Abstract
Ghrelin, a gastric hormone, is a growth hormone-releasing peptide. Its serine-3 acylation with octanoic acid is essential for its orexigenic activity, and therefore, inhibition of the acylation of ghrelin may help in decreasing appetite and preventing obesity. This study aimed to establish a human gastric cell-based assay system to evaluate candidate inhibitors of octanoylated ghrelin production. In human gastric carcinoma AGS cells, obligatory factors for the posttranslational modification of ghrelin, such as certain prohormone convertases responsible for processing of proghrelin to the mature ghrelin and the enzyme-catalyzing acyl-modification of ghrelin, were well expressed, but ghrelin was expressed at low levels. Accordingly, we transfected a ghrelin-expressing vector into AGS cells and isolated a stable ghrelin-expressing cell line (AGS-GHRL8). AGS-GHRL8 cells secreted octanoylated ghrelin in accordance with the concentrations of octanoic acid in the culture medium. Given that ingested heptanoic acid is used for the acyl-modification of ghrelin, we evaluated whether heptanoic acid inhibits production of octanoylated ghrelin in AGS-GHRL8 cells. Butyric acid was used as a control. Indeed, heptanoic acid predictably decreased the secretion of octanoylated ghrelin, whereas butyric acid did not. The AGS-GHRL8 line established in this study will facilitate the screening of inhibitors of octanoylated ghrelin production.
Introduction
Ghrelin, a 28-amino acid peptide that was first purified and identified from rat stomach, is the ligand for the growth hormone secretagogue receptor (GHSR). 1 Ghrelin is predominantly produced in endocrine cells in the gastric mucosa, called X/A-like or ghrelin cells, from where it is secreted into the plasma. In the pituitary gland, ghrelin stimulates growth hormone release and regulates food intake and energy metabolism.1–4
The preproghrelin protein is 117 amino acids in length; from this peptide, the signal peptide is cleaved to form the 94-amino acid proghrelin peptide. Prohormone convertase (PC) 1/3, PC2, or furin, members of the prohormone convertase family, are responsible for this protease processing of proghrelin to the 28-amino acid mature ghrelin peptide within the endocrine cells of the stomach.5,6 Moreover, ghrelin contains an octanoyl modification at the serine 3 residue; this lipid modification is essential for some biological activities of ghrelin. 7 Octanoylated ghrelin can bind to the GHSR and stimulate the secretion of growth hormone. Furthermore, octanoylated ghrelin increases food intake.8–10 Unlike octanoylated ghrelin, des-acyl ghrelin neither binds to GHSR nor stimulates the secretion of growth hormone. It has been reported that des-acyl ghrelin has the ability as a functional antagonist of octanoylated ghrelin. 11 Moreover, it has also been reported that des-acyl ghrelin has such actions as trophic and protective effects on β-cells, decreasing fat mass, increasing muscle regeneration, and enhancing insulin sensitivity. 11 With respect to its effect on food intake, there are two points of view: upon peripheral administration, des-acyl ghrelin decreases food intake or does not affect feeding. 12
Ingested medium-chain fatty acids are directly used for the acylation of ghrelin. 13 Therefore, inhibition of ghrelin octanoylation may suppress hyperphagia and thus contribute to the prevention and improvement of obesity. The enzyme that catalyzes acyl-modification of ghrelin is ghrelin O-acyltransferase (GOAT),14,15 also known as membrane-bound O-acyltransferase 4. It has also been reported that a few synthetic peptides can inhibit GOAT activity16,17; these peptides may therefore be useful for preventing the production of octanoylated ghrelin. However, considering that peptidic compounds generally have poor oral bioavailability, nonpeptidic inhibitors of octanoylated ghrelin production for the prevention of hyperphagia and obesity are currently being explored.
To investigate the inhibitors of ghrelin activation, an in vitro system for the evaluation of octanoylated ghrelin production would be useful. A number of assay systems to determine ghrelin levels and GOAT activity have been developed. 18 As a cell-based assay system, Sf9 insect cells infected with a recombinant baculovirus encoding mouse GOAT and GOAT/proghrelin-transfected HeLa cells have been used for the evaluation of peptidic inhibitors of ghrelin-octanoylation.16,17 Moreover, systems using gastric cells are considered useful for the evaluation of octanoylated ghrelin production because ghrelin is principally produced in the gastric cells and GOAT is largely restricted to the stomach.14,15 However, to date, human gastric cells have not yet been used for the evaluation of candidate inhibitors of the production of octanoylated ghrelin.
In the present study, we established a stable transfectant cell line that constitutively produces ghrelin, by transfecting ghrelin complementary DNA (cDNA) into AGS cells that originated from human gastric carcinoma tissue; we then investigated whether ghrelin production in these cells is influenced by exogenous fatty acids.
Materials and Methods
Cell Culture
Dulbecco’s modified Eagle’s medium (DMEM) was purchased from Invitrogen (Carlsbad, CA). Human gastric carcinoma AGS cells, obtained from American Type Culture Collection (Manassas, VA), were cultured in DMEM containing 10% fetal bovine serum and 100 U/mL penicillin (Invitrogen) at 37 °C in a 5% CO2 humidified atmosphere.
Reverse Transcriptase Polymerase Chain Reaction Analysis
Total RNA was extracted from AGS cells using TRIzol (Invitrogen). Synthesis of first-strand cDNA was performed using a SuperScript III First-Strand Synthesis System (Invitrogen). Primers used for polymerase chain reaction (PCR) were as follows: human ghrelin, sense: 5′-gagccctgaacaccagagag-3′, antisense: 5′-cccagaggatgtcctgaaga-3′; human GOAT, sense: 5′-acagctcgatggctccgacg-3′, antisense: 5′-agcttccaccatcacggccc-3′; human PC1/3, sense: 5′-tagcccagaaggcttttgaa-3′, antisense: 5′-ggagcacttctcagcgtacc-3′; human PC2, sense: 5′-tggtttttgcatctgctgag-3′, antisense: 5′-ggtgtaggctgcgtcttctc-3′; human furin, sense: 5′-acaactatgggacgctgacc-3′, antisense: 5′-tggacacagctcttctggtg-3′; and human glyceraldehyde-3-phosphate dehydrogenase (GAPDH), sense: 5′-gtgtgaaccatgagaagtatg-3′, antisense: 5′-tttggcaggtttttctagacg-3′. PCR was performed using AmpliTaq Gold 360 Master Mix (Applied Biosystems, Foster City, CA). The reaction solutions were prepared in a final volume of 50 µL, containing 1 µL first-strand cDNA, 0.5 µmol/L sense and antisense primers, and 25 µL Amplitaq Gold 360 Master Mix. PCR involved the following steps: an initial denaturation step of 10 min at 95 °C, which was followed by 35 cycles of denaturation for 30 s at 95 °C, annealing for 30 s at 60 °C, and extension for 1 min at 72 °C; this was followed by a final 7-min extension step at 72 °C.
Ghrelin-Expressing Vector Construction
The full sequence of human ghrelin, amplified from AGS cells by PCR, was subsequently inserted into the pcDNA3 vector (Invitrogen). The following primers were used for the PCR reactions: sense, 5′-agctgaggccatgccctccccagggaccg-3′; antisense, 5′-tcacttgtcggctggggcctc-3′.
Isolation of the Stable Cell Line Expressing Ghrelin
AGS cells were seeded into a 100-mm dish. When cells reached 80% confluence, the human ghrelin-expressing vector was transfected into AGS cells using Lipofectamine 2000 (Invitrogen). After 24 h, the medium was changed, and G418 (Wako, Osaka, Japan) was added to the culture medium to a final concentration of 800 µg/mL. Thereafter, cells were cultured in the presence of G418 for 2 weeks. Medium was exchanged every 3 days. Some cells were isolated from the colonies formed in the culture and were seeded into other dishes to establish a stable cell line expressing ghrelin. The expression of ghrelin in each cell line was determined by immunoblot analysis.
Immunoblot Analysis
Complete ethylenediaminetetraacetic acid (EDTA)-free protease inhibitor cocktail was purchased from Roche Diagnostics (Indianapolis, IN). Sodium dodecyl sulfate (SDS) and antibody against β-actin were purchased from Sigma (St. Louis, MO). The recombinant ghrelin protein with His tag (ab89233) was purchased from Abcam (Cambridge, MA). The antibody against ghrelin (clone 1ML-1D7) was obtained from Millipore (Temecula, CA), and horseradish peroxidase (HRP)-conjugated goat anti-mouse antibody was from Nacalai Tesque (Kyoto, Japan). All other reagents for immunoblot analysis were purchased from Wako.
Whole-cell lysates were prepared by lysing AGS cells and ghrelin-expressing AGS cells with detergent buffer (10 mmol/L Tris-HCl [pH 7.5], 5 mmol/L EDTA, 150 mmol/L sodium chloride, 1% [v/v] Triton X-100, 10% glycerol, 1× complete EDTA-free protease inhibitor cocktail, and 1 mmol/L benzylsulfonyl fluoride). Insoluble fractions were removed by centrifugation at 16,000 g for 10 min at 4 °C. Whole-cell lysates were then boiled in a quarter volume of sample buffer (125 mmol/L Tris-HCl [pH 7.5], 25% glycerol, 5% SDS, 0.2% bromophenol blue, and 25% 2-mercaptoethanol). The recombinant ghrelin protein with His tag was used as a standard sample. Proteins in these samples were separated by SDS-polyacrylamide gel electrophoresis (15%) and transferred to a polyvinylidene difluoride membrane (Bio-Rad Laboratories, Hercules, CA). The membrane was incubated for 1 h in Tris-buffered saline (TBS) containing 5% nonfat milk as blocking buffer and then treated overnight with primary antibodies in blocking buffer at 4 °C. The membrane was washed in TBS and then incubated with 1:3000 HRP-conjugated goat anti-mouse IgG antibody in blocking buffer at room temperature for 1 h. The incubations with primary and secondary antibodies were performed on the wave shaker at 10 rpm. It was then washed again in TBS. Antibody binding was visualized using the ECL Plus Western Blotting Detection System (GE Healthcare Bio-Sciences, Buckingham, UK).
Enzyme-Linked Immunosorbent Assay for Octanoylated Ghrelin and Des-acyl Ghrelin
Octanoic acid, heptanoic acid, and butyric acid were purchased from Wako. Cells were seeded into a 12-well plate at a density of 3 × 105 cells/well and cultured for 24 h in DMEM. To examine the effects of exogenous fatty acids on octanoylated ghrelin secretion, culture medium was changed to fresh medium, and each fatty acid was added to the medium. The cells were cultured for 24 h. Octanoic acid up to a concentration of 100 µmol/L was tested, and butyric acid at a concentration of 100 µmol/L and heptanoic acid up to a concentration of 100 µmol/L, respectively, were used with or without octanoic acid. The medium was collected into microtubes and immediately treated with 1/10 volume of 1 mol/L HCl and stored at −80 °C until analysis to maintain the stability of octanoylated ghrelin and des-acyl ghrelin. The levels of octanoylated ghrelin and des-acyl ghrelin in the samples were determined using an Active Ghrelin enzyme-linked immunosorbent assay (ELISA) kit and Desacyl-Ghrelin ELISA kit, respectively (Sceti, Tokyo, Japan), according to the manufacturer’s instructions.
Cell Viability Assay Using 3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide
For this study, 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetra-zolium bromide (MTT) was purchased from Wako. An MTT colorimetric assay was used to determine the relative sensitivity of cell lines to test compounds. Cells were seeded into 96-well plates at 5 × 103 cells/well and cultured for 24 h in DMEM. Cells were treated with octanoic acid, heptanoic acid, and butyric acid at a concentration of 100 µmol/L, respectively. After 72 h of culture in the presence of these substances, 50 µL MTT (1 mg/mL in phosphate-buffered saline) was added to each well, and the cells were cultured for an additional 4 h. The resultant formazan was dissolved with 100 µL DMSO after aspiration of the culture medium. Optical density was read at 595 nm, as previously described. 19
Statistical Analysis
Differences between groups were tested by one-way analysis of variance followed by a Tukey test for multiple comparisons. Data are presented as mean ± standard deviation (SD). Differences were considered significant at p < 0.05.
Results
Characteristics of AGS Cells
It is known that proper processing from proghrelin to octanoylated ghrelin in cultured cells requires GOAT and prohormone convertases, such as PC1/3, PC2, and furin. 5 To assess the characteristics of AGS cells, we first determined messenger RNA (mRNA) expression of ghrelin, GOAT, PC1/3, PC2, and furin in AGS cells by reverse transcriptase PCR (RT-PCR). As shown in Figure 1A , GOAT and furin mRNAs were indeed expressed in AGS cells. Ghrelin mRNA was also expressed, but the level was very low compared with those of GOAT and furin ( Fig. 1A ). Next, we evaluated the ability of AGS cells to secrete octanoylated ghrelin and/or des-acyl ghrelin using ELISA. No octanoylated ghrelin and only a minimal amount of des-acyl ghrelin was detected in the culture medium of AGS cells (data not shown). We also performed immunoblot analysis to examine whether ghrelin peptide expresses in AGS cells. As shown in Figure 1B , ghrelin peptide could not be detected by immunoblot analysis. Both bands in lanes of standard samples appear to be ghrelin, of which the upper is probably the His-tagged version, and the lower is the protein without His tag. These results suggested that the expression level of ghrelin was very low in AGS cells. Therefore, we attempted to generate a stable cell line that constitutively expresses ghrelin using AGS cells.
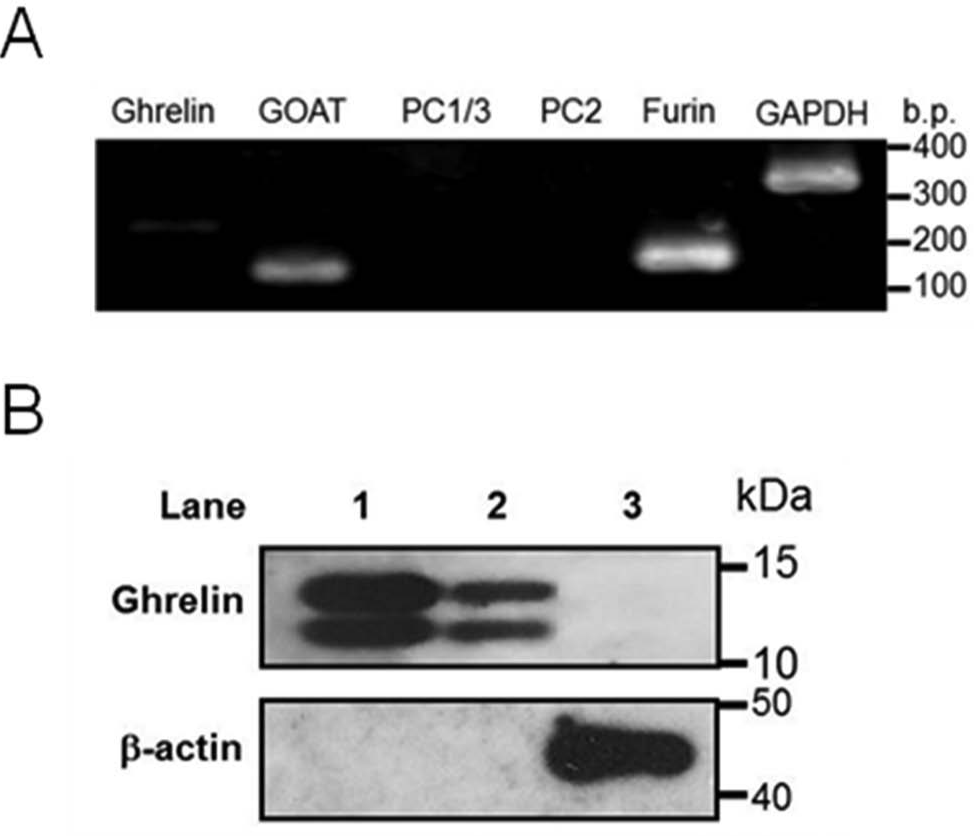
Expression of ghrelin, GOAT, PC1/3, PC2, and furin in AGS cells. (
Establishment of a Ghrelin-Expressing Stable Cell Line from AGS Cells
For generation of a ghrelin expression construct, ghrelin cDNA from AGS cells, encoding 117 amino acids of preproghrelin, was amplified by RT-PCR. The sequence of amplified ghrelin cDNA was confirmed by concordance with that of wild-type human preproghrelin by sequence analysis (data not shown). Amplified ghrelin cDNA was inserted into pcDNA3, an expression vector with neomycin (G418) resistance and cytomegalovirus (CMV) promoter genes. When this ghrelin expression construct was transfected into AGS cells, ghrelin expression was detected by immunoblot analysis ( Fig. 2A ). Next, to screen for a ghrelin-expressing stable cell line in culture using G418, cells were cloned from 10 colonies, and the expression of ghrelin in each line was determined by immunoblot analysis. Ghrelin was detected in 7 of 10 cloned cell lines ( Fig. 2B ). Of these, we selected the line that expressed the highest level of ghrelin and named it AGS-GHRL8. Ghrelin, GOAT, and furin mRNAs were expressed in AGS-GHRL8 cells ( Fig. 2C ). We investigated the ghrelin expression level of AGS-GHRL8 cells with a different passage number. There was no major change between passages 4 and 20 ( Fig. 2D ).
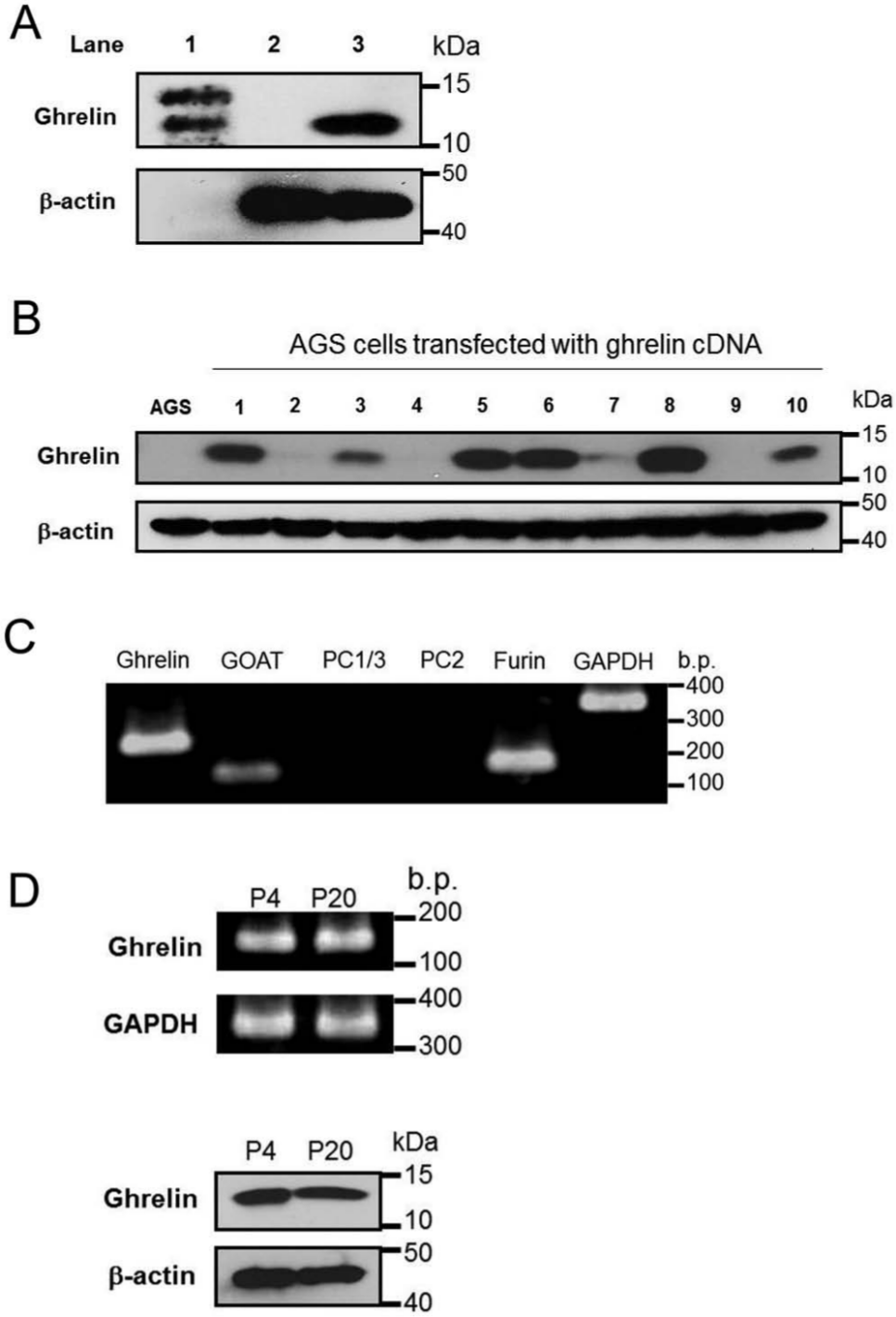
Establishment of a ghrelin-expressing stable cell line by transfection of AGS cells with a ghrelin-expressing vector. (
Secretion of Des-acyl Ghrelin and Octanoylated Ghrelin from AGS-GHRL8
First, we assessed the ability of AGS-GHRL8 to secrete des-acyl ghrelin into the culture medium. After 24-h cultivation in a fresh medium, the levels of des-acyl ghrelin in the AGS-GHRL8 medium were much more abundant than those of AGS cells ( Fig. 3A ). Next, we investigated whether octanoylated ghrelin was secreted from AGS-GHRL8 cells in the presence of octanoic acid in culture medium. The levels of secreted octanoylated ghrelin were measured after 24 h of culture in fresh medium containing octanoic acid at concentrations up to 100 µmol/L. The levels of octanoylated ghrelin in the medium increased with the concentration of octanoic acid ( Fig. 3B ). The concentrations of des-acyl ghrelin were maintained at much higher levels than those of octanoylated ghrelin ( Fig. 3B ).
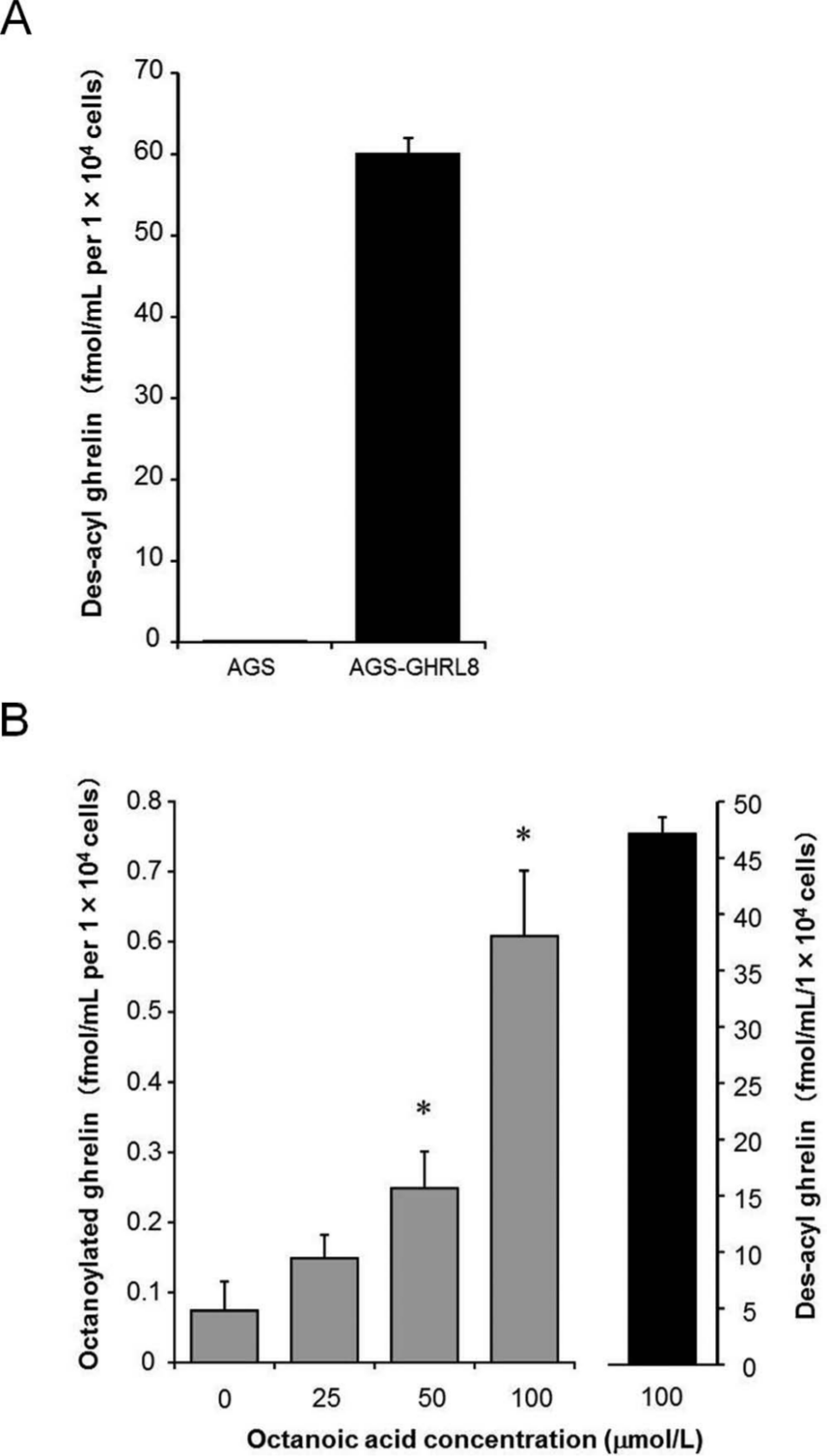
Secretion of des-acyl ghrelin and octanoylated ghrelin from AGS-GHRL8 cells into the culture medium. (
Effects of Heptanoic Acid and Butyric Acid on the Octanoylated Ghrelin Secretion
It has been reported that heptanoyl ghrelin, but not butyryl ghrelin, could be detected in mouse stomach after ingestion of the corresponding fatty acids. 13 These findings suggest that heptanoic acid, but not butyric acid, may suppress production of octanoylated ghrelin by competing with octanoic acid for GOAT-mediated acylation. Therefore, we selected heptanoic acid and butyric acid as test agents and investigated the effects of both fatty acids on octanoylated ghrelin production using AGS-GHRL8. The levels of octanoylated ghrelin after 24-h cultivation in a fresh medium containing octanoic acid and/or each of the test fatty acids were measured. The levels of octanoylated ghrelin in the presence of 100 µmol/L octanoic acid alone were significantly increased compared with the control (0.79 ± 0.13 vs. 0.14 ± 0.09 fmol/mL per 1 × 104 cells; Fig. 4 ). The increase in octanoylated ghrelin levels was dose-dependently suppressed by combined treatment with heptanoic acid, and 100 µmol/L of heptanoic acid completely suppressed the secretion of octanoylated ghrelin (0.14 ± 0.02 fmol/mL per 1 × 104 cells; Fig. 4 ). In contrast, butyric acid did not suppress the secretion of octanoylated ghrelin at a concentration of 100 µmol/L (0.74 ± 0.08 fmol/mL per 1 × 104 cells; Fig. 4 ). The concentrations of des-acyl ghrelin were maintained at much higher levels than those of octanoylated ghrelin in all the treatment groups, and the viability of AGS-GHRL8 cells was not significantly affected by any of treatment groups (data not shown).
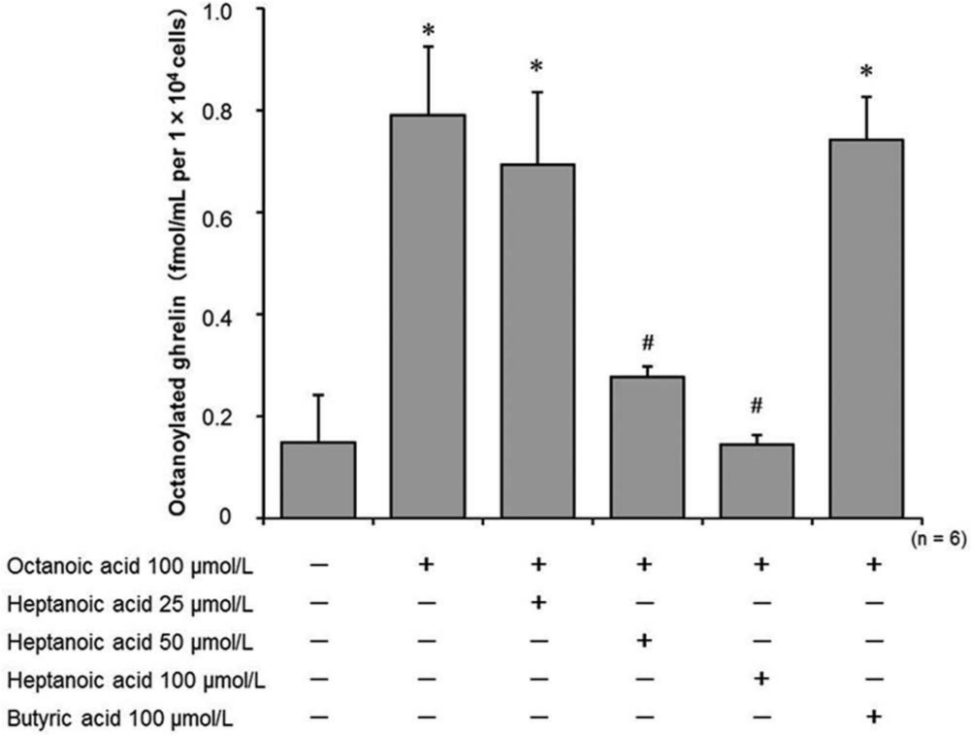
Effects of heptanoic acid and butyric acid on the secretion of octanoylated ghrelin from AGS-GHRL8 cells treated with octanoic acid. Secretion levels of octanoylated ghrelin in AGS-GHRL8 cells treated with octanoic acid and/or heptanoic acid or butyric acid were measured by enzyme-linked immunosorbent assay. The data are expressed as mean ± SD of six independent experiments. *p < 0.01 vs. treatment without any fatty acid. #p < 0.01 vs. treatment with 100 µmol/L octanoic acid alone.
Discussion
In the present study, we aimed to establish a human gastric cell line in which candidate inhibitors of ghrelin octanoylation could be tested. We first investigated whether the human gastric carcinoma cell line AGS itself is useful for exploring inhibitors of octanoylated ghrelin production. RT-PCR analyses revealed that both GOAT and furin mRNAs were expressed well in AGS cells. GOAT and furin can process proghrelin to octanoylated ghrelin in cultured cells. 5 This indicated that AGS cells possess the essential factors required for posttranslational processing and acylation of the proghrelin peptide. On the other hand, ghrelin mRNA itself was expressed at low levels in AGS cells; moreover, ghrelin was not detected by immunoblot analysis and ELISA in AGS cells. It is known that Helicobacter pylori infection downregulates ghrelin production.20–22 In addition, it has been reported that H. pylori can infect AGS cells and facilitate their proliferation, also activating nuclear factor-κB and interleukin-8.23,24 The low expression levels of ghrelin in AGS cells may be caused by infection with H. pylori. Here, we attempted to establish a ghrelin-expressing stable cell line from AGS cells and successfully isolated a ghrelin-stable transfectant line, AGS-GHRL8, which secreted high levels of des-acyl ghrelin into the medium. In addition, treatment of AGS-GHRL8 with octanoic acid caused a dose-dependent increase in the secretion of octanoylated ghrelin. These results suggest that proghrelin is processed to octanoylated ghrelin by furin and GOAT in these cells, and thus, this cell line may be useful for exploring inhibitors of octanoylated ghrelin production.
We then tested the usefulness of this cell line by investigating whether heptanoic acid and butyric acid inhibit the production of octanoylated ghrelin. We hypothesized that heptanoic acid, but not butyric acid, could suppress the octanoylated ghrelin production because heptanoyl ghrelin, but not butyryl ghrelin, was detected in the mouse stomach after ingestion of the corresponding fatty acids. 13 Heptanoic acid at 100 µmol/L completely suppressed the increase in secretion of octanoylated ghrelin caused by addition of octanoic acid, whereas butyric acid did not; thus, our novel gastric cell-based assay system would facilitate in vitro assessment of the suppressive effects of candidate compounds on octanoylated ghrelin production.
It is likely that there are two bottlenecks in the production of octanoylated ghrelin in cells: one is the uptake of fatty acids into cells, and another is the acylation of ghrelin protein by GOAT ( Fig. 5 ). Since extracellularly secreted octanoylated ghrelin is measured by our assay system, it is impossible to discriminate the point at which heptanoic acid inhibited the octanoylated ghrelin production. If heptanoic acid would inhibit the acylation by GOAT, heptanoyl-modified ghrelin might be produced. The investigation of the existence of heptanoyl ghrelin may help to clarify the mechanism of the inhibition of octanoylated ghrelin secretion by heptanoic acid.
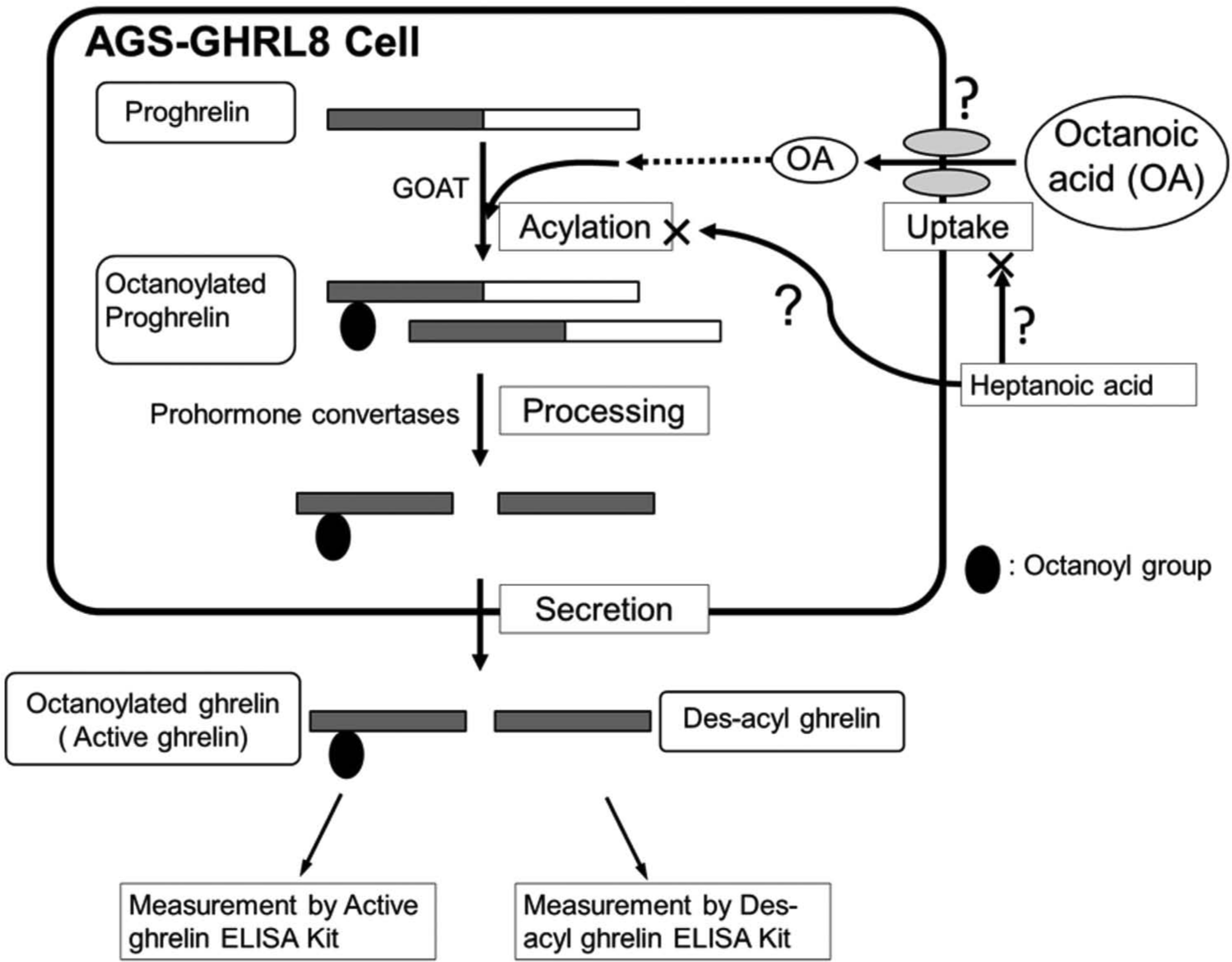
A speculative mechanism for the suppression of octanoylated ghrelin production by heptanoic acid in AGS-GHRL8 cells. ELISA, enzyme-linked immunosorbent assay.
Octanoic acid and heptanoic acid are medium-chain and short-chain fatty acids, respectively. Among the mediators of long-chain fatty acid trafficking into cells is a family of membrane-associated proteins termed fatty acid transport proteins (FATP)25–27; it is not known whether FATP also mediates short- and medium-chain fatty acid trafficking. If octanoic acid and heptanoic acid are transported into cells by the same FATP, it is possible that heptanoic acid may compete with octanoic acid for transport by this protein. Furthermore, FATP may indirectly enhance fatty acid uptake by activating acyl-CoA,25,28 which has recently been reported to be a substrate for ghrelin acylation. 29 Heptanoic acid may compete with octanoic acid for synthesis of an acyl-CoA derivative, but this remains to be investigated. On the other hand, heptanoyl ghrelin has been detected in the stomach of mice after ingestion of either heptanoic acid or glyceryl triheptanoate. 13 GOAT strongly prefers hexanoyl-CoA to octanoyl-CoA as an acyl donor 30 ; if GOAT could mediate acylation by heptanoic acid, heptanoic acid may compete with octanoic acid for acylation by GOAT. Our results also showed that butyric acid did not suppress octanoylated ghrelin production, suggesting that butyric acid would not compete with octanoic acid in the process of producing acylated ghrelin. Although butyryl-modified ghrelin peptides could not be detected after ingestion of butyric acid, 13 serine-3 butyrylated ghrelin has been detected in HEK293 cells transfected with ghrelin and GOAT cDNA. 15 These reports indicate that butyric acid is a substrate for the production of acylated ghrelin, but to a lesser extent than octanoic acid.
In the present study, we successfully established a gastric cell-based assay system for exploring inhibitors of octanoylated ghrelin production. Indeed, our present study demonstrates, for the first time, that heptanoic acid is a candidate inhibitor of the production of octanoylated ghrelin; however, heptanoic acid is not naturally present in food sources and is not naturally synthesized in mammals. Further studies using our gastric cell-based system should be conducted to investigate whether natural fatty acids contained in food oil, such as fish or plant oil, can suppress the production of octanoylated ghrelin, and this cell-based assay system will facilitate the screening of such candidate compounds, thereby contributing to the prevention of obesity.
On the other hand, many reports indicate the stimulating effects of ghrelin on cancer cell growth, for example, stimulating adrenocortical tumor cell growth through an antiapoptotic pathway, 31 stimulating proliferation in the PC3 prostate cancer cell line and activating mitogen-activated protein kinase, 32 and an autocrine proliferative effect via a pathway involving an as yet unidentified ghrelin receptor on leukemic HL-60 and THP-1 cells. 33 Therefore, AGS-GHRL8 cells could also be used for exploring candidate inhibitors of cancer cell growth.
For the evaluation of inhibition of acylated ghrelin production, many cell-based assay systems have been developed recently. 18 Our gastric cell-based assay system, AGS-GHRL8, should be comparable to them.
Footnotes
Acknowledgements
We thank Dr. Tsukasa Fujiki for his helpful technical advice.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Grants-in-Aid for Scientific Research (JSPS KAKENHI Grant number 22500794) from Japan Society for the Promotion of Science.
