 administration on animal weight gain and food and water consumption in male and female rats are presented in Tables 1 through 3. These results indicate that daily administration of up to 109 CFU of each of the 3 probiotic strains for 14 weeks had no adverse effects on animal weight gain or on the consumption of food and water.
Effect of ProBiora3 on Body Weights (g) in Male and Female Rats
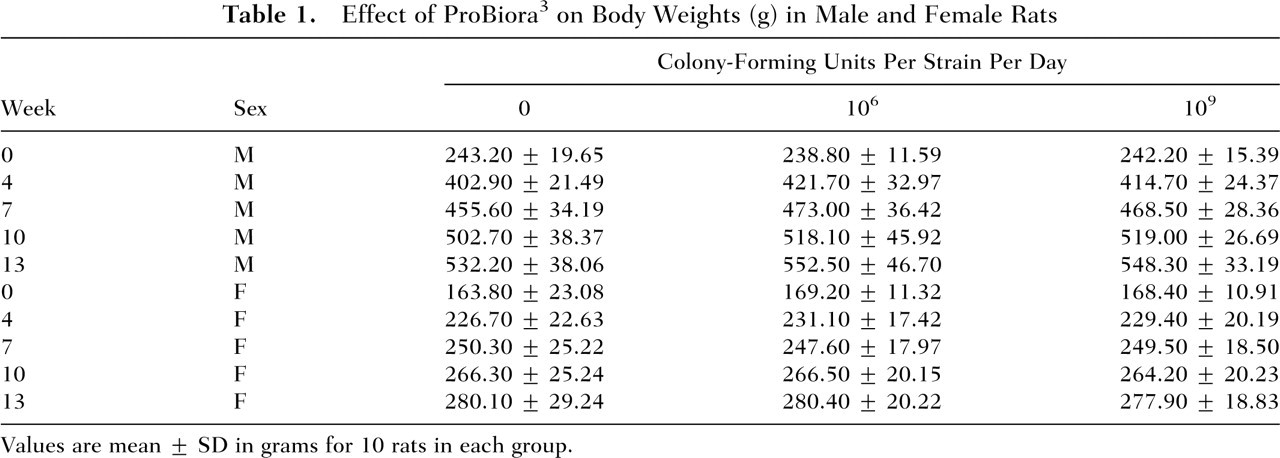
Values are mean ± SD in grams for 10 rats in each group.
Effect of ProBiora3 on Feed Consumption (g) in Male and Female Rats
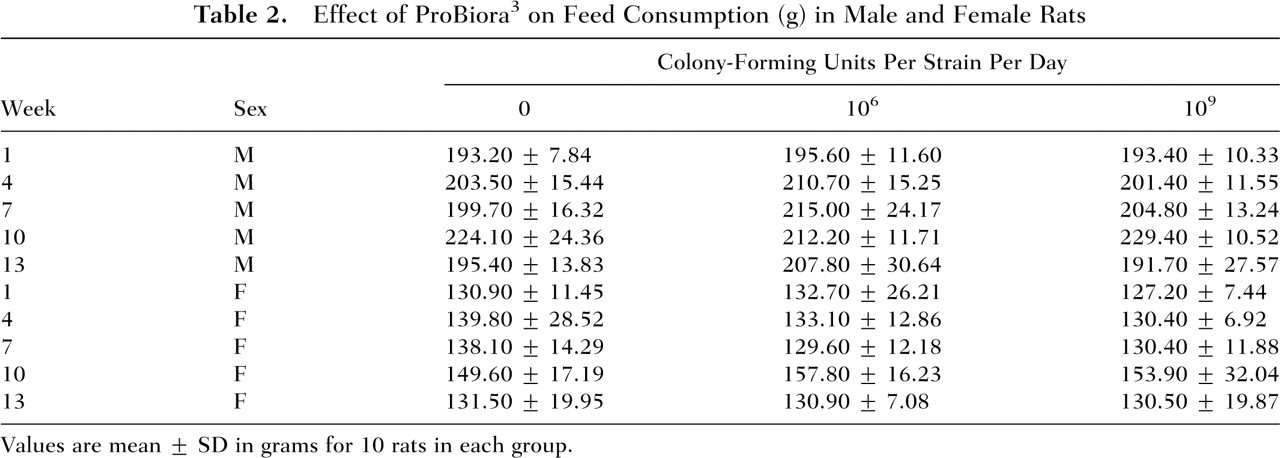
Values are mean ± SD in grams for 10 rats in each group.
Effect of ProBiora3 on Water Consumption (mL) in Male and Female Rats
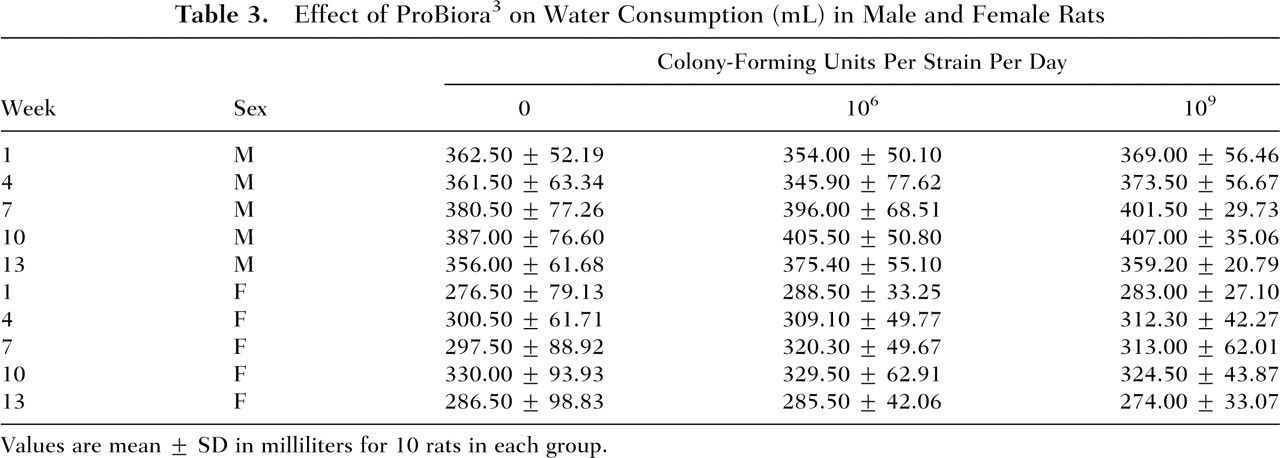
Values are mean ± SD in milliliters for 10 rats in each group.
There were no treatment-related adverse effects of ProBiora3 preparation on hematology and blood chemistry parameters in male and female rats. Table 4 contains group summary data (mean ± SD), separated for male and female animals, of the 20 serum blood tests for the 2 treatment groups and the control group. The red blood cell analysis did not reveal any notable differences or abnormalities in morphology (size, shape, and color) among the 3 groups in this study. Table 5 contains group summary data, separated for male and female animals, of the 4 quantitative white cell differential measures for the 3 groups of animals. Results obtained were compared separately for males and females using a series of 24 one-way ANOVAs to ensure that the low and high doses of the probiotic bacterial treatment did not differ from the control group with regard to the mean blood chemistry and hematology test results. Treatment condition served as the between-group factor. Based on the number of tests (ie, 24), a family-wise α of .0021 was adopted to control for Type I error or the incorrect detection of a false finding attributable to the number of statistical tests conducted. Only CO2 in male rats differed as a function of the treatment (P = 0.001). Follow-up t tests with Bonferroni correction indicated that the control group had significantly reduced CO2 levels relative to the low-dose and high-dose treatment groups (P ≤ .006). With this possible exception, there were no treatment-related, biologically significant effects of ProBiora3 administration on hematology or serum chemistry parameters in male and female rats. Generally, changes in CO2 level suggest loss or retaining of fluid, leading to imbalances in the body’s electrolytes. However, electrolyte analysis did not reveal any significant changes. Additionally, changes in CO2 level were noted in male rats only and not in females. All of these factors indicate that the change in CO2 level or low levels of CO2 in the male control group may not be related to treatment.
Summary of Serum Chemistry Data Following Oral Administration of ProBiora3
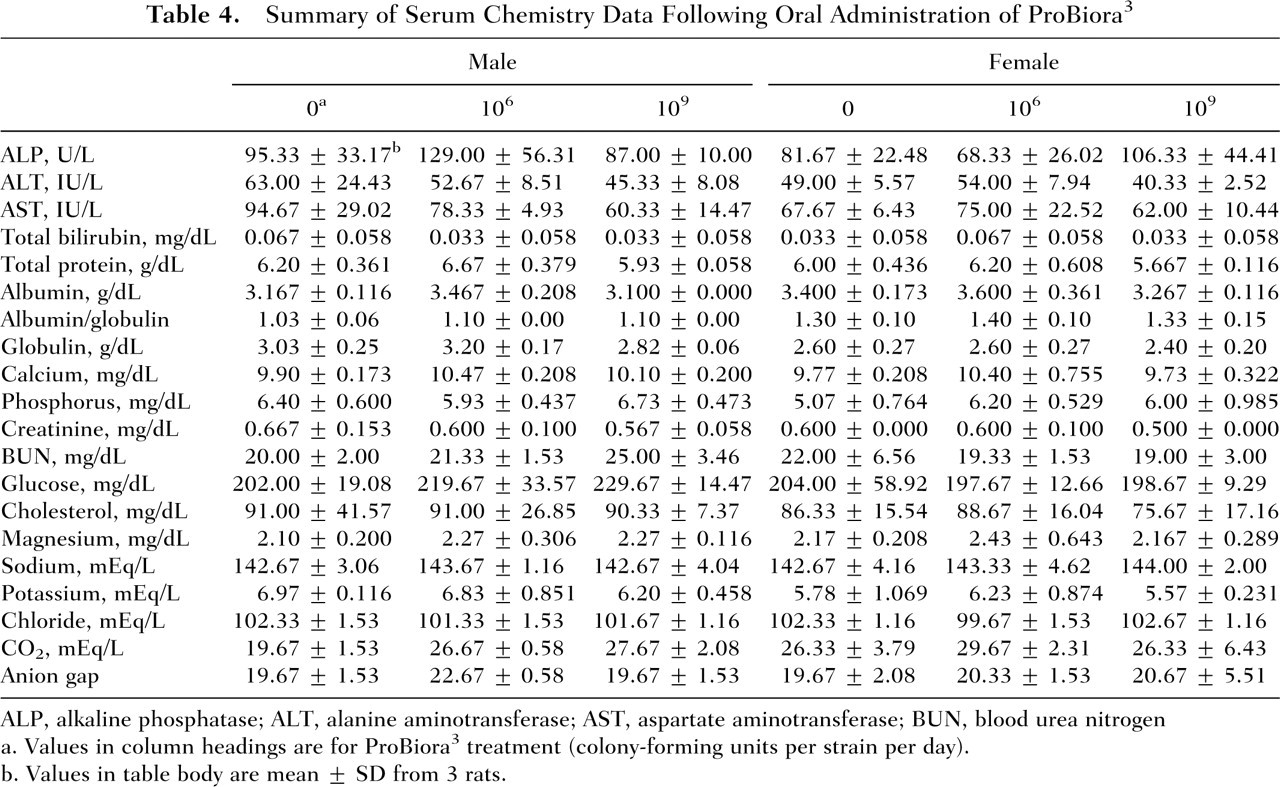
ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; BUN, blood urea nitrogen
Values in column headings are for ProBiora3 treatment (colony-forming units per strain per day).
Values in table body are mean ± SD from 3 rats.
Summary of Complete Blood Count Differential Data Following Oral Administration of ProBiora3
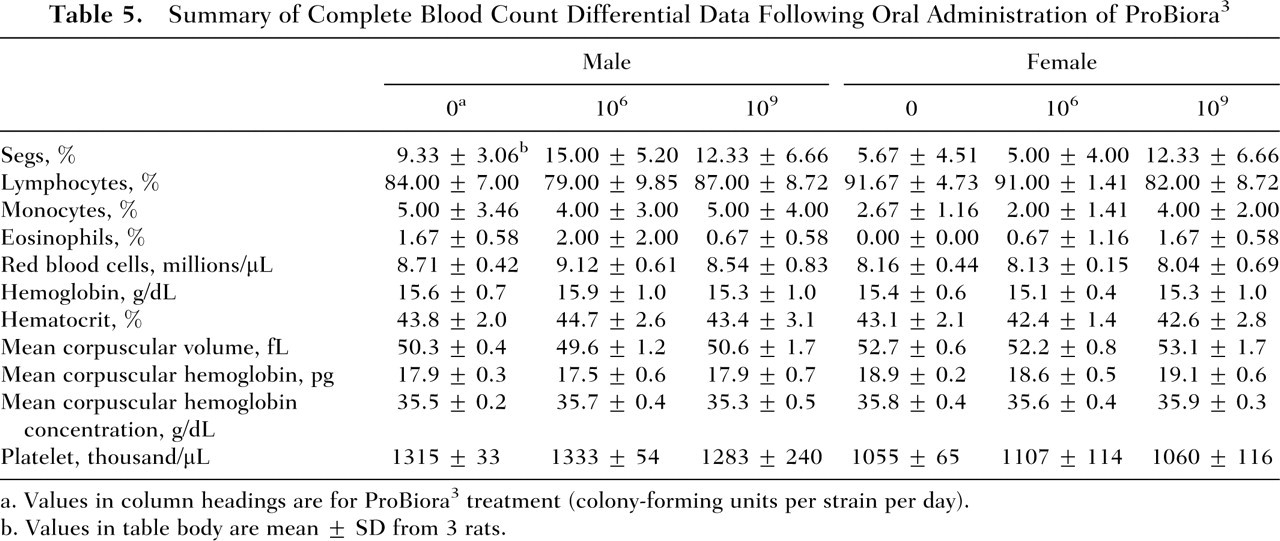
Values in column headings are for ProBiora3 treatment (colony-forming units per strain per day).
Values in table body are mean ± SD from 3 rats.
Administration of ProBiora3 to male and female rats did not cause any treatment-related changes in absolute and relative organ weights (data not shown). There were no treatment-related macroscopic findings. No abnormal findings were noted in size or appearance of the organs or tissues examined during necropsy. A summary of the histopathological findings for relevant tissues and organs that could potentially be affected by the administration of the oral probiotic product is presented in Table 6 . Although exposure to any ingested bacterial strain is most likely limited to the gastrointestinal tract, a histopathological examination of an extensive array of tissues was conducted. Incidental microscopic tissue changes were present in various tissues in representatives of both control and experimental animals, including the liver, thyroid gland, spleen, pancreas, prostate, and Harderian gland. All changes were generally sporadic and minimal to mild, with the exception of splenic hemosiderin accumulation, which occurred in all animals and ranged from minimal to moderate. Evidence of acute congestion and/or hemorrhage was additionally present in the lung, salivary gland, thymus, adrenal gland, and ovary, indicative of blood stasis at necropsy in few animals. Splenic hemosiderin accumulation and hepatic extramedullary hematopoiesis frequently occur in rats and were considered to be incidental findings. It was concluded that there were no pathology findings indicating that treatment with the probiotic test article had any adverse macroscopic or microscopic effects.
Representative Group Summary Data From Histopathology Observations
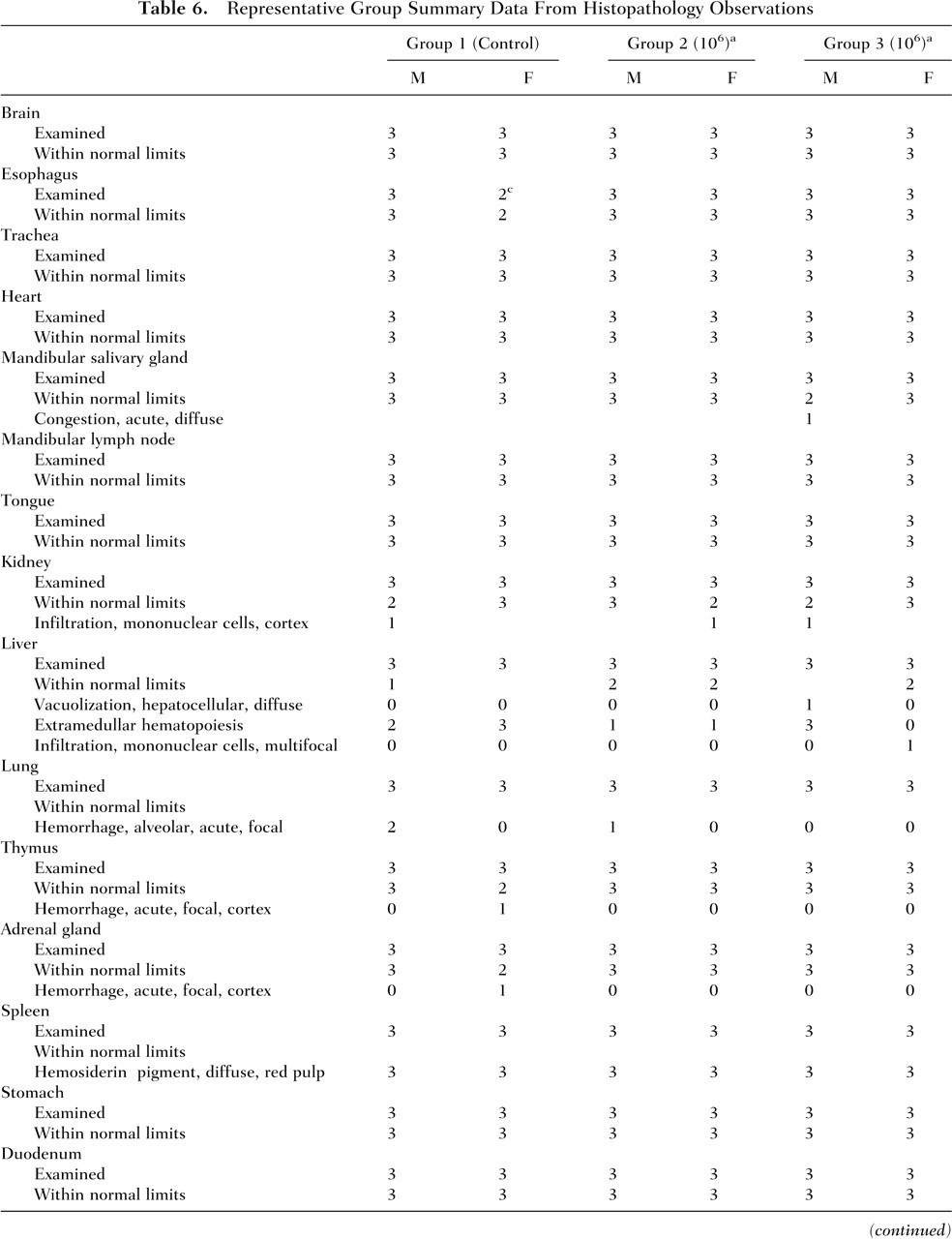
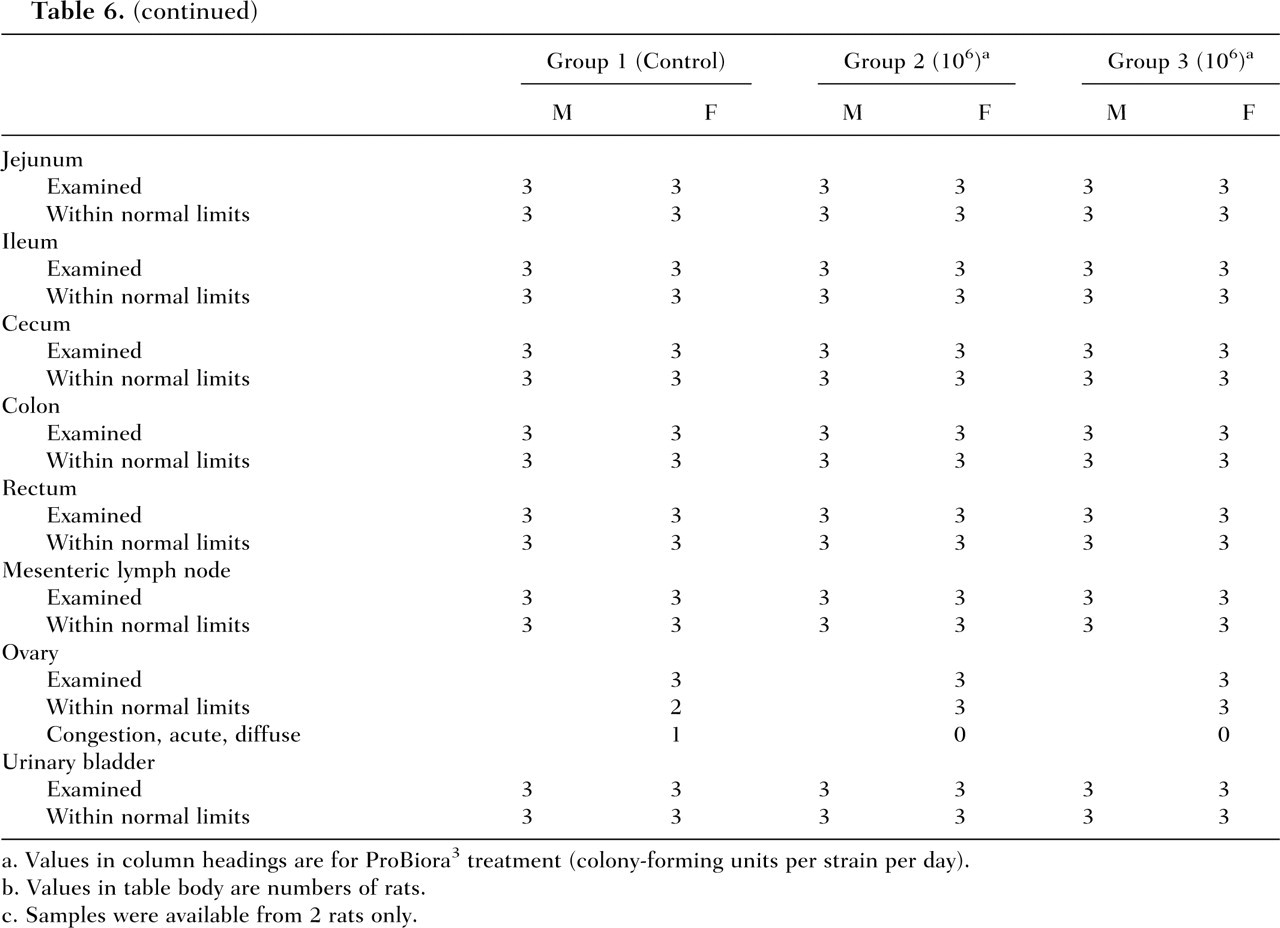
Values in column headings are for ProBiora3 treatment (colony-forming units per strain per day).
Values in table body are numbers of rats.
c. Samples were available from 2 rats only.
Table 7 contains a summary of the test results for MIC and MBC for each of the 3 probiotic strains to a panel of 9 commonly used antibiotics. Based on the experimentally determined MIC values and the Clinical and Laboratory Standards Institute’s interpretive guidelines, KJ2sm, KJ3sm, and JH145 were determined to be susceptible to all of the antibiotics tested.
Summary of the Test Results for Minimum Inhibitory Concentrations (MIC) and Minimum Bactericidal Concentrations (MBC)
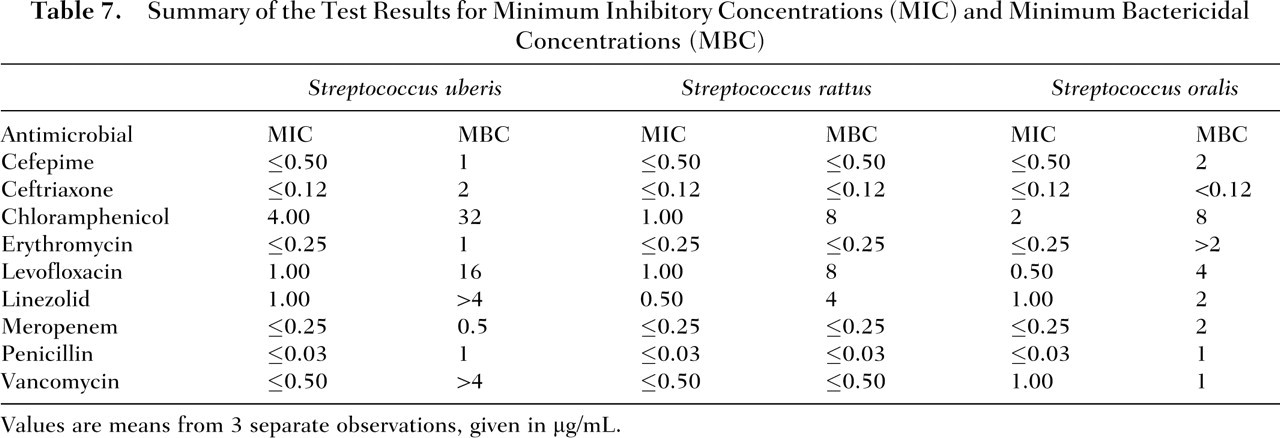
Values are means from 3 separate observations, given in μg/mL.
Discussion
The rapid growth in bacterial resistance to antibiotics has increased research efforts to develop countermeasures to this serious health threat. One preventive approach that has received considerable attention lately involves the use of specific effector strains to colonize human tissues and replace known bacterial pathogens. 22 For example, the ingestion of probiotic bacteria, particularly lactic acid-producing strains, is commonly practiced to promote well-balanced intestinal microflora. In the last several years, probiotics have also been investigated with regard to their ability to help maintain oral and dental health. 23,24 Strains intended to promote gastrointestinal health are not likely to be ideally suited to negatively interact with oral pathogens. For example, studies using strains of Lactobacillus rhamnosus GG 25,26 and Lactobacillus reuteri 27 have found variable effects on the levels of S mutans. Also, the observation that Lactobacillus species are known to promote progression of dental caries lesions 28-30 requires that the pathogenic potential of these strains be carefully addressed.
The 3 bacterial species used in the present study are known to be normal colonizers of the human mouth and, as such, address a major issue for successful application as a probiotic for use in improving oral and dental health. Plaque samples from healthy gingival sulci typically contain large numbers of viridans streptococci, including S oralis and S uberis. These species have been extensively characterized with regard to their ability to interfere with the growth of periodontal pathogens. 11-14 The 2 particular strains used in the present study, S oralis KJ3sm and S uberis KJ2sm, were isolated from the mouth of a human volunteer and have been extensively characterized. 13 Although not as common as S mutans or Streptococcus sobrinus, S rattus is found as a normal resident of the human oral flora. 10 Because of its very similar physiology, S rattus was classified as a member of the S mutans group until relatively recently. 10 The lactic acid-deficient strain, S rattus JH145, used in the present study is a spontaneous mutant of the human S rattus isolate BHT-2. 15 Daily administration of JH145 into the oral cavity of Sprague-Dawley rats was shown 31 to significantly reduce the levels of an indigenous S mutans strain, presumably by competing for attachment sites, essential nutrients, and other unknown factors. In addition to being a normal colonizer of the human mouth, any bacterial strain to be used as a probiotic must be safe. A probiotic microorganism should not be pathogenic, should be genetically stable, and should not possess the ability to transfer antibiotic resistance genes to other indigenous microorganisms. 32 The results of the present safety evaluation of the 3 probiotic strains demonstrate that these particular strains were well tolerated by the experimental animals over the 14 weeks of probiotic administration, at levels up to 109 CFU/d. No biologically significant adverse events or safety problems were observed with any of the physical signs or the blood chemistry (except for CO2), hematology, or histopathology variables measured. All reported test values were within the expected range for healthy male and female adult Sprague-Dawley rats at the conclusion of the study. ProBiora3 treatment resulted in a significant increase in serum level of CO2 (bicarbonate) in male rats. An increased bicarbonate level may result either from a metabolic alkalosis or as compensation to respiratory acidosis. Follow-up t tests with Bonferroni correction indicated that the control group had significantly reduced CO2 relative to the low- and high-dose treatment groups. The CO2 level in the control male group was also lower than in the control female group. In the absence of any other changes (hypokalemia) and lack of effects in female rats, the increases in serum CO2 noted in male rats following administration of ProBiora3 were not considered to be adverse.
With regard to any slight possibility of endocarditis or other systemic infection caused by probiotic administration, the therapy for a diagnosed case of bacteremia would typically involve the administration of a course of antibiotics. The 3 probiotic strains used in this study were shown to be susceptible to a panel of conventional antibiotics, including penicillin. Thus, these probiotic bacteria are not resistant to any of the commonly used antibacterials, and a patient exhibiting signs of bacteremia or endocarditis could be treated effectively if the microorganism causing the infection has been specifically identified as one of the probiotic viridans streptococci. These 3 strains do carry spontaneous mutations that confer resistance to streptomycin sulfate, which is an antibiotic not commonly used in modern medical practice. This resistance marker has been used to permit easy recovery and identification of the 3 probiotic strains in experimental and clinical settings.
The combination of KJ2sm and KJ3sm for maintenance of periodontal health, with JH145 for the maintenance of dental health, constitutes the active ingredients in ProBiora3.
The safety of and tolerance to these 3 probiotic bacteria administered at levels up to 109 CFU per day were demonstrated in the subchronic toxicity study. The absence of antibiotic resistance that was demonstrated in the antibiotic susceptibility study is another important criterion that supports the selection of these orally isolated strains for use as a probiotic treatment. Based on the results of this study, the no-observed-adverse-effect level (NOAEL) of ProBiora3 was 109 CFU per strain, or the highest dose tested in this study. The NOAEL in the present study is based on a 5-day exposure regimen per week, and its extrapolation to a 7-day period will be 109 × 5/7 = 0.7143 × 109 CFU per strain per day. Considering that the average body weight of rats in this study was 330 g, the equivalent NOAEL of the probiotic test material for male and female rats will be 0.7 × 109 × 1000/330 = 2.164 × 109 CFU per kilogram of body weight per day for each strain.
Footnotes
Acknowledgments
ProBiora3, KJ2sm, KJ3sm, and JH145 are trademarks of Oragenics, Inc, Alachua, Florida. J. Hillman, E. McDonell, and R. Zahradnik are employees of Oragenics, the distributor of ProBiora3.
