Abstract
Preeclampsia is a serious disorder of human pregnancy occurring after 20 weeks of gestation. It can be divided into subtypes of early onset (<34 weeks of gestation) and late onset (>34 weeks). Presymptomatic detection to identify those at high risk is important for managing this disease. HtrA3, a serine protease with high expression in the developing placenta, exists in long (HtrA3-L) and short (HtrA3-S) isoforms. They are identical, except HtrA3-S lacks the C-terminal PDZ domain. We have previously shown by Western blot analysis that serum HtrA3 levels at the end of the first trimester are significantly higher in women who later develop preeclampsia than in controls. In this study, using highly specific HtrA3 monoclonal antibodies, we established and fully validated two enzyme-linked immunosorbent assays to detect both HtrA3 isoforms together (HtrA3-T) and HtrA3-L alone in the human serum. We then determined serum HtrA3 at 11 to 13 weeks of gestation in a cohort of singleton pregnancies that proceeded without complications or developed preeclampsia in the third trimester. Compared with controls, those who developed late-onset preeclampsia had significantly higher levels of HtrA3-L, whereas those who developed early-onset preeclampsia had significantly lower ratios of HtrA3-L/HtrA3-T. These data support a potential utility of these HtrA3 ELISAs for early detection of preeclampsia.
Introduction
Preeclampsia (PE) is a life-threatening disorder of human pregnancy, affecting 2% to 8% of pregnancies worldwide. 1 It is one of the leading causes of maternal and neonatal morbidity and mortality. PE is characterized by de novo hypertension and proteinuria after 20 weeks of gestation. PE can be further divided into two distinct subtypes of early onset and late onset, according to the timing of disease presentation and diagnosis. 2 Early-onset PE is defined when the disease presents before 34 weeks of gestation, and late-onset PE occurs after 34 weeks of pregnancy. 2
Currently, there is no effective treatment for PE other than delivery, and it remains one of the major attributing causes of preterm birth. 3 In recent years, attentions have turned to identifying biomarkers to predict the development of PE, preferentially at the end of the first trimester of pregnancy well before the symptoms appear. 4 Correctly identifying women who are at risk early in pregnancy has significant clinical implications, as administration of low-dose aspirin before or at 16 weeks of gestation reduces the incidence of PE, especially the severe cases. 4 However, no definitive method is yet available to predict PE in early pregnancy, although several biomarkers and their combinations with other screening approaches have been proposed. 5 Some of these candidate biomarkers include pregnancy-associated plasma protein A, placental growth factor (PIGF), and soluble FMS-like tyrosine kinase–1.6,7 However, none of these factors alone has shown sufficient predictability when used in the first trimester; they are being tested in combination with one or more biophysical markers such as mean arterial pressure and uterine artery Doppler ultrasound. 8
Furthermore, it is increasingly recognized that the two subtypes of PE have distinct etiology and clinical consequences.9,10 Although early-onset PE poses greater maternal and perinatal risks and often requires earlier intervention, late-onset PE can still be severe and its prediction is as equally important. However, recent studies indicated better prediction for early-onset than late-onset PE. 8 For instance, serum PIGF levels at 12 weeks’ gestation were found to be significantly lower in women who later developed early-onset PE; however, the same study failed to predict late-onset PE with serum PIGF. 8
Our previous studies have identified that high-temperature-requirement A3 protein (HtrA3) is highly expressed in the developing human placenta. 11 HtrA3, a serine protease, belongs to the HtrA family that also includes HtrA1, HtrA2, and HtrA4. All members of this family, with the exception of HtrA2, share a similar domain structure and are secreted out of the cell. 12 We have previously demonstrated that placental HtrA3 is secreted into the maternal circulation during human pregnancy and that serum HtrA3 levels are highest in the first trimester. 11 We have also shown by Western blot analysis that maternal serum levels of HtrA3 at 13 to 14 weeks of gestation are significantly higher in women who subsequently developed PE. 13
Human HtrA3 protein exists in two isoforms, long (HtrA3-L) and short (HtrA3-S), and both isoforms are highly expressed in the developing placenta. 12 They are identical, except HtrA3-S lacks the C-terminal PDZ domain, which is known to be important for protein-protein interaction. 12 Because of this major difference, the two HtrA3 isoforms may have different functions and substrate specificities. 12
Since circulating HtrA3 is detected highest in the first trimester and appears to be regulated differently in women who would proceed with normal or PE pregnancies, establishing a sensitive and widely applicable assay to measure serum HtrA3 in early pregnancy may identify women at risk of developing PE. We have previously generated five highly specific HtrA3 monoclonal antibodies (mAbs). 14 Although all five of these antibodies detect HtrA3-L, three detect both isoforms. In this study, we first used these HtrA3 mAbs and developed two enzyme-linked immunosorbent assays (ELISAs) to detect HtrA3 isoforms in the human serum. We then conducted a retrospective study with these two HtrA3 ELISAs to determine serum HtrA3 isoforms at 11 to 13 weeks of gestation in a cohort of singleton pregnancies that subsequently proceeded with normal pregnancy or developed PE in the third trimester. The ultimate goal of this study was to determine whether the two HtrA3 ELISAs would provide useful tools for early detection of PE.
Materials and Methods
HtrA3 Monoclonal Antibodies
Production and characterization of HtrA3 monoclonal antibodies (mAbs) was previously reported. 14 Among the five HtrA3 mAbs, three (9C9, 2C4, and 10H10) detect both HtrA3-L and HtrA3-S isoforms, whereas the other two (6G6 and 3E6) detect HtrA3-L isoform only ( Suppl. Table S1 ).
Biotinylation of HtrA3 mAbs
Zeba desalting spin 7K MWCO 2 mL columns (Thermo Fisher Scientific, VIC, Australia) were washed with BupH phosphate-buffered saline (PBS; 0.1 M sodium phosphate, 0.15 M NaCl, pH 7.2; Thermo Fisher Scientific). HtrA3 mAbs were buffer exchanged with the above columns and assessed by nanodrop (ND-1000, Biolab Group, VIC, Australia) for concentration. Biotin (Sigma Aldrich, MO) prepared at 5 mg/mL in 100% DMSO (Sigma Aldrich) was added to HtrA3 mAbs at a ratio of 100 µg biotin per 1 mg of antibody. The mAb-biotin mixture was protected from the light and incubated at room temperature for 2 h on a rotating wheel and then passed through a fresh Zeba column by centrifugation at 1000
Establishment of HtrA3 Isoform ELISAs
A sandwich ELISA requires a capture antibody and a detection antibody. We first tested all possible HtrA3 mAb combinations in 96-well plates to select the best pairs for the detection of both isoforms (HtrA3-T) or HtrA3-L alone, respectively. The detection mAb was biotinylated as described in the previous section. To establish an HtrA3-T ELISA, all three HtrA3 mAbs capable of detecting both HtrA3 isoforms ( Suppl. Table S1 ) were tested in all six possible combinations. For an HtrA3-L isoform-specific ELISA, all five HtrA3 mAbs ( Suppl. Table S1 ) in 14 possible combinations were tested. All these antibody combinations were first examined on conditioned media of HEK293 cells (HtrA3 negative) that were transiently transfected for the HtrA3-L or HtrA3-S isoform; pairs showing robust detection were then tested on a pooled serum sample of first trimester of pregnancy to select the best antibody combination for the detection of HtrA3-T and HtrA3-L, respectively, in human serum.
Half-area 96-well plates (Corning, Corning, NY) were coated overnight at 4 °C with 50 uL of HtrA3 mAb at 5 µg/mL in 0.1 M sodium carbonate/bicarbonate buffer, washed with PBS (137 mM NaCl, 2.7 mM KCL, 10 mM Na2HPO4, and 1.8 mM KH2PO4) containing 0.05% Tween 20 (PBS-T), and blocked with 1% bovine serum albumin (Bovogen, VIC, Australia) in PBS for 75 min at 37 °C. The wells were then washed again with PBS-T and incubated with HtrA3-containing analytes for 2 h at room temperature with gentle agitation. The wells were washed again with PBS-T and incubated at room temperature with gentle agitation first with a biotinylated HtrA3 mAb (3 µg/mL in PBS-T) for 1 h, then with streptavidin-HRP conjugate (Dako, Carpinteria, CA; diluted to final concentration of 0.78 µg/mL in PBS-T) for 50 min, and finally with one-step ultra TMB substrate (Thermo Fisher Scientific) for 10 min in the dark. The color reaction was stopped with 1 M sulfuric acid, and the absorbance at 450 nM was measured immediately on an Envision Multilabel Plate reader (PerkinElmer, Waltham, MA). All wash steps were performed using an automated plate washer (ELx50 Washer; Biotek, Winooski, VT).
The best ELISA mAb pairs for the detection of HtrA3-T (both isoforms) and HtrA3-L consisted of 10H10 and 6G6, respectively, as the capture antibodies and biotinylated 9C9 as the detection antibody. These two ELISAs were then optimized further for optimal concentrations of all reagents involved.
The standard curves for the final two HtrA3 ELISAs were constructed by serially diluting purified recombinant HtrA3-L protein 14 in PBS-T and curve fitted with five parameter logistic (5PL) nonlinear regression. For serum studies, the standards also contained 10% fetal bovine serum (FBS; Thermo Fisher Scientific). The specificity of the two ELISAs was validated on conditioned media of HEK293F cells transiently transfected for the HtrA3-L or HtrA3-S isoform and on purified recombinant HtrA family proteins HtrA1 (Biotez, Berlin, Germany), HtrA2 (R&D System, Minneapolis, MN), HtrA3 (Biotez), and HtrA4 (Biotez).
The accuracy and reliability of the final two HtrA3 ELISAs were further examined by determining assay linearity, spike and recovery, and intra-assay and interassay variations on the standard and quality control samples. Intra-assay variation was calculated from one assay of eight replicates, and interassay variation was determined from independent assays performed over multiple days. All serum samples were assayed in duplicates following 1/10 dilution in PBS-T for HtrA3-T and 1/5 dilution for HtrA3-L.
Detection of Serum Concentration of HtrA1 and HtrA4 by ELISA
HtrA1 was determined using a homemade ELISA that was previously established in our laboratory. 10 All serum samples were assayed in duplicates following 1/10 dilution in PBS-T according to the procedure as previously published. 10
Serum HtrA4 level was determined using a commercial ELISA kit (USCN Life Science Inc., China) according to the manufacturer’s protocol. Serum samples were assayed following 1/50 dilution in the standard diluents supplied.
Study Cohort
Maternal sera were collected at 11 to 13 weeks of gestation from a cohort of singleton pregnancies that proceeded with normal pregnancy (
Results and Discussion
Establishment of Two HtrA3 ELISAs
Since the HtrA3-L and HtrA3-S isoforms are identical, except the C-terminal PDZ domain is missing in the HtrA3-S isoform, 12 we aimed to develop two HtrA3 ELISAs, one for the detection of both isoforms together (HtrA3-T) and another for HtrA3-L only. To develop a sandwich ELISA for HtrA3-T, we tested all three mAbs (2C4, 9C9, and 10H10) that can detect both HtrA3 isoforms ( Suppl. Table S1 ), in a total of six possible combinations, with each mAb as the capture or the detection antibody. We first tested all of these combinations on conditioned media of HEK293 cells (negative for HtrA3) transiently transfected for HtrA3-L or HtrA3-S, respectively ( Fig. 1A , top graph). While the combination of 2C4 (capture)/9C9 (detection) did not robustly detect either of the HtrA3 isoforms and the 9C9 (capture)/2C4 (detection) combination did not detect HtrA3-L efficiently, the remaining four combinations showed efficient detection of both isoforms. These four promising pairs were then examined on a pooled serum sample of first trimester pregnancy ( Fig. 1A , bottom graph). The pair with 10H10 as the capture and 9C9 as the detection antibody showed the most robust detection in the human serum. This pair combination was selected for the establishment of an HtrA3-T ELISA for detecting both HtrA3 isoforms in the human serum.
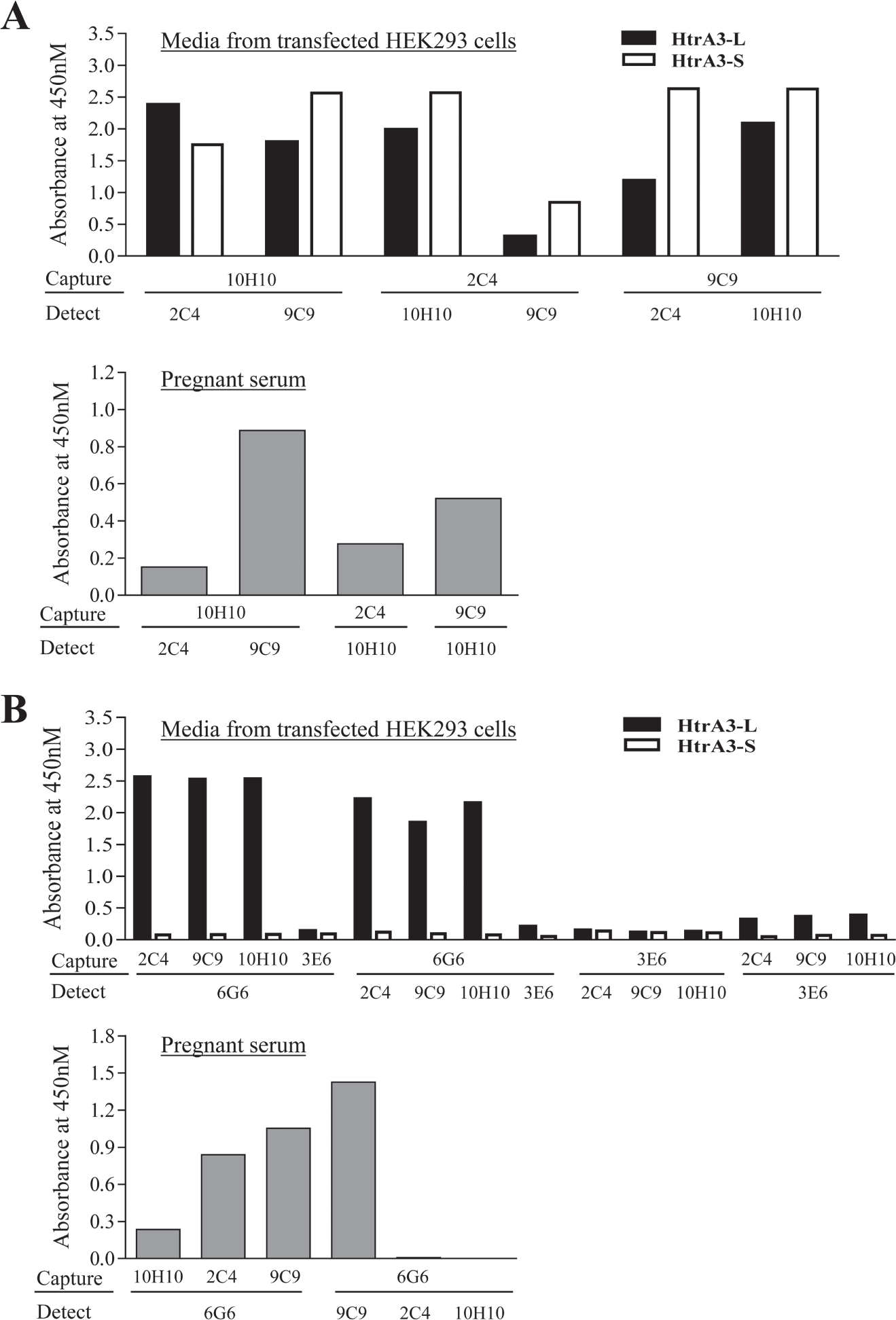
Antibody pair selection for the two HtrA3 enzyme-linked immunosorbent assays. (
To select the best antibody combinations for an HtrA3-L ELISA, we tested all five HtrA3 mAbs that detect HtrA3-L ( Suppl. Table S1 ), in all 14 possible combinations on conditioned media of HEK293 cells transiently transfected for HtrA3-L or HtrA3-S, respectively ( Fig. 1B , top graph). None detected HtrA3-S as expected, while six pairs showed robust detection of HtrA3-L. These six pairs were then tested on the pooled serum sample of first trimester pregnancy ( Fig. 1B , bottom graph). The 6G6 (capture)/9C9 (detection) pair stood out as the best combination for the detection of HtrA3-L in the pregnant serum. This pair was selected for the establishment of an HtrA3-L ELISA for HtrA3-L study in human serum.
Validation of the Specificity of the Two HtrA3 ELISAs
To confirm the specificity of the above two ELISAs, we first tested them on equal amounts of all four recombinant human HtrA family members ( Fig. 2A , B , left panel). Both HtrA3-T and HtrA3-L ELISAs detected HtrA3 only, confirming high specificity. We then tested these ELISAs on conditioned media of HEK293 cells transiently transfected for the HtrA3-L or HtrA3-S isoform, respectively ( Fig. 2A , B , right panel). The HtrA3-T ELISA detected both isoforms ( Fig. 2A , right panel). On identical conditioned media samples, the HtrA3-L ELISA detected the HtrA3-L isoform only ( Fig. 2B , right panel). These data confirmed the high specificity of these two ELISAs for HtrA3 isoforms.
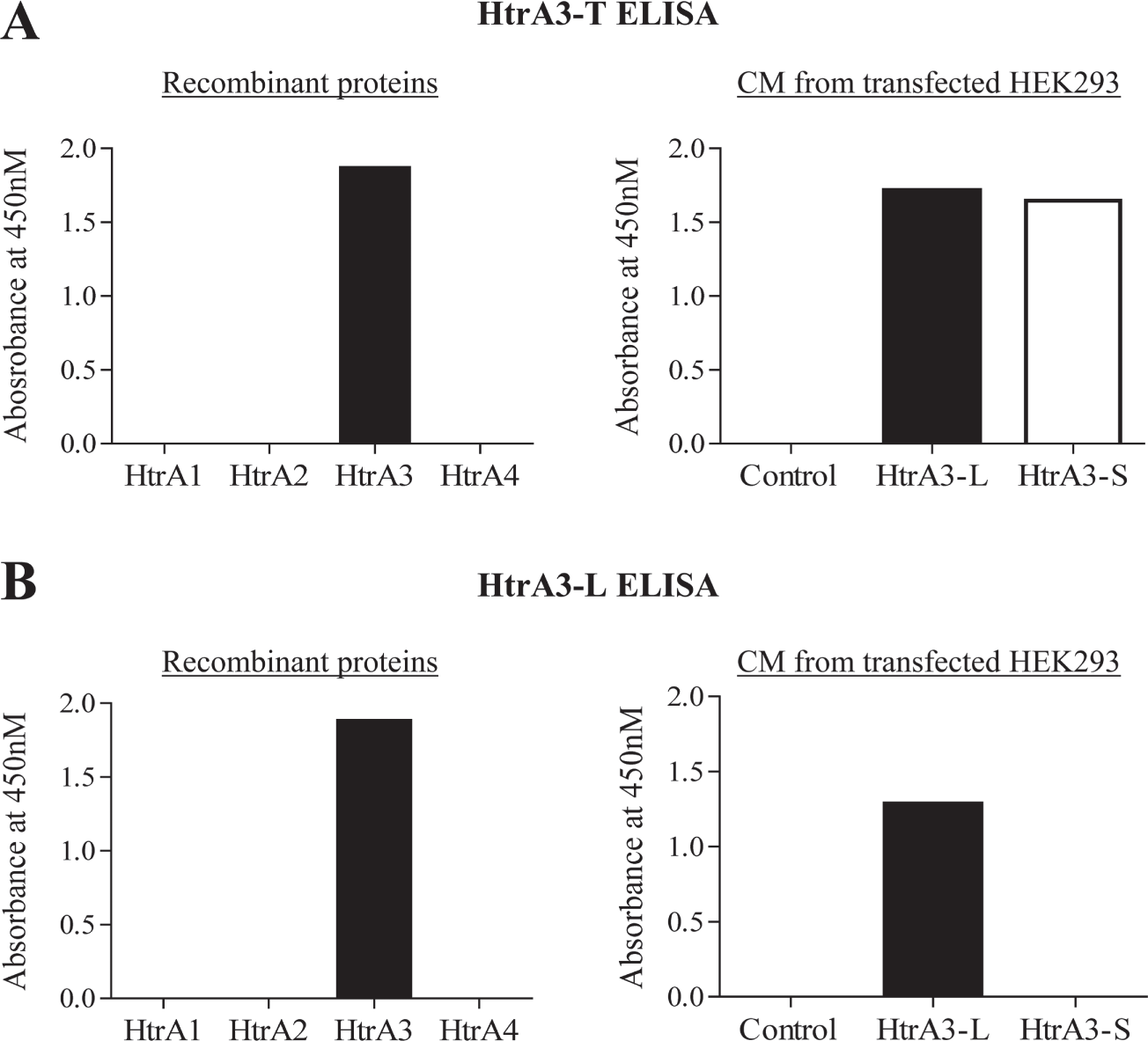
Validation of HtrA3 enzyme-linked immunosorbent assay (ELISA) specificity. Equal amounts (50 ng) of recombinant human HtrA proteins HtrA1, HtrA2, HtrA3 (long isoform), and HtrA4 were assayed, together with conditioned media of HEK293 cells transiently transfected for the HtrA3-L or HtrA3-S isoform. (
Further Optimization of HtrA3 ELISAs for Serum Detection
We first titrated all components involved in each HtrA3 ELISA to optimize their concentrations. Representative standard curves for the final ELISAs are shown in Supplementary Figure S1 (standards in PBS-T). We next tested the effect of the serum matrix on the two ELISAs. Recombinant HtrA3-L protein was serially diluted in standard diluent (PBS-T buffer) containing different concentrations of an HtrA3-negative human serum. Signals inversely correlated to serum amount, and this matrix effect is shown in Supplemental Figure S1 , where 10% serum was absent or present in the standards. We also tested FBS, which is widely available. In general, the matrix effect of FBS was equivalent to the human serum. We thus included either 10% FBS (for HtrA3-T) or 20% FBS (for HtrA3-L) in the standards when assaying human serum samples.
We next tested the linearity of the two ELISAs on conditioned media of transfected HEK293 cells and pooled pregnant serum samples. These samples were diluted to different ranges, and the concentration of HtrA3-L and HtrA3-S was determined to calculate the percentage of recovery after dilution ( Suppl. Table S2 ). For the conditioned media, both ELISAs showed adequate recovery within 1/8 dilution. For the serum samples, the HtrA3-T ELISA showed excellent recoveries even at 1/30 dilution. The lowest acceptable dilution for HtrA3-L ELISA was 1/10.
We then diluted a nonpregnant serum sample containing negligible levels of HtrA3 in PBS-T at 1/10, spiked with three different concentrations of recombinant HtrA3, and determined the recovery efficiency ( Suppl. Table S3 ). Both HtrA3 ELISAs showed excellent recovery.
To further confirm the reproducibility of these HtrA3 ELISAs, their intra- and interassay variations were determined ( Suppl. Table S4 ). Intra-assay variation was calculated from one plate of eight replicates, and interassay variation was obtained from 14 independent assays over a period of 7 days. Quality controls (QC) of pregnant serum samples containing different concentrations of HtrA3 were included in each assay. The detection range of the HtrA3-T ELISA was between 93.76 pM and 1500 pM. Within this range, both the intra- and interassay variations were less than 12% ( Suppl. Table S4 ). For the QC samples, the intra-assay variation ranged between 2% and 4%, and the interassay variation was between 10% and 22%, confirming assay reproducibility with serum samples ( Suppl. Table S4 ). The HtrA3-L ELISA detected between 46.88 pM and 1500 pM HtrA3, and its intra- and interassay variations were <12% and <19%, respectively, on the standards, confirming the reproducibility of the assay ( Suppl. Table S4 ). The QC samples showed <4% intra-assay variation and <9% interassay variation, again confirming assay reproducibility with serum samples.
Serum HtrA3 Isoform Profile at 11 to 13 Weeks of Gestation Is Different between Women Who Proceeded with Normal Pregnancies and Those Who Developed PE
We next assayed human serum samples collected from a cohort of singleton pregnant women who either proceeded with normal pregnancies (
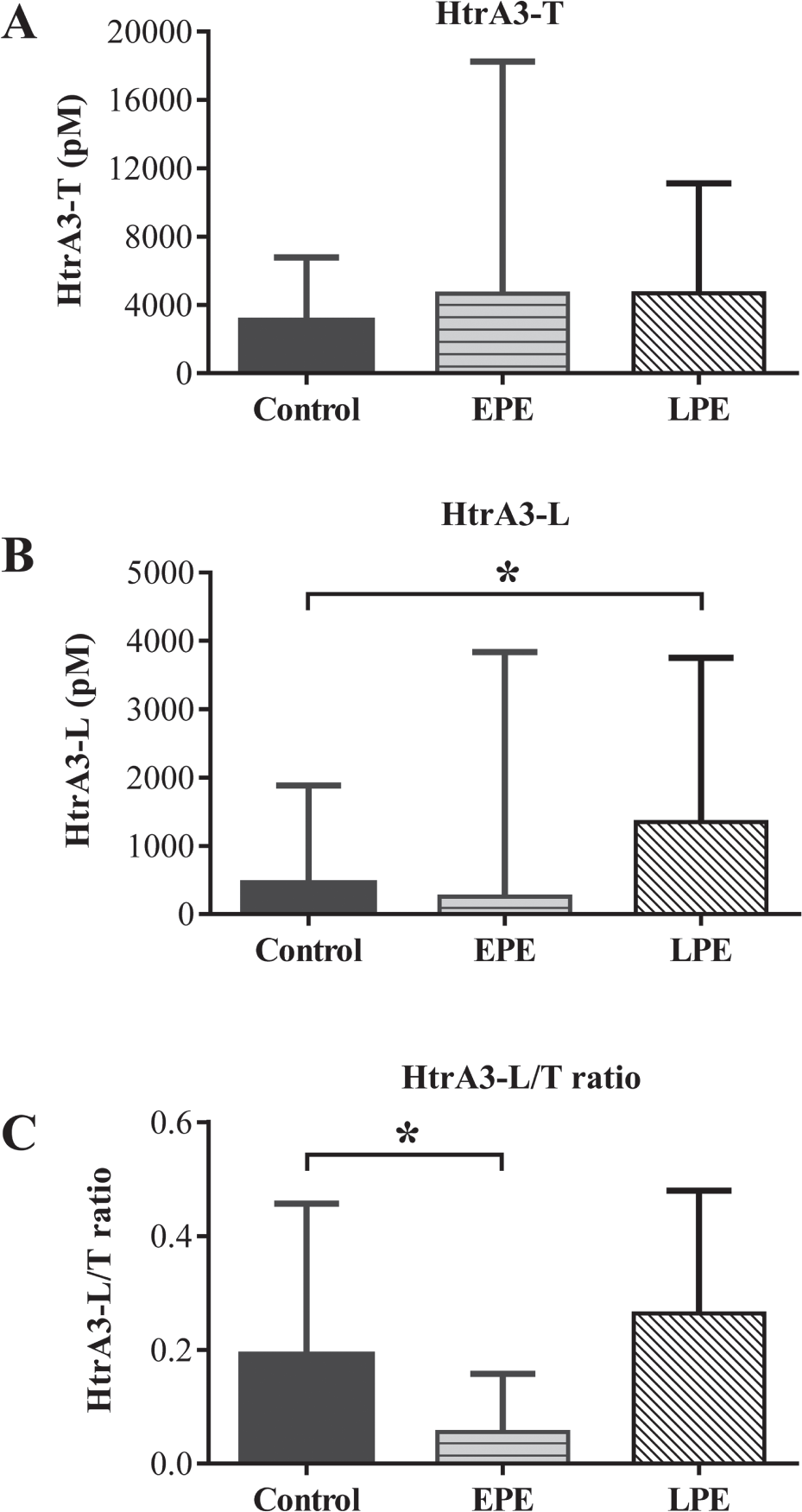
Serum concentration of HtrA3 isoforms at 11 to 13 weeks of gestation in women who proceeded to term without complication or developed preeclampsia (PE) in the third trimester of pregnancy. HtrA3 serum concentrations were determined by the two newly developed HtrA3 enzyme-linked immunosorbent assays. Control,
We then performed receiver-operating curve (ROC) analysis on these data for the prediction of late-onset and early-onset PE ( Fig. 4 ). The area under the curve (AUC) for late-onset PE prediction with HtrA3-L was 0.6096 ( Fig. 4A ). The HtrA3-L/HtrA3-T ratio produced an AUC of 0.7870 for the prediction of early-onset PE ( Fig. 4B ). These data suggest that the ratio of HtrA3-L/HtrA3-T is a potential indicator for early detection of early-onset PE. Future studies will test whether combining HtrA3 isoforms with other makers will increase the predictability of different subtypes of PE.
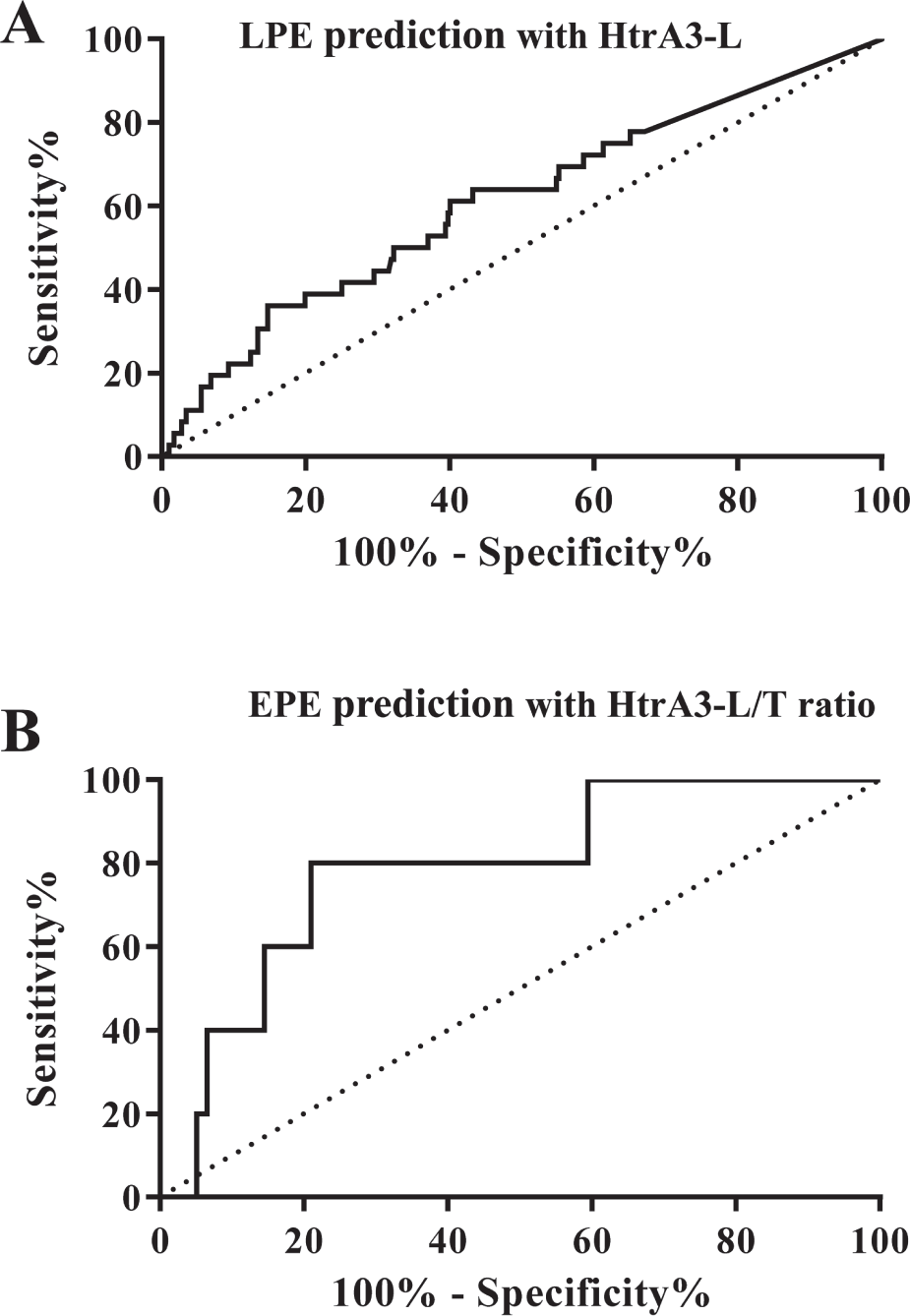
Receiver-operating characteristic curves of serum HtrA3-L or HtrA3-L/T ratio at 11 to 13 weeks of gestation for the prediction of later development of late-onset or early-onset preeclampsia (PE). (
We have previously shown by Western blot analysis of a small serum cohort that HtrA3 levels at 13 to 14 weeks of gestation are significantly higher in women who subsequently developed PE compared with controls. 13 Our HtrA3-L ELISA results confirmed this finding for late-onset PE. The current study also indicates the potential utility of these HtrA3 ELISAs to identify women who are at risk of developing the two different subtypes of PE before the onset of symptoms. Correctly identifying those with a risk of PE in the first trimester has great significance, as it will allow clinicians to initiate appropriate interventions early to reduce the risk.
Serum Levels of HtrA1 and HtrA4 at 11 to 13 Weeks of Gestation Did Not Differ between Women Who Proceeded with Normal Pregnancy and Those Who Developed PE
We also examined the serum levels of HtrA1 and HtrA4 in identical samples used for HtrA3 assays ( Suppl. Fig. S2 ). Neither HtrA1 ( Suppl. Fig. S2A ) nor HtrA4 ( Suppl. Fig. S2B ) showed significant differences between controls and any PE subtypes. We have previously demonstrated that serum concentrations of these two HtrAs are significantly higher in women with early-onset PE at the time of disease presentation.9,10 The current study suggests that serum levels of HtrA1 and HtrA4 change after 11 to 13 weeks of gestation in those who later develop PE and that these two HtrAs may be useful in distinguishing the two PE subtypes at the time of disease presentation but not useful to predict PE in the first trimester.
We have successfully established two highly specific and sensitive HtrA3 isoform ELISAs to determine the serum concentration of both HtrA3 isoforms together and HtrA3-L alone. These two ELISAs showed distinctive serum profiles of HtrA3 isoforms at 11 to 13 weeks of gestation in women who proceeded with normal pregnancy or developed PE in the third trimester. These two HtrA3 ELISAs may offer novel diagnostic tools for early detection of PE, especially the early-onset PE cases. Further studies will determine whether combining HtrA3 isoforms with other biochemical and biophysical markers will offer superior detection of PE in early pregnancy. Furthermore, as HtrA3 levels are reported to be reduced in various cancer cells, 15 these HtrA3 isoform–specific ELISAs may be applicable to other fields, such as cancer research.
Supplemental Material
Wang_JBS_Supplemental_material_-_9th_Nov_2016 – Supplemental material for HtrA3 Isoform–Specific ELISAs for Early Detection of Preeclampsia
Supplemental material, Wang_JBS_Supplemental_material_-_9th_Nov_2016 for HtrA3 Isoform–Specific ELISAs for Early Detection of Preeclampsia by Yao Wang, Ying Li, Jonathan Hyett, Fabricio da Silva Costa and Guiying Nie in Journal of Biomolecular Screening
Footnotes
Acknowledgements
The authors thank all the patients who donated blood samples for this study.
Supplementary material is available online with this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Health and Medical Research Council of Australia (fellowship No. 1041835 and project grant No. 1108365 to G.N.), Bill and Melinda Gates Foundation, and the Victorian State Government Operational Infrastructure Scheme.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
