Abstract
Introduction
Postoperative acute kidney injury (AKI) is a common postoperative complication in cardiac surgery, with varying reported incidences and prognostic factors. Renal hypoperfusion is believed to be a key factor contributing to postoperative AKI. Near-infrared spectroscopy (NIRS) monitoring, which assesses regional tissue saturation (RSO2), has been suggested as a tool to predict postoperative AKI. The aim of this systematic review was to examine the prognostic value of perioperative NIRS monitoring in predicting postoperative AKI in pediatric patients.
Methods and Results
After a systematic search in PubMed, EMBASE, and Cochrane library, twenty studies (1517 patients) were included. The inter-rater agreement on study quality was strong, yet a high risk of bias was identified.
Conclusion
The heterogeneity of the results—in part attributable to several potential confounding factors regarding study population, monitoring technique and the definition of AKI—together with the lack of a clear and consistent association between RSO2 values and AKI, currently preclude recommending NIRS monitoring as a reliable and valid clinical tool to “predict” AKI in the individual patient.
Keywords
Introduction
Postoperative acute kidney injury (AKI) is a common occurrence in pediatric cardiac surgery, with reported incidences ranging from 3.4% 1 to 86%. 2 Although various classification systems have been used in the diagnosis of AKI, the Kidney Disease Improving Global Outcomes (KDIGO) classification is now regarded as the standard diagnostic classification tool. 3 Biomarkers such as Kidney Injury Molecule-1 (KIM-1) and Neutrophil Gelatinase-Associated Lipocalin (NGAL) have shown a higher sensitivity to detect more subtle forms of renal injury, 4 but are still mostly used in research settings. 5
Occurrence of postoperative AKI is dependent on several pre-existing factors, such as young age, chronic kidney disease, anemia, cardiopulmonary bypass duration, multiple cross clamps, and preoperative inotropic support.6-8 Perioperative renal hypoperfusion and hypoxia due to intraoperative hypotension9,10 have been associated with postoperative AKI. 11 It has been shown that children developing AKI are more likely to have prolonged mechanical ventilation,12,13 longer intensive care unit stay, 14 and can evolve to chronic kidney disease. The relevance of AKI as a postoperative complication goes beyond (acute or chronic) kidney injury itself, since AKI has also been linked to an increase in morbidity and mortality, with mortality rates up to 89%.15,16
The use of near-infrared spectroscopy (NIRS) monitoring, which measures regional tissue saturation (RSO2), 17 enables the real-time detection of local tissue hypoxia. Some authors suggest that renal region tissue oxygenation (RrSO2) can be directly monitored by placing NIRS sensors on the flank skin overlying the kidney. 18
Additionally, assessment of cerebral (RcSO2) or somatic (RsSO2) tissue oxygenation can be a surrogate for evaluation of global systemic oxygenation and is considered a useful tool in predicting postoperative AKI.19-22
Over the last decade, various studies tested the hypothesis that low intraoperative RrSO2 values were associated with postoperative AKI, reporting variable findings—and in some centers, RrSO2 measurements are routinely performed in cardiac surgery.19,20,23,24 The aim of this systematic review was to investigate the prognostic value of perioperative RSO2 values to predict postoperative AKI in pediatric patients undergoing cardiac surgery.
Methods
This systematic review was registered publicly in the International Prospective Register of Systematic Reviews (PROSPERO; registration number: CRD42021241770) and is reported according to the 2020 guidelines from the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. 25 In this review we choose to focus on the pediatric cardiac surgery population.
Search Strategy and Article Selection
Three databases were selected for the systematic search (PubMed, EMBASE, and Cochrane Library; supplemental file 1). The database search was conducted on the 18th of February 2021 and repeated on the 18th of February 2024. Since most clinically used AKI definitions have been formulated after 2006,7,26 we have included studies from years 2006–2024.27-29 We also reviewed the bibliographies of the final selected studies, to identify any other potentially relevant study that was not found through the systematic search.
Studies were considered eligible for inclusion if the following a priori established criteria were met: 1. Original clinical studies, performed in the perioperative period in pediatric patients undergoing cardiac surgery (below 18 years of age). 2. Studies investigating either renal region (RrSO2), cerebral (RcSO2), or somatic (RsSO2) NIRS monitoring in the perioperative phase, defined as the day before surgery (preoperative), during surgery (intraoperative) and the days after surgery (post-operative). 3. Studies investigating the association of any of the RSO2 values with postoperative AKI as a primary outcome. 4. Studies using any of the following AKI definitions: - KDIGO criteria
3
or modified KDIGO criteria (based only on serum creatinine, sCr)30,31 - pRIFLE (pediatric Risk, Injury, Failure, Loss, End stage renal disease) criteria
32
- AKIN (Acute Kidney Injury Network) criteria
33
- (fractional) sCr increase
29
- Urine output and/or urinary and serum biomarkers as markers for renal dysfunction (e.g., KIM-1, NGAL, cystatin C, blood urea nitrogen and tissue inhibitor of metalloproteinases-2* insulin-like growth factor-binding protein 7 ([TIMP-2]*[IGFBP7]))
34
- estimated glomerular filtration rate
We excluded studies written in a language other than English, animal studies, case reports, review articles and conference abstracts. The search and bibliographic review were performed independently by two researchers (CKN and DM) using the web application Rayyan. 35
Definitions of RSO2 Values
RSO2 values are measured as percentage of tissue hemoglobin oxygen saturation and were presented as absolute values (%; with either mean ± standard deviation or median [interquartile range]), as decrease from baseline value (%), or as predefined calculations. The following RSO2 values were presented in the included studies: • RrSO2 ◦ Absolute RrSO2 values (%),1,30,36-44 or nadir of absolute RrSO2 value (%).
45
◦ Predefined threshold RrSO2 values (%) regarding total time (≥2 hours or <2 hours)
12
of RrSO2 below 50%,
2
or ≥20% reduction from baseline
40
for at least 60 consecutive seconds,
31
or 20 minutes.
14
◦ RrSO225 score and RrSO265 score (%min): difference between the baseline RrSO2 and the actual RrSO2 value (if decreased ≥25% from baseline or if <65%, respectively), multiplied by the time (minutes) during which RrSO2 is decreased ≥25% from baseline or is <65%, respectively.
13
◦ RrSO2 (%), calculated as a percentage of baseline pulse oximetry saturation (SpO2).
46
• RcSO2 • RsSO2
Data Extraction and Analysis
The primary outcome variables were the RSO2 values and their association with postoperative AKI. The following data were extracted from each study independently: 1. Publication-related data (first author, year, and journal), 2. Number of patients included, 3. Surgery type, 4. NIRS device type and location of sensor(s) placement, 5. RSO2 values, 6. AKI diagnostic criteria, 7. AKI incidence (percentage), and 8. Relation of the RSO2 values with postoperative AKI.
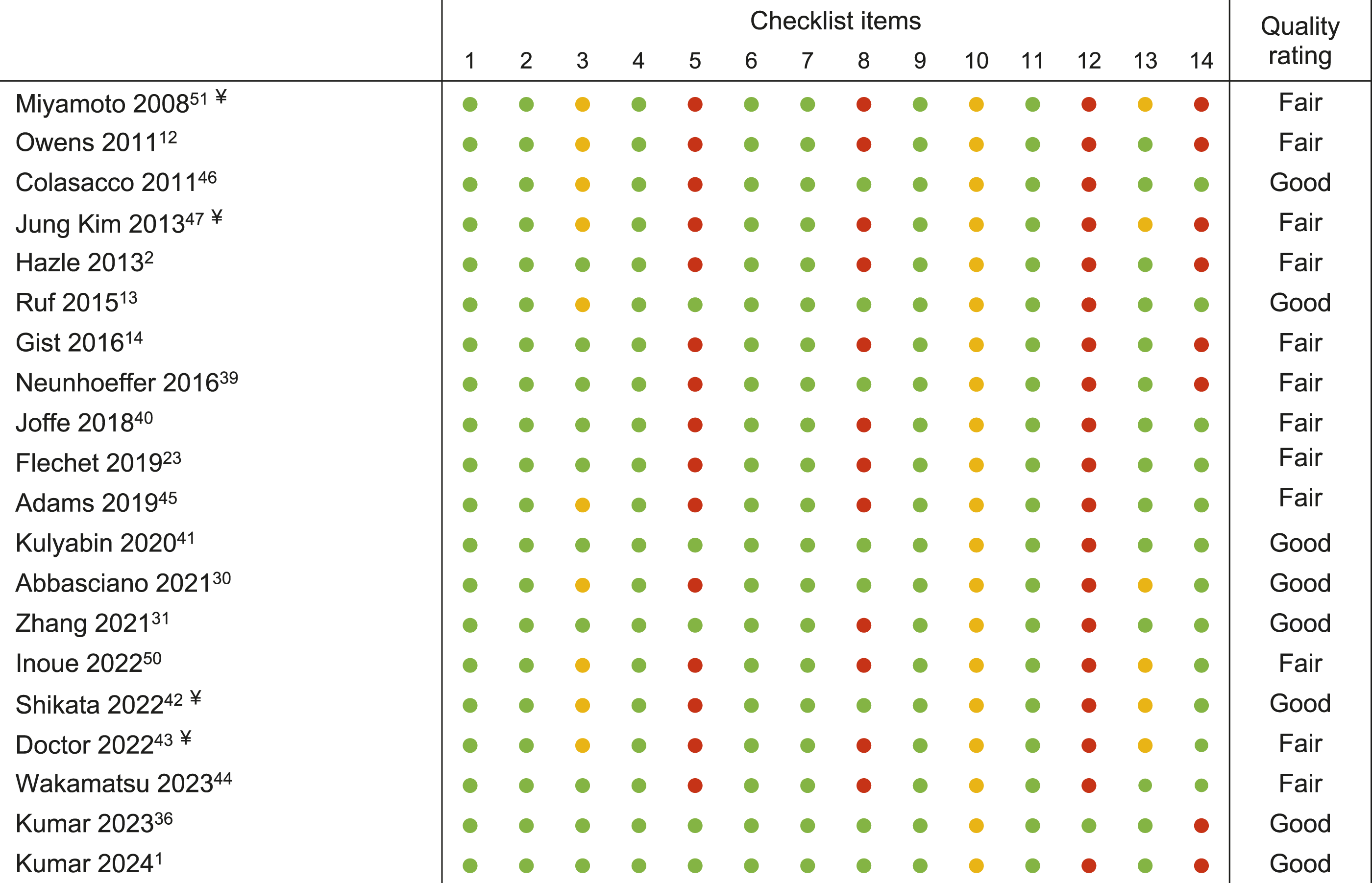
Risk of Bias Check and Overall Rating of the Included Articles as Determined After Reaching Agreement Between the Researchers.
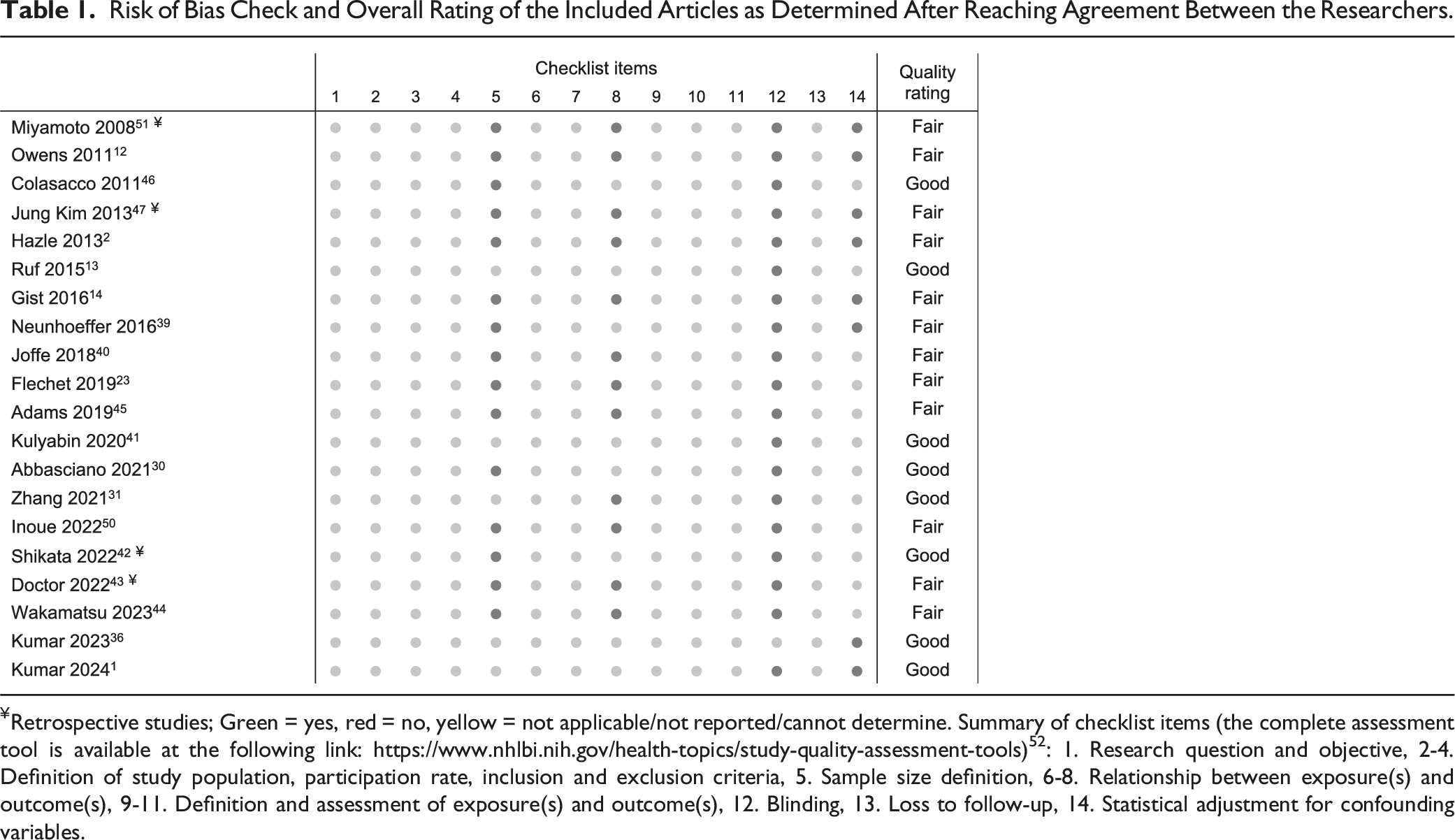
¥Retrospective studies; Green = yes, red = no, yellow = not applicable/not reported/cannot determine. Summary of checklist items (the complete assessment tool is available at the following link: https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools) 52 : 1. Research question and objective, 2-4. Definition of study population, participation rate, inclusion and exclusion criteria, 5. Sample size definition, 6-8. Relationship between exposure(s) and outcome(s), 9-11. Definition and assessment of exposure(s) and outcome(s), 12. Blinding, 13. Loss to follow-up, 14. Statistical adjustment for confounding variables.
Results
The database search retrieved 5304 studies, of which 1221 were duplicates and were removed. We excluded 4049 studies after reviewing title and abstract, and 49 studies were eligible for full text reviewing. Twenty-nine studies were excluded after full text reviewing and 20 studies were included, comprising 1517 patients with a median [interquartile range] of 57 [47–95] patients per study (Figure 1). Flowchart with the inclusion and exclusion of articles in this systematic review.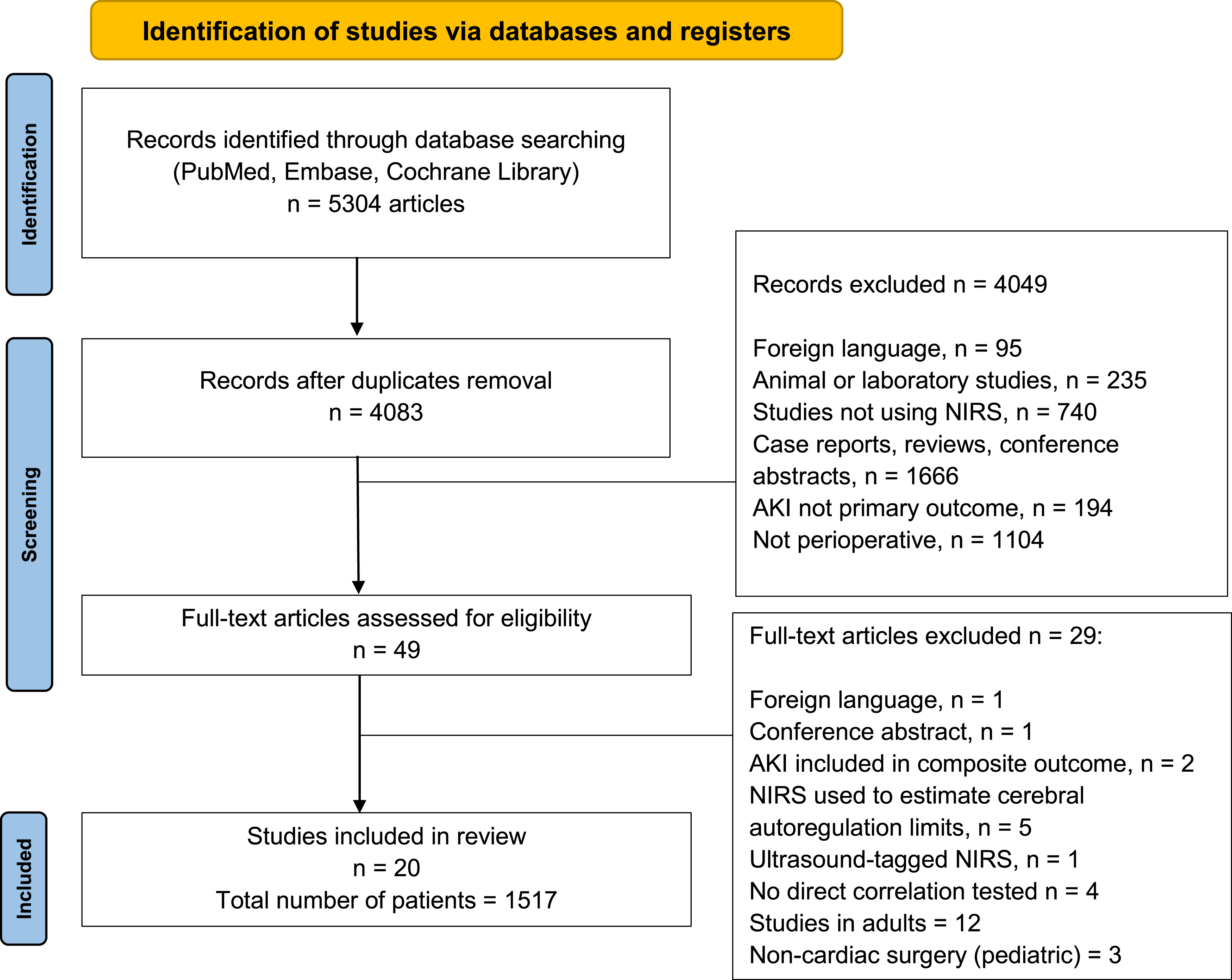
The inter-rater agreement regarding the quality of the studies was “strong” 53 (percent agreement 86.8%, Cohen’s Kappa = 0.72, 95% CI 0.64–0.80, P < .001; Table 1).
A high risk of bias was identified in four main categories: sample size justification, assessment of different levels of exposure, blinding, and statistical adjustment for potential confounding variables.
Most studies included pediatric patients under 12 months of age. Four studies were performed in pediatric patients with non-cyanotic heart disease undergoing aortic arch reconstruction.41,42,47,51 Fourteen studies had a mixed population, including both cyanotic and non-cyanotic pediatric patients, undergoing either corrective or palliative procedures.1,2,12-14,23,30,36,39,40,44-46,50 The reported incidence of postoperative AKI varied from 3.4% 1 and 86%. 2 The diagnostic AKI criteria used were the pRIFLE criteria (n = 7),12-14,39,43,47,50 the KDIGO criteria (n = 7),2,23,40-42,44,45 a modified KDIGO (using only sCr increase (n = 2),30,31 initiation of renal replacement therapy (n = 2),1,36 (fractional) sCr increase (n = 2),12,46 and urine output (n = 1). 51
Renal Region NIRS (RrSO2)
Overview of the Included Studies.
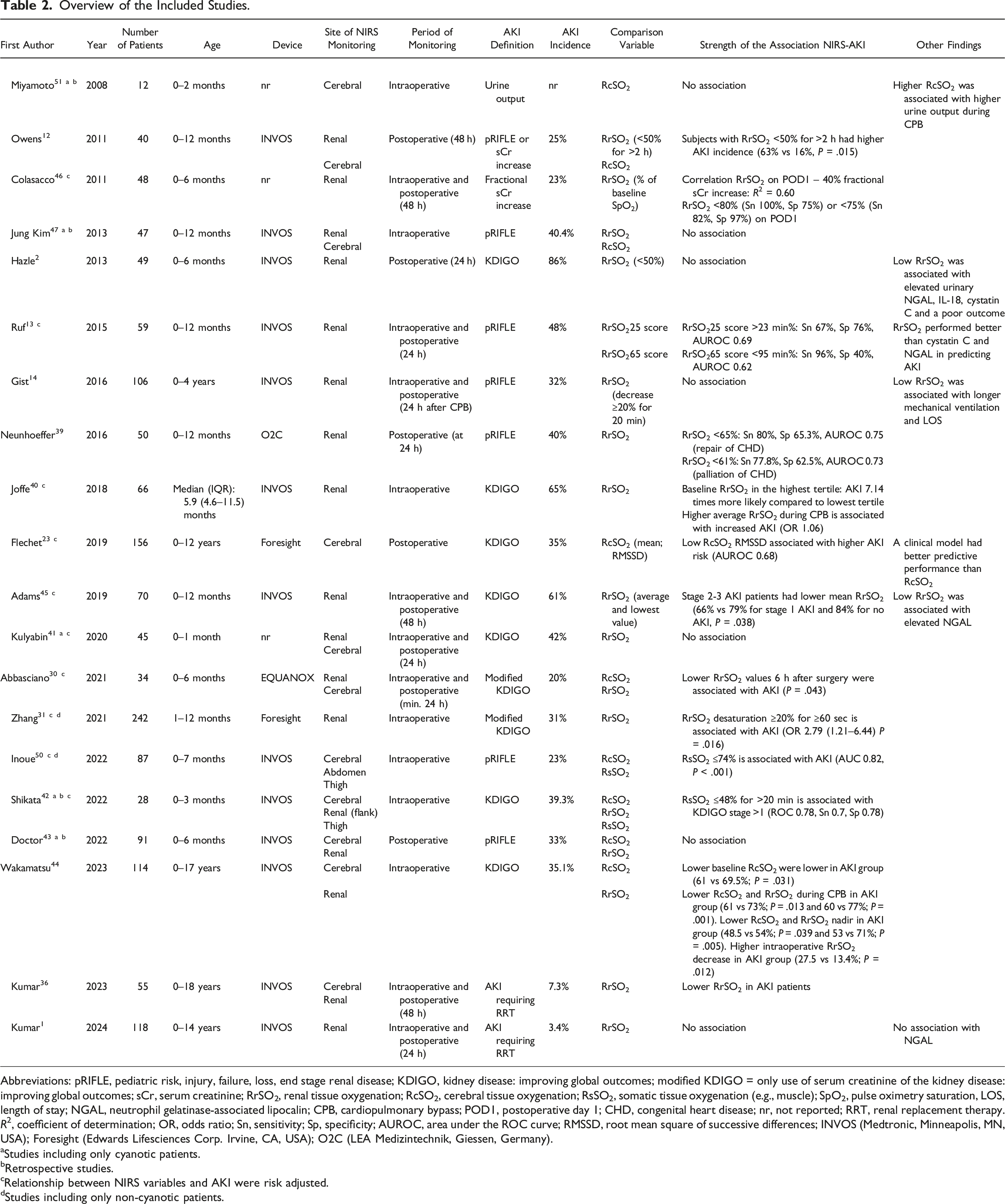
Abbreviations: pRIFLE, pediatric risk, injury, failure, loss, end stage renal disease; KDIGO, kidney disease: improving global outcomes; modified KDIGO = only use of serum creatinine of the kidney disease: improving global outcomes; sCr, serum creatinine; RrSO2, renal tissue oxygenation; RcSO2, cerebral tissue oxygenation; RsSO2, somatic tissue oxygenation (e.g., muscle); SpO2, pulse oximetry saturation, LOS, length of stay; NGAL, neutrophil gelatinase-associated lipocalin; CPB, cardiopulmonary bypass; POD1, postoperative day 1; CHD, congenital heart disease; nr, not reported; RRT, renal replacement therapy.
R2, coefficient of determination; OR, odds ratio; Sn, sensitivity; Sp, specificity; AUROC, area under the ROC curve; RMSSD, root mean square of successive differences; INVOS (Medtronic, Minneapolis, MN, USA); Foresight (Edwards Lifesciences Corp. Irvine, CA, USA); O2C (LEA Medizintechnik, Giessen, Germany).
aStudies including only cyanotic patients.
bRetrospective studies.
cRelationship between NIRS variables and AKI were risk adjusted.
dStudies including only non-cyanotic patients.
The RrSO2 was lower in preoperatively cyanotic patients at baseline12,40 but not during cardiopulmonary bypass (CPB)13,40 and after surgery. 13 RrSO2 values associated with a higher incidence of postoperative AKI were a low perioperative mean RrSO236,44 (66% vs 84% in patients without AKI), 45 intraoperative RrSO2 desaturation ≥20% for ≥60 seconds, 31 an intraoperative RrSO225 score >23 min% 13 or an intraoperative RrSO265 score <95 min%, 13 lower RrSO2 values at 6 hours postoperatively, 30 a postoperative RrSO2 <50% for >2 hours, 12 a postoperative mean RrSO2 <80% (higher sensitivity) or <75% (higher specificity) of individual baseline SpO2, 46 and a postoperative RrSO2 <65% or <61% (corrective or palliative surgical procedures, respectively). 39 The lowest intraoperative RrSO2 value predicted postoperative increases in urinary NGAL and [TIMP-2]*[IGFBP7], 45 and patients spending more cumulative time with postoperative RrSO2 values <50% had increased postoperative levels of urinary biomarkers (NGAL, IL-18 and cystatin C). 2 In one study higher RrSO2 values before or during CPB were associated with the development of postoperative AKI. 40 The remaining seven studies (of which four including solely cyanotic patients41-43,47) did not find an association between RrSO2 and postoperative AKI.1,13,14,41-43,47
Cerebral NIRS (RcSO2)
Pediatric patients who developed AKI had a lower maximum RcSO2 value (76% vs 79%) and a lower RMSSD (variability of RcSO2 that takes gradual shifts in mean into account) as reported by one study. 23 Another study reported lower RcSO2 values before surgery (61% vs 69.5%) and during CPB (61% vs 73%) in patients developing AKI. 44 The other eight studies found no association between RcSO2 values and postoperative AKI in cyanotic patients,42,43,47,51 in a mixed cyanotic and non-cyanotic population,12,30 and in non-cyanotic patients.31,50
Somatic NIRS (RsSO2)
RsSO2 values ≤74% measured at the thigh muscle in non-cyanotic patients undergoing ventricular septum defect repair were associated with postoperative AKI. 50 In aortic arch reconstruction with high-flow regional cerebral perfusion, a RsSO2 ≤48% for >20 min was associated with development of postoperative AKI (KDIGO stage >1). 42
Discussion
The aim of this systematic review was to investigate the prognostic value of perioperative RSO2 values to predict postoperative AKI in pediatric patients undergoing cardiac surgery. The findings of this review preclude the use of NIRS monitoring as a valid clinical tool to predict postoperative AKI.
Key Findings
Low RrSO2 values were associated with postoperative AKI in nine out of seventeen studies. One of these seventeen studies reported an association between higher intraoperative RrSO2 values and postoperative AKI. Only two out of nine studies reported an association of postoperative AKI with low RcSO2 values in pediatric cardiac surgery. Postoperative AKI was associated with lower RsSO2 in both studies reporting this measurement. Threshold RSO2 values under which postoperative AKI was likely to develop were extremely variable, making it difficult to establish threshold values for clinical practice. Therefore, the current evidence precludes recommending the use of NIRS monitoring as a valid clinical tool to predict AKI in the individual patient.
Study Population, Monitoring Site, and AKI Definition
A challenge to drawing comprehensive conclusions stemmed from the inclusion of patients with both cyanotic and non-cyanotic heart disease, which represent distinct pathophysiological profiles with varying baseline RrSO2 values. 40 In studies involving a mixed patient population, low RrSO2 and/or RcSO2 values were associated with AKI in eight and two out of 13 studies, respectively. None of the studies involving cyanotic pediatric patients found an association between RrSO2, RcSO2, and the development of postoperative AKI. In cyanotic pediatric patients, 40 low preoperative arterial oxygen saturation could play a protective role against perioperative kidney dysfunction, by a chronic reduction of metabolic rate of the brain and kidneys (“ischemic preconditioning”) 54 keeping RrSO2 and RcSO2 values close to their preoperative baseline values.
RcSO2 values have been suggested to be a surrogate of tissue oxygenation in general 55 and could be an indirect RrSO2 monitor.24,56 The prognostic value of RcSO2 to predict AKI in the pediatric population seems lower compared with RrSO2 values or RsSO2 values. A possible explanation is that the kidneys and other tissues (like muscles) suffer from hypoperfusion before the brain, due to hierarchical distribution of blood flow 57 and differences in organ blood flow autoregulation. 58
Proper placement of NIRS sensors for measuring RrSO2 may contribute to the variability of the results. Only three studies reported the use of ultrasound imaging when placing the NIRS sensor above the renal region,14,31,39 instead of relying on external anatomy landmarks. Two of these studies found an association between low RrSO2 values and AKI, emphasizing the contribution of contamination of the NIRS signal by extrarenal tissues in acquiring reliable RrSO2 measurements. 18
In addition, the depth of the kidney beneath skin surface is dependent on the patient’s size and weight, and the average depth of penetration of NIRS sensors is generally around 2 cm in children. It is possible that if the kidney is located “too deep” (e.g., in overweight patients or older infants), the NIRS device may not be accurately measuring renal tissue oxygenation. Even if the renal cortex is reached by the near-infrared light, the renal medulla—physiologically more sensitive to hypoxia than the cortex 59 —might remain out of the reach of renal region NIRS monitoring.
Diagnostic criteria used for diagnosing postoperative AKI were highly variable, reflected by the wide range of reported AKI incidences. It is possible that due to these differing criteria, the association between NIRS values and postoperative AKI may have been obscured. In pediatric patients we found some evidence for an association between RrSO2 and novel biomarkers of kidney injury—mainly NGAL and cystatin C—suggesting that RrSO2 might detect subtle alterations in renal function which may go undetected using conventional AKI criteria.
Strengths and Limitations
To the authors knowledge, this is the first systematic review about the use of perioperative RSO2 to predict postoperative AKI in pediatric cardiac surgery. A certified information specialist helped develop a complete search strategy, which would unlikely develop selection bias. We performed a validated quality assessment of the selected studies and determined an inter-rater variability to objectify the differences in opinion. The heterogeneity of the included studies (regarding, for example, methodological differences in NIRS devices and the reporting of NIRS values, and the different AKI definitions used), prevented us from performing a meta-analysis, making this a descriptive review with a systematic search.
Advice for Future Research
In pediatric cardiac surgery, future studies on this topic should classify patients based on cardiac pathophysiology (cyanotic vs non-cyanotic). 60 The focus should be on the monitoring of RrSO2 and RsSO2 values, since they might predict postoperative AKI more consistently than RcSO2. The proper positioning of NIRS sensors in the renal region should be guided by ultrasound imaging. The definition of RSO2 thresholds could be based on deviations from individual RSO2 baseline values rather than on absolute values, which might be less interpretable due to a wide variability in the population. The KDIGO criteria are currently advised in clinical practice 34 and should therefore be applied to detect AKI in future studies. We do not expect that the standardization of the population, monitoring and outcome in other observational studies will allow the definition of a single regional oxygenation threshold capable of predicting AKI, because of the multiple factors interplaying in determining kidney injury. It is likely that recommendations for threshold RSO2 values will continue to rely primarily on consensus and expert opinions, rather than on consistent data from observational studies. We would suggest designing interventional studies to determine if a “NIRS-directed” respiratory and hemodynamic perioperative management could potentially reduce the incidence of postoperative AKI.
Conclusion
Across twenty studies conducted in pediatric patients undergoing cardiac surgery, no clear or consistent association was found between RSO2 values and postoperative AKI. This currently precludes recommending the use of perioperative near-infrared spectroscopy monitoring as a reliable clinical tool for predicting postoperative acute kidney injury in this population.
Supplemental Material
Supplemental Material - Prognostic Value of Perioperative Near-Infrared Spectroscopy Monitoring for Postoperative Acute Kidney Injury in Pediatric Cardiac Surgery: A Systematic Review
Supplemental Material for Prognostic Value of Perioperative Near-Infrared Spectroscopy Monitoring for Postoperative Acute Kidney Injury in Pediatric Cardiac Surgery: A Systematic Review by Cornelia K. Niezen, Marco Modestini, Dario Massari, Arend F. Bos, Thomas W. L. Scheeren, Michel M. R. F. Struys, and Jaap Jan Vos in Seminars in Cardiothoracic and Vascular Anesthesia
Footnotes
Declaration of Conflicting Interests
The research group/department from M.M.R.F Struys received research grants and honoraria (paid to UMCG) from The Medicines Company (Parsippany, NJ, USA), Masimo (Irvine, CA, USA), Becton Dickinson (Eysins, Switzerland), Fresenius (Bad Homburg, Germany), Dräger (Lübeck, Germany), Paion (Aachen, Germany), Medtronic (Dublin, Ireland), Medcaptain Europe (Andelst, The Netherlands), for sponsor initiated research and consulting. T.W.L. Scheeren received research grants and honoraria from Edwards Lifesciences (Irvine, CA, USA) and Masimo Inc. (Irvine, CA, USA) for consulting and lecturing in the past (payments made to institution). TWLS is currently working as Senior Medical Director for BD Advanced Patient Monitoring (Heidelberg, Germany). The other authors declare no conflicts of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.