Abstract
We investigated the use of Adaptive Focused Acoustics (AFA) technology to improve the performance of microtiter plate enzyme-linked immunosorbent assays (ELISAs). Experiments were performed with commercially available AFA instrumentation and off-the-shelf 96-well microtiter plate sandwich ELISAs. AFA was applied over a range of acoustic energies, temperatures, and durations to the antigen/antibody binding step of an ELISA for measuring HIV-1 p24 in tissue culture samples. AFA-mediated antigen/antibody binding was enhanced up to 2-fold over passive binding at comparable temperatures and was superior or comparable at low temperature (8–10 °C) to passive binding at 37 °C. Lower nonspecific binding (NSB), lower inter- and intra-assay coefficients of variation (CVs), higher Z′ factors, and lower limits of detection (LODs) were measured in AFA-mediated assays compared with conventional passive binding. In a more limited study, AFA enhancement of antigen/antibody binding and lower NSB was measured in an ELISA for measuring IGFBP-3 in human plasma. We conclude from this study that application of AFA to antigen/antibody binding steps in microtiter plate ELISAs can enhance key assay performance parameters, particularly Z′ factors and LODs. These features render AFA-mediated binding assays potentially more useful in applications such as high-throughput screening and in vitro diagnostics than assays processed with conventional passive antigen/antibody binding steps.
Introduction
Solid-phase double antibody sandwich enzyme-linked immunosorbent assays (ELISAs) performed in microtiter plates provide a convenient format for applications such as high-throughput screening (HTS) and in vitro diagnostics. However, their utility in these applications is often limited by nonspecific binding (NSB) and poor assay reproducibility, both of which are key to establishing a good Z′ factor. 1
Another limiting factor for the utility of microtiter plate ELISAs is the diffusion coefficient of the solution phase binding partner as it governs the antibody/antigen binding rate after completion of the initial rapid binding of reactants that occurs at the commencement of the incubation. 2 A commonly used procedure for accelerating antibody/antigen binding rates in conventional passive assays is heating at 37 °C for 1 h. However, assay reproducibility and NSB of ELISAs performed at 37 °C for 1 h are typically inferior to assays performed at lower temperatures for longer times.
Use of ultrasound to promote faster binding of antigen to immobilized antibody has been reported. 3 In that study, acceleration of binding of human choriogonadotropin to antibody immobilized on a cellulose support was demonstrated by immersing a test tube containing the solid phase in an ultrasonic water bath. However, ultrasonic water baths operate at frequencies of about 20 KHz, with wavelengths of about 100 mm, and during operation can self-focus in a random manner that creates zones of high acoustic energy that fluctuate in both intensity and position. Consequently, delivery of a uniform acoustic dose to all wells in a microtiter plate cannot be achieved in an ultrasonic water bath. Application of acoustic energy to samples using ultrasonic probes allows more concentrated energy to be imparted to samples than bath sonicators. However, application of ultrasound in this modality to samples in microtiter plates can result in sample cross-contamination and sample contamination by erosion of probe tips.
Ultrasound in the form of surface acoustic waves (SAW) has been reported for removal of nonspecifically bound proteins on microarrays leaving selected antigen receptor interactions bound during the acoustic cleaning process with retention of their efficacy for subsequent antigen capture. 4 SAW is not, however, an acoustic technology that can be readily applied to samples in the wells of a microtiter plate.
In contrast to other ultrasound modalities, Adaptive Focused Acoustics (AFA) allows precise, noncontact, isothermal delivery of acoustic energy to samples in microtiter plate formats. Typically, AFA instruments operate at frequencies in the 0.5 to 1 MHz range and therefore have acoustic wavelengths of a few millimeters. An appropriate design of the transducer surface enables these short acoustic waves to be focused into a narrow zone the length of the transducer. When this zone of tightly focused acoustic waves is applied to a microtiter plate, an entire column of the plate is simultaneously treated with a uniform acoustic dose. This feature enables the acoustic energy to be focused on samples in a noncontact, isothermal, highly efficient mode, thus avoiding contamination of samples and degradation by heat. With these unique design features, a much more precise and reproducible control can be obtained with AFA compared with probe and water bath sonicators. 5
The primary example of the precise control of acoustic energy provided by AFA to biomolecular applications is its widely employed use to precisely shear DNA to desired fragment lengths in an isothermal process for sequencing in next-generation sequencing platforms. 6 AFA has also been applied to RNA sequencing, 7 chromatin shearing, 8 cell lysis, 9 and preparation of monodisperse nano-suspensions of poorly soluble drugs. 10 For the most part, these applications use acoustic energy at the higher power end of the range provided by AFA instrumentation. At the lower power end of the acoustic energy range, AFA has been used to promote efficient mixing of solutions 11 and to remove bacteria adhered nonspecifically to microporous filters. 12
We therefore undertook a study to explore the potential of applying low power level AFA energy to improve the performance of microtiter plate ELISAs by (1) accelerating antigen/antibody binding with improved mixing of reaction solutions and (2) reducing NSB by AFA-mediated removal of nonspecifically bound entities. We demonstrate in this report that application of AFA to antigen/antibody binding reactions in commercially available microtiter plate ELISAs does indeed provide faster antibody/antigen binding kinetics and lower NSB than conventional passive ELISAs. These features, in concert with the excellent reproducibility and precision of AFA-mediated assays demonstrated here and in other applications of AFA, result in lower LODs and higher Z′ factors in AFA-mediated ELISAs versus conventional passive antigen/antibody binding procedures. Some of the findings presented here have been previously reported. 13
Materials and Methods
A microtiter plate sandwich ELISA kit (ABL, Bethesda, MD) that measures HIV-1 p24 antigen levels in tissue culture samples for research purposes only was primarily employed in this study. Kit components were first equilibrated to room temperature per the kit protocol. HIV-1 p24 antigen, supplied in the kit at 1 ng/mL, was serially diluted in polypropylene microfuge tubes into tissue culture medium containing 10% fetal bovine serum (FBS; ATCC, Manassas, VA). HIV-1 p24 dilutions (100 µL) and supplied cell disruption buffer (25 µL) were pipetted into the microwells of the 8-well columns. AFA was applied to samples, as described below, during the first assay step in which solution phase antigen binds to immobilized antibody. For passive binding performed per the kit protocol, samples were placed in a 37 °C incubator. Passive binding controls performed at other temperatures were either incubated at room temperature or placed in the water bath of the AFA instrument but not subjected to AFA treatment. AFA-mediated binding was determined at precisely controlled levels of acoustic energy and compared with passive binding. Standard curves were initially run, per the kit protocol, from 100 to 3.125 pg/mL of HIV-1 p24. After it was observed that linearity of binding was extended below 3.12 pg/mL when binding was mediated by AFA, standards were further diluted to lower concentrations. The tissue culture medium with 10% FBS was used as the zero control. Kinetic and dose-response studies were all performed at 50 pg/mL HIV-1 p24.
In the kinetic studies, samples that would provide the first time point in the assay (typically 5 min) were the last to be pipetted into microwells immediately prior to commencement of AFA or passive binding. At timed intervals thereafter, solution phase was removed from microwells. On completion of the antigen/antibody binding step, samples were processed according to the kit protocol for washing, conjugate binding, reaction with enzyme substrate, and photometric detection. Relative antigen/antibody binding rates were determined from the slopes of the data fit to a second-order polynomial and expressed as absorbance units per minute (AU/min).
A less extensive study than the one conducted with the HIV-1 p24 ELISA was performed with a microtiter plate sandwich ELISA kit (R&D Systems, Minneapolis, MN) that measures IGFBP-3 antigen levels in cell culture supernatants, serum, and plasma. As in the HIV-1 p24 assay, the first antigen/antibody binding step (the capture of IGFBP-3 by immobilized antibody) was studied. Kit components, other than the second antibody–horseradish peroxidase conjugate which was held at 4 °C until use, were equilibrated to room temperature per the kit protocol. IGFBP-3, at 50 ng/mL after reconstitution in the supplied calibrator diluent, was serially diluted into equal volumes of calibrator diluent. The required number of 8-well columns was detached from kit plates and placed in plate frames. First, 100 µL of the supplied assay diluent was added to each well. Then, 100-µL aliquots of standards or human plasma samples diluted 1:200 in calibrator diluent were added to the wells. For antigen recovery (spiking) experiments, 20 µL of 50 ng/mL standard in calibrator diluent was added to 80 µL of diluted plasma sample. Samples were processed for AFA treatment as described for the HIV-1 p24 assay. Passive binding was performed per the IGFBP-3 kit protocol for 2 h in a cold room at 4 °C. On completion of the antigen/antibody binding step, samples were processed according to the kit protocol for washing, conjugate binding, reaction with enzyme substrate, and photometric detection.
Application of AFA to Samples
Immediately following pipetting of samples into microtiter plate wells, the plate frame containing the 8-well columns was placed in the plate holder of an L8 or LE220 AFA instrument (Covaris, Woburn, MA). In a 96-well plate format, the instrument’s transducer focuses the acoustic energy into a line that is simultaneously applied to all wells of an 8-well column. By slight modulation of the frequency and/or raster scan of the focal zone, each well receives the identical amount of acoustic energy. Instrument settings allow an operator to set (1) the peak incident power (PIP), a measure of the amplitude of the acoustic waves emitted by the transducer (Watts); (2) duty factor (df), the percentage of time that the transducer is “on” versus “off”; (3) cycles per burst (cpb) number of ultrasound waves generated during the “on” time; and (4) duration in seconds. Temperature control is maintained by the circulation of cooling water from an external chiller. In the LE220 instrument (500 kHz), the water level is set to reach halfway up the microtiter plate wells. In the L8 instrument (850 kHz), water is applied only during the AFA process to the bottom of the plate. Temperature of samples during AFA treatment was monitored by insertion of a thermocouple wire into control wells.
Results
Faster antigen/antibody binding of AFA-mediated assays versus passive binding was consistently observed throughout this study. A typical example is shown in Figure 1 . A 1-s AFA treatment was applied in an L8 instrument every 22 s at 175 PIP, 5% df, and 200 cpb at 18.8 ± 0.7 °C. As evident from the slopes of the binding curves, the AFA-mediated antigen/antibody binding rate (139 × 10−4 AU/min) at 18.8 ± 0.7 °C was about twice that of the rate (70 × 10−4 AU/min) observed for passive binding at 37 °C.
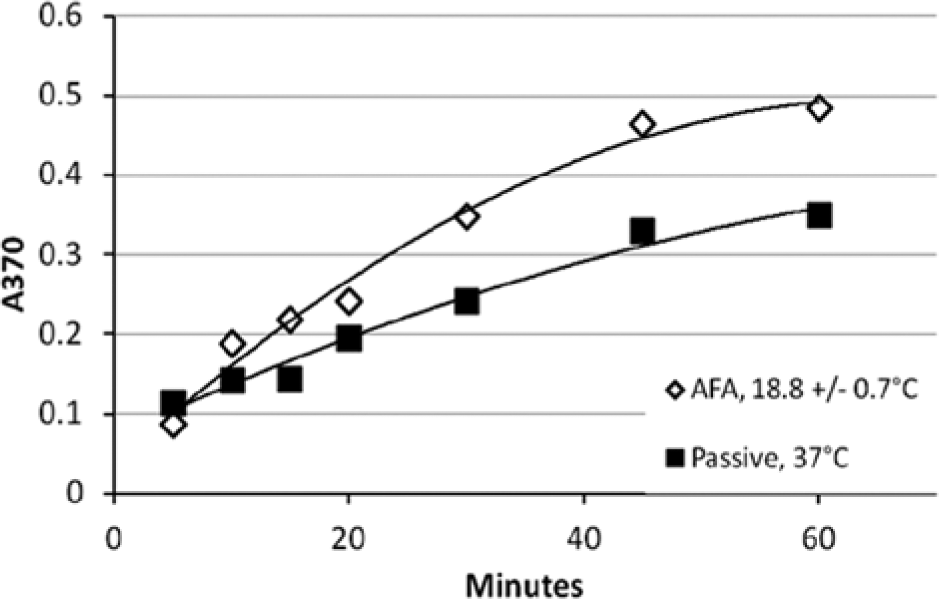
Kinetics of Adaptive Focused Acoustics (AFA)–mediated antigen/antibody binding in HIV-1 p24 enzyme-linked immunosorbent assay compared with passive binding, with typical results for single samples. AFA-mediated binding was performed at 18.8 ± 0.7 °C in an L8 AFA instrument. Samples were subjected to 1 s of AFA applied every 22 s at 175 peak incident power (PIP), 5% duty factor (df), and 200 cycles per burst (cpb). Passive binding was performed per the kit protocol at 37 °C. Data were fit to a second-order polynomial. AFA: slope = 0.0139x, y-intercept = 0.0138, R2 = 0.9844. Passive: slope = 0.0070x, y-intercept = 0.0705, R2 = 0.9782.
Figure 2 shows examples of antigen/antibody binding rates at different levels of AFA compared with passive binding. For example, 1-s pulses of AFA applied every 22 s at 175 PIP, 5% df, 200 cpb in the 18 to 20 °C range (column D) resulted in an approximate 2-fold increase in the rate of antigen/antibody binding compared with conventional passive binding at 37 °C (column B). When the temperature of an AFA-mediated assay was lowered to the 8 to 10 °C range, the resultant HIV-1 p24 antigen/antibody binding kinetics was equivalent or superior to passive binding at 37 °C, as shown in Figure 2 (columns B and C).
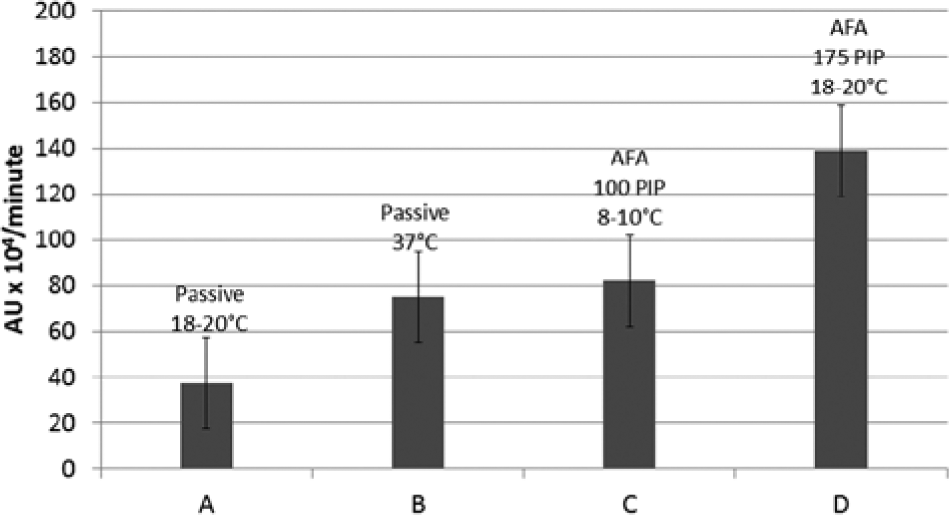
Comparative rates of antigen/antibody binding in HIV-1 p24 enzyme-linked immunosorbent assay. Relative rates of antigen/antibody binding were determined from slopes of binding curves (n ≥ 3) of absorbance values plotted as function of time, as shown in Figure 1 . Column A: passive binding, 20 °C. Column B: passive binding, 37 °C. Column C: Adaptive Focused Acoustics (AFA)–mediated binding, 100 peak incident power (PIP), 5% duty factor (df), 50 cycles per burst (cpb), 1-s pulse every 22 s, LE220, 18 to 20 °C, high-throughput screening (HTS) mode. Column D: AFA-mediated binding, 175 PIP, 5% df, 200 cpb, 1-s pulse every 22 s, L8, 18 to 20 °C, HTS mode.
Comparable or superior binding at low temperature in an AFA-mediated assay compared with passive binding at 37 °C was consistently observed throughout this study. This feature is evident in the data shown in Figure 3 , which shows a typical result for a side-by-side comparison of the HIV-1 p24 ELISA for an AFA-mediated HIV-1 p24 antigen/antibody binding step performed at 9.1 ± 0.1 °C versus passive binding performed at 37 °C. Standard curves with high R2 values were consistently observed with an AFA-mediated antigen/antibody binding step, as shown in Figure 3 , even when only single samples were assayed.
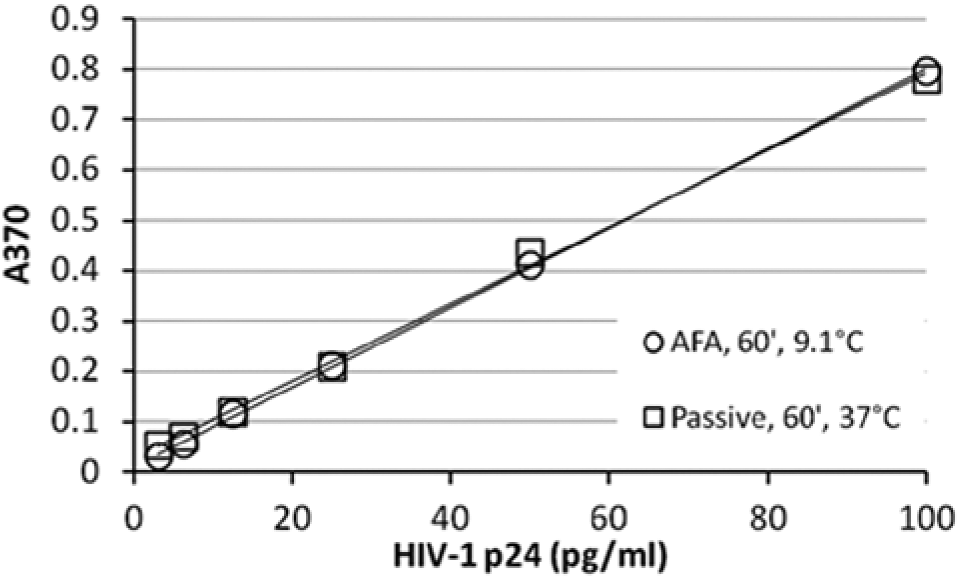
Adaptive Focused Acoustics (AFA)–mediated binding antigen/antibody binding in HIV-1 p24 enzyme-linked immunosorbent assay compared with passive binding, with typical results for single samples. AFA-mediated binding was performed at 9.1 ± 0.1 °C in an LE220 AFA instrument. Samples were subjected to 2 s of AFA every 8 s for 1 h at 150 peak incident power (PIP), 5% duty factor (df), 50 cycles per burst (cpb). Passive binding was performed per the kit protocol for 1 h at 37 °C. Linear data fit. Passive binding: y-intercept = 0.0276, R2 = 0.9975. AFA-mediated binding: y-intercept = 0.0079, R2 = 0.9997.
Throughout this study, NSB levels observed in AFA-mediated antigen/antibody binding assays were consistently lower than those observed in assays run in parallel with conventional passive binding. This feature is evident in the y-intercepts (0.0276 for the passive assay and 0.0079 for the AFA-mediated assay) of the standard curves noted in Figure 3 . Absorbance reading of samples run with zero level of analyte empirically confirmed the lower NSB levels at zero analyte concentration for AFA-mediated samples calculated by the curve-fitting algorithm. Overall, a trend was determined in this study for better signal-to-noise (S/N) ratios for assays performed at lower temperatures. Figure 4 shows a typical result for signal-to-background (S/B) ratios measured in single samples in an AFA-mediated assay versus passive binding assays performed in parallel. As shown by the data, the S/B for the AFA-mediated sample (column A) is significantly superior to the passive assays (columns B–D).
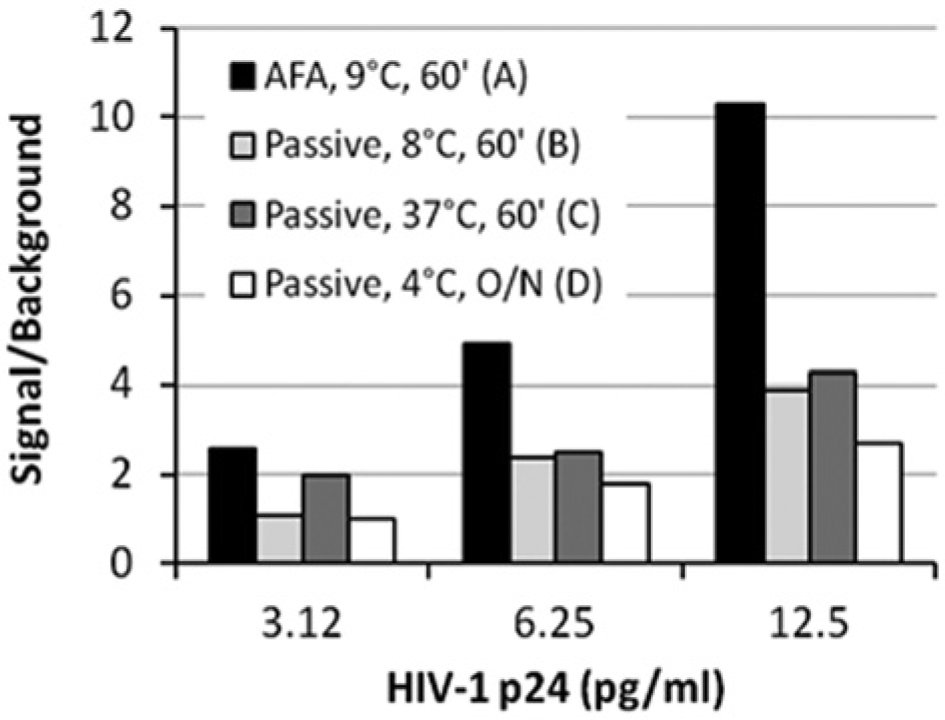
Effect of Adaptive Focused Acoustics (AFA) on signal-to-background ratios of HIV-1 p24 enzyme-linked immunosorbent assay, with typical result for single samples. Column A: AFA-mediated binding at 9.1 ± 0.1 °C in an LE220 AFA instrument. Samples were subjected to 2 s of AFA every 8 s for 1 h at 150 peak incident power (PIP), 5% duty factor (df), 50 cycles per burst (cpb). Column B: passive binding, 60 min, 8 °C. Column C: passive binding, 60 min, 37 °C. Column D: passive binding, overnight, 4 °C.
Results from the less extensive study with the IGFBP-3 ELISA mirrored those with the HIV-1 p24 ELISA. AFA-mediated antigen/antibody binding for 1 h at 11.4 ± 0.2 °C was comparable to the 2-h passive binding incubation at 4 to 8 °C stipulated in the kit protocol. NSB levels—as measured by optical absorbance measurements of zero calibrator samples—in AFA-mediated IGFBP-3 assays were also consistently lower than samples run in parallel with conventional passive binding.
Tight control of sample temperature in AFA-mediated assays was demonstrated throughout this study, as evident from the temperature ranges of control wells measured with a thermocouple noted in figures. Other important performance parameters observed in this study were intra-assay coefficients of variation (CVs) ≤4.6% (n = 12), determined by variations in optical absorbance measurements of triplicate samples; interassay CVs ≤6.4% (n = 28), determined by variations in optical absorbance measured as a function of joules of AFA energy (joules = Watts(PIP) × duty factor × seconds of AFA); a mean CV of 8.8% (n = 12) for determination of control samples spiked with HIV-1 p24; a mean CV of 13.9% (n = 7) in relative rates of binding of HIV-1 p24 to capture antibody; and a mean recovery of 106.5% with a mean CV of 5.8% for HIV-1 p24 spiked into cell culture medium or lysed K562 cells (n = 3) and IGFBP-3 (n = 5) spiked into human plasma.
The lower NSB and CVs of AFA-mediated assays versus passive assays allowed the linearity of the assay to be extended to lower levels. The passive assay, performed according to the kit manufacturer’s protocol, was linear from 3.125 to 100 pg/mL, as stated by the kit manufacturer. In contrast, the AFA-mediated assay was linear from 0.781 to 100 pg/mL.
A lower limit of detection (LOD) was observed in AFA-mediated assays versus passive assays, where LOD is the mean of 10 measurements at 0 pg/mL + 3 standard deviations. A typical result is shown in Figure 5 , where the LOD of the AFA-mediated assays was just below 1 pg/mL compared with ~5 pg/mL for passive assays.
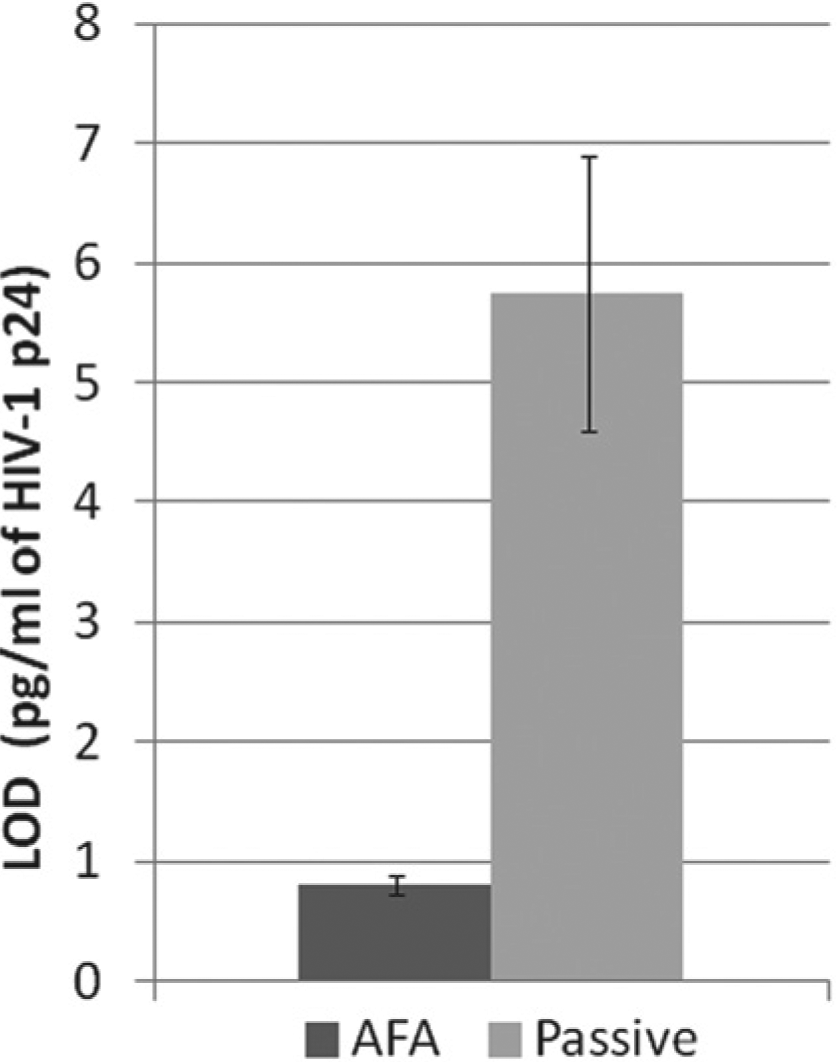
Effect of Adaptive Focused Acoustics (AFA) on limit of detection (LOD) of HIV-1 p24 enzyme-linked immunosorbent assay. Samples were subjected to either 2 s of AFA applied in an LE220 instrument every 8 s for 1 h at 150 peak incident power (PIP), 5% duty factor (df), 50 cycles per burst (cpb) at 9.1 ± 0.4 °C (black column) or to passive binding at 37 °C for 1 h, per the kit protocol (gray column). Ten replicates of samples containing 0 pg/mL HIV-1 p24 were assayed. LOD = mean of 10 replicate samples + 3 standard deviations.
Higher Z′ factors were observed in AFA-mediated assays than in passive assays.
Figure 6 shows the Z′ for assays performed in triplicate over a range of HIV-1 p24 concentrations. Z′ in excess of 0.5 (the target for an assay to have utility in HTS applications) was met by the AFA-mediated assay down to concentrations of 6.25 pg/mL HIV-1 p24. In contrast, the cutoff for the passive assay at 37 °C was 25 pg/mL HIV-1 p24. An improvement in the Z′ of the passive assay was achieved by running the assay at a lower (17.2 °C) temperature. Doing so extended the HTS utility of the passive assay to 12.5 pg/mL HIV-1 p24.
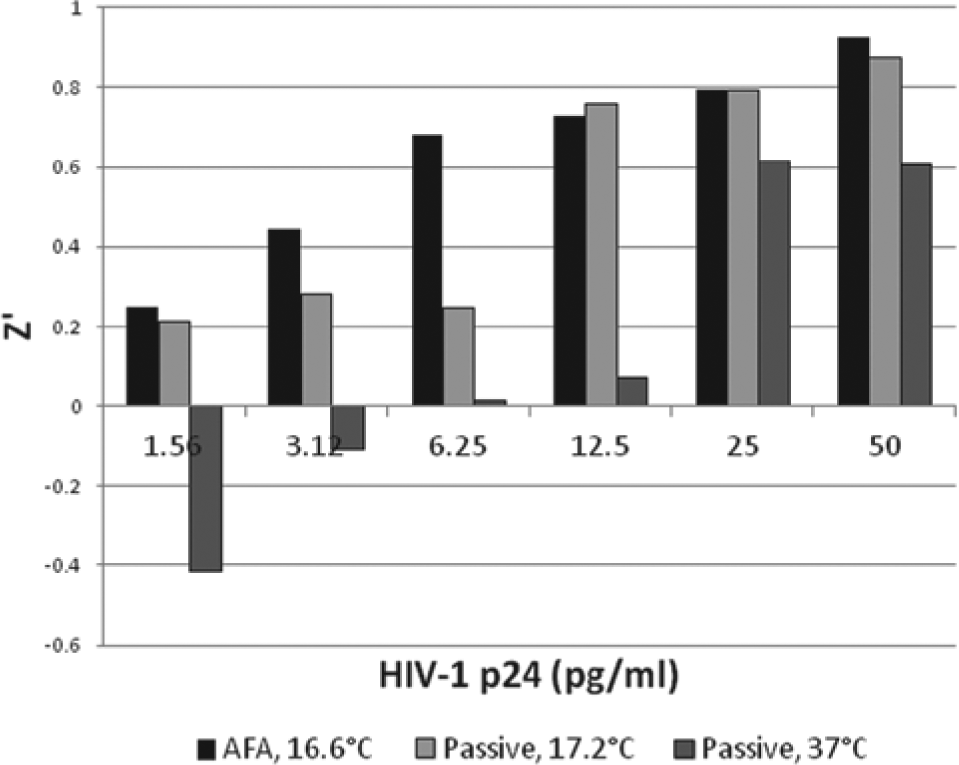
Effect of Adaptive Focused Acoustics (AFA) on Z′ of HIV-1 p24 enzyme-linked immunosorbent assay. Samples were run in replicates (n = 4). Samples were subjected to (1) 1-s pulses of AFA applied in an L8 instrument every 22 s for 1 h at 105 peak incident power (PIP), 5% duty factor (df), 100 cycles per burst (cpb) at 16.6 ± 0.3 °C (black column); (2) passive binding for 1 h at 17.2 ± 0.3 °C (light gray column); or (3) passive binding for 1 h at 37 °C (gray column).
Discussion
As mentioned in the Introduction, the use of ultrasound to enhance antigen/antibody binding rates 3 and to remove nonspecifically bound proteins on microarrays, leaving selected antigen receptor interactions bound with retention of their efficacy for subsequent antigen capture, 4 has been previously reported by others. In the former study, 3 ultrasound was applied to samples in test tubes in an ultrasonic water bath, whereas in the latter study, 4 ultrasound was applied to microarrays by SAW.
The study we report here confirms both (a) ultrasound acceleration of antigen/antibody binding rates and (b) removal of nonspecifically bound proteins from a solid-phase assay. In addition, this study demonstrates that ultrasound applied in the form of AFA provides other important improvements to the performance of microplate double-antibody sandwich ELISAs compared with assays performed with conventional passive binding—namely, lower NSB, excellent intra- and interassay reproducibility, lower LODs, and higher Z′ factors. To our knowledge, this report is the first to demonstrate these multiple benefits of applying ultrasonic energy—in the form of AFA—to microplate ELISAs.
Use of AFA for acoustic enhancement of binding assays eliminates problems experienced with other modalities of acoustic energy that have been tried for this purpose. Low-frequency acoustics, used for example in water bath sonicators, results in collateral thermal energy transfer, which may account for acceleration of binding kinetics observed with unfocused acoustics. The long wavelength of sonicators also results in variable, nonuniform energy zones. The unique features of the AFA process enable the desired mechanical effects to occur under isothermal operating parameters. For example, low-duty-factor, high-efficiency AFA doses require 0.8 W to obtain the desired pressure effects versus 140 W required for a bath sonicator to obtain the same pressure levels (e.g., 1 MPa in water). Another unique feature of AFA is its ability to repeatedly deliver a small, high-density energy zone. This is a direct consequence of the highly efficient switching technology and H-bridge of the electronic circuit (e.g., similar to a Class H amplifier) to drive specialized transducers as designed by Covaris and used in the Covaris AFA apparatus.
The finding in this study that AFA-mediated assays exhibited lower background binding has important significance to the goal of achieving better accuracy in the measurement of low levels of analyte. Moreover, the trend observed in this study for signal-to-noise ratios to be improved at lower temperatures further illustrates the potential advantage of using AFA-mediated antigen/antibody binding reactions in microplate ELISAs as AFA enhancement of antigen/antibody binding occurs equally well at low temperatures. We attribute the lower NSB observed in this study to removal by AFA of antigen that nonspecifically adheres in the assay and is not removed by conventional washing procedures.
Substitution of an AFA-mediated antigen/antibody binding in place of a passive binding step in commercially available microtiter plate ELISAs can be readily performed using AFA instrumentation that is in widespread use for precise isothermal control of focused acoustics in a number of other biotechnological applications,5–11 foremost of which is precision shearing of DNA to desired fragment lengths for next-generation sequencing platforms. 6 For the most part, these applications of AFA are performed at the high-power acoustic energy levels provided by the instrumentation. In contrast, the study reported here employed lower power acoustic energy levels that have been demonstrated to not adversely affect the structural integrity and function of proteins. 14
The findings in this study demonstrate that higher Z′ factors are a benefit that AFA-mediated assays can potentially bring to the performance of ELISAs in HTS assays. Also of significance to HTS assays is the finding that AFA applied for a short period (e.g., for 1 s) consecutively to each column of a microtiter plate allows a whole plate to be simultaneously processed in an automated format. It is important to note that the commercially available ELISAs used in this study were 96-well plates. However, higher density microtiter plates (e.g., 384- and 1536-well plates) can also be used in the AFA instrumentation. For example, in a 384-well plate, 16 wells are simultaneously processed, with each well receiving the same precise amount of acoustic energy.
AFA-mediated binding assays also have potential applications for in vitro clinical diagnostic tests run in a STAT mode rather than the whole-plate sweeping mode described in this report. In the STAT mode, AFA is applied to a single column of a microtiter plate containing sample, calibrators, and controls. The faster rate of AFA application results in even faster rates of antigen/antibody binding than is achieved in the sweeping mode. 13
It is important to note that the AFA instruments used in this study were developed, as mentioned above, for applications such as precision shearing of DNA to desired fragment lengths for next-generation sequencing platforms. 6 For the most part, these applications require higher power levels of acoustic energy than those used in the study reported here. Water baths with external chillers are therefore used to maintain temperature control of samples during application of high-energy AFA. However, the lower power level of acoustic energy required for the acceleration of antigen/antibody binding kinetics allows the AFA apparatus to be configured to enable the desired effect to be accomplished without an open water bath, for example, in a dry format. This feature will likely be of significant importance in applying the findings reported here to the application of AFA to the enhancement of antigen/antibody binding for in vitro diagnostic microtiter plate ELISAs in both HTS and STAT assay formats.
Furthermore, application of AFA to binding assays has the potential to significantly improve the performance of not only microtiter plate antigen/antibody sandwich ELISAs, as demonstrated here, but also other types of binding assays. For example, in preliminary studies, we have demonstrated AFA-mediated improvement of the binding of antibodies to a protein A/G microtiter plate. This finding indicates the potential to use AFA to accelerate the immunoprecipitation step in chromatin immunoprecipitation assays. We have also demonstrated in a preliminary study that AFA can be used to accelerate antibody coating of microtiter plates. Other ways that AFA may have advantageous application in binding assays include acceleration of the binding of hapten-labeled conjugate with antigens in sandwich ELISAs, in enzymatic reactions (e.g., tryptic digests), and in DNA hybridization assays, as well as acceleration of washing steps with concurrent lower NSB. The lower NSB observed in AFA assays in this study may conceivably help enhance image analysis of cell-based assays, thereby improving some high-content screening procedures.
Footnotes
Acknowledgements
The authors gratefully acknowledge helpful discussions with Adrian Palmer, Gary Smejkal, Anna Krowczynska, Hamid Khoja, J-D Herlihy, and Bill Skea.
Declaration of Conflicting Interests
D. J. Green is a consultant, and E. A. Rudd and J. A. Laugharn are employees of Covaris.
Funding
D. J. Green is a consultant, and E. A. Rudd and J. A. Laugharn are employees of Covaris.
