Abstract
Anesthetic management of open thoracoabdominal aneurysm (TAAA) repair poses a number of challenges for even the most experienced of cardiovascular anesthesiologists. This procedure encompasses a large number of unique anesthetic techniques, including one-lung ventilation, invasive hemodynamic monitoring, left-heart bypass, massive transfusion, selective renal and visceral perfusion, and central nervous system monitoring with CSF drainage. In this article, we aim to describe the anesthetic management for thoracoabdominal aortic aneurysm repair, including preoperative workup, intraoperative management, as well as postoperative concerns in the intensive care unit.
Keywords
Definition and Classification
Thoracoabdominal aneurysms are defined as aneurysms that extend from the descending thoracic aorta into the abdominal aorta. Dilation of the aorta can occur as a result of a number of factors, most notably the degradation and weakening of the medial layer of the aortic wall. Over time, hemodynamic shear forces coupled with progression of the underlying disease process can cause further aneurysm dilation.
The most common classification system used to characterize thoracoabdominal aneurysms was first described by Dr Crawford et al, in 1986, now colloquially known as the “Crawford Classification.” In this system, the type of TAAA is defined by the anatomic extent of the defect (Figure 1). Extent I occupies the descending thoracic aorta from the level of the left subclavian artery to the suprarenal abdominal aorta. Extent II also originates at the left subclavian artery; however, it extends further to the femoral bifurcation. Extent III extends from the distal descending thoracic aorta to the femoral bifurcation. Finally, Extent IV is located solely in the abdominal aorta; however, thoracic exposure and lung isolation are necessary for aortic clamp placement. Extent V has been described as an aneurysm located between the level of the diaphragm and the renal arteries.
1
Crawford classification of thoracoabdominal aneurysms with corresponding extents. Used with permission of Baylor College of Medicine.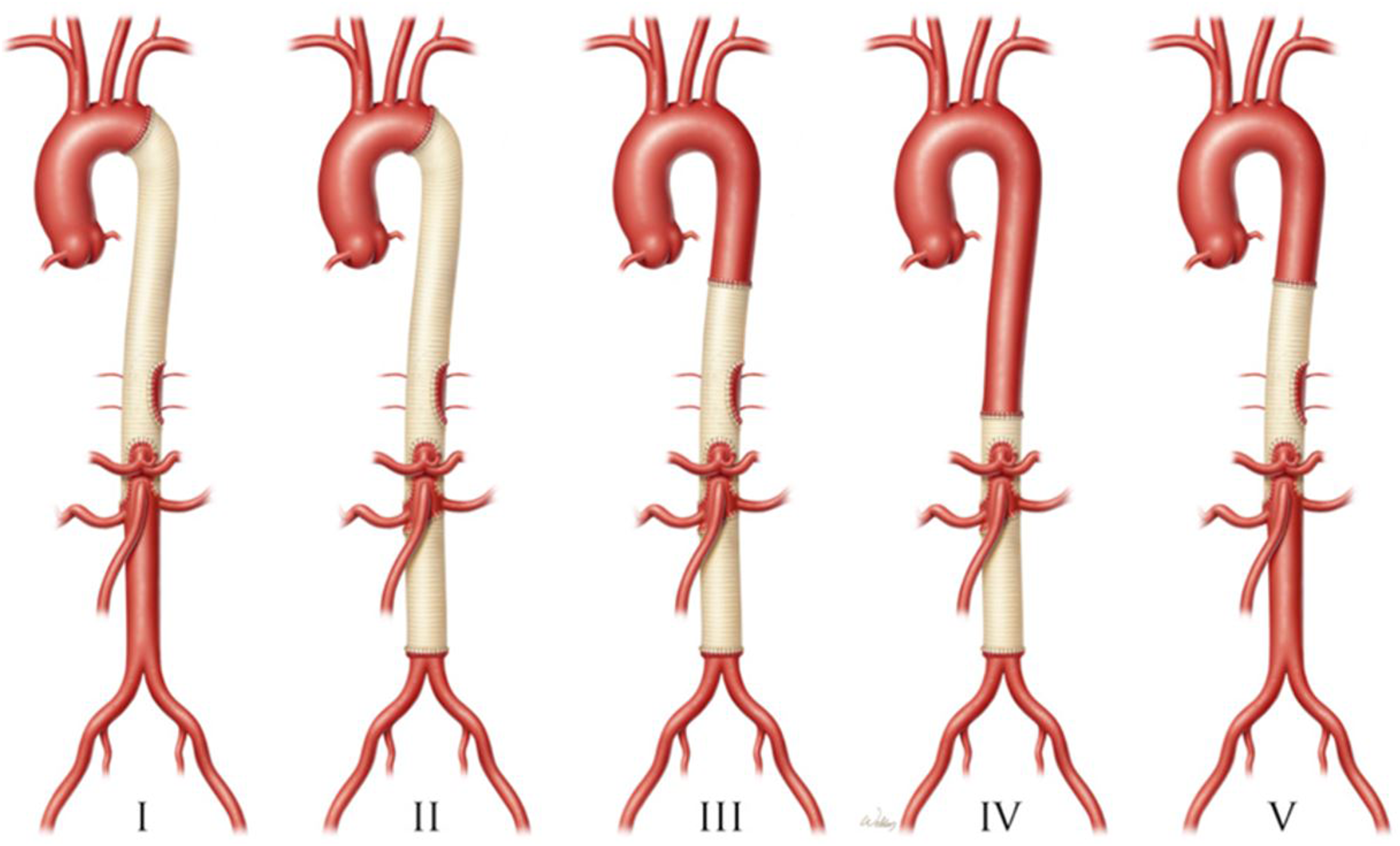
Aortic dissections can be defined according to 3 different classification systems: DeBakey, Stanford, or SVS/STS (Figure 2). The most common system used today is the Stanford Classification. Stanford Type A dissections involve the ascending aorta, proximal to the left subclavian artery. Any aortic dissection distal to the left subclavian artery is defined as a Stanford Type B dissection. In contrast, the DeBakey classification is defined by the exact origin of the intimal tear. DeBakey Type 1 dissections originate in the ascending aorta to the aortic arch. DeBakey Type 2 dissections are limited to strictly the ascending aorta. DeBakey Type 3 dissections originate in the descending aorta and extend distally to either above the diaphragm (Type 3a) or below the diaphragm (Type 3b). Finally, the SVS/STS aortic dissection classification system defines the various regions of the aorta by zone number (Figure 2). Various classification systems for ascending and descending aortic dissections, including the DeBakey system, Stanford system, and SVS/STS system. Used with permission of Baylor College of Medicine.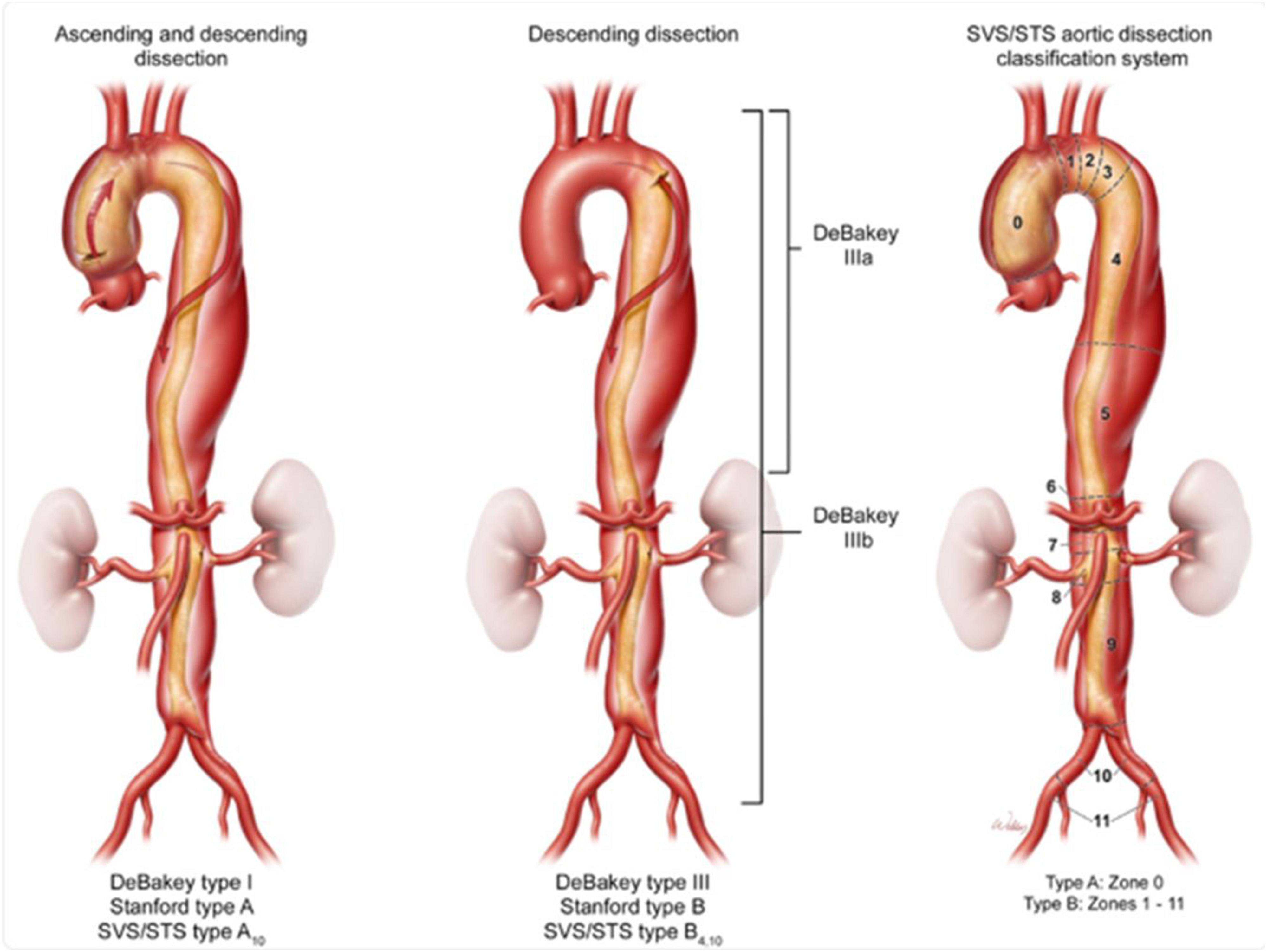
Epidemiology
Thoracoabdominal aortic aneurysms are relatively rare, however can be associated with a high morbidity and mortality without treatment. Based on a systematic review and meta-analysis by Melo et al, the incidence and prevalence of TAAAs were 5.3 per 100,000 individuals/year and 0.16%, respectively. The pooled incidence of ruptured aneurysms was 1.6 per 100,000 individuals/year. Forty five percent of thoracic aneurysms involve the ascending aorta, ten percent the arch, thirty five percent the descending aorta, and the remaining ten percent the thoracoabdominal aorta. 2
Etiology and Disease Progression
Given the relative scarcity of thoracoabdominal aneurysms, risk factors for their development are not well defined. However, the presence of connective tissue disease, genetic disease, smoking, hypertension, and preexisting atherosclerotic disease have all been implicated in the development of thoracoabdominal aneurysms. 3
Connective tissue diseases such as Marfan syndrome, Ehlers–Danlos syndrome, and Loeys–Dietz syndrome are known to be associated with both ascending and descending aortic aneurysms. These conditions are caused by inborn errors of metabolism leading to generalized defects of connective tissue. Thus, the medial layer of the aorta is subject to increased sensitivity to shear stress and subsequent dilation, leading to development of aneurysmal disease at a much younger age than the general population. The most common cause of isolated arch aneurysms and descending aortic aneurysms is atherosclerotic disease, which weakens the intimal layer of the aorta.
The main causes of mortality associated with thoracoabdominal aneurysm include dissection and rupture. If left untreated, the rate of rupture can approach up to 80%. 4 If there is a concomitant aortic dissection, patients are at risk for downstream consequences including stroke, renal injury, bowel and mesenteric ischemia, and spinal cord ischemia. Therefore, it is imperative to seek regular follow-up and potential treatment as soon as possible to prevent further dilation and inevitable rupture.
Diagnosis
The majority of patients with thoracoabdominal aneurysms are diagnosed incidentally through radiographical imaging.
EKG: Although not necessarily specific, patients with thoracoabdominal aneurysms may demonstrate EKG findings representative of the pathologies that contribute to development of aneurysm. For instance, a patient with an aneurysm secondary to uncontrolled hypertension and peripheral vascular disease may display elevated R wave voltages across precordial and limb leads, indicative of left ventricular hypertrophy. Additionally, retrograde extension of a dissection into the coronary artery or involvement of the aortic valve may demonstrate EKG changes, including a variety of ischemic ST/T wave changes.
X-Ray: Patients with thoracic aneurysms can display widening of the mediastinum, deviation of the trachea, and enlargement of the aortic knob. However, this radiographic modality is not the most sensitive or specific of all modalities available, and it is not uncommon for descending aneurysms of various sizes to remain undetected on X-ray.
CT scan: CT scans are perhaps the most widely used radiographic modality in the diagnosis of aortic aneurysms. This technique is useful in all stages of aneurysm treatment, including screening, longitudinal monitoring, preoperative planning, and in emergency assessment. In particular, CT’s high spatial resolution can help surgeons clearly evaluate the aneurysm’s relationship with surrounding structures, the three-dimensional anatomy of aortic branches, as well as aortic wall morphology. 5 Dissections within the aortic root must be examined with EKG-gated CTA to mitigate pulsation artifacts, which can mimic aortic dissection flaps. 6
MRI
MRI has several distinct advantages over other modalities for the characterization of thoracoabdominal aneurysms; however, this comes at the expense of inferior spatial resolution and a markedly lengthier examination time. MRI may have improved sensitivity for early detection of periaortic inflammation in inflammatory or infectious aortic aneurysms. 7
Echocardiography
Echocardiography remains a critical tool in the initial diagnosis and evaluation of all aortic aneurysms. In particular, thoracic and thoracoabdominal aortic aneurysms are frequently associated with aortic valve pathology including bicuspid valve and functional aortic regurgitation. However, CT and MRI provide a better method to accurately assess aortic aneurysm dimensions.
Transesophageal echocardiography can be useful in the intraoperative management of thoracoabdominal aneurysm repair, particularly in assessment of the aortic valve and left ventricular function in the setting of left heart bypass. However, due to the presence of the trachea in close proximity to the aortic arch, certain parts of the aorta are difficult to assess. Furthermore, there can be a substantial risk of aortic rupture with placement of a TEE probe, especially in patients with large aneurysms with weak connective tissue. Institutional practices may vary, and some centers do not employ routine use of TEE intraoperatively.
If an aneurysm is detected incidentally, imaging must be regularly obtained to monitor for interval growth, intramural hematoma, aortic dissection, rupture, and/or leak. 8 Frequency of imaging can vary according to the source or guideline referenced and also depends on patient characteristics including age, comorbidities, and aneurysm size. According to the 2022 ACC/AHA Aortic Disease Guideline, TTE is recommended at the time of diagnosis to evaluate for aortic valve pathology, followed by a CT scan within 6–12 months. If stable, imaging frequency can be anywhere between 6 and 24 months afterward. 9
Treatment
Medical
Upon diagnosis of a thoracoabdominal aneurysm, there are several core recommendations regarding medical management. According to the 2022 ACC/AHA guidelines, 10 it is a class I recommendation to initiate antihypertensive medications if SBP >130 mmHg and DBP >80 mmHg, with the aim of decreasing shear forces on the aorta. It is a class II recommendation to initiate statin therapy without evidence of atherosclerotic disease, upgraded to class I if there is evidence. Finally, all patients are recommended to stop smoking to minimize development of atherosclerotic disease and subsequent weakening of the aortic wall. These risk factors can compound and further increase the risk in patients with underlying connective tissue disease.
Indications for Repair
Symptomatic thoracoabdominal aneurysms carry a high risk of morbidity and mortality; therefore, immediate surgical repair is indicated. In non-symptomatic patients, risk of dissection and rupture increases exponentially at diameters >5.5 cm, and as such it is reasonable to proceed with surgical repair. Patients with connective tissue diseases, such as Marfan’s syndrome, Loeys–Dietz syndrome, or Ehlers–Danlos syndrome, can benefit from earlier repair (diameter >5 cm), as the risk of dissection and rupture of thoracoabdominal aneurysms is much greater than that of the general population. 11
Dissection and rupture can occur nonetheless in patients below the operative size cutoffs. One study found that the majority of patients presenting with acute Type B aortic dissection had an aortic diameter less than 5.5 cm and were not within the guidelines for elective repair. 12 In another study, patients with aortic diameters of 50 mm had a 5.5% incidence of definite adverse aortic events (rupture/dissection) and 8.0% incidence of possible adverse aortic events. 13 Additionally, there is more emerging evidence that aortic sizing should be indexed to body mass area for more accurate treatment. Consideration should be given to patients below the threshold for intervention, particularly if there is an endovascular option.
Since open thoracoabdominal aneurysm repair is an incredibly high risk and complex operation, the decision to proceed with non-emergent and scheduled surgery is multifactorial and may not be limited strictly to aneurysm size. As such, regular multidisciplinary communication between surgeons, cardiologists, anesthesiologists, and other perioperative specialists is essential. Scheduled surgical repair of thoracoabdominal aneurysm is typically offered whenever patients are experiencing symptoms secondary to the aneurysm. 14 If a patient is symptomatic, one can expect to experience the consequences of mass effect of the aneurysm on adjacent thoracic structures. 15 For instance, patients may experience dyspnea, cough, or respiratory tract obstruction secondary to compression of the trachea or mainstem bronchi; dysphagia as a consequence of esophageal compression; or hoarseness from irritation of the recurrent laryngeal nerve. Dissections of the thoracoabdominal aorta can present with sharp back pain, abdominal pain (from mesenteric ischemia), as well as laboratory indicators of end-organ ischemia (elevations in lactate, creatinine, etc). Any Type A aortic dissection is immediately classified as a surgical emergency.
There are 2 surgical methods for thoracoabdominal aneurysm repair: endovascular and open. Endovascular repair was first described in 2001, reserved for patients with poor cardiovascular reserve. Over the past few decades, there has been a significant improvement in graft technology (including the development of fenestrated, branched, parallel grafts, etc), leading to a higher incidence of patients receiving primary endovascular repair. Endoleak remains the chief complication of endovascular repair, with incidence ranging between 15% and 66%, with reintervention rate ranging from 3% to 33%. 16 As such, open technique is generally preferred due to a lower frequency of reintervention rate. There are currently no prospective randomized trials comparing endovascular vs open technique, and as such, more research is indicated before drawing definite conclusions.
Preoperative Evaluation
Prior to undergoing a TAAA repair, it is necessary to complete a comprehensive preoperative assessment for medical optimization as well as risk stratification. Multiple standardized procedures have been provided by professional organizations including the American Society of Anesthesiologists. Preoperative functional status has been shown to be a strong independent predictor of perioperative death, and as such, should be evaluated along with baseline mobility. 17 It is not uncommon to have patients present with evidence of previous stroke. Neurologic assessment should include a physical exam to determine baseline deficits, carotid artery Doppler ultrasonography, and computed tomography scan in select patients. Angiographic studies should be considered in order to evaluate the size of the aorta as well as variations in the attachment of major spinal cord branches or the presence of collateral circulation that could impact surgical strategies.
Tobacco use as well as pulmonary disease is prevalent in patients undergoing TAAA repair, including chronic obstructive pulmonary disease (COPD). COPD has been associated with aortic aneurysm rupture and is more frequent in patients with aortic aneurysm. 18 The presence of an aortic aneurysm can also cause compression of the left mainstem bronchus or pulmonary vessels resulting in pulmonary edema or dyspnea. Pulmonary function should be evaluated with spirometry and for diffusion capacity for carbon monoxide. Additionally, this is important in order to assess the patient’s ability to withstand single lung ventilation during the procedure. High-risk patients may benefit from pulmonary optimization with inhaled bronchodilators and possibly steroids. If severe dysfunction is present, an alternative modality such as an endovascular approach may be considered.
A comprehensive cardiac exam is also essential, as coexisting coronary artery disease is prevalent amongst this patient population. In 1 case series, 15% of patients undergoing TAAA repair had prior coronary artery bypass grafting. 19 The presence of coronary artery disease may impact the timing of repair and revascularization strategy. Aortic cross-clamping, particularly without left heart bypass, can increase afterload and myocardial oxygen demand. Patients should also undergo electrocardiography, echocardiography, and if needed a myocardial perfusion study or cardiac catheterization. Patients can be further risk stratified using the Revised Cardiac Risk Index or Society of Thoracic Surgery Adult Cardiac Surgery Database risk calculator.
Acute kidney injury (AKI) is a common complication associated with TAAA repair. Postoperative dialysis rates have been reported to be up to 12%. 20 Chronic kidney disease is a significant risk factor for mortality and need for postoperative dialysis. Preoperative assessment of kidney function is essential, and, if present, consultation with a nephrologist and perioperative placement of dialysis access may be necessary to allow for continuous renal replacement therapy postoperatively. This may help curb large hemodynamic changes and mitigate risk for hypotension and spinal cord ischemia.
Intraoperative Anesthetic Considerations
Induction of anesthesia for patients with thoracoabdominal aneurysms requires tight hemodynamic control to minimize risk of rupture and/or dissection. Patients undergoing open repair require at minimum a right radial arterial line, as left-sided radial lines are often inadequate in the setting of proximal aortic clamping: if the aortic clamp is placed proximal to the left subclavian, one may lose blood pressure monitoring capability. As a result, the redundancy of bilateral radial arterial lines may be beneficial, especially if direct axillary cannulation is employed. Furthermore, cases involving left heart bypass require a femoral arterial line, which provides a more direct estimation of distal aortic anastomosis pressure. However, placement of a femoral arterial line may be limited in patients with severe peripheral arterial disease or extensive Type B Aortic Dissection. Central venous access with 1 or 2 large bore central cannulas is important to facilitate transfusion of blood products. Although not routinely needed, a pulmonary artery catheter can be useful for monitoring right-sided afterload, left-sided filling pressures, continuous cardiac output, and mixed venous oxygenation, particularly in the setting of proximal aortic clamping and left heart bypass. A pulmonary artery catheter can also be employed in postoperative ICU care for the patient, allowing for monitoring of filling pressures, cardiac output, mixed venous oxygenation, and to assist in volume management.
Hemodynamic shear stress on the aorta must be minimized during induction of anesthesia and intubation. To do so, the patient must maintain a relatively low blood pressure and low heart rate. Propofol, opioids, calcium channel blockers (nicardipine and clevidipine), vasodilators (nitroglycerin and nitroprusside), and short acting β-blockers such as esmolol may be used judiciously to facilitate these goals. However, agents that do not cause reflex tachycardia are preferred to further minimize hemodynamic shear stress. In general, we aim for a systolic blood pressure of less than 120 mmHg and a heart rate less than or equal to 60–70 bpm.
Airway Management
Lung isolation is mandatory to allow for surgical access to the descending thoracic aorta. This is most reliably accomplished with a double-lumen endotracheal tube; however, alternative techniques such as bronchial-blockers or EZ blockers may be used as well. In patients with arch involvement, one must be cognizant of the potential for recurrent laryngeal nerve involvement and postoperative vocal cord paralysis. Furthermore, in patients with exceptionally large descending aortic aneurysms, mass effect of the aneurysm on to the airway increases the risk of tracheomalacia, which can be devastating in the setting of traumatic intubation. Lastly, due to the potential for airway edema in the setting of massive transfusion, we generally do not exchange double-lumen endotracheal tubes for single-lumen endotracheal tubes at the conclusion of the case, proceeding to the intensive care unit with the DLT in situ. This also allows for rapid lung isolation if needed postoperatively in the setting of lung/airway injury, hemorrhage, or need for re-operation or re-exploration. However, management of a DLT outside of the operating room setting can be challenging, as other medical staff including nurses, respiratory therapists, and intensivists must feel comfortable to troubleshoot any issues that may arise. As such, depending on institutional preferences, the risks of airway exchange to a single-lumen ETT may outweigh the benefits of managing a DLT postoperatively. Tube exchange should be performed cautiously with an exchange catheter and video laryngoscopy given the high likelihood of airway edema.
Positioning
Positioning for thoracoabdominal aneurysm must be done properly to facilitate adequate surgical exposure, as well as for the prevention of position-related complications. As shown in Figure 3,
21
the patient is placed in a modified right lateral decubitus position with the hips rotated 30° clockwise and the shoulders 60° clockwise. An axillary roll is placed to prevent brachial plexus injury, and multiple sheets must be placed under the head to prevent cervical spine injury. The arms are secured with arm boards.
22
Schematic of proper positioning for open TAAA repair. Used with permission of Baylor College of Medicine.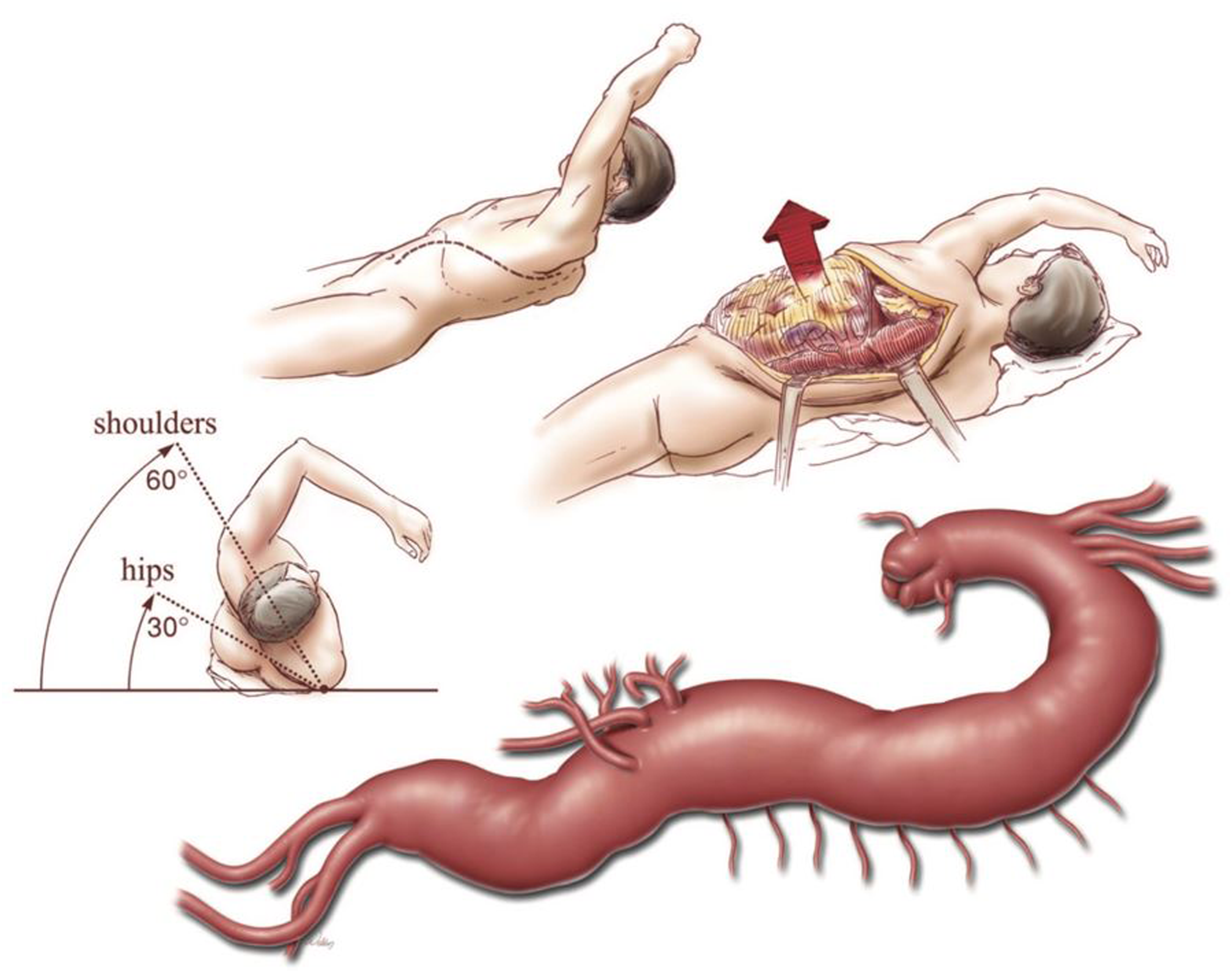
Blood Management
Open TAAA repair has a high probability of blood loss, with the potential of exceeding the patients’ starting blood volume. Hemorrhage is a major cause of death in patients that do not survive thoracic aneurysm repairs. Aside from known surgical risk of hemorrhage, many patients with thoracoabdominal aortic disease have concurrent hepatic and renal dysfunction that can contribute to systemic coagulation abnormalities. Furthermore, there is a well-described chronic consumptive coagulopathy that can affect patients with aortic aneurysms and acute aortic dissections. 23 Preoperatively, we recommend obtaining baseline coagulation labs including PT, PTT, INR, fibrinogen, and platelet count. Clear communication with blood banks preoperatively is imperative, as massive transfusion protocols are routinely needed. Also, it is recommended to use antifibrinolytic therapy such as ε-aminocaproic acid or tranexamic acid to minimize degradation of clots. Blood loss can be significant in these cases due to back bleeding from the intercostal arteries, which are often ligated when the aorta is opened. Cell-scavenging devices are commonly utilized and have reduced the need for allogeneic blood transfusions. Heparinization is performed prior to aortic clamping, with a typical dose of 1 to 1.5 mg/kg and goal Activated Clotting Time (ACT) of 220–270 seconds for heparin-bonded circuits without a membrane oxygenator or cardiotomy reservoir. However, should the patient require a membrane oxygenator or cardiotomy reservoir, a higher ACT goal of >400 seconds is required. 24 Shed whole blood harvested from the field via suction can be returned to a cardiotomy reservoir and subsequently to a rapid infusion device. The rapid infuser can then be managed by the anesthesiologist to return autologous whole blood back to the patient. Acid-citrate-dextrose (ACD) solution is added to the cardiotomy blood to prevent clot formation, and calcium supplementation may be necessary. After the conclusion of left-heart bypass, further coagulopathy can ensue as a consequence of systemic inflammation, thrombocytopenia, and mechanical degradation of clotting factors. 25 Coagulopathy after cases requiring deep or moderate hypothermic circulatory arrest can be quite profound and requires diligence on the anesthesiologists’ part to ensure appropriate availability of blood products. We recommend guiding transfusion with point of care testing including TEG, ROTEM, as well as traditional analyses including CBC, fibrinogen, PT, and PTT.
Mechanical Circulatory Support
In the setting of a proximal aortic cross-clamp, particularly in Extent I and II repairs, mechanical circulatory support is a useful adjunct in the preservation of end-organ perfusion. Two main modalities exist to achieve this goal: left heart bypass and partial cardiopulmonary bypass. Regardless of the exact circuit used, both methods withdraw oxygenated blood from the left-sided circulation, usually via the left inferior or superior pulmonary veins, and return blood into the distal descending thoracic aorta (Figure 4). Physiologically, this technique aims to reduce left ventricular afterload, improve thoracic spinal perfusion, and preserve visceral and renal perfusion. Schematic of cannulation for left heart bypass. Inflow cannula is placed in the left inferior or superior pulmonary vein, and outflow cannula is placed in the distal descending aorta. Used with permission of Baylor College of Medicine.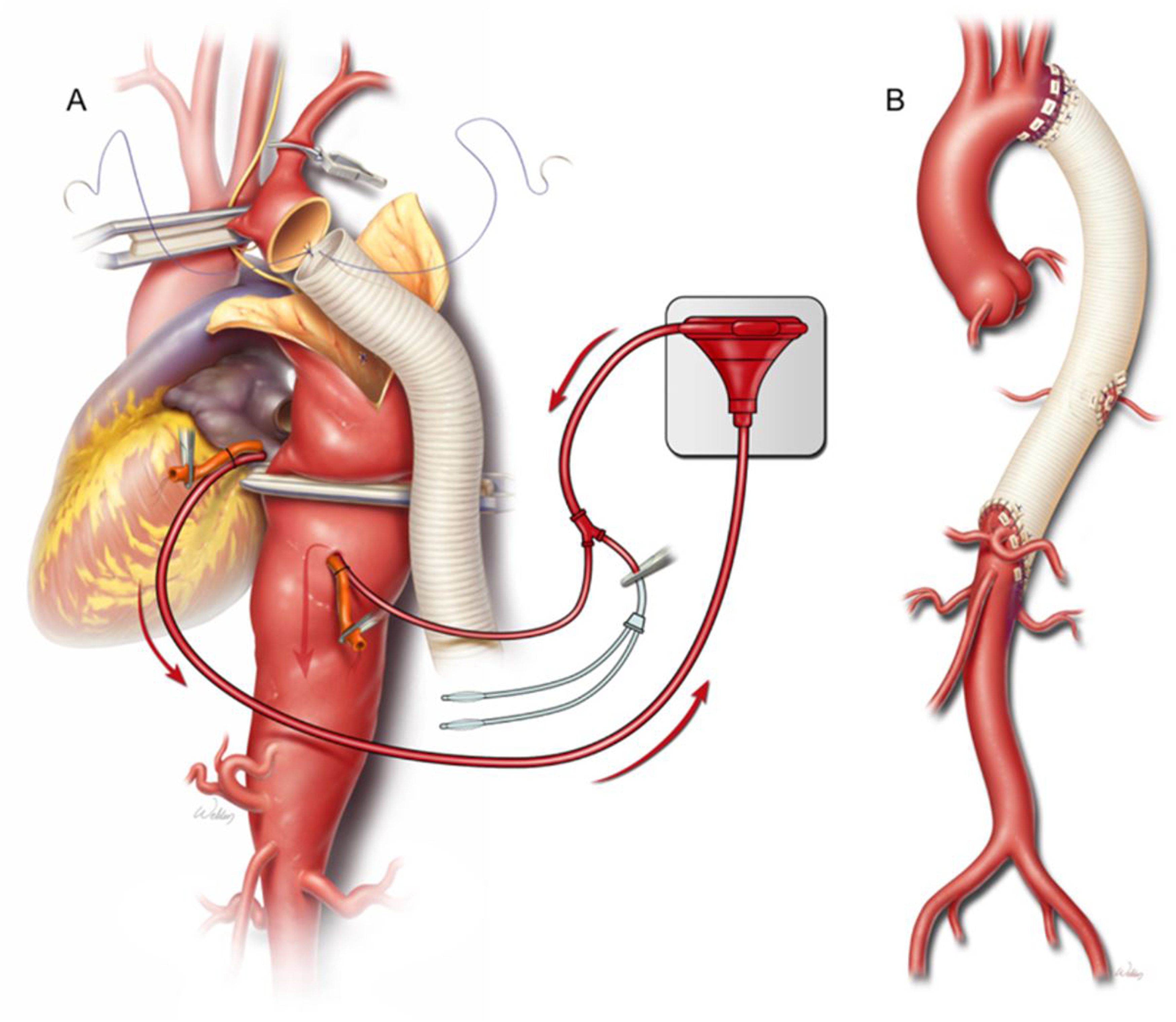
The main difference between left heart bypass and partial cardiopulmonary bypass lies in the circuit used. Left heart bypass is typically a heparin-bonded ECMO circuit without a cardiotomy reservoir or membrane oxygenator. Systemic heparinization is required, typically with an ACT goal of ∼250 seconds. The lack of a reservoir or membrane oxygenator helps eliminate air–blood contact and reduce the subsequent inflammatory response. Typically, this strategy is employed with the use of cell-saver and a rapid infusion system. Patients under left heart bypass must be able to tolerate single right lung ventilation in order to maintain adequate oxygenation. Communication between the anesthesiologist, surgeon, and perfusionist during left heart bypass is crucial, as flows may need to be manipulated depending on hemodynamics. Increasing flow rate will shunt more blood away from the left heart towards lower-extremity circulation, reducing proximal blood pressure and flow to the upper extremities and brain.
Partial cardiopulmonary bypass circuits maintain similar cannulation strategies; however similar to a complete cardiopulmonary bypass circuit, a cardiotomy reservoir and membrane oxygenator are used. As such, a higher ACT goal of >400 seconds is necessary to prevent thrombosis. This strategy can be particularly helpful in patients with preexisting pulmonary disease, particularly if a patient is unable to tolerate selective right lung ventilation due to poor function or reserve. Regardless of the circuit used, flow rate is adjusted to minimize proximal hypertension and improve visceral perfusion, approximately 1 liter/min of flow. Care must be taken to avoid hypoperfusion of the carotid arteries in the setting of high flow, through close monitoring of cerebral oximetry (Figure 5). Selective perfusion of renal or visceral arteries utilizing a pump and oxygenator/heat exchanger to deliver warm blood. Used with permission of Baylor College of Medicine.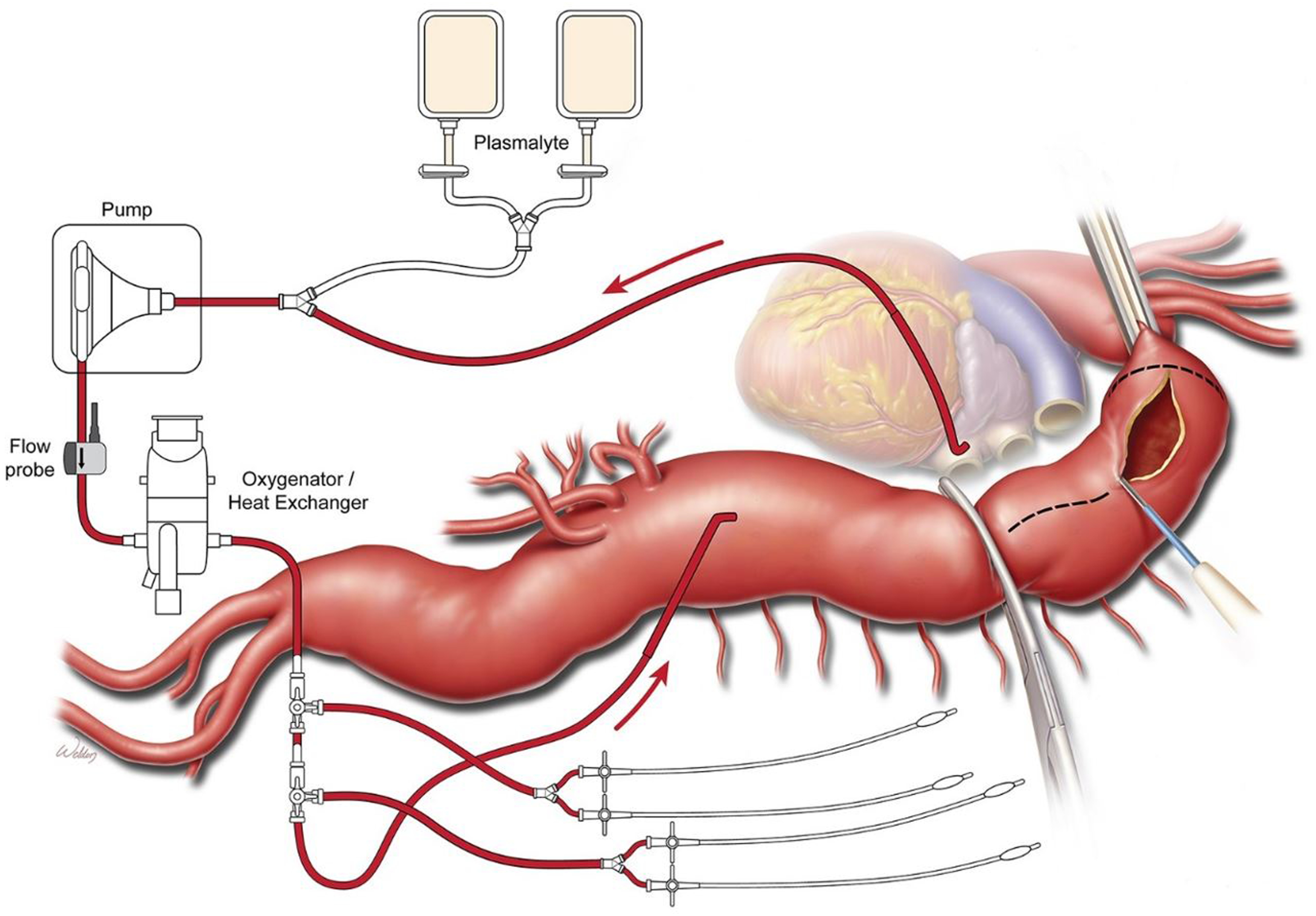
Furthermore, as various arterial branches are reconnected to the aorta, many centers advocate for a sequential clamping technique. By progressively adjusting clamp position distally, we can allow for physiologic pulsatile perfusion of the intercostal, visceral, and renal arteries, which can minimize ischemic time and its subsequent complications including bowel ischemia, spinal cord ischemia, and acute kidney injury (Figure 6(C) to (H)). With each subsequent release of the aortic clamp, the anesthesiologist must be acutely aware of the physiologic changes associated with the reperfusion of high vascularized and metabolic organ systems. One can expect a large bolus of deoxygenated, acidotic, and hypothermic blood to return into circulation, which manifests physiologically as hypotension, bradycardia, acidosis, and hypercarbia. This brief period of instability can be tempered with a sodium bicarbonate infusion, calcium chloride, hyperventilation, vasopressor, and inotropic support. Completed repair of a DeBakey type III aortic dissection. A four-branched graft was used to aid Crawford extent II thoracoabdominal aortic aneurysm repair. Alternates to this approach include a single visceral patch that incorporates the celiac axis, superior mesenteric artery, and both renal arteries (upper inset) or a three-vessel patch that incorporates the celiac axis, superior mesenteric artery, and right renal arteries, with the left renal artery reattached as a button (lower inset). Used with permission of Baylor College of Medicine.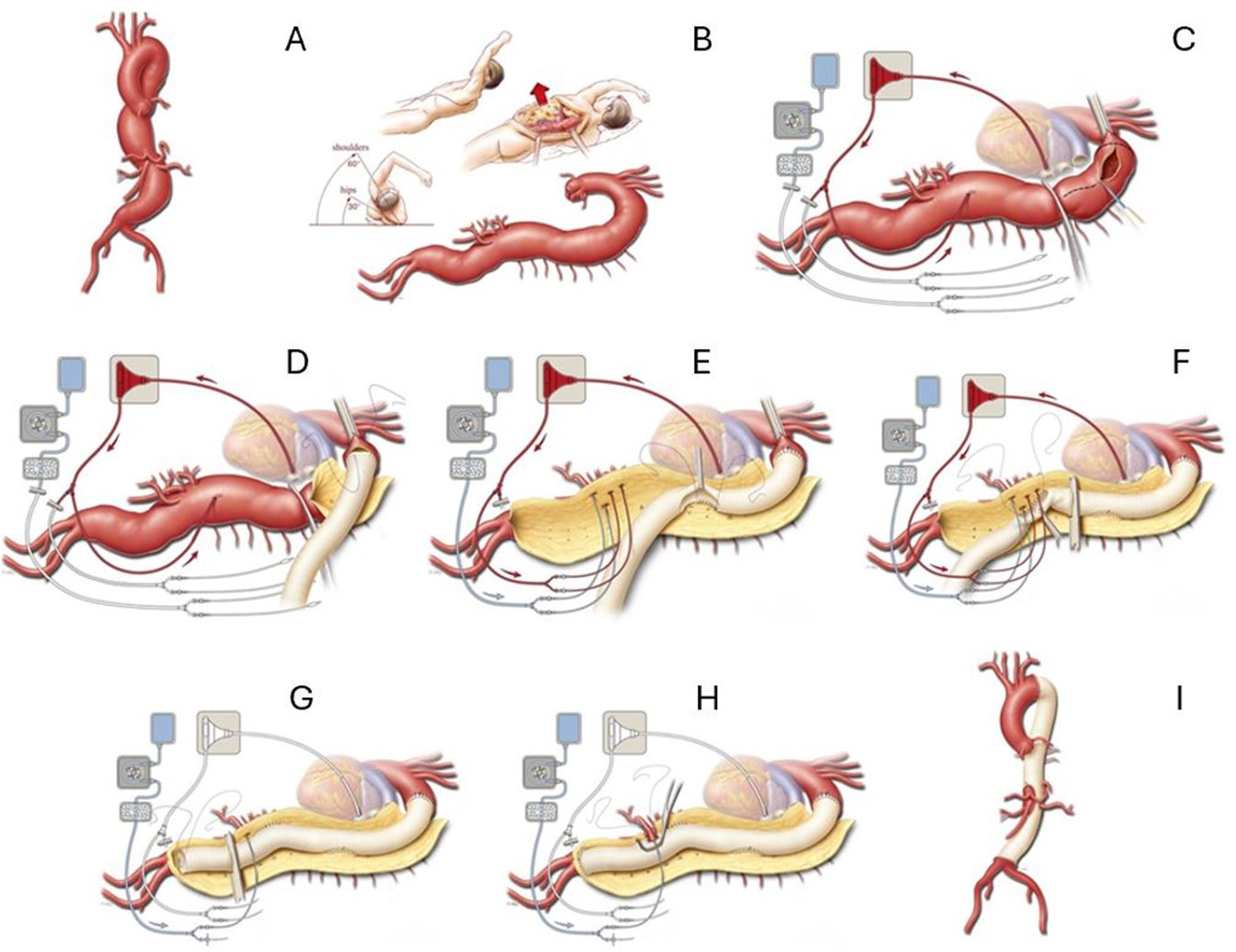
In addition to facilitating antegrade perfusion of aortic branches, it is possible to directly cannulate the renal arteries and splanchnic circulation. Preservation of end organ perfusion is imperative: according to a study from Texas Heart Institute in 2013, acute renal dysfunction occurred in up to 12% of patients undergoing open TAAA repair, 7% required hemodialysis at discharge, and bowel ischemia in approximately 1%. 26 More specifically, Extent II and III repairs were especially implicated in postoperative renal dysfunction. Within the left heart bypass circuit, surgeons can directly cannulate the renal arteries and visceral circulation to maintain perfusion with isothermic blood and/or cold crystalloid solution. In a single-center study published in 2002, selective cold crystalloid perfusion of the renal arteries was superior to normothermic blood in the prevention of acute kidney injury. 27 Alternatively, it is possible to utilize VA ECMO with an oxygenator to selectively perfuse the viscera and renal arteries with warm oxygenated blood (Figure 5); however, further research is needed to compare this technique to cold crystalloid perfusion.
CNS Monitoring and Spinal Cord Ischemia
One of the unique and dreaded consequences of open thoracoabdominal surgical procedures is ischemia to the spinal cord. As shown in Figure 7, the thoracic spinal cord is supplied directly by a number of direct branches of the aorta, most notably the artery of Adamkiewicz. This artery usually originates on the left side of the aorta between T9 and T12, supplying the anterior spinal cord directly. Extent I and II thoracoabdominal aneurysm repairs inevitably require a replacement of this crucial part of the aorta, and as such, it is imperative to monitor and prevent anterior spinal cord ischemia. If not protected adequately, patients may experience postoperative motor weakness and paraplegia. Anatomic diagram of the thoracic spinal cord, with special attention to the perfusion of the anterior spinal cord. Used with permission of Baylor College of Medicine.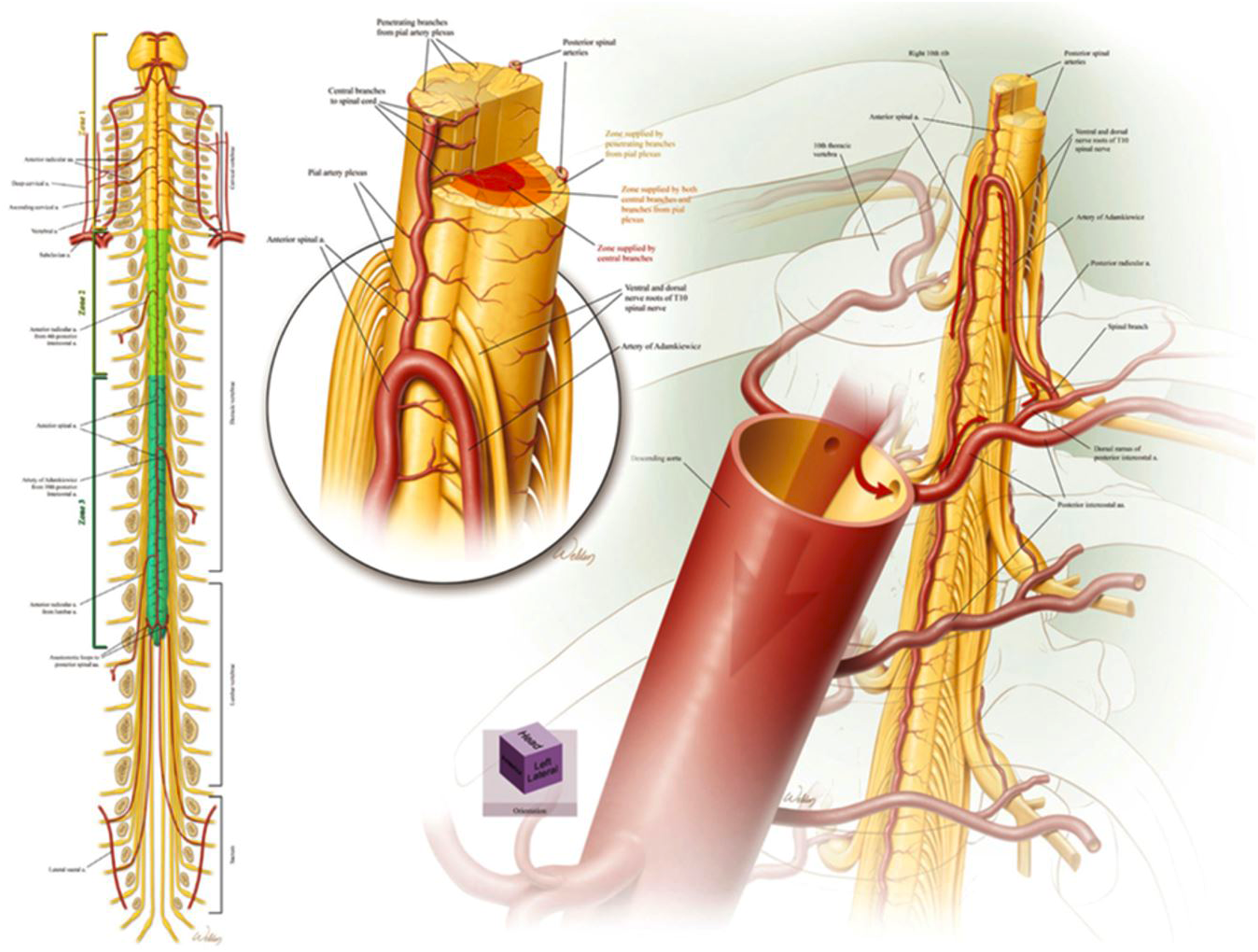
Various neuromonitoring techniques have been utilized to protect the spinal cord during TAAA surgery and prevent ischemia. These include somatosensory-evoked potentials (SSEPs), motor-evoked potentials (MEPs), and near-infrared spectroscopy (NIRS). MEP signals are detected with the use of surface electrodes and are caused by electrical stimulation of the cerebral cortex resulting in muscle contractions. Several considerations must be taken if this modality were to be used. The use of an inhaled agent may interfere with MEP neuromonitoring signals. Muscle relaxant administration typically is ceased after induction and intubation, as the amplitude of signals will be decreased. Deep hypothermia, if utilized, can have significant influence on MEP signal amplitude. Studies have demonstrated that MEP monitoring has very high sensitivity and specificity. 28 One study demonstrated that intraoperative MEP signal loss was a risk for immediate and delayed spinal cord ischemia and that a lower incidence of immediate spinal cord ischemia was observed when MEP neuromonitoring was utilized. 29 SSEP neuromonitoring reflects spinal dorsal column function but is known to have a higher rate of false positivity and delayed detection of spinal cord ischemia. Concomitant monitoring of MEPs is recommended to detect intraoperative changes and help direct anesthetic or surgical care. NIRS is a non-invasive technique to measure oxygenation levels of tissue locally under the optical sensor. The NIRS sensors can be positioned bilaterally at the upper and lower thoracic or lumbar paraspinal region. Anesthetics do not impact NIRS measurements at the level of the spinal cord. Paraspinal NIRS monitoring may have great utility in postoperative monitoring and may be feasible in detecting delayed spinal cord ischemia, but further studies are still required. 30
There are a number of techniques utilized to optimize spinal cord perfusion. First and foremost, according to the 2022 ACC/AHA Guideline for the Diagnosis and Management of Aortic Disease, it is a class I recommendation to place a lumbar CSF drain for Extent I and Extent II thoracoabdominal aneurysm repairs. By decreasing the CSF pressure, we can maximize patients’ spinal cord perfusion pressure (SCPP = MAP - ICP). In patients with severe degenerative disc disease, scoliosis, or arthritis, it may benefit the patient to have the CSF drain placed under fluoroscopy to minimize risk of bleeding or nerve damage. CSF drains may be placed awake pre-induction in either the sitting or lateral positions, or post-induction in the lateral position. We avoid placing CSF drains postoperatively secondary to systemic coagulation derangements which may increase risk of epidural hematoma. We recommend continuously transducing ICP intraoperatively, draining approximately 10cc of CSF per hour as needed to maintain ICP <15 mmHg. A higher rate of CSF drainage may be complicated by subdural hemorrhage. In addition, for Extent I and Extent II repairs, left heart bypass is utilized to allow blood to bypass the proximally located aortic clamp and prevent proximal hypertension, thereby maintaining distal thoracic spinal cord perfusion. In select cases where the distal arch or proximal descending aorta cannot be clamped, deep hypothermic circulatory arrest is used, with anterograde or retrograde cerebral perfusion employed to ensure adequate cerebral oxygenation and protection. 31 From a surgical perspective, the intercostal arteries must be meticulously and thoroughly anastomosed to the new graft to maximize perfusion to the distal descending aorta, 32 which can be achieved through an island graft technique or end to end.
Postoperative Management
Highly specialized and monitored ICU care is of paramount importance within the first 72 hours of open thoracoabdominal aneurysm repair. Immediate goals include a comprehensive neurologic assessment, respiratory assessment, and peripheral vascular assessment. Mean arterial pressure should be maintained between 80 and 90 mmHg to maximize spinal cord, renal, and visceral perfusion, but care must be maintained not to place too much unneeded shear stress on critical suture lines. Although patients may have intact neurologic function, spinal cord ischemia may present several days postoperatively. The exact causes of delayed cord ischemia have not been clearly defined; however, it is thought that postoperative hypotension and a subsequent decrease in CPP may trigger a delay in recovery of spinal cord ischemia within watershed areas. As a result, CSF drains are typically maintained for 72 hrs, aiming for a mean ICP of 10–15 mmHg. In general, we drain a maximum of 10 mL per hour to minimize risk of subdural hematoma. Coagulopathy must be corrected prior to removal of the CSF drain to decrease risk of epidural hematoma.
Pulmonary dysfunction represents the most common postoperative complication, with incidence of up to 20%–40%. This source of morbidity is multifactorial, including prolonged one-lung ventilation time of up to 6–8 hours and intense postoperative pain secondary to a massive incision. Pain control is of utmost importance and can be achieved from a variety of multimodal techniques including opioids; anti-inflammatory medication; ketamine and/or dexmedetomidine infusions; and regional anesthesia including intercostal cryoablation, erector spinae plane blocks, intrathecal opiates, or epidural analgesia. Open TAAA repair can also be complicated by TACO, TRALI, and laryngeal edema secondary to massive transfusion, which may lengthen postoperative intubation, and may require a double lumen tube to remain in situ longer than expected.
Final remarks
In conclusion, the anesthetic management of thoracoabdominal aneurysm repair remains tremendously complex, encompassing virtually every organ system. This procedure has a high intraoperative probability of hemodynamic instability, fluid shifts, and blood loss, requiring intense diligence from the anesthetic team. Furthermore, it is imperative to maintain a high standard of postoperative care in the ICU, as this procedure has unique challenges and complications that must be managed by an experienced multi-disciplinary team. Although the majority of thoracoabdominal aneurysms are now managed through endovascular techniques, open repair still remains the gold-standard among certain patient populations.
Footnotes
Acknowledgments
The authors thank Scott A. Weldon, MA, CMI, FAMI, of the Michael E. DeBakey Department of Surgery at Baylor College of Medicine, for creating several of the illustrations. Mr. Weldon’s work is partly supported by the E. Stanley Crawford Endowment. Additional thanks to Susan Y. Green, MPH, of the Office of Surgical Research, Michael E. DeBakey Department of Surgery at Baylor College of Medicine, for assisting with image selection.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
