Abstract
Comprehensive care services (SAIs) play a pivotal role in implementing preventive strategies by offering treatments that have demonstrated efficacy, as exemplified by dolutegravir (DTG). This medication has been employed in various clinical settings because of its pronounced genetic barrier and high tolerance. To characterize the clinical and epidemiological use of dolutegravir in people living with HIV (PLHIV) treated for SAI in Honduras. Cross-sectional studies and secondary analyses of the 2020 to 2021 phenotypic database were performed with SPSS (version 20.0). Ethics Committee approval (057-2021). Out of 2,984 records with sociodemographic data, 44% were women, and 56% were men. The population included 2,221 general population patients, 684 men who have sex with men (MSM), 185 Garifuna, 36 trans women, 8 bisexuals, 6 pregnant women, and 2 prisoners. The average age was 39.5 years. First-line ARVs are the most commonly used type of SAI. The National Cardiopulmonary Institute was the center with the highest use of dolutegravir, with 37.7% of patients receiving it. Among the 3,533 antiretroviral therapy (ART) patient records, 80.1% achieved complete viral suppression (viral load <1,000 copies), with a 95% confidence interval of 78.7%-81.3%. High viral loads were observed in PLHIV receiving ART, regardless of the population type. Monitoring of antiretroviral resistance through phenotypic and genotypic testing is crucial for optimizing HIV treatment in Honduras.
The expansion of antiretroviral coverage has been a fundamental pillar in the fight against the HIV epidemic; however, the emergence of resistance to these drugs has become an increasing challenge that requires the implementation of effective measures (UNAIDS, 2023; Zaniewski et al., 2024). In the context of Latin America, and particularly in Honduras, comprehensive care services (SAI) have emerged as a crucial element in the HIV response. The creation of the first SA in 2002, with the support of the Global Fund, marked a significant change in the region’s response to the epidemic, particularly in the implementation of antiretroviral therapy (ART) and pharmacological surveillance of resistance (SESAL, 2022).
According to the 2023 report of the Joint United Nations Program on HIV/AIDS (UNAIDS), HIV prevalence in the population aged 15 to 49 years is 0.7%, although significantly higher rates are observed in certain demographic groups, such as sex workers (FSWs) (2.5%), men who have sex with men (MSM) (7.5%), and transgender people (10.3%). According to the 2013 National Population and Housing Census conducted by the National Institute of Statistics (INE), the Garifuna population was estimated at 46,789 people in 2018, with significant settlements in urban areas; however, most are in coastal communities along the Atlantic coast. The prevalence of HIV infection in this population is, in fact, significantly higher than in the general population, with prevalences reaching 4.5%, and prevalences close to 8% have been mentioned (Paz-Bailey et al., 2009).
National resistance surveys as a World Health Organization (WHO) strategy for monitoring and surveillance of HIV drug resistance (HIVDR) have been adopted in 48 countries between 2004 and 2018. Preliminary findings from resistance surveys in at least 12 countries reported 10% resistance to first-line ART. As ART coverage expands globally, the prevalence of FRHIV to NRTI drugs has increased. Some publications in East and Southern Africa report increases in 29% and 23%, respectively; other prevalences on the African continent, in the West and Central region, 17%, and in Latin America, prevalences of up to 15% are estimated, and in Asia, 11% (WHO, 2021). HIVDR is a problem in Latin American countries, and national survey data show a trend of increasing resistance, especially to NNRTIs, reaching moderate to high levels in countries such as Mexico, Argentina, Nicaragua, and Panama (Aguilar et al., 2022; Cecchini et al., 2025; WHO, 2021). Results from the antiretroviral resistance survey in Honduras (2016–2017) showed 29.1% of people with primary HIV infection were resistant to non-nucleoside reverse transcriptase inhibitors (NNRTIs), which are used as first-line treatments (SESAL, 2018). This finding was an alarming paradigm shift in guidelines at the national level, adopting WHO recommendations (2016–2018) to incorporate dolutegravir (DTG) as the preferred first-line option in adults and adolescents. Dolutegravir (DTG) has proven its efficacy as a key component of comprehensive HIV care services and is used in a variety of combinations and clinical settings due to its high genetic barrier to resistance and tolerability (Cruciani & Parisi, 2019).
Despite advances in the incorporation of DTG in the first line of treatment in Honduras, there is limited information on its impact on virologic suppression, especially in key populations and vulnerable groups. This study analyzes the implementation of DTG in the country’s SAI’s and its relationship with virological suppression rates reported from national registries to support decision-making and strategies in the national response to HIV.
Methodology
This retrospective observational study is based on the analysis of secondary data from the main comprehensive care services (SAI) in Honduras and was conducted in accordance with STROBE guidelines for observational studies (Figure 1). The objective of this study was to provide a detailed clinical and epidemiological characterization of DTG use in people living with HIV (PLHIV) in Honduras. Using a cluster sampling method, with selection of SAIs from regions with the highest reported prevalences of antiretroviral drug resistance (ARVDR).
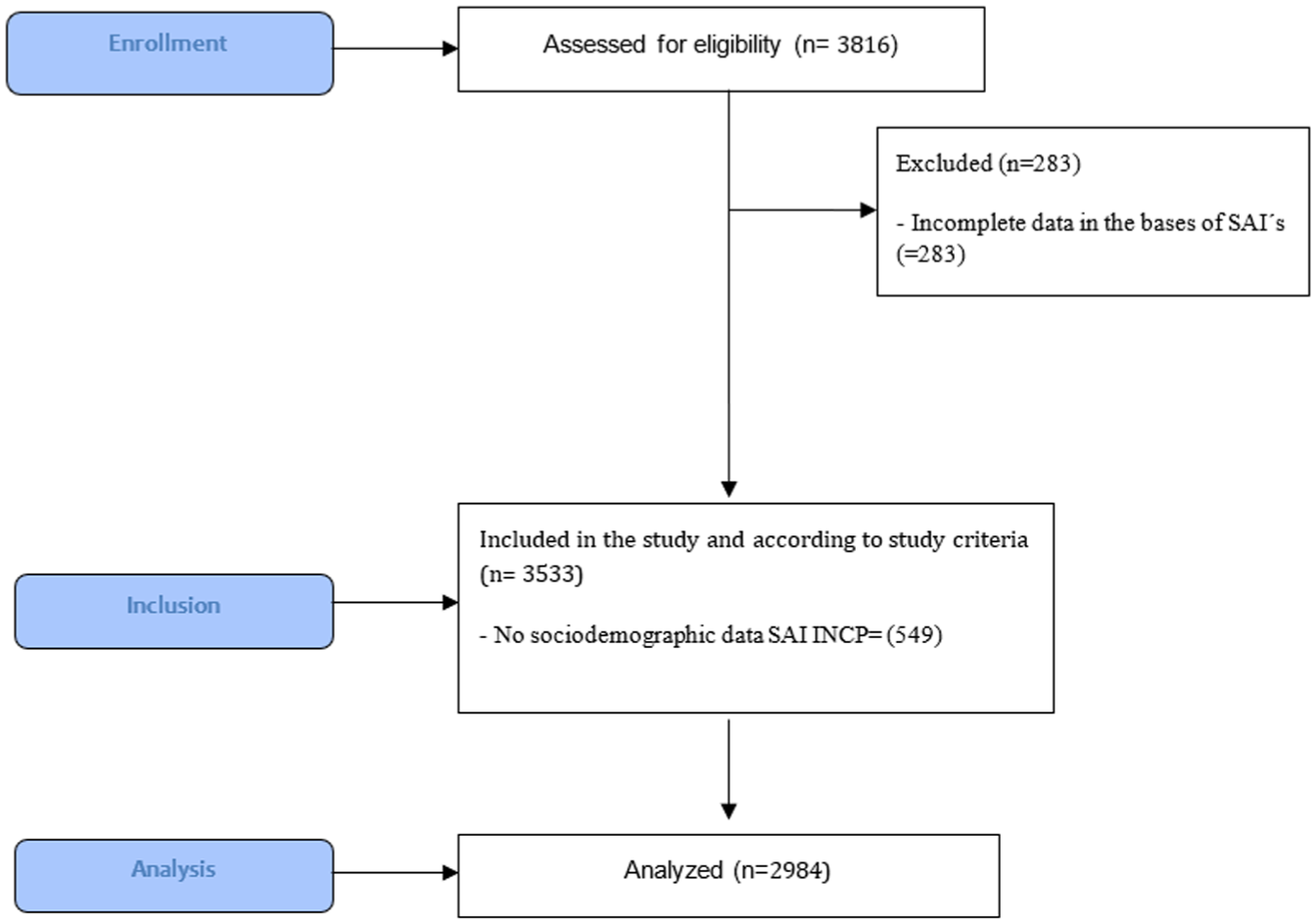
Flow Diagram, Missing Data Handling
The selection was made from the following facilities: Miguel Paz Barahona (SAI-MPB), Ceiba Metropolitana (SAI Ceiba), Hospital Escuela (SAI HE), and the National Cardiopulmonary Institute (SAI INCP). Which allowed us to examine the impact of DTG adoption in centers where resistance to other ARVs was more prevalent, the following criteria were taken: (1) prevalence of HIVDR, selecting sites with high reported rates of ART resistance, and (2) representation of diverse populations, including individuals from different demographic groups.
The analysis of the main sociodemographic variables, type of population, and virological suppression was performed. The main reason for excluding the SAI INCP from the sociodemographic analysis was incomplete sociodemographic variables, such as sex, ethnicity, and type of population. We did not report sex/gender-specific suppression because the sociodemographic fields were incomplete at one center. Virological suppression in each SAI was evaluated using a viral load (VL) <1,000 copies/mL. However, it was not possible to estimate suppression by ART duration (12 ± 3 and 48 ± 3 months) due to incomplete data. In resource-limited settings, the <1,000 copies/mL cutoff is commonly used as a practical balance between accessibility and diagnostic accuracy (SESAL, 2023).
In contrast, thresholds of <200 or <50 copies/mL are internationally preferred for long-term monitoring. However, the <1,000 copies/mL cutoff was used in alignment with the national ART monitoring framework and the practical constraints of the Honduran health system and available registries. Statistical analysis was performed using SPSS software (version 20.0). The Kolmogorov-Smirnov test was used to evaluate the normality of the continuous variables. The descriptive analysis of the study variables was performed using the Student’s
Results
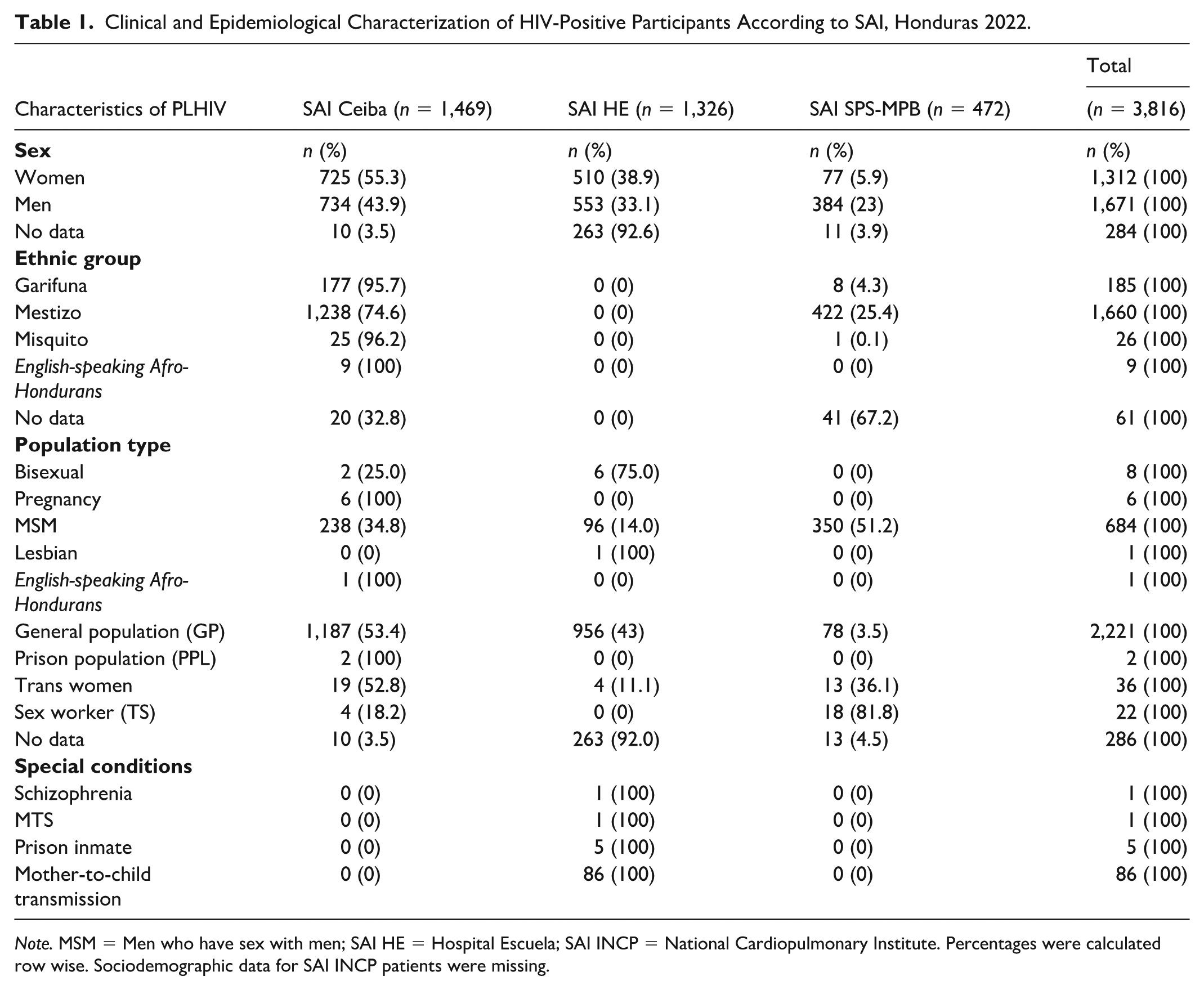
Data from the main referral SAIs nationwide were considered, including sociodemographic variables such as sex, ethnicity, and population type among people living with HIV (PLHIV). Of these, 38.5% were affiliated with SAI Ceiba-Metropolitano, 34.7% with SAI Hospital Escuela, 14.4% with the National Cardiopulmonary Institute, and 12.4% with SAI SPS. The cohort from the National Cardiopulmonary Institute (n = 549 PLHIV) was excluded from the sociodemographic analysis due to the lack of sociodemographic data.
Of the 2,984 complete records containing sociodemographic data, 44% were women and 56% men. Regarding the categorization of risk groups, the study included 2,221 individuals from the general population (GP), 684 MSM, 185 Garifuna, 36 trans women, 8 bisexuals, 6 pregnant women, and 2 prisoners. The mean age was 39.5 years (37.2 years for men and 42.3 years for women), indicating a representative distribution of the population served in the SAI’s. (Table 1)
Clinical and Epidemiological Characterization of HIV-Positive Participants According to SAI, Honduras 2022.
Regarding the use of ART, SAI Ceiba Metropolitana used 48.32% of first-line ARVs, SAI Instituto Cardiopulmonar used 37.24% of third-line ARVs, and SAI Hospital Escuela used 56.14% of second-line ARVs (Table 2). With respect to dolutegravir (DTG), the Instituto Nacional Cardiopulmonar was the institution with the highest use of DTG, with 37.7% of its patients receiving this treatment (Table 3).
Antiretroviral Therapy Used in SAI, Honduras 2022.

SAI SPS-MPB did not provide treatment line data for the PVHI cohort.
Use of Dolutegravir (DTG), Data Analysis From a Sample of SAIs, Honduras 2022.

Source: SAI database, SESAL. HIV Resistance Platform, Honduras 2022.
Of a total of 3,533 medical records of patients on ART, 2,828 medical records were found to have a VL of less than 1,000 copies, with a total virological suppression of 80.1%, with a 95% confidence interval (78.7%–81.3%). Positive differences in the centers that migrated to DTG: Virologic suppression by type of population: MSM: 73.4% (95% CI: 69.8%–76.8%), GP: 50.7% (95% CI: 48.6%–52.9%), trans women: 68.7% (95% CI: 51.4%–82.0%), and Garifuna: 83.4% (95% CI: 76.9%–88.3%) (SAI Ceiba).
Regarding VL suppression, SAI-Ceiba Metropolitana reported the lowest percentages 55.7%; SAI HE 87.9%, SAI Instituto cardiopulmonar 87.3%, and SAI SPS 89.9% with the best performance. In the ART centers that have implemented DTG, the results show notable differences in virologic suppression: Ceiba ART Center: 55.7% (95% CI: 53.2%–58.2%), HE ART Center: 87.9% (95% CI: 85.8%–89.7%), SAI Cardiopulmonary Institute: 87.3% (95% CI: 84.8%–89.4%), SAI SPS: 89.9% (95% CI: 86.6%–92.5%)
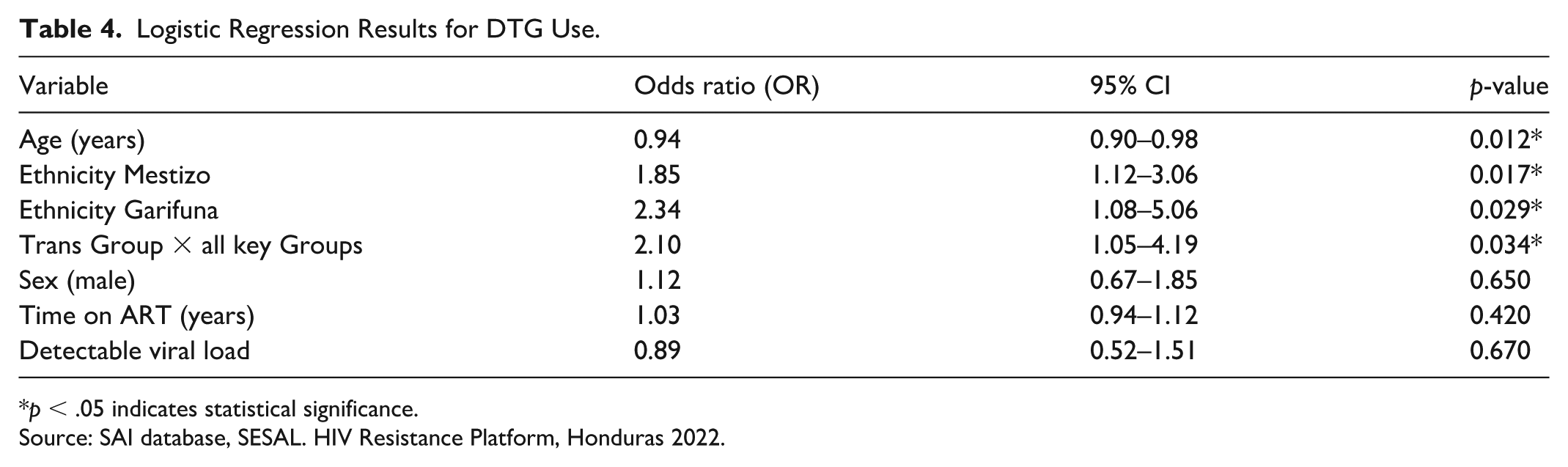
The logistic regression model revealed that for each additional year of age, the odds of using DTG decreased by approximately 6%, holding all other variables constant. In contrast, sex, time on ART, and VL were not significantly associated with DTG use (
Logistic Regression Results for DTG Use.
Source: SAI database, SESAL. HIV Resistance Platform, Honduras 2022.
Discussion
The present study addresses the interpretation of findings derived from secondary analysis of national databases, with the purpose of identifying the main demographic characteristics, risk factors, treatment regimens, and treatment resistance of people living with HIV (PLHIV) attending comprehensive care services (SAI) in Honduras (Alkhawaldeh et al., 2023).
In Honduras, most people with HIV rely on the public health system, which centralizes care in tertiary hospitals and reflects persistent barriers to access. However, STI surveillance services (VICITS) and SAIs have played a key role in monitoring the epidemic, providing essential prevalence data from the populations they serve and strengthening national HIV surveillance. Although this analysis does not directly estimate the association between DTG use and virologic suppression at the individual level, the approach adopted allows the identification of trends and patterns of virologic suppression linked to DTG implementation. A higher proportion of virologic suppression was observed in centers that better incorporated dolutegravir (Zaniewski et al., 2024). It also allows stratification of demographic groups, providing essential information for decision-making and the design of strategies aimed at mitigating HIV transmission.
Evidence supports the use of DTG for its efficacy and good tolerability in diverse populations, with most patients achieving viral suppression, making it a viable therapeutic option for HIV treatment, achieving virologic suppression rates close to 95% among users of combination regimens (Bowman et al., 2023; Doro Altan et al., 2025; Gandhi et al., 2025; Majula & Mweya, 2024). Multicenter studies such as GEMINI-1 and 2, phase 4 STAT and D2ARLING trials have demonstrated the high efficacy of DTG plus lamivudine as a first-line regimen for ART initiation (Cordova et al., 2025; Schneider et al., 2024).
In the present cohort, most patients seen in the SAI used first-line antiretrovirals, followed by third-line and second-line antiretrovirals. The observed differences in DTG use among the different populations can be attributed mainly to factors of age, sex, reproductive status, comorbidities and presence of viral resistance, as well as access policies and regulatory recommendations (Chandasana et al., 2024). These results highlight that sociodemographic and identity-related factors play a significant role in DTG use.
The logistic regression model showed a significant effect of age on the use of DTG. The younger the age, the greater the probability of receiving this drug, odds ratio (OR = 0.94). Pharmacokinetic studies show that age does not significantly affect DTG metabolism. Efficacy and safety in the young population is comparable among different age groups with similar rates of virologic suppression and adverse events (Jacobs et al., 2024). Other variables, such as sex, time on ART, and VL, did not show statistically significant associations.
On the other hand, mestizo people presented a higher proportion of DTG use compared to other ethnic groups. This underscores the importance of developing specific interventions and inclusive health strategies that take into account age, gender diversity, and ethnicity, especially among historically marginalized populations. The GP showed a viral suppression rate of only 50.7%, despite the widespread implementation of ART, which shows important barriers to achieving sustained virologic control in this group. Likewise, differences were observed in the implementation of DTG in the different care centers. The chi-square analysis confirmed a significant association between DTG use and care center (χ² = 530.3,
The case of SAI La Ceiba stands out, where 58% of people do not have DTG incorporated into their treatment regimens, with a virological suppression rate of only 55.7%. In contrast, SAI Instituto Cardiopulmonar and HE, which have incorporated DTG into their regimens, report higher suppression rates (87.9% and 87.3%, respectively). These differences demonstrate the urgent need to promote its inclusion in accordance with WHO recommendations and the scientific evidence supporting its positive impact on treatment efficacy (Figueroa et al., 2025; Paton et al., 2021).
Honduras relies on programmatic data from VICITS and SAI’s to monitor response in key populations, which means that surveillance only captures people accessing these facilities. According to programmatic data from Honduras, HIV prevalence among individuals aged 15–49 is 0.30%. However, rates are substantially higher in key groups, including MSM (6.9%–11.7%) and transgender women (17.2%–33.3%), with incidences up to 1% higher among MSM (José, 2019; Kim et al., 2013). Nevertheless, we recognize the importance of including these groups in analyses, as they may have distinct clinical and epidemiological characteristics that merit attention. In low- and middle-income countries, various socioeconomic factors influence key outcomes and targets related to HIV reduction and spread (Doblado-Maldonado et al., 2025; Inzaule et al., 2019; Vitoria et al., 2018). These results further demonstrate that, within specific populations such as the Trans and Garifuna populations, viral suppression levels are notably higher (68.7% and 83.4%, respectively) and are associated with higher odds of DTG use or adherence (OR = 2.10 and 2.34, respectively). The higher rates of suppression in these groups are encouraging, considering the high prevalence of HIV, and reflect the positive impact of health care strategies aimed at improving access and adherence (Paz-Bailey et al., 2012).
Although Garifuna in general showed a higher percentage of viral suppression compared to the GP, the data should be interpreted with caution, as the magnitude of incomplete records could lead to underestimation of the actual levels of viral suppression in this group. This underscores the need to address inequalities and population-specific issues in a differentiated manner to optimize the impact of the national HIV program (Fonseca et al., 2025; McAllister et al., 2017).
The positive results observed in the Trans population maintain their relevance, although in our study the sample was limited and not fully representative. The restricted availability of registries and underreporting in health information systems are limitations recognized in secondary analyses of national and international databases. The response to DTG in this key population group has a favorable safety profile despite consideration of possible interactions between DTG and hormonal therapy (Frola et al., 2023). Significant percentages of risky sexual practices, lower use of protection, discrimination, and limited access to HAIs have been described in this population that expose them to a higher risk of HIV transmission (Paz-Bailey et al., 2012).
Taken together, these findings reflect both progress made and persistent gaps in the implementation of DTG in comprehensive care services in Honduras. The marked variability between sites underscores the need to strengthen the standardization of registration systems such as monitoring HIV programs to ensure access to highly effective therapies.
The universal incorporation of DTG not only represents a strategy aligned with international recommendations but also constitutes a concrete opportunity to improve virological suppression rates, reduce HIV transmission and mitigate the risk of drug resistance in both general and vulnerable populations.
Conclusions and Recommendations
This study represents the first national analysis of dolutegravir use in Honduras and highlights the significant variability in SV outcomes among treatment centers. Centers that adopted dolutegravir (DTG), such as SAI HE and SAI Instituto Cardiopulmonar, showed significant improvement in viral suppression compared to those that did not fully implement it. This underscores the importance of adhering to current WHO guidelines on the use of dolutegravir in HIV treatment, given its positive impact on public health outcomes and its low rate of resistance. In addition, it is critical to continue to monitor treatment resistance at the national level and to optimize ART adherence strategies, especially in the most vulnerable populations.
It is crucial to target future efforts and seek collaborations with LGBTQIA+ organizations and community groups to gain trust and engage transgender women in HIV studies, as well as support SAIs to ensure that they are fully equipped to offer appropriate care, based on respect, education, and provide all necessary resources in HIV prevention and treatment, as well as adequate follow-up.
To maximize dolutegravir (DTG) effectiveness in resource-limited settings, broader access across all SAIs—especially in high-resistance areas—is essential. Expanding resistance testing and strengthening adherence support, including mHealth and community strategies, can further improve ART outcomes. Public health education for providers and patients remains key to promoting early diagnosis, adherence, and long-term treatment success (Kalungwe et al., 2022; WHO, 2021).
Limitations
This analysis used routine program data with heterogeneous SAI database structures and non-standard variable coding, which may affect comparability across sites. Missing fields, most notably the absence of sociodemographic data at SAI INCP and incomplete capture of sociodemographic characteristics or treatment outcomes, limit consistency, introduce selection/information bias, and constrain generalizability. In addition, the cluster sampling prioritizing regions with higher reported ART resistance may underrepresent lower prevalence areas and the broader Honduran population. Despite these constraints, the study provides program-relevant insights into dolutegravir use across key HIV care settings. Incomplete capture of sex/gender and key population fields precluded national disaggregation; this will be addressed in future analyses after data harmonization.
Footnotes
Acknowledgements
Expressions of gratitude are extended to the organizations and institutions that facilitated the realization of this study, namely the heads of the health regions of the departments of Atlantida, Yoro, Cortes, Francisco, Comayagua, and Choluteca. Special acknowledgment is reserved for Dr. Diana Varela (HE) and Dr. Wendy Moncada (INCP) for their indispensable contributions. In addition, sincere appreciation is directed toward the administrative personnel of the care centers, secretaries, laboratory staff, and microbiologists. To Global Communities, as the primary recipient of the HIV grant, and to all those who collaborated with administrative and logistical support. In addition, we acknowledge the leadership and dedication of the staff from the various departments of the Honduran Ministry of Health (SESAL), the directors of the SAI, the Bioethics Committee of the Faculty of Medical Sciences (CEIB), and all the NGOs that provide support to individuals with an HIV diagnosis at the national level, their leaders, all the field staff involved in the development of the study, and the representatives of external cooperation who participated in different stages of the research project. We would like to express our deepest gratitude to the entire LGBTQIA+ community, who generously gave their time and trust in the noble purpose of this study at every stage. We are also indebted to the staff at Global Communities, who provided invaluable support and guidance in the study’s development. Finally, we would like to acknowledge the crucial contributions of the administrative staff at FUNDAUNAH.
Authors’ Note
The study findings were disseminated through a poster presentation at the II ISPE LATAM Pharmacoepidemiology Congress, held in Brazil from October 10 to 12, 2025. Subsequently, the study was presented at the XXX Scientific Conference of the Faculty of Medical Sciences, National Autonomous University of Honduras, in June 2023.
ORCID iDs
Ethical Considerations
The study was carried out under the Helsinki Declaration. The preparation and execution of this research fully complied with the ethical principles of autonomy, justice, beneficence, and non-maleficence. The approval number (057-2021) was granted by the Biomedical Research Ethics Committee (CEIB) at the National Autonomous University of Honduras (UNAH) in December 2021.
Consent for Publication
He current article processing fees (publication fees) were funded by the National Autonomous University of Honduras (UNAH), Tegucigalpa, MDC, Honduras, Central America.
Author Contributions
Study design: ICF, IL, FML, MS, MC, ALS, LIZ, Data collection: ICF, IL, LIZ, Data analysis: ICF, EET, HNC, IL, MS, LIZ, ALS, LIZ, Writing: ICF, EET, HNC, IL, FML, MS, RLG, MC, ALS, JA, CRM, LIZ all authors have read and approved the published version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was fully funded by Global Communities as part of the grant “Moving HIV and Tuberculosis Responses Toward 90-90-90 and Sustainability with Focus on General and Vulnerable Populations,” a project funded by the Global Fund and administered by FUNDAUNAH.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data presented in this study are available upon request from the corresponding author.
