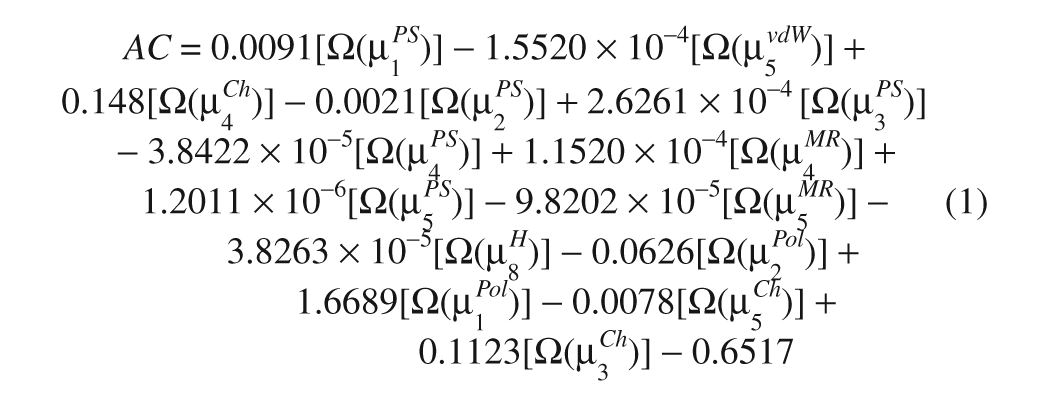Abstract
Flavonoids have been reported to exert multiple biological effects that include acting as pro-oxidants at very high doses. The authors determined a structural alert to identify the clastogenic activity of a series of flavonoids with pro-oxidant activity. The methodology was based on a quantitative structure–activity relationship (QSAR) study. Specifically, the authors developed a virtual screening method for a clastogenic model using the topological substructural molecular design (TOPS-MODE) approach. It represents a useful platform for the automatic generation of structural alerts, based on the calculation of spectral moments of molecular bond matrices appropriately weighted, taking into account the hydrophobic, electronic, and steric molecular features. Therefore, it was possible to establish the structural criteria for maximal clastogenicity of pro-oxidant flavonoids: the presence of a 3-hydroxyl group and a 4-carbonyl group in ring C, the maximal number of hydroxyl groups in ring B, the presence of methoxyl and phenyl groups, the absence of a 2,3-double bond in ring C, and the presence of 5,7 hydroxyl groups in ring A. The presented clastogenic model may be useful for screening new pro-oxidant compounds. This alert could help in the design of new and efficient flavonoids, which could be used as bioactive compounds in nutraceuticals and functional food.
Introduction
Dietary phenolic compounds are known to exhibit pro-oxidant and cytotoxic properties under appropriate conditions. Antioxidant/pro-oxidant activities of phytophenols depend on different factors such as metal-reducing potential, chelating behavior, pH, and solubility. 1 Flavonoids are a family of phenolic compounds that represent different constituents of the nonenergetic part of the human diet. They are naturally found in vegetables, berries, fruits, wine, and beer. Flavonoids have been reported to have multiple biological effects due to their antioxidant and free radical scavenging abilities. They have also been reported to exert other different effects, which include acting as pro-oxidants at very high doses. Despite the apparently beneficial health effects, several studies have described their mutagenicity and genotoxicity in both bacterial and mammalian experimental systems. 2 There are two possible reasons: their activity as pro-oxidants, 3 generating free radicals that damage DNA, and their inhibition of DNA-associated enzymes, such as topoisomerase. Unrepaired or misrepaired DNA oxidative damage can result in DNA strand breaks, mutations, 4 or chromosomal aberrations. 5 A significant number of studies reported that the flavonoids’ biological activities may play a dual role in mutagenesis and carcinogenesis. They can act, at the same time, as antimutagens and promutagens, as well as antioxidants and pro-oxidants. Indeed, in the toxicological realm, there is little information about these biochemical mechanisms and the structure–activity relationships. On the other hand, beneficial attributes are commonly overemphasized. The structure–activity (antioxidant or pro-oxidant) relationship for flavonoids has been analyzed by predictive methods based on the growth number of quantitative structure–activity relationship (QSAR)–investigated predictive methodologies.6,7 Therefore, the aim of this study was the development of a QSAR methodology as a fast screening assay to (1) identify structural alerts of clastogenic (chromosomal aberrations) pro-oxidant flavonoid activity, using topological molecular descriptors, and (2) calculate the main fragments that will be connected to structure–activity relationships.
Materials and Methods
QSAR study using TOPS-MODE approach
Descriptor generation and feature selection
The topological substructural molecular design (TOPS-MODE) approach represents a useful platform for the automatic generation of structural alerts. 8 It is based on the calculation of spectral moments of molecular bond matrices appropriately weighted, taking into account the hydrophobic, electronic, and steric molecular features. Spectral moments are the trace of the kth power of a matrix (i.e., the sum of all the main diagonal entries of such matrices). 9 A bond matrix is a square symmetric matrix in which nondiagonal entries are 1s or zeros if the corresponding bonds have a common atom or not, respectively. These matrices represent the molecular skeleton without taking into account hydrogen atoms. Bond weights are placed as diagonal entries of such matrices and represent quantitative contributions to different physicochemical properties. Among bond weights currently in use in our approach, we have standard bond distance (SD), standard bond dipole moments (DM), hydrophobicity (H), polar surface area (PS), polarizability (Pol), molar refractivity (MR), van der Waals radii (vdW), and Gasteiger-Marsili charges (Ch). Descriptors were generated to each SMILE (simplified molecular input line entry specification) reported substance with pro-oxidant activity using MODesLAB version 2.0. The MODesLAB software package has been proved to provide highly predictive models for different pharmaceutical and toxic properties.10,11
Classification model
We used the theoretic classification model that coded topological information in each spectral moment (µ) bond weight (molecular descriptors). 12 This is the orthogonal classification model validated and created by these authors:
The clastogenic activity (AC)–relationship model was conformed by 372 organic compounds, including known carcinogens, drugs, food additives, agrochemicals, cosmetic materials, medicinal products, and household materials. Here, Ω is used to indicate that the corresponding variable in brackets was orthogonalized with respect to the rest of the variables included in the model. The classification model obtained is given below, together with the statistical parameters of the linear discriminate of the squared analysis, where λ is the Wilks’s statistic, D2 is the Mahalanobis distance, and F is the Fisher ratio (Wilks’s λ = 0.629; F(14, 194) = 8.148; D2 = 2.353; p < 0.0000).
Virtual screening
Pro-oxidant reported molecules were screened, and reports were obtained from experimental data published by different authors. The virtual screening was sustained on the hypothesis that the damage to DNA could generate substances with pro-oxidant activity and could be studied through the clastogenicity. 13 The linear discriminate analysis (LDA), implemented in the statistics software, was used to generate the classification (active/inactive). The probability that a case belongs to a group (active/inactive) is known as posterior probability. From the two corresponding values to each case, the greater percentage was used to generate the classification (active/inactive). This result allows interpreting a classified compound as “active” or “inactive,” as a percentage of good classifications, in one or another group. A compound with a probability of 50.0% ± 2.5% is not classified (NC). Therefore, a classified compound with a value greater than 52.5% is considered a “good” independent classification from the active/inactive group, whereas that below 47.5% is considered a “bad” classification. This screening was the external validation of the model. With the aim of estimating the percentage of good classifications, the prediction generated by the model was corroborated with experimental reports.
Structural alerts generation and bond contribution
To generate the structural alert, we sorted the analyzed functional group effect rules into flavonoid subclasses. We needed to generate a “compound design” and another way to report flavonoids for screening. Bond contribution was calculated using “fragment contributions” in the MODesLAB software module.
Results
Classification model and virtual screening
The substances reported as pro-oxidants are shown in Table 1 , with 83.9% of the data containing substances presented in food. Many were also reported as antioxidant dietaries, such as vitamins and/or polyphenols. The other 16.1% are substances such as drugs, toxins, and so on. We obtained the first 15 spectral moments of all studied molecules, appropriately weighted to take into account the hydrophobic, electronic, and steric molecular features. The theoretic classification model for the reported flavonoid compounds and “compound design” is shown in Table 2 . It coded topological information in each spectral moment bond weight. The “active” compounds are represented as “G_2:1,” with probabilities measured in percentages. Data set predictions have more compounds as “active” (G_2: 1) than “inactive” (G_1: –1). This prediction was compared with experimental in vitro reports, shown in Table 3 . The propilgalate, (+)catechin, caffeine, naringin, β-carotene, and α-tocopherol model predictions were similar to the experimental results. The myiricetin compounds were also analyzed. Kaempferol and galangin were also studied, being a total of 10 studied compounds. As can be observed, nine compounds have been correctly classified. This result contributes to the percentage of the good classifications that the model is able to predict. Only the classification generated for ascorbic acid did not correspond to the experimentally observed data, a fact that is analyzed in detail later. It can be said that the clastogenic model predicted 90% of the good classifications that the external prediction set used.
Pro-oxidant Reported Substances

Abbreviations: CAS, Chemical Abstracts Service; SMILE, simplified molecular input line entry specification.
Classification Obtained from the Virtual Screening
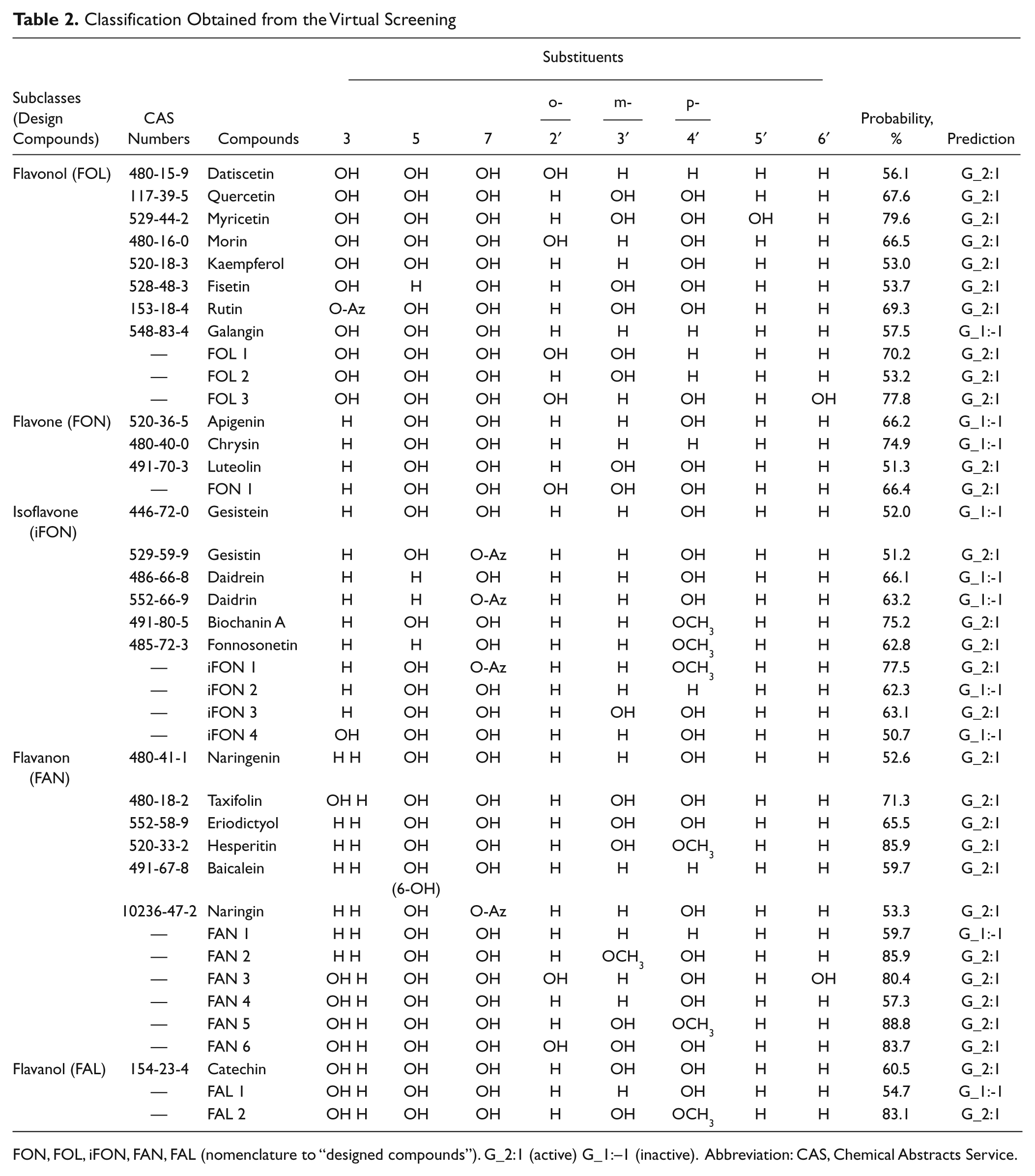
FON, FOL, iFON, FAN, FAL (nomenclature to “designed compounds”). G_2:1 (active) G_1:–1 (inactive). Abbreviation: CAS, Chemical Abstracts Service.
Model Prediction for the Compounds Tested In Vitro
Structural alerts generation and bond contribution
It was possible to study the contribution of different fragments at a substructural level, allowing definition of the structural alerts for the flavonoids. They are compounds with low molecular weight, which share a common diphenylpyran (C6-C3-C6) skeleton. Flavonoids are formed by two phenyl rings (A and B) joined through a pyran ring (heterocyclic ring C). The carbon atoms in rings A and C are numbered from 2 to 8 and in the ring B from 2′ to 6′. The basic structure of the hydrocarbon skeleton is shown in Figure 1 . We analyzed the functional group effect for flavonoid subclasses with reported pro-oxidant activity and compared them with 12 “none reported” flavonoids and 16 “designed compounds” (see Table 2 ). It was possible to analyze all the flavonoids’ skeleton rings to generate the structural alert rules. This included a hydroxyl group effect at C3, in rings A, B, and C; a methoxyl group effect; a C2:C3 phenyl group effect; a C2:C3 double-bond ring C effect; and a carbonyl group at ring C effect. This was corroborated with the bond contributions. The most important examples are shown in Table 4 .
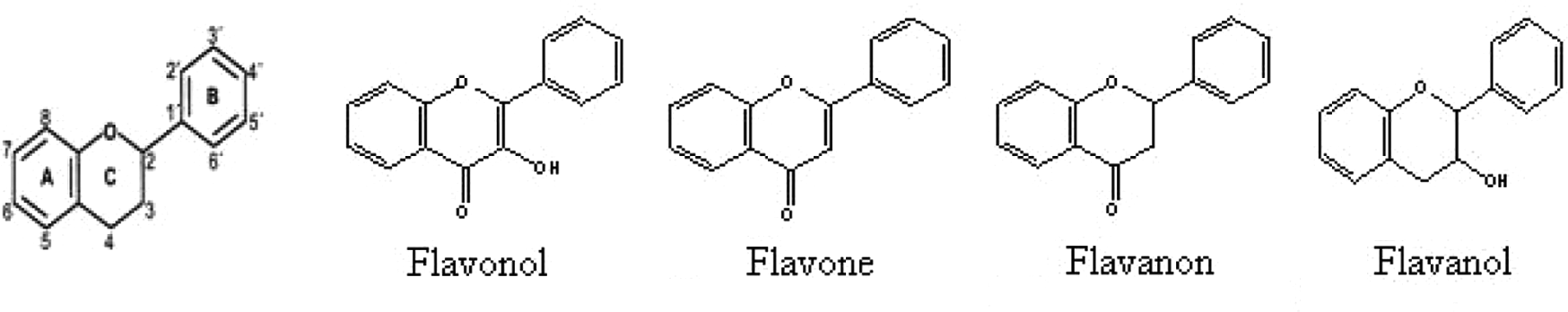
Numeration system of the flavonoids and the reported pro-oxidant subclasses.
Representative Examples of the Bond Contributions Obtained from the TOPS-MODE Classification Model
TOPS-MODE, topological substructural molecular design.
Discussion
Polyphenolic pro-oxidant compounds (flavonoids, cinnamic acid derivates, and benzoic acid derivates) were reported in this article. The 47.4% of them are flavonoids. Representations of the numeration system of flavonoids and the reported pro-oxidant subclasses are shown in Figure 1 . Flavonoids, as described before, are polyphenolic compounds that occur ubiquitously in plants, with some used as food. They present a C6-C3-C6 carbon skeleton, with some structural variations in ring C. These changes characterize the different types or subclasses of flavonoids: flavonols, flavonones, isoflavones, flavanones, and anthocyanins. The principal criterion for including the different molecules in this study was the pro-oxidant activity, reported by any experimental method. Being isomers or ions was a criterion for exclusion. Anthocyanin compounds have been widely reported in the literature. 14 Unfortunately, because of their peculiar ion structure, it was not possible to study them. This subclass of compounds is responsible for the organoleptic characteristics of dietary vegetables.
The “active” compounds have a probability of having clastogenic properties and damaging DNA. One of these relevant toxicological end points is the chromosome aberration or clastogenicity produced by chemicals. Chromosome aberrations are DNA changes generated by different repair mechanisms of DNA double-strand breaks, which are microscopically visible. 15 They are consequences of human exposure to ionizing radiation or to mutagenic chemicals. 16 The frequencies of chromosome aberrations in peripheral lymphocytes show a positive correlation with the later onset of cancer in humans. 15
The theoretical results were compared with experimental analyses of other studies. The activity of 10 compounds is an important reason to confirm this prediction. Gaspar et al 17 demonstrated that myiricetin and kaempferol are pro-oxidants and clastogenics in vitro. The classification and magnitude of these compounds were similar in our study. Myricetin was experimentally more active than kaempferol, and the prediction was from 79.6% and 53.0%, respectively. Galangin was experimentally classified as being not clastogenic and predicted as “inactive.” A similar association is possible with the other six compounds (see Table 2 ), with the exception of ascorbic acid. In this study, ascorbic acid was considered a “false positive.” Its prediction contradicted what was experimentally observed, affecting the percentage of the good classifications that the model is capable of generating. This compound is a dietary antioxidant that has been the objective of numerous studies. In the present study, it takes part in a series of external predictions due to its reported pro-oxidant activity. However, differences between its experimental pro-oxidant activity in vitro and in vivo were observed, perhaps due to the methodologies. The data indicate that in physiologic conditions, the compound is not pro-oxidant. 18
In this sense, it has been shown that vitamin C readily scavenges reactive oxygen and nitrogen species and may thereby prevent oxidative damage to important biological macromolecules such as DNA, lipids, and proteins. Vitamin C also reduces redox active transition metal ions in the active sites of specific biosynthetic enzymes. The interaction of vitamin C with free catalytically active metal ions could contribute to oxidative damage through the production of hydroxyl and alkoxyl radicals. Whether these mechanisms occur in vivo, however, is uncertain. To examine this issue, 18 studies have investigated the role of vitamin C, both in the presence and absence of metal ions, in oxidative DNA, lipid, and protein damage. They found compelling evidence for antioxidant protection of lipids by vitamin C in biological fluids, animals, and humans, both with and without iron co-supplementation. The data on vitamin C and DNA oxidation in vivo are inconsistent and conflicting, but some of the discrepancies can be explained by flaws in experimental design and methodology. These and other important issues need to be addressed in future studies of the role of vitamin C in oxidative damage.
All the data confirm the model predictive quality (90% of good classifications) when it is necessary to predict clastogenic activity of compounds with reported pro-oxidant activity. If we accept the chromosome aberration theory as an end point from oxidative DNA damage, 19 then it is possible to predict new substances with pro-oxidant activity (with DNA damage probability) using this model. This may be of great interest for studies related to the topic of cancer.
Structure alert
The basic chemical structure of flavonoids allows many substitution patterns in ring C. The reported pro-oxidant flavonoids present hydroxyl substituents on both benzene rings (A and B) and the heterocyclic ring C. However, depending on the concentration and the hydroxyl substituent patterns, these polyphenolic compounds can also act as pro-oxidants. 20 Also, functional group effects in DNA damage by clastogenic activity are analyzed in Table 2 .
Hydroxyl group effect at C3
The C3 hydroxyl group effect was analyzed when it was positioned at the sp3 and sp2 hybridization carbon. First, the flavonone subclass was compared with equivalent flavonols (apigenin vs. kaempferol and luteolin vs. quercitin). The results are shown in Table 2 . The classification as “active” is more probable in the flavonol subclass. The substituents blending to the sp2 carbon are positive to the activity.
The hydroxyl group at the C3 effect (sp3 hybridization, double bond 2:3 not present) was verified by studying eriodictyol and taxifolin. In the prediction, they are active compounds with high probability values (65.5% and 71.3%). A hydroxyl group at the sp3 carbon is also a positive contribution to the activity. A similar result was observed 21 when the hydroxyl group at the C3 position increased quercetin and kaempferol oxidation capacity with copper. These results demonstrated that the removal of the C3 hydroxyl group from the structure (e.g., luteolin) produces a probable decline in the activity. 22 The pro-oxidant activity’s increment is associated with a loss of stability by p-π conjugations. The C2:3 double-bond removal is associated with high probability values.
Hydroxyl group effect at ring B
Different compounds of flavonol, flavone, and isoflavone subclasses were compared. At the flavonol subclass, kaempferol, quercetin, and myricetin have one, two, or three substituents, respectively. In the case of galangin, there are no hydroxyl groups in the chemical structure. It was observed that the active probability is proportional to the number of hydroxyl groups present in this ring. Three hydroxyl groups increased the probabilities to 27%. Each hydroxyl group increased the probability to 14%. A comparison of chrysin, apigenin, and luteolin found the same results as for the flavonol compounds. The increase in the probability of being “active” is higher when hydroxyl groups in ring B are increased. Hydroxyl substitutions at 2′, 4′, and 6′ positions are characteristic of the FOL 3, and they are different from other compounds. The “active” exhibited probability showed a high value (77.8%), with this molecule being an optimum candidate. Similar results were observed in isoflavones and flavanonol compounds.
Hydroxyl group effect at rings A, B, and C
The presence of the same amount of hydroxyl groups in flavonoid rings A, B, and C was analyzed. Kaempferol, fisetin, and FOL 2 were compared. Quercetin and morin, featuring five hydroxyl groups in the system, presented the same results. It can be predicted that the activity is proportional to the hydroxyl groups’ contribution.
Methoxyl group effect
Isoflavone and flavanon subclasses were compared, and the methoxyl group effect was analyzed. As shown in Table 3 , gesistein (characterized by having no methoxyl groups) is an “inactive” compound, contrasting with the biochanin A model classification (75.2%). Daidrein and fonnosonetin predictions are similar. Eriodictyol versus hesperitin and eriodictyol versus FAN 2 were compared and also showed similar results. As a result, the effect of the methoxyl groups can be associated with an increase in activity.
C2:C3 phenyl group effect
The isoflavones, which contain a C3 phenyl group, are different from other subclasses. iFON 3 (design compound) was compared to luteolin (reported compound). The phenyl groups in the C3 position increase the activity probability through an electrostatic effect between the benzene ring and the carbonyl group by nearing the maximum possible.
C2:C3 double bond on ring C effect
A double bond between C2 and C3 is present in all subclasses, except for the flavanon and flavanol families. The compared compounds are shown in Table 3 . The double bonds’ absence is important to the activity. For instance, eriodictyol (65.5%) is “more active” than luteolin (51.3%), suggesting that aromatic removal can be positive to the activity.
Carbonyl group on ring C effect
The carbonyl group in the C4 position of ring C is common in all analyzed subclasses, except for the flavanol compounds. Comparing the catechins with the taxifolin, they have probability values indicating that the carbonyl group is relevant to modulate the activity. The probability for taxifolin is 10.8%, which is more than when the carbonyl group is not present.
In summary, the criteria for maximal clastogenicity of pro-oxidant flavonoids are as follows:
The presence of the 3-hydroxyl group in ring C
The hydroxyl groups at ring B, especially in the ortho position and agglutinated
The presence of methoxyl and phenyl groups
The presence of a 4-carbonyl group at ring C
The absence of a 2,3-double bond in ring C
The structural alerts: flavonol, flavonol, isoflavone, flavanol, and flavone
Fragment contributions
Bond contributions are numeric characterizations of such bonds, which permit the identification of some groups or regions of a molecular framework that can be responsible for a property/activity. This procedure consists of transforming a QSAR model into a bond additive scheme in which you can calculate the property under study as the sum of bond contributions of a molecule. When the function between the spectral moment and activity was obtained, we could determine the bond contribution to the activity of each structural fragment or substitute group. Table 4 shows examples of the calculated bond fragment. The selected compounds were “active” in the predictions. The hydroxyl group contribution is positive when C3 appears in ring C, corroborating the alert analysis. The 5,7-hydroxyl groups in ring A have 0.172 and 0.153 fragment contributions, respectively, and are more than the hydroxyl groups in ring C. This is very interesting because these substitutions are present in 15 of the 24 tested flavonoids (62.5%). It can be concluded that 5,7-hydroxyl substitutions are very important for the activity, so the methoxyl and hydroxyl groups have similar bond contributions.
The 2:3 double bond at ring C is an example of a negative fragment contribution (–0.111) of the “active” molecules. Only one fragment does not determine the biologic activity. Also, the additional collaborative effect at the molecule is needed.
As described before, flavonoids have been reported as molecules that present pro-oxidant and clastogenic activity. The QSAR study allows a fast virtual screening to predict biological activity. The QSAR model also allows the identification of clastogenic compounds with great structural variability. This model not only allows the classification of chemicals as clastogenic or nonclastogenic but also permits the identification of the molecular regions responsible for the clastogenic activity of the pro-oxidant flavonoid compounds. The substances can be classified as “active” or “inactive” according to the model. The topological information was based on the identified structural alerts that are flavonol, flavonol, isoflavone, flavanol, and flavone. Therefore, the structural criteria for maximal clastogenicity of pro-oxidant flavonoids are as follows: (1) the presence of the 3-hydroxyl group in ring C; (2) the maximal hydroxyl groups in ring B, especially agglutinated and in the ortho position; (3) the presence of methoxyl and phenyl groups; (4) the presence of the 4-carbonyl group at ring C; and (5) the absence of the 2,3-double bond at ring C. The calculated fragment contributions demonstrated that 5,7-hydroxyl groups at ring A are also important to the activity. The clastogenic model may be useful for screening new pro-oxidant compounds. This alert could be helpful for the design of new and efficient flavonoids that could be used as bioactive compounds in nutraceuticals and functional foods.
Footnotes
Acknowledgements
The authors thank the creator of the MODesLAB software. The authors also thank Aldo Hernández, Ph.D., and Luis Ledesma, Ph.D., from the University of Havana.