Abstract
Background
Pulmonary vasodilators, including oxygen, have not shown consistent beneficial effects on pulmonary hypertension due to valvular heart disease (PH-VHD). Therefore, the study aimed to assess the effect of 100% fractional inspiration of oxygen (FiO2) on pulmonary and systemic hemodynamics in patients with combined pre- and post-capillary pulmonary hypertension (CpcPH) and isolated post-capillary pulmonary hypertension (IpcPH) due to PH-VHD.
Methods
This prospective study was conducted among patients with PH-VHD undergoing mitral or aortic valve replacement or repair. The study was conducted after induction of anesthesia and pulmonary artery catheterization. Cardiac output was obtained using thermodilution and all direct, and derived hemodynamic variables were obtained at 30% and 100% FiO2. The patients were stratified a priori into responders {(≥10 mmHg fall in mean pulmonary artery pressure (MPAP)} and non-responders.
Results
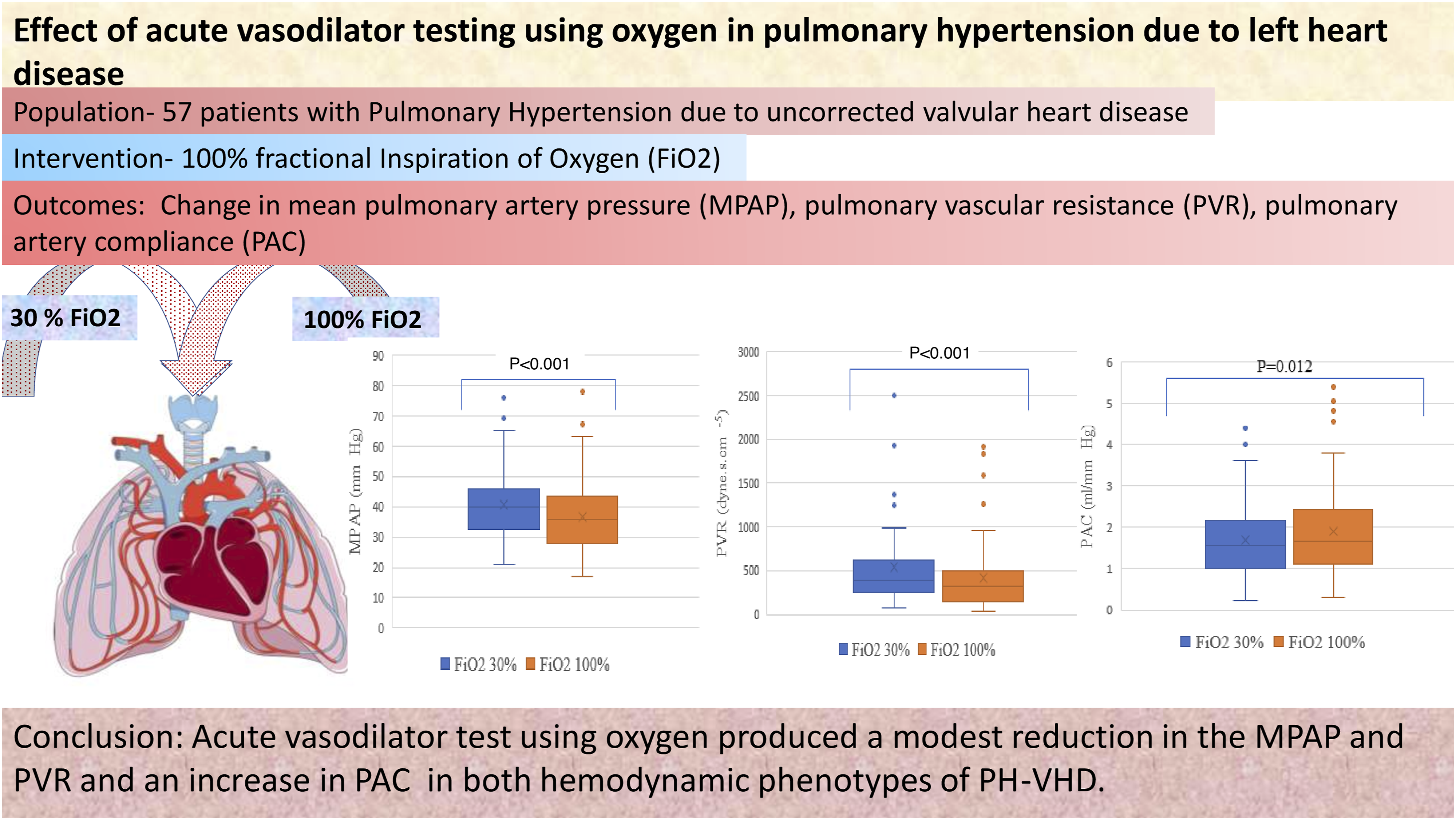
Fifty-seven patients completed the acute vasodilator test. The mean age and body mass index of the study population was 41.8 ± 14.1 years and 21.4 ± 4.6 kg/m2, respectively. There was a significant decrease in MPAP (40.77 ± 12.07 mmHg vs 36.74 ± 13.3 mmHg; P < .001) and pulmonary vascular resistance (PVR) {(median; Interquartile range (IQR); 388; 371 vs 323; 362 dynes sec.cm−5; P < .001) at 100% FiO2. Transpulmonary gradient (TPG) and diastolic pulmonary gradient (DPG) also decreased significantly (P < .001 and P < .001). Cardiac output did not change significantly. The magnitude of decrease in MPAP, PVR, TPG, DPG, and pulmonary artery compliance (PAC) between CpcPH and IpcPH was comparable. Responders did not show a significantly greater fall in MPAP, PVR, TPG, DPG, and PAC after surgery.
Conclusion
Hyperoxia may lead to reduction in MPAP and PVR in both hemodynamic phenotypes of PH-VHD. A larger sample size is required to support or refute the findings of this study.
Keywords
Introduction
The World Health Organization (WHO) has classified pulmonary hypertension (PH) into five distinct categories, and PH due to left heart disease (PH-LHD) is the most common variant reported in the literature. 1 Among individuals with left heart disease, the prevalence of PH has been estimated between 23% and 80%.2,3 The three clinical phenotypes in WHO group 2 PH are due to heart failure with preserved ejection fraction (HFpEF), heart failure due to reduced ejection fraction (HFrEF), and valvular heart disease (VHD). 4
Pulmonary vasodilators have generally produced unpredictable hemodynamic change in PH-LHD.5,6 Additionally, pulmonary hemodynamic characterization using acute vasodilator testing has not been done in PH due to valvular heart disease (PH-VHD). Nevertheless, the acute vasodilator test in PH-VHD (PH-LHD) could be helpful to differentiate between reversible or irreversible combined post and pre-capillary pulmonary hypertension (CpcPH).7,8 The two hemodynamic phenotypes of PH-VHD may not lead to a predictable right ventricular and pulmonary vascular hemodynamic parameter after reduction in pulmonary capillary wedge pressure (PCWP). Therefore, hemodynamic characterization and response to vasodilator in PH-VHD would help in finding suitable alternatives for definitive management in PH and prognostication before surgical correction. Moreover, preoperative reduction in pulmonary artery pressure and pulmonary vascular resistance (PVR) could improve postoperative outcomes.
Among the commonly used vasodilators, oxygen appears to be safe and potent. The specific pulmonary vasodilator effect of oxygen is unlikely to cause significant systemic hemodynamic changes. 9 Extrapolating ambiguous pulmonary vascular effects of oxygen in one category of PH to other categories of PH is incorrect as the pathophysiology in each category of PH is likely to differ. 9 We hypothesize that the extent and magnitude of reactive pulmonary vasoconstriction is partly responsible for isolated post-capillary pulmonary hypertension (IpcPH) and the transition from IpcPH to CpcPH. Hyperoxia is likely to reduce the severity of reactive pulmonary vasoconstriction and may reduce reversible components of pulmonary vascular resistance (PVR) and pulmonary artery pressure. Moreover, in pediatric cardiac catheterization laboratories, oxygen remains a standard method of pulmonary vasodilator testing. 10 Therefore, the study aimed to assess the effect of 100% fractional inspiration of oxygen (FiO2) on pulmonary and systemic hemodynamics among patients with PH-VHD. Additionally, we hypothesized that the decrease in the MPAP and PVR after surgical correction would be significantly higher among those who respond to 100% FiO2.
Materials and Methods
This is a prospective, pre-post study performed in a large tertiary care center. The Institute Ethics Committee approved the protocol (protocol no.
Adult patients (>18 years) with right ventricular systolic pressure ≥40 mmHg (assessed by transthoracic echocardiography) scheduled for elective valvular heart surgery were included in the study. Exclusion criteria of this study included presence of a shunt lesion, right-sided valvular or infundibular stenosis, history of pneumectomy or lobectomy, and patients receiving pulmonary artery vasodilators. Patients requiring an inotrope to maintain hemodynamic stability before or during the study procedure were withdrawn from the study. The patients were also withdrawn if the measured value of the mean pulmonary artery pressure was <20 mmHg during pulmonary artery catheterization. Hemodynamic instability was defined as the requirement of vasoactive or inotropic medication to maintain systolic blood pressure >90 mmHg.
The routine monitoring devices included electrocardiogram, invasive arterial pressure, central venous pressure, temperature, airway pressure, end-tidal carbon dioxide, trans esophageal echocardiography, and Bispectral Index (BIS).
Anesthesia induction was performed with midazolam (.05 mg/kg), fentanyl (2-5 mcg/kg), thiopentone (3-5 mg/kg), and vecuronium (.01 mg/kg) in 50:50 ratio of oxygen and air. FiO2 was immediately lowered to 30% after endotracheal intubation to maintain end-tidal O2 <30%. A pulmonary artery catheter (Edwards Lifesciences Corp, One Edwards Way, Irvine, CA) was introduced through the sheath placed in the right internal jugular vein. The transesophageal echocardiographic probe was inserted before pulmonary artery catheter manipulation. Adequate wedging of the pulmonary artery catheter was confirmed by inflation and deflation of the balloon. Anesthesia was maintained with sevoflurane (1minimum alveolar concentration) with the supplemental dosage of fentanyl. The ventilator settings were adjusted to keep end-tidal CO2 between 35-40 mmHg. The PEEP was kept at 5 cm H2O. The first part of the study began after maintaining patients on 30% FiO2 for at least 15 min. No physical or surgical stimulus was allowed during the study procedure. Core body temperature was maintained with the help of a forced air warmer. Intermittent thermodilution (10 mL cold saline) was used to measure cardiac output using the InteliView MX550 hemodynamic monitor (Philips Medizin System, Boeblingem GmbH, Hewlett-Packard- Str-2, 71034 Boeblingen, Germany). Cardiac output was obtained in sets of three, and the average value was used to calculate derived hemodynamic variables.
The FiO2 was increased to 100% after hemodynamic and respiratory variables were obtained at FiO2 of 30%. The second set of cardiac output measurements, direct hemodynamic data and derived hemodynamic data were obtained 10 min later on 100% FiO2. During the course of the study, no additional fluids were given, and BIS was maintained between 50-70. Surgical procedures began once the acute vasodilator test was over. Dobutamine (5-10 mcg/kg/min) and adrenaline infusion (.05-.1) were used during weaning from cardiopulmonary bypass. All the measurements were repeated at FiO2 of 40% after 1 h stay in the intensive care units.
Patients who had a decrease in MPAP by ≥10 mmHg accompanied by a reduction in absolute value below 40 mmHg without or minimal increase in cardiac output were labeled responders. 1
PH-LHD, CpcPH, and IpcPH were defined according to the European Society of Cardiology/European Respiratory Society (ESC/ERS) guidelines. 1 PH-LHD was defined as MPAP >20 mmHg and PCWP >15 mmHg. The CpcPH was characterized by MPAP >20 mmHg, pulmonary vascular resistance (PVR) >3 Wood Units (240 dynes.s.cm−5) and PCWP >15 mmHg, while IpcPH was characterized by MPAP >20 mmHg, PVR ≤3 Wood units and PCWP >15 mmHg. 4
Statistical Analysis
We obtained a mean difference of 3 ± 7.5 mmHg in MPAP during a pilot study on 15 patients with PH-VHD, which was consistent with a previous study using oxygen among adult patients with pulmonary artery hypertension. 11 Fifty-two patients were required to find a similar mean difference with a power of 80% and type I error of .05 using paired t-test. We decided to increase the sample size by 10% to compensate for the withdrawal. Data were assessed for normality using Kolmogorov–Smirnov and Shapiro–Wilk test. Normally distributed data were analyzed for differences at 30% FiO2 and 100% FiO2 using the Student’s paired t-test. Wilcoxon signed-rank test was used for non-normally distributed data. Between-group comparisons (responders vs non-responders and CpcPH and IpcPH) were evaluated using an independent t-test or Mann-Whitey U test, as appropriate. A P value <.05 was considered significant. Data were analyzed using SPSS 24.0 version.
Results
Demographic Parameters (n = 57).
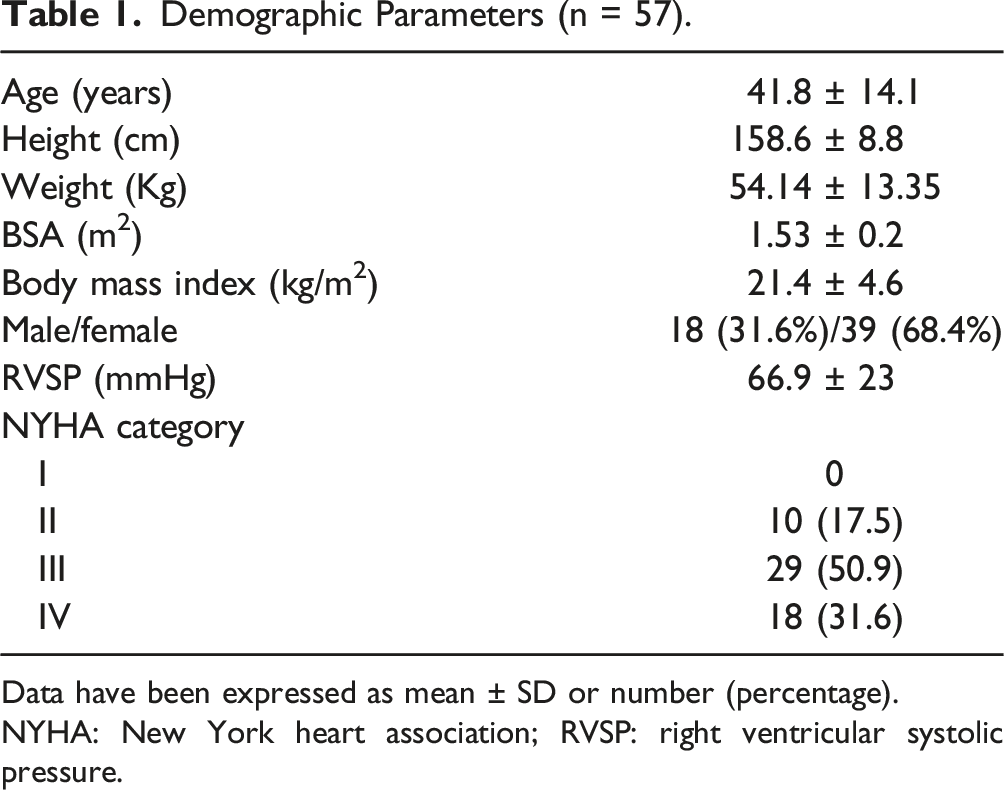
Data have been expressed as mean ± SD or number (percentage).
NYHA: New York heart association; RVSP: right ventricular systolic pressure.
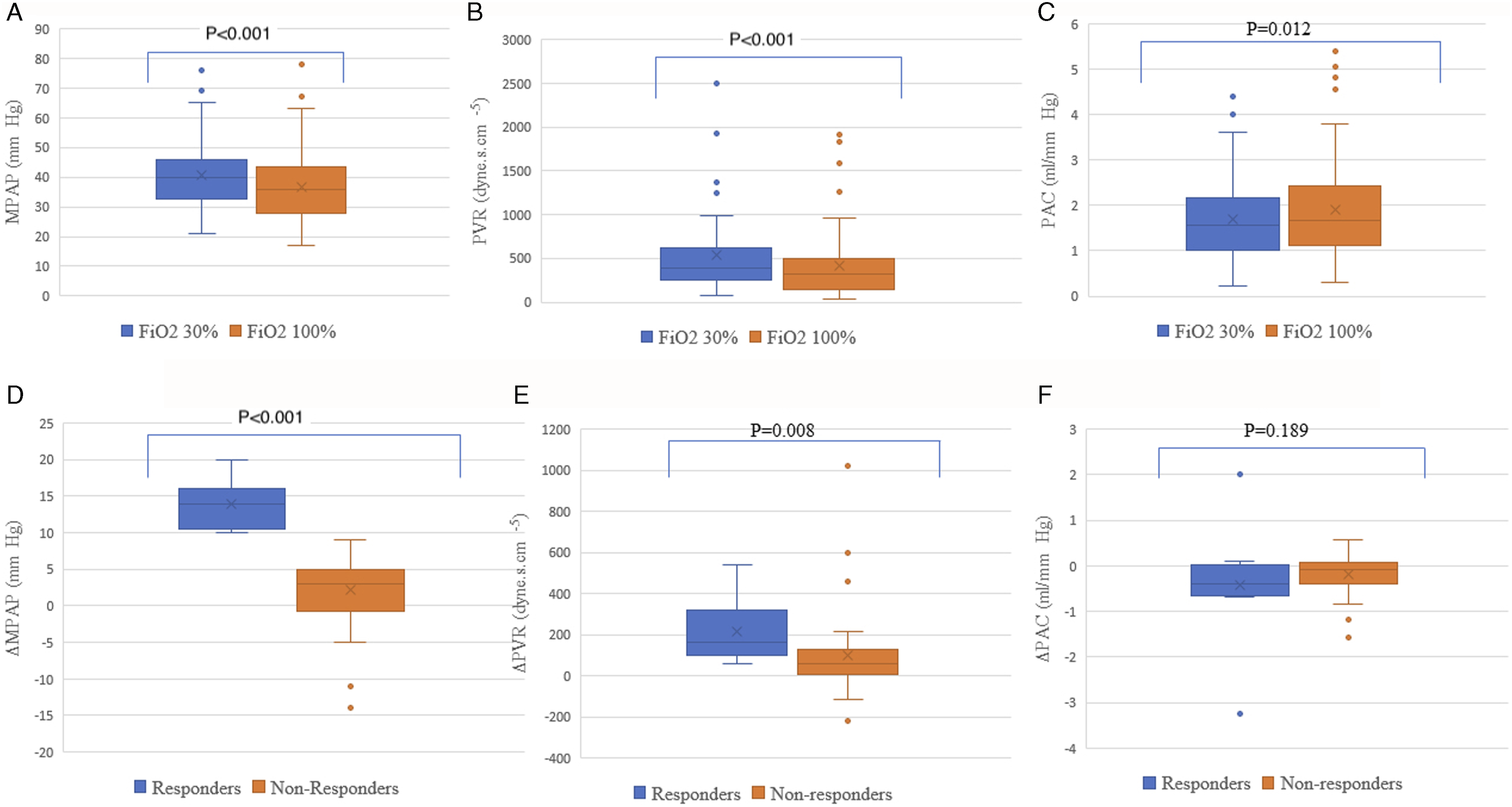
Panel (a). Box and whisker plot showing change in mean pulmonary artery pressure (MPAP) from 30% FiO2 (fractional inspiration of oxygen) to 100% FiO2. Panel (b). Box and whisker plot showing change in pulmonary vascular resistance (PVR) from 30% FiO2 (fractional inspiration of oxygen) to 100% FiO2. Panel (c). Box and whisker plot showing change in pulmonary artery compliance (PAC) from 30% FiO2 (fractional inspiration of oxygen) to 100% FiO2. Panel (d). Box and whisker plot comparing differences in mean pulmonary artery pressure (Δ MPAP = MPAP at 30% FiO2-MPAP at 100%) among responders and non-responders. Panel (e). Box and whisker plot comparing differences in pulmonary vascular resistance (Δ PVR = PVR at 30% FiO2-PVR at 100%) among responders and non-responders. Panel (f). Box and whisker plot comparing differences in pulmonary artery compliance (Δ PAC = PAC at 30% FiO2-PAC at 100%) among responders and non-responders.
Hemodynamic and Respiratory Variables at Both FiO2 Levels (n = 57).
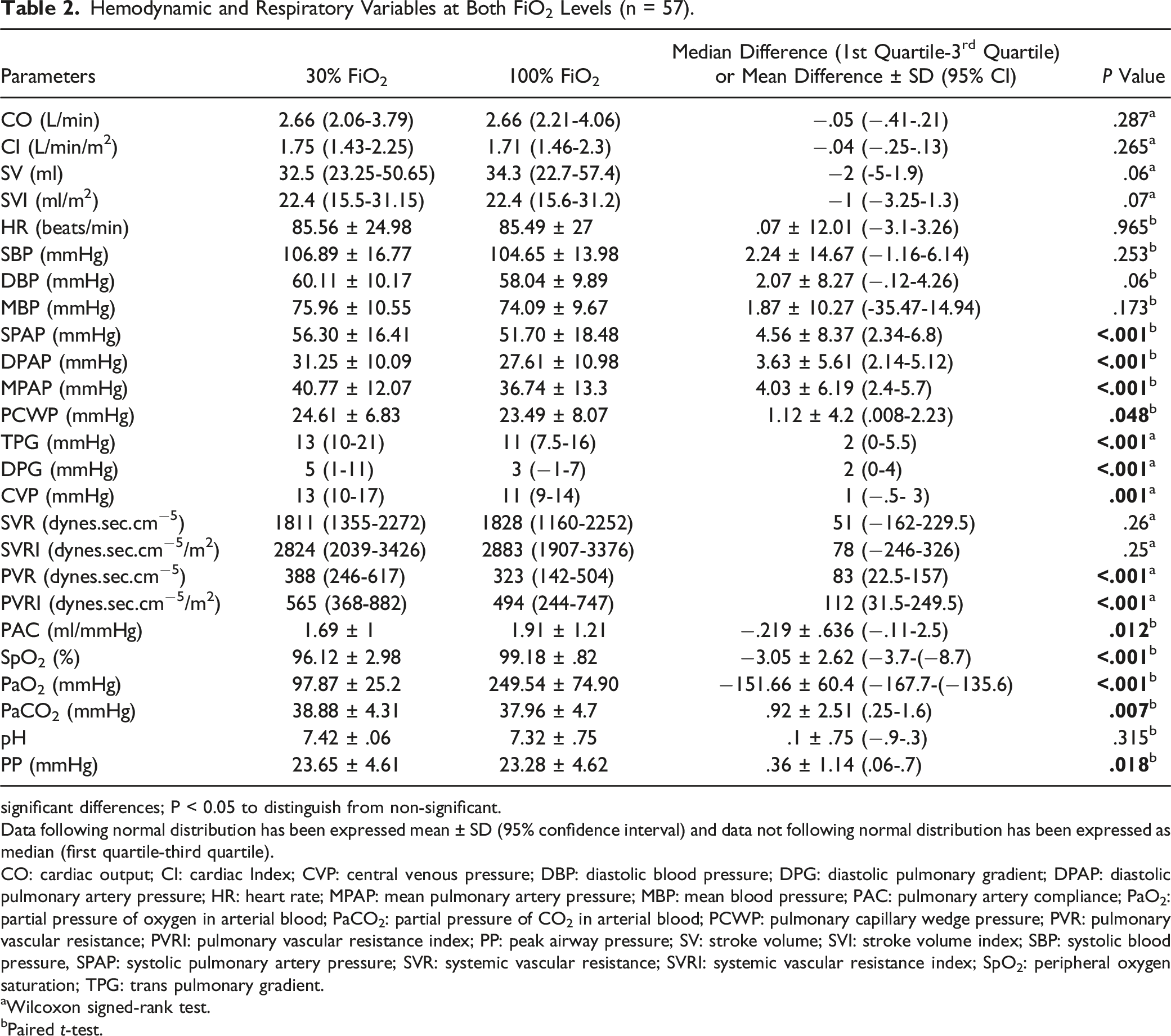
significant differences; P < 0.05 to distinguish from non-significant.
Data following normal distribution has been expressed mean ± SD (95% confidence interval) and data not following normal distribution has been expressed as median (first quartile-third quartile).
CO: cardiac output; CI: cardiac Index; CVP: central venous pressure; DBP: diastolic blood pressure; DPG: diastolic pulmonary gradient; DPAP: diastolic pulmonary artery pressure; HR: heart rate; MPAP: mean pulmonary artery pressure; MBP: mean blood pressure; PAC: pulmonary artery compliance; PaO2: partial pressure of oxygen in arterial blood; PaCO2: partial pressure of CO2 in arterial blood; PCWP: pulmonary capillary wedge pressure; PVR: pulmonary vascular resistance; PVRI: pulmonary vascular resistance index; PP: peak airway pressure; SV: stroke volume; SVI: stroke volume index; SBP: systolic blood pressure, SPAP: systolic pulmonary artery pressure; SVR: systemic vascular resistance; SVRI: systemic vascular resistance index; SpO2: peripheral oxygen saturation; TPG: trans pulmonary gradient.
aWilcoxon signed-rank test.
bPaired t-test.
Responders vs Non-Responders
Hemodynamic and Respiratory Changes Among Responders and Non-Responders.

significant differences; P < 0.05 to distinguish from non-significant.
Data has been expressed mean ± SD or median (first quartile-third quartile).
CO: cardiac output; CI: cardiac Index; CVP: central venous pressure; DBP: diastolic blood pressure; DPG: diastolic pulmonary gradient; DPAP: diastolic pulmonary artery pressure; HR: heart rate; MPAP: mean pulmonary artery pressure; MBP: mean blood pressure; PAC: pulmonary artery compliance; PaO2: partial pressure of oxygen in arterial blood; PaCO2: partial pressure of CO2 in arterial blood; PCWP: pulmonary capillary wedge pressure; PVR: pulmonary vascular resistance; PVRI: pulmonary vascular resistance index; PP: peak airway pressure; SV: stroke volume; SVI: stroke volume index; SBP: systolic blood pressure, SPAP: systolic pulmonary artery pressure; SVR: systemic vascular resistance; SVRI: systemic vascular resistance index; SpO2: peripheral oxygen saturation; TPG: trans pulmonary gradient.
aMann–Whitney U test.
bIndependent t-test.
CpcPH and IpcPH
Hemodynamic and Respiratory Changes Among CpcPH and IpcPH.
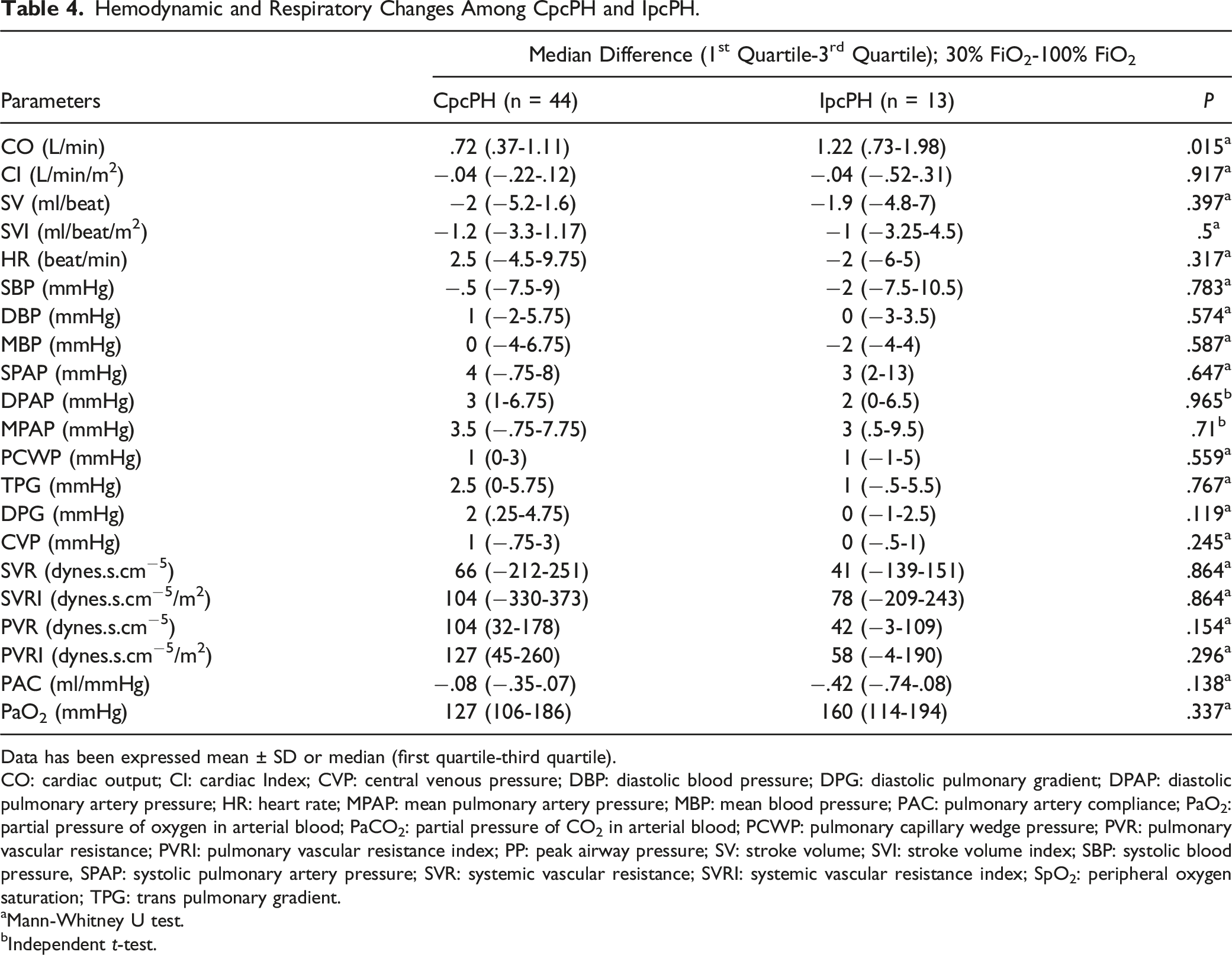
Data has been expressed mean ± SD or median (first quartile-third quartile).
CO: cardiac output; CI: cardiac Index; CVP: central venous pressure; DBP: diastolic blood pressure; DPG: diastolic pulmonary gradient; DPAP: diastolic pulmonary artery pressure; HR: heart rate; MPAP: mean pulmonary artery pressure; MBP: mean blood pressure; PAC: pulmonary artery compliance; PaO2: partial pressure of oxygen in arterial blood; PaCO2: partial pressure of CO2 in arterial blood; PCWP: pulmonary capillary wedge pressure; PVR: pulmonary vascular resistance; PVRI: pulmonary vascular resistance index; PP: peak airway pressure; SV: stroke volume; SVI: stroke volume index; SBP: systolic blood pressure, SPAP: systolic pulmonary artery pressure; SVR: systemic vascular resistance; SVRI: systemic vascular resistance index; SpO2: peripheral oxygen saturation; TPG: trans pulmonary gradient.
aMann-Whitney U test.
bIndependent t-test.
After Surgery (All Patients)
The MPAP and PVR after surgery were (mean ± SD; 30.4 ± 9.8 mmHg) and mean ± SD; 276.7 ± 150.7 dynes.s.cm−5), respectively. The PCWP, TPG and DPG after surgery were (mean ± SD; 14.4 ± 5.4), (mean ± SD; 15.8 ± 7.4) and (mean ± SD; 7 ± 4.6), respectively. 32 patients required dobutamine alone, while 20 patients required dobutamine and adrenaline. 5 patients did not require inotrope.
Responders and Non-Responders After Surgery
Hemodynamic and Respiratory Changes Among Responders and Non-Responders After Surgery.
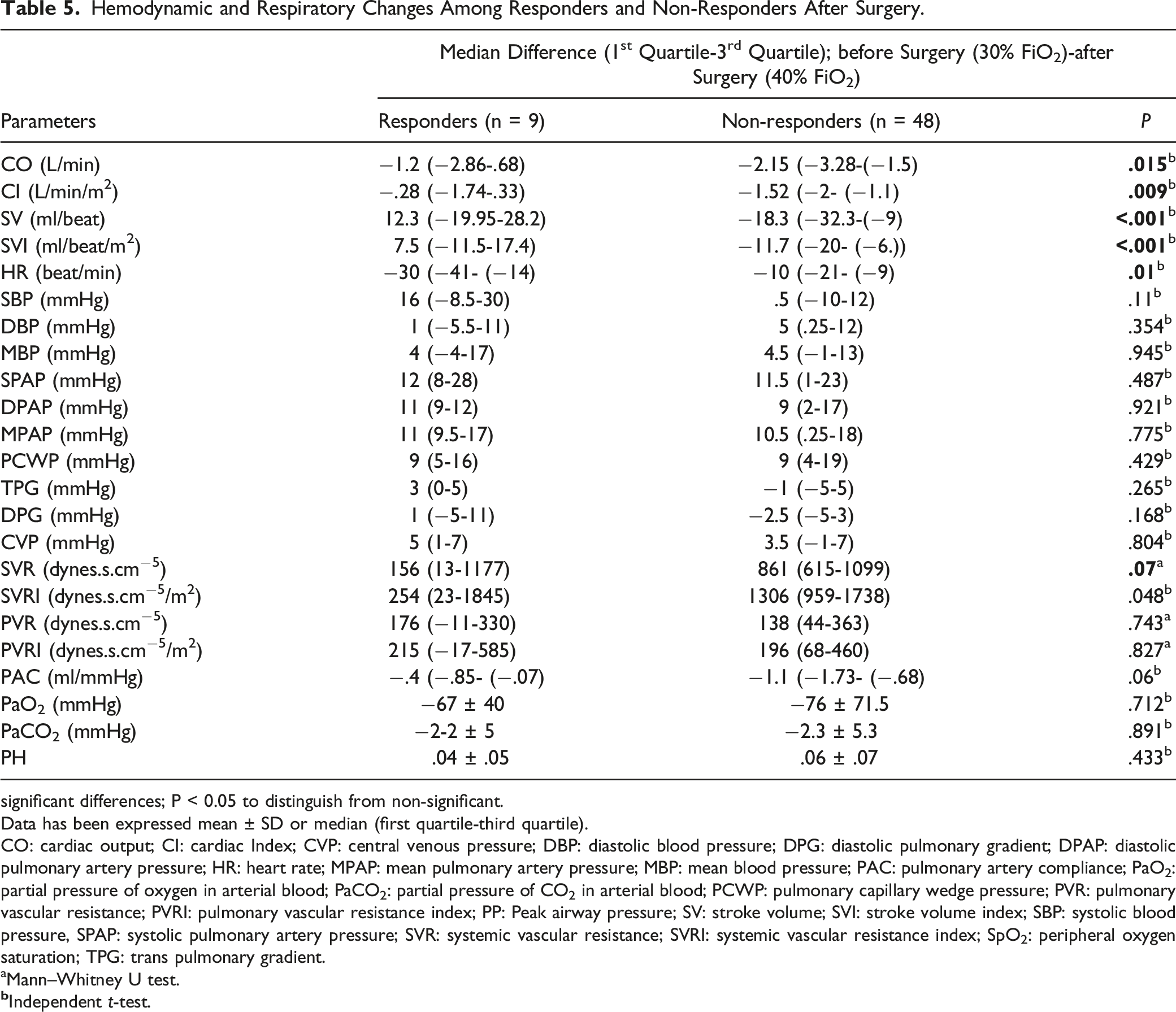
significant differences; P < 0.05 to distinguish from non-significant.
Data has been expressed mean ± SD or median (first quartile-third quartile).
CO: cardiac output; CI: cardiac Index; CVP: central venous pressure; DBP: diastolic blood pressure; DPG: diastolic pulmonary gradient; DPAP: diastolic pulmonary artery pressure; HR: heart rate; MPAP: mean pulmonary artery pressure; MBP: mean blood pressure; PAC: pulmonary artery compliance; PaO2: partial pressure of oxygen in arterial blood; PaCO2: partial pressure of CO2 in arterial blood; PCWP: pulmonary capillary wedge pressure; PVR: pulmonary vascular resistance; PVRI: pulmonary vascular resistance index; PP: Peak airway pressure; SV: stroke volume; SVI: stroke volume index; SBP: systolic blood pressure, SPAP: systolic pulmonary artery pressure; SVR: systemic vascular resistance; SVRI: systemic vascular resistance index; SpO2: peripheral oxygen saturation; TPG: trans pulmonary gradient.
aMann–Whitney U test.
CpcPH and IpcPH After Surgery
The magnitude of increase in CO {median (first quartile-third quartile); 2.2 (3.44- (−1.5) l/min vs-1.31 (−2.49- (−.5) l/min; P = .035} and SV {18.8 (−32.5- (−10) ml/beat vs 4.6 (-15-11)) ml/beat; P = .006} were significantly higher in the CpcPH group. The MPAP decreased by 12 (4-18) mmHg and 5 (−2.5-15) mmHg while PVR decreased by 209 (109-378) and 6 (-67-51) dynes.s.cm−5, respectively in the CpcPH and IpcPH group (Table S5; Supplemental digital content).
Discussion
In this study, we observed a modest decrease in MPAP (4.03 ± 6.19; P = <.001) and PVR {(median (IQR);83 (180); P = <.001} accompanied by a rise in PAC (.219 ± .636; P = .012) after 100% FiO2 among patients with PH due to uncorrected valvular heart disease (
Unlike pediatric heart catheterization practice patterns, 100% oxygen has rarely been used as a vasodilator in the hemodynamic characterization of PH among adults. 12 In a series of adult patients with PH due to mixed etiology, 100% oxygen led to a reduction in MPAP and PVR by 3 mmHg and 2.5 Wood units, respectively. 11 Acute vasodilator testing using oxygen in PH-VHD has been rarely reported in the literature. Therefore, the recommendation of the recent guidelines not to use oxygen in vasodilator testing is not based on credible evidence.1,9 Conceivably, improved alveolar PO2 after 100% oxygen is likely to reduce PVR and MPAP by inducing a fall in reactive pulmonary vasoconstriction. In this study, PaO2 increased from 98 mmHg to 250 mmHg after 100% FiO2 and could have mitigated reactive pulmonary vasoconstriction. Moreover, in a review of nine studies of PH cohorts due to mixed etiology and population, eight studies observed a reduction in PVR and MPAP by 20-30% and 10-20%, respectively. 9
With respect to PH-LHD, hemodynamic characterization using acute vasodilator testing and vasodilator therapy has been mainly targeted in PH due to left ventricular systolic or diastolic dysfunction. In patients with heart failure with preserved ejection fraction, vasodilator testing using sodium nitroprusside/nitroglycerin led to a significant decrease in MPAP, PVR, and CO. 13 Concerningly, perioperative PH characterization and targeted vasodilator therapy in the clinical phenotype of PH-LHD due to uncorrected valvular heart disease have often been ignored or have yielded little success in improving clinical outcomes.
Though our intended goal was to perform hemodynamic characterization only, a long-term oxygen supplementation in the perioperative period in PH-VHD may improve clinical outcomes by reducing the MPAP and PVR. High FiO2 could have produced a higher reduction in TPG in responders due to pulmonary vasodilatation. The reduction in PVR and a rise in PAC after oxygenation is likely to improve right ventricular function by reducing the static and dynamic load of the right ventricle. This study raises the possibility of using other vasodilators in the perioperative management and acute vasodilator testing in this clinical phenotype of PH.
The incidence of responders (15.8%) in this study is consistent with previously published reports among patients with idiopathic pulmonary artery hypertension using different vasodilators during acute vasodilator testing.14,15
Interestingly, we noted a similar magnitude of change in MBP, MPAP, PVR, SVR, TPG, DPG, PCWP, and PAC among responders and non-responders after surgery. Additionally, no significant differences were observed after oxygenation among patients with CpcPH and IpcPH. Of note, a better improvement in PAC among IpcPH is consistent with a previous report. 13 In the beginning, elevation in the left atrial or pulmonary venous pressure causes dilatation in pulmonary circulation and fall in PVR. This fall in PVR is usually prevented by reactive pulmonary vasoconstriction. 16 The obliterative changes in pulmonary vasculature due to alveolar edema or hemorrhage may lead to a progressive increase in PVR. 17 Plausibly, both CpcPH and IpcPH could be at risk of an increase in PVR after the reduction in PCWP. In this study, we did not observe an increased PVR after surgery in both groups. Nevertheless, DPG rose significantly in the IpcPH group after surgery. Therefore, hyperoxygenation may benefit both categories of PH after surgical correction. The PCWP fell below 15 mmHg, and all patients in the CpcPH group came under the pre-capillary PH group. The utility of oxygen in pre-capillary PH has been abundantly evaluated with reasonable success. 1
The extent and mechanism of reactive pulmonary vasoconstriction are unclear, may vary at different pathophysiological stages of the disease, and could be higher in CpcPH. The reduction in PVR and MPAP after oxygenation in this study could have been due to alleviation of hypoxia-induced reactive pulmonary vasoconstriction. The pathophysiological mechanism of reactive pulmonary hypertension or irreversible component of PVR could be further explored using other pulmonary artery vasodilators during acute vascular testing in this clinical phenotype of PH-LHD.
A homogenous patient population is the major strength of this study. We were able to compare and analyze the effect of 100% FiO2 in both hemodynamic phenotypes of a particular clinical phenotype (uncorrected VHD) of PH-LHD. The other major strength includes rigorous inclusion, exclusion and withdrawal criteria. We believe this is the largest vasoreactivity study performed in patients with PH-VHD utilizing oxygen. The previous trials using pulmonary artery vasodilators have not yielded satisfactory clinical outcomes in patients with elevated PCWP (>15 mmHg).5,6 Hyperoxygenation improved pulmonary hemodynamic without altering PCWP, and the magnitude of change in PVR and MPAP was similar in both CpcPH and IpcPH. This study’s findings might help explore the effect of limited oxygen therapy in reducing PVR and MPAP in the perioperative period. However, long-duration hyperoxemia may have unintended consequences. Hyperoxia may affect multiple organ systems by producing reactive oxygen species that may have cytotoxic and proinflammatory effects. The lungs may develop diffuse alveolar damage, reabsorption atelectasis, fibrosis, and mucociliary dysfunction. Systemic, cerebral, and coronary artery vasoconstriction may lead to neurologic and cardiac toxicity. 18
The smaller sample size and single-center design of this study are the most notable limitations. Though we observed a statistically significant decrease in MPAP, the magnitude of the decrease (4 mmHg) may not be physiologically significant. Nevertheless, in patients with severe pulmonary hypertension, any reduction in MPAP and PVR would likely improve right ventricular function and cardiac output. Our study population comprises younger adults without any obvious lung parenchymal lesions and hemodynamic instability. Therefore, the findings of this study may not be generalizable to older patients and patients with hemodynamic instability due to severe pulmonary hypertension and right ventricular dysfunction. Older patients may have parenchymal lung lesions and chronic obstructive pulmonary disease. 19 These older patients are more prone to baseline hypoxemia, and hyperoxia could produce a higher magnitude of reduction in MPAP and PVR. Anesthesia or sedation would have altered pulmonary and systemic hemodynamics. However, we obtained all hemodynamic measurements under anesthesia only. The effect of anesthesia would have been negated, the only difference was intervention (30% FiO2 and 100% FiO2). We observed the effect of oxygen under a stable and controlled environment (similar BIS value, PaCO2, and temperature). No physical or surgical stimulus was allowed during the study. The fluid infusion was also avoided. Additionally, the study intended to withdraw patients in the event of hemodynamic instability or inotropic requirement. The same study can be done in awake patients to find the magnitude of change in hemodynamics after 100% oxygenation. However, administering 100% oxygen by mask for 10 minutes is indeed challenging and it is difficult to ensure 100% oxygenation without curtailing anxiety and sympathetic stimulation. Notably, the majority of the catheterization studies and vasoreactivity studies in infants and children is done under sedation or anesthesia. A larger study would help to validate the findings of this study in PH-VHD. The potential benefit of oxygen supplementation in severe pulmonary hypertension has to be weighed against the possible harm associated with hyperoxia. We have to assess the benefit and harm associated with oxygen supplementation vis a vis outcome in severe pulmonary hypertension. Whether the harm associated with oxygen outweighs the benefit in severe pulmonary hypertension requires further study.
Conclusions
Hyperoxia may lead to reduction in MPAP and PVR in both hemodynamic phenotypes of PH-VHD. Responders to oxygen did not have a significantly higher fall in MPAP and PVR after surgery. A larger sample size is required to support or refute the findings of this study.
Supplemental Material
Supplemental Material - Effect of Acute Vasodilator Testing Using Oxygen in Pulmonary Hypertension Due to Left Heart Disease
Supplemental Material for Effect of Acute Vasodilator Testing Using Oxygen in Pulmonary Hypertension Due to Left Heart Disease by Sundararaj Rajkumar, Ajay Kumar Jha, Satyen Parida, Chitra Rajeswari, Sakthirajan Panneerselvam, and Sreevathsa K. S. Prasad in Journal of Seminars in Cardiothoracic and Vascular Anesthesia.
Footnotes
Author Contributions
Name: Sundararaj Rajkumar, MBBS, MD. Contribution: This author helped with intellectual content, study design, data collection, writing and editing all sections of manuscript, and approving manuscript for publication. Name: Ajay Kumar Jha, MD, DM. Contribution: This author helped with intellectual content, study design, writing, and editing all sections of manuscript, and approving manuscript for publication. Name: Satyen Parida, MBBS, MD. Contribution: This author helped with intellectual content, study design, writing and editing all sections of manuscript, and approving manuscript for publication. Name: Chitra Rajeswari, MBBS, MD. Contribution: This author helped with intellectual content, study design, writing, and editing all sections of manuscript, and approving manuscript for publication. Name: Shakthirajan Paneerselvan, MBBS, MD. Contribution: This author helped with intellectual content, study design, writing, and editing all sections of manuscript, and approving manuscript for publication. Name: Srivatsa KS Prasad. Contribution: This author helped with intellectual content, study design, writing, and editing all sections of manuscript, and approving manuscript for publication.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Intramural funding of the Institute (Jawaharlal Institute of Postgraduate Medical Education and Research, Puducherry, India.
Ethical Statement
Clinical Trial Registration
Clinical Trial Registry India number: CTRI/2019/09/021441 (https://ctri.nic.in) URL: https://ctri.nic.in/Clinicaltrials/pmaindet2.php?EncHid=MzcwMzU=&Enc=&userName=.
Data Availability Statement
The data underlying this article will be shared on reasonable request to the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
