Abstract
Thoracic aorta pathologies, especially those of the ascending aorta and aortic arch, were traditionally approached via open surgical repair. This carries risk of ischemic end-organ damage and other complications. Endovascular repair of ascending aorta and aortic arch pathologies is becoming more successful and widespread, thereby posing numerous challenges to the anesthesiologist. This article reviews the anesthesia-pertinent pathophysiology, repair techniques, preoperative evaluation, intraoperative management, and postoperative care of patients presenting for endovascular repair of thoracic aorta pathologies.
Introduction
2022 ACC/AHA Aortic Disease Guidelines (Summarized from Isselbacher, et al). 6

Pathophysiology and Epidemiology
The molecular pathogenesis and biomechanics of aortic disease are complex processes. Aortic wall function is dependent upon the interaction between aortic cells, the extracellular matrix, and various signaling and contractile proteins and their responses to hemodynamic environmental changes.7,8 Genetic mutations such as those associated with connective tissue disease (CTD) or acquired conditions such as hypertension interfere with these components and their adaptive mechanisms, producing death of smooth muscle cells and damage to the extracellular matrix. The loss of normal aortic elasticity, recoil, stiffness, and strength impairs the accommodation of ventricular ejection and pulsatile blood pressure and results in aortic dilation, which may progress to dissection and rupture with catastrophic consequences (Supplemental Figures).9,10
Two principal pathologies of the thoracic aorta carrying significant morbidity and mortality requiring repair are aneurysms and dissections (Figure 1).
11
Aortic aneurysms are traditionally defined as permanent, localized dilations of greater than fifty percent of the expected diameter, although exact numbers vary by age, gender, associated heritable diseases, and other factors.6,12,13 These changes may initially be adaptive responses to injury or stress but eventually become maladaptive and result in increasing wall tension and ultimately rupture.
14
The pooled incidence of thoracic aortic aneurysms (TAA) has been estimated at 5.3/100 000 individuals per year with a prevalence 0.16%, and the pooled incidence of ruptured aneurysms has been estimated at 1.6/100 000 individuals per year; however, the paucity of population-based studies makes this difficult to capture.6,15 Various aortic pathologies, including penetrating aortic ulcer, dissection, intramural hematoma, and aneurysm. Used with permission of Baylor College of Medicine.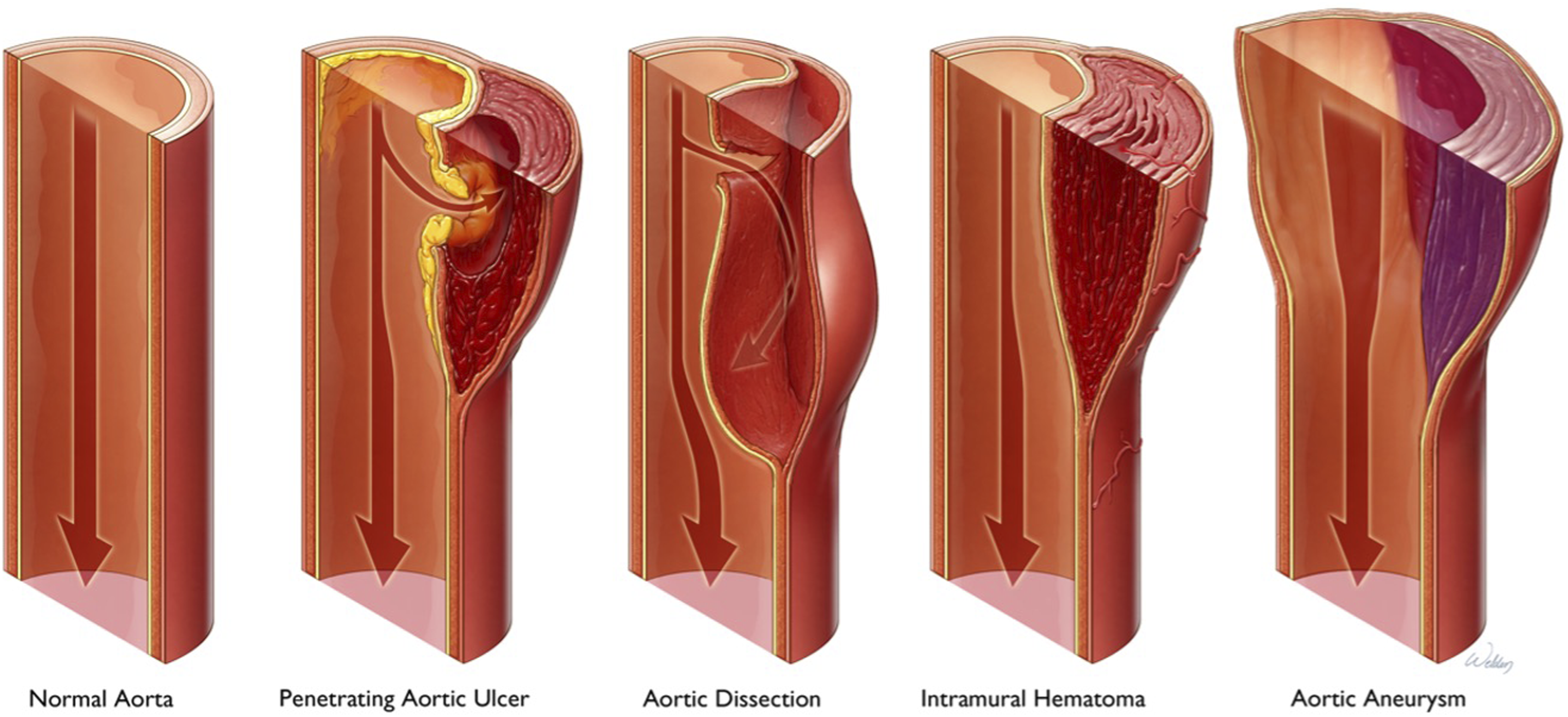
Risk Factors for Thoracic Aortic Aneurysms (TAA) and Dissections (Summarized from Sen, et al). 17
Brief Overview of Surgical Endovascular Techniques
Guidelines
Several societies have published clinical practice guidelines regarding aortic disease. The Society for Thoracic Surgery published guidelines in 2013 regarding ascending aorta management that highlight the significant impact of endovascular aortic repair and the promise of feasibility studies for ascending aortic and arch repair. Also noted is the need for ongoing study of device development and disease process, especially for proximal aortic repairs. 18 The Society for Vascular Surgery published guidelines in 2020 on descending thoracic endovascular aortic repair (TEVAR) but have not yet published regarding endovascular ascending aortic or arch repair. 5 Most recently, the American College of Cardiology/American Heart Association (ACC/AHA) published extensive guidelines in 2022 regarding the diagnosis and management of aortic disease. While there is discussion on management of descending aorta disease and TEVAR, they acknowledge limitations in design and application of current technologies and give limited recommendations for endovascular repair of the ascending aorta or aortic arch. 6
Purpose and Aortic Zones
The purpose of endovascular repair is to functionally exclude a lesion from circulation with a stent graft that is proximally and distally anchored to non-diseased tissue.
6
The aorta is divided into eleven anatomic segments or zones (Figure 2). The thoracic aorta includes zone 0 (beginning at the aortic root) through zone 5 (ending at the distal descending aorta at the level of the diaphragm).
19
The identification of a sufficiently long landing zone (as dictated by the patient’s pathology, aortic diameter, and type of stent graft) is critical to avoid inadvertent arterial take-off occlusion, stent migration, stent collapse, and endoleaks.
20
Landing zones of the thoracic aorta, including zone 0 (aortic root to just beyond the innominate artery), zone 1 (innominate artery to just beyond the left common carotid artery), zone 2 (left common carotid artery to just beyond the left subclavian artery), zone 3 (first two centimeters of the descending aorta distal to the left subclavian artery), and zone 4 (end of zone 3 to mid-descending aorta at approximately T6). The green segments delineate the aortic arch; proximal to the arch is the ascending aorta and distal to the arch is the descending aorta 14 used with permission of Baylor College of Medicine.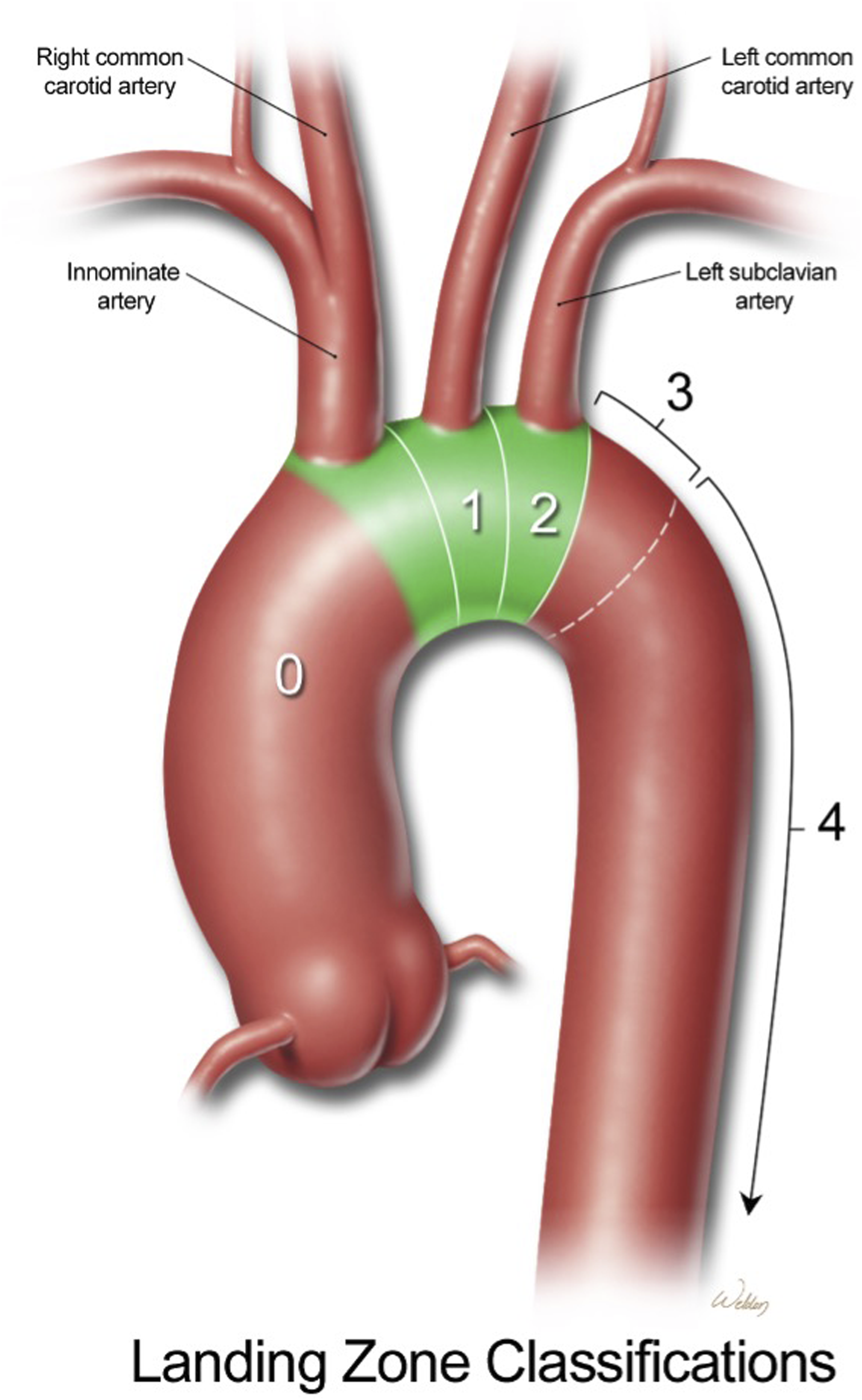
Open vs Endovascular Repair
While many patients who present for thoracic aorta repair are at high-risk for poor surgical outcomes, some patients may be candidates for both open and endovascular approaches. Endovascular repair is preferred for elective repair of descending thoracic aneurysms and emergent repair of descending thoracic ruptures due to decreased morbidity, hospital length-of-stay, and short-term mortality. 5 Notably, survival at 5 years is significantly lower in endovascular repair than open repair. 6
American College of Cardiology/AHA guidelines give several recommendations for open vs endovascular repairs of various aortic zones and pathologies previously described (Table 1). Recommendations are graded by both class (or strength) and level (or quality) of evidence. Important to note is the lack of high-quality evidence in most categories. 6 In the absence of guidelines, consideration must be given to the patient’s age, co-morbidities, life expectancy, and desired quality of life as well as repair feasibility, repair durability, surgeon experience, and institutional practice.
Total Endovascular Surgical Approaches
Endovascular surgical repair includes approaches that are entirely minimally invasive and hybrid repairs that have components of open surgical repair. Total endovascular surgical approaches may involve parallel stent grafts (i.e., multiple stents placed side by side), in situ fenestrations (i.e., creating openings in the stent graft during the procedure), and custom-made devices (e.g., scalloped, fenestrated, or branched stent grafts specifically designed for the patient’s anatomy). 21 Devices are placed via large-bore delivery sheaths, typically via the femoral arteries, which may be obtained with a surgical cutdown or percutaneous access. Other access sites include carotid and axillary arteries and the left ventricular apex.
There are several considerations for endovascular techniques. Adequate seal zones of non-diseased aortic tissue adjacent to the lesion (>25 mm for most endovascular repairs) accounts for potential disease progression, aortic growth, and aortic remodeling. 22 Durability of the device is important given the rapid rate of change in pressure over change in time (dP/dT) during systole (particularly in the aortic arch), which may predispose a weaker graft to more wear, fracture, and kinking and thereby compromise device performance. 23 Proper alignment and deployment of the device is dependent on high resolution imaging, which helps minimize graft manipulation in diseased segments and reduce the risk of embolic stroke, device migration, and device misalignment (the last of which could result in branch artery coverage, stent branch misalignment, and fenestration misalignment). Finally, special attention must be paid to the aortic valve as stent positioning may require crossing the aortic valve several times with guidewires and exposing the valve to potential injury. For this reason, mechanical or tissue aortic valves are considered contraindications to procedures requiring such guidewire manipulation such as endovascular aortic arch repair. In the event of difficult guidewire passage through a tortuous aortic arch, transseptal puncture via the foramen ovale or left ventricular apical access with subsequent navigation of the guidewire through the ventricle and into the aorta under transesophageal echo (TEE) guidance may be considered. 24
Hybrid and Debranching Procedures
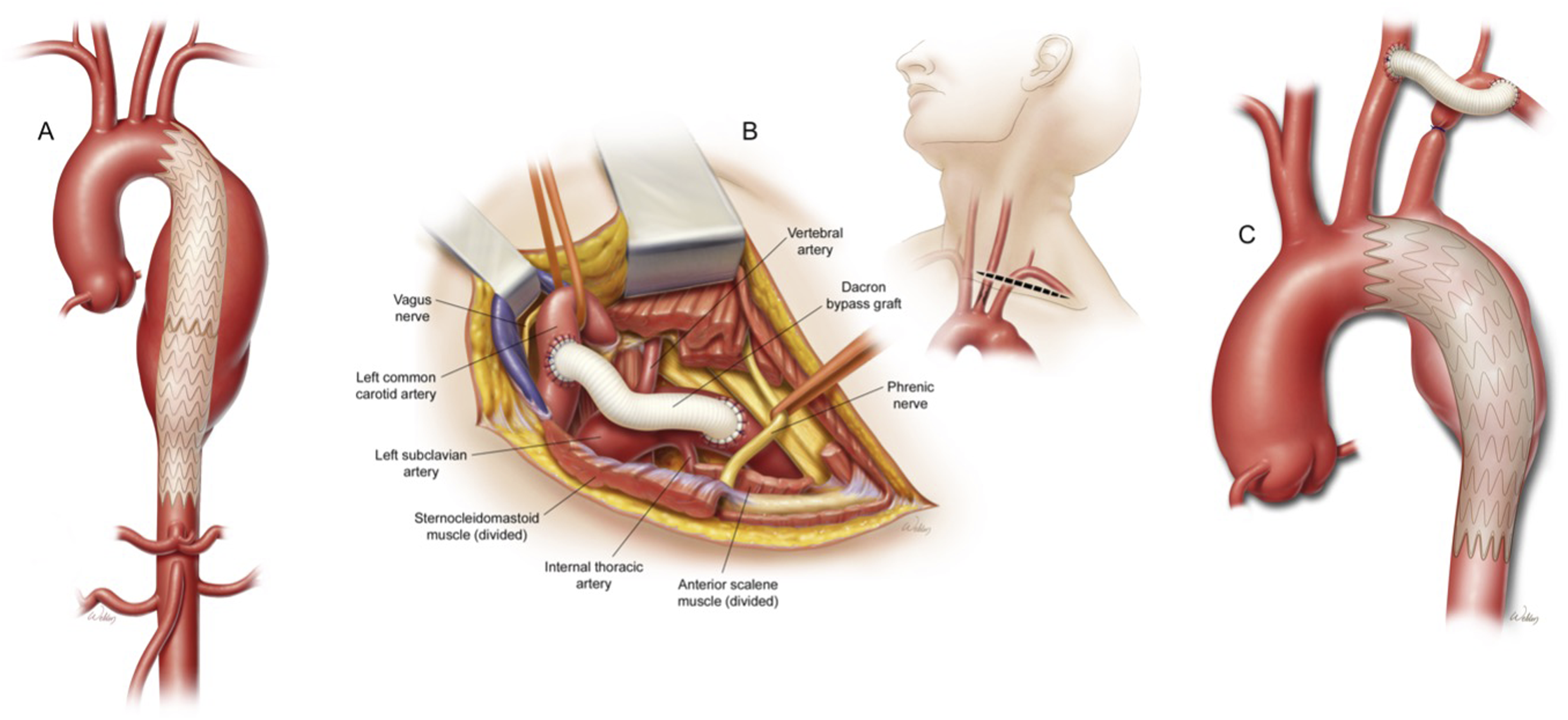
Endovascular management of arterial take-off branches is often difficult and may require some degree of open management. Hybrid repairs combine open surgical exposure and large artery management as needed with endovascular graft placement and are often staged. Debranching procedures serve to ligate and re-route arterial take-offs from the main circulation to maintain perfusion, prevent competitive flow, and prevent retrograde leaks (Figure 3). For example, coverage of the left subclavian artery with descending thoracic grafts in patients with left-dominant vertebral artery circulation or prior left internal mammary artery harvest may result in cerebral or myocardial ischemia. This may be addressed with a left common carotid artery to left subclavian artery bypass graft via a supraclavicular incision prior to endovascular graft placement (Figure 4).
5
American College of Cardiology/AHA guidelines recommend pre-emptive revascularization of the left subclavian artery in patients undergoing descending TEVAR with planned left subclavian artery coverage to prevent SCI and possibly decrease stroke risk (class 1 recommendation, level B-NR).
6
In descending TEVAR patients who develop SCI refractory to blood pressure augmentation and cerebrospinal fluid (CSF) drain placement, left subclavian artery revascularization may be an option (class 2b recommendation, level C-LD).
6
Similarly, a left to right common carotid artery bypass may be performed to extend the proximal seal zone for a descending thoracic graft.
5
Open total debranching via median sternotomy may also be performed.
25
An important complication of open debranching procedures is the risk of aortic dissection from placement of the side-biting aortic clamp.
26
Alternatives to open left subclavian artery bypass include endovascular placement of thoracic branch endoprostheses.
27
Endovascular repair is typically accomplished with retrograde stent deployment through a large-bore delivery system placed in the femoral or iliac arteries. However, this might be difficult or impossible in patients with severe peripheral vascular disease or inadequate stretches of disease-free tissue needed for graft fixation. Alternative approaches are antegrade stent deployment via subclavicular or axillary approaches, mini-sternotomy, or sternotomy (as part of a debranching procedure) into the aorta under direct visualization or over a guidewire with fluoroscopic and/or TEE confirmation.
28
(A) Aortic arch and descending aortic aneurysm; (B) Hybrid repair with surgical debranching of the head vessels and antegrade deployment of descending endograft stent; (C) Completed hybrid repair with deployed endograft stent. Used with permission of Baylor College of Medicine. (A) Descending thoracic aortic aneurysm status post stent graft without coverage of the head vessels; (B) Left common carotid to left subclavian artery bypass graft; (C) Descending thoracic aortic aneurysm status post stent graft with left subclavian artery coverage and left common carotid to left subclavian artery bypass graft. Used with permission of Baylor College of Medicine.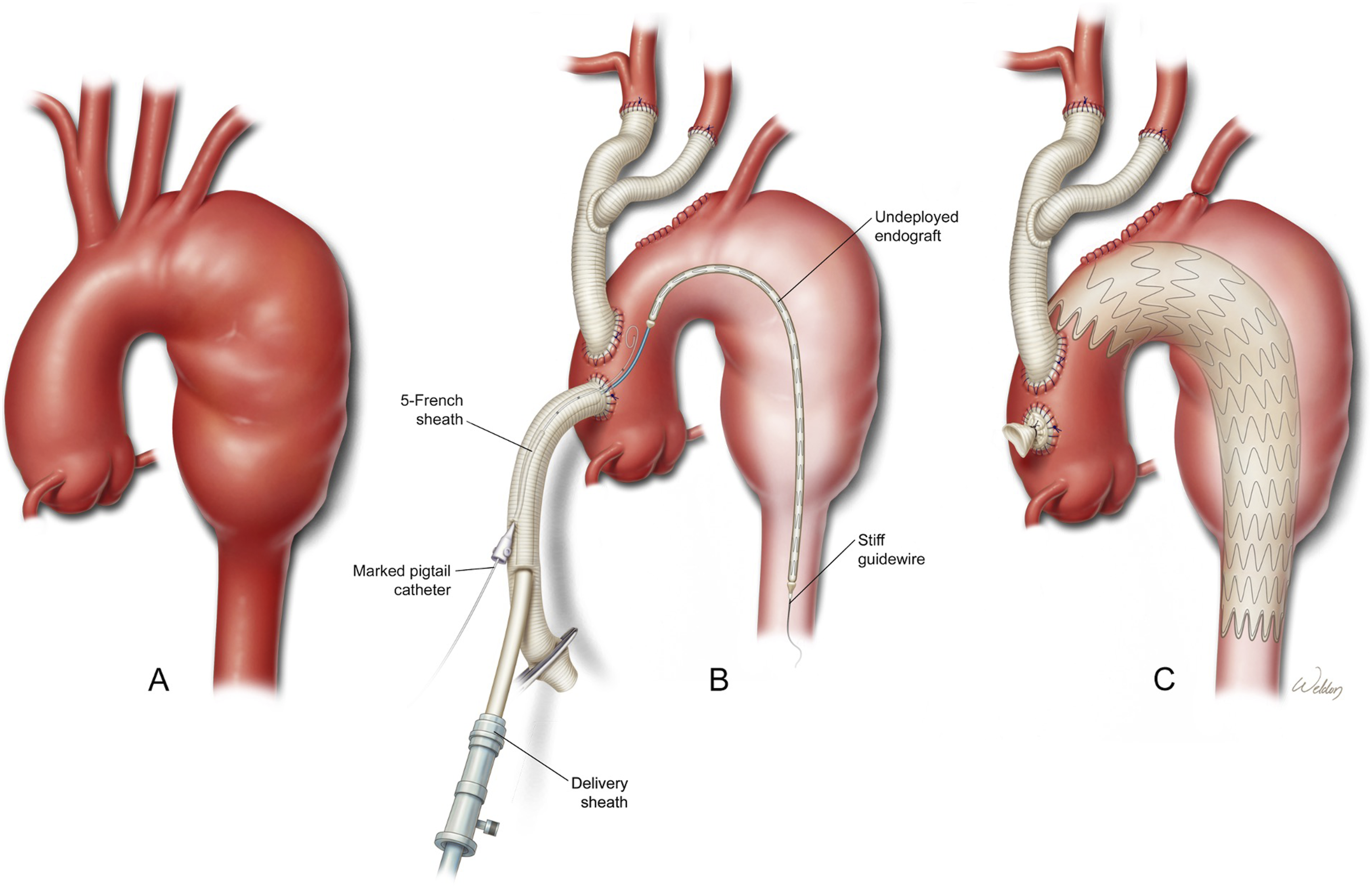
Staged Procedures
Endovascular repair of the descending aorta may be performed as a second stage procedure after open repair of ascending and aortic arch pathologies that extend distally resulting in malperfusion syndromes. In one recent case series in patients with type A ascending aortic dissections, performing a descending TEVAR approximately one month after the central open surgery resulted in better resorption of false lumen thromboses, smaller false lumens, larger true lumens, effective treatment of malperfusion syndromes, and possibly less risk of SCI than if the procedures were performed as a single stage. 29 Hybrid repair of aortic arch disease has been shown to have similar short- and medium-term outcomes as open surgical repair with fewer complications associated with long cardiopulmonary bypass and hypothermic circulatory arrest times.30,31 However, open surgical appears to have superior long-term outcomes according to the current data. As with total endovascular techniques, hybrid repairs appear to most benefit patients at high risk for poor outcomes with open repair.30,31
Ascending Aorta Repair
Two principal indications for endovascular repair of the ascending aorta are aneurysms (frequently in the setting of CTD, bicuspid or unicuspid aortic valves with resultant dilation, aging, and atherosclerosis) and dissections (in the setting of hypertension and a pre-existing aneurysm) (Figure 5). Other indications include aortic ruptures, intramural hematomas, and penetrating atherosclerotic ulcers.
1
(A) Development of an ascending aortic pseudoaneurysm from a prior cannulation site; (B) Completed repair with endograft stent. Used with permission of Baylor College of Medicine.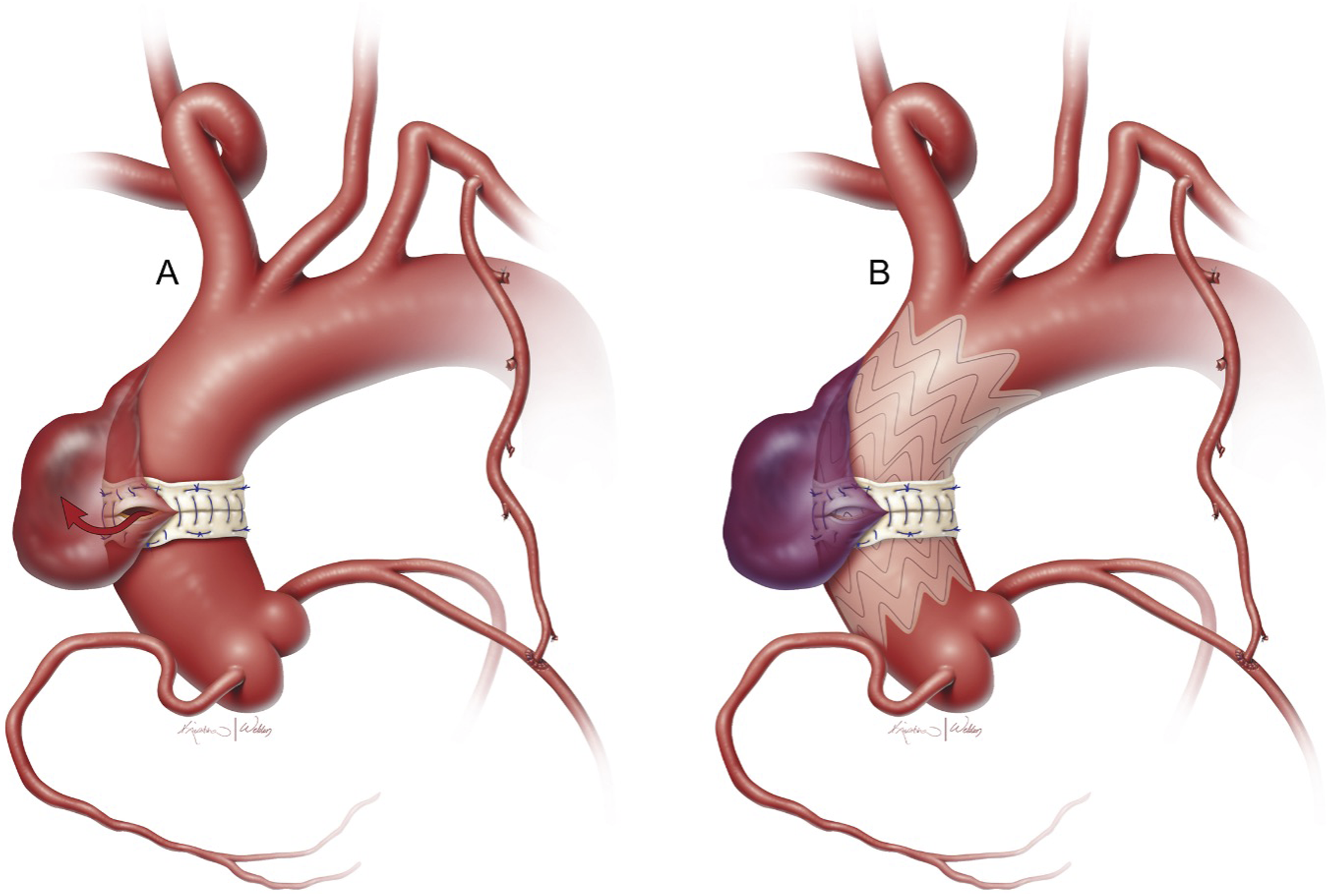
Endovascular repair of the ascending aorta presents numerous challenges. First is the selection of graft type. Currently available grafts include single branch and multi branch devices that were designed for the aortic arch or descending aorta but are used in the ascending aorta. A number of manufacturers including Cook, Relay, and Gore, have created grafts specific to the ascending aorta and are in various stages of development for approved use in the United States. 1 Thoracic endovascular and abdominal aortic cuffs and otherwise unspecified or off-label grafts have been utilized in the ascending aorta. In addition to custom-made grafts (which may be difficult to obtain in an emergency), surgeons may opt to alter pre-existing grafts for their patients. 4 The geometry of the inner and outer aortic curvatures is difficult to replicate with current prosthetic grafts, and many of the grafts currently available were created for the descending aorta and are too long for the ascending aorta.1,4 Replacing a section of the ascending aorta with prosthetic graft creates a compliance mismatch as well as altered sheer stress at the junction of the graft and native tissue, which may increase the risk of dissection propagation. As with other endovascular grafts, there must be an adequate landing zone proximally and distally to the diseased tissue so as to not occlude important arterial take-offs and risk distal ischemia in the absence of collateral blood supply (e.g., coronary artery coverage causing myocardial ischemia or brachiocephalic coverage causing cerebral ischemia) or to otherwise obstruct forward blood flow and decrease cardiac output (e.g., interference with aortic valve causing functional stenosis or regurgitation). Considering the complexities of potential coronary and valvular involvement in such pathologies, endovascular repair of the ascending aorta is currently limited to disease distal to the sino-tubular junction. 1 Therefore, patients presenting emergently with concomitant tamponade, aortic valvulopathy, or coronary ischemia currently pose a significant challenge to endovascular repair. 25 Finally, as with any endovascular procedure, achieving vascular access suitable for delivery sheath placement and graft deployment in this population may be difficult. 4
Preliminary successes and drawbacks with endovascular repair of ascending aortic lesions have been documented in various studies. For example, the prospective, multi-center ARISE feasibility trial tested a novel stent graft specifically designed for ascending aortic dissections with encouraging results. 32 Phase II trials are underway for an ascending aortic graft manufactured by Gore. 33 Additional studies and reviews have demonstrated feasibility and acceptable outcomes of such repairs.4,34-36
Aortic Arch Repair
Anatomic and Physiologic Criteria for Endovascular Aortic Arch Repair (Summarized from Haulon, et al). 41


Aortic arch branched endovascular stent grafts. Left, Nexus Aortic Arch Stent Graft System with integrated innominate artery branch by Endospan (image courtesy of Endospan). Right, TAG Thoracic Branch Endoprosthesis with left subclavian artery branch by Gore (image courtesy of Gore Medical).
Descending Aorta Repair
Much has been written about descending TEVAR and there are many reviews available for reference.42,43 Indications for endovascular repair of the descending aorta (including the thoracoabdominal aorta) include aneurysms (fusiform or saccular), type B dissections, blunt traumatic injuries, and infectious etiologies, though they are less common.5,42 Risk factors for aneurysms and dissections include old age, hypertension, tobacco use, CTD, and family history of aortic aneurysms. 44 Several manufactured grafts exist, including those made by Gore, Medtronic, Cook, and Bolton. 20 A significant challenge is management of branch arteries (e.g., left subclavian, celiac, superior mesenteric, renal arteries) due to the primary disease process or graft coverage. Patients undergoing descending TEVAR with planned left subclavian artery coverage are at increased risk of stroke and spinal cord ischemia (SCI), and preoperative revascularization is recommended to decrease these risks and prevent other ischemic complications (e.g., coverage of patent left internal mammary graft, left arm dialysis access). As previously mentioned, obtaining vascular access in these patients may be challenging and require additional instrumentation. 6
Preoperative Evaluation
Patients presenting for thoracic aorta endovascular repair tend to be critically ill with multiple co-morbidities. Those with acute or acute-on-chronic aortic disease may present with hemodynamic instability or shock due to pericardial effusion, tamponade, acute aortic valve insufficiency, or coronary artery dissection as well as altered mental status or stroke symptoms due to carotid or vertebral artery dissection. These acute sequelae currently limit endovascular repair of the ascending aorta and aortic arch. Mesenteric and renal ischemia are also possible depending on the extent of the pathology. Patients may present with dyspnea, wheezing, hoarseness, hypoxemia, or respiratory distress from recurrently laryngeal nerve compression, tracheal compression, or pulmonary hemorrhage. Those with chronic aortic disease may have CTD and tend present at younger ages. Older patients may have coronary artery disease, heart failure, chronic respiratory or renal disease, diabetes, or neurologic or cognitive deficits. For those patients not presenting urgently or emergently and at high risk of obstructive coronary artery disease, preoperative evaluation by a cardiologist including possible coronary angiography and/or revascularization may be necessary per ACC/AHA guidelines.44,45 Ganapathi et al 46 investigated the role of preoperative cardiac testing in this arena noted a low risk (∼2.4%) of postoperative adverse cardiac events after descending TEVAR and suggested that assessment of the patient’s symptoms along with resting electrocardiogram or transthoracic echocardiogram was appropriate for initial screening for most of these patients. Given the proximal location of ascending and aortic arch endovascular repairs, preoperative evaluation is similar to that for patients undergoing open repair in the absence of dedicated guidelines. Therefore, preoperative workup should include laboratory testing (e.g., complete blood count, complete metabolic panel, coagulation tests, arterial blood gas and lactate, blood type and cross) and imaging as needed for cardiac screening and surgical planning (e.g., EKG, echocardiogram, chest radiograph, computed tomography or magnetic resonance imaging of the chest and major vessels, and carotid ultrasound).4,42 Fortunately, much of the required preanesthetic evaluation is accomplished with the labwork and imaging necessary to determine the appropriate surgical course, including evaluation of ventricular and valvular function, coronary anatomy, and aortic anatomy. A baseline neurologic exam, including movement of bilateral lower extremities, is important to guide postoperative assessment of neurologic function.
Anesthetic Management
Lines and Monitors
Thoracic endovascular repair procedures have unique perioperative considerations to optimize outcomes and avoid specific complications. In addition to standard monitors, these procedures require at least one arterial line for invasive beat-to-beat blood pressure monitoring (usually an upper extremity or femoral), although multiple arterial lines may be justified depending on endograft and procedural arterial access locations. Central venous pressure monitoring may assist in determining volume status in conjunction with TEE. Insertion of a pulmonary artery catheter or other cardiac output monitoring may be advantageous in patients with pulmonary hypertension or reduced ventricular function. Large-bore peripheral intravenous lines and/or central venous access for volume resuscitation and vasoactive medication infusion are needed. As with arterial lines, venous line location may need to be modified depending on procedural access. 47 Cerebral oximetry may help identify coverage of major arterial take-offs if using grafts without fenestrations or collateral blood flow. 48
Intraoperative TEE may be advantageous for several reasons, including continuous monitoring (e.g., volume status, ventricular function, valvular function) and pathology characterization (e.g., extent of dissection flaps, true and false lumens, involvement of aortic valve, branch arteries, and other critical structures). Additionally, it may supplement the use of fluoroscopy and provide procedural assistance (e.g., guidewire manipulation, device advancement), detection of procedural complications (e.g., iatrogenic/retrograde aortic dissection from graft deployment, cardiac tamponade, endoleaks, valve injury, ventricular perforation), and inform management in the event of hemodynamic decompensation.49,50
Certain endovascular repairs have unique considerations. In ascending and aortic arch repairs, it may be advantageous to induce hypotension with rapid ventricular pacing during balloon expansion of the endograft stent, especially if multiple ballooning episodes may be needed. While there are no guidelines regarding this practice, a recent study demonstrated safety and efficacy of this practice. 51 Surgical and anesthetic experience with rapid pacing has grown tremendously, as this technique has been utilized in endovascular aortic valve replacement. Transvenous pacemaker placement may be performed by the surgeon or anesthesiologist depending on institutional preference. For aortic arch procedures, European guidelines suggest multiple arterial lines in the upper and lower extremities with one dedicated to anesthetic monitoring. 52 Furthermore, cerebral oximetry seems reasonable for neuromonitoring, and use of somatosensory evoked potentials (SSEPs) and motor evoked potentials (MEPs) must be based on patient and institutional factors. 52 For descending TEVAR, bilateral radial arterial lines and cerebral oximetry are useful to detect possible left subclavian artery coverage. Acute changes in SSEPs, MEPs, and near-infrared spectroscopy (NIRS) trends may help indicate risk of spinal cord ischemia (SCI) and allow for maneuvers to improve spinal cord perfusion.53,54 Placement of CSF drains in descending TEVAR may be considered for monitoring via transduction of CSF pressure as well as end-organ protection via drainage of spinal fluid to optimize spinal cord perfusion pressure (SCPP) (see “End-organ Protection” section for further discussion). 6 Ultimately preoperative discussion with the surgeon is very helpful in determining the anesthetic plan.
Anesthetic Type
Anesthetic type for endovascular repair is largely based on patient factors and institutional practice given lack of high-quality comparative data. 5 General anesthesia is often preferred for the benefits of a secured airway, controlled ventilation (especially for planned breath holds, possible acid-base disturbance, and minimization of motion artifact), facilitation of TEE probe placement, patient immobility, and potential conversion to open repair.44,47 If SSEPs and MEPs are performed, then maintenance of general anesthesia may require a primarily intravenous technique given the known effects of inhaled anesthetics on evoked potentials. 55 Barring procedural complications or cardiopulmonary instability, general anesthesia should be titrated with short-agent agents to facilitate prompt emergence and extubation in the operating room (known as “fast-tracking”) to perform a neurological exam, especially in those patients at high risk of complications. 47 One recent study noted that “fast-track” general anesthesia for descending TEVAR was safe and decreased ICU and hospital length of stay. 47 Local anesthetic infiltration has been successfully utilized as the primary anesthetic with patient and surgeon cooperation and allows for direct neuromonitoring via physical exam. 56
End-organ Protection
Protection of end-organs by maintaining perfusion is of great importance during endovascular repair. One of the most feared complications of endovascular repair is SCI due to inadequate SCPP (mean arterial pressure [MAP] – CSF pressure or CVP, whichever is greater). 6 Spinal cord ischemia with descending TEVAR results from coverage of the left subclavian and thoracic aortic take-offs and placement of large-bore iliofemoral sheaths. Older patients with hypertension, chronic obstructive, pulmonary disease, chronic renal disease, and those presenting with concomitant abdominal aortic aneurysms are at increased risk. 42 Per ACC/AHA guidelines, interventions to optimize spinal cord perfusion pressure and therefore mitigate the risk of SCI include volume resuscitation, maintenance of serum hemoglobin > 10 g/dL and MAP > 100 mmHg, cardioversion for tachyarrhythmias, and insertion of a CSF drain. 6 Increased MAP and CSF drainage are often considered the two most important interventions to reduce the risk of SCI, although evidence for routine prophylactic placement for all descending TEVARs is lacking.57,58 Drawing from open thoracoabdominal repair literature, indications for CSF drain insertion in patients at high risk of SCI include those requiring more than 20 cm of aortic coverage or coverage of the distal third of the thoracic aorta, prior abdominal aortic aneurysm repair, and occlusion of the left subclavian or internal iliac arteries, among others. 57 Additional considerations concerning placement include abnormal coagulation studies or systemic anticoagulation that would increase the risk of neuraxial hemorrhage and hematoma, pre-existing infection, and prior back surgeries. 59 A common management approach is to drain CSF in 10 mL increments to maintain a CSF pressure of 10–15 mmHg with close monitoring of neurologic exams post-procedurally to determine appropriate timing for removal. 57 Notably, recent evidence has suggested that routine prophylactic CSF drains may not be necessary in low-risk patients undergoing fenestrated-branched endovascular repair in the aortic arch and descending aorta. 60
As mentioned, trends in SSEPs, MEPs and NIRS provide insight into spinal cord perfusion and acute changes from baseline, particularly with vessel cannulation or endograft deployment, may indicate immediate SCI and can be used to direct interventions to improve perfusion and potentially avoid permanent defects.54,61 A recent large review indicated that acute changes in SSEPs and MEPs intraoperatively during TEVAR had a 93% sensitivity and 96% specificity for risk of SCI (for example, insertion of venous sheath with delayed drops in MEPs that resolve with removal of the sheath). However, whether detection of these changes was linked to fewer incidences of lower extremity weakness was not necessarily demonstrated. The authors did highlight operative time and extent of aortic coverage as risk factors for SCI. 54 Perioperative cerebral protection may be best served by avoiding drastic changes in cerebral perfusion and utilizing neuromonitoring techniques as previously mentioned. It is known that patients with prior strokes and chronic renal disease undergoing descending TEVAR with left subclavian artery coverage are at higher risk of stroke due to coverage of a dominant vertebral artery, and close monitoring of neurologic exams is important postoperatively. 42
Perioperative cardiovascular protection includes maintenance of sufficient preload, afterload, rate, rhythm, and contractility to maintain cardiac output with utilization of cardiac point of care ultrasound or TEE for guidance as needed. Perioperative pulmonary protection includes lung-protective ventilation strategies if intubated, early extubation, non-invasive positive pressure ventilation, and pulmonary toilet. 42 Perioperative renal protection includes avoidance of new or worsening renal injury with adequate hydration in anticipation of contrast administration, avoidance of nephrotoxins, and judicious continuation of angiotensin-converting enzyme inhibitors and angiotensin receptor blockers.42,44 Important to consider in all endovascular repairs is anticoagulation. Heparin or another anticoagulant is administered in anticipation of aortic manipulation, is titrated to an activated clotting time of approximately 250–300 s (less than required for institution of cardiopulmonary bypass) and may be reversed with successful procedure completion. Risks of partial or full anticoagulation reversal must be balanced with patient-specific clotting risk factors and merits discussion with surgeons.
Complications
Anesthesiologists should be aware of complications of endovascular repair as they may affect nearly all major organs. 42 Major neurologic complications include cerebral and spinal cord ischemia. Perioperative stroke incidence in TEVAR patients ranges from 1.2–8.2% and may result from coverage of carotid and/or vertebral arteries, dislodgement and embolization of existing plaques, retrograde dissection, and decreases in global cerebral perfusion.25,62 Serial neurologic exams should be performed postoperatively. In the event of a focal or global deficit, diagnostic imaging in conjunction specialty consultation dictates management. 42 The incidence of SCI has been estimated at from 3.4% in descending TEVAR vs 8.2% after open aortic repair.42,43 Early postoperative detection with serial neurologic exams focusing on lower extremity movement are of utmost importance. Establishment of postoperative SCI management protocols may assist in swift initiation of treatment. One treatment algorithm consists of volume resuscitation targeting a CVP 10–12 mmHg and serum hemoglobin of 9.5–10.5 g/dL, blood pressure augmentation with vasopressors as needed to MAP 90–100 mmHg (or 100–110 mmHg in severe cases), and CSF drainage to achieve a CSF pressure 10–12 mmHg with drainage of up to 15 mL per hour or 50 mL over four hours. Pharmacologic adjuvants including scheduled mannitol and dexamethasone and bolus doses of lidocaine, and magnesium sulfate are also administered with close monitoring of serum electrolytes, glucose, urine output, and volume status. 42
Conclusion
The feasibility and success of endovascular repairs of the thoracic aorta continue to improve. American College of Cardiology/AHA guidelines recommend endovascular repair for certain pathologies (e.g., descending thoracic aneurysm without CTD) and recommend consideration of endovascular repair for select proximal aortic pathologies (e.g., high-risk patients with asymptomatic arch aneurysms). 6 Endovascular repair has several considerations of great importance to anesthesiologists. Preoperative assessment includes evaluation of the patient’s co-morbidities (especially cardiac, respiratory, and neurological) and thorough review of imaging. Preoperative discussion with surgeons and familiarity with the various endografts and surgical approaches may assist in determining intraoperative line and monitor placement. Knowledge of common complications of these procedures may help reduce the risk of adverse events after surgery. Endovascular repair of certain descending thoracic aortic pathologies has shown to have improved morbidity, short-term mortality, and hospital length-of-stay compared to open repair. 5 Whereas preliminary data shows favorable outcomes in certain patients undergoing endovascular ascending and aortic repair, additional studies are needed to further identify patients and clinical scenarios that would most benefit from these newer techniques. In closing, a multidisciplinary approach with close communication and coordination with surgeons and intensivists is essential in optimal perioperative care for patients presenting for endovascular repair of thoracic aortic disease.
Supplemental Material
Supplemental Material - Anesthetic Considerations for Endovascular Repair of the Thoracic Aorta
Supplemental Material for Anesthetic Considerations for Endovascular Repair of the Thoracic Aorta by Olivia M. Valencia, Thomas Powell, Ali Khalifa, Vicente Orozco-Sevilla, and Daniel A. Tolpin in Seminars in Cardiothoracic and Vascular Anesthesia.
Footnotes
Acknowledgments
The authors thank Scott A. Weldon, MA, CMI, FAMI, of the Michael E. DeBakey Department of Surgery at Baylor College of Medicine, for creating several of the illustrations. Mr. Weldon’s work is partly supported by the E. Stanley Crawford Endowment. Additional thanks to Susan Y. Green, MPH, of the Office of Surgical Research, Michael E. DeBakey Department of Surgery at Baylor College of Medicine, for assisting with image selection.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Valencia declares no conflicting interests. Dr Powell declares no conflicting interests. Dr Khalifa declares no conflicting interests. Dr Orozco-Sevilla consults and participates in clinical trials for Terumo Aortic, Cook Medical, and W.L. Gore & Associates. Dr Tolpin declares no conflicting interests.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

