Abstract
The epithelial sodium channel (ENaC) plays a crucial role in salt and water homeostasis and is primarily involved in sodium reabsorption in the kidney and lung. Modulators of ENaC function, particularly within lung epithelia, could offer potential treatments for a number of diseases. As a constitutively active sodium channel, ENaC expression at the cell membrane is highly regulated through rapid turnover. This short half-life of the channel at the membrane and cytotoxicity from overexpression pose a problem for reagent generation and assay development in drug discovery. We have generated an HEK293 stable cell line expressing ENaC β and γ subunits containing the PY motif trafficking mutations found in Liddle’s syndrome to overcome rapid channel turnover at the membrane. A BacMam virus was used to transiently express the ENaC α subunit to reconstitute channel function to reduce the toxicity associated with long-term overexpression. We have configured a 384-well FLIPR membrane potential antagonist assay for high-throughput screening and an IonWorks Quattro electrophysiology antagonist assay that is predictive of potency values derived from primary lung epithelial cell short-circuit measurements. The triage strategy for compound screening and profiling against this target using these assays has resulted in the discovery of novel chemotypes.
Introduction
The epithelial Na+ channel (ENaC) is a member of the ENaC/degenerin ion channel family. 1 ENaC is located on the apical membrane of polarized epithelial cells in the lung, distal nephron, distal colon, sweat ducts, salivary glands and taste buds, where it plays a key role in the reabsorption of Na+.1–4 It is critical in maintaining Na+ and K+ haemostasis 1 as well as the ionic content, volume, and osmolarity of extraepithelial fluids in the lung. 5
ENaC is composed of three homologous subunits, termed α, β, and γ, that are encoded by distinct genes. 6 Although functional channels can be formed by assembly of α subunits alone, β and γ are required for maximal expression and relevant channel activity on the cell surface. 6 The precise structure of the native channel remains unresolved, but based on crystallographic studies of the homologous ASIC1 channel, ENaC is believed to have 1:1:1 α, β, γ subunit stoichiometry.7,8 Importantly, ENaC is constitutively active, so wherever the channel is functionally expressed, it continuously conducts Na+. The surface density of ENaC, and hence the transmembrane Na+ flux, is under a complex hormonal control involving transcriptional and post-transcriptional regulation of its synthesis and insertion as well as retrieval of active channels from the apical membrane. 9 Under normal conditions, ENaC has a membrane half-life of less than 1 h, because of binding of ubiquitin ligase Nedd4-2 to C-terminal domains of the channel, which target the protein for lysosomal degradation.10,11
Mutations in the C-terminus PY motif of the β and γ subunits disrupt this interaction and lead to accumulation of active channels on the cell surface and a subsequent increase in Na+ reabsorption and K+ loss. 12 These mutations are clinically manifested as autosomal dominant Liddle’s syndrome, characterized by elevated blood pressure, hyperkalemia, and pseudohyperaldosteronism. 13 Such gain-of-function mutations in ENaC may also contribute to the pathophysiology of cystic fibrosis. 14 Conversely, loss of function mutations in ENaC cause autosomal recessive severe systematic pseudohypoaldosteronism (type 1: PHA-1), which is characterized by hypotension with renal insensitivity to aldosterone. 13 Accordingly, modulators of ENaC channels may have useful therapeutic benefit. Cystic fibrosis is most widely pursued through mucus hydration and clearance by aerosol delivery of channel inhibitors delivered directly to the lung.15–19
To date, efforts to identify novel channel modulators have been hampered by the availability of suitable in vitro screening methods. Although both conventional patch clamp electrophysiology and short-circuit current measurements in epithelial cells using Ussing chambers can yield highly relevant functional measures of ENaC, each lack the throughput and ease of use to support wider-scale screening and compound profiling. Here, we report the development, optimization, and validation of novel fluorescence (FLIPR membrane potential [Vm]) and automated patch clamp electrophysiology assays for human ENaC. In combination, these assays enable high-throughput and relevant screening for novel ENaC modulators.
Materials and Methods
Compounds and Materials
Three standard ENaC small-molecule inhibitors, amiloride, benzamil, and phenamil, were purchased from Sigma Aldrich (St. Louis, MO). A series of novel ENaC blockers first described by Collingwood and Smith 20 and Hirsh et al.15,16 were synthesized by GSK Medicinal Chemistry or obtained from the GSK compound collection. All compounds were initially solubilized in 100% DMSO to a concentration of 10 mM, with the exception of amiloride (30 mM DMSO stock for FLIPR assays). Compounds were further diluted in buffer before addition to the assay. Membrane potential indicator dyes and other FLIPR reagents, as well as all cell culture materials, were purchased from Invitrogen (Carlsbad, CA), unless stated otherwise.
Recombinant Expression of ENaC in HEK293 Cells
ENaC subunit cDNAs were amplified from human kidney (α and β) and human brain (γ) marathon libraries. The sequences are identical to GenBank references NM_001038, NM_000336, and NM_001039, respectively. The α subunit cDNA fragment was placed under the control of the CMV promoter in the pBacMire shuttle vector and transfected into SF9 insect cells, and BacMam viruses were generated as previously described by Condreay et al. 21 The β and γ subunits were subcloned into pBacMire (neomycin/geneticin resistance) and pBacMireHyg (hygromycin resistance) respectively, under the control of the CMV promoter. HEK293 cells were co-transfected with β and γ subunit constructs, and stable cell lines were established under the selection of 0.5 mg/mL geneticin and 0.2 mg/mL hygromycin. Cells were maintained in DMEM Ham’s F12 medium supplemented with glutamax and 10% fetal bovine serum (FBS), at 37 °C with 5% CO2, dissociated with TrypLE, and split 1/5 to 1/10 for propagation. Mutations in the β (from CCC-AAC-TAT to GCC-AAC-TTA, corresponding to P618A Y620L) and γ (from CCC to TAG, corresponding to P624Stop) subunits (ENaC β [P618A Y620L; β*] and γ*: ENaC γ [P624Stop; γ*], respectively) were created using the Stratagene QuikChange XL Site-Directed Mutagenesis Kit (cat. No. 200516) according to the manufacturer’s instructions. HEK293 ENaC βγ and β*γ* stable cell lines were maintained under a constant selection of 0.5 mg/mL geneticin and 0.2 mg/mL hygromycin. For functional experiments, the α subunit was introduced to form the full ENaC channel via a BacMam virus added 18 to 24 h prior to the start of the experiment.
FLIPR Membrane Potential Assay
Cells expressing ENaC β and γ subunits were plated onto 384-well, black, clear-bottomed assay plates (Greiner, Monroe, NC) for 18 to 48 h at 37 °C in culture media supplemented with ENaC α subunit BacMam virus (multiple of infection [MOI] = 2.4) at a density of 15,000 cells per well. The medium was then aspirated using a Tecan Plate Washer, leaving 10 µL residual volume in each well. Twenty microliters of FLIPR membrane potential dyes from Molecular Probes (Eugene, OR) (Blue: FMP [cat. No. R8042] or FMP2 [cat. No. F8181]; Red [cat. No. R8126] or DiBac2 [cat. No. B438]), diluted at the manufacturer’s recommended concentrations in Tyrode’s buffer (Sigma cat. No. T2145 supplemented with sodium bicarbonate: in mM, NaCl 136.9, KCl 2.7, H2NaO4P 0.4, CaCl2 1.8, MgCl2 1.1, D-glucose 5.6, NaHCO3 11.9; pH adjusted to 7.35 with NaOH), was then added to each well using a Multidrop and incubated at room temperature for 1 h. Compound plates with 384 wells were prepared by adding 50 µL of Tyrode’s buffer containing 0.04% pluronic acid into each well preloaded with 1 µL of 1/3 serial dilution of compounds in DMSO. 10 μL of the diluted compounds was transferred to each well in the assay plate using the dispensing head of a FLIPR (Molecular Devices) while simultaneously monitoring whole-well fluorescence (excitation/emission λ 510–545/565–625 nm). Amiloride (150 µM) and DMSO were used as high (100% channel block) and low (vehicle) controls, respectively. The kinetic changes in fluorescence intensity following compound addition were recorded using SoftMax Pro software. The average steady-state fluorescence intensity values were measured before and after drug addition and exported for data analysis. Curve fitting and –lg(IC50 at [M] concentration) (pIC50) determination were carried out using a four-parameter logistic model (Excel XC50 Module). For Na+ add-back experiments, Na+-free buffer contained (mM) N-methyl-D-glucamine 132, KCl 2, CaCl2 2, MgCl2 1, HEPES 10, and D-glucose 5, pH 7.3 with HCl.
IonWorks Planar Array Electrophysiology
HEK293 ENaC β*γ* stably expressing cells were seeded at ~3 × 106 cells in T175 cm2 flasks and grown for 18 to 24 h. ENaC α subunit BacMam was added at an MOI of 25, and cells were maintained for a further 18 to 24 h in medium containing 70 mM NaCl to minimize channel-mediated cellular toxicity (50:50 mix of standard growth medium and DMEM/F12 without NaCl, supplemented with 10% FBS).
Planar array electrophysiology recordings were made on an IonWorks Quattro instrument using both single-cell (HT) and population patch-clamp (PPC) configurations.22–26 Cells were prepared immediately prior to experimentation as previously described 26 and resuspended in warm external buffer (in mM, Na-gluconate 120, KCl 5, NaCl 20, CaCl2 1, MgCl2 2, HEPES 10, pH adjusted to 7.35 with NaOH, 0.2 µm filter sterilized) to yield a final cell density of ~2 × 106 cells per mL. Internal buffer was composed of (mM) K-gluconate 120, NaCl 5, KCl 20, HEPES 10, CaCl2 2, MgCl2 1, pH adjusted to 7.2 with KOH (0.2 µm filter sterilized). Cell perforation was achieved using internal buffer supplemented with 0.2 mg/mL amphotericin B.
Individual cells (or wells for PPC) were voltage clamped and the test protocols applied in groups of 48 using the electronics (E-) head. 26 A holding potential (Vm) of −40 mV was applied for 5 s followed by a ramp (250 ms, −100 mV to +100 mV with a 20 ms hold at each extremity) and step (50 ms, 0 mV; 50 ms, +30 mV) protocol. The corresponding currents were recorded first in the absence (pre-) and then the presence (post-) of amiloride, test compound, or vehicle (compound incubation time of 3–5 min). To reduce any potential compound carryover effects due to compound sticking to the fluidics (F-) head pins, the 50% DMSO solvent wash facility was used in these experiments. Leak subtraction was disabled, and currents were measured either at 0 mV (where the contribution of nonbiological leak is negligible 24 ) or −100 mV (and referenced to 100% block amiloride controls in PPC). Curve fitting and pIC50 determinations were carried out by fitting to a four-parameter logistic equation (Microsoft Excel XC50 Module).
Ussing Chamber Short-Circuit Current Recording
Human airway epithelium cultures (HEAC) grown in air-liquid interface (AIR-100-SNP, 12 tissues in Snapwell inserts) were purchased from MatTek Corp (Ashland, MA). On the day of use, Snapwell inserts containing EpiAirway tissue (1 cm2 diameter) were placed in Ussing chambers containing Krebs bicarbonate buffer (KBS; composed of [mM] NaCl, 118; KCl, 5.4; NaH2PO4, 1.2; MgSO4, 1.2; CaCl2, 2.5; NaHCO3, 25.0; dextrose, 11.1, and gassed with 95% O2 and 5% CO2, pH 7.4) for bioelectric measurements. Throughout the experiment, each half chamber contained 5 mL of KBS maintained at 37 °C and bubbled with 95% O2/5% CO2. Tissues were equilibrated under open-circuit conditions until transepithelial potential was stable (typically within 45 min) with washing every 15 min.
Bioelectric measurements were made with an epithelial voltage clamp (model VCC MC6) and a self-contained Ussing chamber system (both purchased from Warner Instruments, Inc., Hamden, CT), digitized by a DataQ DI-720 series real-time Data Acquisition System (Physiologic Instruments, San Diego, CA) and displayed and recorded using Acquire and Analyze 2.3 Software (Physiologic Instruments, version 2.3.177). Once transepithelial potential was stable, transepithelial voltage was clamped at zero, and the resulting short-circuit current (Isc ) was recorded prior to and following the addition of ENaC blocker compounds that were added in a series of cumulative increasing concentrations (threefold increments). ENaC blockers were prepared as a 10 mM stock solution in DMSO and stored at −20 °C. Working solutions were prepared daily as a series of 10-fold dilutions in KBS. Data analysis was carried out using GraphPad Prism (version 4.03).
Results
Pharmacology of ENaC in Human Airway Epithelia
Prior to assembling high-throughput assays, the pharmacology of ENaC was determined by short-circuit current (ISC) measurements in HEAC grown at an air-liquid interface. 27 We considered this HEAC assay the gold standard because human ENaC channels, presumably composed of relevant subunits and auxiliary regulatory proteins, are endogenously expressed under conditions that most closely resemble the environment in native polarized epithelia. The absolute and relative potencies of 17 known inhibitors (amiloride, benzamil, phenamil, published and patent literature compounds) were quantified and used as the target pharmacology profile to which we attempted to align (correlate) higher-throughput assays. Under the experimental conditions, HEAC exhibited large, stable ISC of 31.4 ± 12.7 µA/cm2 (n = 45). Addition of amiloride (100 µM) reduced the ISC by >95% to 1.4 ± 1.6 µA/cm2 (n = 6). In cumulative concentration-response curve assays, all 17 inhibitors produced a substantial, concentration-dependent reduction in ISC ( Fig. 1 ). The pIC50 value for amiloride was 6.47 ± 0.03 (n = 6, Hill slope = 0.97) with a maximum percentage inhibition of 96.0 ± 5.0 (n = 6) that was shown to be reversible on washout (86.3% ± 6.3%, n = 6). These data were consistent with previously published values. 28 The ENaC inhibitor Novartis-5 (mean pIC50 values of 8.7 ± 0.04, n = 2, Hill slope = 0.72, maximum inhibition = 93.6% ± 2.7%, reversibility = 14.5% ± 0.3%) was >100,000-fold more potent than the least potent compound trimethoprim (pIC50 = 3.46, n = 1, Hill slope = 0.75, maximum inhibition = 68.4%, reversibility = 80.6%).
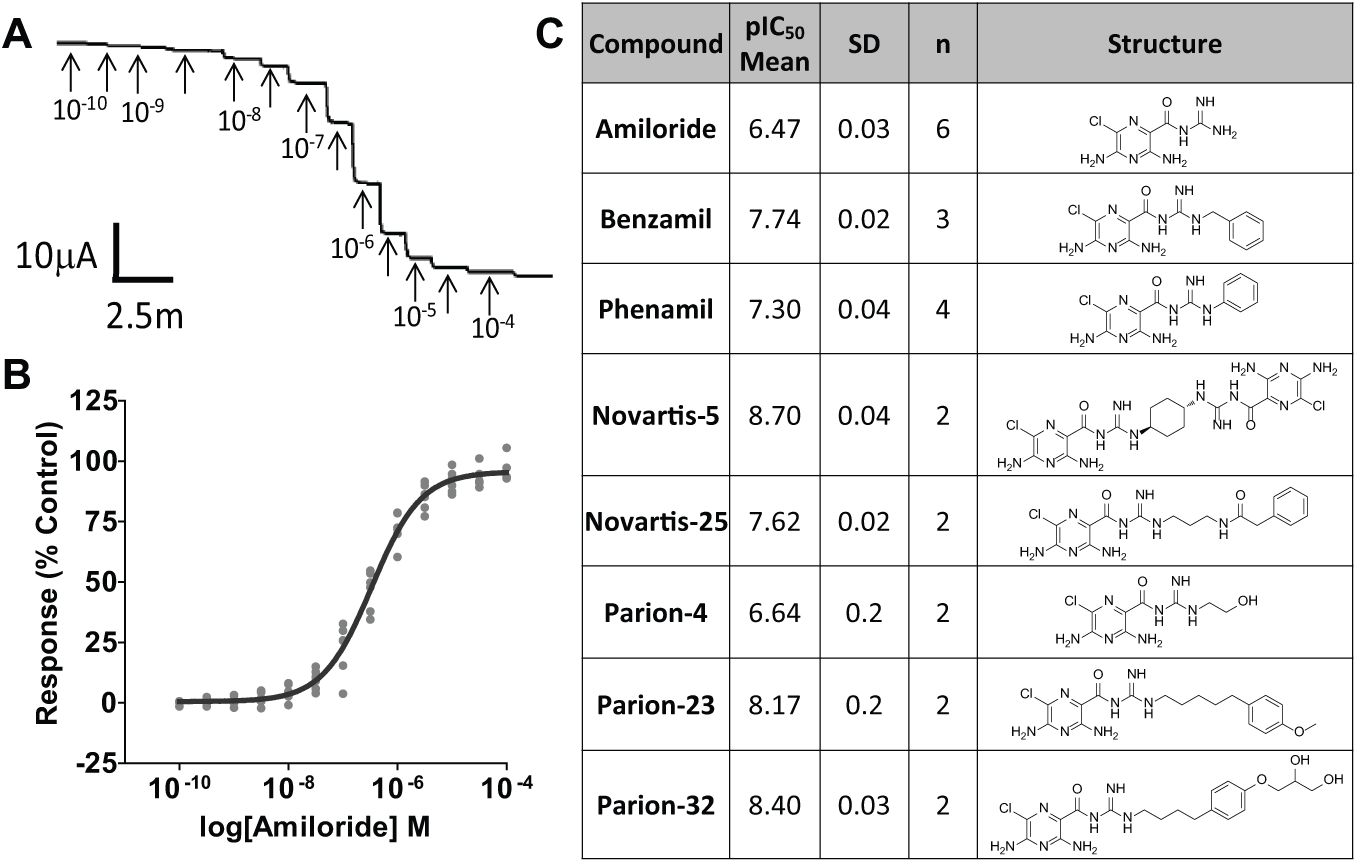
Ussing chamber recordings of human airway epithelium. (
Analysis of ENaC Activity in Lung- and Kidney-Derived Epithelial Cell Lines
As ENaC is known to be present in lung and kidney epithelia, we first examined a range of lung- and kidney-derived epithelial cell lines for the possibility of developing high-throughput assays using endogenously expressed ENaC. These cell lines included human bronchial epithelia (16HBE 14o- [kindly provided by Dr. D. C. Gruenert, University of California, San Francisco, California, USA] and Beas 2B [ATCC CRL-9609]), human lung carcinoma A549 (ATCC CRL-185), human lung adenocarcinoma NCI-H441 (ATCC HTB-174), human epithelial kidney HEK293 (ATCC CRL-1573), and dog kidney MDCK (ATCC CCL-34). Amiloride-sensitive whole-cell currents were measured using single-hole planar array electrophysiology but were found to be too small to conduct detailed pharmacology studies upon: the largest signal obtained with H441 cells was <150 pA at 0 mV ( Table 1 ). Efforts to increase the functional expression by pretreating cells with dexamethasone, forskolin, or 1-methyl-3-(2-methylpropyl)-7H-purine-2,6-dione were unsuccessful (data not shown).
Characterization of lung- and kidney-derived epithelial cell lines for ENaC activity measured by high-throughput automated electrophysiology.
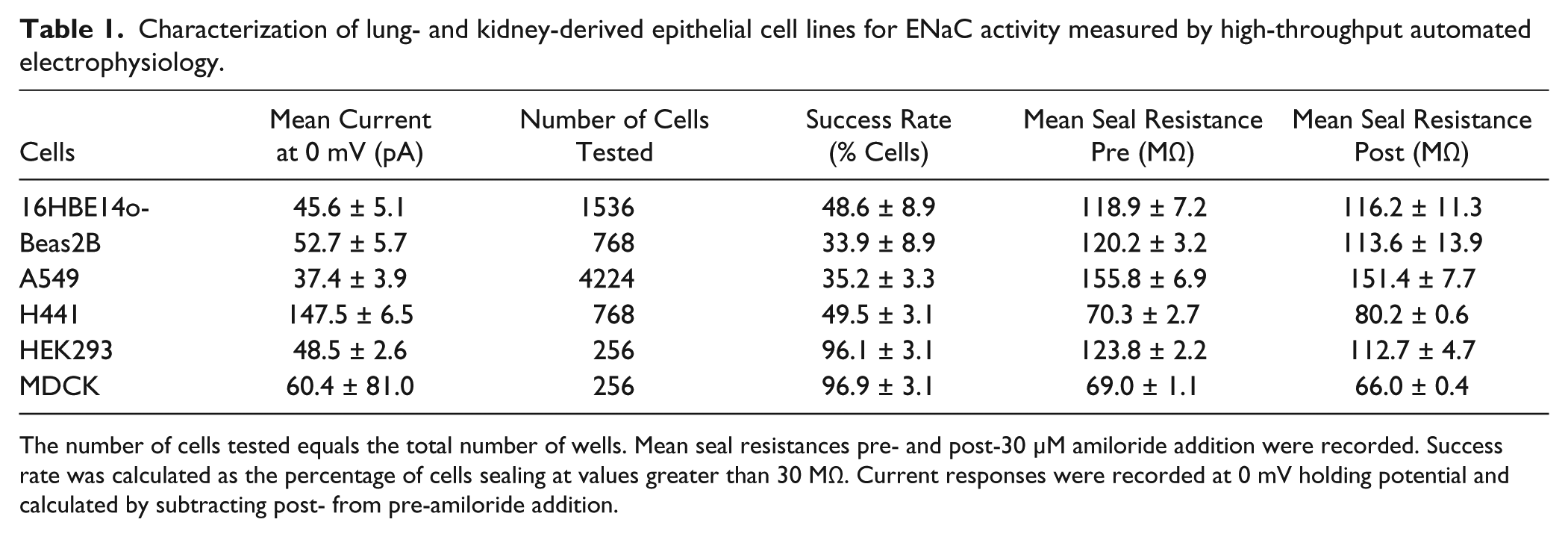
The number of cells tested equals the total number of wells. Mean seal resistances pre- and post-30 µM amiloride addition were recorded. Success rate was calculated as the percentage of cells sealing at values greater than 30 MΩ. Current responses were recorded at 0 mV holding potential and calculated by subtracting post- from pre-amiloride addition.
Functional Expression of Recombinant ENaC
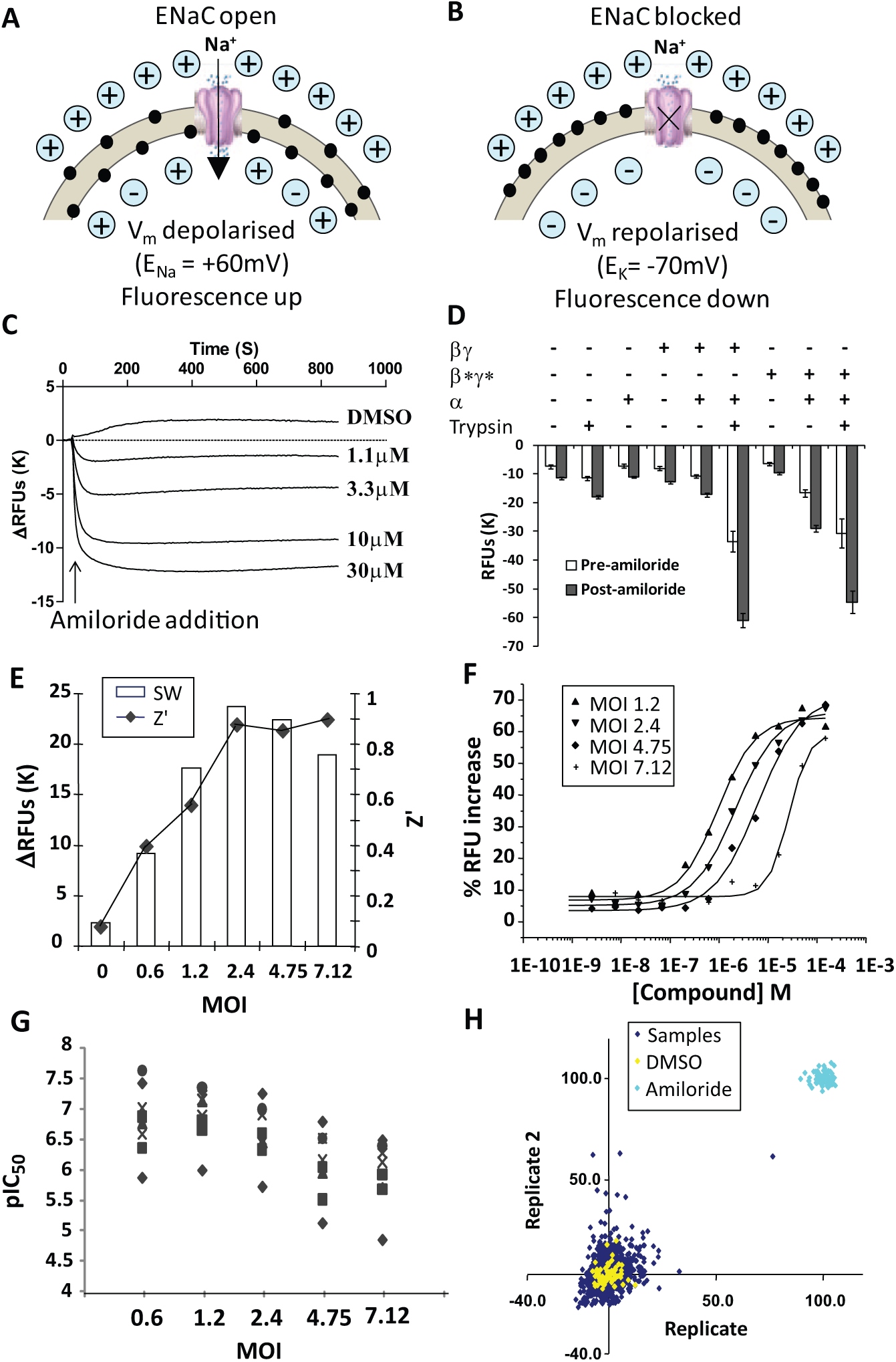
We next explored expression of human recombinant ENaC in mammalian host cells. As the channel is constitutively open, long-term overexpression is likely to be associated with cellular toxicity due to excess Na+ influx. Thus, an HEK293 stable cell line co-expressing the β and γ subunits was cultivated, and the fully functional channel constituted by transient introduction of the α subunit via BacMam viral transduction (for 18–24 h prior to the functional assay). To validate this expression strategy, ENaC activity was initially assessed by determining the magnitude of the hyperpolarization response to amiloride in a fluorescence-based membrane potential assay on the FLIPR (
Fig. 2A
(
To overcome this, mutations were introduced to the PY motif of β (P618A Y620L) and γ (P624Stop) subunits (referred to as β* and γ*, respectively) to mimic those found in Liddle’s syndrome, which have been shown to significantly increase the cell surface expression and stability of ENaC. 11 Expression of the α subunit into a β*γ* stably expressing cell line yielded significantly higher basal fluorescence than observed with wild-type subunits, indicating an ENaC-mediated cellular depolarization. Consistent with this, the amiloride-evoked hyperpolarization was >2-fold higher in αβ*γ* compared with αβγ-expressing cells ( Fig. 2D ).
Interestingly, despite the apparent low functional activity of ENaC αβγ, the basal fluorescence and amiloride-induced hyperpolarization were dramatically enhanced when cells were treated with trypsin, a serine protease enzyme. Similar observations were made with α β*γ* cells ( Fig. 2D ). Proteolytic treatment of ENaC increases channel open-state probability by releasing Na+-“self-inhibition,” and PY motif mutations further enhance this effect.29,30 We did not pursue this approach further because preliminary studies indicated that trypsin treatment attenuated the effects of ENaC blockers (data not shown) and would likely yield discrepant pharmacology values.
To verify functional activity and basic biophysics of ENaC, α β*γ* automated planar array electrophysiology methods were applied prior to subsequent FLIPR assay optimization and screening (discussed later in this article).
Development and Optimization of ENaC FLIPR Membrane Potential Assays
A range of membrane-potential sensitive fluorescent dyes were evaluated as suitable reporters for high-throughput (384-well) FLIPR assays. Pilot studies with DiBAC and FMP Red were unsuccessful because of a high background fluorescence and low amiloride-sensitive signal window in ENaC α β*γ* HEK293 cells. With the FMP Blue kit, clear hyperpolarizing responses to amiloride were observed, but the potency (mean pIC50 value 5.22) was 24-fold lower than observed in the HEAC Ussing chamber assay (mean pIC50 value of 6.58). Strikingly, the highly potent inhibitor Novartis-5 (HEAC pIC50 8.7) was >1000-fold less active in this FMP Blue membrane potential assay (pIC50 <5). Switching to a second-generation FMP2 Blue Kit produced some improvement. Although the measured potencies of amiloride and phenamil remained unchanged, we observed a large increase in the effect of Novartis-5 (pIC50 6.6 FMP2) and some other ENaC blockers (
Fig. 3
). Interestingly, the FMP Blue and FMP2 Blue kits differed in the results obtained in Na+ substitution and add-back experiments. When switching from Na+-containing Tyrodes to an NMDG-based Na+-free solution, an amiloride-evoked reduction in fluorescence (hyperpolarization) was observed with FMP (
Fig. 3A
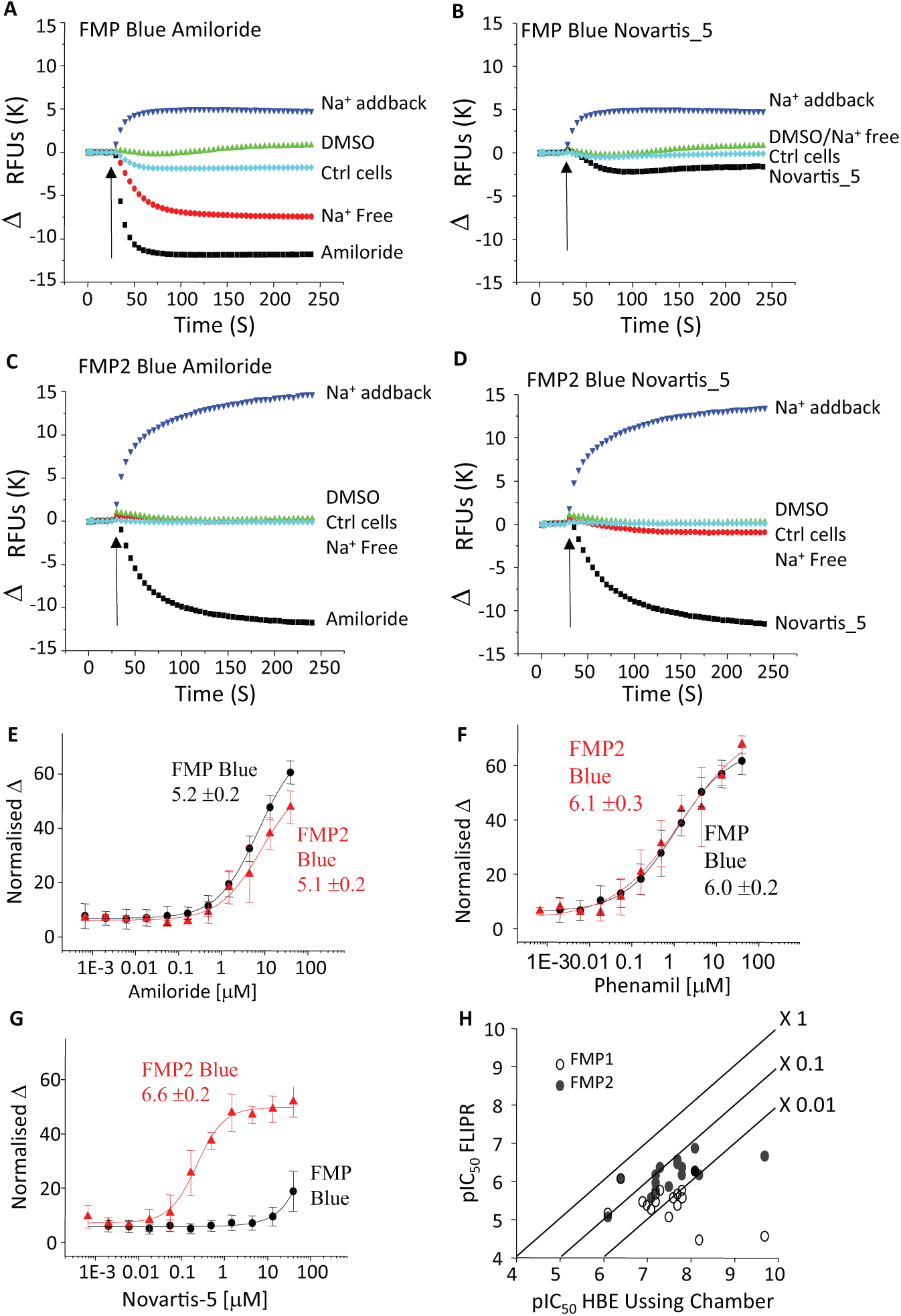
Comparison of dyes for the FLIPR assay using HEK293 β*γ* stable cell line with α subunit BacMam. Fluorescence (relative fluorescent units) was recorded online by FLIPR. Cells were loaded with FMP Blue (
In an attempt to address the underestimation of ENaC-blocking activity, we considered the nonlinearity of channel block with the change in membrane potential. Theoretically, with high levels of functional activity of ENaC, a substantial proportion of channels will need to be blocked before the membrane will begin to hyperpolarize. To explore this, we titrated the expression levels of ENaC by modifying the multiplicity of infection of the BacMam for the α-subunit. Across the range of 7.12 to 1.2 MOI, we observed a clear increase in compound potency as the MOI was reduced (i.e., fewer channels were expressed;
Fig. 2E
As a final test of robustness, four plates of test compounds (352 samples per plate at a final assay concentration of 10 µM, 16 high [150 uM amiloride], 16 low [0.5% DMSO] controls) were tested in duplicate. The Z′ value was 0.80 ± 0.05 (SD, n = 8), and the activity of the test samples was centered on 0% inhibition for both replicates ( Fig. 2H ).
Development and Optimization of ENaC IonWorks Patch Clamp Electrophysiology Assay
Taking learnings from assays for other non–voltage-gated channels,24,26 we next developed a planar array patch clamp electrophysiological assay for ENaC using IonWorks Quattro. In single-hole experiments on control HEK293 β*γ*-expressing cells, grown in standard media without the α-subunit, the measured seal resistance was 197.1 ± 63.6 MΩ (n = 366). A voltage step to −100 mV (20 ms holding prior to ramp) produced a small inward current of 320 ± 140 pA (n = 356) that was relatively unchanged in the presence of amiloride (data not shown). Figure 4A shows that the voltage ramp (200 ms, −100 to +100 mV) produced a small linear current (615 pA at −100 mV in this example), reversing close to 0 mV, which was insensitive to amiloride (10 µM; <10% block at −100 mV). This current was similar to that observed in wild-type (untransfected) cells and is likely a (contaminating) non-ENaC leak current ( Table 1 ). In the presence of the co-expressed α-subunit (BacMam), functional ENaC channels can be formed. Figure 4B shows that the voltage ramp (200 ms, −100 to +100 mV) produced a much larger linear current (2712 pA or 2.71 nA at −100 mV), reversing between +20 and +30 mV, which was highly sensitive to amiloride (10 µM; 76% block at −100 mV). The ENaC current, measured by subtracting the amiloride-resistant current from the control (no-amiloride) signal, displayed inward rectification and reversed toward the Na+ equilibrium potential (+58 mV under the ionic conditions). When compared in HT mode, large, inward amiloride-sensitive currents (mean value of −0.91 ± 1.26 nA; n = 153, values ranging from −6.62 to +1.06 nA) were observed in some but not all cells ( Fig. 4C ). In these cells, the initial seal resistances were highly variable but overall lower ( Fig. 4D ; mean HT value of 58.4 ± 50.8 MΩ, n = 191). Application of amiloride markedly increased the (membrane) resistance in cells that had low initial values (see Fig. 4D ; mean HT value 123.7 ± 57.5 MΩ, n = 190), indicating constitutive ENaC activity.
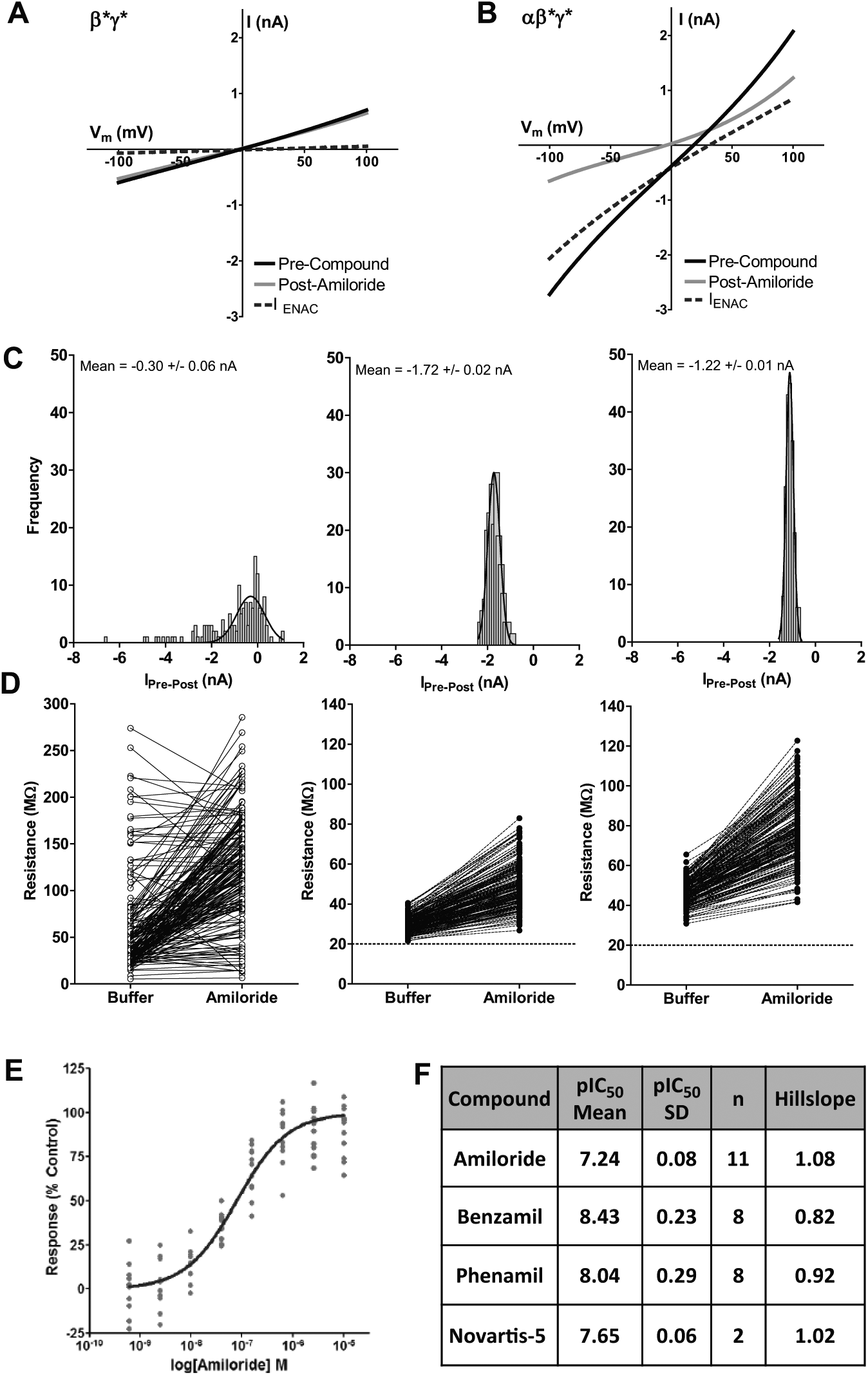
Population patch-clamp planar array electrophysiology recordings of epithelial sodium channel (ENaC)–derived currents in human recombinant ENaC αβ*γ*-expressing HEK cells. (
To overcome the large cell-to-cell variability in ENaC current amplitude, further studies were conducted in PPC mode in which the ensemble (average) current from up to 64 cells per well was measured. We also explored whether reducing the Na+ concentration in the growth/transfection medium for the HEK293 αβ*γ* cells would improve assay quality, hypothesizing that a high basal ENaC-mediated Na+ flux may be detrimental to cell health for electrophysiology recordings. When cultivated in standard sodium-containing culture media, the mean seal resistance of the cells in PPC was 29.5 ± 4.0 MΩ, n = 192, and was increased to only 49.5 ± 11.0 MΩ (n = 192) in the presence of amiloride ( Fig. 4D ). After culturing in low-sodium-containing media (half that of standard culture media), improved seal resistances (46.2 ± 6.0 MΩ and amiloride = 77.8 ± 16.4 MΩ [n = 191] for basal and amiloride recordings, respectively) were observed, likely owing to improved cell health. The amplitude of the ENaC-specific currents was not increased under low-sodium conditions; however, the consistency was greatly improved across the test plate (see Fig. 4C , Gaussian curve fitting).
Figure 4E shows the pharmacology of amiloride recorded under final assay conditions (PPC, low sodium) and plotted as percentage of maximum control (10 µM amiloride). The pIC50 value for amiloride was 7.24 ± 0.08 (n = 11) with a Hill slope of 1.08. The pharmacology of amiloride was unchanged in the presence of low-sodium versus normal media conditions (data not shown). The pharmacology of other known ENaC blockers is summarized in Figure 4F . Because of the variability of the assay, each data point was tested in quadruplicate on the plate (pseudo 96-well plate format). Average robust Z′ values of >0.1 were observed for this assay, with a cutoff of >0 applied to data for screening. All compounds were tested with a final assay concentration of 1% DMSO. This assay has previously been shown to be tolerable of ≤2% DMSO final concentration (data not shown).
Comparative Pharmacology, Hit Identification, and Triage
Using this IonWorks assay format, potency values for a set of known ENaC blockers were obtained and correlated well with that from the Ussing chamber (R2 = 0.81), without a noticeable general shift in potency (
Fig. 5A
). Correlation with FLIPR was also good (R2 = 0.83;
Fig. 5C
). The FLIPR assay understated the pIC50 values for the more potent compounds, by about 1 log unit (
Fig. 5B
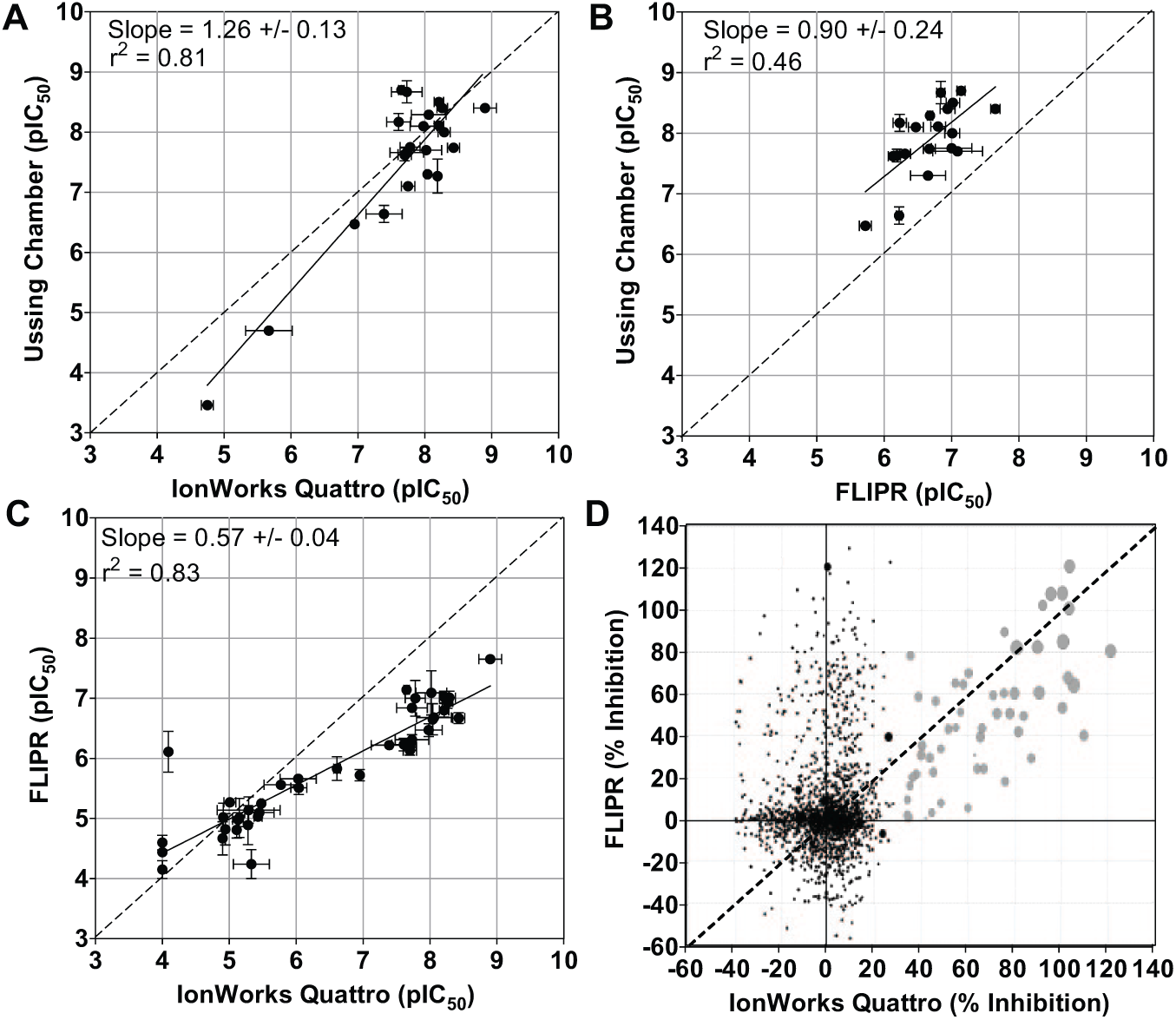
Correlation plots for assay comparison. (
We next compiled a focused set of ~2000 compounds based on homologies to several known ENaC blockers and profiled with the FLIPR and IonWorks assays in parallel initially in single-well format at 10 µM. pIC50 values for the hits were determined in subsequent experiments. The results are illustrated in Figure 5D . When 30% cutoff of response was applied, a hit rate 1.8% were obtained using IonWorks. Most of these hits were also positive in the FLIPR assay using the 30% cutoff, representing 1.4% of the total compounds, and this was increased to 1.7% when the 20% cutoff was applied. This result shows that the FLIPR Vm assay is capable of detecting most of the genuine hits but also reported a high rate of hits unconfirmed by IonWorks (10.9% and 14.4% with 30% and 20% cutoff, respectively), perhaps as a result of lower assay sensitivity and specificity.
Discussion
Owing to its rate-limiting role in regulating Na+ absorption and Na+/K+ balance, ENaC is clearly a potential point for therapeutic intervention for a number of diseases and has long attracted interest from academics and pharmaceuticals alike. It is therefore rather surprising that, to our knowledge, no high-throughput FLIPR or electrophysiological assay for this channel has been documented. An obvious answer might be the rather low level of channel activity of native epithelial cell lines that express wild-type ENaC ( Table 1 ). A high level of and uniform expression of ion channels is critical for the development of robust high-throughput assays, particularly planar array electrophysiology such as IonWorks. 31 Our approach has addressed these difficulties in several ways. First, the β and γ subunits were expressed in a stable cell line using IRES, a technology that enables uniform high-level expression in stable cell lines. 31 Second, mutations in the β and γ subunits that mimic those found in Liddle’s syndrome were introduced, which are known to prevent fast internalization and degradation as well as to increase the open probability of the channel. 30 Third, functional channels were reconstituted only transiently following the addition of α subunit BacMam, to avoid long-term functional overexpression, which may cause cell toxicity due to excessive cell swelling.
The mutations introduced to the PY motif of ENaC β and γ subunits did not appear to affect compound potency values generated by IonWorks. The values were close to that by measuring ISC of HEAC ( Fig. 5A ), despite the large differences in the level of channel expression, cell background, and the technology between the two platforms. This is likely due to the fact that the PY motif is close to the extreme C-termini of the proteins, which are intracellular and far away from the channel pore, 8 and are mainly involved in the regulation of trafficking rather than the biophysical properties of the channel. Existing tool compounds such as amiloride interact directly with the pore without the need to enter the cell.8,32
Despite the difference in functional FLIPR responses under normal assay conditions, trypsinization significantly elevated the activity of not only αβ*γ* mutant ENaC but also αβγ wild-type channel, generating equivalent maximal fluorescent responses in the FLIPR assay ( Fig. 2 ). Proteolysis is a well-documented mechanism of controlling the open probability of the channel.33,34 The cause for the equivalent maximal responses of the wild-type and mutant ENaC requires further investigation. If dye saturation is not the cause, alternative explanations could include the differences in proteolytic regulation between wild-type and mutant ENaC carriers and the implications for health and disease in these populations.
A reliable FLIPR assay was possible only with the second-generation FMP2 Blue dye, which overcomes several limitations of the first-generation FMP formulation by the ability to detect Na+-dependent depolarization and respond to all of the known ENaC blockers tested. The potency values were about 1 log unit lower than obtained with the Ussing chamber. The reason for this is unclear, but this level of potency shift is often true for membrane potential FLIPR assays. It is unlikely due to the buffering of compounds by excess amounts of nonfunctional ENaC, as potency values from IonWorks patch clamp recordings using the same cells were in much better agreement with the Ussing chamber. An interesting observation is that the potency of these compounds in FLIPR was quite sensitive to the amount of viral load to the cells, which may reflect the level of functional ENaC expressed on the cell surface.
The viral load of the IonWorks assay was about 10 times that of FLIPR, which would lead to significantly higher Na+ influx during cultivation, causing cellular swelling and compromising membrane integrity. This was resolved by growing the cells in low-Na+ medium. The change of growth conditions did not lead to alterations in the I-V relationship or the potency of amiloride, suggesting that the channel properties per se are unlikely altered.
The IonWorks Quattro assay using the ENaC recombinant cell line is highly predictable of compound potencies of HEAC ISC determined by the Ussing chamber. The FLIPR assay, on the other hand, was not only less sensitive but also reported a high degree of false-positives. Nevertheless, it detected the majority of the high-potency IonWorks positives in the focused screen. The significantly lower cost and simplicity of the FLIPR assay merit its consideration as a frontline assay to screen or profile large numbers of compounds. The IonWorks assay is best suited for postscreen activities, such as confirming target engagement and driving SAR campaigns. The HEAC ISC assay would be conducted prior to in vivo tests. A scheme of lead discovery that incorporates these assays is outlined in
Technology is perhaps now ripe for the systematic screening and profiling of diversity compound libraries for novel modulators for diseases either directly or indirectly involving ENaC function or dysfunction.
Footnotes
Acknowledgements
The authors would like to acknowledge Sergio Senar-Sancho, Oyee Chiu, Sandra Arpino, Simon Dowell, and Jeff Clare for general and technical support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
