Abstract
Connexin 43 (Cx43), the predominant gap junction (GJ) protein, directly interacts with the A-kinase-anchoring protein (AKAP) Ezrin in human cytotrophoblasts and a rat liver epithelial cells (IAR20). The Cx43-Ezrin–protein kinase (PKA) complex facilitates Cx43 phosphorylation by PKA, which triggers GJ opening in cytotrophoblasts and IAR20 cells and may be a general mechanism regulating GJ intercellular communication (GJIC). Considering the importance of Cx43 GJs in health and disease, they are considered potential pharmaceutical targets. The Cx43-Ezrin interaction is a protein-protein interaction that opens possibilities for targeting with peptides and small molecules. For this reason, we developed a high-throughput cell-based assay in which GJIC can be assessed and new compounds characterized. We used two pools of IAR20 cells, calcein loaded and unloaded, that were mixed and allowed to attach. Next, GJIC was monitored over time using automated imaging via the IncuCyte imager. The assay was validated using known GJ inhibitors and anchoring peptide disruptors, and we further tested new peptides that interfered with the Cx43-Ezrin binding region and reduced GJIC. Although an AlphaScreen assay can be used to screen for Cx43-Ezrin interaction inhibitors, the cell-based assay described is an ideal secondary screen for promising small-molecule hits to help identify the most potent compounds.
Keywords
Introduction
Connexin 43 (Cx43) is a gap junction (GJ) protein that makes hexameric pores in the cell membrane that dock together and form functional GJs between adjacent cells. The junctions are permeable for molecules smaller than 1 kDa, including cyclic adenosine monophosphate (cAMP), cyclic guanosine monophosphate and Ca2+. GJ intercellular communication (GJIC) plays important roles in many physiological cell processes. It has been linked to cancer progression, where Cx molecules are implicated in tumor progression/suppression and metastasis. 1 Cx43 is the predominant Cx in many different primary cells and cell lines. Because of the importance of GJs in health and disease, they are considered as potential pharmacological targets. There are clinical trials currently testing compounds regulating Cx43 GJ formation, reviewed by Grek and others. 2 However, because of Cx ubiquity and complexity in expression, concerns about the drug’s specificity, selectivity, and off-target effects mean further chemical space has to be explored.
GJIC regulation is a highly controlled process, and GJ permeability depends on numerous factors including Cx43 phosphorylation. 3 A-kinase anchoring proteins (AKAPs) are crucial for spatial and temporal organization of many signalosomes. 4 One such AKAP, Ezrin, is shown to be involved in regulation of GJ gating. Pidoux et al. 5 reported for the first time on a Cx43-Ezrin-protein kinase (PKA) complex that controls GJIC in human placental trophoblasts. They proposed a model of regulation of GJIC in which Ezrin-anchored PKA phosphorylates Cx43, which opens the GJ pores. In placental cytotrophoblasts, the GJ opening is necessary to promote cytotrophoblast cell fusion where fusogenic signals are transferred via GJs. This is a human chorion gonadotropin–driven, cAMP-mediated process. 5 We recently showed that this complex regulating GJIC is not specific just for fusogenic placental cells but also has been found in rat liver epithelial cells (IAR20), which form stable GJs. 6 IAR20 cells express Cx43, which forms GJ plaques in the membrane. To our knowledge, Cx43 is the only Cx isoform in this cell line.7,8 We performed gap-fluorescence recovery after photobleaching (FRAP) experiments to test the hypothesis that GJ regulation occurs in the same manner as in cytotrophoblasts, leading to channel opening when Cx43 is phosphorylated by Ezrin-anchored PKA. 6 The Gap-FRAP assay measures the recovery of fluorescence in a cell that is bleached with a strong laser, where the recovery occurs due to the influx of calcein dye through GJs from neighboring, nonbleached cells. Cx43 knock-down experiments with specific small interfering RNA showed complete loss of communication between IAR20 cells, indicating that the communication between IAR20 cells is most likely due to GJs formed by Cx43. 6
The Cx43-Ezrin interaction is a direct protein-protein interaction with known regions of interaction: 510-DDRNEEKR-517 in Ezrin and 366-RASSR-370 in Cx43. It was shown that residues D510 and R517 in Ezrin and R370 in Cx43 are required for binding, whereas substitutions D510I, R517V, and R370E abolished the interaction completely. 5 The fact that the complex is not only found in placental fusogenic cells but also in nonfusogenic IAR20 cells 6 and neonatal and adult rat cardiomyocytes as well as H9C2 and HEK cell lines 8 suggests that regulation of GJIC by Ezrin-anchored PKA is a general mechanism and opens a possibility for targeting the complex with disruptor peptides or small molecules. Consequently, it is essential to have quantitative, functional, yet high-throughput assays for testing potential GJIC inhibiting molecules or Cx43-Ezrin interaction disruptors. There are several commonly used methods for assessing GJIC in live and fixed cells: assays involving microinjection, patch clamping, scrape loading, as well as flow cytometry and imaging techniques such as gap-FRAP and LAMP. The limitation of the listed methods is the number of cells and conditions that can be tested at the time, which means that these assays are informative but not compatible with high-throughput screening (HTS). High-throughput techniques that have been reported include microfluidic chip technology9,10; intracellular Ca2+-based, codon-optimized aequorin expressed assay 11 ; and automated fluorescence imaging assay. 12 An HTS assay that has a fast and scalable experimental protocol that allows the quantification of response and lends itself to data mining is necessary in order to screen large libraries of small molecules.
Here we used the IAR20 cell line expressing the Cx43-Ezrin complex that regulates GJ communication to develop a high-throughput cell-based assay for inhibitors and therefore opening the possibility for further screening.
Materials and Methods
Cell Culture
The rat liver epithelial cell line (IAR20) was a gift from Edward Leithe at the Institute of Cancer Research, Oslo University Hospital–Radiumhospitalet, Oslo, Norway. Cells were cultured in an incubator under 37 °C and 5% CO2 conditions in DMEM high-glucose GlutaMAX medium (Gibco, Waltham, MA) supplemented with 10% fetal bovine serum (Gibco) and 1% PenStrep (Gibco). Cells were passaged twice a week by 1:10 dilution. All the experiments were done with ~90% confluent cells in 384-well plates (Greiner Bio One, Kremsmünster, Austria).
Chemical Compounds
GJ blockers: α-Chlordane, ƴ-Chlordane, Chlordane (technical mix), and 18β-glycyrrhetinic acid were purchased from Sigma (St. Louis, MO). All compounds were dissolved in DMSO and stored as 10 mM stock. The cell-permeable cAMP analog 8-CPT-cAMP was from Biolog (Hayward, CA). None of the compounds are cytotoxic.
Peptides
All peptides used were synthesized on an Intavis MultiPep robot (Intavis Bioanalytical Instruments AG, Köln, Germany), uncoupled and verified by high-performance liquid chromatography. The concentrations of the peptides were determined by amino acid analysis using an amino acid analyzer from Thermo Scientific Dionex (Waltham, MA). Ht31 R11-DLIEEAASRIVDAVIEQVKAAGAY; ppHt31 R11-DLIEEAASRPVDAVPEQVKAAGAY; PKI: R9-TYADFIASGRTGRRNAI; Cx43 wt (±R9)-DQRPSSR ASS
Dye Transfer Assay
IAR20 cells were plated in T175 flasks 2 days prior the experiments, trypsinized, washed with phosphate-buffered saline (PBS) and counted. The cell number was adjusted to 1.5 million per milliliter. One half of the cells were stained with calcein acetoxymethyl ester (calcein AM; 3 µM, Invitrogen, Carlsbad, CA), and the second half was left unstained. Calcein AM was incubated for 10 min. Unstained and calcein-stained cells were further treated the same way; washed with DPBS (+Ca2+, +Mg2+) four times with spinning in between (600g, 5 min, room temperature) to remove excess calcein. The cells were resuspended in clear DMEM medium (high glucose, HEPES, no phenol red, 21063-029; Gibco) supplemented with 10% fetal bovine serum, in the same starting volume. Compounds were dispensed in 384-well plates using the Echo automated liquid-handling system, which uses acoustic energy to eject precisely sized droplets from a source onto a microplate, without altering the compounds (Labcyte, Sunnyvale, CA). Before plating, the two cell populations were mixed at a 1:1 ratio and dispensed using a Multidrop 384 dispenser (Labsystems, Waltham, MA) with 40 µL in each well of a 384-well poly-d-lysine coated plate (Greiner).
IncuCyte ZOOM
IncuCyte ZOOM is a real-time quantitative 384-well live-cell imager with the advantage of nonperturbing imaging. Moreover, the 10× objective allows imaging the whole well surface in 384-well format, preventing any loss of information. Because the field stays the same over time, it enables the comparison between different time points and normalizing the data to itself in the same well. Another advantage of the IncuCyte ZOOM system is short image acquisition time; it takes less than 20 min for the green fluorescent protein (GFP) channel and phase contrast imaging of the whole 384-well plate. As the instrument has six positions for 384-well plates, the capacity allows for HTS.
The first image (0 h time) was acquired immediately after plating and centrifugation using the IncuCyte ZOOM (Essen BioScience, Ann Arbor, MI). Subsequent image acquisition then occurred after 2 and 4 h. Images were analyzed using ImageJ by converting them first to eight-bit and then applying an Autotreshold plugin to differentiate background from fluorescent signal. At this point, the number of particles was counted. The particle count was decided to be the most suitable parameter because calcein dye fluorescence slightly fades away over time. During the 4 h, the general fluorescence intensity was reduced; therefore, measuring and comparing fluorescence between time 0 h and 4 h was excluded as a parameter. An ImageJ macro and a Python script were created to automate the analysis. For each well, Δ particle number was calculated between times 0 h and 4 h. All experiments were repeated three times, showing very similar values for Δ particle count and with a calculated Z′ value of 0.7 from a typical assay. Variations may occur because of minor differences in the number of cells per well, time of readout, the IncuCyte ZOOM imager used and drifting during 20 min of readout time. Limitations of the assay are in use of autothreshold for defining the fluorescent signal. Furthermore, Δ particle count is an estimation of the difference in cell number between time 0 h and 4 h, which could introduce variation and reproducibility between wells with a Z′ value of 0.7.
AlphaScreen
The assay was performed as previously described. 13 Δ1-232 Cx43 was expressed with a GST tag and immobilized on donor streptavidin beads, whereas Biotin-His-Ezrin (full length) was immobilized on glutathione acceptor beads. Inhibitory peptides competed with the beads when mixed together. Three separate experiments with three replicates were performed.
Statistical Analysis
Statistical analysis was carried out using GraphPad Prism using an unpaired t test or analysis of variance where appropriate. All the data are shown as mean ± SEM (*p < 0.05; **p < 0.01; ***p < 0.005; ****p < 0.001).
Results
IncuCyte ZOOM System for Simple and Fast Imaging
The IncuCyte ZOOM instrument has been used for different applications, including cell migration, invasion, and wound healing. Here we show that the system can be used for monitoring GJIC and report its validation using reference compounds.
IAR20 cells were loaded with calcein acetoxymethyl ester (AM) dye, which is a cell-permeant dye. In live cells, the nonfluorescent calcein AM is converted to a green fluorescent calcein after acetoxymethyl ester hydrolysis by intracellular esterases. When fluorescent calcein is formed inside the cell, it will retain in the cytoplasm. Because of its small molecular weight (622 Da), it has been shown that the dye passes through GJ channels and enters other cells.
14
This mechanism allows the possibility to assess the rate of intercellular communication by testing different reagents that could potentially disturb the GJ permeability. Mixed cell populations were plated in 384-well plates with compounds dispensed in the plates via acoustic dispensing. We acquired images at time T0, immediately after plating and centrifuging the plates and then subsequently at 2 and 4 h from T0 (
Fig. 1
) to assess the GJIC over time. It was previously reported that cells need 2 h to form functional GJs.
11
Green fluorescence images were extracted and analyzed in ImageJ software (
Fig. 1
;
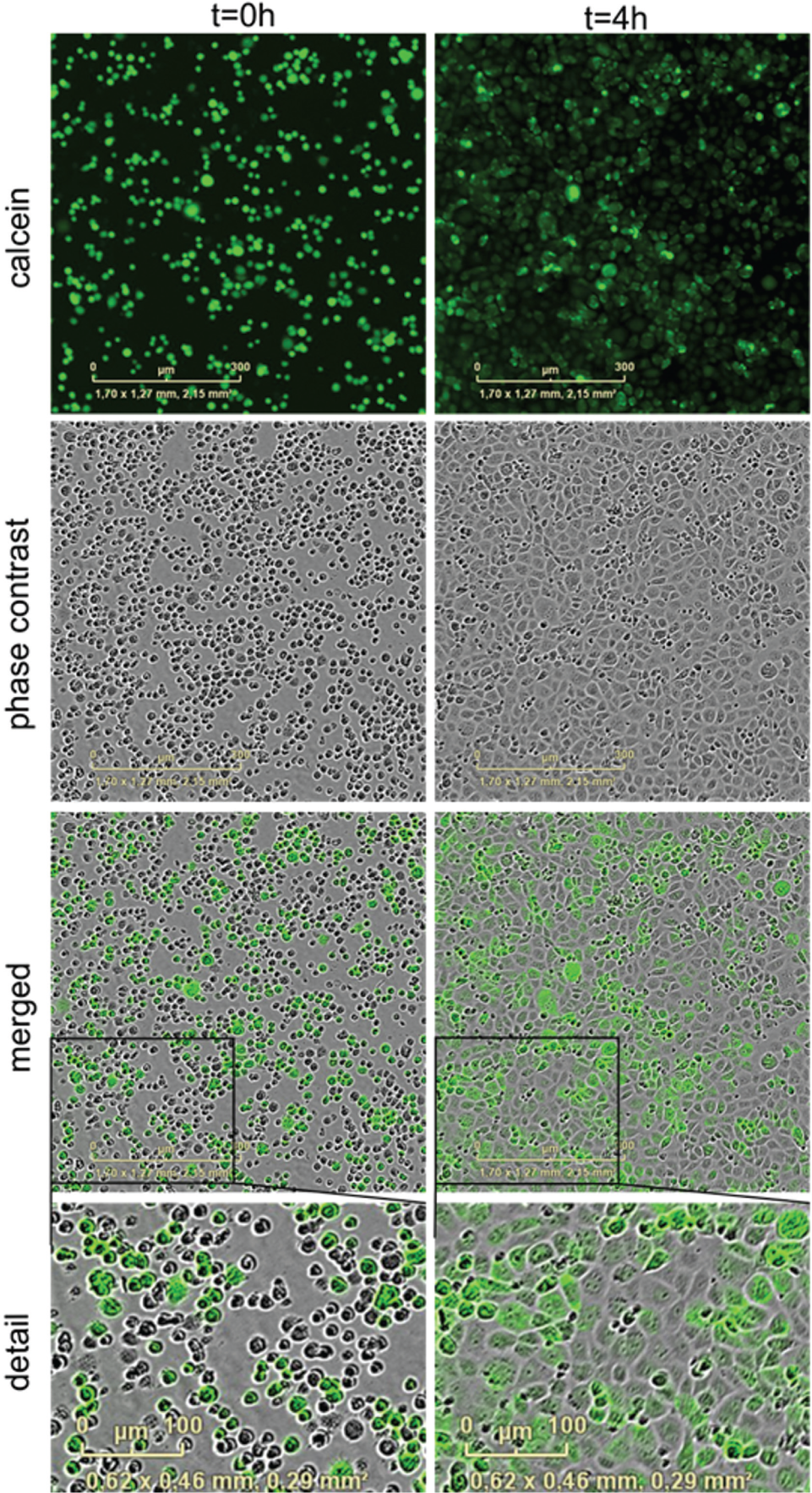
Calcein dye staining and imaging with the IncuCyte ZOOM system. IAR20 cells were stained with calcein AM dye (green) and mixed with unstained cells (1:1 ratio), at 4000 cells/well (384-well plate). Images were acquired with IncuCyte ZOOM Imager using the 10× objective. (
GJ Communication Inhibition by Known GJ Blockers
To validate the assay using known GJ blockers, IAR20 cells were treated with the range of concentrations of different compounds. Chlordane and 18-α- and 18-β-glycyrrhetinic acids (β-GA) have been shown to block GJIC in a number of assays.15–17 We selected α and ƴ Chlordane as well as Chlordane racemate, Chlordane technical mixture, and β-GA for testing ( Fig. 2A–E ). Data were expressed as Δ particle count, plotted, and concentration-dependence curves were generated. IC50 values were calculated showing potencies of the applied inhibitors similar to published results.11,12,17 In addition, cells were treated with 100 µM of each compound and compared with DMSO controls. Single data points are shown on the graph with the mean value labeled ( Fig. 2F ). A statistically significant decrease was detected with all compounds tested (p < 0.001), although ƴ Chlordane, α+ ƴ Chlordane, and β-GA were more efficacious at this concentration ( Fig. 2F ).
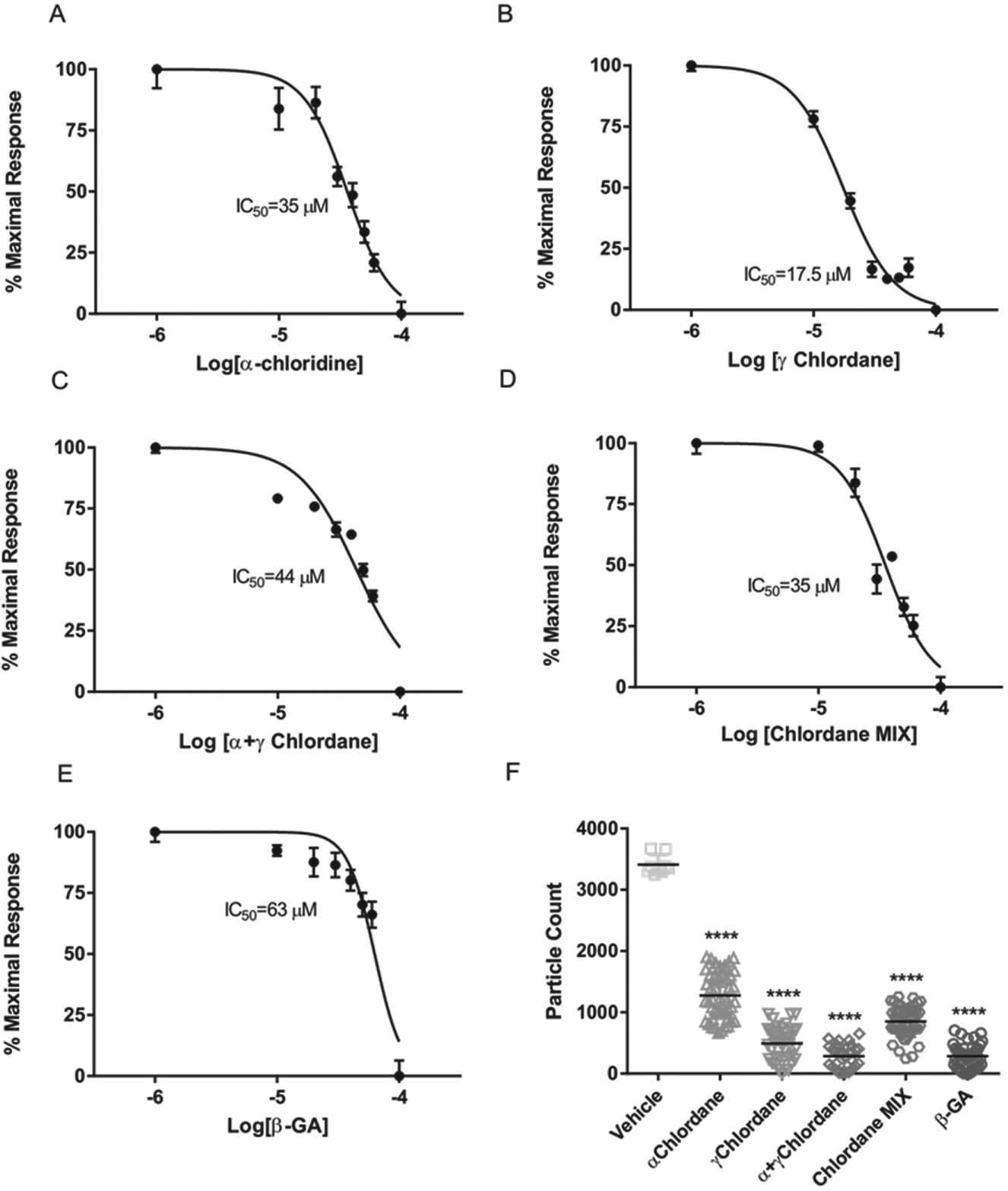
Inhibitory effect of known gap junction blockers on gap junction intercellular communication (GJIC). (
PKA Activation or Displacement Affects GJ Communication
The cAMP signaling pathway plays a critical role in many physiological processes. The Cx43-Ezrin-PKA complex is important for regulation of GJIC in different cell systems, such as fusogenic cytotrophoblasts and IAR20 cells, and may provide a general mechanism of regulation. cAMP stimulation leads to activation of PKA, which is then able to phosphorylate Cx43 when anchored to Ezrin. Thus, the calcein-based assay was used to test the effect of cAMP on GJIC. Increasing concentrations of cAMP (10 µM and 100 µM) significantly potentiated the communication compared with the basal level ( Fig. 3A ). The data are in accordance with our previous work on IAR20 as well as from other studies.18,19 cAMP treatment showed changes in Cx43 phosphorylation expressed by a shift in mobility of isoforms of Cx43 analyzed by Western blot analysis applying different antibodies against the unphosphorylated/phosphorylated form as well as a phospho-PKA antibody. 6 In cytotrophoblasts, cAMP analogues promoted formation of syncytiotrophoblast by cytotrophoblast fusion. In addition, the PKA inhibitors H89 and PKI impaired the process. 5
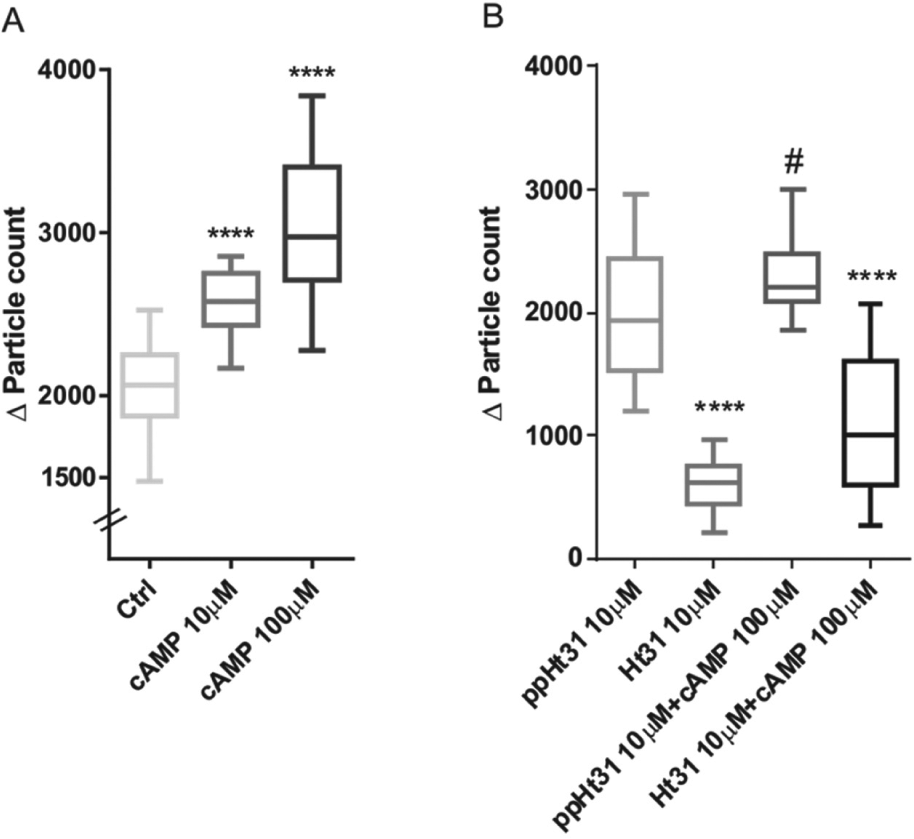
Effect of cAMP and Ht31 peptide on gap junction intercellular communication (GJIC). Effect of cAMP (
PKA is in close proximity to its substrate, here Cx43, when anchored to Ezrin, and by displacing PKA from the AKAP, the phosphorylation process is disabled. 5 Specific peptides called anchoring disruptors have the ability to displace PKA from AKAPs. The Ht31 peptide is known to be dual-specific, meaning that it is effective toward both PKA regulatory subunits, RI and RII. Ezrin is a dual-specific AKAP binding both RI and RII 20 ; therefore, we tested Ht31 in our assay. When compared with the control peptide, ppHt31, the inhibitory effect of Ht31 on dye transfer and communication is highly significant (p < 0.001; Fig. 3B ). Moreover, the use of Ht31 together with cAMP yielded a similar result (p < 0.001) when compared with control (ppHt31+cAMP; Fig. 3B ). Ht31 displacement also reduced the GJIC when used in gap-FRAP experiments on IAR20 cells. 6 In addition, the proximity ligation assay signal was inhibited when IAR20 cells were pretreated with Ht31 but not ppHt31. 6
Cx43-Ezrin Protein-Protein Interaction Inhibition with Peptides
Besides possibilities to stimulate or inhibit a specific enzyme (e.g., PKA) for potential therapeutic application, other approaches are currently investigated. Specificity problems may occur when inhibiting enzymes because they belong to different signalosomes. On the contrary, targeting protein-protein interactions with peptides or small molecules provides a high degree of specificity with potential for avoiding side effects. The interaction between Cx43 and Ezrin was proven to be a direct protein-protein interaction. In addition, binding regions of both Cx43 and Ezrin were defined by multiple approaches. The minimal binding motifs in Ezrin and Cx43 are 510-DDRNEEKR-517 and 366-RASSR-370, respectively. Furthermore, amino acids required for binding were defined; residues D510 and R517 in Ezrin and R370 in Cx43 were found to be critically important, and substitutions D510I, R517V, and R370E abolished the binding completely. 5 Based on these data, we considered designing peptides that disrupt the interaction and furthermore would be expected to affect GJIC. The Cx43 mimicking peptide contains 18 amino acids (aa) plus an arginine tag enabling cell permeability (Cx43 wt), whereas the Ezrin-mimicking peptide has 17aa plus an arginine tag (Ezr wt). Both represent the extended core region, which was previously defined. To make valid controls, peptides were designed with the above-described substitutions that abolish binding (Cx43 mut and Ezr mut).
The peptides were tested first in the AlphaScreen assay. The assay was designed in a way that streptavidin donor beads interact with glutathione acceptor beads when the distance is less than 200 nm, which produces an optimal signal. 13 We used Δ1-232 Cx43 with a GST tag and Biotin-His-Ezrin (full length) immobilized on glutathione and streptavidin beads, respectively. Cx43 and Ezrin peptides competed in the assay, and signal reduction occurred in concentration-dependent manner. Cx43 wt peptide competed and significantly reduced the signal compared with a control peptide, with IC50 values of 13.3 µM for Cx43 wt and 83.4 µM for Cx43 mut ( Fig. 4A ). This proved the ability of the wt peptide to inhibit the interaction, whereas core mutations reduced that effect. In contrast, Ezrin peptide created a strong background signal in the Alphascreen assay as it bound directly to anti-GST beads, without any GST-tagged protein present, meaning that it could not be used in this assay.
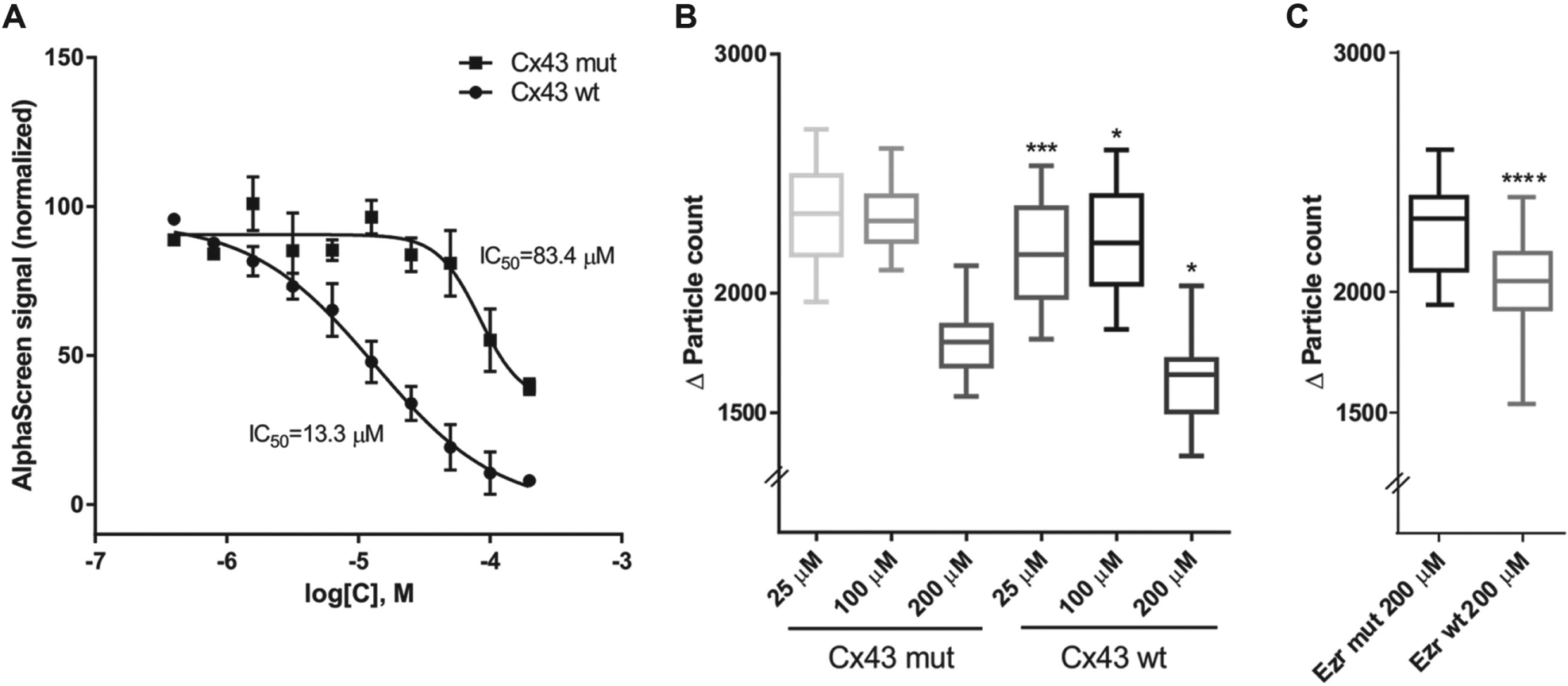
Effect of Cx43-Ezrin protein-protein interaction disruptor peptides. (
Cx43 peptide disruptors were subsequently used in the cell-based assay. The concentration required to show the most significant decrease in dye transfer was 25 µM (p < 0.005; Fig. 4B ). Furthermore, a significant difference compared with the control was seen with higher concentrations (100 and 200 µM) as well (p < 0.05; Fig. 4B ). Although we could not obtain valuable data from AlphaScreen testing of the Ezrin competitor peptides, because of their unspecific binding to GST, they were tested in the cell-based assay. High concentrations of the Ezrin peptides (200 µM) showed significant inhibition of the GJIC with wt peptide compared with mut (p < 0.0001; Fig. 4C ).
Discussion
Targeting protein-protein interactions has become an approach of high interest because of many advantages over conventional approaches. Enzyme inhibitors are highly effective and specific for a particular enzyme or isoform, but the specificity might not be limited to an individual type of complex because enzymes usually belong to multiple signalosomes. Commonly used RNAi research tools, which give the possibility of targeting any specific gene, became attractive for developing RNAi-based therapeutics; however, targeted delivery in vivo is the obstacle that remains to be overcome, as reviewed by de Fougerolles et al. 21 Disrupting the binding region between two proteins by designed peptides or small molecules is another promising approach for drug discovery, as it overcomes specificity issues and therefore potentially limits side effects.
GJIC is involved in many cell processes. Cx43 is the most abundantly present Cx and the most studied one. The fact that Cx43 knockout mice survive only hours after birth emphasizes the importance of this molecule. 22 Therefore, deficiency or mutations in Cx43, as well as impaired GJIC, have been linked to several diseases.23–25 Physiological importance led to an interest in designing pharmacological tools to modulate Cx43 synthesis, assembly, stabilization, and degradation, and there are currently clinical trials in process testing molecules regulating Cx43 GJ formation, reviewed by Grek et al. 2 Considering Cx43’s ubiquitous expression, further approaches are needed that will increase selectivity, tissue specificity, and off-target effects.
In the heart, Cx43 GJs enable the cardiac action potential to spread through cardiac muscle and are needed for synchronous contraction of cardiomyocytes during each heartbeat. 26 Truncation of the Cx43 C-terminus, which is mainly responsible for regulation of GJ permeability, leaves the channel constitutively open. Knock-in mice lacking the C-terminus showed increased tissue damage after myocardial infarction in animals that were also more susceptible to arrhythmias. 27 Furthermore, EMT and MET transformation in cancer cells has been shown to involve GJs, as reviewed by Yu and Elble. 28 Therefore, channel closure by specific peptides/small-molecule disruptors could be of benefit in these conditions.
The Ezrin-Cx43 interaction is a direct protein-protein interaction 5 that could possibly be targeted by peptides disruptors and small molecules. It has been shown previously that the AlphaScreen assay can be used in an HTS setting for inhibitory peptides that bind to AKAPs.13,29 AlphaScreen, being a biochemical assay with high sensitivity and strong signal-to noise ratio, gives valuable information about the binding efficiency of the disruptors but not about function ( Fig. 4A ). Therefore, AlphaScreen assays can serve as a primary screening assay, but to further characterize peptides or small molecules, a high-content screening (HCS) assay is needed. Cell-based assays and the multiparametric information acquired are more suitable for secondary screens. Considering that Cx43 is a GJ protein and the role of the Cx43-Ezrin-PKA complex is to regulate GJ opening, 5 a cell-based assay assessing the GJIC is necessary to test if the screened molecules are effective in live cells.
Here we designed a GJ assay that could measure the effect of putative Cx43-Ezrin disruptor molecules that is compatible with HTS and HCS modalities. We validated a calcein dye transfer-based assay ( Fig. 1 ) using reference compounds that were GJ blockers ( Fig. 2F ), giving IC50 ( Fig. 2A–E ) values in the same range as published values.11,12,17 The main advantage of our assay is in the high level of automation with very few manual steps. We used assay-ready plates prepared by the Echo acoustic liquid-handling system and then applied the cells directly. The Echo system results in low final DMSO content because compounds are dispensed directly in nanoliter-range volumes from high concentration 100% DMSO stock solution, avoiding intermediate dilutions and thus compound precipitation. The automation gives strength to an assay when it comes to scaling up for screening libraries of small molecules. Because it is a cell-based assay, it is important to have a simple and fast sample preparation protocol keeping the cells in viable state. Our assay is based on the use of just one fluorescent dye whose diffusion is known to be GJ dependent. 14 Automated imaging of the phase-contrast channel together with the GFP (green) channel, in high resolution, gave further information about the compounds tested such as cell viability, changes in morphology, and other properties. By using high-content imaging analysis, we could compare the same cell populations over time, acquiring images at several time points and observing multiple parameters.
We tested molecules that will specifically disturb the Cx43-Ezrin supramolecular complex under study. It has been shown that Ezrin-anchored PKA phosphorylates Cx43, which increases GJ permeability in trophoblast as well as in IAR20 cells. 5 PKA-activating reagents were previously shown to potentiate the GJIC,18,19 which we confirmed in our study. However, displacement of PKA from Ezrin with Ht31 peptide reduced the communication, in accordance with our data on IAR20 cells, 6 demonstrating that peptide inhibitors could be assayed. Following from that study, we proceeded with a new peptide design strategy. Peptides and peptidomimetics are appealing because of their high selectivity, specificity, and affinity to the targets. Furthermore, the synthesis is relatively simple as is optimization and evaluation. Most importantly, there is minimal immune response and low toxicity, reviewed by Cochran. 30 Pidoux and Tasken 5 identified the amino acids in Cx43 and Ezrin crucial for binding and moreover mutations that abolished the binding completely. Following this, we synthesized peptides that were efficient in reducing GJIC when compared with peptides with the loss-of-function mutations ( Fig. 4B , C ).
Peptides do, however, have a number of problems if they are to be used as human therapeutics. They can be metabolically cleaved and rapidly cleared from the body, and intracellular delivery may be a problem, as discussed by Cochran. 30 Thus, small molecules have advantage over peptides in more easily optimizable pharmacokinetic properties, better stability, and the potential for higher oral bioavailability, as reviewed by Calejo and Tasken. 31 The challenge is having enough knowledge about the interaction between the proteins of interest, which could be a limiting step. The contact surface is frequently quite big, and the topology of the interacting proteins is often relatively flat but with key binding properties residing in certain residues (“hot spots”); hence, the detail understanding is a necessary requirement for therapeutic targeting. 32 Moreover, crystal structures of an active compound bound to the target or a nuclear magnetic resonance approach are important methods used in drug discovery. Targeting the Cx43 and Ezrin interaction could be promising, but further studies are required to determine Cx43 and Ezrin protein structures and their interaction. We believe that our assay is suitable for a secondary screen of available small-molecule libraries and, considering its simplicity, it can easily be adjusted to other cell types. The PKA-Ezrin-Cx43 GJIC regulation is an important mechanism in a number of endogenous and disease state processes, and our assay enables discovery of new pharmacological tools or potential therapeutics targeting this complex.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Norwegian Cancer Society, Research Council of Norway, the K.G. Jebsen Foundation, and the Novo Nordic Foundation.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
