Abstract
Smoking negatively impacts lung function and cardiovascular capacity, making physical activity (PA) more challenging and contributing to mental health issues. This study investigates the role of PA in mitigating the links between smoking, sleep disorders, and depressive symptoms in adults in the United States. Data from the 2017 to 2020 National Health and Nutrition Examination Survey (NHANES) were examined. Participants were categorized by PA adherence (≥ 900 metabolic equivalent tasks [METs]/week or not) and smoking status based on serum cotinine levels (< 0.05 for non-smokers, 0.05–15 for secondhand smokers, and > 15 ng/mL for active smokers). Relationships between sleep disorders, depressive symptoms, and PA/smoking were evaluated using survey logistic regression, with non-smokers meeting PA guidelines as the reference group. Stratified analyses were conducted by age group (21 - 39, 40 - 59, 60+ years). Active smokers not meeting PA guidelines had a significantly higher likelihood of moderate to severe depressive symptoms (odds ratio [OR] 2.21, 95% confidence interval [CI] = [1.34, 3.65]). Young active smokers meeting PA guidelines had greater odds of sleep disorders (OR = 2.86, 95% CI = [1.38, 5.93]). Among middle-aged adults, those not meeting PA guidelines exhibited elevated odds of sleep disorders (OR = 2.18, 95% CI = [1.33, 3.59]) and severe depressive symptoms (OR = 3.01, 95% CI = [1.11, 8.18]). For older adults, increased odds of depressive symptoms were observed (OR = 3.37, 95% CI = [1.22, 9.29]). These findings underscore the potential of PA to mitigate mental health risks among individuals exposed to smoking. Longitudinal research is required to confirm these associations and inform targeted interventions.
Smoking remains a significant public health concern in the United States, contributing to various chronic diseases and premature mortality (Carter et al., 2015; OSG, 2010; Steinberg et al., 2010). In 2020, approximately 47.1 million (19.0%) of U.S. adults were current smokers, with prevalence highest among middle-aged adults (17.4%), followed by young adults (14.8%) and older adults (8.4%) (Cornelius, 2022). Smoking is the most common preventable cause of mortality globally and is linked to diseases such as lung cancer (85% of all cases), cardiovascular diseases, chronic obstructive pulmonary disease (COPD), and other respiratory illnesses (Lushniak et al., 2014; Siegel et al., 2022).
Sleep disorders, including insomnia and sleep apnea, impact overall health and affect about 30% of adults (Karna et al., 2020; Ohayon, 2011). Smoke exposure negatively influences sleep quality, causing issues like difficulty falling asleep and sleep dissatisfaction (Jaehne et al., 2012). In contrast, regular moderate-to-vigorous physical activity (MVPA) improves sleep quality and duration (Grandner et al., 2011), whereas sedentary behavior increases sleep disorder risks (Kredlow et al., 2015). Poor sleep is linked to adverse health outcomes, including cardiovascular diseases, obesity, diabetes, and mental health issues (Grandner, 2022), emphasizing the need for effective management.
Similarly, depressive symptoms, characterized by sadness, hopelessness, and reduced interest in activities, are significant public health concerns (Lam, 2018). These symptoms, ranging from mild to severe, affect daily functioning (Christensen et al., 2020; Kazama et al., 2011) and impact approximately 21.0 million (8.3%) of U.S. adults annually (NIMH, 2022). Effective management of depressive symptoms is vital, as it is associated with high mortality and morbidity, and successful treatment improves quality of life, reduces comorbidities, and lowers suicide risk (WHO, 2017). Specifically, research consistently demonstrates a significant association between tobacco smoking and depressive symptoms (Fan et al., 2022; Pasco et al., 2008). Nicotine dependence and associated biochemical changes can worsen mood and exacerbate depressive symptoms (Fluharty et al., 2016). However, regular PA is linked to reduced depression risk, as it promotes neurogenesis, lowers inflammation, and enhances mood-regulating neurotransmitters (Schuch et al., 2018). Thus, regular PA may influence preventing the onset of depressive symptoms.
PA guidelines provide evidence-based recommendations regarding the type and amount of PA necessary for maintaining health and preventing diseases (Pedersen & Saltin, 2015). Current research suggests that adults should perform muscle-strengthening exercises 2 or more days a week in addition to 300 min of moderate-intensity aerobic PA or 150 min of vigorous-intensity aerobic PA (Lee et al., 2022). Adherence to these guidelines is associated with multiple health benefits, including reduced risk of cardiovascular diseases (Ades et al., 2001; Wenger et al., 1995), Type 2 diabetes (Hamman et al., 2006), specific cancers (Li et al., 2016), and mental health conditions including depression and anxiety (Warburton & Bredin, 2017). However, exposure to smoke, particularly from tobacco, can impair lung function and cardiovascular capacity, potentially making PA more challenging and less enjoyable (Lushniak et al., 2014). Previous research has highlighted regular engagement in MVPA among smokers has been found to partially counteract some negative health effects associated with smoking, such as poor cardiovascular outcomes and decreased lung capacity (Kaczynski et al., 2008). Although the evidence still remains inconsistent and incomplete, regular PA could mitigate some of the adverse effects of smoking by enhancing cardiovascular health and lung function (Audrain-McGovern & Benowitz, 2011).
In addition, the intersection of PA, smoking, depressive symptoms, and sleep disorders represents a critical area of study due to their interconnected physiological and psychological pathways. Smoking exacerbates depressive symptoms through neurochemical changes and increased inflammation, which in turn negatively impact sleep quality (Colrain et al., 2004; Costa & Esteves, 2018). Conversely, PA mitigates depressive symptoms by enhancing mood-regulating neurotransmitter levels (Phillips & Fahimi, 2018) and improving overall mental well-being (Granero-Jiménez et al., 2022; Mahindru et al., 2023), which also positively influences sleep quality and duration (Peach et al., 2016; Vaingankar et al., 2020). Despite these established connections, there remains a gap in understanding the combined impact of PA and smoking status on depressive symptoms and sleep quality, particularly across different age groups and exposure levels, necessitating further comprehensive research.
This study aims to comprehensively investigate the protective role of PA against sleep disorders and depressive symptoms among U.S. adults, focusing on variations across different smoking statuses (i.e., active smokers, secondhand smokers, and non-smokers). By conducting subgroup analyses based on both PA and smoking status across different age groups (i.e., young, middle-aged, and older adults), the study seeks to contribute to the broader understanding of how lifestyle intervention, particularly PA, can be employed to reduce the adverse health outcomes associated with smoking.
Method
Study Design and Population
The National Health and Nutrition Examination Survey (NHANES), which was carried out between 2017 and 2020, provided the data for this investigation. To evaluate the health and nutritional status of the American people, the CDC administers the NHANES, a nationally representative survey, using a sophisticated, multistage, random sample approach (Stierman et al., 2021). Laboratory testing, physical examinations, and demographic and behavioral questionnaires were all used in the data collection process. NHANES 2017—March 2020 pre-pandemic data was produced by merging data from the 2019–March 2020 NHANES cycle with data from the 2017 to 2018 NHANES cycle due to the disruption of data collection during the COVID-19 pandemic (Stierman et al., 2021). All participants gave written informed consent, and the National Center for Health Statistics (NCHS) Ethics Review Board examined and approved the study procedures. This study was classified as exempt, as the data utilized were de-identified and publicly available, which did not require further institutional review board (IRB) approval at our institution.
Measures
Weekly PA levels were gathered using the Global Physical Activity Questionnaire (GPAQ) (Keating et al., 2019). Participants reported their daily PA through questions such as, “How many days per week do you engage in moderate-to-vigorous intensity PA?” and “How many hours per day?” (Keating et al., 2019). PA data were transformed into weekly metabolic equivalent tasks (METs) in accordance with WHO (2020) Analytical Guidelines. This study quantified PA using the formula “METs level × minutes × number of activities per week” for each intensity level, with 6.0 METs assigned to vigorous PA and 3.0 METs to moderate PA (Ainsworth et al., 2000). Adults are advised to perform 300 min of moderate or 150 min of vigorous PA per week (900 METs), including a combination of both (Lee et al., 2022). Therefore, this study used a threshold of 900 METs per week to distinguish between participants who adhered to PA guidelines and those who did not.
Serum cotinine level is widely recognized as a reliable indicator of tobacco smoke exposure due to its extended half-life (15 - 20 hrs) (Benowitz, 1996; Kunno et al., 2024). This study used isotope dilution high-performance liquid chromatography/atmospheric pressure chemical ionization tandem mass spectrometry to evaluate the levels of serum cotinine (NHANES, 2024). Recent nicotine exposure was defined as cotinine levels above 0.015 ng/mL, the limit of detection (LOD) divided by the square root of two was used for levels below the LOD. (NHANES, 2024). Smoke exposure was categorized as non-smokers (< 0.05 ng/mL), secondhand smokers (0.05 - 15.0 ng/mL), and active smokers (> 15.0 ng/mL) (Kim, 2019). Utilizing serum cotinine levels offers an objective approach to assessing tobacco smoke exposure, reducing the likelihood of misclassification that can occur with self-reported smoking data (Wells et al., 1998).
The adherence to PA guidelines was classified into two groups (meet PA guidelines and did not meet guidelines) based on calculated METs, and smoke exposure was categorized into three groups (non-smokers, secondhand smokers, and active smokers) based on measured serum cotinine levels. These classifications were then combined to create six distinct groups: (1) active smokers who did not meet PA guidelines, (2) secondhand smokers who did not meet PA guidelines, (3) non-smokers who did not meet PA guidelines, (4) active smokers who met PA guidelines, (5) secondhand smokers who met PA guidelines, and (6) non-smokers who met PA guidelines (reference group).
Sleep disorders were assessed using responses to the NHANES (2020) questionnaire item, “Have you ever told a doctor or other health professional that you have trouble sleeping?” with response options of “Yes,” “No,” “Refused,” or “Don’t know.” Participants who selected “Refused” or “Don’t know” were coded as missing data and subsequently excluded from the analysis.
The Patient Health Questionnaire-9 (PHQ-9), a nine-item screening instrument based on the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition criteria, was used to measure depressive symptoms (Kroenke et al., 2001). The PHQ-9 has demonstrated strong psychometric qualities when used to screen for depressive symptoms in the American population (Beard et al., 2016; Kroenke et al., 2010). Each question had four possible responses based on experiences over the past 2 weeks: 0 for “not at all,” 1 for “several days,” 2 for “more than half the days,” and 3 for “nearly every day.” Participants answered each question based on their experiences during the previous 2 weeks. The overall PHQ-9 score, which is determined by adding the answers to all items, runs from 0 to 27. Higher scores indicate more severe depressive symptoms. Scores are classified as follows: 0 – 4 denotes little depressive symptoms, 5 – 9 mild depressive symptoms, and 10 or more denotes moderate to severe (clinically significant) depressive symptoms (Kroenke et al., 2001). To include individuals with mild symptoms and capture a broader spectrum of depressive symptomatology, in this study, depressive symptoms were defined as a PHQ-9 score of 5 or higher (Guo et al., 2024). This cutoff was chosen to include mild depressive symptoms and include a broader spectrum of participants who might otherwise be overlooked. Participants were therefore categorized into two groups: those who had no depressive symptoms (0–4) and those who had depressive symptoms (5 or higher).
Covariates
Gender (male, and female), age (21 - 39, 40 - 59, 60+ years old), race/ethnicity (non-Hispanic White, non-Hispanic Black, Hispanic/Latino, and other), poverty-to-income ratio (PIR, < 1.3, 1.3 – 3.5, and > 3.5), employment (employed, disabled, retired, and not employed), education level (high school or less, some high school/2-year degree/no degree, and 4-year degree or more), marital status (married/partnered, never married, and divorced/separated/widowed), body mass index (BMI, underweight/normal, overweight, and obese), and medical condition (stroke, heart attack, angina/angina pectoris, congestive heart failure, coronary heart disease, arthritis, thyroid problem, and any liver condition) were included as covariates. Alcohol consumption was included in covariates in this study because alcohol intake has been shown to have a negative impact on sleep quality and depressive symptoms (Boden & Fergusson, 2011; Jaehne et al., 2012).
Statistical Analysis
For MVPA and serum cotinine levels, descriptive statistics were calculated at all levels, including frequencies, percentages, means, and standard deviations. One-way analysis of variance (ANOVA) was used to examine mean differences for continuous variables, and chi-square tests were employed to investigate proportionate differences in sleep disorders and depressive symptoms across categorical categories. The adjusted odds ratios (OR) and 95% confidence intervals (CI) for sleep disorders and depressive symptoms were then estimated for each exposure group using survey logistic regression, specifically focusing on adherence to PA guidelines and serum cotinine levels. The referent group in this study consisted of non-smokers who met PA guidelines, as this group represents the lowest-risk behavioral profile in the population and provides a meaningful, consistent benchmark for understanding how different combinations of smoking exposure and PA adherence contribute to health risks. The initial model evaluated these associations for the entire adult sample, followed by age-based stratified analyses for young adults (ages 21 – 39), middle-aged adults (ages 40 – 59), and older adults (ages 60+). To properly account for the intricate sampling design and sampling weights used in NHANES, variables for cluster, stratum, and weights were incorporated into the model. SAS Version 9.4 (SAS Institute, Inc., Cary, North Carolina) was used for statistical analyses. A p-value of less than 0.05 was considered statistically significant for all analyses.
Results
The 2017 – 2020 cycle included 8,965 participants, of whom 3,718 were eligible adults. After removing 202 participants with missing data on sleep disorders or depressive symptoms as determined by the PHQ-9 and 508 participants with insufficient data on covariates like socioeconomic status, alcohol consumption, BMI, and chronic conditions, 3,008 participants remained in the final analysis. A comparison of participants included in the final sample and those excluded due to missing data on key demographic characteristics revealed no statistically significant differences. Therefore, listwise deletion was employed to retain a complete case analysis approach. Figure 1 shows the flowchart for the selection of participants included in the final study sample.
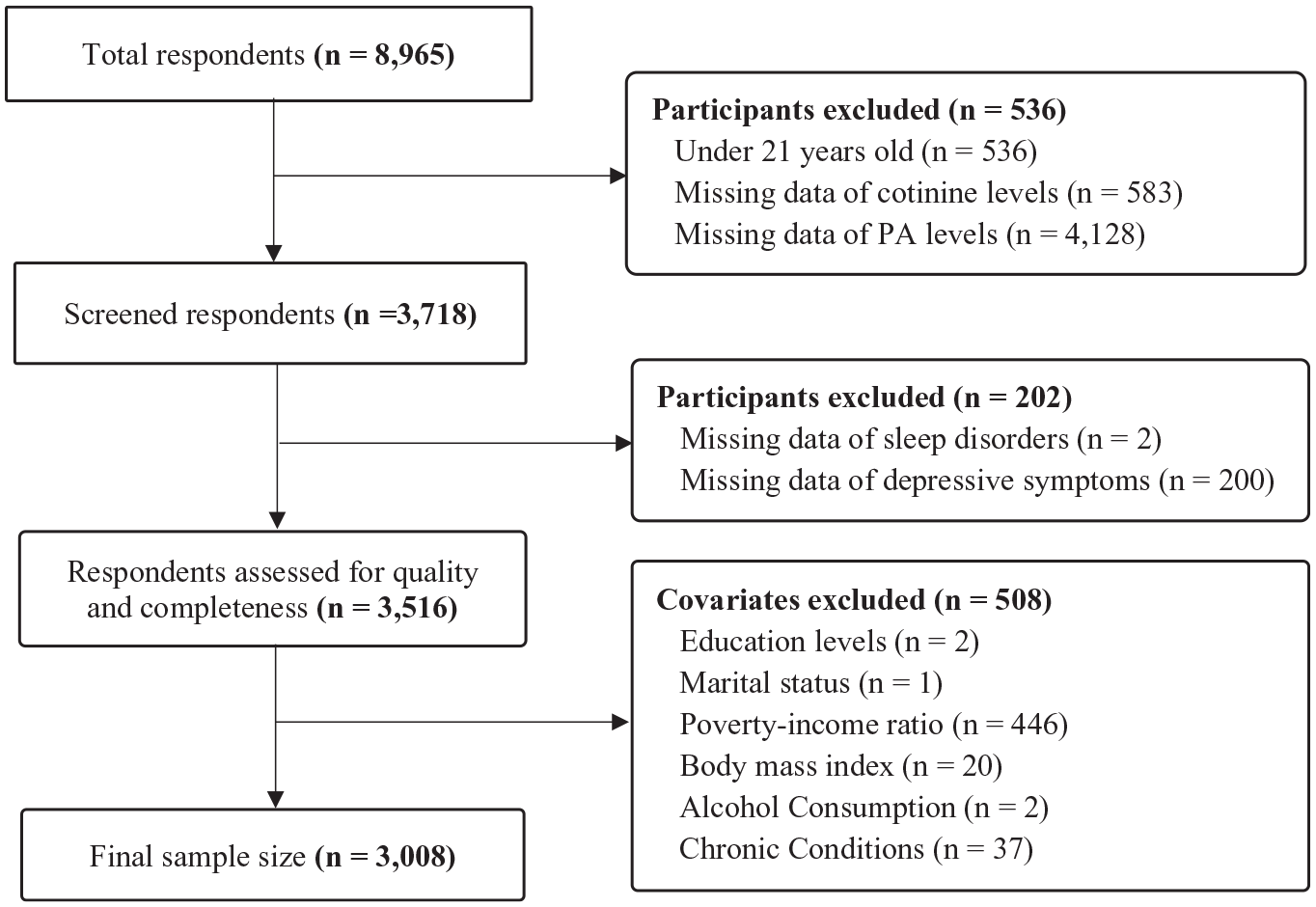
Flowchart of Participant Selection for the Final Study Sample.
The descriptive analysis reveals significant associations between sleep disorders and several demographic variables (Table 1), including age (p < .01), race/ethnicity (p < .01), employment status (p < .01), BMI (p < .01), marital status (p = .02), serum cotinine levels (p = .034), and MVPA (p < .01). The prevalence of sleep disorders was highest among middle-aged adults (40 - 59 years, 30.2%), followed by older adults (≥ 60 years, 32.0%) and younger adults (21 - 39 years, 21.7%). Among participants with sleep disorders, the largest proportion were non-Hispanic Whites (33.7%). Marital status also showed a significant relationship, with divorced/separated/widowed adults (36.5%) having a higher prevalence of sleep disorders compared with other groups, particularly married adults (26.3%) and those who were never married (23.4%). Employment status revealed that disabled individuals had the highest prevalence of sleep disorders (59.0%), followed by retirees (34.6%), the unemployed (26.7%), and those who were employed (23.8%). Obesity was positively associated with sleep disorders, with obese individuals having the highest prevalence (34.3, followed by overweight (25.6%) and normal-weight individuals (21.2%). Regarding serum cotinine levels, sleep problems were more common in active smokers (32.4%) compared with non-smokers (27.5%) and secondhand smokers (23.3%). The prevalence of sleep disorders was higher among participants who did not meet PA guidelines (31.2%) than among those who did (23.8%). Table 1 displays the characteristics of the subjects categorized based on whether they had sleep disorders or depressive symptoms.
Characteristics of Study Participants Stratified by Category of Sleep Disorders (n = 3,008).
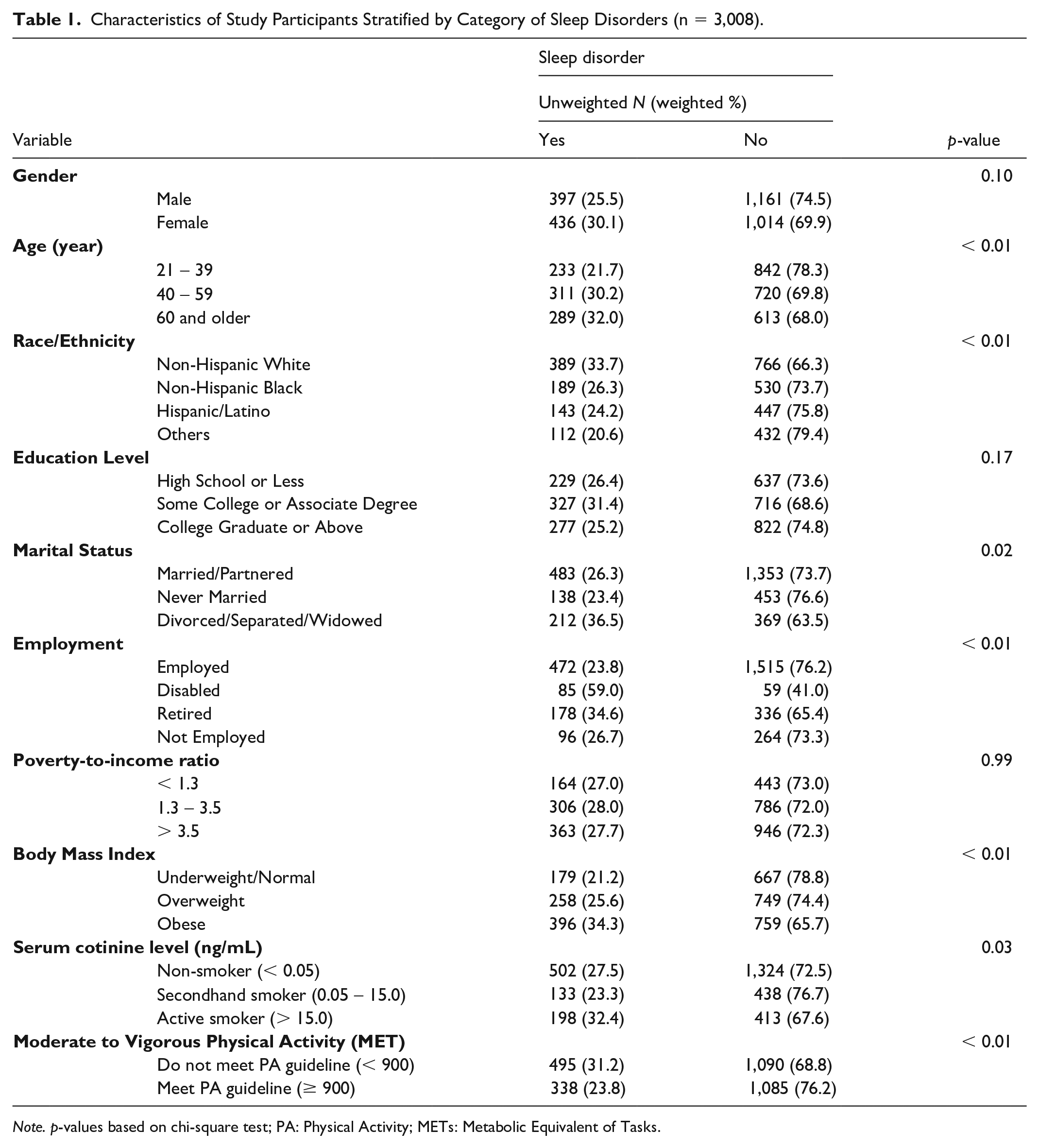
Note. p-values based on chi-square test; PA: Physical Activity; METs: Metabolic Equivalent of Tasks.
The analysis presented in Table 2 also revealed significant associations between depressive symptoms and several covariates, including age (p = .04), education level (p <.01), employment status (p < .01), marital status (p < .01), PIR (p < .01), BMI (p = .02), and serum cotinine levels (p < .01). Participants with a college degree or above exhibited a higher proportion of individuals without depressive symptoms (86.7%) compared with those with lower educational attainment. Divorced/separated/widowed adults had higher rates of depressive symptoms (26.9%) compared with never married (25.4%) and married/partnered individuals (16.9%). Employed adults had the lowest prevalence rates of depressive symptoms (16.8%) compared with those who were retired (17.3%), unemployed (33.1%), and disabled (50.7%). The lowest PIR was linked to higher rates of depressive symptoms (31.6%), with the lowest rates observed in those with the highest PIR (13.8%). Consistent with the findings on sleep disorders, obese participants had the highest prevalence of depressive symptoms (24.3%).
Characteristics of Study Participants Stratified by Category of Depressive Symptoms (n = 3,008).
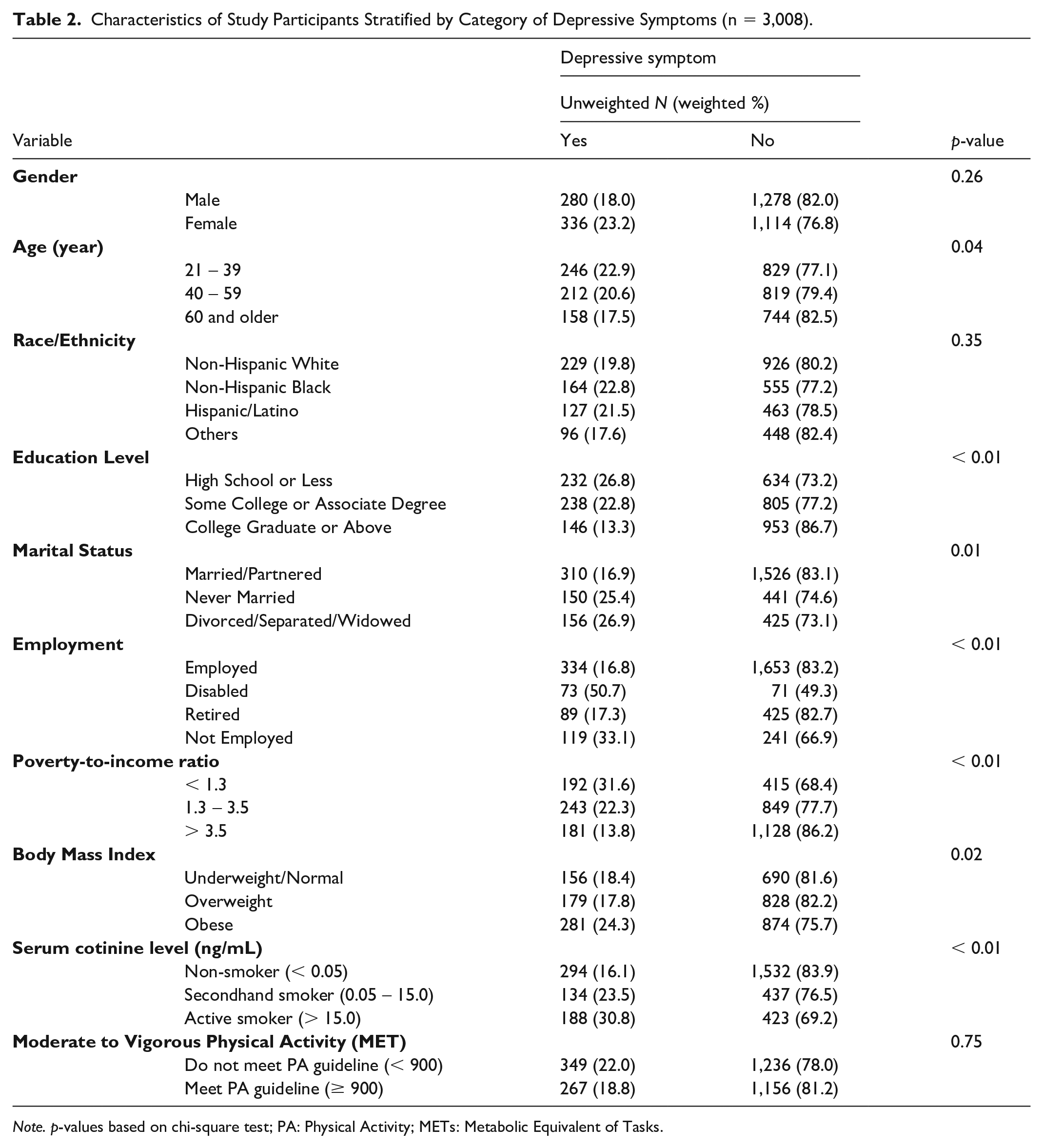
Note. p-values based on chi-square test; PA: Physical Activity; METs: Metabolic Equivalent of Tasks.
The descriptive analysis found that males, regardless of smoking status, were substantially more likely to adhere to PA recommendations compared with females (p < .01). Younger participants consistently exhibited higher adherence to PA guidelines across all smoking statuses. Among active smokers, the likelihood of not meeting PA criteria was significantly higher with increasing age and among non-Hispanic Whites (p < .01). Interestingly, non-Hispanic Whites who adhered to PA guidelines were more frequently exposed to secondhand smoke compared with those who did not (p < .01). Those with lower educational attainment had a higher likelihood of exposure to smoking environments, regardless of PA guideline adherence (p < .01). Among married individuals, active smokers who adhered to PA guidelines were more prevalent than those who did not (p < .01). Employment status revealed that, although employed adults had greater exposure to smoking environments, those who were exposed were less likely to adhere to PA guidelines (p < .01). Higher-income levels were associated with greater adherence to PA guidelines, even among those exposed to smoking environments, including both active and secondhand smokers (p < .01). Adults with a normal BMI were more likely to adhere to PA guidelines despite exposure to smoking environments, whereas overweight or obese individuals were less likely to meet PA guidelines under similar conditions (p < .01). Participants who did not meet PA guidelines, regardless of smoking status, had a greater prevalence of sleep disorders than those who met the guidelines. Moreover, adherence to PA guidelines was associated with fewer depressive symptoms, even among those exposed to smoking environments. Serum cotinine levels were markedly higher among active smokers who did not meet PA guidelines (227.51 ng/mL ± 154.50) compared with those who met the guidelines (221.67 ng/mL ± 163.67). In addition, active smokers who met PA guidelines had significantly higher MET values (3956.84 ± 1802.07) compared with those who did not meet the guidelines (419.42 ± 219.68). See Table 3 for the characteristics of study participants based on their adherence to PA guidelines and serum cotinine levels, with a focus on active smokers, secondhand smokers, and non-smokers.
Characteristics of Study Participants Stratified by Category of Physical Activity and Serum Cotinine Levels (n = 3,008).
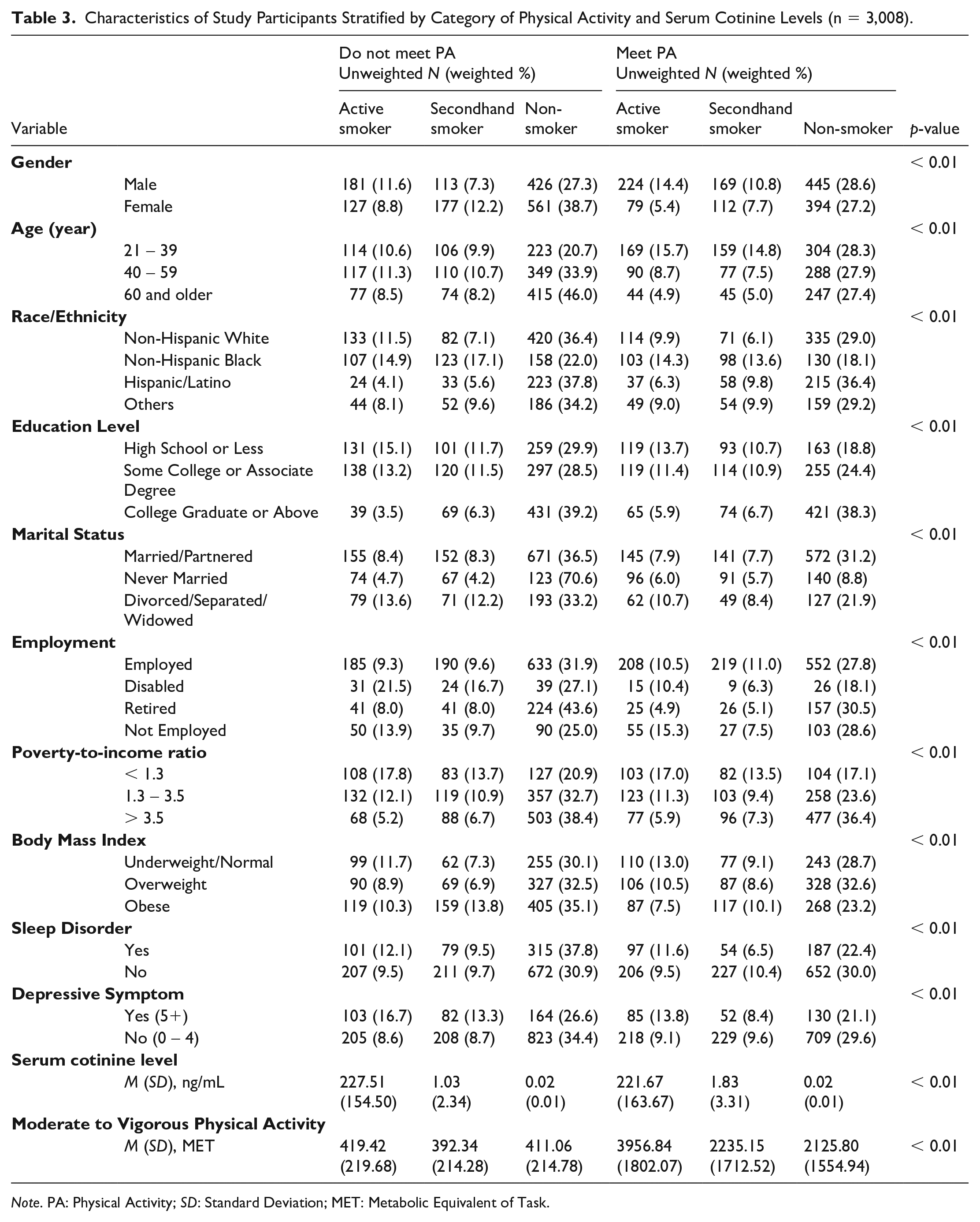
Note. PA: Physical Activity; SD: Standard Deviation; MET: Metabolic Equivalent of Task.
The survey logistic regression includes the entire sample and is further stratified by age groups: young adults (21 - 39 years), middle-aged adults (40 - 59 years), and older adults (60+ years). Across the entire sample, active smokers who did not meet PA guidelines had significantly higher odds of depressive symptoms (OR = 2.21, 95% CI = [1.34, 3.65], p < .01), however, the odds ratio was lower among active smokers who adhered to PA guidelines (OR = 1.87, 95% CI = [1.15–3.05], p < .05) than active smokers who did not meet PA guidelines. In addition, non-smokers who did not meet PA guidelines (OR = 1.39, 95% CI = [1.01, 1.91], p < .05) and active smokers who met PA guidelines (OR = 1.89, 95% CI = [1.17, 3.04], p < .01) were more likely to report sleep disorders. Among young adults, active smokers who met PA guidelines had significantly higher odds of sleep disorders (OR = 2.86, 95% CI = [1.38, 5.93], p < .01). In middle-aged adults, active smokers who did not meet PA guidelines had significantly higher odds of both sleep disorders (OR = 2.18, 95% CI = [1.33, 3.59], p < .01) and depressive symptoms (OR = 3.01, 95% CI = [1.11, 8.18], p < .05). For older adults, active smokers who did not meet PA guidelines had higher odds of depressive symptoms (OR = 3.37, 95% CI = [1.22, 9.29], p < .05). See Table 4 for the results of survey logistic regression analyses examining the relationships between sleep disorders, depressive symptoms, smoking status (active smokers, secondhand smokers, non-smokers), and adherence to PA guidelines.
Adjusted Odds Ratios (ORs) for Sleep Disorders and Depressive Symptoms by Exposure Smoke Status and Adherence to PA Guidelines.
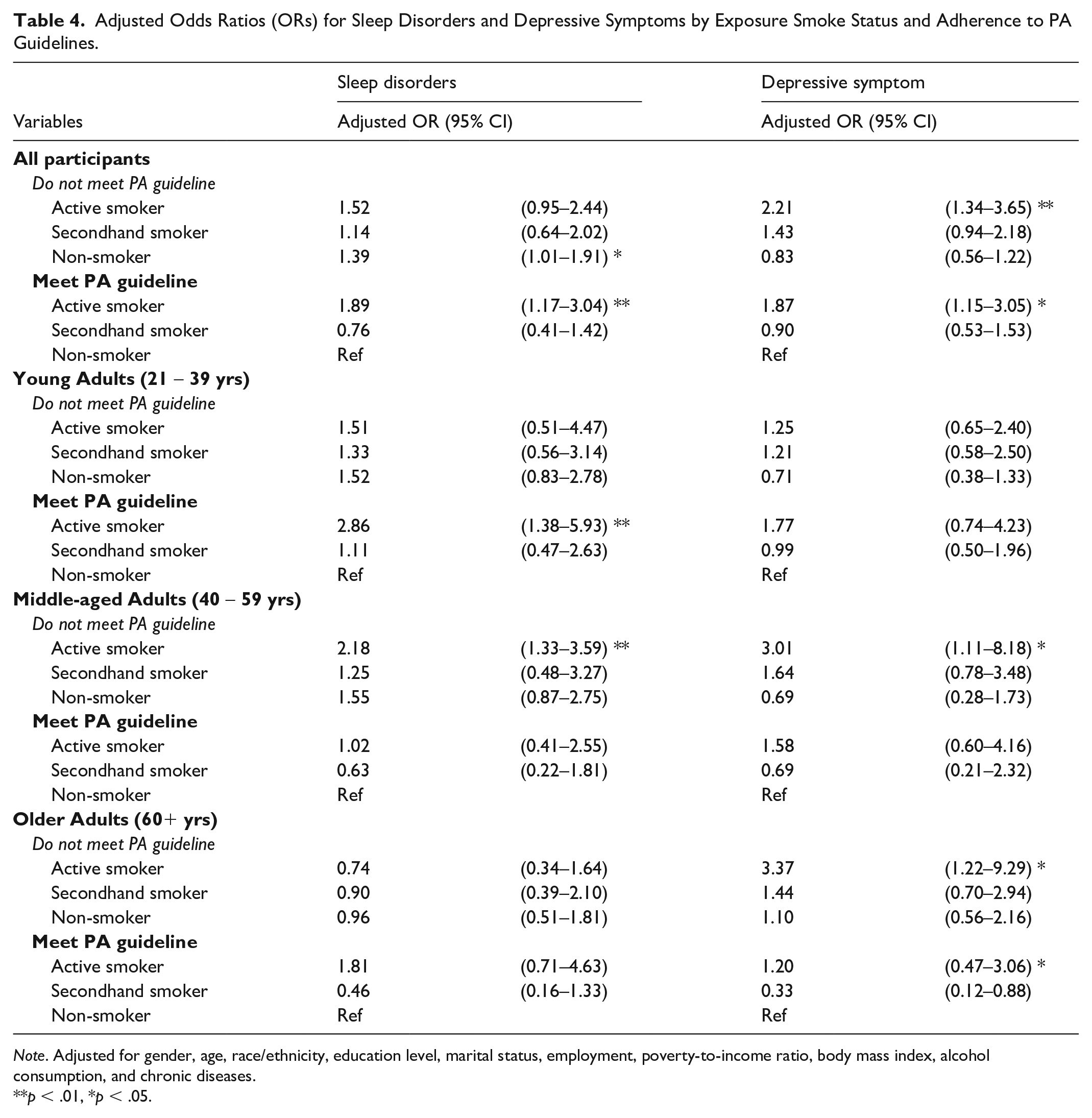
Note. Adjusted for gender, age, race/ethnicity, education level, marital status, employment, poverty-to-income ratio, body mass index, alcohol consumption, and chronic diseases.
p < .01, *p < .05.
Discussion
In this study, the combined effects of smoking status and PA on the prevalence of sleep disorders and depressive symptoms among U.S. adults were examined. The findings demonstrated that adherence to PA guidelines was linked to a lower prevalence of both sleep disorders and depressive symptoms, regardless of smoking exposure levels (Table 3). Participants who met the recommended levels of MVPA experienced fewer sleep disturbances and depressive symptoms compared with those who did not. However, these benefits were moderated by smoking status; active smokers reported higher odds of these conditions despite meeting PA guidelines (Table 4). Age also influenced outcomes, as different age groups demonstrated varied responses to the PA-smoking interaction concerning mental health and sleep quality.
This study found that PA was protective against depressive symptoms across all smoking statuses. Regular PA is well-established for enhancing mental well-being by promoting neurotransmitters such as serotonin, dopamine, and endorphins, crucial for mood regulation (Di Liegro et al., 2019; Marques et al., 2021; Schuch et al., 2018). The neurochemical impact of PA, particularly in releasing these neurotransmitters, has been linked to reduced sadness, better emotional stability, and lower depressive symptoms. Nevertheless, the study suggests that PA’s protective effect on depressive symptoms was weaker in active smokers. Nicotine, the addictive substance in cigarettes, disrupts mood regulation through its impact on the brain’s reward pathways, leading to dependency and increased mood disturbances (Benowitz, 2010; Laviolette, 2021). Smoking has been shown to alter serotonin and dopamine functions, both key to mood stability, potentially counteracting PA’s positive effects on mental health. These findings align with prior research showing that smoking can worsen depressive symptoms and interfere with neurochemical processes critical to mood stabilization (Hahad et al., 2021; Nunes et al., 2013). Thus, while PA may be associated with lower levels of depressive symptoms, active smokers, especially those who do not meet PA guidelines, remain vulnerable due to the complex neurobiological interplay between nicotine addiction and mood regulation. In addition, our results showed that lower educational attainment and income levels were associated with higher rates of both smoking and physical inactivity, consistent with previous studies demonstrating that socioeconomic disadvantage is a key driver of unhealthy behaviors (Foster et al., 2023; Pampel et al., 2010). Individuals with lower socioeconomic status may face more barriers to engaging in regular PA, such as limited access to safe recreational spaces or health resources and are also more likely to be exposed to smoking environments (Hiscock et al., 2012). Furthermore, mental health conditions are known to cluster with both low PA and tobacco use, possibly due to shared social and psychological determinants, including chronic stress and reduced social support (Harwood et al., 2007; Schuch et al., 2018). Therefore, interventions targeting PA and smoking cessation should be considered to address these underlying socioeconomic and psychosocial factors.
Significant associations were observed between PA and sleep disorders, with the descriptive analysis indicating that, within each smoking status group, the prevalence of sleep disorders was higher among those who did not meet PA guidelines compared with those who did. Previous studies suggest that smoking affects sleep quality, likely through nicotine’s stimulating effects on the central nervous system (Grigoriou et al., 2024; Jaehne et al., 2009). Nicotine can shorten the duration of sleep, delay the onset of sleep, and alter sleep architecture, disrupting normal sleep patterns (Branstetter et al., 2016). These physiological impacts may be more pronounced in smokers with low PA, as regular PA is known to improve sleep quality by promoting circadian rhythm regulation and reducing sleep latency (Purani et al., 2019). However, even among active smokers adhering to PA guidelines, the odds of disrupted sleep remained elevated, indicating that the adverse effects of nicotine on sleep may persist despite regular PA. Although PA benefits sleep by promoting deeper and more restful sleep, nicotine’s stimulating properties may override these effects, leading to continued issues with sleep initiation, continuity, and sleep stages. This indicates that while PA can improve sleep quality, its effectiveness is reduced in regular smokers, particularly those with high nicotine dependence. Smokers may require additional interventions, such as smoking cessation, to fully restore healthy sleep patterns.
An important and unanticipated finding in this study is that among all participants and particularly young adults, smokers who met PA guidelines exhibited higher odds of sleep disorders compared with smokers who did not meet PA guidelines. This contrasts with the descriptive findings shown in Table 3, where the prevalence of sleep disorders was generally greater among those who did not meet PA guidelines regardless of smoking status. Several factors may explain this discrepancy between unadjusted and adjusted analyses. First, it is important to recognize that the adjusted models account for a range of covariates, including demographic, socioeconomic, and health-related factors, that may confound the relationship between PA, smoking, and sleep disorders. Smokers who meet PA guidelines may represent a distinct subgroup characterized by heavier nicotine dependence or more intense smoking behaviors, which may not be fully captured by cotinine levels alone. For instance, individuals who are physically active despite heavy smoking might use exercise as a compensatory behavior to mitigate smoking-related health risks but still suffer from the neurophysiological and respiratory consequences of tobacco use that disrupt sleep (Kaczynski et al., 2008; Purani et al., 2019). This is consistent with prior research indicating that while PA improves sleep quality, the stimulating effects of nicotine and smoking-induced oxidative stress can independently impair sleep architecture (Jaehne et al., 2012; Lavie, 2015). Second, the nature and timing of PA might also play a role. Although adherence to PA guidelines was defined based on weekly MET minutes, the timing (e.g., evening exercise) and type of PA were not assessed and could influence sleep outcomes differently. Some evidence suggests that vigorous PA close to bedtime can transiently increase arousal, potentially worsening sleep latency in certain individuals (Kredlow et al., 2015). Smokers may be more susceptible to these effects due to their altered autonomic regulation. Finally, active smokers adhering to PA guidelines might experience higher stress or engage in riskier health behaviors (e.g., irregular sleep schedules, caffeine or alcohol use) that counteract the benefits of PA on sleep (Costa & Esteves, 2018; Schuch et al., 2018). Furthermore, confounding by unmeasured variables such as mental health conditions or the severity of sleep apnea may also contribute. Therefore, the adjusted models reveal nuanced associations that may not be apparent in unadjusted descriptive analyses. These results can call for caution in interpreting PA as a uniformly protective factor against sleep disorders among smokers and emphasize the importance of considering multiple interacting behavioral and physiological factors.
Among young adults (21 - 39 years), exposure status was most strongly associated with sleep disorders, whereas among older adults (60 years or older), the associations were more pronounced for depressive symptoms. In these groups, the prevalence of sleep problems was higher among active smokers who followed PA guidelines than among non-smokers, although PA provided some protection against depressive symptoms. This finding suggests that while PA’s mental health benefits, particularly in reducing depressive symptoms through improved brain-derived neurotrophic factor levels and enhanced emotional resilience (Phillips, 2017), are substantial, its effect on mitigating smoking’s impact on sleep is limited. Smoking may interfere with sleep via oxidative stress and inflammation, which can damage neural pathways responsible for sleep regulation (Cohrs et al., 2014; Lavie, 2015; Van Eeden & Sin, 2013) and reduce respiratory function, contributing to issues like sleep apnea (Crinion & McNicholas, 2014). Persistent sleep disturbances among young adults, despite regular PA, highlight the challenge of counteracting nicotine’s effects solely through lifestyle modifications. Thus, although PA benefits mental health across age groups, nicotine exposure from smoking presents significant barriers to achieving optimal sleep health.
For middle-aged adults (40 - 59 years), the findings were particularly notable, as this group is at increased risk for the cumulative effects of smoking and physical inactivity, often resulting in chronic health conditions. The risks of moderate-to-severe depressive symptoms and sleep disturbances were considerably greater for active smokers in this age group who did not adhere to PA guidelines. The study also revealed that middle-aged secondhand smokers who did not meet PA guidelines had a heightened experience of depressive symptoms, underscoring the mental health risks associated with indirect smoke exposure. Secondhand smoke contains many of the same toxic chemicals as direct smoking, triggering inflammation and disrupting mood-regulating neurochemical pathways (Flor et al., 2024; Flouris et al., 2010; IOM, 2010). In middle-aged adults, these effects may be exacerbated by physical inactivity, increasing the likelihood of depressive symptoms. This age group often faces added stressors, such as job and caregiving demands (Aldwin & Levenson, 2001; Infurna et al., 2020), which may heighten the mental health effects of secondhand smoke. The increased depressive symptoms in this subgroup point to a need for public health initiatives that target not only smoking cessation but also raise awareness about the risk of secondhand smoke.
One of the study’s strengths is a nationally representative sample from NHANES, which allows generalization to the broader U.S. adult population. In addition, the use of serum cotinine levels to objectively measure smoking exposure minimizes the risk of misclassification compared with self-reported measures. However, this study has several limitations. Our final analytic sample, due to study-specific inclusion and exclusion criteria, may differ from the original NHANES sample, and this may limit the generalizability of our findings to some extent. In particular, the exclusion of participants with missing PA data may introduce selection bias if those individuals differ systematically from those included in the analysis, which could affect the generalizability of our results. While serum cotinine is a validated and objective biomarker of recent nicotine exposure, it cannot distinguish between combustible tobacco use, nicotine replacement therapy, or exposure to nicotine from vaping products; therefore, some degree of misclassification may exist for participants using alternative nicotine delivery systems. Causal inferences are not possible due to the cross-sectional design, as it is unclear whether PA reduces depressive symptoms and sleep disorders or whether individuals with fewer mental health issues are more likely to engage in PA. The reliance on self-reported data on PA, sleep disorders, and depressive symptoms also introduces potential biases, and the study did not account for smoking intensity or duration, which may affect sleep and mental health outcomes. Future research should employ longitudinal designs to clarify causal links between PA, smoking, and mental health issues and to examine the effects of different PA types and intensities.
Conclusion
This study highlights the important association between smoking and PA status in connection to sleep disorders and depressive symptoms in U.S. adults. Although smoking was associated with a higher prevalence of depressive symptoms and sleep disorders, our results indicate that its harmful effects are attenuated among those who meet PA guidelines, particularly with respect to sleep quality. However, these findings suggest that while PA may help mitigate the adverse effects of nicotine exposure on mental health, smokers still experience worse outcomes compared with non-smokers. The increased risk of depressive symptoms among middle-aged secondhand smokers, particularly those who are physically inactive, emphasizes the need to raise awareness of the mental health risks associated with indirect smoke exposure. Public health strategies should not only promote PA but also work on reducing both direct and secondhand smoke exposure for improved overall health outcomes. Future research should investigate tailored interventions that incorporates optimal PA intensity and smoking cessation to address these concerns effectively.
Footnotes
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
