Abstract
We present the case of a 73-year-old female with chemotherapy-induced cardiomyopathy, severe mitral regurgitation status post bioprosthetic mitral valve (MV) replacement one year prior, and atrial fibrillation, admitted for evaluation for left ventricular assist device (LVAD) implantation. While her preoperative transthoracic echocardiogram (TTE) did not reveal a discrete mass, it showed a mean mitral gradient of 7 mmHg; intraoperative transesophageal echocardiography (TEE) identified a 2 cm × 1.2 cm thrombus on the bioprosthetic MV. Given these findings, the surgical team decided to replace the mitral valve concurrently with LVAD implantation. Although this approach is not routine, the goal was to improve her quality of life and reduce postoperative complications. Post-procedure TEE showed an improved MV mean gradient of 3 mmHg with no evidence of paravalvular leak. The patient was subsequently discharged home in stable condition without any thromboembolic events. This case highlights the challenges of managing complex valvular pathology in patients undergoing LVAD placement.
Keywords
Introduction
HeartMate 3 left ventricular assist devices (LVADs) have become more common as either a bridge to transplantation or destination therapy for patients with severe heart failure. 1 Heart failure often coexists with valvular pathology. At this time, replacement of a pre-existing functioning mitral bioprosthetic valve at the time of mechanical support device is not recommended as routine practice. 2 We present a patient with extensive mitral valve (MV) thrombosis identified on intraoperative transesophageal echocardiogram (TEE) who underwent concurrent MV replacement with her LVAD implantation. This report discusses the diagnostic challenges and surgical planning involved in managing patients undergoing LVAD implantation with concurrent complex valvular pathology.
Materials and Methods
As the case report is devoid of patient identifiable information, it is exempt from IRB review requirements in accordance with University of Texas Southwestern policy. Written informed consent was obtained from the patient for publication of this case report and accompanying images.
Case Presentation
A 73-year-old female with a history of chemotherapy-induced cardiomyopathy was transferred from an outside hospital for consideration for HeartMate 3 LVAD implantation. Her medical history included severe mitral regurgitation status post bioprosthetic MV placement one year prior, atrial fibrillation on amiodarone, breast cancer status post lumpectomy with chemotherapy and radiation therapy, and Graves’ disease status post radioactive iodine. The patient was compliant with her home anticoagulation regimen of apixaban 5 mg twice a day. While admitted to the hospital, her apixaban was held for 10 days pending workup for advanced therapies. In the interim, she developed a myocardial infarction with non-obstructive coronary arteries secondary to a small 6 mm left ventricular (LV) apical thrombus. She was re-started on a heparin drip with a targeted therapeutic range of anti-factor Xa level of 0.5–0.7 IU/mL. After discussion with the patient and Cardiothoracic Surgery, she was posted for HeartMate 3 LVAD implantation. Preoperative transthoracic echocardiogram (TTE) showed an ejection fraction (EF) less than 20% and a well-seated MV with a mean gradient of 7 mmHg. The TTE did not identify a discrete echogenic mass. The absence of visualization may be attributable to technical limitation, including a restricted acoustic window from previous chest surgery or artifact generated by the bioprosthetic mitral valve (Figure 1). Parasternal long axis view on preoperative TTE did not demonstrate a visible echogenic mass near the mitral valve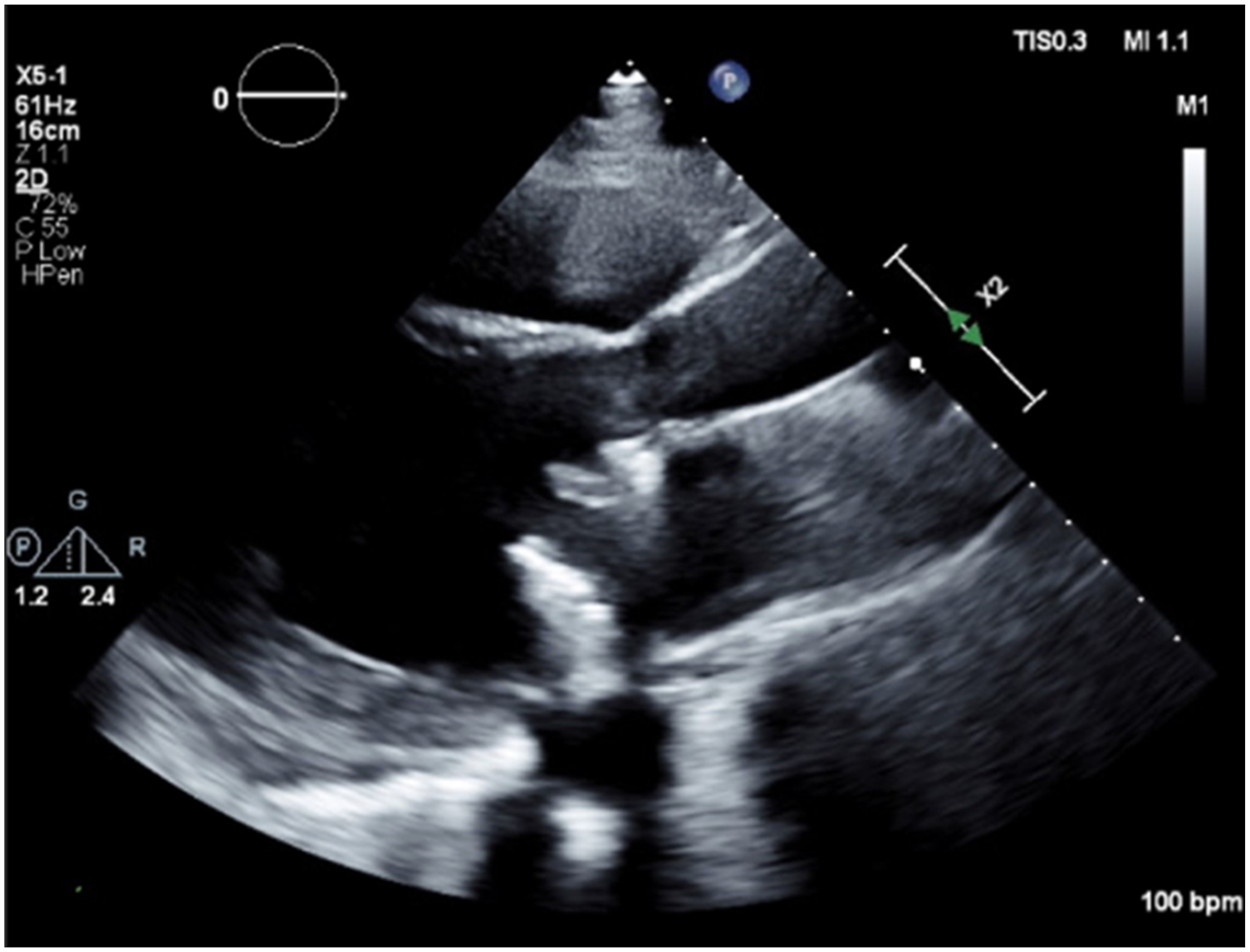
Intraoperative TEE demonstrated a 2 cm × 1.2 cm echogenic mass on the posterior leaflet and annular ring of the bioprosthetic MV with a mean gradient of 9 mmHg across the mitral valve (Figure 2). After discussion with the surgical team and family, the decision was made to replace the MV concurrently with LVAD implantation. Intraoperative TEE in mid-esophageal 4-chamber view demonstrating a 2 cm × 1.2 cm echogenic mass on the posterior leaflet of the bioprosthetic mitral valve as well as a mean pressure gradient of 9 mmHg across the mitral valve by continuous-wave Doppler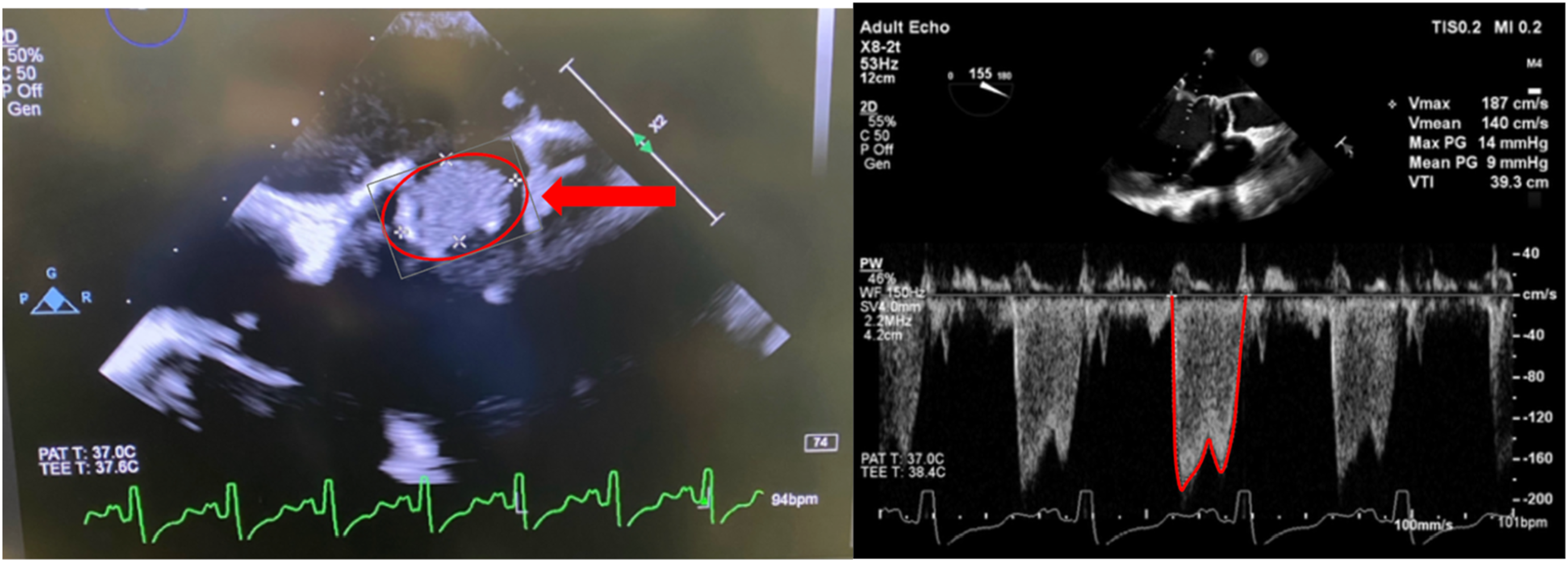
Upon surgical exposure, the thrombus was visualized along the prior MV suture line and leaflets (Figure 3). Surgical view of the prior bioprosthetic mitral valve showing some of the thrombus attached to the prior MV suture line and valve leaflets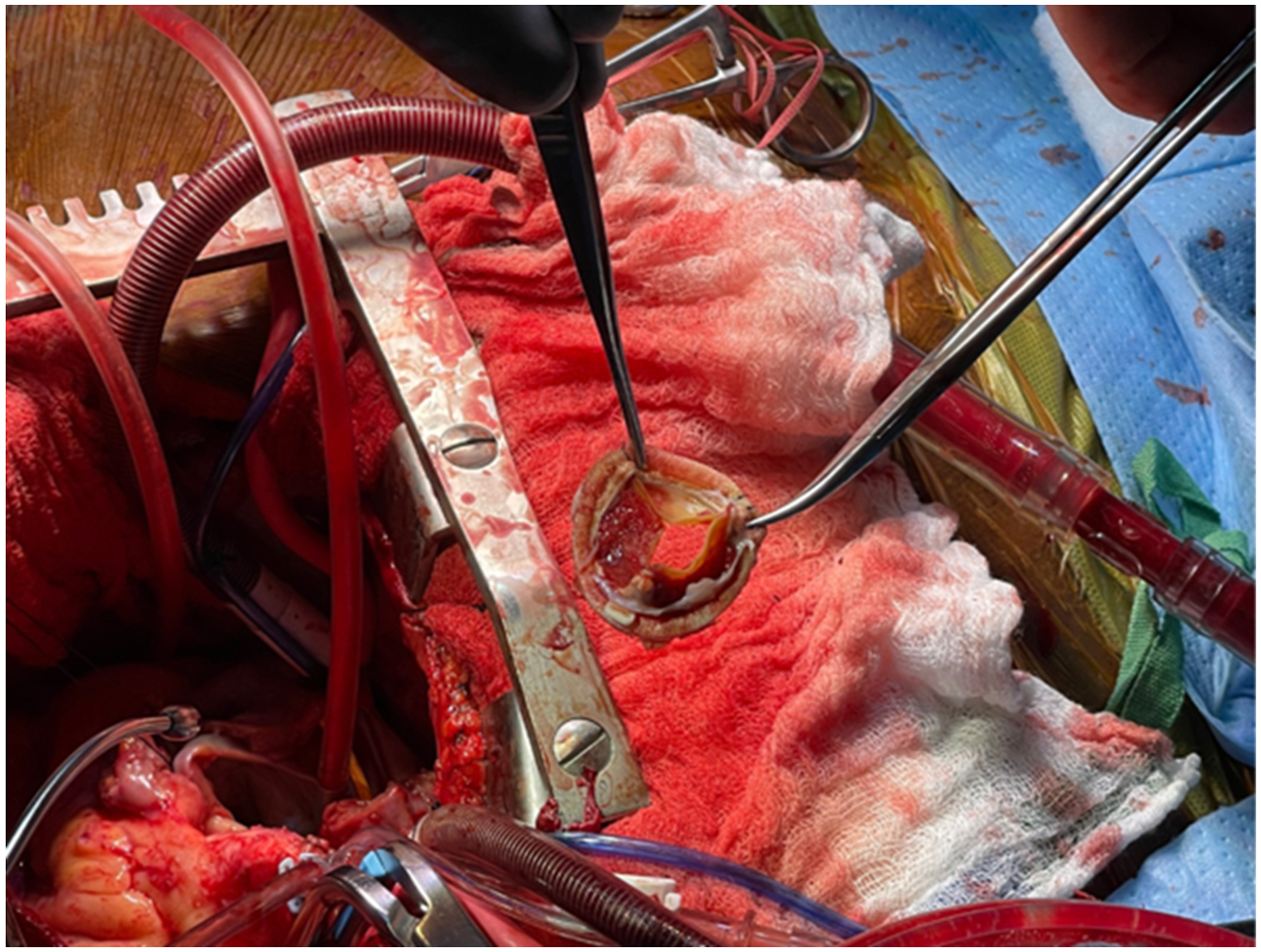
The bioprosthetic MV was replaced prior to LVAD implantation, and the left atrial appendage was occluded. Post-implantation, TEE showed proper inflow cannula positioning and moderate right ventricular dysfunction necessitating inotropic and vasoactive support along with intravascular volume resuscitation (6 units of packed red blood cells, 6 units of fresh frozen plasma, 4 pooled units of platelets, and 20 units of cryoprecipitate). Intraoperative course also complicated by vasoplegia with weaning of cardiopulmonary bypass that improved after hydroxocobalamin (Cyanokit). She was anticoagulated with heparin (400 units/kg) in preparation for cardiopulmonary bypass (CPB). Activated clotting time post-CPB after protamine was 121. Total CPB time was 188 minutes. Post-procedure TEE showed an EF of 10% with mild to moderate right ventricular dysfunction. MV mean gradient was 3 mmHg (Figure 4), and there was no evidence of paravalvular leak. She was transferred to the intensive care unit with epinephrine, dobutamine, and vasopressin drips. Post cardiopulmonary bypass TEE demonstrating a mean gradient of 3 mmHg across the replaced bioprosthetic mitral valve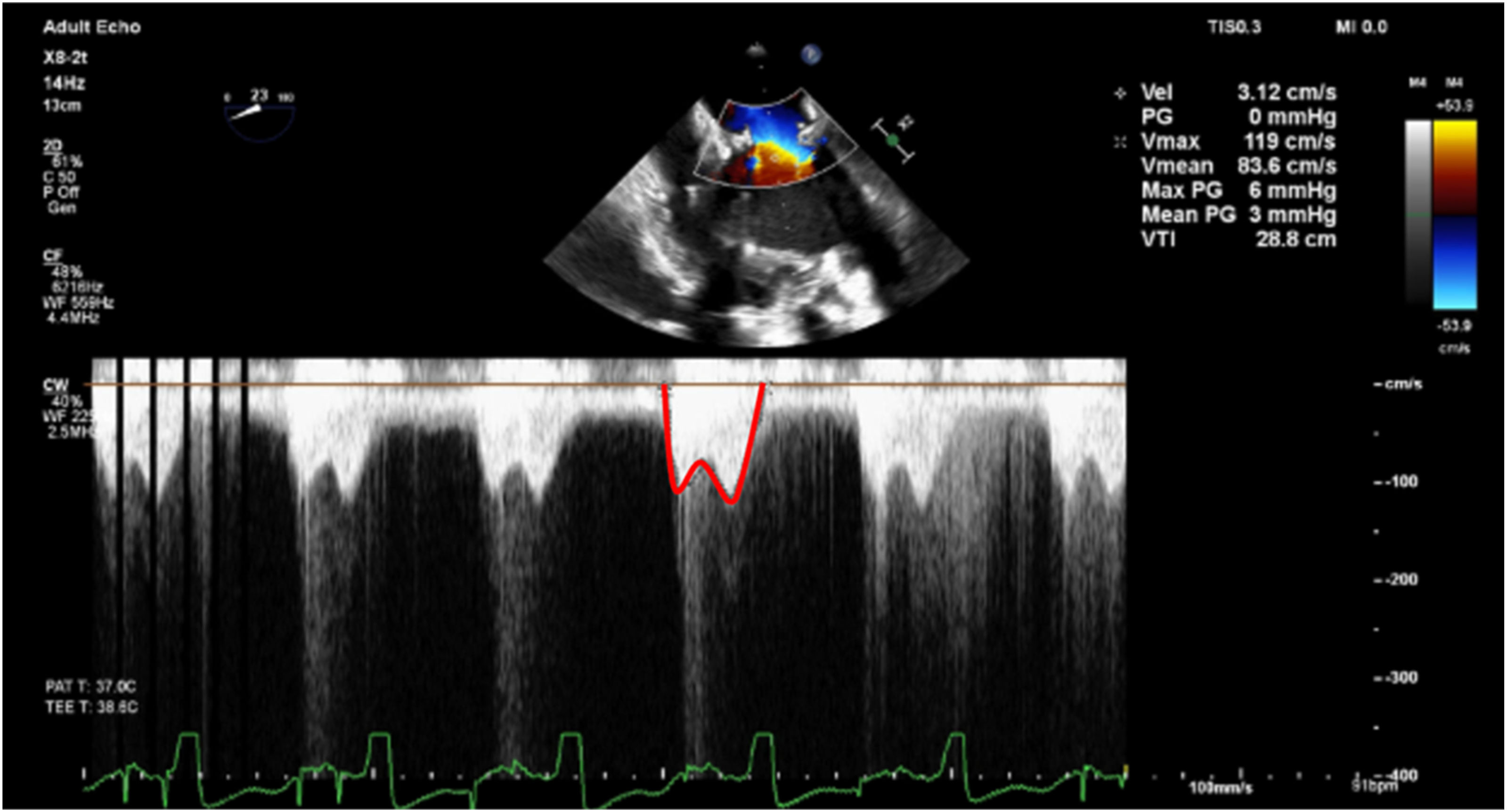
Her hospital course was complicated by right heart failure requiring sildenafil therapy. Right heart catheterization demonstrated a right atrial/pulmonary capillary wedge pressure ratio of 0.9 and a thermodilution cardiac index of 1.74 L/min/m^2. She was bridged with heparin and transitioned to warfarin on postoperative day (POD) 7. Later during her hospitalization on POD 34, she had an interval development of subacute subdural hematoma. She subsequently received a dose of intravenous Vitamin K 10 mg and underwent bilateral middle meningeal artery embolization. Her INR goal was adjusted from 2–2.5 to 1.8–2.2. She was discharged to inpatient rehabilitation on POD 49 with a warfarin regimen of 2 mg on Monday, Wednesday, Friday, and Saturday and 1 mg on Tuesday, Thursday, and Sunday. She was discharged to home on POD 64 in stable condition. She continues to follow up routinely with the Heart Failure clinic and has not demonstrated any subsequent thromboembolic complications since the surgery.
Discussion
The incidence rate of mechanical valve thrombosis is approximately 0.4 per 100 patient-years, with mitral valve thrombosis slightly higher at 0.5 per 100 patient-years. The greatest thromboembolic risk occurs within the first three months after valve replacement. Additional risk factors include lack of anticoagulation, mitral valve replacement, previous thromboembolism, and increasing age.3,4 In our patient, the thrombus was discovered within one year of her mitral valve replacement. Her risk factors included her interrupted anticoagulation regimen, mitral valve replacement, a history of LV thrombus, and advanced age. The etiology is thought to involve Virchow’s triad: endothelial injury from surgery, hypercoagulability due to thrombogenic prosthetic material, and blood stasis from cardiac dysfunction and areas of low-velocity flow. 3
In this case, the mitral prosthetic thrombus was missed on TTE, while TEE was instrumental in diagnosing the thrombus and guiding intraoperative surgical planning. Although TTE is more accessible and less invasive, its utility can be limited by patient anatomy, operator experience, and ultrasound artifacts. Specifically, thrombi can be obscured by artifact and near-field clutter. 5 Several case reports have highlighted the use of intraoperative TEE to diagnose intracardiac thrombi and guide management during major surgeries, such as liver transplantation. TEE provides valuable diagnostic details—including size, location, leaflet motion, and hemodynamic effects of the thrombus—guides hemodynamic management, and aids in assessing resuscitative efforts.3,6 Notably, thrombi smaller than 0.8 cm2 on TEE are associated with lower complication risks after thrombolytic therapy. 7 However, our patient’s thrombus measured 2 cm × 1.2 cm and was located at the prior mitral valve suture line and leaflets. The real-time capabilities of TEE allowed efficient communication with the surgical team and guided management without delays in care.
An intracardiac thrombus is clinically significant as it independently predicts stroke and mortality within six months post-LVAD implantation. 8 The devastating impact of stroke is underscored by data from the Interagency Registry for Mechanical Assisted Circulatory Support (INTERMACS), which showed that 45% of ischemic stroke patients and 70% of hemorrhagic stroke patients did not survive beyond one year post-LVAD. 9 Therapeutic options include intraoperative heparin administration, thrombolytic therapy, or surgical clot removal. However, no randomized controlled trials directly compare medical therapy with surgery, so treatment decisions are typically based on surgical risk, patient-specific factors, and thrombus characteristics on imaging. 3
The 2023 International Society for Heart and Lung Transplantation Guidelines for Mechanical Circulatory Support does not recommend replacing a functioning bioprosthetic valve at device implantation (Class I, Level C evidence). Conversely, removal of an intracardiac thrombus at the time of device implantation is Class I, Level C evidence.2,10 In our patient’s case, extensive thrombus on the bioprosthetic mitral valve and evidence of moderate mitral stenosis (mean gradient 9 mmHg) on intraoperative TEE prompted a change in the surgical plan. The team engaged the Cardiothoracic Surgery service and the patient’s family to discuss next steps. Cardiology was urgently consulted in the operating room to evaluate management options for the LV thrombus, the bioprosthetic valve, and the feasibility of proceeding with LVAD placement. The patient’s medical power of attorney (POA) affirmed that she would prefer to proceed with surgery rather than delay. Ultimately, the decision was made to replace the valve and remove the thrombus during LVAD implantation, which is uncommon but was a necessary approach to ensure the patient received the mechanical support needed to improve her quality of life.
Unlike aortic and tricuspid valve disease, where concurrent valve replacement is indicated in select patients, concurrent mitral valve replacement is relatively uncommon, especially as a redo procedure on a bioprosthetic valve. 11 Uncorrected moderate to severe mitral stenosis at time of LVAD implantation can lead to impaired ventricular filling and limit pump flow. This valvular pathology can subsequently have downstream effects of pulmonary hypertension and RV dysfunction. 12 Performing valvular surgery alongside LVAD implantation carries increased risk. A single-center study reported higher in-hospital mortality and lower 1-year survival in patients undergoing combined procedures. 13 Additionally, combined surgeries frequently require longer cardiopulmonary bypass times, which are associated with an increased risk of vasoplegia. Patients with moderate to severe vasoplegia have a higher risk of mortality (hazard ratio of 2.12). 14 This patient did develop vasoplegia post-bypass, which was treated with hydroxocobalamin (Cyanokit). In her case, the risks associated with prolonged bypass time had to be weighed against the elevated risk of stroke and death (hazard ratios of 2.45 and 2.36, respectively) linked to the presence of an intracardiac thrombus.8,14 Although some centers are exploring off-pump implantation of HeartMate 3 via thoracotomy, this approach was not suitable for our patient given her prior mitral valve replacement and intracardiac thrombus. An additional disadvantage with off-pump LVAD implantation, particularly in this case, is the inability to fully visualize the LV cavity to identify any thrombus or trabeculations that could impede device function. 15
Most literature on concomitant mitral valve replacement focuses on severe mitral regurgitation, limiting direct comparison to our patient’s case. While these studies have not demonstrated a survival benefit, some evidence suggests reduced rates of right heart failure, fewer hospital readmissions, and improved quality of life. 16
This case is notable for the combination of a redo bioprosthetic mitral valve replacement and concurrent removal of an intracardiac thrombus during LVAD implantation. Dogan et al. reported that when thrombus is appropriately managed intraoperatively, short-term outcomes (including 30-day survival, stroke free survival, and absence of pump thrombosis) are comparable between LVAD patients with and without thrombus. 17 Consistent with these findings, our patient did not experience any postoperative thromboembolic complications.
Had the thrombus been detected preoperatively, it is possible that the patient’s prior recovery from MV replacement would have influenced her willingness to undergo a repeat sternotomy. Ultimately, the decision in this case was guided by her POA. However, this case highlights the need to integrate a patient’s previous surgical experiences and the anticipated recovery trajectory into shared decision-making for advanced heart failure interventions.
Routine preoperative TEE prior to LVAD implantation is not universally performed, and how often it is utilized prior to LVAD implantation varies from institution to institution. According to ASE guidelines, TTE is generally the first-line imaging modality used to screen LVAD candidates for structural or functional abnormalities that may be absolute or relative contraindications to device implantation. 18 While TTE serves as the standard initial evaluation, its diagnostic sensitivity may be limited in patients with suboptimal acoustic windows or prosthetic valve shadowing. Therefore, institutions may selectively obtain preoperative TEE in patients with suboptimal TTE imaging or patients at elevated risk for an intracardiac thrombus.
The intraoperative identification of a thrombus on a bioprosthetic mitral valve also underscores the critical importance of multidisciplinary collaboration among cardiac anesthesiologists, cardiac surgeons, and cardiologists in the management of complex cardiac surgeries. In such scenarios, the cardiac anesthesiologist plays a pivotal role through the use of TEE to detect unexpected pathology in real time. Prompt communication of these findings facilitates immediate interdisciplinary collaboration. Input from the cardiologist offers valuable context regarding the patient’s underlying structural pathology, anticoagulation history, and thromboembolic risk, while the cardiac surgeon must rapidly integrate this information into operative decision-making. Together the team must weigh the risks of embolization or valve obstruction, reassess surgical strategy, and determine the safest and most effective course of action. The collective expertise of all specialties is essential to reassessing surgical priorities, optimizing hemodynamic management, and mitigating risk intraoperatively. This case exemplifies the indispensable role of team-based decision-making in enhancing patient safety and ensuring adaptive intraoperative care of cardiac surgical patients.
Conclusion
While prior literature describes successful management of intracardiac thrombus during LVAD implantation, our case is notable due to thrombus involvement of the suture line and leaflets of a functioning bioprosthetic mitral valve. Although routine replacement of a functioning bioprosthetic valve during LVAD surgery is generally discouraged, the thrombus’s size and location necessitated concurrent valve replacement to prevent further complications and enhance the patient’s quality of life. This case underscores the challenges and considerations in managing complex valvular pathology during LVAD implantation.
Footnotes
Ethical Considerations
As the case report is devoid of patient identifiable information, it is exempt from IRB review requirements in accordance with University of Texas Southwestern policy.
Consent for Publication
Written informed consent was obtained from the patient for publication of this case report and accompanying images.
Author Contributions
JH: writing and editing original draft and research. LS: supervision, writing and editing original draft, and research.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
