Abstract
Ion channels play critical roles in physiology and disease by modulation of cellular functions such as electrical excitability, secretion, cell migration, and gene transcription. Ion channels represent an important target class for drug discovery that has been largely addressed, to date, using small-molecule approaches. A significant opportunity exists to target these channels with antibodies and alternative formats of biologics. Antibodies display high specificity and affinity for their target antigen, and they have the potential to target ion channels very selectively. Nevertheless, isolating antibodies to this target class is challenging due to the difficulties in expression and purification of ion channels in a format suitable for antibody drug discovery in addition to the complexity of screening for function.
In this article, we will review the current state of ion channel biologics discovery and the progress that has been made. We will also highlight the challenges in isolating functional antibodies to these targets and how these challenges may be addressed. Finally, we also illustrate successful approaches to isolating functional monoclonal antibodies targeting ion channels by way of a number of case studies drawn from recent publications.
Introduction
Ion channels form a large family of polytopic membrane proteins that function by allowing passage of ions across cellular membranes. They are present in all cell types, and they play critical roles in biological processes and disease as a consequence of their modulation of functions such as electrical excitability, secretion, cell migration, and gene transcription. Ion channels are widely regarded as attractive targets to exploit by therapeutic drugs and comprise the third best-selling group of prescribed drugs with estimated worldwide sales of $12 billion. 1 Despite this success in the use of ion channel modulators, only a few of the estimated 400 annotated ion channel genes predicted in the human genome have been targeted to date.2,3 The majority of the drugs that have been marketed or are under development are small chemical entities with the most notable exceptions being the peptide toxin, ziconotide, and the proanthocyanidin oligomer, crofelemer.4,5 Alternative modalities of targeting ion channels comprise the use of biological toxins derived from venomous species and the use of biologics such as antibodies.6,7 Despite the attractiveness of targeting ion channels with antibodies due to their high specificity, no antibody-based molecule has been developed to date that has progressed to clinical use. In this article, we will review the current state of ion channel biologics drug discovery with a particular focus on antibodies. We also highlight the challenges in isolating functional antibodies to these targets and how these challenges are being addressed.
Overview of Ion Channels as Drug Targets
Ion channels are complex polytopic transmembrane proteins that control the transfer of ions across biological membranes via a gated pore. The family of ion channels is involved in a range of diverse biological processes, which include nerve and muscle excitability, sensory and cognitive function, signal transduction, blood pressure regulation, and cell proliferation. 2 This wide range of activities and the modulation of ion channel function have translated into links to a variety of pathophysiologies that include cardiac and neurological disorders, perception of pain, kidney failure, blindness, and oncogenesis.8–15 A large number of channelopathies have also been described in which rare genetic variants give rise to disease that is linked to point or nonsense mutations in ion channels. 16 In addition, a number of autoimmune disorders exist in which antibodies are directed to ion channels leading to adverse clinical effects. Examples of such autoimmune disorders include Lambert–Eaton myasthenic syndrome and Isaacs’ syndrome in which autoantibodies target voltage-gated calcium channels and voltage-gated potassium channels, respectively.17–20 Given the importance of ion channels in modulation of cellular function and their role in disease, it is clear that ion channels are recognized as important therapeutic targets.
Analysis of the human genome shows that several hundred genes encode ion channels. The ion channel superfamily is classified broadly into two types (voltage-gated or ligand-gated) depending on the factors that control opening and closing of the ion channel pore.21,22 Examples of modulation of both channel types by small molecules have been described. This can be exemplified by the action of lidocaine on voltage-gated sodium channels, the action of diazepam on the ligand-gated γ-aminobutyric acid (GABA) class A channel, and the action of sulphonylureas such as glipizide on the KATP channel. 2
Ion channel drug development throughout the past 50 years has focused on the discovery of small-molecule therapeutics. The discovery of these drugs ranges from opportunistic observations made during in vivo testing of compounds to the use of high-throughput compound screening.2,23 These efforts have given rise to approximately 20 marketed compounds targeting a very limited number of ion channels. 2 Although the pace of new drug approvals against this target class slowed in the past decade, two new drugs targeting ion channels have been approved for clinical use in 2012. These new drugs are Ivacaftor (Kalydeco), which targets the cystic fibrosis transmembrane regulator (CFTR) chloride channel, and crofelemer (Fulyzaq), which inhibits both CFTR and the calcium-activated chloride channel TMEM16A.5,24,25 These new drug approvals provide renewed confirmation of the value of ion channel targets and a significant stimulus to the field of ion channel drug discovery.
Given the importance of this target class, a significant opportunity exists to target ion channels with biologics such as antibodies or antibody fragments. Antibodies demonstrate exquisite specificity against their targets, and their affinity and potency against a target can be further optimized by protein engineering. 26 In addition, the effector function and pharmacokinetics of antibodies can be modulated to provide further control of their mode and duration of action.27,28 To date, however, there has been limited progress in identifying antibodies with the appropriate properties to modulate ion channel function in a pharmacologically relevant way. To some extent, this reflects the challenges in identifying functional antibodies against this complex and diverse target class.
Targeting Ion Channels with Antibody Therapeutics
Antibodies and related molecules represent the fastest-growing class of therapeutic drugs. 29 In 2012, 34 approved monoclonal antibodies were described in a compendium of marketed therapeutic antibodies. 30 As a result, antibody therapeutics are a well-established drug class, and they benefit from a high success rate from their first time in human studies through to regulatory approval. Several features of antibodies make them attractive as potential therapeutics for ion channels that address some of the barriers to small-molecule drug discovery such as difficulties in achieving specificity to highly homologous ion channel subtypes. Antibodies typically bind to their target antigens with very high specificity allowing them to be used for targeted therapies. Antibodies can also be generated that have high antigen-binding affinities in the nanomolar to subpicomolar range, and options exist for optimizing antibody affinity via protein-engineering strategies targeting the antibody paratope.27,31 This affinity maturation typically leads to large reductions in the dissociation rate of the antibody and may also lead to increased biological potency against a target. 27 These features of antibodies reside in the variable-domain regions of antibodies.
In addition to the variable domains of antibodies mediating binding to target antigens, features of the antibody constant domains affect the effector function of antibodies (e.g., antibody-dependent cellular cytoxicity) and also the pharmacokinetics exhibited with antibodies typically having a long plasma half-life (several weeks) allowing for reduced frequency of administration. 28 These properties of the constant region are also amenable to protein engineering, and it is possible to “tune” the properties of antibodies to increase or decrease effector functions such as antibody-dependent cellular cytotoxicity and enhance the half-life of antibodies to affect dosing frequency and duration of action. One other feature of antibodies is that their distribution is peripherally restricted, which lends itself to targeting ion channels in which the functional effect needs to be limited to the periphery to avoid CNS side effects. In contrast, technologies are emerging by which antibodies can be modified and targeted to cross the blood–brain barrier, which holds the promise of being able to devise strategies to target ion channels in the CNS.32–34
A variety of alternative antibody formats also exist and add to the options for targeting ion channels. Although not specifically the focus of this review, scFv’s, Fab fragments,35,36 nanobodies,37,38 and a variety of alternative non-antibody scaffolds39,40 have been described that all have potential to target ion channels. Alternative non-antibody scaffolds and nanobodies are smaller in size than intact immunoglobulin G (IgG) molecules, and these smaller binding protein formats may be useful to target small epitopes and binding pockets on ion channels that may not be accessible to full IgGs. These formats also allow multiple different epitopes to be targeted by linking two or more binding proteins together as a fusion protein. This strategy has been described by Ablynx in conference proceedings (Collaborations in Ion Channel Drug Discovery, Cambridge, UK, 2014) in which variable domains of heavy-chain-only camelid antibodies (nanobodies) have been isolated that show function against the Kv1.3 ion channel when combined in a multivalent format. 41
Challenges in Identifying Functional Antibodies to Ion Channels
Although small-molecule approaches have been successful in delivering marketed therapeutic drugs targeting ion channels, targeting ion channels with functional antibodies offers significant opportunities. Generating antibodies against these integral membrane proteins does, however, pose some challenges. Ion channels comprise a very diverse collection of structural and functional types with selected examples shown schematically in Figure 1 . Ligand-gated ion channels feature large extracellular domains that have defined ligand-binding pockets to allow for activation of the channel following ligand binding.42,43 In principle, these large extracellular domains are accessible for antibody binding, and examples of antibodies targeting these ion channels have been described. 44 Identifying antibodies that target voltage-gated ion channels may be more challenging particularly because the extracellular loops of these channels are typically small and therefore less accessible to antibodies. In addition to these structural considerations, a number of general features of ion channels linked to the difficulty of expressing and manipulating ion channels and the complexities of functional screening also add complexity to the isolation and characterization of antibodies. These challenges are described in the following sections.
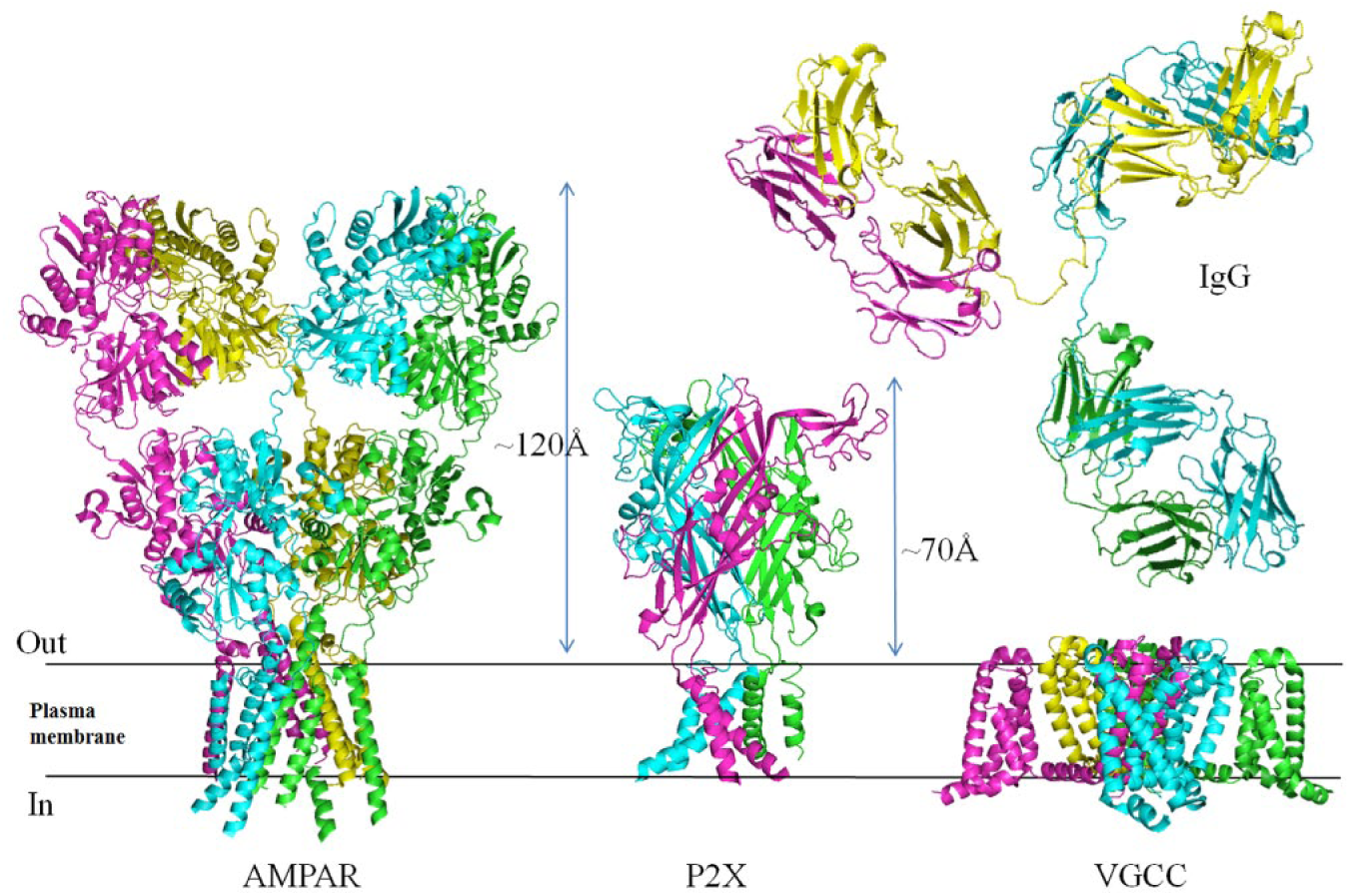
Illustration of the diversity of transmembrane topologies of voltage-gated and ligand-gated ion channels. Representations of three types of ion channel to demonstrate their diverse structures and the relative differences in extracellular loops and domains that could be targeted by antibodies. Shown here are two ligand-gated ion channels [AMPA receptor (AMPAR) and P2X receptor] and a voltage-gated calcium channel (VGCC). Structures were generated by PyMOL software using information in the Protein Data Bank [AMPAR 3KG2, 106 P2X receptor 3I5D, 42 VGCC 4MTO, 107 and immunoglobulin G (IgG) 1IGT 108 ]. The ligand-gated ion channels both have large extracellular domains that bind agonist ligands. The voltage-gated ion channels typically have four homologous domains that are arranged around a central pore. Voltage-gated ion channels have relatively small extracellular loops in comparison to the ligand-gated ion channels represented here. A structure of an IgG antibody is shown approximately to scale.
Antigen and Immunogen Formats
Identification of functional antibodies directed to specific targets fundamentally relies on the selection of a suitable antigen and its presentation to elicit antibody generation.45,46 The aim of immunization methodologies is to present an antigen to the host in a form that stimulates a strong and specific immune response. To isolate antibodies from technologies such as phage display, the aim is to present a pure antigen to allow selection of a diverse panel of binding and functional antibodies. Although the precise format of the antigen will affect the selection and identification of binding antibodies, the production of antigens for soluble proteins such as cytokines, growth factors, protein subdomains, and globular extracellular domains of receptors is relatively straightforward. 47 A plethora of methods exist for the expression and preparation of these antigens in a pure, well-characterized, and correctly folded state.
By contrast, methods for the expression and purification of ion channel antigens face a number of challenges that are linked to the complex topology of ion channels and the intimate association with the membrane lipid bilayer. In the following sections, we will highlight some of the challenges and strategies that may be used to overcome the challenges.
Synthetic peptides represent the simplest antigen format that can be used for both immunizations and selection-based antibody discovery strategies. 48 Typically, peptides require conjugation to a carrier protein such as keyhole limpet hemocyanin or bovine serum albumin prior to use as an immunogen. Peptides may be used as surrogates for proteins and result in a highly specific targeted epitope antigen. Peptides may be used in a linear format, or more complex peptides may be synthesized when chemical methods are used to constrain the conformation of single or multiple peptides as defined loops. Examples of the use of chemically constrained, more native-like peptides exist for generating antibodies targeting G-protein coupled receptors (GPCRs). GPCRs exhibit similar issues to ion channels with respect to the challenges faced in isolating functional antibodies. Biotinylated cyclic peptides have been used to isolate functional scFv antibodies to CCR5 by phage display methods.49,50 Functional antibodies to CXCR7 have also been reported to have been raised using CLiPS (chemical linkage of peptides to scaffolds) peptide technology that is marketed by Pepscan. 51 Generation of functional antibodies to GPCRs using constrained peptides suggests that this type of method could be extended to ion channels. The ability to modify peptides by chemical methods to introduce moieties such as biotin facilitates phage display panning methods and detection methods by allowing capture onto beads or solid-phase surfaces coated with streptavidin. 48
A particularly successful strategy for obtaining polyclonal antibodies to ion channels has been the use of peptides derived from the E3 pore loop region of a variety of channels. 52 Several channel classes, including voltage-gated ion channels (with Na, K, and Ca selectivities) and the transient receptor potential (TRP) channels, exhibit a common structural theme of six transmembrane spanning helices (S1–S6) per subunit with intracellular amino and carboxyl termini and three extracellular loops (E1–E3). The E3 loop is implicated in the ion selectivity filter for ion channels, and because it is typically relatively long, it lends itself to being an accessible epitope for antibodies ( Fig. 2 ). Using this loop as an immunogen, antibodies have been generated to Nav1.5, Cav2.1/2, Kv1.2, Kv3.1, Eag1, TRPC1, TRPC5, TRPM3, and TRPV1.53–55 It is noteworthy that the majority of antibodies generated using E3 loop antigens have been polyclonal antibodies, but they have nevertheless shown functionality in blocking channel function, albeit in a partial manner. In one case (TRPV1), both polyclonal and monoclonal antibodies were isolated with only the polyclonal antibodies showing activity. 55 Recent studies, however, have demonstrated that monoclonal antibodies targeting the E3 loop can be effective antagonists. A functional monoclonal antibody against Eag1 has been generated by using the E3 loop strategy, and this example is exemplified as one of the case studies in this review. 54 More recently, Miller et al. 109 explored the feasibility of blocking the transient receptor channel TRPM8 by evaluating the functional effects of commercially available polyclonal and monoclonal antibodies raised against E3 loop peptides. In this study, a polyclonal antibody (ACC-049; Alomone Laboratories, Jerusalem, Israel) and a monoclonal antibody (MBS609041; My Biosource, San Diego, CA) were reported to completely inhibit ligand activation of human TRPM8. Although the authors stated that the potencies of the antibodies were not sufficient for therapeutic consideration, this study supports the feasibility of generating monoclonal antibodies targeting the pore loop that block channel activation.
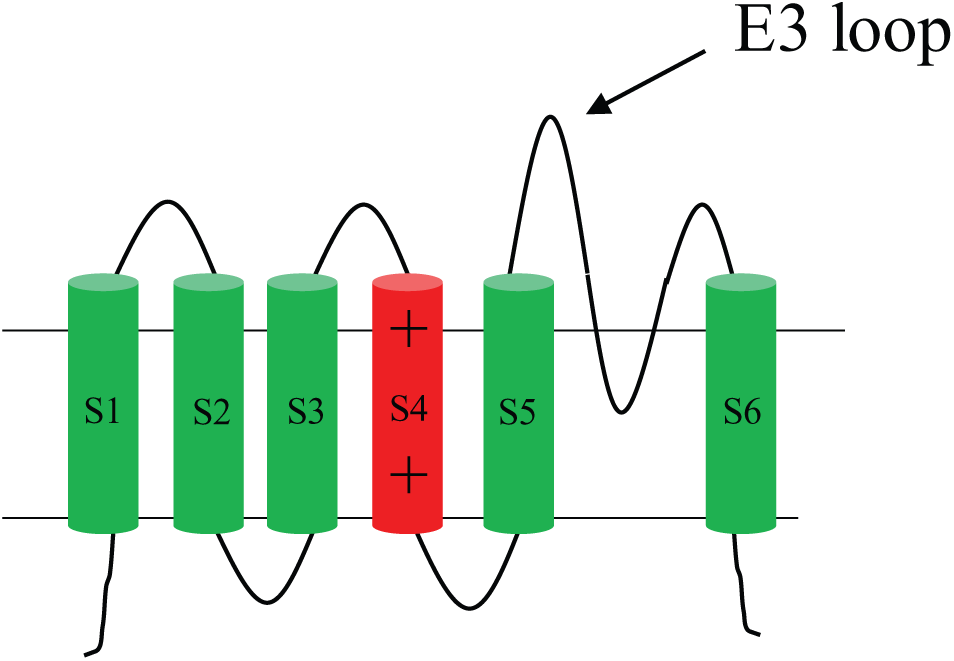
Schematic representation of the functional unit of a voltage-gated ion channel. Transmembrane folding topology of a single domain of the voltage-gated Nav or Kv ion channel, illustrating the position of the E3 extracellular loop. Sodium channels are single polypeptides in which four repeated domains, each with six transmembrane helices, are arrayed around a single ion-conducting pore. Potassium channels with a similar six-transmembrane topology contain four identical subunits that assemble to form a pore. A similar topology is exhibited by transient receptor potential channels such as TRPV1.
As an alternative to using peptides as immunogens, whole cells expressing ion channels of interest can be used for immunizations and in vitro display technologies. 44 The use of cells in this way is affected by some general challenges raised by expressing ion channels. Ion channels are integral membrane proteins and are often found in low abundance. Family members within specific ion channel classes often exhibit high sequence identities. Overexpression of ion channels can be difficult to achieve because methods for monitoring or selecting for high expression are compromised by the lack of effective tool antibodies to check for expression and the difficulty of assaying for channel function with relatively low-throughput electrophysiology methods. High-level expression of ion channels can also be difficult because the overexpression can sometimes result in toxicity to cells, precluding the generation of stable cell lines. 56 It is possible to address some of these concerns by using methods that enhance detection of ion channels or that result in transient, regulated expression of the ion channels. To improve detection of ion channels, a wide range of strategies have been used to introduce epitope tags into ion channel sequences to facilitate detection using methods such as flow cytometry. Epitope tags including, but not limited to, Flag, hemagglutinin (HA), V5 epitope, 6-His, and c-myc tag have been used to facilitate ion channel detection. 57 Green fluorescent protein (GFP) has also gained significant utility in tagging ion channels to monitor expression in cells and to facilitate ion channel purification.58–60 The use of epitope tags has been comprehensively reviewed by Maue. 57 Controlling the timing of gene expression is another method that can be used to allow overexpression and reduce the issues associated with channels that cause toxicity. In this approach, the ion channel gene of interest is placed in a vector that allows for inducible promoter expression by administration of agents such as tetracycline. 61 A number of commercial products are available for controlling gene expression in mammalian cells that include the T-Rex, Tet-On 3G, and Cumate inducible systems.61–63 Finally, it is also possible to prepare significant quantities of cells by transient transfection, which may also assist high-level expression. This can be done using polycation or lipid-based transfection reagents or by the use of systems such as the Maxcyte electroporation system that allow electroporation of gene expression vectors into large batches of cells. 64
In addition to considering methods to achieve overexpression of ion channels, the cell host chosen for expression also represents a significant factor. Ion channels may be expressed in a variety of cell types (e.g., HEK293, CHO, and U2OS). In each case, subsequent screening on cells for isolated antibodies will require both the parental host cell and the overexpressing cell line. When using host cells expressing ion channels as immunogens, an immune response will also be mounted to other proteins in the host cell. To maximize the opportunity for a specific antibody response to the target of interest, human target genes can be overexpressed on a host cell that is syngeneic to the immunization host strain (e.g., BALB/C mouse). 44 In this case, a significantly reduced background immune response would be expected with an improved response to the target of interest and increased yield of monoclonal antibodies against the target protein.
If overexpression of ion channel genes can be achieved in cells, another potential option is to generate virus-like particles (VLPs) as an immunogen.65,66 In this approach, cells overexpressing the channel are transfected with vectors encoding a viral protein such as Gag, which causes budding of the cell membrane with the release of VLPs. These small particles are often enriched for the gene of interest, compared with the host cell, and can be used to support both immunization and phage display antibody isolation methods. 67 Recent conference reports indicate that this approach has been successful in the isolation of antibodies against ion channels with functional antibodies to P2X3 reported by Integral Molecular and Crystal Biosciences. 67
Purified, correctly folded, and functional protein provides one of the best and most versatile of antigens for generating functional antibodies. Purified protein can be used to support immunization strategies as well as support isolation of antibodies by in vitro display methods. In the case of ion channels, significant challenges are posed by the integral membrane location of the channels and the requirement for extraction and purification of the proteins in detergents. Solubilization of membrane proteins by detergents typically requires screening of multiple factors to extract functional protein and can often result in protein instability, unfolding, and aggregation that are all detrimental to production of quality antigen. It is clear that progress is being made in the purification of ion channel proteins as demonstrated by the increasing number of crystal structures of these integral membrane proteins, but success still requires a diverse array of methods to achieve well-behaved, correctly folded, stable protein.42,68 Methods to achieve this include using species homologues of human proteins and engineered proteins that effectively “screen” for proteins with amino acid sequences that have well-defined behavior in detergents. Examples of this include the use of bacterial homologues of potassium channels 68 and species variants of P2X channels. 42 Although this demonstrates that ion channel purification is feasible, it does not solve the issue that human proteins are required to support therapeutic antibody discovery. Other features of detergent extraction that may affect antibody isolation include the fact that longer-chain detergents typically required for stabilization of membrane proteins may mask critical epitopes. Purified proteins will also have both intracellular and extracellular loops available, which may result in antibodies being isolated to intracellular epitopes.
One other approach that may improve our ability to purify ion channels is the use of mutagenesis to create stabilized ion channel proteins as has been demonstrated in the case of GPCRs and transporters.69–71 The use of a stabilized receptor protein for functional antibody generation has been validated for the β1-adrenergic receptor. 72 This approach appears feasible for ion channels, but in some cases (e.g., mammalian voltage-gated sodium channels), the large size of the protein makes the sequence space that needs to be explored challenging.
Given the challenges described in expression and purification of these complex antigens, an alternative strategy for antibody isolation involves DNA immunization. 73 In this approach, expression plasmids encoding a particular gene of interest are used for immunization, and DNA is delivered by injection or via a gene gun into a suitable host.73,74 This method would typically require that a check is made prior to immunization that the plasmid supports gene expression, and there is still a requirement to have purified antigen, high-expressing cell lines, or functional assays for the ion channel of interest to allow effective screening for binders and functional antibodies. Improved methods for DNA immunization have been described recently, leading to successful antibody isolation against a membrane transporter, which is a target with similar challenges to ion channels. This report described the use of hydrodynamic tail vein injection of DNA encoding the transporter gene in conjunction with immune modulators. 75 This method shows great promise for complex membrane proteins such as ion channels. In a further extension of this DNA immunization strategy, it is possible to also use viruses encoding the ion channel as immunogens. This particular approach has shown some success using adenovirus, although it is not entirely clear in the published work which of several strategies led to the final functional monoclonal antibody. 76 Examples of strategies that have been used to isolate functional monoclonal antibodies described in the public domain are described in the case studies in this review.
Challenges Facing Ion Channel Biologics Screening
The field of ion channel screening for biologics is still in its infancy with a limited number of successful campaigns resulting in examples of functional antibodies against ion channels. 77 In general, identifying antibodies against ion channels can be achieved through a variety of approaches, and the range of commercial antibodies is increasing rapidly, highlighting the feasibility of identifying binding nonfunctional and polyclonal antibodies to ion channels. 78 Identification of functionally active antagonist or agonist antibodies is more challenging and requires the use of screening assays that add additional complexity along with the requirement for investment in technology and expertise. Historically, there is a high attrition between identifying binding antibodies and those exhibiting functional activity as evidenced by the limited number of functional antibodies reported in Table 1 . To reduce this attrition, many antibody screening campaigns implement functional assays as a first screen, and this can be successful for cell surface targets such as GPCRs, in which difficulties such as antibody expression, media tolerance, and antibody potency can be overcome. 79
Key Information on Published Functional Monoclonal Antibodies That Form the Basis of the Case Studies Described in This Review.
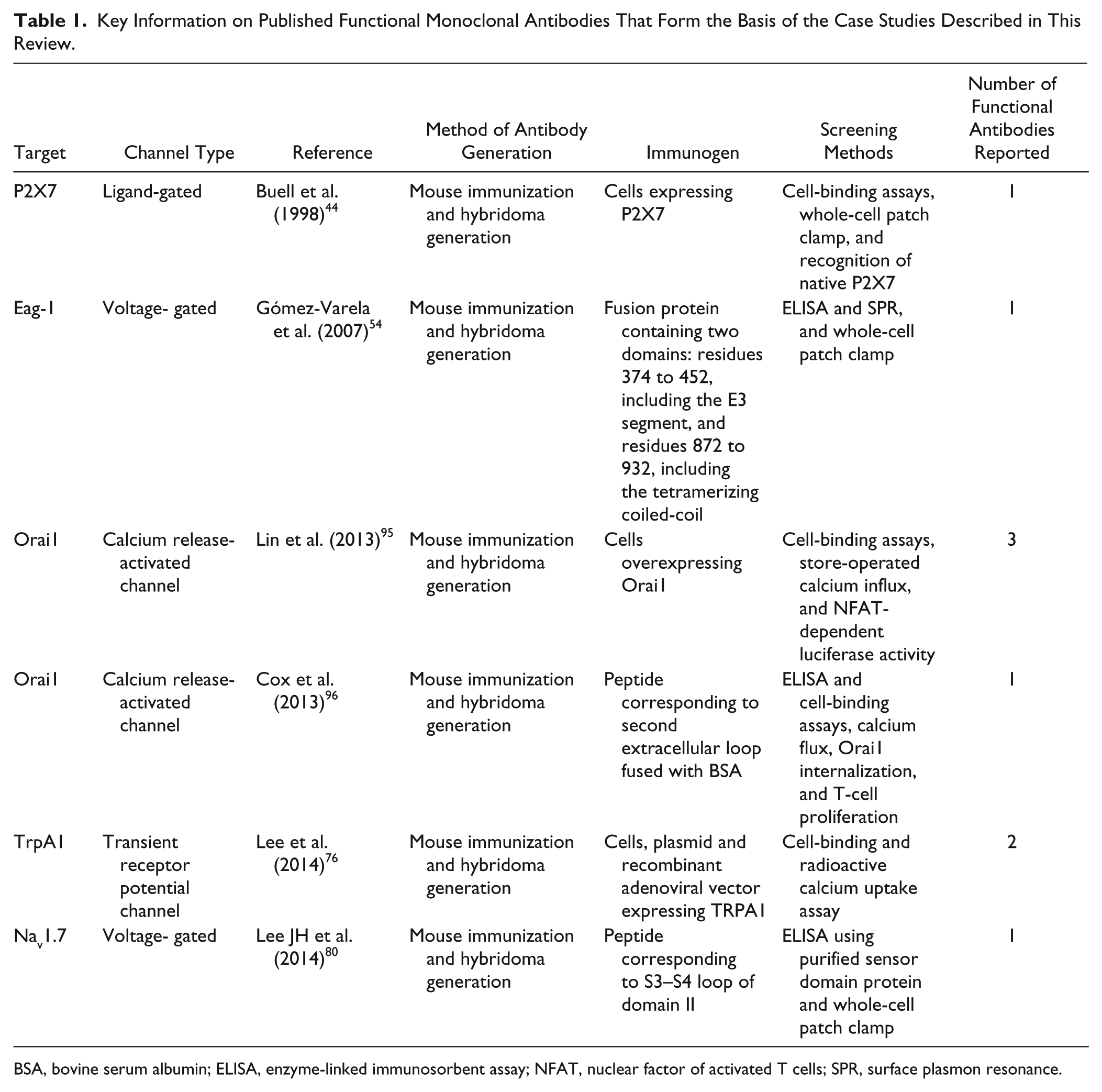
BSA, bovine serum albumin; ELISA, enzyme-linked immunosorbent assay; NFAT, nuclear factor of activated T cells; SPR, surface plasmon resonance.
Outputs from phage display selection using naïve antibody libraries typically require expression of scFv in
Ideally, cell-based binding assays can be implemented in which the ion channel is more likely to be in a native, active conformation due to its location in the plasma membrane. This format requires cell lines that either stably express the target of interest or can be induced to express the target following the activation of an inducible promoter. 82 Typically, it has been challenging to achieve high copy numbers of ion channels because overexpression of the channel can be detrimental to cell health, possibly as a result of compromised membrane integrity. Poor cell viability in a screening assay will increase identification of false positives due to nonspecific binding and uptake of fluorophore labels into the transfected cells. Therefore, the ion channel expression level needs to be optimized to maintain cell health and to allow detection of relatively low-affinity clones in phage libraries. The benefit, however, of cell-binding assays is that they are normally more tolerant to sample media and can provide significant sensitivity. 83 Once antibodies have been identified by this process, they need to be tested for function in a relevant assay. Due to the issues mentioned above regarding assay interference, the antibodies are purified and prepared in a buffer compatible with downstream functional assays.
Patch clamp electrophysiology remains the gold standard for measurement of the effects of molecules on ion channel function. The classic manual patch clamp (see Neher and Sakmann 84 and Neher et al. 85 ) has too low a throughput for meaningful drug discovery efforts, and whereas there are non–patch clamp high-throughput screening assay technologies such as fluorescent imaging plate reader assays, which use either membrane potential or voltage-sensitive dyes, these do not measure the effects of modulators directly on the ion channel. There are limited options to screen ion channels in other assay formats, and the development of reporter assays, internalization assays, and downstream readouts that incorporate signal amplification is still lacking for ion channel targets. Small-molecule screening has historically required the testing of extensive chemical libraries in excess of 1×106 compounds, and it has necessitated higher-throughput screening approaches. Biologics screening, specifically the identification of monoclonal antibodies that modulate ion channels, uses far smaller subsets of molecules because the original libraries are essentially screened by phage display followed by standard binding assays, resulting in greatly reduced numbers of screening samples. This allows the direct screening of all binding monoclonal antibodies with automated patch clamp platforms described below.
There are multiple automated patch clamp systems available, ranging from full 384-well screening platforms down to simplified manual patch clamp equipment, and these have been recently reviewed (see Dunlop et al. 86 and Trevedi et al. 87 ). Screening for functional antibodies against ion channels has a unique set of issues to overcome, including buffer tolerance (typically overcome by purification) and incubation times required to see an effect. Monoclonal antibodies likely exert a modulatory effect via allosteric mechanisms on ligand-gated ion channels due to their size and lack of access to the ligand-binding site as well as via inhibition of ion channel domain movement on activation. To address these issues, the ability to perform baseline readings followed by prolonged incubation times before a test stimulus is applied as a prerequisite for screening. This requirement moves antibody screening toward lower-throughput systems such as QPatch and PatchXpress, which offer more control over the experimental protocols than higher-throughput systems such as the Ionworks Quattro. The introduction of higher-throughput automated patch clamp platforms such as the Ionworks Barracuda (Molecular Devices), SyncroPatch 384PE (Nanion), and Qube (Sophion) allows more detailed 384-well patch clamp assays to be performed. The Ionworks Barracuda has been validated for screening of focused small-molecule compound libraries (in the range of 20,000–100,000 compounds) against voltage-gated calcium channels. 88 Such studies show there is a significant opportunity to use these automated patch clamp platforms for biologics screening, and time will tell if they are suitable for screening and identification of functional antibodies directed against ion channels.
As the number of successful campaigns increases, so will our knowledge regarding the best technologies to use and the most sensitive assays to implement for each type of ion channel. Until that time, the assays used for biologics screening for ion channel targets will require a mixture of assays from standard antibody isolation methods to any cell surface target (reviewed by Cariuk et al. 89 ) and the use of assays previously developed for screening large small-molecule libraries (reviewed by McManus 96 and Falconer and Smith 91 ). This, however, can lead to assumptions regarding how antibodies may modulate ion channels, and it could lead to screening campaigns that are biased to certain mechanisms or, worse still, assays not sensitive enough to identify any antibody modulators. Nevertheless, the case studies described in the following section demonstrate that progress is being made in addressing screening methods to identify functional antibodies.
Case Studies of Isolation of Functional Monoclonal Antibodies to Ion Channels
Despite the challenges outlined in previous sections to isolate antibodies against ion channel targets, there have been some significant advances in the past decade. In this section, we describe a number of case studies taken from published literature that highlight advances in the field. Table 1 summarizes key information from these publications, and more detailed descriptions follow below.
P2X7
P2X receptors are ligand-gated ion channels that are activated by extracellular adenosine triphosphate (ATP). Activation results in opening of a cation channel that allows entry of calcium and depolarization of the cell membrane. 92 P2X7 receptors have been implicated in the immune response with P2X7 upregulation on macrophages and microglial cells following lipopolysaccharide or interferon-γ stimulation.
Buell et al. 44 successfully isolated a functional monoclonal antibody directed to P2X7. This was achieved by immunization of mice with a mouse myeloma cell line (XS63) stably transfected with human P2X7. Following hybridoma generation, screening for binding antibodies was performed by flow cytometry using XS63 cells expressing P2X7 and a parental control XS63 cell line to check for specificity of binding to P2X7. The specificity of the binding was also confirmed by assessing binding to HEK293 cells expressing P2X1, P2X4, and P2X7. The monoclonal IgG2b antibody isolated was specific for P2X7 in these assays. The antibody was also shown to bind endogenous P2X7 on human monocytes and macrophages. Further studies demonstrated that the antibody was functional and was shown to inhibit ATP-stimulated release of interleukin 1β (IL-1β) from THP-1 cells. Interestingly, the monoclonal antibody was able to immunoprecipitate P2X7 from XS63 cells overexpressing the receptor, whereas the antibody was nonfunctional in western blotting. This suggests that the antibody recognizes a conformational epitope.
Eag-1
Potassium channels are increasingly thought to play an important role in cellular processes such as excitability, cell cycle progression, and cellular proliferation. Human Eag 1 is a voltage-gated potassium channel modulated throughout the cell cycle, and it is believed to be involved in tumorigenesis. 13 Observations linking high expression levels of Eag1 in tumor tissues have led to the suggestion that Eag1 is involved in progression of malignant disease and may represent a valid target for therapeutic intervention.
The high sequence homology between voltage-gated potassium channel subtypes presents a significant hurdle in the development of selective inhibitors. In the case of Eag1, the closest family member is Eag2, which has 73% sequence identity with Eag1. Specificity is particularly important for Eag because it is related to the human ether-à-go-go related channel (hERG), whose inhibition is linked to severe and dangerous adverse cardiac events. 93 Given this background, monoclonal antibodies targeting the Eag1 channel offer the potential for highly specific binding.
Gómez-Varela et al. 54 isolated a monoclonal antibody to Eag1 by immunizing mice with a fusion protein composed of the linker between the S5–S6 transmembrane segments (E3 loop, residues 374–452) fused to the C-terminal tetramerization domain of the channel (residues 872–932). This strategy was performed to mimic the native tetrameric state of the potassium channels. Following immunization and hybridoma generation, screening was performed using a sequence of steps. First, an ELISA was used to confirm binding of antibodies to the recombinant antigen followed by surface plasmon resonance analysis to confirm those antibodies binding to the E3 loop and those binding selectively to Eag1 over Eag2. Function was then confirmed by testing the effect of the antibodies on Eag1 currents in Xenopus oocytes. This gave rise to a single monoclonal antibody named mAb56.
Further analysis of mAb56 showed that it recognized a linear epitope (GSGSGKWEG), and the antibody was able to bind to HEK293 and CHO cells expressing Eag1. This binding could be inhibited by co-incubation of the synthetic peptide epitope. The antibody was also able to inhibit Eag1 currents in HEK cells expressing the channel by ~40%, and mAb56 did not inhibit hERG currents demonstrating selectivity. The authors also demonstrated that mAb56 was able to inhibit anchorage-independent cancer cell growth in vitro.
Orai1
Calcium release-activated calcium channels are activated in response to the depletion of intracellular calcium stores in the endoplasmic reticulum (ER). The channel has two components: stromal interaction molecule 1 (STIM1), which acts as a calcium store sensor in the ER, and Orai1, which is a pore-forming protein subunit located in the plasma membrane. 94 These two proteins are responsible for store-operated calcium influx, which is responsible for refilling intracellular calcium stores following cell activation involving release of calcium as a second messenger. STIM1 acts as a calcium sensor detecting reduced calcium concentration in the ER, which subsequently activates Orai1 in the plasma membrane resulting in calcium influx. Orai1 and STIM1 are present in T-cells, and the calcium entry following antigen stimulation of T-cells is critical for NFAT-mediated T-cell activation. Human mutations in STIM1 and Orai1 that affect protein expression or function give rise to a hereditary form of severe combined immunodeficiency syndrome. 94 Autoreactive T-cells play a key role in autoimmune diseases such as rheumatoid arthritis, and this observation highlights Orai1 as a potential target for therapeutics that inhibit autoimmune disease and chronic inflammation as well as for potential therapeutics for use in organ transplantation. Although small-molecule antagonists have been developed and studied, none have advanced to therapeutic use. This resulted in Lin et al. 95 and Cox et al. 96 pursuing strategies to develop monoclonal antibodies as alternative antagonists of Orai1 function.
Lin et al. 95 obtained fully human antibodies targeted to Orai1 by immunization of the Xenomouse with U2OS cells overexpressing human Orai1. The Xenomouse is a transgenic system in which human immunoglobulin loci are introduced into the germ line of mice. 97 One hundred and two fully human antibodies were isolated that showed binding to CHO-AM1 cells stably overexpressing both human Orai1 and a YFP-tagged STIM1. From these antibodies, 11 unique monoclonal antibodies were expressed and purified for more detailed study. Antibody 2C1.1 was studied in some detail and was shown to bind to Orai1 expressed in HEK293 cells and also to endogenously expressed Orai1 in Jurkat cells by flow cytometry. A total of four antibodies were described (2C1.1, 2D2.1, 2B7.1, and 5F7.1), which were all shown to bind Orai1 with high affinity by the KinExA instrument with dissociation constants (Kd) of ~20–100 pMolar. The antibodies isolated were unable to bind to rat and mouse Orai1, and the authors used this information to suggest putative epitope sites for the antibodies. Extracellular loop 2 (ECL2) shows only 60% identity between human and rat/mouse Orai1 species variants, suggesting this may be the epitope location. Human/Mouse chimeric proteins were constructed in which the ECL2 of human Orai1 was replaced with the mouse ECL2 and a reverse construct was made with Mouse Orai1 containing the human Orai1 ECL2. Binding studies on cells overexpressing the chimeras showed that the monoclonal antibodies were unable to bind to the Human Orai1 containing mouse ECL2 and that antibodies did bind to Mouse Orai1 containing the human ECL2. These results confirmed ECL2 as the binding epitope for the Orai1 antibodies. Further studies based on site-directed mutagenesis further defined the epitope to the region of amino acid residues 210–217, with some subtle differences in the precise epitope targeted by the four antibodies.
Subsequent studies demonstrated that the antibodies showed inhibition of Orai1 function. This was demonstrated by dose-dependent inhibition of cytokine release from thapsigargin-treated whole blood (2D2.1, 2C1.1, and 2B7.1) and dose-dependent inhibition by the 2C1.1 antibody of IL-17, IL-4, and IL-10 release from phorbol myristate acetate and ionomycin-stimulated whole blood. Antibody 2C1.1 was also shown to inhibit Orai1 current measured by PatchXpress recordings in HEK293 cells stably expressing Orai1 and STIM1. It is noteworthy that the antibody only caused partial inhibition of currents in this experimental format.
Cox et al. 96 adopted a more direct strategy to target the extracellular loops of Orai1 by using peptides spanning the first and second extracellular loops to immunize mice. Although both peptides gave rise to immune responses as detected by peptide ELISA, only ECL2 gave immune titers that recognized native Orai1 expressed on cells. A single Orai1 monoclonal antibody (10F8) was identified following hybridoma generation and was characterized in more detail. 10F8 was shown to bind to Orai1 overexpressed in a Ba/F3 host cell but did not bind to Ba/F3 cells expressing the related family members Orai2 and Orai3. The antibody was also able to bind to endogenous Orai1 expressed in Jurkat cells, and this was confirmed by reduced binding of the antibody to cells in which the Orai1 gene was knocked down using short hairpin RNA interference.
Further studies with the 10F8 monoclonal antibody showed that thapsigargin-induced calcium flux was specifically inhibited, confirming the antibody was functional. Interestingly, the authors also demonstrated that antibody-mediated internalization of Orai1 contributes to the functional inhibition observed and 10F8 induced internalization of Orai1 in primary T-cells. The antibody was also shown to inhibit T-cell proliferation and inhibited IL-2 and interferon (IFN)-γ production in both αCD3/αCD28-stimulated peripheral-blood mononuclear cells from healthy donors and stimulated synovial-fluid mononuclear cells obtained from rheumatoid arthritis patients. These studies were extended to demonstrate that antibody 10F8 was efficacious in vivo in a human T-cell-mediated graft-versus-host disease mouse model (GvHD). This was demonstrated by a significant delay in time to and incidence of GvHD measured by weight loss, reduction in CD4+ and CD8+ T-cell numbers, and reduced IFN-γ plasma levels compared to isotype controls.
These two studies both indicate the importance of ECL2 as a target epitope to generate functional monoclonal antibodies and illustrate that these antibodies can be successfully obtained either by strategies using whole-cell immunizations or via direct use of ECL2-derived peptides. An important feature of both studies was the ability to screen for binding to native Orai1 on cells stably expressing Orai1. A common theme that also emerges from the studies is that the monoclonal antibodies obtained only recognized the human Orai1 with no observed cross-reactivity to rodent Orai1. This illustrates an important consideration for the drug discovery process because the lack of cross-reactivity will affect the ability to conduct experimental in vivo studies for efficacy. In this case, it is known that the human Orai1 ECL2 has relatively poor sequence identity to rodent Orai1 ECL2 (60%), and this makes obtaining cross-species recognition with a monoclonal antibody challenging.
TRPA1
The transient receptor potential ankyrin 1 (TRPA1) channel is a nonselective cation channel that has been implicated in pathophysiologies that include asthma, cough, itch, and inflammatory pain. 98 Agonists of the channel include molecules such as mustard oil and allicin, which cause pain and pain-related behavior. TRPA1 antagonists are therefore being pursued as potential therapeutics.
Lee et al. 76 recently described the isolation of antagonist monoclonal antibodies that inhibit activation of TRPA1 by a variety of stimulants including chemical irritants, cold, and osmotic effects. The strategy used to isolate antibodies involved immunization of mouse strains with cells, plasmids, and adenoviruses expressing TRPA1 followed by generation of hybridomas and screening of conditioned media from hybridoma lines to identify mAbs showing selective binding to U2OS cells overexpressing TRPA1. The TRPA1-overexpressing cells were isolated by inserting a FLAG tag into the predicted extracellular loop 1 of TRPA1, which enabled fluorescent cell sorting to be used to enrich for high-expressing cells. These cells were also checked to confirm that the insertion of the epitope tag did not affect ion channel activity detected by 45 Ca influx. Sixteen antibodies were identified that showed greater than twofold specific binding to TRPA1 U2OS cells compared to parental control cells. Purified antibodies were then tested for binding to a second cell line (CHO) expressing TRPA1 and were also tested for their ability to inhibit allyl isothiocyanate (AITC)-stimulated 45 Ca uptake. Two hybridoma lines (2B10 and 2D1) were identified that showed greater than 50% inhibition of AITC-induced TRPA1 activation, and the antibodies were subcloned to confirm monoclonality. These antibodies were further tested and shown to cause concentration-related inhibition of AITC-induced 45 Ca uptake, cold-induced 45 Ca uptake, and osmotic pressure–induced 45 Ca uptake. Although detailed epitope mapping was not done, the authors suggested that the antibodies were targeting ECL3 (which is part of the pore) based on considerations of activity against both human and rat TRPA1 and sequence identities in the loop 3 region.
This successful isolation of functional antibodies can be attributed to the immunogenicity of the pore loop sequence and the very effective screening strategy using direct binding assays to cells (with the added benefit of a positive control due to the FLAG tag) and subsequent activity assays. Interestingly, the authors do state that the potency of the antibodies is too weak to consider for therapeutic applications, and it is also noteworthy that the antibodies are unable to completely block the channel unlike a small-molecule inhibitor, AMG9090.
Nav 1.7
Voltage-gated sodium channels (Nav) are responsible for action potential initiation and propagation in excitable cells with nine highly homologous subtypes encoded by the human genome.99,100 The pore-forming unit of Nav channels consists of a single polypeptide with four non-identical but homologous repeat domains (domains I–IV). Each repeat consists of six transmembrane helices (S1–S6) with the first four helices (S1–S4) acting as a voltage sensor domain (VSD) and the S5–S6 segments assembling together as a tetramer in the membrane to form the ion-conducting pore. 103 In the VSD, S4 contains arginine residues that sense membrane potential changes and together with S3 form a unit known as the voltage sensor paddle, which moves in response to membrane potential changes and is coupled to pore opening and closing. This voltage sensor paddle has been the target of a number of natural peptide toxins isolated from venomous species of spiders and scorpions that cause potent inhibition of Nav channels.104,105 Each subtype is implicated in various physiological and disease processes.
Nav1.7 has generated particular interest because loss-of-function mutations in SCN9A, the gene encoding Nav1.7 in humans, lead to congenital inability to sense pain without affecting other sensory pathways.99,101 There are also gain-of-function mutations in SCN9A that lead to episodic pain disorders such as erythromelalgia and paroxysmal extreme pain disorder. 102 Together, these observations support Nav1.7 as a potential analgesic target for a variety of pain disorders.
In 2014, Lee at al. 80 described the first monoclonal antibody targeting Nav1.7. This seminal paper describes the use of an elegantly simple strategy to isolate the functional antibody. Isolation of a monoclonal antibody against the channel was achieved by immunizing mice with a peptide (VELFLADVEG) that corresponds to the loop between helices S3 and S4 in domain II. Thus, the antibody response was directed to the voltage sensor paddle of the channel. The hybridomas derived from the mice were screened to identify antibodies that bind to the recombinant domain II VSD. A single functional IgG1 (SVmab1) was obtained by this approach. A negative control monoclonal antibody (CTmab) was raised by targeting the peptide sequence, HHPMTEEFKN, in the S1–S2 loop. The S1–S2 loop does not move in response to changes in membrane potential and is therefore not likely to be involved in channel gating. It is also interesting to note that the peptide sequence used as antigen to obtain SVmab1 is part of the recognition site for two inhibitory scorpion toxins (Huwentoxin IV 104 and Hainantoxin III 105 ). This highlights the importance of this amino acid sequence region for functional activity and as a potential functional antibody epitope.
SVmab1 and CTmab were tested in patch clamp experiments on HEK293 cells transiently expressing human Nav1.7. SVmab1 significantly reduced the sodium currents (75% inhibition at 100 nM), whereas CTmab had no effect on the currents at 1 µM. The specificity of S3–S4 loop targeting was confirmed by demonstrating that the inhibitory effects of SVmab1 on the sodium currents was eliminated in the presence of the peptide that was used as the antigen. Further characterization of SVmab1 demonstrated that it was Nav1.7-specific and did not inhibit other sodium channel subtypes with the exception of Nav1.6, in which it caused a partial inhibition of currents. This latter observation is expected because the S3–S4 loop of Nav1.6 contains a peptide sequence that is 80% identical to the antigen. Following the demonstration of Nav 1.7 inhibition, the authors used the monoclonal antibody in a variety of in vivo models of pain and itch and were able to demonstrate suppression of inflammatory and neuropathic pain and inhibition of acute and chronic itch.
Discussion and Perspectives
Ion channels are widely regarded as important therapeutic targets that to date have been targeted mainly by small molecules. Antibodies represent a significant and growing class of successful therapeutic drugs and are well established as effective treatment options for diseases such as cancer, inflammatory disease, and autoimmune disorders. A significant literature has demonstrated that antibodies can be used as experimental tools to study ion channel function, but to date no antibodies suitable for therapeutic intervention have been developed. Nevertheless, antibodies represent a significant opportunity by virtue of their specific properties, which include exquisite specificity, options for tailoring their affinity and potency, and the ability to modulate their effector function and pharmacokinetic properties by engineering of the Fc region. It is also possible now to consider modifying antibodies to favor delivery of molecules across the blood–brain barrier to enable targeting of not only peripheral ion channels but also those located in the CNS.
Although no therapeutic ion channel antibodies are described to date, there are a number of new studies reporting functional monoclonal antibodies that give rise to some optimism that effective drugs are on the horizon. Challenges remain, however, and issues relating to effective antigen generation, expression of ion channels, and screening methods suggest that antibody screening campaigns will require a portfolio of techniques to isolate effective functional antibodies. It is also noteworthy that in several recently reported cases of functional antibodies, relatively few functional antibodies were isolated from immunization campaigns and only partial inhibition of ion channel activity was achieved. This further confirms the challenging nature of the task and suggests that the epitopes targeted by the antibodies were suboptimal. Many reports to date also describe mouse monoclonal antibodies, and this raises a further requirement for humanization and possibly further optimization of binding properties before clinical use can be considered.
In summary, the feasibility of identifying functional antibodies to ion channels has taken significant steps forward in the past decade, and advances in the field of ion channel biochemistry and structural understanding coupled with advances in antibody discovery and screening technologies strongly suggest that effective functional antibodies may be developed with significant opportunities for therapeutic treatment of diseases covering a wide spectrum of pathophysiologies.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received support through their employment at MedImmune for the research, authorship, and/or publication of this article.
