Abstract
Surgical repair of the proximal aorta is a complex endeavor, requiring cardiopulmonary bypass (CPB) and often the use of hypothermic circulatory arrest (HCA). In addition to the normal considerations for patients undergoing cardiopulmonary bypass, additional challenges include cerebral and end-organ protection during periods of circulatory arrest. This review aims to provide an up-to-date, evidence-based review on anesthetic management for proximal aortic repair.
Keywords
Introduction
Surgical repair of the proximal aorta is a complex endeavor, requiring cardiopulmonary bypass (CPB) and often the use of hypothermic circulatory arrest (HCA). In addition to the normal considerations for patients undergoing cardiopulmonary bypass, additional challenges include cerebral and end-organ protection during periods of circulatory arrest. This review aims to provide an up-to-date, evidence-based review on anesthetic management for proximal aortic repair.
Anatomy and Definitions
The aorta may be divided into 5 anatomical segments: the aortic root, the ascending thoracic aorta, the aortic arch, the descending thoracic aorta, and the abdominal aorta. 1 The aortic root is sometimes referred to as the sinus segment and extends from the aortic valve annulus to the sinotubular junction (STJ). The ascending thoracic aorta extends from the STJ to the innominate artery. The aortic arch extends from the innominate artery to the left subclavian artery. The term proximal aortic repair throughout this manuscript is used to describe the repair of any of the structures between and including the aortic root and the transverse aortic arch.
Normal values for ascending aortic size vary depending on patient gender, height, and body surface area. The traditional definition of an arterial aneurysm is a diameter 1.5 times the normal diameter. However, specifically in the case of the ascending aorta, it has been shown that patients may experience increased risk of complications at smaller aortic diameters.2,3 This phenomenon has been termed the aortic size paradox. For example, compared with aortic diameter <3.5 cm, a diameter of 4.0–4.4 cm conferred an 89-fold risk of dissection (despite being well under the traditional definition of 1.5 times the normal diameter) and a diameter >4.5 cm conferred a 6000-fold increased risk. 4 Consequently, the most recent guidelines have advocated referring to ascending aorta diameter of 4.0–4.4 cm as “dilated,” and ascending aorta with diameter >4.5 cm as “aneurysmal.” 1
The spectrum of aortic disease is shown in Figure 1. The aortic wall is comprised of 3 layers: a thin inner intima, the media (consisting of over 50 layers of smooth muscle cells, elastic fibers, collagen proteins, and polysaccharides), and a thin outer adventitia.
1
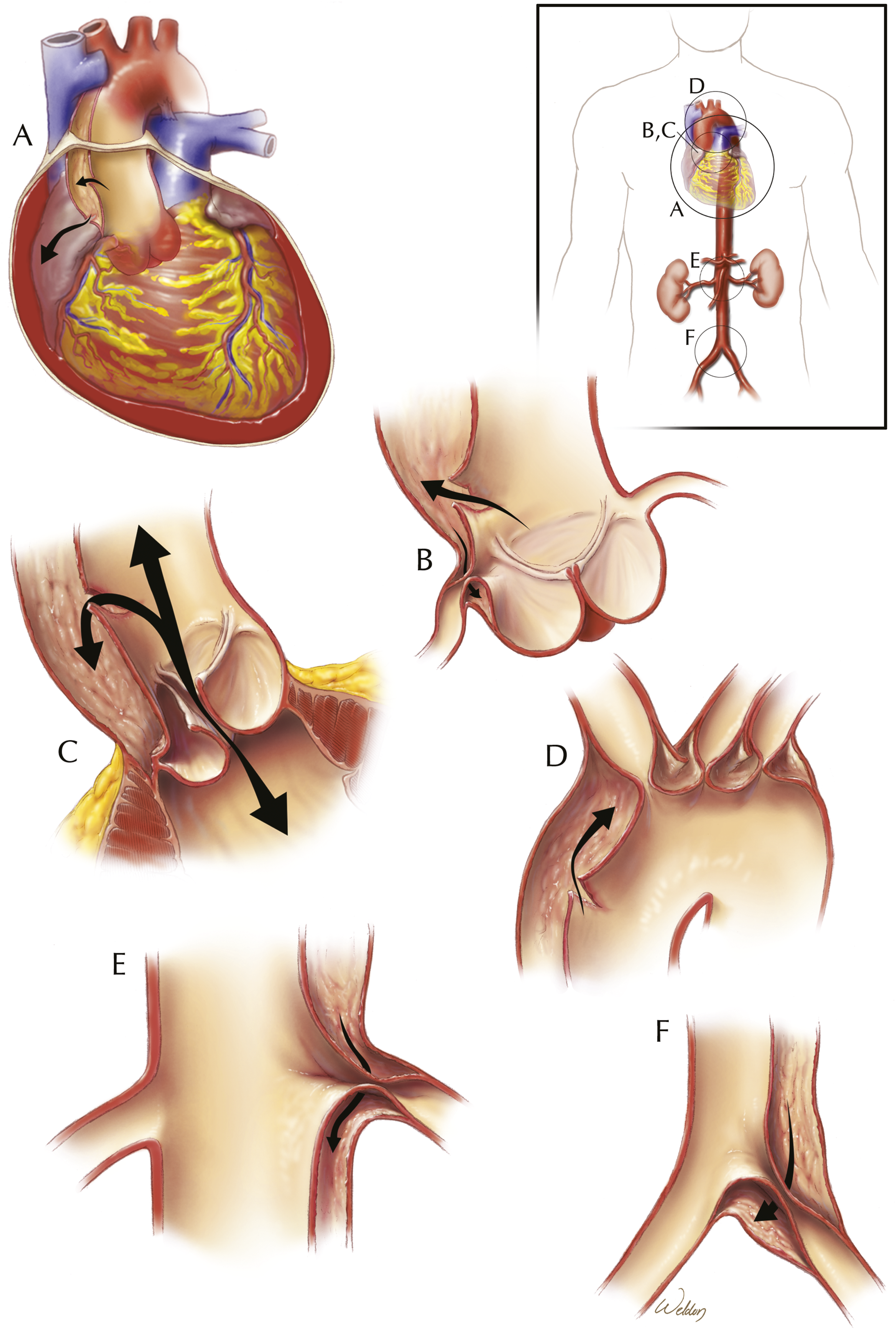
Acute aortic dissection (AAD) occurs when an intimal tear allows blood to spread through the tear into the media, creating a false lumen that is separated from the true lumen by a thin dissection flap. The dissection may continue to propagate along the aorta in either an anterograde or retrograde fashion due to increased hydrostatic pressure, potentially leading to life-threatening complications such as acute aortic valvular insufficiency (AI), myocardial ischemia from coronary involvement, tamponade, acute cerebral ischemia, renal and/or visceral malperfusion, and lower extremity ischemia.1,5 These are summarized in Figure 2. Besides AAD, other acute aortic syndromes (AAS) include intramural hematoma (IMH), characterized by hemorrhage within the media of the aortic wall without overt intimal disruption (e.g., due to rupture of the vasa vasorum), and penetrating atherosclerotic ulcers (PAU), where ulceration of an atherosclerotic plaque allows blood to enter the media but scarring of the aorta keeps the blood confined to a localized area, resulting in a localized dissection or pseudoaneurysm.
1
Spectrum of aortic pathologies that require aortic repair. Normal aorta and pathologic variants of aortic disease that require surgical repair. The normal aortic wall is composed of several layers—an innermost intima, the media (composed of smooth muscle cells, elastic fibers, and collagen), and an outer adventitia. In penetrating aortic ulcer (PAU), there is ulceration of an atherosclerotic lesion that penetrates the aortic intima and progresses through the internal lamina into the media. In aortic dissection, a tear in the intima allows blood to expand into the media, creating a false lumen. Intramural hematoma (IMH) is characterized by hemorrhage within the media without an overt disruption of the intima. When involving the proximal aorta, PAU, dissection, and IMH are collectively termed acute aortic syndromes and are considered surgical emergencies. Finally, aortic aneurysm is the either fusiform or saccular dilatation of the aorta in response to increased wall shear stress. Used with permission from Baylor College of Medicine. Abbreviations: IMH, intramural hematoma; PAU, penetrating atherosclerotic ulcer. Potential complications of aortic dissection. Acute aortic dissection is associated with a number of potential complications, shown here. (A) Pericardial effusion with or without tamponade, caused by extravasation of fluid from the false lumen into the pericardium. (B) Coronary obstruction by the dissection flap, which can lead to myocardial ischemia. (C) Disruption of normal aortic root geometry by the dissecting aneurysm, leading to aortic valvular insufficiency. (D) Obstruction of the great vessels by the dissection flap, leading to cerebral ischemia and stroke-like symptoms. (E) Obstruction of branch vessels by the dissection flap, such as the celiac artery, superior mesenteric artery, or renal arteries, leading to visceral malperfusion. (F) Obstruction of the common iliac artery by the dissection flap, leading to lower extremity malperfusion. Used with permission from Baylor College of Medicine.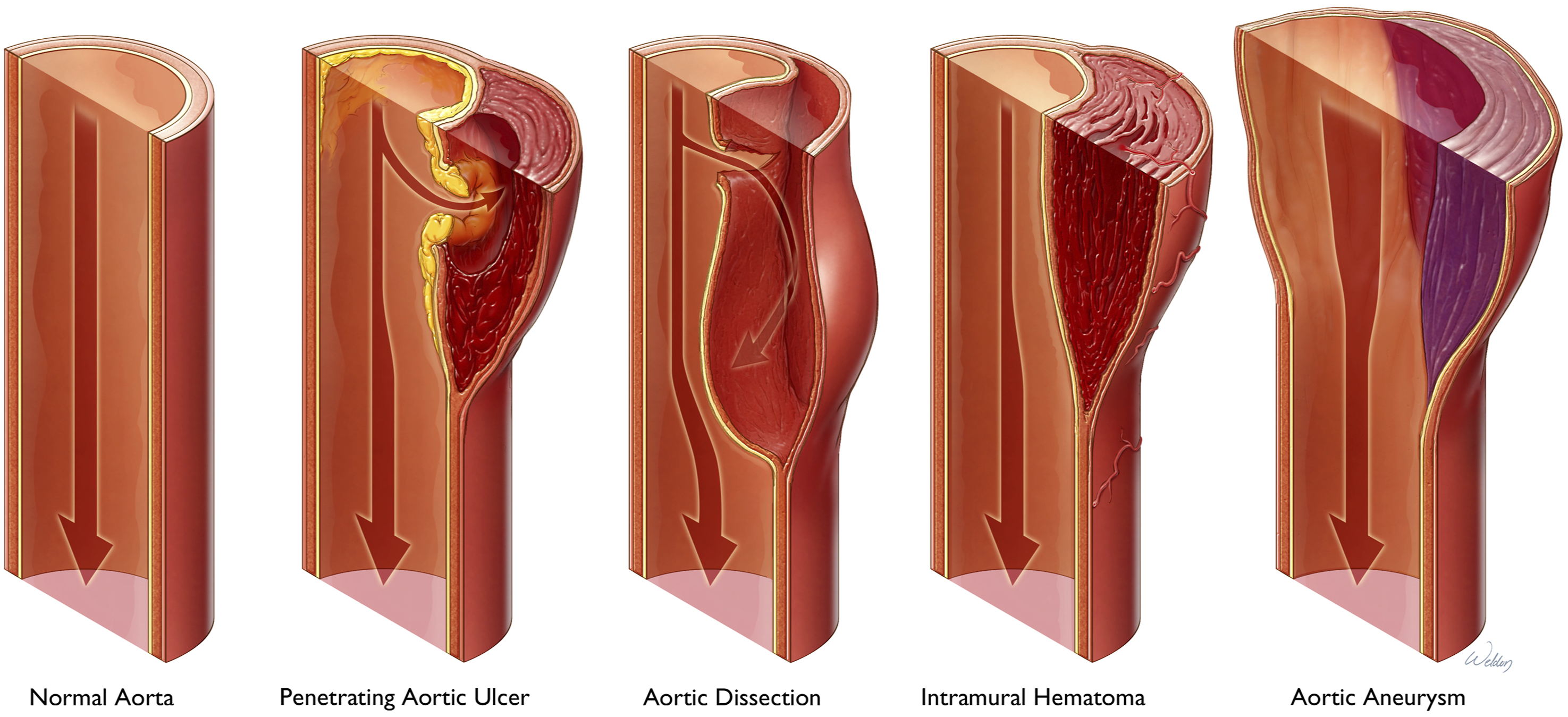
Aortic dissections are most frequently classified using the Stanford and/or DeBakey classification systems. Stanford type A aortic dissections (DeBakey type I and II) involve the ascending aorta and are more likely to present acutely, whereas Stanford type B dissections (DeBakey type III) arise distal to the left subclavian artery and primarily involve the descending thoracoabdominal aorta. Additional guidelines for the categorization of type B dissections were proposed in 2020 by the Society of Vascular Surgeons and Society of Thoracic Surgeons (STS), which incorporates descriptors for the location of the intimal tear as well as the proximal and distal extent of the dissection. 6 Even more recently, the STS and European Society for Cardiothoracic Surgery released guidelines advocating for the use of the TEM (Type, Entry, Malperfusion) classification for AAD. 7
AADs involving the ascending aorta are considered time-sensitive surgical emergencies. Although early mortality with Stanford type A AADs has traditionally been considered to be as high as 1–2% per hour after symptom onset, 8 in the modern era, with advances in medical management, mortality is now thought to be closer to 0.5% per hour. 9 Nevertheless, when diagnosed, prompt surgical repair is the gold standard of care, and preferably at a high-volume center.1,10
Preoperative Evaluation and Management
Patient Risk Factors and Comorbidities
Thoracic aortic aneurysms occur in 5–10 per 100,000 person-years, and the incidence of acute dissection is 5–30 cases per million person-years. 1 Common risk factors for thoracic aortic disease (TAD) involving the proximal aorta include hypertension, smoking, hyperlipidemia, and the presence of syndromic heritable thoracic aortic diseases (HTAD), non-syndromic HTAD, and biscupid aortic valve-associated aortopathy. Additionally, drug use (especially cocaine) is a risk factor for dissection.
HTADs are conditions in which weakness of the aortic wall predisposes patients to aortic dilatation, aneurysm formation, and acute aortic syndromes. 11 These may be classified as either syndromic or non-syndromic. Examples of syndromic HTADs include Marfan syndrome, Loeys-Dietz syndrome, vascular Ehlers-Danlos syndrome, and Turner syndrome. Non-syndromic HTAD include mutations of ACTA2, MYH11, PRKG1, and MYLK, among others.
Anesthetic considerations for Marfan syndrome, 12 Ehlers-Danlos syndrome,13,14 Loeys-Dietz syndrome,15,16 and Turner syndrome 17 have been published elsewhere previously, and should be heeded.
Imaging Assessment of the Proximal Aorta
The proximal aorta can be imaged using a variety of different modalities, including echocardiography, magnetic resonance imaging (MRI), and computed tomography (CT).1,7 Arterial phase contrast-enhanced CT has a very high sensitivity and specificity for AAS and has very high spatial resolution, which facilitates the identification of coronary involvement, great vessel involvement, hemopericardium, and the location of intimal tears. MRI can visualize aortic wall changes in the setting of inflammation and offer physiologic assessment of ventricular and valvular function but has a lower spatial resolution than CT. Echocardiography is particularly useful for assessment of the aortic root and in delineating the function of the aortic valve. In patients with a suspected AAS, CT is recommended for initial diagnostic imaging, given its accuracy, speed, and the extent of anatomic information it provides. 1
For the anesthesiologist, preoperative review of the patient’s imaging is mandatory. One should identify the diseased portions of the proximal aorta and take into consideration the anticipated surgical plan, including likely cannulation sites for CPB, the degree of aortic valvular insufficiency, and whether circulatory arrest will be required for a hemi-arch or total arch repair. In the case of aortic dissection, it is critical to identify the extent of the dissection flap, confirm patency of the cerebral and subclavian vessels, and identify evidence of malperfusion. All this information should inform the anesthetic plan, especially in regard to the application and interpretation of monitors and in the hemodynamic management of the patient.
Indications for Surgical Repair
Indications for proximal aortic repair include acute aortic syndromes, chronic dissection, or chronic aneurysmal disease of the thoracic aorta. Acute aortic syndromes, including acute dissection, IMH, and PAU with or without rupture of the aorta, frequently require emergent repair.
Indications for Aortic Repair in Chronic Aneurysmal Disease.
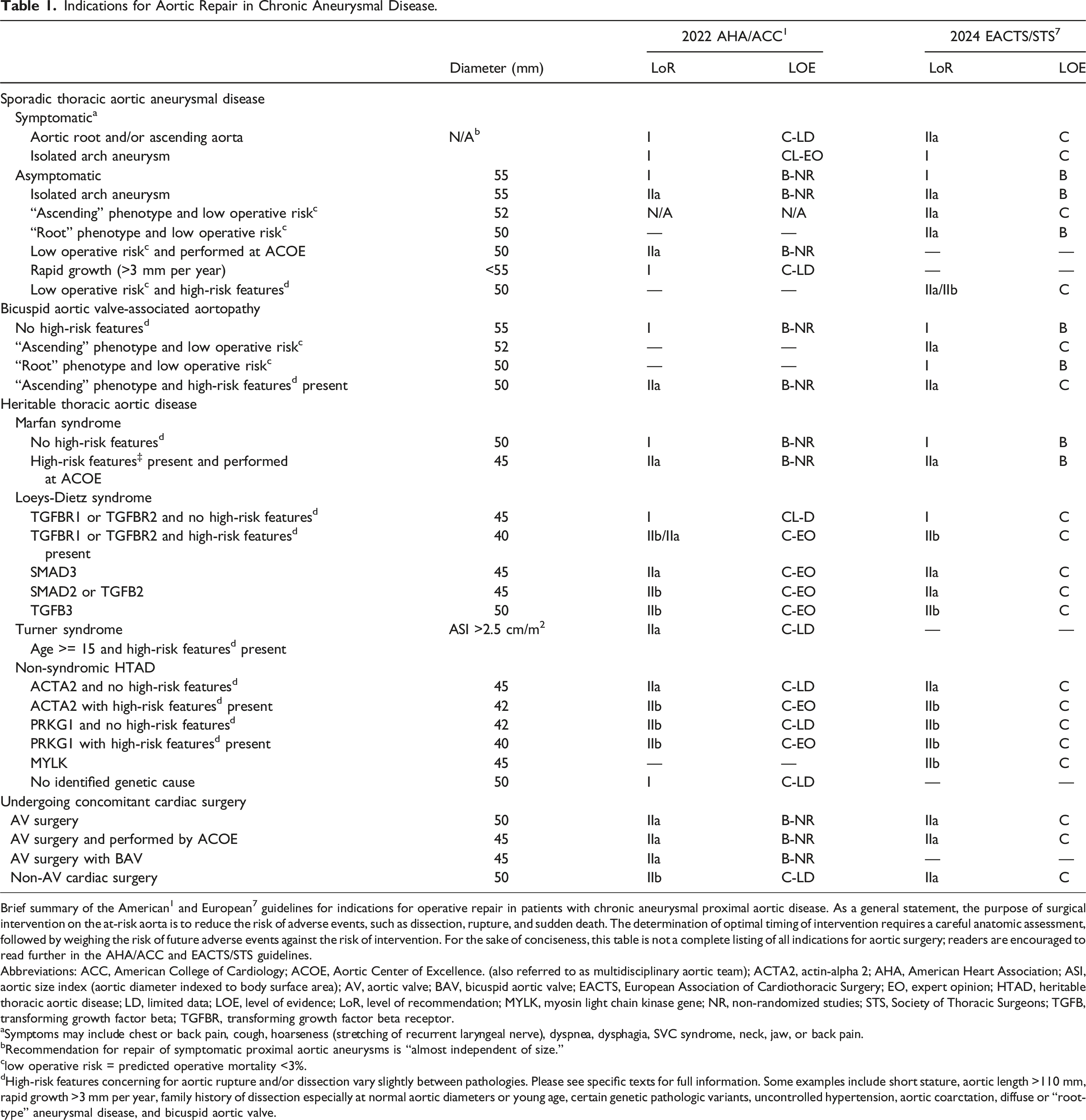
Brief summary of the American 1 and European 7 guidelines for indications for operative repair in patients with chronic aneurysmal proximal aortic disease. As a general statement, the purpose of surgical intervention on the at-risk aorta is to reduce the risk of adverse events, such as dissection, rupture, and sudden death. The determination of optimal timing of intervention requires a careful anatomic assessment, followed by weighing the risk of future adverse events against the risk of intervention. For the sake of conciseness, this table is not a complete listing of all indications for aortic surgery; readers are encouraged to read further in the AHA/ACC and EACTS/STS guidelines.
Abbreviations: ACC, American College of Cardiology; ACOE, Aortic Center of Excellence. (also referred to as multidisciplinary aortic team); ACTA2, actin-alpha 2; AHA, American Heart Association; ASI, aortic size index (aortic diameter indexed to body surface area); AV, aortic valve; BAV, bicuspid aortic valve; EACTS, European Association of Cardiothoracic Surgery; EO, expert opinion; HTAD, heritable thoracic aortic disease; LD, limited data; LOE, level of evidence; LoR, level of recommendation; MYLK, myosin light chain kinase gene; NR, non-randomized studies; STS, Society of Thoracic Surgeons; TGFB, transforming growth factor beta; TGFBR, transforming growth factor beta receptor.
aSymptoms may include chest or back pain, cough, hoarseness (stretching of recurrent laryngeal nerve), dyspnea, dysphagia, SVC syndrome, neck, jaw, or back pain.
bRecommendation for repair of symptomatic proximal aortic aneurysms is “almost independent of size.”
clow operative risk = predicted operative mortality <3%.
dHigh-risk features concerning for aortic rupture and/or dissection vary slightly between pathologies. Please see specific texts for full information. Some examples include short stature, aortic length >110 mm, rapid growth >3 mm per year, family history of dissection especially at normal aortic diameters or young age, certain genetic pathologic variants, uncontrolled hypertension, aortic coarctation, diffuse or “root-type” aneurysmal disease, and bicuspid aortic valve.
Preoperative Anesthetic Evaluation
Prior to the preoperative interview, the anesthesiologist should personally review the patient’s imaging paying special attention to the patient’s anatomy, location of aneurysm/dissection and presence of great vessel involvement as this will impact the surgeon’s approach, line placement and potential complications.
In elective cases, during the preoperative interview by the anesthesiologist, a thorough history and physical should be performed. Pulses in all extremities should be palpated. It is important to auscultate for decrescendo early-diastolic blowing murmur indicative of aortic insufficiency (AI) and therefore valve involvement. In patients with aortic dissection, a thorough baseline neurologic assessment should be performed, as well as an assessment for evidence of malperfusion. As for any major cardiovascular surgery, baseline labs, including a complete blood count, complete metabolic panel, coagulation studies, and a type and screen, as well as a baseline EKG and chest X-ray should be obtained. 18
In cases where the patient must proceed emergently to the OR, such as type A dissection, it may not always be possible to perform a thorough preoperative evaluation. At a minimum, it is immensely helpful to have knowledge of the extent of dissection (whether by review of the imaging or through a discussion with the surgeon) and perform a focused assessment for complications arising from the dissection (Figure 2) since that information may impact line placement, interpretation of arterial readings, and anesthetic management.
Overview of the Surgical Approach for the Anesthesiologist
The authors of this review firmly believe that having a strong grasp of surgical approaches, considerations, and rationale make for a better cardiovascular anesthesiologist. Therefore, a review of surgical techniques, with an emphasis on the details pertinent to the anesthesiologist, is presented here.
Techniques of Open Surgical Repair
Aortic root repair requires resection and replacement of the aortic root, reimplantation of coronary buttons, and depending on the status of the aortic valve, either resuspension of the aortic valve leaflets or aortic valve replacement. Different strategies for repair of the aortic root are shown in Figure 3. Types of aortic root replacement. Depending on the pathology, anatomy, and other patient factors, a number of options are clinically available for aortic root replacement. Replacement of the aortic root with a composite graft with mechanical valve is also known as a Bentall procedure. Valve-sparing root replacement, in which the aortic valve leaflets are resuspended in the graft, is also known as a David procedure. A homograft is a cadaveric aortic root and ascending aorta; it is used most frequently in the setting of endocarditis, as compared with the other options, it has the lowest risk of infection. Used with permission from Baylor College of Medicine.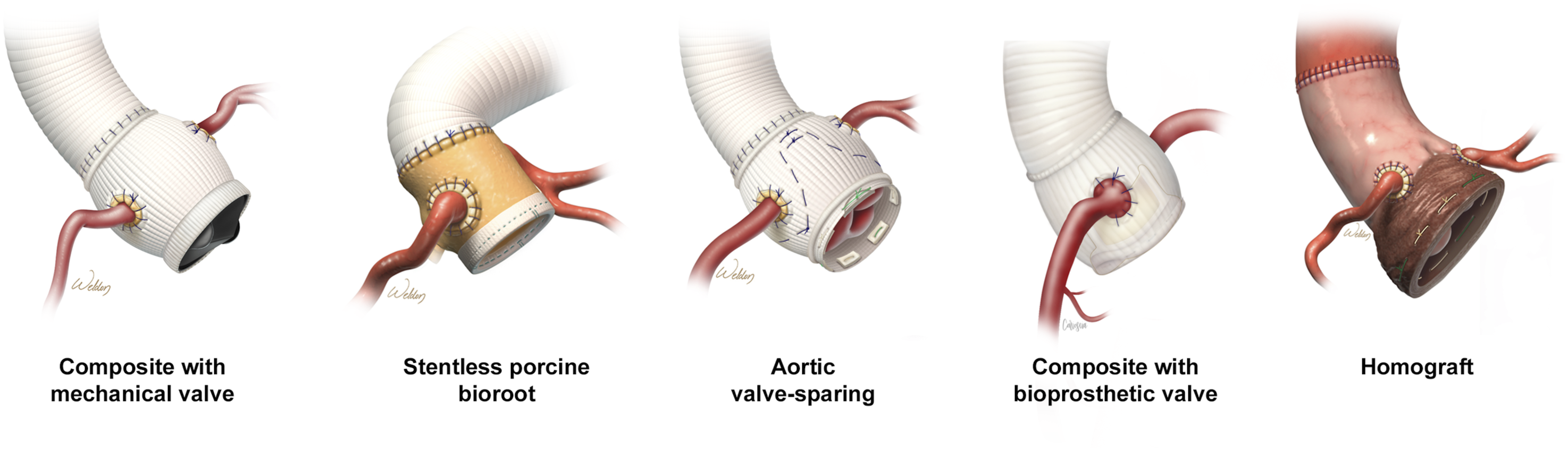
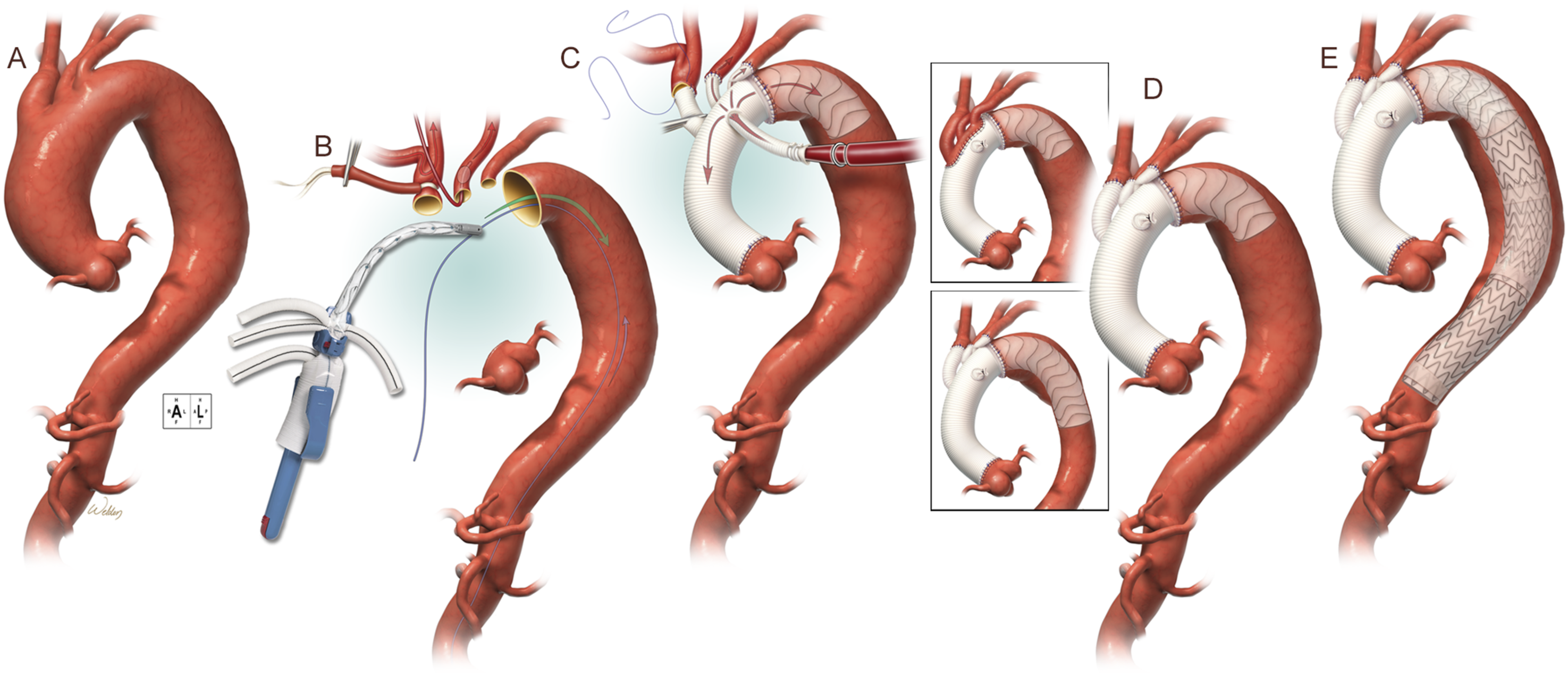
Repair of the tubular portion of the ascending aorta is straightforward and involves replacement with an interposition graft during CPB. However, If the distal portion of the ascending aorta or portions of the transverse aortic arch requires replacement, then HCA is required in order to maintain a bloodless surgical field. Different types of proximal aortic repairs are shown in Figure 4 and one option for hybrid repair of the aortic arch is shown in Figure 5. Types of proximal aortic repair. Final appearance of a variety of different proximal aortic repairs. The type of repair performed depends on the extent of diseased aorta, presence of dissection, patient anatomic factors, and other patient factors such as presence of a connective tissue disorder associated with aortopathy or planned future interventions. Used with permission from Baylor College of Medicine. Technique for hybrid repair of the aortic arch. One approach for hybrid total aortic arch repair is shown. (A) Aneurysmal disease of the ascending aorta, aortic arch, and descending aorta are shown. While repair of the ascending aorta is the immediate priority, future repair of the descending aorta will be needed. In this case, a frozen elephant trunk technique, with deployment of a stent graft in the descending aorta, can facilitate future repair. (B) After performing a median sternotomy and establishing cardiopulmonary bypass via right axillary artery cannulation, the aorta was transected proximally at the sinotubular junction and distally just beyond the left subclavian artery. A balloon catheter was inserted into the left common carotid artery to provide antegrade cerebral perfusion. A guide wire for the deployment procedure was inserted into the femoral artery and advanced retrograde with subsequent retrieval from the opening of the descending thoracic aorta. The tip of the device was threaded onto the guide wire and advanced into position in the descending thoracic aorta. (C) The endograft portion of the device was deployed, and the delivery system was separated and removed from the graft. The device was secured by suturing the collar to the distal native aortic remnant. In this patient without aortic root disease, the proximal anastomosis was completed at the level of the sinotubular junction. Aortic perfusion was provided via a side branch. The brachiocephalic arteries were anastomosed. Reattachment of the innominate artery might necessitate ceasing perfusion through the right axillary artery as the tourniquet is removed. (D) The completed stage 1 repair using the thoraflex hybrid plexus device is shown. The brachiocephalic arteries were replaced with branch grafts. The thoraflex hybrid ante-flo device may be used to reattach the brachiocephalic arteries as an island patch (inset). (E) 2 weeks after the initial repair, a stage 2 extension procedure was performed via retrograde deployment of 2 stent grafts to provide definitive repair of the descending thoracic aorta. There is considerable overlap between the endograft portion of the hybrid device and the first stent graft. Used with permission from Baylor College of Medicine.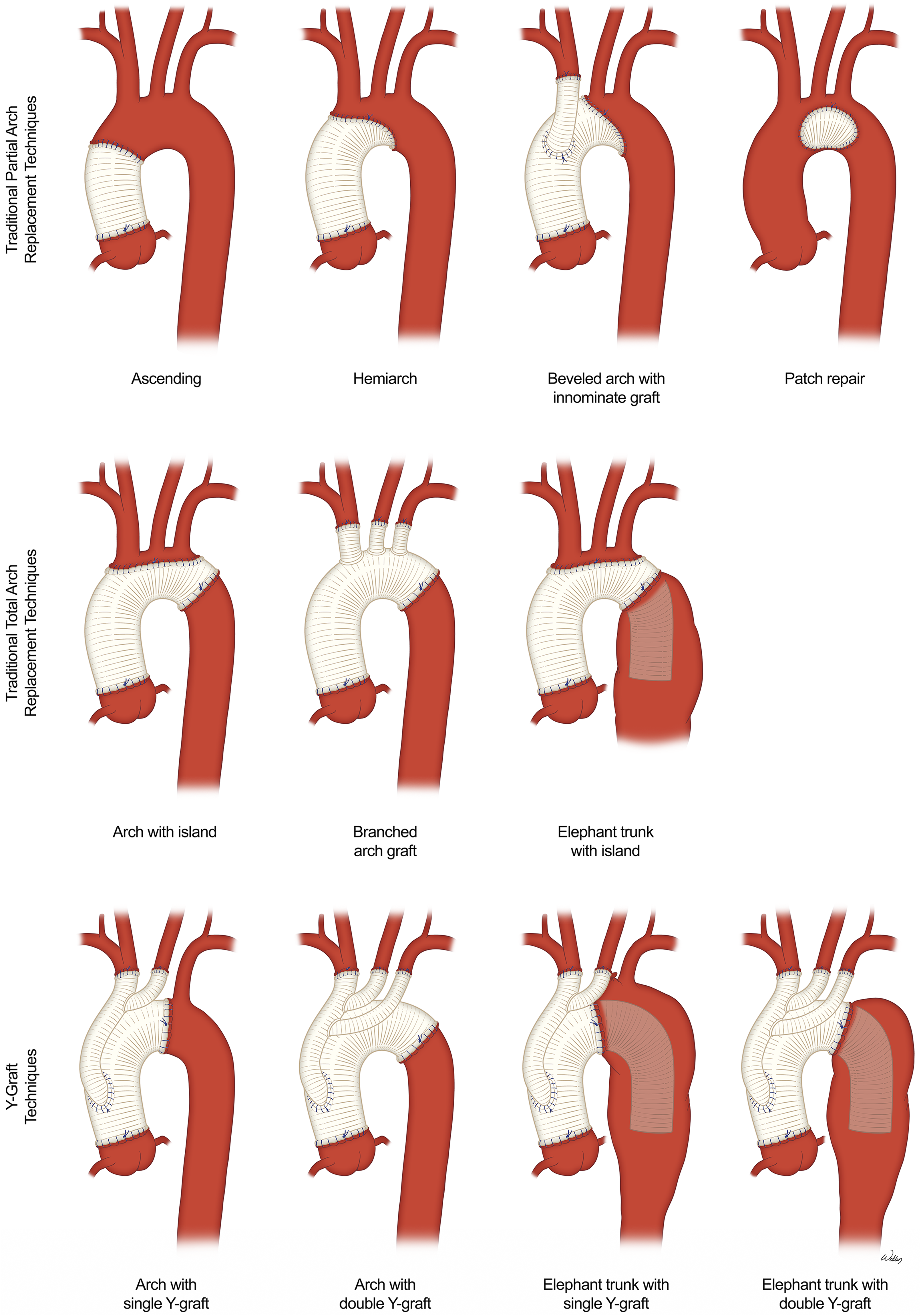
Cannulation for Cardiopulmonary Bypass
Due to the impact on arterial pressure monitoring location and management, it is important to be familiar with different cannulation strategies that may be encountered in proximal aortic surgery. Venous cannulation for CPB is straightforward and may be done either centrally or peripherally.
Advantages and Disadvantages of Various Arterial Cannulation Sites.
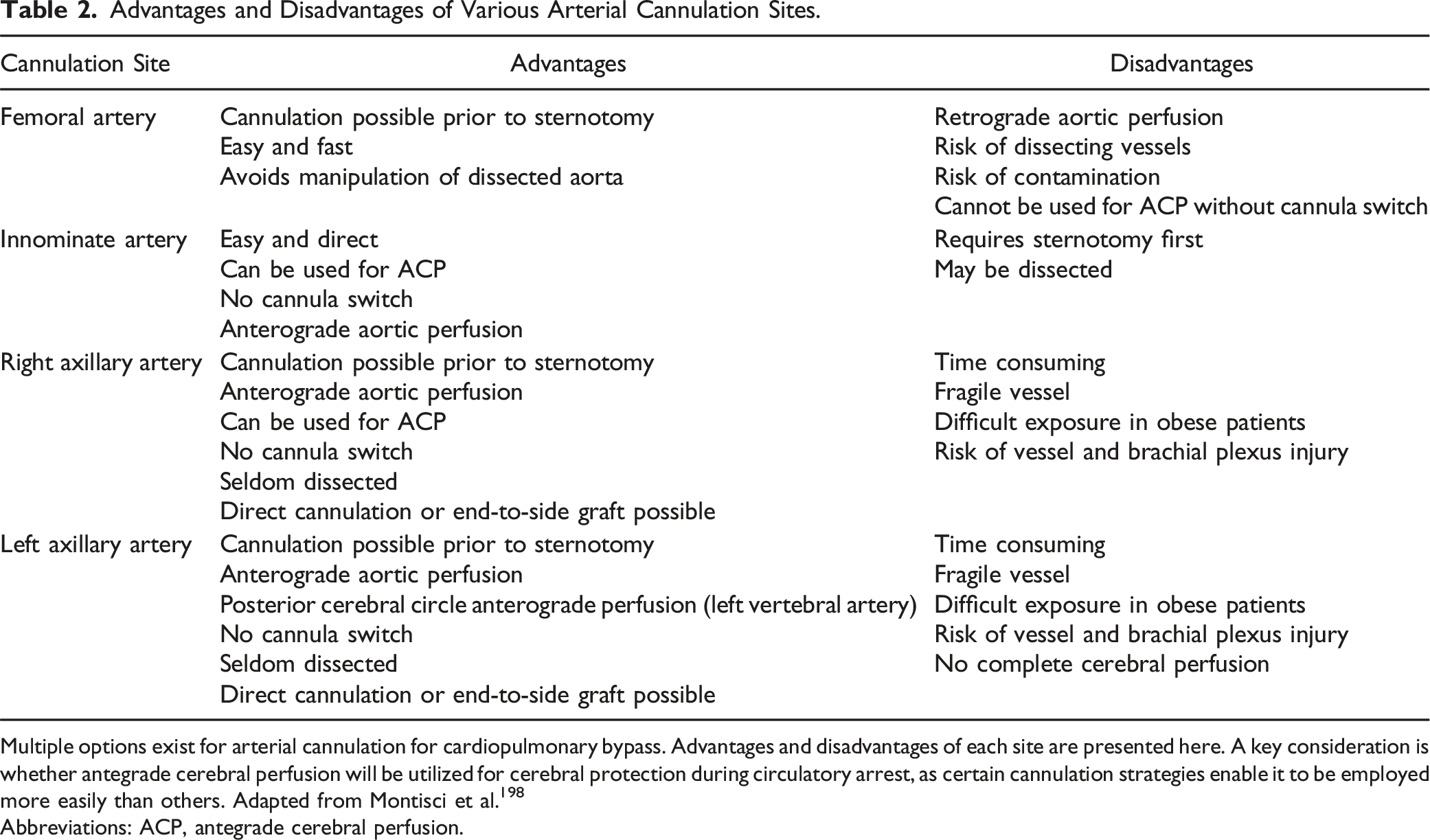
Multiple options exist for arterial cannulation for cardiopulmonary bypass. Advantages and disadvantages of each site are presented here. A key consideration is whether antegrade cerebral perfusion will be utilized for cerebral protection during circulatory arrest, as certain cannulation strategies enable it to be employed more easily than others. Adapted from Montisci et al. 198
Abbreviations: ACP, antegrade cerebral perfusion.
As mentioned previously, repair of the aortic arch must be done under circulatory arrest. Due to the limited accessibility to the distal arch and descending aorta through a sternotomy approach, in patients who must undergo repair of the proximal transverse arch (i.e., “hemi-arch” repair) or the complete transverse arch (i.e., “total arch” repair), it is immensely difficult to provide systemic and/or cerebral blood flow by the arterial CPB cannula while the arch is being operated upon. Therefore, adjunctive perfusion techniques are required (described below). For the same reason, central aortic cannulation is not possible in patients undergoing aortic arch repair, since the path of arterial CPB outflow would be interrupted when the arch is opened, and high rates of blood flow would obscure the field.
Instead, when the aortic arch requires repair, either the innominate artery or the right axillary artery is often the preferred cannulation site. While the femoral artery may be selected for arterial cannulation for CPB, the innominate or axillary artery is typically preferred due to the ease with which full CPB can be converted to selective antegrade cerebral perfusion (ACP) during circulatory arrest, simply by the application of a tourniquet at the base of the innominate artery and changing the pump flow rate. A randomized study comparing the safety and efficacy of axillary vs innominate cannulation showed a similar degree of neuroprotection between the 2 approaches; therefore, either is acceptable. 19 At the authors’ institution, the innominate artery is the preferred approach, unless it is dissected or unless the patient is undergoing redo sternotomy, in which case the right axillary artery is utilized (before sternotomy).
For arterial cannulation of either the right axillary or innominate artery, an 8–10 mm Dacron graft may be sewn on to the artery in an end-to-side fashion, as shown in Figure 6. The arterial CPB limb is then connected to this graft. Cannulation strategies for CPB that facilitate ACP. Cannulation strategies for cardiopulmonary bypass in proximal aortic repair when antegrade cerebral perfusion will be utilized. (A) Cannulation of the right axillary artery with a Dacron graft. A cutdown is performed to expose the right axillary artery. Care is taken to avoid the brachial plexus during dissection. After administration of heparin (3000–5000 units), a side-biting clamp is placed on the axillary artery and an 8 mm Dacron graft is sewn onto the artery in an end-to-side fashion. The clamp is then removed, and once hemostasis is confirmed, the arterial limb of the CPB circuit is connected to the graft using a 3/8-inch perfusion connector. At the authors’ institution, this approach is favored in redo sternotomies or if the innominate artery is dissected. (B) Cannulation of the innominate artery with a Dacron graft. After performing median sternotomy and dissection to facilitate exposure, heparin (typically 5000 units) is administered. A clamp is placed on the innominate artery and an 8- or 10-mm Dacron graft is sewn onto the artery in an end-to-side fashion. Close attention is paid to the NIRS during this time. Once the graft is sewn on, the clamp is removed, and the arterial limb of the CPB circuit is connected to the graft with a 3/8-inch perfusion connector. At the authors’ institution, this is the favored approach. (C) Configuration for HCA and administration of ACP with an axillary arterial graft. The patient is placed on full cardiopulmonary bypass and cooled to the target temperature. Once adequately cool, neuroprotective drugs are administered and ice is placed on the patient’s head. A Rummel tourniquet is placed at the base of the innominate artery and snared at the same time that CPB flows are dropped to 10 mL/kg/min. This marks the onset of HCA with unilateral ACP. The standard approach at the author’s institution is the use of bilateral ACP. To accomplish this, during setup for the procedure, an additional Y-limb is placed on the arterial CPB circuit. The additional Y-limb is connected to a balloon-occludable perfusion cannula which is placed up into the left common carotid artery after the aorta is opened for bilateral ACP. This perfusion cannula is left in place while the distal anastomosis is completed and removed for the final couple of sutures. Using this approach, the patient remains on bilateral ACP for all but 1–2 minutes of HCA. (D) Configuration for HCA and administration of ACP with an innominate arterial graft. Similar to cannulation of the right axillary artery, at the onset of HCA, a Rummel tourniquet is placed at the base of the innominate artery and flows are dropped to 10 mL/kg/min for unilateral ACP. An extra Y-limb on the arterial CPB circuit is used with a balloon-occludable perfusion cannula to perfuse the left common carotid artery during the distal aortic anastomosis. Used with permission from Baylor College of Medicine. Abbreviations: ACP, antegrade cerebral perfusion; CPB, cardiopulmonary bypass; HCA, hypothermic circulatory arrest.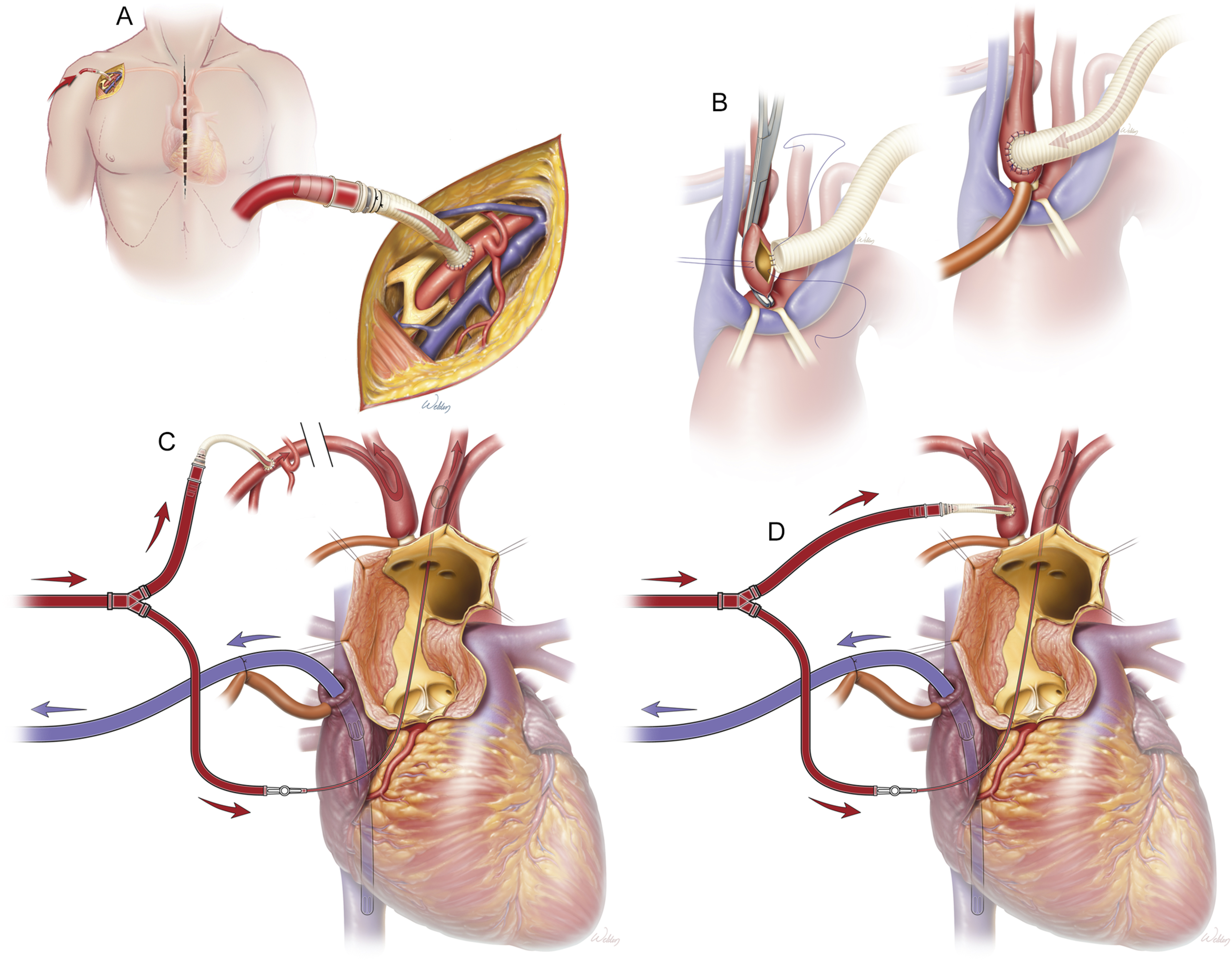
Surgical Aspects of Cerebral Protection During Circulatory Arrest
Rationale for Cerebral Protection During Circulatory Arrest
The brain has a high metabolic rate, a constant oxygen requirement, and is extremely vulnerable to ischemic injury if its oxygen supply is interrupted, even for brief periods. 20 Consequently, the brain is at significant risk during operative repair of the aortic arch when interruption of the great vessels and circulatory arrest is required. Although the incidence of clinically overt stroke after aortic surgery with HCA is between 5% and 8%,21,22 new cerebral lesions are seen much more frequently on MRI—as high as in 35–40% of patients. 19 Although they may not responsible for clinically overt focal neurologic deficits, these lesions likely play a pathophysiologic role in neurocognitive dysfunction that occurs postoperatively and may have a significant impact on patient morbidity.
During periods of cerebral ischemia, levels of high-energy metabolites such as adenosine triphosphate (ATP) and phosphocreatine decrease within seconds after oxygen supply to the brain is interrupted. 23 The breakdown of ATP and the switch to anaerobic glycolysis leads to an increase in intracellular levels of phosphate and lactate, as well as both intracellular and extracellular acidosis. Failure of ATP-dependent pumps in neuronal cells ultimately leads to a rapid intracellular accumulation of calcium. 24 Calcium sequestration induces mitochondrial dysfunction and activates several intracellular enzyme pathways leading to neuronal depolarization and release of the excitatory neurotransmitter glutamate. Prolonged exposure to glutamate induces a permanent state of hyperexcitability (termed the excitotoxic cascade) which leads to additional injury and cell death. 25
This injurious cascade begins within minutes following the initial injury and may last for many hours or days. The consequences of these pathways include excitotoxicity (as described above), neuroinflammation, apoptosis, free radical production, blood-brain barrier disruption, capillary leak, and seizure activity, among numerous others. 25
The need to maintain cerebral perfusion during periods of circulatory arrest has long been recognized,26,27 and effective cerebral protection remains the principal concern during contemporary aortic arch surgery. 28 Strategies for cerebral protection during aortic surgery with HCA are aimed at (1) limiting the duration of cerebral ischemia, (2) reducing cerebral oxygen consumption (CMRO2), and (3) providing flow of oxygenated blood to the brain during periods of circulatory arrest.
Minimizing Cerebral Ischemic Time
Cerebral ischemic time may be minimized by (1) making the circulatory arrest period as short as possible and (2) minimizing time periods without cerebral perfusion. It is well known that the duration of circulatory arrest is associated with neurologic adverse outcomes. 29 However, there is no consensus about the maximal safe duration of deep hypothermic circulatory arrest (DHCA). 30 Typically, in hemi-arch repair, only the distal anastomosis is done under circulatory arrest. After the distal anastomosis is completed, a clamp may be applied to the proximal portion of the aortic graft, and systemic and cerebral flow are restored while attention is turned to the proximal aortic anastomosis. For a total arch replacement, blood flow to the body may be restored after completion of the distal anastomosis, but full cerebral blood flow can only be restored after the innominate and left common carotid anastomosis are completed, which is done either as an island or by direct reimplantation.
One strategy that has relatively recently emerged to limit cerebral ischemic time in total arch replacement is preoperative debranching of the left subclavian artery, which is accomplished by performing a staged transposition or bypass of the left subclavian artery onto the left common carotid artery several days or weeks prior to aortic arch repair. 31 Carotid-subclavian bypass has traditionally been performed prior to thoracic endovascular aortic repair (TEVAR) in cases where coverage of the takeoff of the LSCA by the endostent is anticipated, 32 but is now increasingly being utilized prior to open total arch procedures so that one less anastomosis needs to be sewn into the arch, thus shortening the cerebral ischemic time and simplifying critical time-sensitive portions of the repair.33,34
Reducing CMRO2: Temperature Management
Hypothermia is an indispensable component of cerebral protection during aortic surgery and has been described in aortic surgery for almost 50 years.
35
A key mechanism by which hypothermia confers cerebral protection is by its reduction of cerebral metabolism, slowing both the utilization of oxygen (i.e., CMRO2) and of glucose, and therefore prolonging the tolerable ischemic period (Figure 7). For every 1°C drop in brain temperature, CMRO2 decreases by approximately 6–7%.
25
Signifying its reduction of cerebral metabolism, hypothermia alone can produce electrocerebral silence (ECS) at low temperatures. Effect of temperature on cerebral metabolic rate and safe duration of HCA. Hypothermia leads to a reduction in CMRO2 in a temperature-dependent fashion (red). Due to the slower metabolic rate and other protective factors initiated by hypothermia (see text for full description), hypothermia increases the allowable “safe” duration of HCA (teal). The data presented here for HCA duration are for HCA without any form of cerebral perfusion. Adapted from McCollough et al.
20
Abbreviations: CMRO2, cerebral metabolic rate of oxygen consumption; HCA, hypothermic circulatory arrest.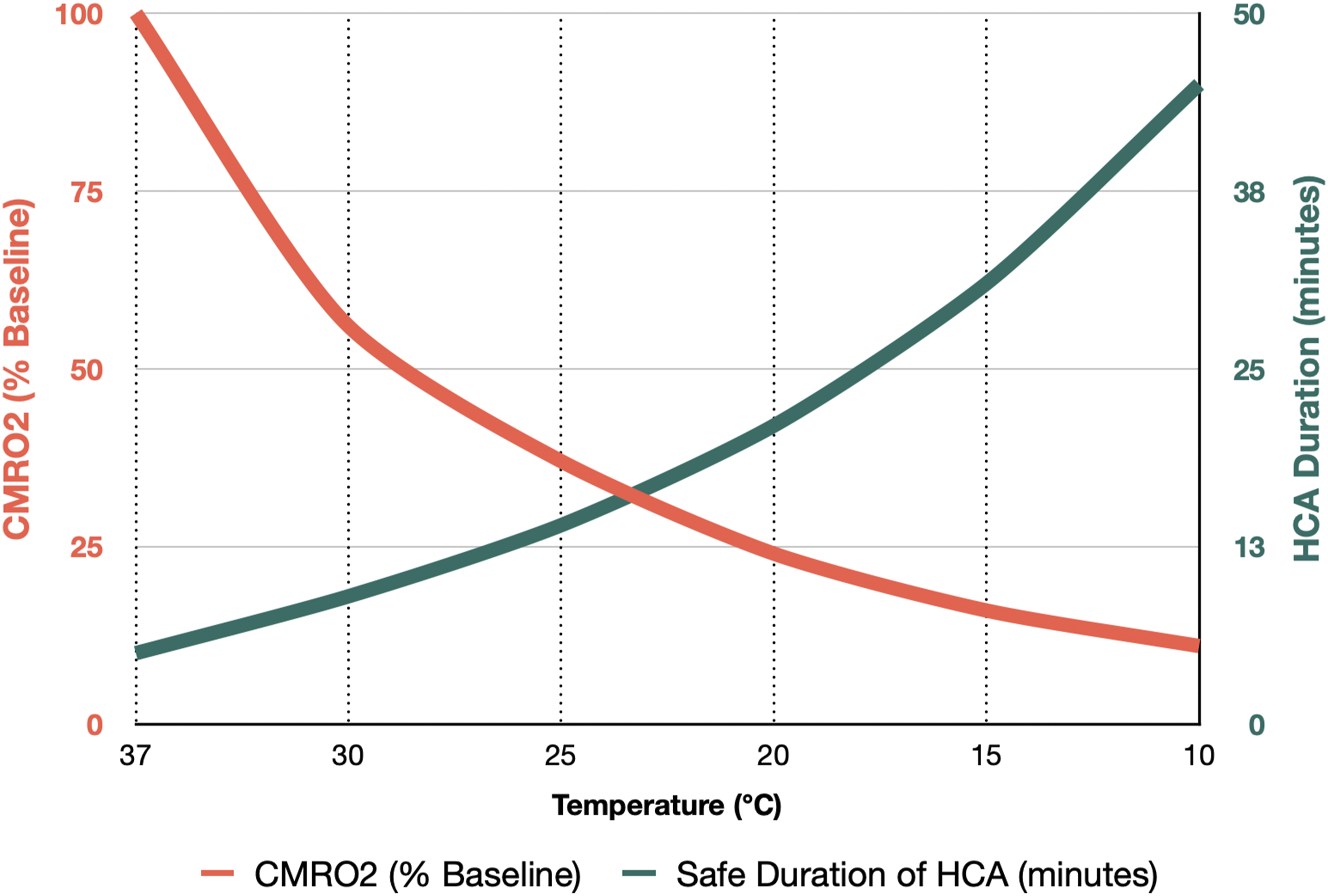
Interestingly, the mechanisms underlying the protective effects of hypothermia in the ischemic brain are quite complex and may actually not be principally related to the decrease in metabolic rate. The harmful cascades leading to intracellular calcium accumulation discussed above are temperature-dependent, and hypothermia can mitigate the involved mechanisms while also stimulating protective systems within the brain.25,36 Animal studies have shown that hypothermia can even prevent various steps in the destructive neuroexcitatory cascades, including calcium influx and the accumulation of glutamate. 37 Ion homeostasis can be significantly improved even with only mild decreases in temperature; conversely, elevated temperatures upregulate these harmful pathways.
However, more profound degrees of hypothermia are associated with a dose-dependent increase in adverse effects in other organ systems, including arrhythmias, vasoconstriction, coagulopathy, impaired wound healing, and electrolyte disorders. 38 Therefore, the selected temperature must balance the protective effects of hypothermia on the brain while minimizing the negative side effects in other organ systems.
Consensus Hypothermia Definitions for Aortic Surgery.

In order to standardize nomenclature regarding the use of hypothermia in aortic surgery, these consensus definitions were developed in 2013 for defining the thresholds of mild, moderate, deep, and profound hypothermia. Adapted from Yan et al. 39
The trend in recent years has been the adoption of warmer temperatures during circulatory arrest, often mild to moderate hypothermia in conjunction with selective ACP. For instance, acceptable neurologic outcomes have been reported by groups using mild to moderate hypothermia,41-43 though the lack of a control group in these studies limits analysis. A retrospective study of 221 patients showed that moderate hypothermia compared with deep hypothermia was associated with lower mortality, shorter CPB duration, and fewer neurologic sequelae. 44 A large network meta-analysis of more than 12,000 patients comparing deep, moderate, and mild levels of hypothermia during aortic surgery showed a lower incidence of stroke in the mild and moderate hypothermia groups compared with deep, and lower mortality in mild vs moderate or deep groups. 45 A prospective multicenter randomized trial of 251 patients undergoing aortic arch surgery evaluated cognitive outcomes and showed that low-moderate hypothermia (20.1–24.0°C) was noninferior to deep hypothermia for global cognitive scores, though in domain-specific analysis, low-moderate hypothermia was associated with lower verbal memory scores. 46 A prospective randomized trial comparing mild vs moderate hypothermia (both with ACP) during hemi-arch repair is currently enrolling patients. 47
Antegrade and Retrograde Selective Cerebral Perfusion
Despite the established beneficial effects offered by hypothermia on the ischemic brain, hypothermia alone can only provide so much protection to the brain. This is evidenced by the limits of “safe” circulatory arrest at various temperatures when hypothermia is used without adjunctive cerebral perfusion techniques, as seen in Figure 7. 20 Therefore, several techniques have been developed to provide for cerebral perfusion during aortic surgery with HCA, including antegrade cerebral perfusion (ACP), and retrograde cerebral perfusion (RCP). Thorough reviews of selective cerebral perfusion techniques have been published elsewhere and the interested reader is encouraged to review these sources.48-50 The optimal approach to cerebral protection during circulatory arrest is not known and remains an evolving field. 51
Retrograde Cerebral Perfusion
In RCP, blood is directed through the superior vena cava in a retrograde fashion to the internal jugular veins and then the brain. This is commonly accomplished with bicaval venous cannulation for CPB. At the time circulatory arrest is initiated, a snare is placed around the superior vena cava (SVC) at the level of the cannula to prevent back bleeding, and then a Y-connector off the CPB arterial cannula is connected to the SVC cannula. The aortic cannula is then clamped and CPB flow is administered solely through the SVC cannula to deliver RCP. Oxygenated blood travels retrograde through the cerebral circulation and re-enters the surgical field through the open innominate and common carotid arteries. Blood is returned to the CPB reservoir via cardiotomy suction placed in the open thoracic aorta, blood suctioned from the surgical field, and via the inferior vena cava cannula. RCP may be applied either continuously or intermittently during circulatory arrest. Some clinicians apply a single instance of RCP very briefly at the end of an HCA period solely for the purpose of flushing embolic debris from the cerebral vasculature.
During RCP, the venous pressure is monitored either by a central venous catheter in the SVC or by a jugular bulb catheter. Rather than targeting a specific flow, RCP flows are titrated to target specific pressures. On the one hand, adequate perfusion pressures are required to generate retrograde flow through the cerebral circulation. However, if venous pressures become too high, it can precipitate the development of cerebral edema. In most centers, the flow is adjusted to maintain a veinous pressure in the range of 15–25 mmHg,52-54 but pressures up to 40 mmHg have been described. 55 Typically these target pressures are achieved with flow rates in the range of 300–500 mL/min. 56 One animal study suggested that driving pressures of 25–35 mmHg provided optimal flow, as assessed by brain tissue blood flow (colored microspheres), regional brain pH (photometry), and vertebral surface blood flow. 57 However, another animal study showed excess cerebral edema at a pressure of 30 mmHg. 58 Pressures exceeding 30–40 mmHg should be avoided due to the potential for inducing cerebral edema.
Monitoring of the efficacy of cerebral perfusion during RCP may be performed with near-infrared spectroscopy (NIRS), transcranial Doppler assessment of the middle cerebral artery, or even central retinal artery flow.55,59
Clinical outcome studies comparing RCP and DHCA have yielded mixed results. Retrograde cerebral perfusion has been found to be associated with comparable mortality rates by some investigators,60-62 while others have shown reduced neurologic morbidity, mortality, and extension of the allowable duration of circulatory arrest.63-68 Based on the capillary flow data in animal studies, it is unlikely that RCP provides sufficient perfusion to completely support cerebral metabolism. 49 Both clinical and animal data suggest that RCP provides between 20% and 60% of baseline cerebral blood flow when administered through the superior vena cava (SVC) at jugular pressures of 20 to 25 mmHg.69-71 While higher flows may be achievable, the potential for inducing cerebral edema limits the deliverable flow rate.
Advantages of RCP include that it provides bilateral perfusion of the brain, can flush out emboli due to retrograde flow, and it does not require axillary cannulation, therefore decreasing operative time and complexity. 72 Disadvantages include that RCP can lead to cerebral edema and it provides less cerebral flow compared to ACP, likely providing incomplete cerebral metabolic support and inadequate neuroprotection.
Antegrade Cerebral Perfusion
To set up cannulation for administration of ACP, a Dacron graft may be sewn in an end-to-side fashion onto either the right axillary or innominate artery, and the arterial CPB outflow connected to the graft using a tubing connector. In some cases, the right common carotid artery may be preferred. This cannula can be used for full CPB to deliver oxygenated blood to the brain and the body. At the time circulatory arrest is initiated, a clamp or tourniquet is applied to the base of the innominate artery, so that all arterial outflow is directed to the right cerebral circulation. Simultaneously, the CPB flows are dropped to lower rates since only the cerebral circulation needs to be supported. This is the preferred approach at the authors’ institution. Alternatively, instead of using a graft, the chosen artery may be directly cannulated with the arterial CPB cannula, while ensuring that the cannula is directed towards the right common carotid artery. ACP may also be established, regardless of the initially chosen cannulation site, by balloon-occludable perfusion catheters placed in the innominate artery and/or LCCA. The addition of ACP changes the concept of HCA from total body circulatory arrest to only lower body circulatory arrest, as it provides perfusion of the brain, upper extremities, and spinal cord (anterior and posterior spinal cord arteries via the vertebral artery).
During HCA with ACP, most centers perfuse the brain at a rate of 10–15 mL/min/kg body weight, which correlates to approximately 0.6 mL/min/g of cerebral tissue. 73 It has been shown that flows of 8–10 mL/min/kg are sufficient to restore the regional oxygen saturation (rSO2) during HCA to awake baseline levels. 74 Studies examining various flow rates during ACP have demonstrated that high flow rates significantly increase cerebral blood flow and the intracranial pressure, resulting in cerebral edema with no cerebral metabolic benefit. 75 However, higher flow rates may be needed in neonates and infants. 76
The approach described above provides unilateral ACP, as cerebral perfusion occurs only through the right common carotid and right vertebral arteries. It is possible to perform bilateral ACP by placement of a balloon-occludable perfusion cannula into the left common carotid artery once the aortic arch has been opened. This requires adding an additional Y-connector on the arterial limb of the CPB circuit. However, despite the potential advantage of bilateral cerebral perfusion, the extra manipulation of the aortic arch vessels required for placement may increase the risk for embolic stroke; the advantage of unilateral ACP is that there is less manipulation of arch vessels. The key question is whether contralateral cerebral perfusion occurs to a sufficient degree when only unilateral ACP is utilized.
Important to this discussion is that a significant number of patients do not have a fully intact circle of Willis (CoW). For example, anatomic studies have shown that approximately 15–20% of the general population does not have a complete CoW and that anatomic variants are exceedingly common, especially involving the posterior cerebral circulation.77-79 However, anatomic studies may be limited in that they underestimate the importance of collateral vessels, such as the ophthalmic artery, leptomeningeal vessels, and external carotid arteries. 48 These collateral vessels may augment contralateral cerebral perfusion during ACP, even when the CoW is incomplete. Illustrating the point that there may be poor correlation between anatomic studies and clinical outcomes, in one review of 99 patients undergoing arch surgery with unilateral ACP, preoperative CT angiography showed that 40% of patients had abnormal CoW, but intraoperative monitoring indicated good cross-perfusion in all patients. 80 In this cohort, only one patient had a minor embolic stroke.
In contrast with anatomic assessment of the CoW, functional assessment (i.e., assessing flow during ACP) may be superior. However, when functional assessment of the CoW was performed using transcranial Doppler in one cohort, only 27% of patients had a functionally fully intact Circle of Willis. 81 These authors classified different functional variants as being “safe,” “moderately safe,” and “unsafe” for unilateral ACP and found that 28% of patients had cerebral vascular anatomy that was either unsafe or only moderately safe for unilateral ACP.
The optimal approach for ACP—either unilateral or bilateral—is still debated, and additional high-quality evidence is still needed before definitive conclusions can be reached.82,83 For example, a meta-analysis of over 6000 patients undergoing aortic arch surgery comparing unilateral ACP vs bilateral ACP showed no significant difference in mortality or permanent neurologic dysfunction. 84 Some authors have found that bilateral ACP confers a benefit over unilateral ACP only when HCA duration exceeds 40–50 minutes. 85 Other groups advocate that bilateral ACP should be the standard approach in all aortic surgery as it provides more physiologic perfusion.86,87 Yet other groups have found that bilateral ACP is associated with worse neurologic outcomes when compared with unilateral ACP. 42 Unfortunately, much of the data in this area is retrospective and a randomized trial would be beneficial.
Given that it is not possible or practical to perform preoperative cerebral imaging on all patients undergoing aortic repair, and given the significant heterogeneity in cerebral vascular anatomy, the routine practice at the authors’ institution is to use moderate hypothermia with bilateral ACP in cases where HCA is required. Left common carotid arterial perfusion is provided by adding a balloon-occludable perfusion catheter along with an extra Y-connector on the arterial CPB limb.
ACP vs RCP
Numerous studies have compared ACP vs RCP in aortic surgery and evidence is conflicting. 30 Compared with DHCA alone, both antegrade and retrograde cerebral perfusion techniques appear to offer similar benefits and reduce neurologic morbidity and extend the safe duration of HCA.22,88,89 However, a large meta-analyses comparing ACP with RCP have not shown any significant difference in stroke, mortality, or transient neurological dysfunction between the 2 approaches.89,90
In most aortic centers, ACP (whether unilateral or bilateral) has emerged as the technique of choice for cerebral perfusion during HCA. This is in part due to evidence that RCP may not fully meet cerebral metabolic demands, as well as that ACP provides more physiologic antegrade flow to the brain.
Anesthetic Management
Given the lack of high-quality data regarding optimal anesthetic management for proximal aortic surgery, significant inter-institutional variation in approaches exists, regarding monitoring techniques, management during HCA, and other aspects. This manuscript aims to describe some of the available strategies and their evidence or rationale. Additionally, since there is significant overlap in the anesthetic considerations for proximal aortic repair with a standard CPB case, this article will focus on aspects more specific to proximal aortic repair with HCA and only briefly discuss general CPB considerations.
Prior to arrival to the operating room, blood product availability should be confirmed and a perfusion team with a primed pump should be present due to the risk of hemodynamic collapse. The main focus prior to surgical repair is on maintaining normal hemodynamics with an emphasis on avoiding increases in blood pressure that could lead to aortic rupture. Preinduction, at a minimum, the patient must have large-bore venous access and invasive blood pressure monitoring. Pressors and inotropes should be in line in case of aortic rupture and allogenic blood should be available.
Lines and Monitoring
Invasive arterial blood pressure monitoring is mandatory; however, the location(s) utilized vary between centers. 51 Each arterial site has specific considerations, which are presented here, and many advocate for arterial monitoring at more than one site. 91 Additional consideration should be made if the preoperative CT shows dissection flap involving any of the aortic arch vessels, since the dissection flap may impact arterial readings in the more distal portions of the affected branch vessels.
Arterial monitoring of the left upper extremity (at the radial, brachial, or axillary arteries) has the advantage of uninterrupted readings during clamping of the innominate or right axillary artery for CPB cannulation. An arterial catheter in right upper extremity will lose its readings during this period, but has the advantage of measuring cerebral perfusion pressure during HCA with ACP, which some centers utilize in their management of HCA. 73 However, during full CPB with innominate or right axillary outflow, right radial arterial pressure reading will be impacted. If an end-to-side graft is used, then right radial pressure measurements may overestimate systemic perfusion pressures due to its close proximity to CPB arterial outflow.91,92 If the right axillary artery is directly cannulated, the right radial artery no longer provides an accurate assessment of cerebral perfusion pressures due to occlusion of the arterial lumen by the cannula. Being further away from the arterial cannulation site, left upper extremity and femoral pressure readings may better reflect systemic perfusion pressures during CPB when the innominate or right axillary artery is cannulated. Femoral arterial pressure readings may better approximate central aortic pressures compared with radial arterial measures, especially after CPB.93-95
At the authors’ institution, the customary approach for hemi-arch repair is for using a combination of left radial and femoral arterial pressures. For total arch repair, bilateral radial arterial lines and a femoral arterial line are placed. If the patient has preoperatively undergone LCCA transposition or bypass, it is common for left radial pressures to be 10–20 points lower than right radial measures.
Large-bore central venous access is mandatory for the administration of fluids, vasopressors, and inotropic agents. There is limited evidence of benefit for the use of PA catheters in aortic surgery with circulatory arrest, 96 but many groups, including the authors of this manuscript, routinely employ PA catheters, as the additional hemodynamic data are felt to be beneficial in the intraoperative and postoperative management of these patients. 97
Cerebral Monitoring
Due to the high risk of neurological sequelae during and after HCA, neurologic monitoring is an essential component of anesthetic monitoring. Cerebral monitoring is usually accomplished using near-infrared spectroscopy (NIRS), transcranial Doppler (TCD), or electroencephalography (EEG).
NIRS allows for noninvasive assessment of the rSO2 in the frontal cortex by measuring the difference in absorption of near-infrared light by hemoglobin and oxyhemoglobin.98,99 For this modality to be the most useful and to establish a true baseline, the NIRS monitor should be applied prior to pre-oxygenation & induction, while the patient is on room air. Average baseline values range 50–75%, though thresholds for normal and abnormal values of cerebral rSO2 are poorly defined.100-102
Advantages and Disadvantages of NIRS for Cerebral Monitoring During Hypothermic Circulatory Arrest.
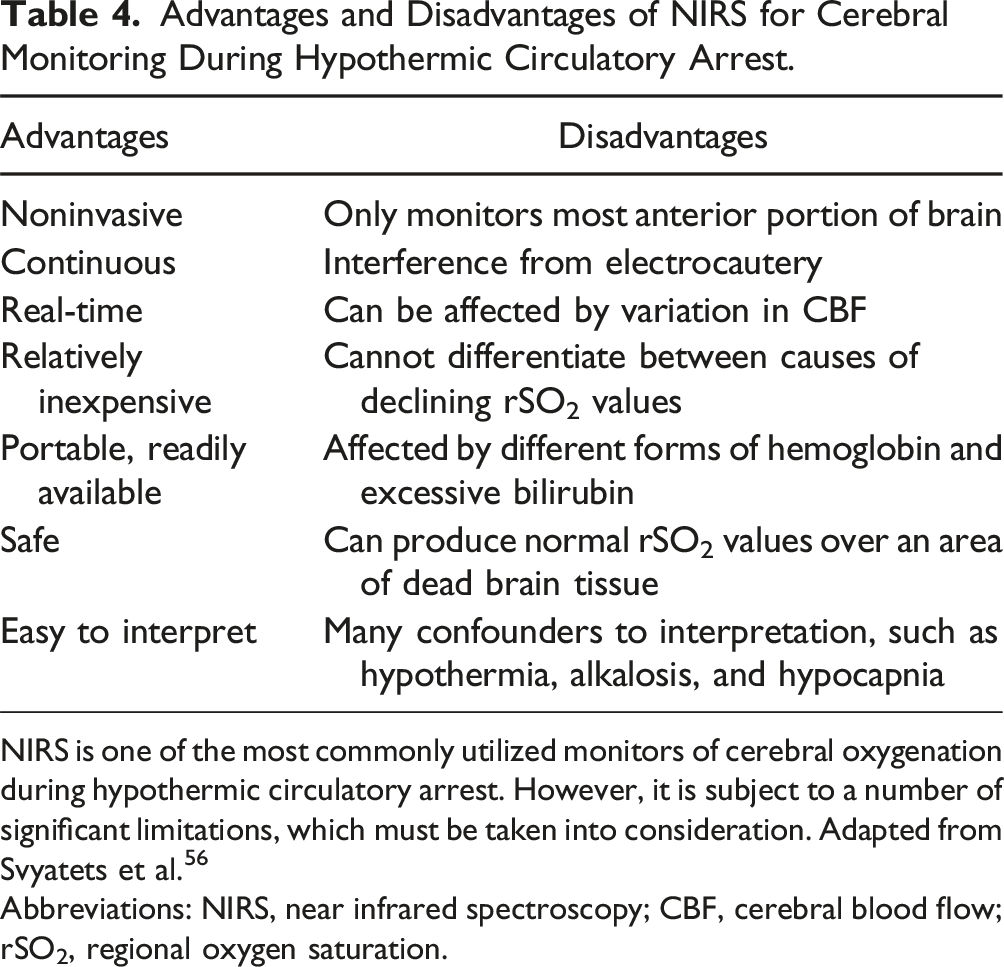
NIRS is one of the most commonly utilized monitors of cerebral oxygenation during hypothermic circulatory arrest. However, it is subject to a number of significant limitations, which must be taken into consideration. Adapted from Svyatets et al. 56
Abbreviations: NIRS, near infrared spectroscopy; CBF, cerebral blood flow; rSO2, regional oxygen saturation.
Despite these limitations, NIRS remains one of the most commonly utilized modalities for monitoring cerebral oxygenation during HCA.51,73,105,106 One 2015 survey of several hundred centers in Europe indicated that 2 thirds of centers routinely employ NIRS. 73 This is because, in part, the application of NIRS is much more simple than TCD or a full EEG. Additionally, NIRS can detect global hypoxia with reasonable fidelity. The most important time periods during which NIRS values should be actively followed are during innominate arterial clamping and during HCA; in fact, during HCA, it is the only vital sign of any import. Though strict thresholds have not been developed or validated, 107 NIRS values below ∼50% (or a drop of more than 20% from the baseline) should warrant concern and prompt an assessment for inadequate cerebral perfusion, and NIRS values below ∼40% should be considered critically low. 105
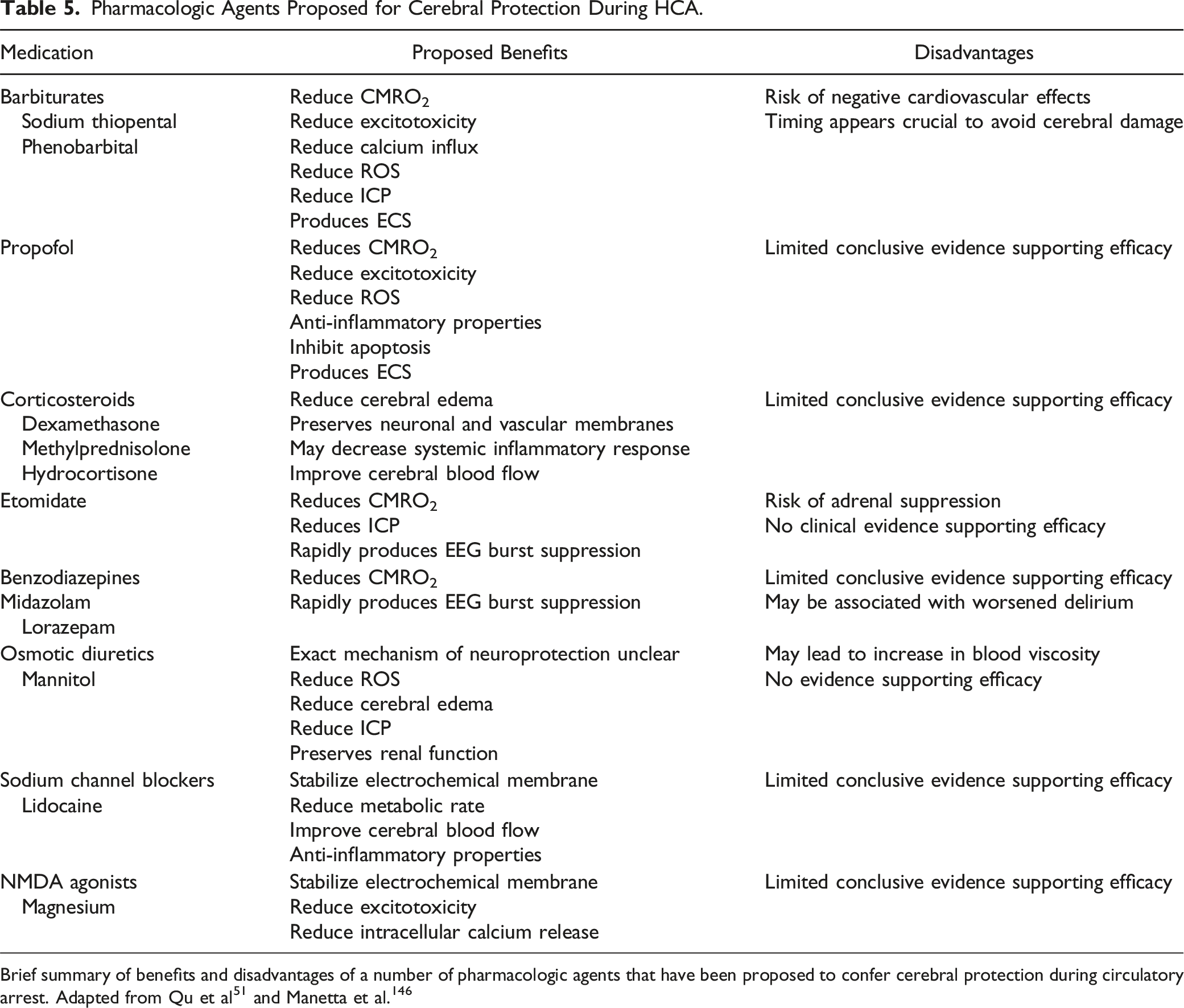
Pharmacologic Agents Proposed for Cerebral Protection During HCA.
EEG provides direct assessment of cerebral electrical activity, from which the state of the brain may be surmised. In aortic surgery, EEG may be used to assess for ECS, a state indicative of minimal cerebral metabolic demand, prior to the initiation of circulatory arrest.113-115 EEG may also detect abnormal recovery or seizure activity after HCA and during rewarming, 116 which are associated with neurologic morbidity. However, full EEG monitoring during proximal aortic surgery is uncommon due to the complexity of monitoring and additional logistic challenges, such as the need for personnel with specialized training to interpret EEGs. More commonly, processed EEG such as the bispectral index (BIS) or patient state index (Sedline) are utilized to assess for ECS prior to and during HCA. The “simplified” monitors used by processed EEG are easier to apply than a full EEG array, but processed EEG monitors are prone to a number of artifacts common in patients undergoing cardiac surgery. 117
Transesophageal Echocardiography (TEE)
Transesophageal echocardiography (TEE) provides high-resolution images of most of the thoracic aorta, apart from a short segment of the distal ascending aorta just proximal to the innominate artery. This “blind spot” is attributable to acoustic shadowing from the trachea or left mainstem bronchus. 118 TEE should be utilized for all patients who undergo proximal aortic repair, barring any contraindications.119-121 In a propensity-matched retrospective analysis of over 870,000 patients in the STS adult cardiac surgical database who underwent cardiac valve or proximal aortic surgery, the use of TEE was found to be associated with improved 30-day mortality. 122
Prior to CPB, TEE can be used to assess the extent of aortic disease and help with operative planning (Figure 8).123,124 In particular, focus should be paid to the extent of aneurysmal disease or dissection, biventricular function, the presence of regional wall motion abnormalities, and the status of the aortic valve, which may frequently be affected by proximal aortic aneurysmal disease or by dissection, leading to aortic valvular insufficiency (AI). In patients with type A dissection, TEE is particularly helpful for confirming the diagnosis, assessment of the true and false lumens, and in evaluation of associated complications, such as acute AI, pericardial effusion, tamponade, and/or coronary involvement. Transesophageal assessment of the ascending aorta. Transesophageal echocardiography plays a critical role in the evaluation of the aortic valve, aortic root, ascending aorta, and aortic arch in patients undergoing proximal aortic repair. A variety of pathologies can be seen. (A) Midesophageal log axis view showing aortic root aneurysm (6.5 cm). (B) Midesophageal long-axis view showing acute type A dissection. Dissection flap can be visualized in the ascending aorta, originating near the sinotubular junction. (C) Upper esophageal ascending aortic short axis view with simultaneous biplane imaging showing a long-axis view. This image shows an intramural hematoma causing near-complete obstruction of the proximal aorta. (D) Midesophageal long-axis view in a patient with aortic root aneurysm showing severe aortic valve insufficiency.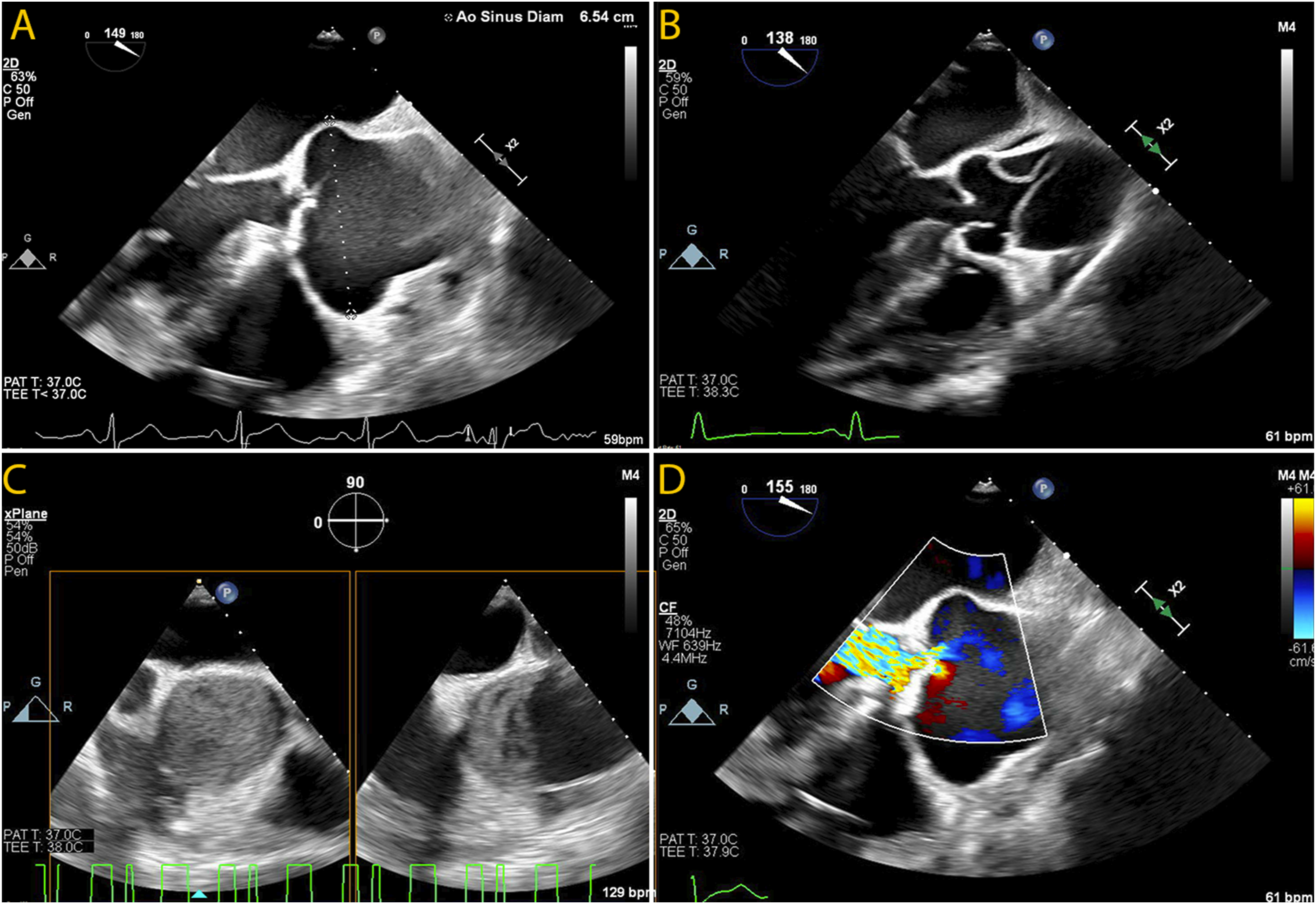
A particularly thorough assessment of the aortic valve is warranted, and more in-depth reviews can be found elsewhere.125-127 Functionally, the aortic annulus, cusps, and the STJ all contribute to the valvular mechanism of AI. Each component should be evaluated when assessing the mechanism of AI, especially when a surgical aortic valve repair is being considered. For surgical correction to be effective, both cusp and root abnormalities must be evaluated and corrected. 125 Aortic insufficiency should be classified using TEE with a repair-oriented functional classification, emphasizing the contribution of different components of the aortic root. Categorization schemes for the aortic valve analogous to the Carpentier classification system for mitral regurgitation have been proposed, by grouping pathologies with normal leaflet motion, excessive leaflet motion, and restrictive leaflet motion together. 128 For assessing AI severity, guidelines from the American Society of Echocardiography outline the approaches for assessing the severity. 129
Additional echocardiographic measures that are helpful in assessing the aortic valve, in particular with reference to achieving a successful and durable aortic valve repair, include the coaptation height (cH) and the effective height (eH). 126 The cH (mean: 4–5 mm) is the length of apposition of 2 cusps in diastole. The eH (mean: 8–10 mm) 130 is the orthogonal distance from the aortic annulus to the middle of the free margin of the cusp. In a tricuspid aortic valve, each leaflet may have its own eH and each leaflet pair its own cH. The use of three-dimensional echocardiographic datasets with multiplanar reconstruction can be helpful in measuring these values for each cusp.126,131 The intraoperative post-repair assessment of eH is associated with improved long-term survival and freedom from AI. 132 An eH less than 11 mm after valve-sparing aortic root replacement is associated with increased likelihood of at least moderate AI. 133 Similarly, aortic valve repair with resultant cH less than 4 mm are more likely to fail and have recurrent AI. 134
Induction and Intubation
The goal of induction is to prevent the sympathetic response to airway instrumentation. Hypertension and tachycardia increase shear stress on the fragile aortic wall which can cause progression of aortic dissection or, in the worst-case scenario, aortic rupture. For the hemodynamically stable patient, preferred induction agents include midazolam, fentanyl, lidocaine, propofol, esmolol, and rocuronium, though any agents that produce an adequately anesthetized patient, adequate intubating conditions, and avoidance of hemodynamic perturbations above may be used. For the hemodynamically unstable patient, lidocaine, etomidate, rocuronium, and pressor boluses can be used to maintain a proper mean arterial pressure. Endotracheal intubation should be achieved with a single lumen tube.
Hemodynamic Management
As mentioned above, the hemodynamic goals during the beginning phase of the procedure prior to institution of CPB center primarily around heart rate and blood pressure control, in order to maximally reduce aortic wall shear stress (WSS). 135 The goal is to provide enough oxygen delivery and perfusion pressure to avoid end-organ dysfunction, while preventing hypertension, tachycardia, or any hemodynamic conditions that impose additional stress on the aorta. Additional considerations may be warranted in specific pathologies (e.g., dissection with coronary involvement, tamponade, acute AI, etc.).
WSS is defined as the shear force produced by tangential blood flow on the vessel wall as a result of blood viscosity and is related to the gradient of velocity in the surface normal direction.
136
Essentially, WSS can be thought of as the product of the dynamic viscosity of the fluid (η) and the velocity gradient near the wall (
Strategies to minimize WSS include tight regulation of heart rate (often with β-blockers) and blood pressure (often with anesthetic agents and/or arterial vasodilators). Permissive hypotension or the minimum blood pressure able to support end-organ perfusion can be employed until the surgeon gains access to the aorta. At the author’s institution, during this period, we are much more comfortable with hypotension rather than hypertension (provided that hypotension is not due to aortic rupture).
The one exception to this general rule during the pre-CPB phase is during the period when the arterial graft is being sewn onto the innominate artery. To sew the graft onto the artery, the surgeon must apply a side-biting cross-clamp, which frequently completely occludes the innominate artery due to the artery’s relatively small size. Therefore, there is the potential for decreasing cerebral perfusion. Frequently, a small amount of heparin (3000–5000 units) may be given prior to innominate cross-clamp placement. During the time when the innominate artery is clamped, the perfusion pressure should typically be increased, since the completeness of the patient’s CoW is usually not known, and a significant number of patients do not have an intact CoW.79,81 However, excessive hypertension during this period can lead to disastrous consequences in the setting of a weakened and/or dissected aorta, including aortic rupture. Hemodynamic management during this period should be guided by a measure of cerebral perfusion (NIRS is used at the authors’ institution) and by the severity of the patient’s aortic disease. If the axillary artery is to be cannulated, then the above hemodynamic considerations do not apply, since the placement of a cross-clamp on the axillary artery does not significantly impair cerebral perfusion.
Following separation from CPB, strict hemodynamic control remains important. Hypertension (systolic blood pressure greater than 130–140 mmHg) should be avoided as it will stress fresh suture lines and may be associated with worsened bleeding. Additionally, some patients may have residual dissection in the descending aorta, which may be worsened by hypertension. However, blood pressure should be kept sufficiently high to ensure adequate coronary and end-organ perfusion.
If a frozen elephant trunk is performed as a part of total arch repair, the stent graft inevitably covers some intercostal branches of the aorta, and the risk of spinal cord injury ranges from approximately 1–9%. 137 In such cases, higher blood pressures (e.g., mean arterial blood pressure 90–100 mmHg) are targeted to ensure spinal cord perfusion.
Management of Cardiopulmonary Bypass
Standard practices for management of CPB should be employed, including with regards to anticoagulation, 138 conduct during CPB,139,140 temperature management, 141 and patient blood management. 142
One special scenario will frequently occur during cooling, depending on whether the surgeon is able to place an aortic cross-clamp on the distal portion of the aorta. In the event that the surgeon is able to place a cross-clamp, this may be done, followed by the administration of cardioplegia (typically through a combination of antegrade, retrograde, or direct ostial, depending on the degree of AI) and arrest of the heart. The surgeon may then be able to inspect the aortic valve or even begin portions of more proximal repair while still cooling the patient down to an adequate temperature for HCA.
However, due to the patient’s anatomy, it may not be able to place an aortic cross-clamp. In that case, the patient is cooled down to the desired temperature and then circulatory arrest is initiated. During cooling to moderate or deep levels of hypothermia, the heart will spontaneously fibrillate. 143 Ventricular fibrillation, even during full CPB support, leads to several deleterious consequences. First, oxygen utilization of the heart is increased. 144 Second, since the left ventricle is not ejecting in a coordinated fashion, it results in ventricular distention, which can impair coronary perfusion pressure and compromise subendocardial perfusion. 144 As such, it is common practice at the authors’ institution to administer anti-arrhythmic agents during cooling to try to delay the onset of ventricular fibrillation, despite limited evidence for the practice. These agents include lidocaine (1–1.5 mg/kg), amiodarone (150 mg), magnesium (2 grams), and a β-blocker (e.g., esmolol, 0.5–1 mg/kg). In addition, the surgeon may place a vent in the left ventricle, through the pulmonary vein, to minimize left ventricular distention.
If the patient is on any vasopressors during CPB to maintain perfusion pressure, these should be turned off prior to the institution of HCA, assuming ACP is utilized. The goal of ACP is to maximize cerebral perfusions and continuation of vasoconstrictors would be counterproductive to that goal.
Cerebral Protection During Circulatory Arrest
Pharmacologic Protection
Numerous drugs have been used in attempt to confer protection to the brain and spinal cord during HCA, and given the limited quantity and quality of evidence, significant inter-institutional variation exists.73,124 Many centers routinely administer pharmacologic agents for cerebral protection during HCA, despite the fact that their efficacy remains unproven.73,120,145-147 Table 5 presents a summary of various medications that have been proposed for cerebral neuroprotection. The most commonly used medications include barbiturates and propofol (to produce ECS and reduce CMRO2), mannitol (to prevent swelling and scavenge oxide free radicals), and corticosteroids (to stabilize neuronal membranes).
Barbiturates such as sodium thiopental were initially considered to be the standard neuroprotective drug due to their ability to lower CMRO2, reduce cerebral blood flow, and reduce intracranial pressure. Preclinical research has shown that barbiturates may be associated with inhibition of apoptosis and improved neuronal protection during hypoxemia. 148 However, clinical studies have not shown a benefit, though most are retrospective and have limited quality. Other authors, in their review, concluded that thiopental is beneficial, but if given too early before HCA may be associated with worse neurologic outcomes. 149
A registry review of over 2000 patients from 2013 failed to show any benefit of barbiturates or mannitol in reducing mortality-corrected new permanent neurologic dysfunction, though these authors did find that steroids were associated with a statistically significant improvement. 150 These authors were unable to demonstrate a significant protective effect of any neuroprotective drug on 30-day mortality, or rates of neurologic dysfunction following prolonged (≥30 min) cerebral ischemia. In addition, steroids also have the potential to worsen hyperglycemia, which may lead to worsened neurologic outcomes following cerebral ischemia.
Other agents may be administered empirically based on theoretical benefit, though high-quality evidence in humans is lacking. One example is lidocaine. In preclinical studies lidocaine at a dose of 4 mg/kg prior to HCA (plus an additional 2 mg/kg at reperfusion) has been shown to reduce CMRO2 by blocking sodium channels, essentially mimicking the effect of hypothermia. 151 Clinical studies in humans have had mixed results. In one study, a. lidocaine infusion of 4 mg/min throughout CPB and HCA showed improved short-term neurologic outcomes, 152 while in another study, authors found worsened outcomes in some patient populations, including diabetics. 153 Magnesium, which blocks voltage-sensitive calcium channels and N-methyl D-aspartate-activated calcium channels, leading to reduced intracellular calcium, has been show to confer neuroprotective benefit in preclinical studies, 154 though evidence in cardiac surgery has been mixed.155,156 Dexmedetomidine has been shown in animal studies to be neuroprotective in both focal and global cerebral ischemia, 157 but no studies in humans have been performed assessing its neuroprotective effect in humans undergoing aortic repair with HCA.
Topical Cooling: Ice on the Head
Another area which is debated is the use of topical cooling by placing ice packs on and around the patient’s head during HCA.158-160 Although many centers use this routinely, many do not, thus highlighting the clinical equipoise as to the utility of this technique.
Proponents of the technique point out that its significant advantages include that it is not confined by the cerebral circulation (a significant limitation of using hypothermic arterial CPB outflow to cool the brain, especially in patients with cerebrovascular disease); its use does not carry the embolic or hyper-perfusion risks; it is an inexpensive intervention; and it can promote focal cooling of those body regions more likely to realize benefit from colder temperatures. 158 In one animal study conducted under DHCA (without any cerebral perfusion), the use of topical cooling was found to be associated with a significant decrease in brain temperature. 161 Another animal study found that in patients in whom topical cooling was used, the recovery of CMRO2 to baseline was 50% more rapid when topical cooling was used, reflecting less cerebral metabolic debt accumulation during the arrest. 162
Others argue that even animal studies have shown mixed results. 163 Given the mixed evidence for benefit, the potential for interference with cooling techniques on important monitoring modalities such as NIRS or EEG, and possible risks of topical cooling (such as ischemic retinopathy or frostbite), the routine use of topical cooling is not warranted. 159 These authors also argue that the use of ACP even further reduces any potential benefit of topical cooling, since cerebral perfusion is maintained.
Glycemic Management
Although evidence in patients undergoing aortic repair with HCA is lacking, there is very clear evidence that hyperglycemia is associated with worsened neurologic outcomes in patients with other forms of cerebral injury. This is likely because hyperglycemia exaggerates numerous damaging processes, including intracellular acidosis, the accumulation of extracellular glutamate, cerebral edema formation, and blood-brain barrier disruption. 164
Even non-diabetic patients are prone to hyperglycemia during the intraoperative period due to the upregulation of counter-regulatory hormones like glucagon, decreased insulin production, and increased insulin resistance that occur as part of the neuro-hormonal stress response to surgery. 165 Guidelines from the STS recommend targeting blood glucose levels less than 180 mg/dL in cardiac surgical patients. 166 Separately, guidelines for nonsurgical patients with ischemic stroke recommend maintaining serum glucose between 140–180 mg/dL, because hyperglycemia in acute stroke is associated with worse outcomes. 167 Given that evidence in aortic surgical patients is lacking, and given the potential pathophysiological overlap in patients with ischemic stroke, it is reasonable to target similar serum glucose levels throughout the perioperative period.
Blood & Fluid Management
Acute normovolemic hemodilution (ANH) is one strategy that has been shown to minimize blood transfusion requirements in aortic surgery. 168 It involves removing some of the patient’s own whole blood at the beginning of the case, replacing the blood volume with colloid or crystalloid and then transfusing the whole blood after CPB and protamine has been administered.
In addition to potential transfusion-sparing effects of ANH, the authors routinely employ this technique as a part of managing hematocrit during HCA. Blood viscosity, the primary determinant of which is hematocrit, increases at lower temperatures.169,170 For example, when temperature is decreased from 37°C to 22°C, viscosity increases by 26% from the baseline.
171
As seen from the Hagen–Poiseuille formula, viscosity also plays an important role in vascular resistance (eq. (2)).
In this equation, R is resistance, η is viscosity, L is the circuitry length, and r is the radius of the tubing. Though this equation is only intended for estimating the resistance of Newtonian fluids in straight tubing, the impact of viscosity can still be seen. Therefore, at higher viscosities, cerebral microcirculatory blood flow may be diminished not only due a delay in the passage of red blood cells and increased microcirculatory resistance.170,172,173 Other studies have shown that optimal microcirculatory perfusion occurs at a hematocrit of approximately 30% at normothermia. 174 Therefore, due to the theoretical benefits outlined above, the aim of this approach is to reduce the hematocrit to improve cerebral microcirculatory perfusion, even though there is little direct clinical evidence supporting this strategy. It remains unknown what the optimal hematocrit during moderate HCA or DHCA is.
Following separation from CPB, a principal concern is the assessment and correction of coagulopathy. Due to lower nadir temperatures with proximal aortic surgery compared with standard cardiac procedures, greater degrees of platelet dysfunction, coagulation factor depletion, fibrinogen depletion, and increased fibrinolysis should be anticipated.175,176 In addition, patients who have aortic dissection frequently have preoperative derangements in coagulation due to exposure of tissue factor within the false lumen of the aorta, which must be accounted for.177,178
In order to assess coagulopathy, point of care viscoelastic tests such as rotational thromboelastometry (ROTEM) or thromboelastography (TEG) should be utilized to guide transfusion and administration of factor concentrates. 124 The use of viscoelastic studies has been associated with reduced rates of transfusion, cost, and rates of adverse events including rates of surgical re-exploration in aortic surgery.179,180 Though little clinical data are available to drive transfusion decision making in aortic surgery, the optimal approach likely includes the use of viscoelastic testing, aggressive goal-directed replacement of fibrinogen, platelets, and factor deficiencies, the use of antifibrinolytic agents, and the use of factor concentrates to augment blood component therapy. 124
In addition to bleeding and transfusion, ongoing assessment of volume status is critical after separation from CPB. Dynamic intravascular fluid shifts occur after separation from CPB due to rewarming-associated vasodilation and inflammation-associated capillary leak. 181 Rewarming does not occur homogenously, and after separation from CPB, there is ongoing equilibration of core and peripheral temperatures, with the result of ongoing peripheral vasodilation as heat redistributes from the core to the periphery. Volume status should be assessed and reassessed in a reiterative fashion, using TEE and the invasive hemodynamic monitors which are routinely applied in aortic surgery.
Weaning From CPB and Post-CPB Management
Following completion of the repair, the patient is warmed on CPB. Rewarming should occur gradually—typically 0.5°C per minute or less. 141 This practice helps reduce the formation of gaseous microemboli, and reduces the likelihood of cerebral hyperthermia, which is unequivocally harmful for the brain. 182 Slower rates of rewarming are associated with improved neurocognitive outcomes. 183 In addition, it should be noted that CPB arterial outflow temperature may underestimate cerebral temperatures,184,185 and this should be taken into consideration in avoiding cerebral hyperthermia.
Following release of the aortic cross-clamp, the heart should be given an adequate duration to rest before attempting to wean from CPB. Frequently, one or more inotropic agents may be required. Right ventricular dysfunction is common due to the long CPB duration, cold temperatures utilized, and use of retrograde cardioplegia, which provides suboptimal right ventricular protection. More complete reviews of management of weaning from CPB have been presented elsewhere and the interested reader is encouraged to review those sources.186,187
Following separation from CPB, the heart and aorta are assessed with TEE. In particular, attention should be paid to evaluating biventricular function for global and regional wall motion, especially if a root replacement, which requires reimplantation of coronary buttons, is performed. In addition, TEE is valuable in assessing the aortic valve and the visible portions of the aorta to check the quality of the repair assess for any residual AI. Assuming that cardiac and valvular function are adequate, protamine is administered slowly to reverse heparin. As mentioned above, blood components are also frequently required to treat coagulopathy.
Postoperative Care
Postoperative Complications
While proximal aortic repair is potentially life-saving, there may be significant perioperative morbidity. Common early complications following proximal aortic repair include hypothermia, coagulopathy, delirium, respiratory failure, cardiovascular instability, metabolic disturbances, renal failure, and stroke. Perioperative mortality varies depending on the extent and circumstances of the procedure. For example, the overall mortality for aortic valve, aortic root, and ascending aortic repair is approximately 1–5%. 120 Mortality is slightly higher for total aortic arch repair, with estimates ranging 2–6%. 120 For acute type A dissection, a review of the STS database found mortality is as high as 17%. 188
Myocardial ischemia occurs in approximately 6% of patients who underwent root replacment. 189 The incidence of clinically overt stroke is approximately 3.5–5%, but may be as high as 13% for type A dissection. 190 Visceral ischemia may occur during circulatory arrest, but with the use of moderate or deep hypothermia, is fortunately well-tolerated. Vocal cord dysfunction may occur postoperatively, especially following operations on the distal aortic arch. It arises due to injury of the recurrent laryngeal nerve, either due to stretch from distal arch aneurysmal disease or due to injury during total arch repair. 120
Postoperative ICU Management
In general, much of the postoperative management following proximal aortic repair mirrors that of other cardiac surgical procedures performed with CPB, and this topic has been extensively reviewed elsewhere.191,192
However, special focus should be paid to several aspects. Given the neurologic risks of HCA, it is prudent to obtain a neurologic exam as soon as possible, even if for a brief assessment while the patient remains intubated. In the early postoperative period, patients may require additional rewarming, management of sedation and analgesia, fluid resuscitation, titration of vasopressors and inotropes, management and weaning of mechanical ventilation, ongoing assessment of kidney function and urine output, correction of coagulopathy and management of bleeding, and correction of metabolic derangements. Specifically in patients with acute aortic dissection, an ongoing assessment for malperfusion should occur. Patients with malperfusion syndrome may have mortality as high as 25–42%, depending on the number of affected organ systems.193-195 Additional interventions may be required to restore perfusion.
Later in the postoperative course, blood pressure management is exceedingly important, specifically with regards to heart rate and anti-impulse control. The use of beta-adrenergic blockers, which reduce heart rate, blood pressure, and aortic wall stress, is associated with reduced long-term mortality.196,197 Patients also require routine postoperative surveillance with CT, as aortic events are a major cause of late morbidity and mortality.
Footnotes
Acknowledgments
The authors would like to express their gratitude to Susan Y. Green, MPH and Scott A. Weldon MA, CMI, FAMI for their assistance in preparing the manuscript with special regard to the figures used.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
