Abstract
Vascular adhesion protein–1 (VAP-1), also known as semicarbazide-sensitive amine oxidase (SSAO) or copper-containing amine oxidase (AOC3, EC 1.4.3.6), catalyzes oxidative deamination of primary amines. One endogenous substrate has recently been described (Siglec 10), and although its mechanism of action in vivo is not completely understood, it is suggested to play a role in immune cell trafficking, making it a target of interest for autoimmune and inflammatory diseases. Much of the enzymology performed around this target has been conducted with absorbance, fluorescent, or radiometric formats that can have some limitations for high-throughput screening and subsequent compound profiling. The authors present the use of a bioluminescent assay, originally developed for monoamine oxidase enzymes, in a high-throughput format. It can be used for related SSAOs such as AOC1 given their substrate similarity with VAP-1. The authors also demonstrate that it is compatible with different sources of VAP-1, both purified recombinant and VAP-1 overexpressed on live cells.
Introduction
V
The active sites of SSAOs contain an essential topoquinone (TPQ) cofactor, which is modified from tyrosine in a copper-dependent manner, and so these enzymes are also called copper-containing amine oxidases (AOCs). VAP-1 can oxidize a variety of amine substrates, including methylamine and aminoacetone (physiological) and benzylamine (nonphysiological). Siglec 10 has recently been described as a leukocyte ligand. 7 The enzymatic reaction involves reduction of the primary amine with formation of a transient Schiff base via the TPQ with the release of an aldehyde. The reduced TPQ is then hydrolyzed with oxygen, and subsequently hydrogen peroxide and ammonia are released. There are four SSAO family members, including VAP-1 (AOC3), AOC1 (soluble), AOC2 (retina specific), and AOC4 (inactive pseudogene).
Valley and colleagues 8 have recently reviewed the inadequacies of current methods of assaying amine oxidases; high-performance liquid chromatography (HPLC) and radiochemical detection are time-consuming and expensive; fluorescence and absorbance assays are rapid and sensitive but may be susceptible to compound interference. They also disclosed a luciferase-based assay for monitoring monoamine oxidase (MAO)–A and MAO-B activity via bioluminescence. We investigated this bioluminescent assay and predicted that it would be suitable for measuring VAP-1 activity as MAOs and AOCs share common substrates and inhibitors, 3 but nonetheless it was important to demonstrate as we evaluated our high-throughput screening (HTS) screening options.
We therefore present our VAP-1 bioluminescent assay development steps and summarize the performance of the assay used for initial HTS, counterscreening, and cellular profiling. We believe it provides a convenient, versatile, and robust HTS format for screening multiple amine oxidases and offers some advantages over other screening formats (e.g., radiometric, absorbance). Here, we also show the utility of this bioluminescent assay to measure the activity of multiple amine oxidases from different sources, such as recombinant purified proteins and as AOC enzymes expressed on the surface of live Chinese hamster ovary (CHO) cells, allowing the assay to be used broadly in monitoring the optimization of compounds throughout a drug discovery program.
Materials and Methods
Construct design
Human VAP-1 (41-755), murine VAP-1 (41-755), rat VAP-1 (41-755), and human AOC1 (20-751) with a mutated glutathione S-transferase (GST) N-terminal fusion tag (with cysteines at positions 85 and 138 and serine at position 178) were expressed in the pcDNA-Dest40 Gateway Vector (Invitrogen, Carlsbad, CA). The native signal sequence was replaced with a murine IgG heavy-chain signal peptide, and an human rhinovirus (HRV) protease cleavage site was introduced after the mutated GST fusion tag.
Cell line generation
HEK 293 cells were grown in RPMI 1640 Medium (Invitrogen) supplemented with 10% fetal bovine serum (FBS; Invitrogen). They were transfected with the expression vectors (human VAP-1, mouse VAP-1, or rat VAP-1) using Freestyle MAX (Invitrogen) transfection reagent according to the manufacturer’s protocol and selected with 500 µg/mL G418 (Invitrogen). 9 The relative protein expression levels from pools of stably expressing cells were determined by Western blot. The stable pools were adapted to suspension conditions in FreeStyle 293 serum-free medium (Invitrogen) and scaled up for 10- or 20-L runs with a Wave bag bioreactor using the FreeStyle medium without G418.
CHO cells were cultured in RPMI 1640 with glutamine from Gibco/Invitrogen, 10% FBS from HyClone (Logan, UT), and 1 mg/mL (human clones) or 0.5 mg/mL (murine clones) G418 (Gibco/Invitrogen) at 37 °C, 5% CO2. Full-length constructs of human (GenBank Accession #NM_003734), mouse (GenBank Accession #NM_009675), and rat (GenBank Accession #NM_031582) VAP-1 in pcDNA DEST 40 were generated (Geneart, Inc., Ontario, Canada) and transfected into CHO K1 cells using Lipofectamine 2000 (Invitrogen) according to the manufacturer’s instructions. Stable cell lines were established by limiting dilution under G418 antibiotic selection for 2 weeks. Antibiotic-resistant clones were screened for VAP-1 expression by flow cytometry (Guava Technologies, Inc., Hayward, CA) using VAP-1-specific monoclonal antibodies: TK8-14 (Santa Cruz Biotechnology, Santa Cruz, CA) for human VAP-1 expression, clone 174-5 (Hycult Biotechnology, Uden, The Netherlands) for rat VAP-1, and clone 7-88 (Hycult Biotechnology) for mouse VAP-1 expression. Secondary antibodies for the anti-human and anti-mouse antibodies were goat anti-mouse IgG-FITC (cat. sc-2010; Santa Cruz Biotechnology) and goat F(ab1)2 anti-rat IgG-PE (R&D Systems, Minneapolis, MN), respectively. Isotype controls included mouse IgG2a κ pure, mouse IgG2b κ isotype control, and mouse IgG1 κ isotype control (BD Biosciences, San Jose, CA). Multiple clones expressing >98% VAP-1-positive cells were selected, expanded, and cryopreserved for each line. For enzymatic analysis, cells were grown to confluence; washed with phosphate-buffered saline (PBS) prior to treatment with Accutase (Millipore, Billerica, MA) for 10 min at 37 °C, 5% CO2; resuspended in media; and then washed twice and resuspended in assay buffer.
Purification of GST-VAP-1 and GST-AOC1
Both GST-VAP-1 (VAP-1) and GST-AOC1 (AOC1) were purified from fresh biomass by affinity chromatography using Glutathione Sepharose 4B resin (Amersham Biosciences, Piscataway, NJ) in the presence of 0.1% Tween-20. Postdialysis, 0.1% Tween-20 and 10% glycerol were added. Protein concentration was determined by Bradford assay. Total protein yield was approximately 1 to 1.4 mg/L.
Luminescence assay
The AOC luminescence assay for recombinant and cell surface VAP-1 (CHO-VAP-1) was the Promega MAO-Glo Assay (cat. V1401; Promega, Madison, WI) with the following modifications; assay plates were white, medium binding, flat-bottom plates (96-well; cat. 655075; Greiner Bio One, Monroe, NC). The assay buffer was modified from the “physiological HEPES” 10 and consisted of 50 mM HEPES (pH 7.4), 5 mM KCl, 2 mM CaCl2, 1.4 mM MgCl2, 120 mM NaCl, 0.001% Tween-20, and 100 µM TCEP (Thermo Scientific, Rockford, IL).
For assay development with recombinant VAP-1, AOC1 and CHO-VAP-1 ( Figs. 1 and 2 ), the reaction volume was 50 µL (25 µL diluted enzyme or cells plus 25 µL diluted substrate) with a 60-min 37 °C catalysis reaction followed by the addition of 50 µL detection reagent (100 µL total) and a 20-min 37 °C detection reaction, after which relative light units (RLU) were counted on an LJL BioSystems Analyst AD (Molecular Devices, Sunnyvale, CA). The Promega MAO-Glo substrate ((4S)-4,5-dihydro-2-(6-hydroxybenzothiazolyl)-4-thiazolecarboxylic acid) was solubilized in DMSO (16 mM). For experiments titrating substrate, buffers were adjusted such that all samples contained the same final DMSO concentration of 1%. For IC50 determinations, compound inhibitors were solubilized in 100% DMSO and serially diluted in DMSO prior to being diluted into assay buffer. They were preincubated with enzyme (20 min) prior to the catalysis reaction. There was a 1% final DMSO concentration from compounds in the recombinant VAP-1 assay and 0.25% DMSO in the CHO-VAP-1 assay, which was mimicked in controls and backgrounds. For compound assays, the substrate concentration used varied depending on the respective AOC Km such that all IC50 data are comparable. Buffer blanks with buffer plus substrate were subtracted from sample and control values. The IC50 curves for the recombinant VAP-1 assay were generated by SigmaPlot regression fitting using the equation category “Sigmoidal” and the equation name “Hill 3 Parameter.” For the CHO-VAP-1 assay data, SigmaPlot 9.0 (Systat Software, San Jose, CA) was used to graph substrate concentration versus RLUs fitting the data to the rectangular hyperbolic curve according to the Michaelis-Menten equation. XLfit was used to calculate compound concentration versus percent activity (% of control activity, no inhibitor). IC50 values were calculated by fitting the data to the four-parameter logistic curve in SigmaPlot 9.0. The compounds LJP 1586 and LJP 1207 were synthesized as described 11 except for the last two steps, which were combined into one step as follows: 9.0 g of the boc-amine was dissolved in 2 M HCl in doxane (25 mL). After approximately 30 min, a solid began to form. This was left to stir for 18 h, and the solid was collected by vacuum filtration to afford both isomers of the final product. The solid was purified by reverse-phase HPLC to afford 1.7 g (29%) of the desired z-isomer after lyophilization. The structure was identified and confirmed using 1H nuclear magnetic resonance (HNMR) and mass spectral information.
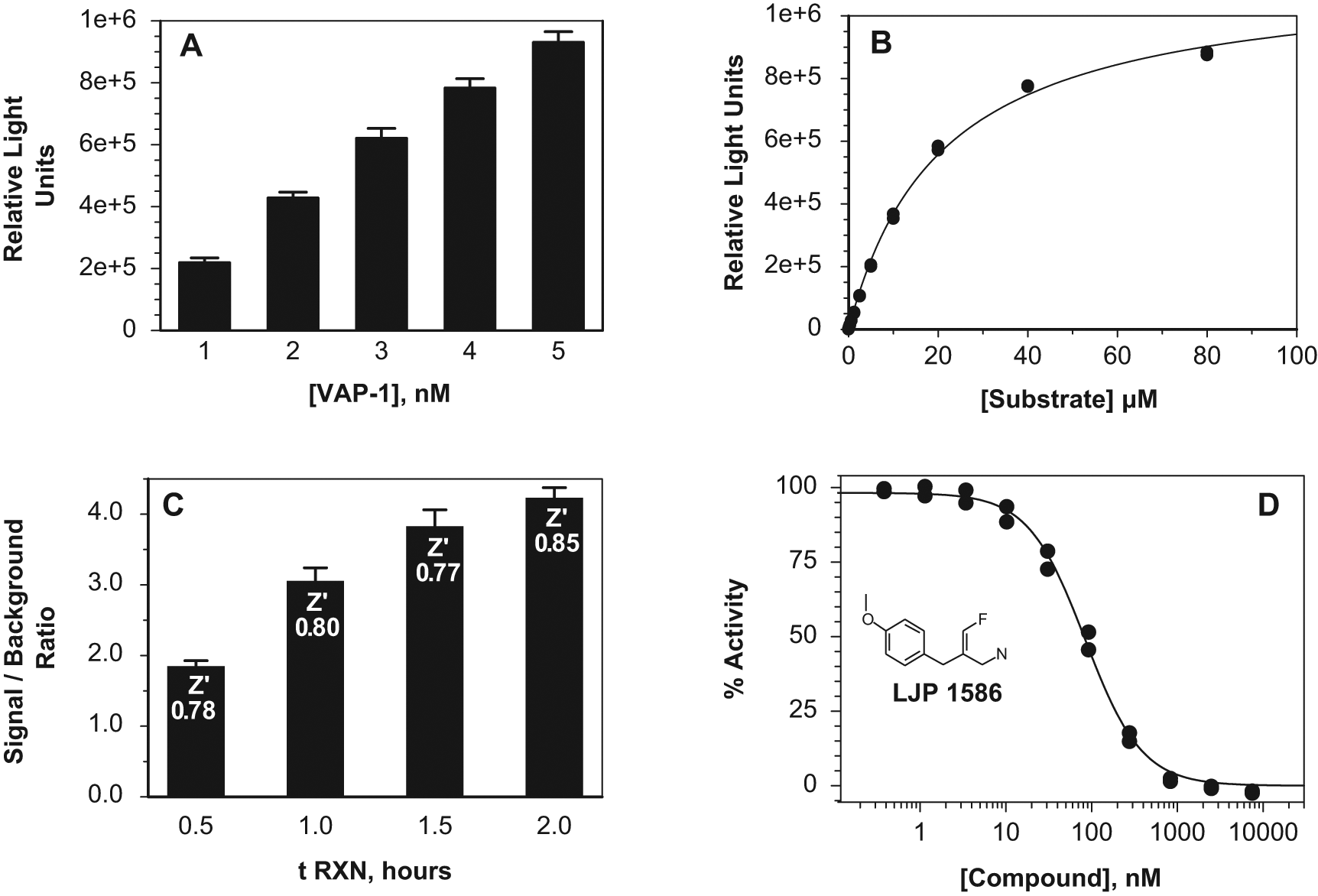
Vascular adhesion protein–1 (VAP-1) assay development. (
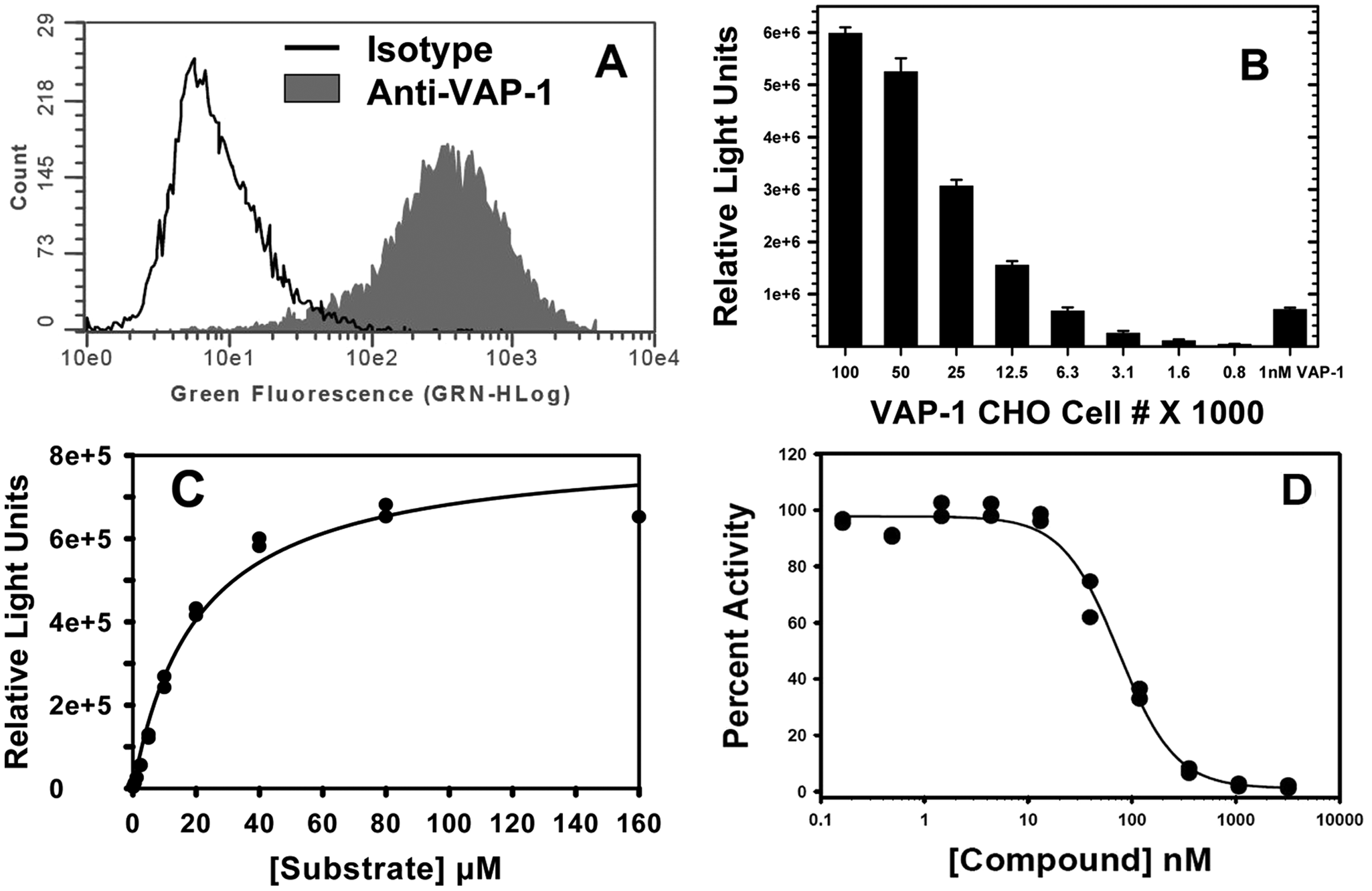
Vascular adhesion protein–1 (VAP-1) Chinese hamster ovary (CHO) cell characterization and assay development. (
For HTS ( Fig. 3 ), the VAP-1 assay was miniaturized to 30 µL total volume in a 384-well white nonbinding surface plate (#3673; Corning, Corning, NY) in a reaction comprising 3 µL VAP-1 protein, 2 µL test compound, 10 µL substrate, and 15 µL MAO-Glo detection reagent. Final concentrations in the screening assay were 2 nM VAP-1, 27 µM substrate, and 5 µg/mL test compound. The assay was automated on the Allegro robotics system (Caliper, Hopkinton, MA) using a Sciclone pipettor to deliver enzyme and compound (Caliper) and Multidrop Combi’s (Thermo Fisher Scientific, Waltham, MA) to deliver substrate (1-h incubation) and detection reagents (15-min incubation). The plates were incubated in a humidified incubator (Liconic, Woburn, MA) at 37 °C, and luminescence was read on an Analyst HT (Molecular Devices). The percent of control (POC) and IC50 values were calculated relative to a negative control containing complete reaction minus VAP-1 and a positive control containing VAP-1 buffer with 1% DMSO in place of test compound, using Activity Base (IDBS, Guildford, UK).
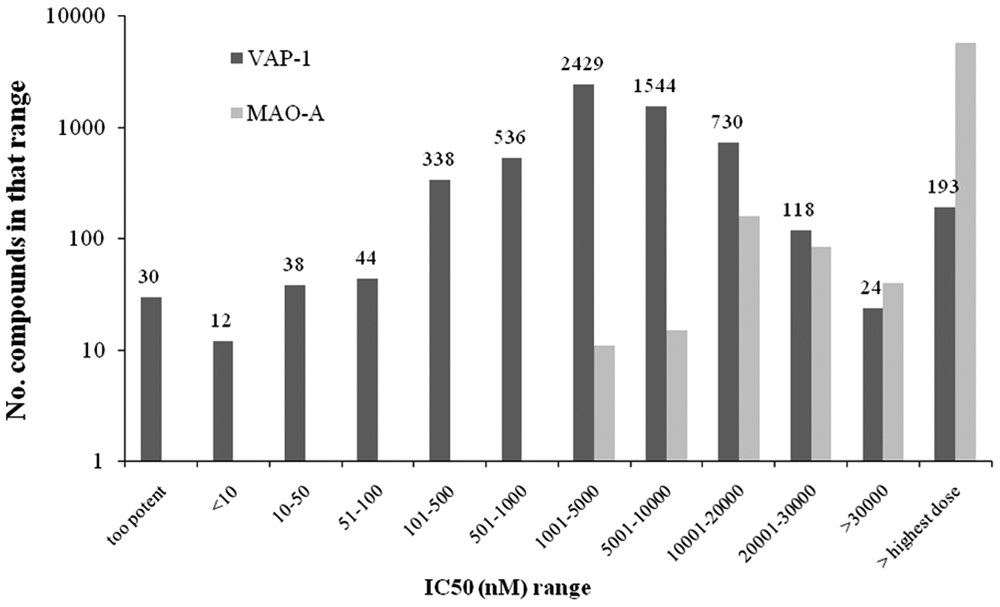
High-throughput screening of vascular adhesion protein–1 (VAP-1). Distribution of IC50s in the VAP-1 and monoamine oxidase (MAO)–A assays.
Results and Discussion
The luminescence assay was modified from the Promega MAO-Glo assay (see Materials and Methods) and extensively tested with different SSAO enzymes, from recombinant and cellular sources, and shown to be a robust bioluminescent assay, which can be used to identify and characterize VAP-1 inhibitors. We describe herein the details of the assay characterization, including evaluation of known VAP-1 inhibitors and its use in an HTS screen. Recent reports describing inhibitors of VAP-1 have used radiometric, a combination of fluorometric and radiometric, or absorbance assay formats to evaluate and compare this class of enzymes.12–14 We determined that the bioluminescent format both enables a direct comparison of IC50s for the different amine oxidases from different sources and uses a convenient HTS-compatible method (rapid, robust, without compound interference, and nonhazardous) and present it as an additional format for measuring this class of enzymes.
We also analyzed a peroxidase-coupled fluorometric assay (Amplex Red Monoamine oxidase kit from Invitrogen [data not shown]), which is a two-step assay based on utilization of the hydrogen peroxide by-product of benzylamine conversion to benzaldehyde by the target amine oxidase. Hydrogen peroxide then oxidizes the substrate ADHP (10-acetyl-3, 7- dihydroxyphenoxazine) to produce fluorescent product. We chose to develop the bioluminescent assay as the Amplex Red reagent is reported by Invitrogen to be unstable in the presence of reducing agents (such as dithiothreitol and 2-mercapto-ethanol at >10 µM) and at high pH (>8.5). Also, it has been reported that the assay can give a positive result due to mobilization of NADH or glutathione. 15
Figure 1 shows the steps in the development of the assay for human recombinant VAP-1 protein, produced as described in the Materials and Methods. Enzyme-dependent product formation was determined by titrating human recombinant VAP-1 in the presence of 27 µM substrate ( Fig. 1A ). Under these conditions, data were linear up to 4 nM. To calculate substrate-dependent product formation, 1 nM recombinant human VAP-1 was assayed versus 11 concentrations of substrate up to 80 µM (including 0 µM; Fig. 1B ). The simulated Vmax is 1.14 × 106 RLU (60-min reaction), and the derived apparent Km(Promega Substrate) value is 20.7 µM. Under the same assay conditions, the derived apparent Km (Promega Substrate) values for MAO-A and MAO-B were determined to be 18 and 1.8 µM, respectively (data not shown).
The VAP-1 assay time course was conducted using 2 nM enzyme and 27 µM substrate and was shown to be robust from 30 min to 2 h ( Fig. 1C ). All conditions resulted in a percent coefficient of variation (CV%) ≤5 and Z′ determinations of >0.77. A 1-h reaction time was therefore chosen for compound profiling. Known VAP-1 inhibitors were profiled in the assay and included LJP 1586, IC50 84 nM ( Fig. 1D ) and LJP 1207, IC50 23 nM ( Table 1 ). These data are consistent with the literature11-13 and serve to validate the assay. The human recombinant VAP-1 assay was therefore determined to be a robust bioluminescent assay that could be used to identify and characterize VAP-1 inhibitors.
Summary of AOC Substrate and Inhibitor Binding Affinities

AOC, copper-containing amine oxidase; CHO, Chinese hamster ovary; ND, not determined; VAP-1, vascular adhesion protein–1.
Three different lots of material were tested, and binding affinities were 26.9, 19.4, and 20.7 µM.
Assayed with a stable pool of mouse VAP-1-expressing cells.
The assay was miniaturized for use in a high-throughput format, as described in the Materials and Methods, and used to profile approximately 890 000 compounds. The average Z′ for the screen was 0.78 with signal-to-background ratios between 7 and 8 (data not shown) and demonstrated that the assay was robust over approximately 2800 plates and 18 screening days. By comparing the potency of hits for VAP-1 to those of MAO-A, we demonstrated that specific inhibitors could be identified across a range of IC50s ( Fig. 3 ).
The assay was further modified to measure the activity of VAP-1 expressed on the surface of cells. As described in the Materials and Methods, full-length human, mouse, and rat VAP-1 were expressed on CHO cells. Flow cytometry was used to determine the surface expression level of each protein on the stable cell pool. Clones expressing >98% VAP-1-positive cells ( Fig. 2A ) were selected, expanded, and analyzed for corresponding enzyme activity. Cell number was titrated to be in the linear range of RLUs for the assay and comparable to the values obtained with 1 nM recombinant VAP-1 ( Fig. 2B ), and 6000 cells/well were used for further experiments. This allowed for better comparison of substrate Km values and IC50 values of compounds between the molecular and cellular assays. Apparent substrate Km values were determined for human, mouse, and rat VAP-1-transfected CHO cells by titrating various concentrations of substrate ( Fig. 2C ).
The Promega substrate Kmapp was determined for full-length human, mouse, and rat VAP-1 CHO cells: 25 µM (human), 26 µM (mouse), and 23 µM (rat). Vmax values were 8.22 × 105 RLU (human), 1.9 × 106 RLU (mouse), 1.7 × 106 RLU (rat). IC50 values for compounds were derived at substrate Km for each species using a constant cell number. A positive control using 1 nM recombinant VAP-1 was included in all experiments. The assay was validated by generating IC50 values for reference compounds LJP 1586 and LJP 1207, as shown in Table 1 and Figure 2D , and were 75 nM (human), 8 nM (mouse), and 26 nM (rat) for LJP 1586 and 26 nM (human), 2 nM (mouse stable pool) for LJP 1207.
The assay was also used to evaluate a related SSAO, AOC1. The substrate Km was determined to be 4.4 µM (see Materials and Methods and Table 1 ), and it was used to profile compounds, including LJP 1586 (not active) and LJP 1207 (IC50 526 nM). The assay was further evaluated to measure VAP-1 activity from mouse and human tissue lysates and plasma. Although the signal to background was smaller (~2), the assay could be used to evaluate enzyme inhibition (data not shown).
These assays allowed us to compare data generated with the truncated recombinant GST-VAP-1 fusion protein (VAP-1) to data generated with full-length cell surface–expressed VAP-1 across three species. We saw an excellent correlation of the data from the two different assays for human VAP-1 with 311 compounds evaluated (IC50s within threefold for 302 compounds, lower IC50s in the cellular assay for the remaining nine compounds, suggestive of cytotoxicity; data not shown).
