Abstract
Monoacylglycerol acyltransferase (MGAT) activity catalyzes the synthesis of diacylglycerol (DAG) from fatty acyl-CoA and monoacylglycerol as substrates. It is important for the resynthesis of triacylglycerol (TAG) in the intestine. In the present study, we developed a MGAT enzymatic assay of human intestinal microsomes using a high-throughput mass spectrometry (MS)–based detection system. After screening with small-molecular-weight libraries for compounds exhibiting inhibitions against DAG and the consequent TAG syntheses, we identified multiple compounds that specifically inhibit intestinal MGAT activity. The inhibitory activities of these compounds were correlated to those determined using a recombinant human MGAT2 enzyme. An aryl-sulfonamide compound T1 showed potent inhibitory activity toward human intestinal MGAT and recombinant human MGAT2, with selectivity over MGAT3. This high-throughput MS-based assay provides a novel platform for the discovery of DAG or TAG synthesis inhibitors. The identified aryl-sulfonamide compound T1 is a promising starting compound for optimization studies of inhibitors with selectivity toward MGAT2.
Keywords
Introduction
Obesity is a metabolic disorder resulting from the increase in body fat. The prevalence of obesity has been steadily increasing in developed countries, with important consequences to health and society. 1 Although obesity is closely linked to genetic backgrounds, environmental factors have significant impacts on the development of obesity. Among others, imbalance between energy intake and expenditure leads to metabolic abnormalities such as obesity and metabolic diseases. In fact, the consumption of a high-fat diet increases the likelihood of obesity. 2 Therefore, the modification of fat intake is considered to be a potential therapy for the prevention of obesity.
Fat is a major source of energy in the human diet, and more than 95% of dietary fat is long-chain triacylglycerol (TAG). 3 TAG is efficiently absorbed into the body through sequential metabolism involving multiple successive and coordinated processes. First, TAG is hydrolyzed to monoacylglycerol (MAG) and free fatty acids by pancreatic lipase before its absorption into intestinal enterocytes. Next, MAG is solubilized and absorbed into intestinal enterocytes, followed by reesterification by intracellular enzymes, where diacylglycerol (DAG) and TAG are sequentially synthesized.4,5 Synthesized TAG is secreted into the systemic circulation as chylomicrons to provide and store energy in other tissues. Multiple enzymes or proteins are involved in the metabolism of TAG, and these have attracted drug discovery research for the treatment of metabolic diseases. 6
Monoacylglycerol acyltransferases (MGATs) (EC 2.3.1.22) are involved in the reesterification of MAG in the intestine. Three homologous genes have been identified in human or rodent genomes (i.e., MGAT1, MGAT2, and MGAT3), 7 which possess DAG synthesis activity, using MAG and fatty acyl-CoA as substrates. Sequential synthesis of TAG starting from the MGAT reaction with the acylation of MAG is known as the MGAT pathway, in contrast to the glycerol 3-phosphate pathway. The MGAT pathway is considered to be important for TAG synthesis in each tissue. 5 However, it has been shown that each MGAT subtype possesses distinct substrate specificity and synthesizes DAG, TAG, or both. 8 These features of intestinal MGAT subtypes have complicated the identification of the subtype responsible for MGAT activity in the intestine. Therefore, intestinal MGAT activity remains to be clarified and attributed to each MGAT subtype.
MGATs are considered to be potential targets for small-molecule inhibitors. Conventionally, the enzymatic activity of MGAT has been investigated by radioisotope-based assays, including the scintillation proximity assay (SPA) 9 and thin-layer chromatography (TLC) separation, or by the thiol detection assay. 10 Although the TLC assay is unique among the conventional assays in its ability to detect specific reaction products, it is a low-throughput, labor-intensive, and high-cost method that is not suitable for investigations requiring a high-throughput assay, particularly where there is a need to screen against chemical libraries with millions of compounds. Therefore, a robust assay system is required to facilitate the discovery and development of MGAT inhibitors.
Mass spectrometry (MS) combined with the RapidFire System (Agilent Technologies, Lexington, MA) is an emerging technique for high-throughput screening. The MS-based evaluation methods for MGAT2 inhibitors in cellular and biochemical assays have recently been reported.11,12 We applied the high-throughput MS system to identify intestinal MGAT inhibitors. We used crude human intestinal microsomes as an enzyme source and performed random screening in a physiologically relevant setting. This novel system enabled the simultaneous detection of DAG and TAG and the identification of intestinal MGAT inhibitors.
Materials and Methods
Materials
The substrates 2-oleoyl-glycerol and 13C18-oleoyl-CoA and an internal standard 1,2-dioleoyl-sn-glycerol were purchased from Sigma-Aldrich (St. Louis, MO). Mixed human intestine microsomes were purchased from XENOTECH (Lenexa, KS).
Protein Preparation
The full-length coding sequences of human MGAT2 and human MGAT3 are identical to NCBI accession numbers NM_025098 and NM_001109436, respectively. The respective sequences were subcloned into the pcDNA3.3 vector (Life Technologies, Carlsbad, CA) to generate expression plasmids in mammalian cells. To prepare overexpressed membranes, the expression vector was transiently transfected into COS-7 cells. After culture for 2 days, cells were collected and homogenized in ice-cold 20 mM Tris-HCl buffer (pH 8.0) containing 1 mM EDTA and 300 mM sucrose. Cell homogenates were centrifuged (2000 rpm, 10 min, 4°C), and the supernatant was recovered. Total membrane fractions were isolated by ultracentrifugation (45,000 rpm, 60 min, 4°C). Pellets were resuspended in the same buffer and stored at −80°C. The protein concentration was determined using the BCA Protein Assay Kit (Pierce Biotechnology, Rockford, IL) according to the instruction manual.
RapidFire Mass Spectrometry Assay
For the assays of human intestinal microsomes, recombinant human MGAT2, and recombinant human MGAT3, the enzymes were preincubated for 15 min with a tested compound in 10 µL of an assay buffer (100 mM Tris-HCl [pH 7.5], 5 mM MgCl2, 200 mM sucrose, 0.01% Tween 20, 2 mM DTT, 0.01% bovine serum albumen [BSA], and 5% DMSO), followed by addition of 10 µL of the substrate mixture containing 10 µM 13C18-oleoyl-CoA (Sigma-Aldrich) and 10 µM 2-oleoyl-glycerol (Sigma-Aldrich) to start the reaction in a 384-well assay plate. The substrate 2-oleoyl-glycerol was dissolved in ethanol at 10 mM and added to the substrate mixture.The human intestinal microsomes were reacted at 1 µg/mL. Human MGAT2 and MGAT3 were reacted at 10 µg/mL. After the enzyme reactions for 30 min at room temperature, 5 µL of stop solution (8.3% formic acid in the assay buffer) was added, followed by 50 µL of 2-propanol containing 167 nM 1,2-dioleoylglycerol (Sigma-Aldrich) and 167 nM trioleoylglycerol (MP Biomedicals, Santa Ana, CA) as internal standards. The samples were mixed and centrifuged to precipitate the proteins. High-throughput online solid-phase extraction was performed using the RapidFire 300 System (Agilent Technologies, Santa Clara, CA). The samples were loaded onto an SPE C4 cartridge (Agilent Technologies) using 10 mM ammonium formate and 0.2% formic acid in acetonitrile/deionized water (30/70 v/v) at a flow rate of 1.5 mL/min and were eluted using 5 mM ammonium formate and 0.2% formic acid in acetonitrile/acetone/deionized water (75/25/2 v/v/v) at a flow rate of 1.25 mL/min. The injection needle was washed with deionized water, followed by acetonitrile. The aspiration time (injection volume 10 µL), load/wash time, elution time, and reequilibration time were adjusted to 500, 2500, 5000, and 1000 ms, respectively, for a total cycle time of approximately 10 s. For the kinetics measurements, the same reaction conditions were used as those described. The reaction samples were subjected to a high-throughput liquid chromatography–mass spectrometry (LC/MS) assay, where separation conditions were based on the RapidFire assay, except that the combination of an LC system (Shimadzu, Tokyo, Japan) and a switching valve was used.
Mass spectrometric analysis of analyte formation was performed using an API-5000 triple quadrupole mass spectrometer (AB SCIEX, Framingham, MA) equipped with an electrospray ion source (TurboIon Spray) operated in positive selected reaction monitoring (SRM) mode. The SRM transitions for 13C18-dioleoylglycerol as a reaction product and 1,2-dioleoylglycerol as an internal standard were set as 656.6/339.2 and 638.5/339.2, respectively. The SRM transitions for 13C36-trioleoylglycerol as a reaction product and trioleoylglycerol as an internal standard were set as 939.3/621.5 and 903.2/603.2, respectively. Typically, the SRM conditions for DAG species were set as follows: declustering potential (DP), 161 V; collision energy (CE), 25 V; and collision exit potential (CXP), 12 V. Parameters were optimized as follows: capillary temperature, 500°C, and ion spray voltage, 5.5 kV. The collision gas, curtain gas, ion source gas 1, and ion source gas 2 pressures were set at 4, 15, 60, and 60 psi, respectively. Analytical data were acquired and analyzed using Analyst software (version 1.5.0; AB SCIEX).
The inhibitory activity was calculated as follows: % inhibition = (A − B)/(A − C) × 100, where A, B, and C are the peak area ratio (reaction product divided by its internal standard) with vehicle, with the test sample, and with full inhibition, respectively. The dose–response data were then fitted to a four-parameter logistic curve using GraphPad Prism (GraphPad Software, San Diego, CA) to determine IC50 values.
Results
Reaction Progress Curves of Recombinant Human MGAT2 and Intestinal MGAT Activity
Multiple reaction monitoring (MRM) conditions were established to monitor 1,2-dioleoylglycerol and trioleoylglycerol at m/z 638.5/339.2 and 903.2/603.2, respectively. The limits of quantitation for 1,2-dioleoylglycerol and trioleoylglycerol were 200 and 67 fmoles on column with a signal/background ratio of more than 10, respectively. These were used as internal standards at concentrations giving robust signals compared with the backgrounds within dynamic ranges (
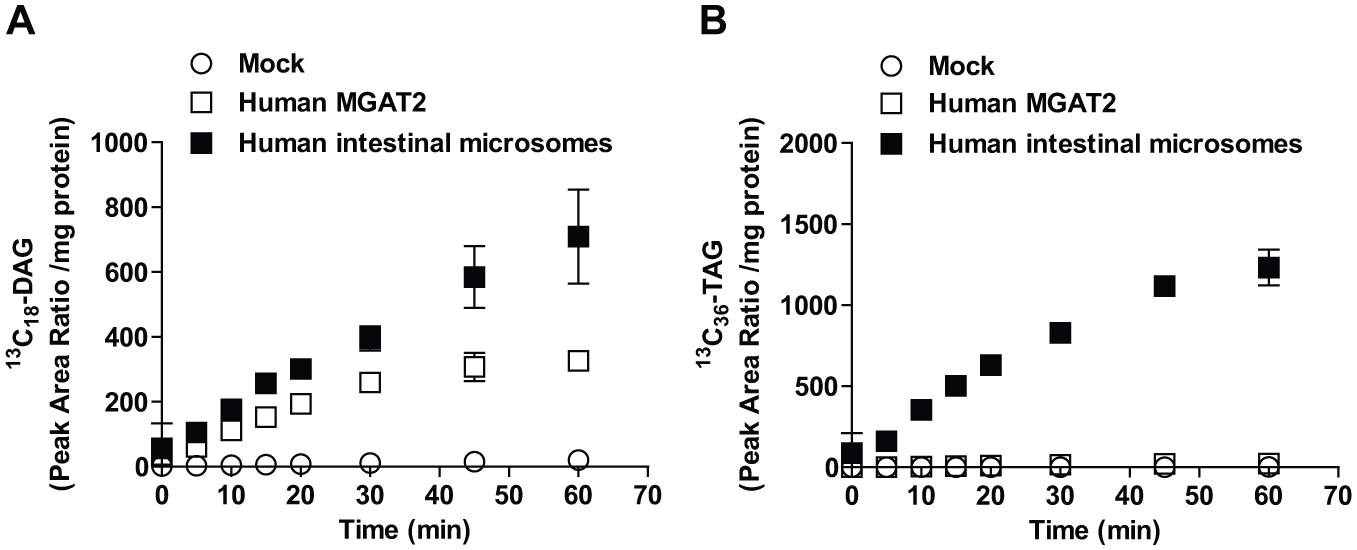
Detection of MGAT activity of recombinant human MGAT2 and human intestinal microsomes using the LC/MS assay, which monitored DAG and TAG. (
Characterization of Recombinant Human MGAT2 and Intestinal MGAT Activity
To further characterize recombinant and intestinal MGAT reactions, saturation curves with oleoyl-CoA were analyzed using MS-based monitoring of DAG and TAG with 2-oleoyl-glycerol as an acyl donor to monitor MGAT reactions. Recombinant human MGAT2 reacted with oleoyl-CoA with a Michaelis–Menten kinetic constant of 2.3 µM (95% CI 1.7–2.9) for Km in the DAG synthesis reaction ( Fig. 2A ), although no saturable enzyme activity was observed in TAG synthesis ( Fig. 2B ). In contrast, human intestinal MGAT showed Km values of 0.60 µM (95% CI 0.38–0.81) in the DAG synthesis reaction ( Fig. 2A ) and 6.8 µM (95% CI 5.6–8.0) in the TAG synthesis reaction ( Fig. 2B ). Thus, human intestinal microsomes showed a distinctive character in the robust TAG synthesis activity compared with recombinant human MGAT2. In the screening and following assays, MGAT reactions were conducted within the linear range of progress curves using 5 µM oleoyl-CoA and 5 µM 2-oleoyl-glycerol as substrates.
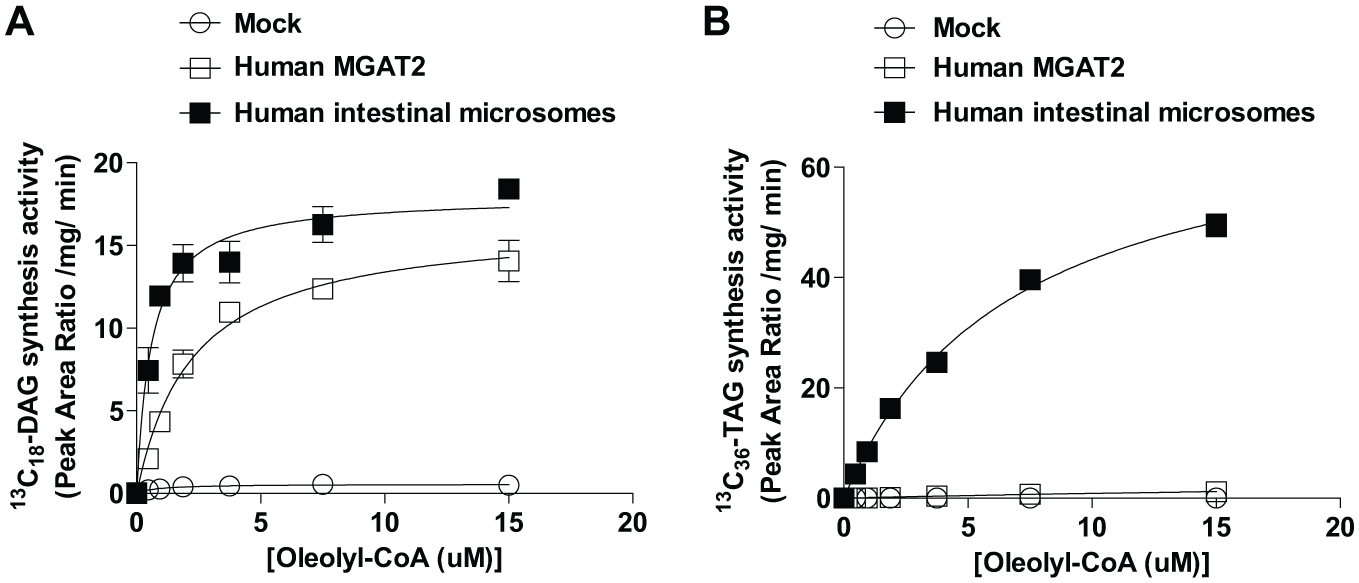
Determination of the kinetics constants for oleoyl-CoA with MGAT reactions of recombinant human MGAT2 and human intestinal microsomes. (
High-Throughput Screening for MGAT Inhibitors Using RapidFire MS Assay
The screening campaign was conducted with the MGAT activity of human intestinal microsomes on more than 500,000 compounds at a single concentration of 1 μM. RapidFire assays were performed with an average Z′ factor ± standard deviation of 0.70 ± 0.053 and 0.83 ± 0.059 for the detections of DAG and TAG, respectively. In addition, the signal/background ratio was greater than 8.0 for both assays during the screening campaign. Dual measurement with MS detection enabled the identification of specific MGAT inhibitors, with separation from diacylglycerol acyltransferase (DGAT) inhibitors. MGAT inhibitors were identified by the characteristic inhibitory activities in both DAG and TAG synthesis, while DGAT inhibitors were characterized by the accumulation in DAG (data not shown). After the screening, compounds with inhibitory activities in both DAG and TAG synthesis (DAG > 20% and TAG > 30%) were selected for further evaluations.
Characterization of Diverse Intestinal MGAT Inhibitors with MGAT Subtype Assays
Selected compounds included diverse chemical structures and potencies. In the dose–response assay, the compounds showed inhibitory activities for both DAG and TAG synthesis of intestinal MGAT reactions with correlated potencies (r 2 = 0.88, p < 0.0001; Fig. 3A ). This characteristic is a distinctive feature of MGAT inhibitors rather than DGAT inhibitors. Next, compounds were subjected to recombinant human MGAT2 and MGAT3 assays to examine their inhibitory activities to those MGAT subtypes. Notably, inhibitory activities in the intestinal MGAT assay were significantly correlated to those in the recombinant human MGAT2 assay (r 2 = 0.69, p < 0.0001; Fig. 3B ) but not to those in the recombinant human MGAT3 assay (no significant correlation; Fig. 3C ). These results suggested that the screened compounds were MGAT2 inhibitors with their inhibitory activities correlated to those in human intestinal microsomes.
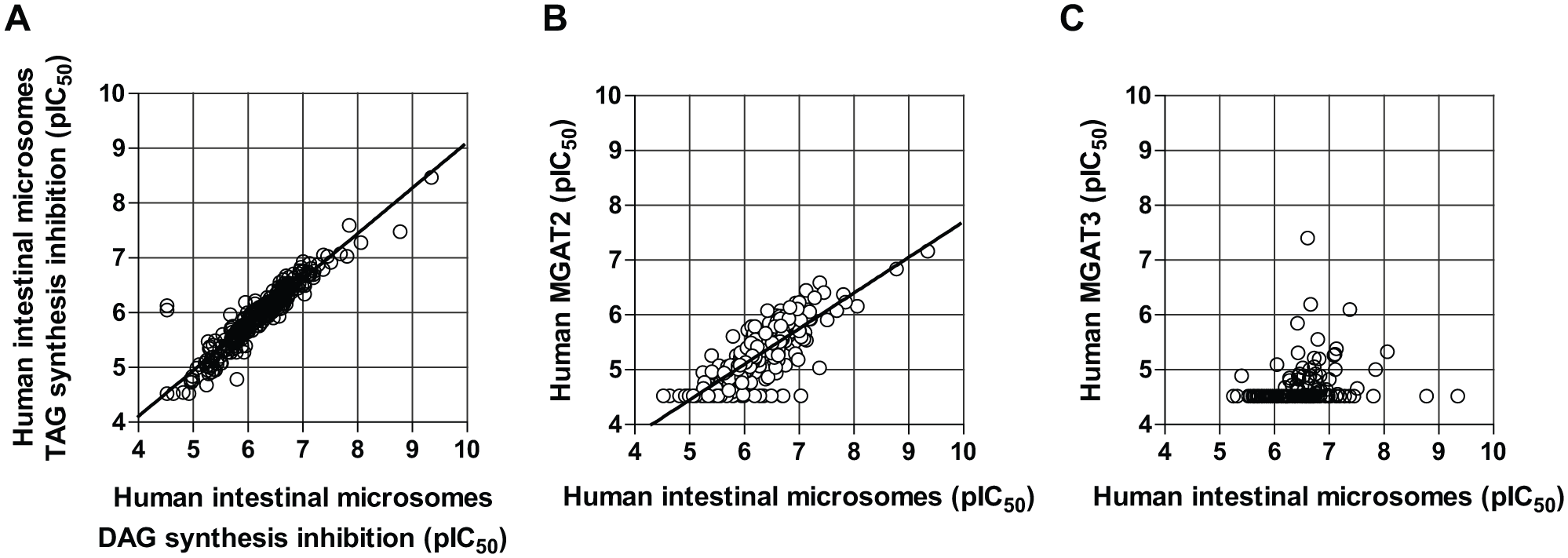
Comparison of inhibitory activities against human intestinal MGAT, recombinant human MGAT2, and recombinant human MGAT3. (
A Novel Aryl-Sulfonamide MGAT Inhibitor as a Representative Hit Compound
As described above, the RapidFire assay identified multiple compounds with diverse chemical structures and potencies from an in-house compound library. Among them, an aryl-sulfonamide compound T1 ( Fig. 4 ) showed potent inhibitory activity against human intestinal MGAT with an IC50 value of 21 nM (95% CI 12–34) ( Table 1 ). T1 showed comparable inhibitory activity against recombinant human MGAT2 with an IC50 value of 83 nM (95% CI 67–100). The inhibitory activity against MGAT3 was less than 50% at 30 µM, showing its selectivity over MGAT3.
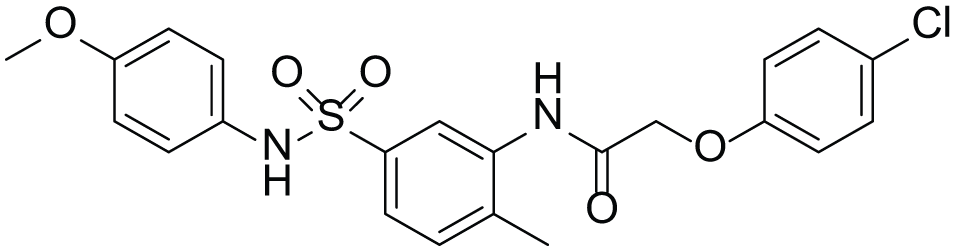
Chemical structure of compound T1.
Inhibitory Activities of Compound T1 against Related MGAT Enzymes.

IC50 values obtained from quadruplicate experiments are presented with 95% confidence intervals in parentheses.
Discussion
The high-throughput MS assay is a powerful technique to monitor enzyme activity. The MS-based enzymatic assay is characterized by simultaneous and specific detection of DAG and TAG in a single reaction sample. In the present study, this feature of MS detection was crucial because crude microsomes were used as an enzyme source with complex matrices that contained MGAT activity. Furthermore, the MS-based MGAT assay was more robust than a fluorescence assay using a reducing reagent. Consequently, the screening campaign was conducted with remarkable stability and robustness.
The enzymatic activities of recombinant human MGAT2 and intestinal microsomes were compared. Human intestinal microsomes showed successive TAG synthesis activity compared with recombinant human MGAT2 when 2-MAG was used as an acyl acceptor ( Fig. 1 ). This feature might reflect the status of MGAT activity in the human intestinal microsomes that we used. One possible explanation for the different behavior is that other endogenous acyltransferase activities are coupled with MGAT activity in human intestinal microsomes. 13 The presence of downstream activity such as DGAT activity might affect the overall DAG and TAG synthesis activities. 14 It is noteworthy that the MS-based assay enabled robust and specific detection of MGAT reaction products and the use of more physiologically relevant enzyme sources with an excellent screening performance.
From the results of our screening campaign, we gained insights into the role of MGAT2 in human intestinal microsomes. We identified intestinal MGAT-specific inhibitors from our compound library rather than DGAT inhibitors by monitoring both TAG and DAG synthesis. Importantly, selectivity assays for these MGAT inhibitors showed a positive correlation of inhibitory activities of diverse MGAT inhibitors against recombinant human MGAT2, but not against recombinant human MGAT3 ( Fig. 3 ). Weakened potencies and variations may have been the result of differences in inhibition modes or solubilities among diverse compounds. It is noteworthy that we were not able to detect the enzymatic activity of referenced human MGAT1 (NM_058165) in our assay, while we were able to detect enzymatic activities of human MGAT2 and MGAT3. Collectively, we assume that the inhibitory activities of intestinal MGAT inhibitors are attributable to their inhibitory activities against human MGAT2 enzyme in intestinal microsomes. In accordance with this, MGAT2 has been reported to be highly expressed in the human intestine. 15 Therefore, in our assay conditions, we consider MGAT2 to be a promising molecular target among human MGAT subtypes to effectively suppress human intestinal MGAT activity.
Compound T1 was identified as a potent intestinal MGAT inhibitor on the basis of in-house library screening ( Table 1 ). Among the acyltransferases examined, the most potent inhibitory activity of compound T1 was shown with MGAT2. Compound T1 showed a more than 360-fold selectivity over MGAT3. Thus, the aryl-sulfonamide hit compound T1 was considered to be a good starting point to develop MGAT2 inhibitors.
Recently, MGAT2 inhibitors have been reported by multiple groups, including us.11,16–18 Notably, our screening campaign using human intestinal microsomes identified novel MGAT2 inhibitors with confirmed inhibitory activities against human intestinal MGAT. Therefore, the lead compound is a useful chemical scaffold for further optimization of MGAT2 inhibitors for the suppression of lipid synthesis in the intestine.
In conclusion, we have shown that dual detection of TAG and DAG in the crude intestinal microsomes by high-throughput MS is a powerful strategy to identify intestinal MGAT inhibitors. As a result of the screening, we have identified diverse intestinal MGAT inhibitors that possess comparable inhibitory activities against the human MGAT2 subtype. Developing small-molecule inhibitors of MGAT2 with sufficient potency and selectivity is considered to be a viable approach to target intestinal MGAT activity.
Footnotes
Acknowledgements
We acknowledge Satoko Minami for the data analysis, Shoichi Okubo for the plasmid preparation, and Junji Matsui and Naoki Tarui for their support of this study.
Supplementary material for this article is available online.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
