Abstract
Since their initial approval by the Food and Drug Administration in 2016, leadless pacemakers have become increasingly prevalent. This growth has been driven by an improved adverse effect profile when compared to traditional pacemakers, including lower rates of infection, as well as eliminated risk of pocket hematoma and lead complications. More recently, technology enabling leadless synchronized atrioventricular pacing in patients with atrioventricular block has vastly expanded the indications for these devices. Anesthesiologists will increasingly be relied upon to safely care for patients with leadless pacemakers undergoing non-electrophysiology procedures and surgery. This article provides an overview of the technology, evidence base, current indications, and unique perioperative considerations for leadless pacemakers.
Introduction
Despite increased sophistication in programming parameters and capabilities, the basic design of permanent pacemakers has varied little since the first implantation in 1958. 1 Food and Drug Administration (FDA) approval of the first leadless pacemaker in 2016 heralded a major break with this initial design, with a capsule-sized device incorporating generator, battery, and electrode system in a single unit. 2 With an improved side effect profile over traditional pacemakers and expanding capabilities for atrioventricular synchrony, these exciting advances in pacemaker technology provide unique challenges for anesthesia providers.3,4 While data for the safety of these devices continues to grow, societal guidelines for perioperative care have yet to be developed.5,6 We present a review of the current literature to provide the best opportunity to safely care for patients with leadless pacemakers in the perioperative period.
Leadless Pacemaker Overview
Historical Context
In 1958, Swedish surgeon Dr. Ake Senning implanted the world’s first permanent pacemaker into a 43-year-old man suffering from high grade atrioventricular block and refractory syncope. Senning implanted a generator into the patient’s abdominal wall and tunneled 2 leads directly into the myocardium via left thoracotomy. Despite requiring 5 lead repairs and 22 generator exchanges, the device remained functional and allowed the patient to survive to the age of 86. 7 Modern pacemakers have advanced considerably since Senning’s sentinel placement. Smaller generators with complex pacing algorithms are now placed in the subcutaneous chest tissue and are connected to the heart with transvenous leads. 8 Despite the increased sophistication in programming, however, Senning’s fundamental pacemaker design—a pulse generator connected to the heart with leads—has persisted for over 60 years. 1 Accordingly, the introduction of the leadless pacemaker in 2016 represents a disruptive breakthrough in pacemaker technology with immediate clinical consequences for patients undergoing anesthesia.
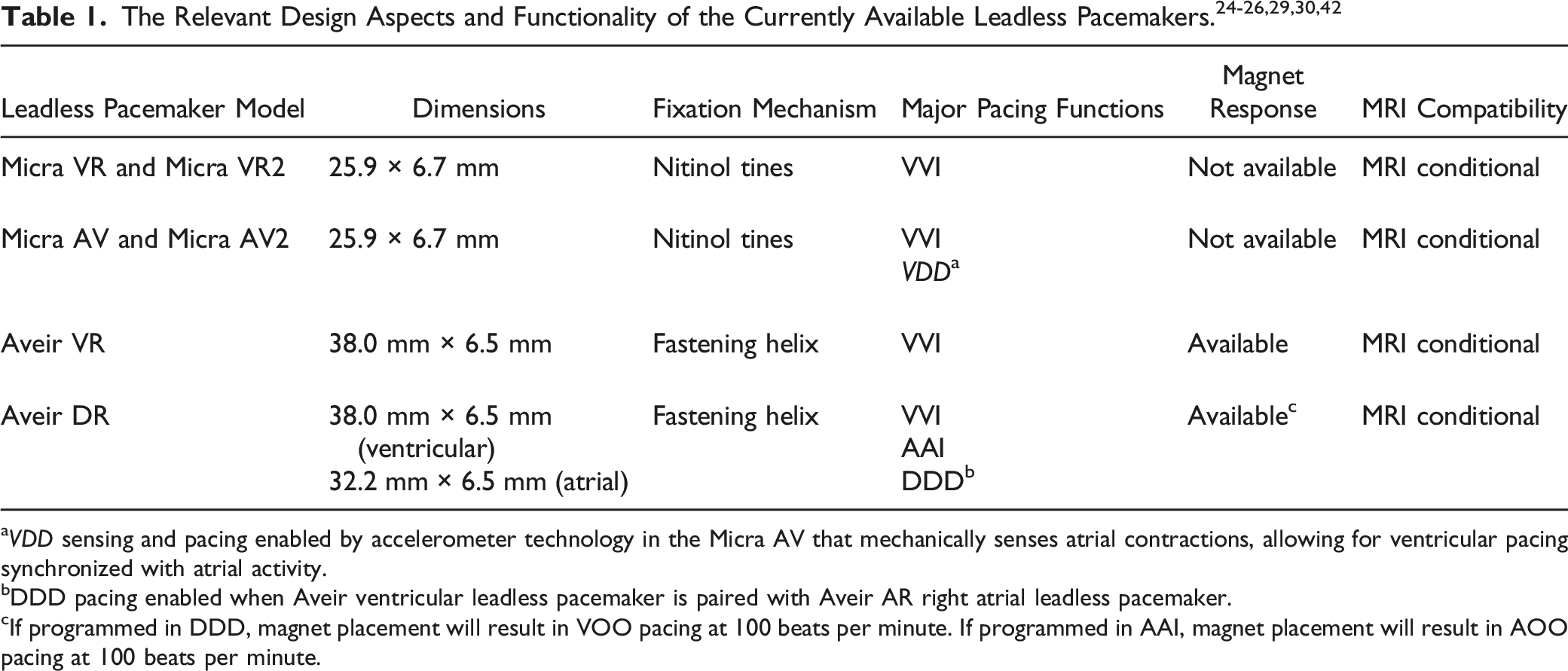
Leadless Pacemaker Device Overview
Leadless pacemakers are capsule-sized devices that contain a generator, battery, and electrode system in a single, self-contained pacing unit. They are implanted percutaneously into the septal wall of the right ventricle, typically through the right femoral vein. 9 The device is permanently affixed to the right ventricular myocardium using either tines or a fastening helix. In this secured position, an electrode is placed in direct contact with the myocardium, obviating the need for traditional pacemaker leads. The leadless pacemaker provides both ventricular sensing and pacing capabilities. Additionally, the most recent leadless pacemaker versions also incorporate atrial electrical activity. The estimated leadless pacemaker battery life is approximately 5–15 years, comparable to that of a traditional transvenous pacemaker. 10 Differences in pacing burden account for variability in battery duration, with more pacer-dependent patients experiencing a shorter battery life.
By nature of their design, leadless pacemakers have an improved long-term side effect profile as compared to traditional transvenous pacemakers.6,11 The devices have a reduced risk of infection and eliminate the possibility of lead-related adverse effects, including fracture, tricuspid regurgitation, and thoracic vessel stenosis.6,12,13 However, leadless pacemakers also have inherent limitations related to their novel design. Prior to the release of the first dual chamber leadless pacemaker in 2023, leadless pacemakers were limited to single chamber ventricular pacing. 4 Additionally, leadless pacemakers cannot perform cardiac resynchronization therapy (CRT) or conduction system pacing to promote ventricular synchrony.14,15 Finally, unlike implantable cardioverter-defibrillators (ICD’s) that also perform transvenous pacing, leadless pacemakers have no cardioverter-defibrillator or anti-tachycardia pacing function.
Implantation
Leadless pacemakers are percutaneously implanted by electrophysiologists in a fluoroscopy procedure suite. The vast majority are implanted using local anesthesia, sedation, or a combination of the 2 techniques. 16 The right femoral vein is the preferred leadless pacemaker insertion site due to its straight path to the inferior vena cava and ability to accommodate the large introducer sheath. 17 The electrophysiologist uses fluoroscopic guidance to advance the device through the introducer sheath toward the right ventricular septum. After implanting the device in the myocardium, the electrophysiologist tests the pacing and sensing function. To ensure stable device implantation, gentle outward traction is applied prior to removal of the catheter system. 18 Pending acceptable results, the delivery catheter and introducer sheath are retracted and the venotomy access site is closed with suture or a vascular closure device. 19 Rare intraprocedural complications include atrial fibrillation (3.3%), transient complete heart block (3.0%), transient device dislodgement (1.7%), bleeding from vascular access sites (.6%), and pericardial effusion (.7%). 4
Extraction
To facilitate percutaneous extraction at end of battery life, leadless pacemakers have a proximal removal device at their non-adhered end that serves as an attachment dock for a catheter to ensnare and remove the pacemaker. While safe percutaneous removal of leadless pacemakers is well documented in small observational studies, encapsulation by endocardial tissue over time can complicate the removal of longstanding leadless pacemakers.20,21 In this circumstance, the electrophysiologist may elect to simply place a second leadless pacemaker without extracting the original encapsulated pacer.22,23
Available Devices and Capabilities
aVDD sensing and pacing enabled by accelerometer technology in the Micra AV that mechanically senses atrial contractions, allowing for ventricular pacing synchronized with atrial activity.
bDDD pacing enabled when Aveir ventricular leadless pacemaker is paired with Aveir AR right atrial leadless pacemaker.
cIf programmed in DDD, magnet placement will result in VOO pacing at 100 beats per minute. If programmed in AAI, magnet placement will result in AOO pacing at 100 beats per minute.
In May of 2023, Medtronic received FDA approval for updated versions of its existing leadless pacemakers system. The Micra VR was upgraded to the Micra VR2 (Medtronic, Minneapolis, MN), and the Micra AV was upgraded to the Micra AV2 (Medtronic, Minneapolis, MN). 27 These devices are similar in size, design, and pacing function to the original models. According to preliminary analyses performed by Medtronic, the new devices are distinguished by improved battery longevity and enhanced atrioventricular synchrony in the Micra AV2 at higher heart rates. 28
The second major leadless pacemaker system approved for use in the United States is the Aveir Leadless Pacemaker (Abbott, Abbott Park, IL). The Aveir device has 2 available models, a first generation single chamber leadless pacemaker and a second generation dual chamber leadless pacemaker. The first generation model, the Aveir VR (Abbott, Abbott Park, IL), received FDA approval in 2022. The Aveir VR device is comparable in size to the Micra VR (38 mm × 6.5 mm) and secures directly to the right ventricular myocardium with a helix apparatus. 29 The Aveir VR is limited to right ventricular pacing and sensing. Abbott’s second generation leadless pacemaker, the Aveir DR (Abbott, Abbott Park, IL), received FDA approval in July, 2023. The Aveir DR is the first and only FDA-approved dual chamber leadless pacemaker system. One device is implanted into the right ventricle and a slightly smaller one, 32.2 mm in length, is implanted into the right atrium. 30 The 2 leadless pacemakers communicate wirelessly using low-voltage electric signals transmitted through the blood and cardiac tissue, obviating the need for connecting leads. 31 This allows the Aveir DR system to perform multiple pacing configurations: AAI, VVI, and DDD. With the ability to provide both atrial and ventricular pacing, the Aveir DR system vastly expands the patient population eligible for a leadless pacemaker and will likely continue to accelerate the rate of implantation.
Transvenous and Leadless Pacemaker Safety Profiles
Traditional transvenous pacemakers are associated with numerous complications stemming from the device insertion process, the subcutaneous pocket where the generator resides, and the pacemaker leads. In a prospective, multi-center cohort study published in 2012 that followed 1517 patients for an average of 5 years after traditional pacemaker implantation, 12.4% of patients experienced a complication within 2 months. 32 The most frequent short-term complications included lead dislocation, lead disconnection, and subcutaneous pocket hematoma. Pneumothorax also occurred in 2.2% of patients. During long-term follow-up averaging 5.8 years, 9.2% of patients experienced a complication, primarily due to lead malfunction and subcutaneous pocket discomfort. Approximately .2% suffered lead endocarditis, a highly morbid infection that often requires device explantation. Additionally, venography studies demonstrate severe venous obstruction in approximately 10% of patients following pacemaker lead implantation. 13 Finally, pacemaker leads traversing the tricuspid valve can interfere with coaptation of the valve apparatus, increasing the risk of tricuspid regurgitation progression following placement.12,33 Notably, pacemaker lead-associated tricuspid regurgitation has been associated with worse clinical outcomes, including higher rates of heart failure hospitalizations as well as mortality in certain studies.34,35
A series of progressive trials over the last decade have established the improved safety profile of leadless pacemakers as compared to traditional transvenous devices. The first trial, published in 2013, was a prospective, multi-center study that followed 33 patients for 3 months following leadless pacemaker implantation. This small study reported a 6% complication rate at 3 months, significantly lower than the complication rate associated with transvenous pacemakers. 36 This small feasibility study paved the way for 2 larger trials published in the New England Journal of Medicine, LEADLESS II and the Micra Transcatheter Pacing Study.5,37 These sentinel trials each studied a cohort of 300 patients for 6 months after leadless pacemaker insertion. At 6-month follow-up, only 4.0% of patients in the Micra Transcatheter Pacing Study and 6.7% of patients in Leadless II experienced a pacemaker complication, significantly less than the complication rate seen with traditional transvenous pacemakers.5,37 Post-hoc analysis of the Micra Transcatheter Pacing Study also demonstrated that patients receiving leadless pacemakers had fewer hospitalizations and fewer system revisions due to device complications as compared to a matched cohort with traditional pacemakers. 38 In both the Micra Transcatheter Pacing Study and a post-approval registry study of the Micra device, there were no recorded incidences of leadless pacemaker infection requiring explantation. 6 Together, these sentinel safety studies validated the notion that the leadless pacer design decreased device complications compared to traditional pacemakers and provided the basis for FDA approval for the first two single chamber ventricular pacemaker devices: the Micra VR and Aveir VR.
These studies also highlighted a unique set of procedural risks associated with leadless pacemaker implantation. While the femoral percutaneous approach with leadless pacemakers eliminated the risk of pneumothorax and subcutaneous pocket hematoma, approximately 1% of patients suffered femoral vascular complications related to the large sheaths required for leadless pacemaker insertion.5,37 Additionally, the risk of cardiac perforation from leadless pacemaker implantation in early studies was 1.5%, nearly double the rate established for transvenous pacemakers. 39 Fortunately, the rate of cardiac perforation has decreased over time with increased operator experience and a practice shift to implant leadless pacemakers into the right ventricular septum, rather than the apex. In the most recent large leadless pacemaker trial, the rate of cardiac perforation was .7%, nearly half of the previously reported rate. 4
Leadless Pacemaker Indications
The advent of the first dual chamber leadless pacemaker, the Aveir DR, has greatly expanded the clinical indications for leadless pacemakers. As demonstrated in the sentinel Aveir DR i2i study, the Aveir DR provided safe and efficacious pacing for patients with common dual chamber pacemaker indications including sinus node dysfunction and atrioventricular block. 4 Accordingly, the Aveir DR is now approved for use for a wide variety of pacing indications including sick sinus syndrome, high grade atrioventricular block, sinus node dysfunction, significant bradycardia, and sinus arrest. 43 This significant expansion in clinical indication will likely continue to accelerate the rate of leadless device implantations.
In addition to the electrophysiologic indications, there are 2 special patient populations that favor the placement of a leadless pacemaker over a traditional transvenous pacer. The first is patients with prior transvenous pacemaker infections requiring device explantation. Due to their smaller surface area, device encapsulation, and absent leads, leadless pacemakers are at markedly decreased risk of infection as compared to transvenous pacemakers. 44 Implanting a new, leadless pacemaker in this patient population within 30 days of explant is safe, with no instances of recurrent infection requiring leadless pacemaker removal reported over approximately 8 months of follow-up. 11 The second patient population are end-stage renal disease (ESRD) patients. Patients with ESRD often have compromised upper extremity venous systems from multiple prior access attempts or arteriovenous fistulas that preclude transvenous pacemaker leads. 3 In this population, the insertion of a leadless pacemaker through the femoral vein offers a viable alternative. 45
Importantly, despite the expanding indications for leadless pacemakers, numerous clinical situations still favor the placement of a transvenous pacemaker. For example, leadless pacemakers are currently incapable of providing CRT, a cornerstone of pacing therapy for patients with reduced ejection fraction, heart failure symptoms, and widened QRS.46,47 Similarly, leadless pacemakers cannot perform conduction system pacing, an emerging alternative to CRT achieved by pacing the bundle of His or left bundle branch. Finally, for patients meeting indications for ICD placement, leadless pacemakers lack defibrillation capacity, although rare case reports have described concomitant leadless pacemaker and subcutaneous ICD placement. 48
Leadless Pacemaker Contraindications
The contraindications to leadless pacemakers are largely anatomic and related to placement issues. An inferior vena cava filter and mechanical tricuspid valve both preclude safe percutaneous placement of leadless pacemakers. Tortuous femoral vasculature is a relative contraindication, although there are multiple case reports of percutaneous placement using upper extremity veins.49,50 Additionally, patients with severe obesity are not candidates, as the increased soft tissue prevents reliable telemetry communication with the implanted device. Finally, young patients require special consideration for leadless pacemakers, as these patients will likely require multiple attempted device extractions and replacements during their lifetime.
Perioperative Considerations for Non-Electrophysiology Surgery
There is a dearth of both clinical research and medical societal guidelines to guide the perioperative management of leadless pacemakers. Published in 2011, the most recent combined Heart Rhythm Society and American Society of Anesthesiologists (ASA) expert consensus on perioperative management of patients with pacemakers predates FDA approval of the first leadless pacemaker. 51 Accordingly, the perioperative management of leadless pacemakers is largely based on clinical experience accrued from the management of traditional transvenous pacemakers in the perioperative setting with a few notable exceptions. Anesthesiologists must verify adequate device function prior to surgery, manage the impact of electromagnetic interference (EMI), and understand how to manage the devices in hemodynamic emergencies.
Preoperative Preparation
It is imperative that the anesthesiologist ascertains the pacemaker device type, manufacturer, magnet response, and indication for leadless pacemaker placement preoperatively. If there is uncertainty as to whether a patient has a leadless pacemaker, a recent chest radiograph can be reviewed. Leadless pacemakers are easily visualized on chest radiograph as small, cylindrical radiopaque devices in the right ventricle. A standard chest radiograph depicting a recently placed Micra AV2 leadless pacemaker is depicted in Figure 1. The ASA Practice Advisory and Heart Rhythm Society both recommend pacemaker interrogation within 6–12 months of scheduled surgery.51,52 While these recommendations were devised for traditional transvenous pacemakers, they remain reasonable for leadless pacemakers given the similarity in reliability and battery life between the 2 device types. Finally, the anesthesiologist must determine whether the patient is dependent on their leadless pacemaker. For pacemaker-dependent patients, interrogation on the day of surgery allows the anesthesiologist to reprogram the device to an asynchronous mode, or, to verify that a magnet response setting is enabled. Chest radiograph of a Micra AV2 leadless pacemaker implanted in the right ventricular septal wall (arrow).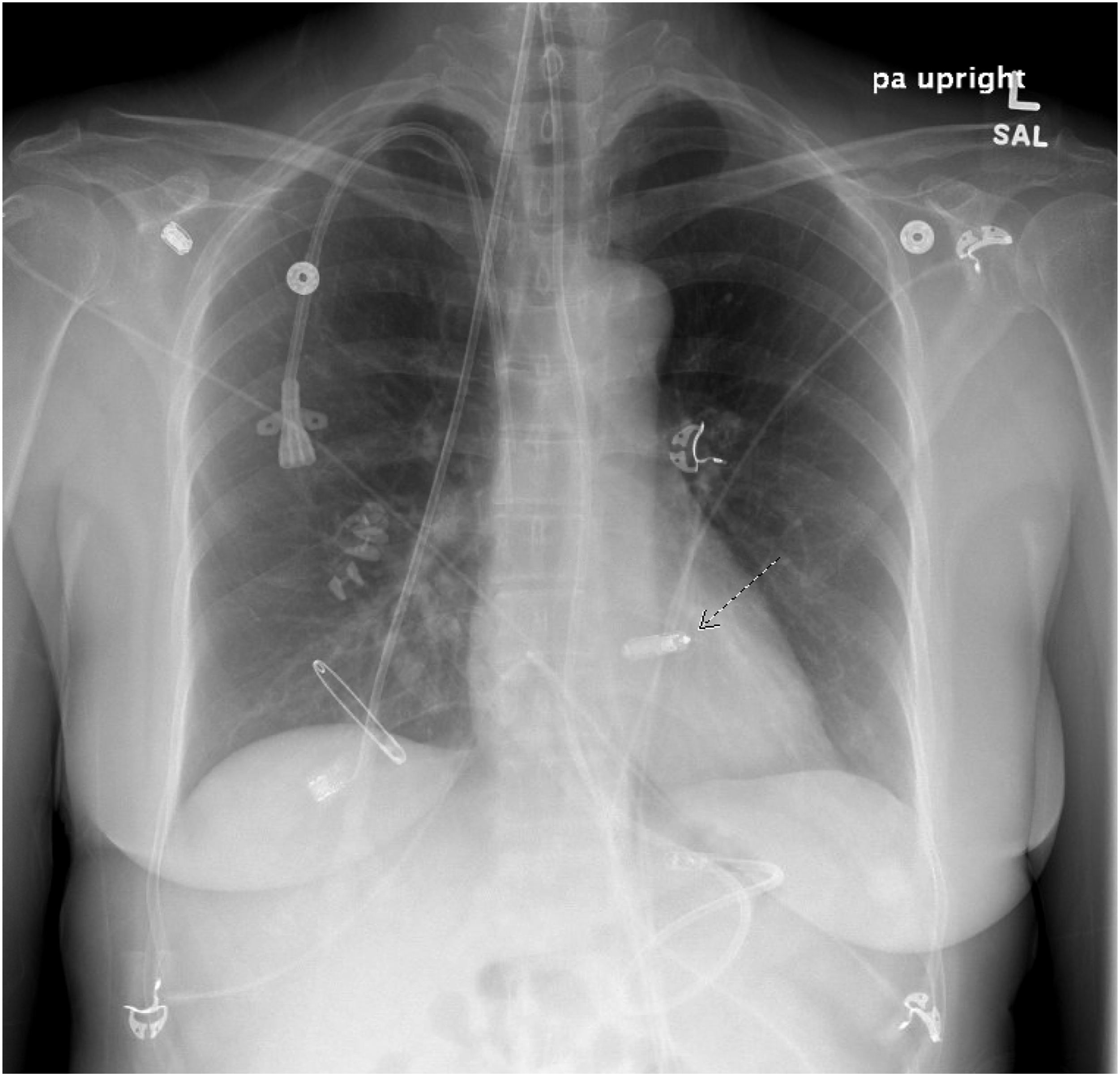
Electromagnetic Interference and Intraoperative Management
As demonstrated by Schulman et al., the use of monopolar electrocautery above the umbilicus can produce EMI disruptive to traditional pacemakers and ICDs. 53 There have been no comparable trials studying the risk of EMI for leadless pacemakers. In a very small case series, Gifford et al. demonstrated no instances of clinically significant EMI in 6 patients with leadless pacemakers receiving various surgeries, including abdominal and neck procedures with monopolar cautery. 54 Nonetheless, given the limited data available for leadless pacemakers and known impact of electrocautery on traditional transvenous devices, anesthesiologists should maintain a low threshold to preoperatively reprogram leadless pacemakers to an asynchronous mode in pacemaker-dependent patients undergoing surgery with expected EMI.
An additional consideration for perioperative management of leadless pacemakers is that only certain models respond to the application of a magnet. For example, the Abbott Aveir system defaults to asynchronous pacing between 85 and 100 beats per minute in the presence of a magnetic field. 55 However, the device’s magnet response setting must be enabled, which requires verification with device interrogation preoperatively. Additionally, the obligatory amount of separation between the intracardiac leadless pacemaker and cutaneous magnet introduces the possibility of inadequate magnetic field strength to trigger the device. Acknowledging this, the manufacturer recommends adding a second magnet stacked on top of the first to increase the strength of the magnetic field in situations of inadequate pacer response. 29 While the manufacturer does not specify an exact site on the chest wall for magnet placement, positioning the magnet over the estimated location of the right ventricular septum ensures optimal proximity to an implanted leadless device. In contrast to the Aveir system, the Medtronic Micra leadless pacemakers are not equipped with a magnet sensor and have no response to cutaneous magnet placement. The inability to reliably convert a patient to an asynchronous mode with intraoperative magnet placement is an additional reason to consider preoperatively reprogramming leadless pacemakers for pacemaker-dependent patients.
For emergent surgery precluding leadless pacemaker reprogramming, preparations should be taken to minimize EMI and to prepare for backup transcutaneous pacing, particularly for pacer-dependent patients. The surgeon should be encouraged to utilize bipolar cautery, and for unipolar cautery, the electrosurgery dispersive electrode should be positioned to direct the current path at least 15 centimeters away from the device. 56 A transcutaneous pacing system should be available in the operating room for instances of sustained, inappropriate pacing inhibition.
Postoperative Management
For postoperative management of traditional transvenous pacemakers, the ASA Practice Advisory and Heart Rhythm Society recommend scheduling routine device interrogation within 1 month. 52 In the absence of clear guidelines for leadless pacemakers, applying this guideline to patients with leadless pacemakers is reasonable. Situations that merit immediate postoperative interrogation include the use of monopolar cautery, emergency surgery, cardioversion, or situations where a patient requires heart rate modulation for hemodynamic instability. 56
Advanced Cardiac Life Support
Similar to the approach to advanced cardiac life support (ACLS) for patients with a traditional transvenous pacemaker, there are no specific modifications to ACLS for patients with leadless pacemakers. Although leadless pacemakers are designed to withstand defibrillation and cardioversion, there is a paucity of data regarding the impact of high energy shocks on leadless pacemakers. 26 The few available case reports demonstrate that defibrillation and cardioversion do not impair leadless pacemaker programming or function.57,58 Cardioversion and defibrillation should not be withheld when emergently indicated. In asystole, the leadless pacemaker will provide a backup paced rate.
Device Dislodgement Risk
In the largest safety study available, the Micra post-approval registry, only .13% of 726 patients experienced device dislodgement. 6 However, this registry follow-up period was limited to 30 days. The incidence of device dislodgement beyond this period is unknown, although the paucity of case reports describing dislodgement of longstanding leadless pacemakers is reassuring. 59 Regardless, caution should be taken when introducing a pulmonary artery catheter or percutaneous right ventricular assist device in close proximity to the leadless pacemaker. Case reports of device dislodgement at the time of insertion emphasize the potential dire cardiopulmonary consequences of device embolism to the pulmonary arteries. 60 Gradual encapsulation of the leadless pacemaker by endocardial tissue may be protective against embolization, though the time frame for this is unclear.
MRI Compatibility
All currently available leadless pacemakers are designated as “MRI conditional.” This is the same classification given to the majority of traditional pacemakers and signifies that the devices are safe and compatible with clinical MRI scans, assuming certain conditions are met. These conditions include patient factors, MRI machine specifications, and device parameters. Notable disqualifying criteria include recent device implantation (<6 weeks) and poor capture thresholds. 61 A recent trial studying the impact of MRI on 14 patients with Micra devices further reaffirmed the safety of MRI scans in patients with leadless pacemakers. Examining planned device interrogations extending to 3 months following an MRI scan, the authors noted no significant changes to pacing thresholds, sensing capability, or battery life. 62
Prior to an MRI scan, leadless pacemakers must be reprogrammed to an MRI compatible setting. Both the Micra and Aveir leadless pacemaker systems contain specific MRI modes that can be enabled. These modes are designed to prevent inappropriate sensing that can result from the strong electromagnetic field emitted by the MRI machine. As such, the MRI compatible modes typically default to an asynchronous pacing mode at a programmable rate.63,64 For pacer-dependent patients, maintaining an asynchronous pacing mode is essential during an MRI scan, since the pacemaker’s sensing function is rendered inaccurate by the MRI. For patients who are not pacer-dependent, the pacing function is typically turned off when activating the MRI compatible mode prior to a scan. This avoids competitive pacing that risks triggering ventricular tachycardia or ventricular fibrillation. Following completion of the MRI, the MRI compatible mode should be turned off and the device returned to its baseline settings.
Conclusion
As our perioperative experience with these devices grows, expertise in management will continue to evolve. More rigorous studies looking at the effects of EMI in particular will be vital to perioperative care. Given the rapid expansion of leadless pacemaker technology and a favorable risk profile, it seems likely these devices will become more common in the operative space and societal guidelines from the ASA and Heart Rhythm Society will be pivotal in reducing variability in practice. Despite these new challenges, leadless pacemakers are an exciting step forward for patients with conduction issues, especially those with limited options for implantation.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author’s Note
We confirm that this work is original and has not been submitted for consideration to any additional journal or book. We have no competing interests to disclose. This manuscript was not supported by any funding source.