Abstract
Development and progression of colon cancer may be related to cytokines. Cytokines with diagnostic value have been identified individually but have not been implemented into clinical praxis. Using a multiplex protein array, the authors explore a panel of cytokines simultaneously and compared its performance to carcinoembryonic antigen (CEA) and carbohydrate antigen 19-9 (CA 19-9). Serum concentrations of 12 cytokines were simultaneously determined by multiplex biochip technology in 50 colon cancer patients and 50 healthy controls. Serum levels of interleukin-8 (IL-8) and CEA were significantly higher in cancer patients than in healthy controls. Areas under the receiver operating characteristic curves (AUCs) were largest for IL-8, followed by CEA, vascular endothelial growth factor (VEGF), and CA 19-9. Analyses regarding marker combinations showed an advantage over single marker performance for CEA, VEGF, and CA 19-9 but not for IL-8. Multiplex biochip array technology represents a practical tool in cytokine and cancer research when simultaneous determination of different biomarkers is of interest. The results suggest that the assessment of IL-8, CEA, VEGF, and possibly CA 19-9 serum levels could be useful for colon cancer screening with the potential of also detecting early stage tumors. Further validation studies using these and additional markers on a multiplex array format are encouraged.
Introduction
C
There is reasonable hope and emerging evidence that the presence of malignant disease could be detected by specific changes in the composition of serum proteins. Comprehensive serum proteome profiling for such tumor-specific markers has therefore become a field of intensive research, not only for CRC. 8 For instance, determination of serum levels of prostate-specific antigen (PSA) for the detection of prostate cancer, despite issues regarding specificity and sensitivity, has become routine clinical practice. 9
For CRC, serum markers in current clinical use are carcinoembryonic antigen (CEA) and carbohydrate antigen 19-9 (CA 19-9). Both markers are widely used during surveillance and as prognostic measures for disease-free survival but not for diagnostic or screening purposes because of insufficient sensitivity and specificity.9–11 Unfortunately, the use of single or a combination of these serum markers has failed to deliver diagnostic tests for colon cancer in clinical routine use so far. 12 Hereby, the measurement of a disease-specific protein panel would be deemed more successful as it provides a more comprehensive description of molecular networks and pathophysiological conditions of diseases than individual biomarkers. Against this background, the method of multiplexing, allowing simultaneous concentration assessments of several biomarkers, has become of increasing interest, especially when volumes of clinical samples are limited. Multiplexing technologies provide opportunities for integrated approaches, in contrast to conventional technologies such as enzyme-linked immunosorbent assays (ELISAs) or electrochemiluminescence immunoassays (ECLIAs). A group of serum markers with potential diagnostic value for various malignancies are cytokines.13–16 Cytokines are a group of heterogeneous proteins of high biological activity that control and regulate cell functions such as apoptosis, migration, and invasion. They are released in picomolar amounts and are often overexpressed during tumor development and progression.17–20 Therefore, we found it intriguing to investigate the diagnostic potential of serum cytokines for colon cancer screening by a multiplex biochip array using the simultaneous assessment of 12 different serum cytokines. Serum cytokines were analyzed individually and in concert to define their sensitivity and specificity and compared to the discriminative performance of CEA and CA 19-9 in serum samples of 50 patients with colon cancer and 50 healthy controls.
Materials and Methods
Material and study group
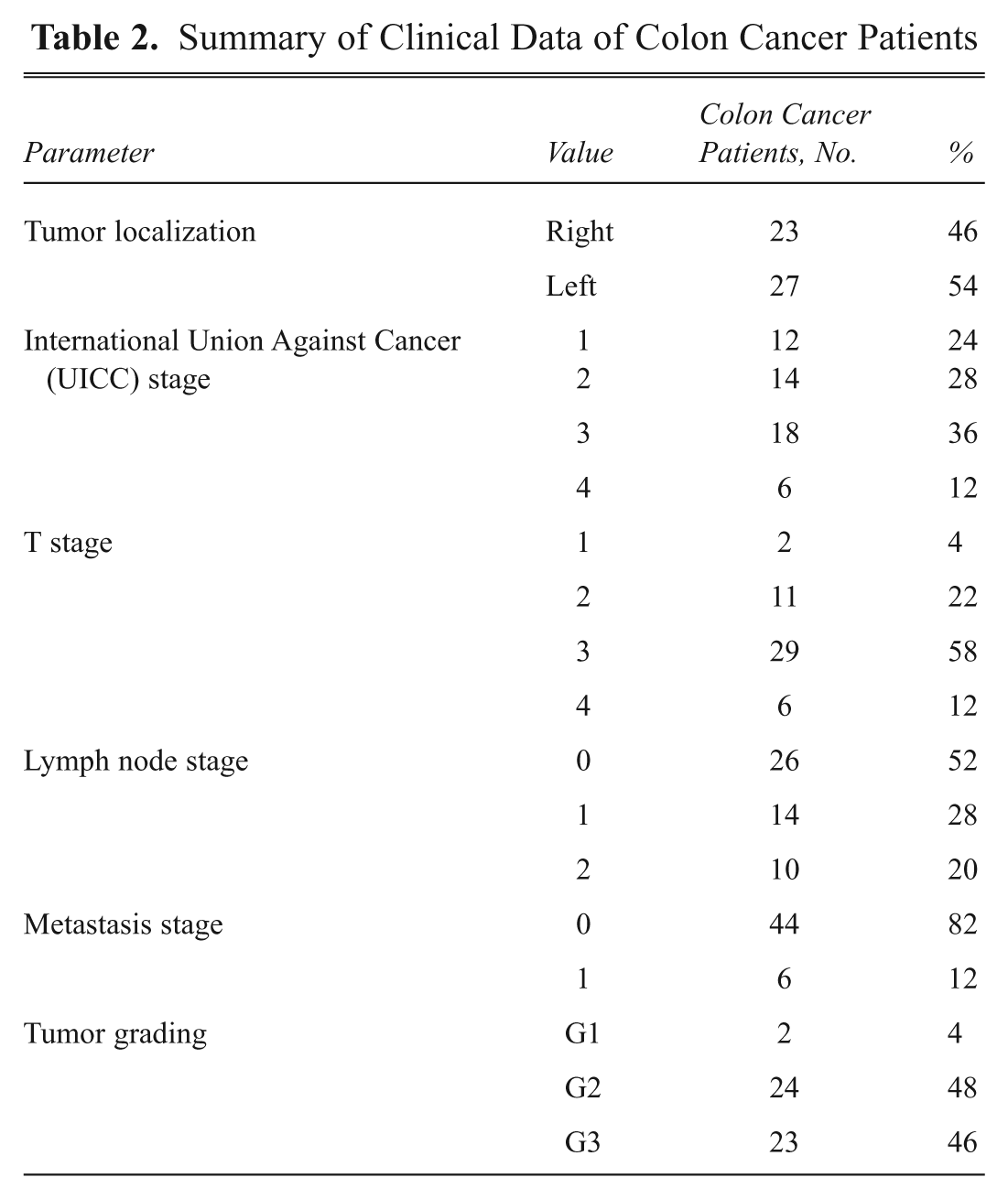
This study comprises 100 serum samples that were randomly selected out of 1161 serum samples of colon cancer patients and healthy controls collected at the University Clinic Schleswig-Holstein, Campus Lübeck, Germany, between 2007 and 2009. This serum collection belongs to the ColonBiomics biomaterial bank, which is an integral part of the Surgical Center for Translational Oncology–Lübeck (SCTO-L), University of Lübeck, and the DKH e.V.–funded network North German Tumorbank of Colorectal Cancer. Serum samples were collected adhering to the guidelines of the local ethical review board (Medical University of Lübeck, #07-124). The 100 samples comprised 50 patients with histologically confirmed colon cancer (31 men and 19 women, aged 38–88 years) and 50 healthy controls (21 men and 29 women, aged 22–90 years; Table 1 ). For control patients, phlebotomy was performed before full colonoscopy, which confirmed that no signs of premalignant (polyps, adenomas, dysplasia) or malignant (carcinoma in situ, carcinoma) lesions were present in this cohort. For cancer patients, phlebotomy was performed before neoadjuvant chemo- or radiotherapy and/or surgery. Tumors were classified according to UICC staging, revealing 12 UICC I, 14 UICC II, 18 UICC III, and 6 UICC IV stage carcinomas. Patients with UICC I and II carcinomas were summarized as early stage and patients with UICC III and IV carcinomas as late-stage cancer patients. Detailed clinical data of colon cancer patients are summarized in Table 2 .
Summary of Clinical Data of the Study Group Consisting of Colon Cancer Patients and Healthy Control Patients
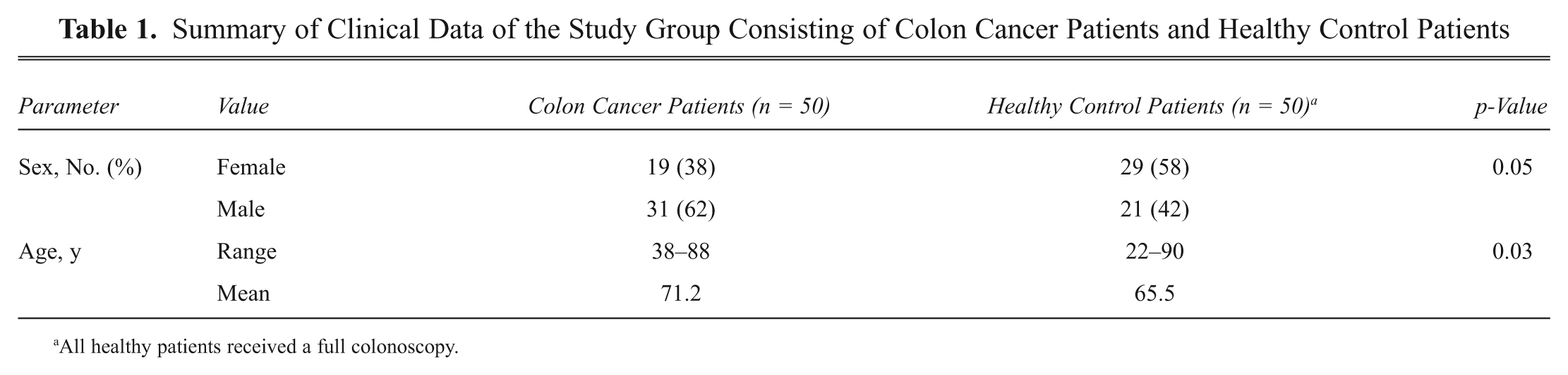
All healthy patients received a full colonoscopy.
Summary of Clinical Data of Colon Cancer Patients
Methods
All venous blood samples were obtained using serum gel-monovettes (#01.1602; Sarstedt AG & Co, Nürnbrecht, Germany) and centrifuged at 1550 g for 10 min at 4 °C to separate the serum. Aliquots of serum samples were stored at −196 °C within 30 min after phlebotomy. Samples were thawed on ice before simultaneous, parallel assessment for the 12 serum cytokines, CEA, and CA 19-9.
Determination of cytokine serum levels
For simultaneous quantitative detection of multiple cytokines from a single patient sample, we used a cytokine and growth factor multiplex biochip array in combination with the Evidence Investigator readout equipment (Randox Laboratories Ltd., Crumlin, UK). This biochip comprises interleukins IL-1α, IL-1β, IL-2, IL-4, IL-6, IL-8, and IL-10; vascular endothelial growth factor (VEGF); epidermal growth factor (EGF); tumor necrosis factor–α (TNF-α); interferon-γ (IFNG); and monocyte chemoattractant protein–1 (MCP-1). Assays were performed following the manufacturer’s instructions. The multiplex array operates methodically on the basis of an enzyme-linked immunoassay platform as previously described.21–23 In brief, simultaneous sandwich immunoassays are employed, and the biochip functions as the solid phase and reaction vessel for the immunoreactions ( Fig. 1A ). The biochip setup allows processing up to 54 samples (6 × 9 wells) simultaneously ( Fig. 1B ). In our setup, three controls and nine calibration samples were run in parallel with 42 patient samples. The inclusion of controls and calibration samples avoids the need for technical replicates for patient samples. A total of 100 µL of undiluted serum samples was applied to each reaction well on the biochip, allowing binding of particular serum cytokines to their specific primary antibody. Suitable horseradish peroxidase (HRP)–labeled secondary antibody, presented as a multiconjugate, was added and cytokines were quantified by chemiluminescence using a super-cooled charge-coupled device (CCD) camera being part of the Evidence Investigator’s imaging system as described previously.21,22
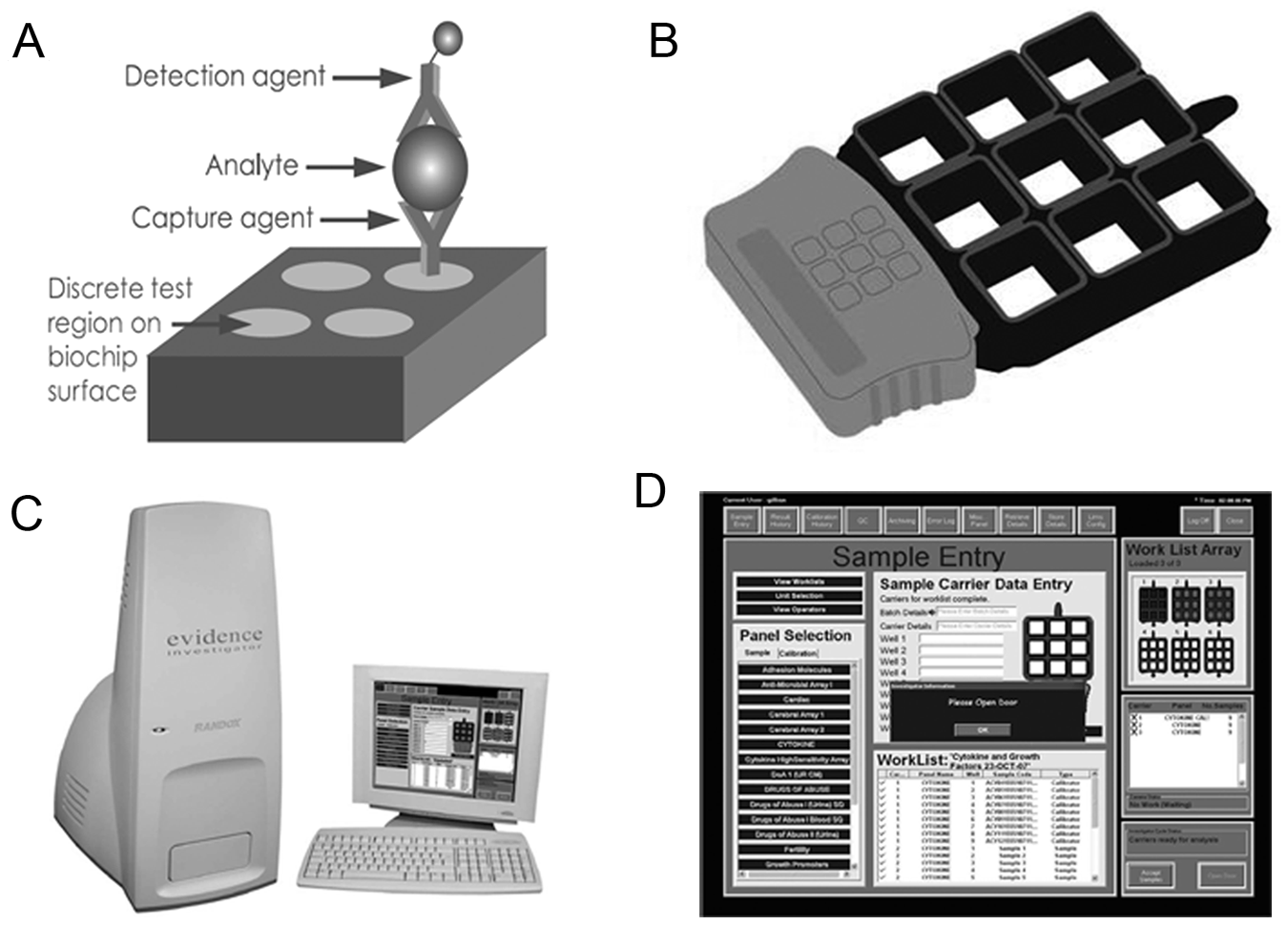
(
The readout equipment routinely assesses the quality of assay performance and generates calibration curves as previously described by FitzGerald et al. 21 The analyte’s concentration present in the sample was calculated automatically using routinely generated calibration curves (Evidence Investigator Software version 1.4). Results of individual patient sample measurements were retrieved as Excel files for further statistical analyses.
Determination of tumor markers CEA and CA 19-9
In addition, tumor markers CEA and CA 19-9 serum levels were blindly investigated by means of ECLIA kits (Roche Diagnostics, Mannheim, Germany). Assay automation was performed using the Elecsys analyzer (Roche Diagnostics). Reference cutoff values (the 95th percentile) were previously established in the Department of Clinical Chemistry (University of Lübeck, Germany) by examining sera of healthy blood donors. According to the manufacturer’s instructions, lower detection limits for the assays were 0.20 µg/L for CEA (clinical cutoff value at 4.7 µg/L) and 0.60 KIU/L for CA 19-9 (clinical cutoff value at 37.0 KIU/L).
Statistical analysis
Using SAS version 9.1 (SAS Institute, Cary, NC), we described the serum levels of the individual markers in colon cancer cases and healthy controls with respect to median levels, interquartile ranges, and overall ranges and performed nonparametric tests to compare median levels between both patient groups. We assessed the correlation between serum levels and age and sex as well as between the different markers using Spearman’s correlation coefficient. To assess the discriminatory power of the individual markers, we calculated diagnostic sensitivities when fixing the cutoff level at the 95th percentile of values measured among controls, yielding specificities of about 95%. Furthermore, we constructed receiver operating characteristic (ROC) curves and calculated the area under the ROC curves (AUC) and the corresponding 95% confidence interval (CI) using MedCalc for Windows, version 9.6.4.0 (MedCalc Software, Mariakerke, Belgium). To assess the potential of marker combination, we developed a multivariate logistic regression model by backward deletion of the least significant marker, using the Akaike information criterion. The discriminative ability of the regression model was assessed by the AUC. Given the problem of overfitting, we assessed the internal validity of the regression model by using bootstrapping techniques to estimate optimism-corrected performance. Random bootstrap samples were drawn with replacement from the full sample (200 replications), and a regression model was developed in each bootstrap sample. At each step, model performance in the bootstrap sample was compared to the performance of the respective bootstrap model when applied in the original sample to quantify the optimism. 24 The analyses on marker combinations were done using R software version 2.10.0.
Results
Here, we simultaneously assessed 12 serum cytokines using a multiplex biochip array in comparison to CEA and CA 19-9 to identify clinically applicable serum markers for colon cancer screening. In addition, markers were tested in a highly standardized serum collection of cancer and control samples stored at −196 °C within 30 min after phlebotomy following strict standard operation procedures (SOPs).
Analysis of multiplex biochip and ECLIA data
The multiplex and ECLIA analyses revealed that serum levels of IL-8 and CEA were significantly higher in colon cancer patients than in healthy controls (
Table 3
). The median level (interquartile range) for IL-8 was 10.6 pg/mL (7.29–15.76) in controls and 17.6 pg/mL (11.74–35.55) in cases (p = 0.01). The median level (interquartile range) for CEA was 1.6 µg/L (1.02–3.45) in controls and 2.8 µg/L (1.29–7.80) in cases (p = 0.02;
Figure 2
). Even though serum levels for VEGF and CA 19-9 were higher in colon cancer patients than in controls, the difference was not statistically significant (p = 0.23 and p = 0.19, respectively). All other cytokines and growth factors did not discriminate between patient groups (
Serum Levels of IL-8, VEGF, and Tumor Markers CEA and CA 19-9 in Colon Cancer Patients
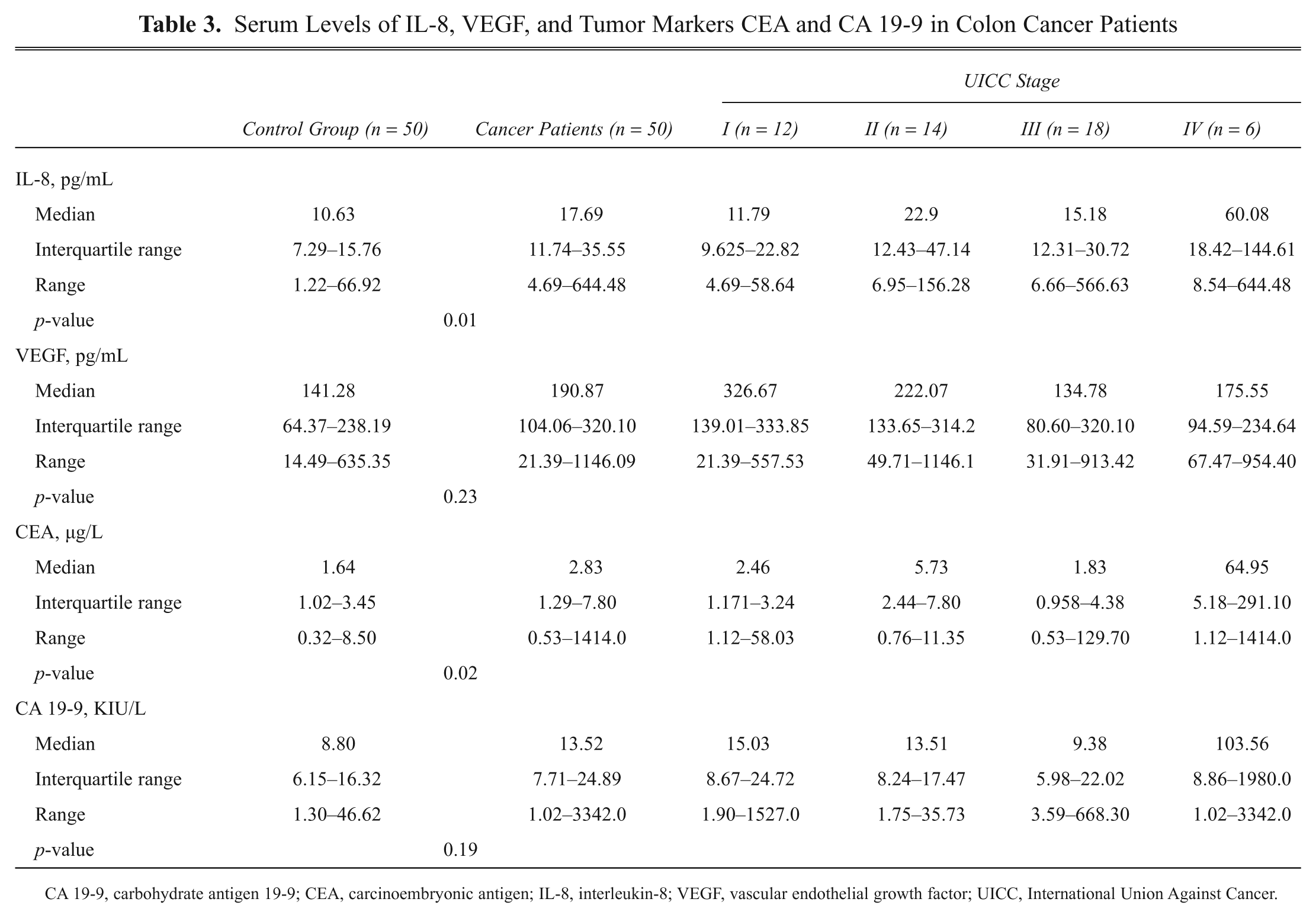
CA 19-9, carbohydrate antigen 19-9; CEA, carcinoembryonic antigen; IL-8, interleukin-8; VEGF, vascular endothelial growth factor; UICC, International Union Against Cancer.
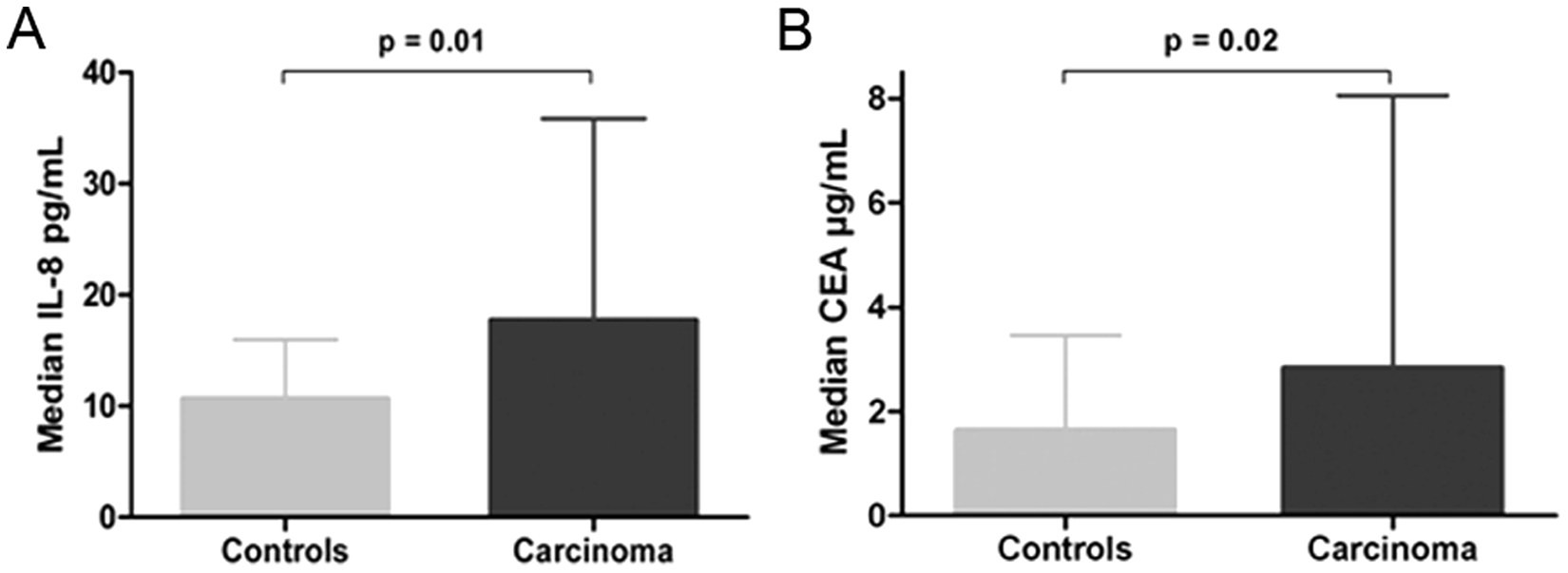
Serum levels of cytokines and cancer markers between colon cancer and controls for (
Correlation among biomarker levels and clinicopathological issues
We were particularly interested to see if the most promising markers show tumor stage–dependent expression changes. However, when comparing median serum levels of IL-8, CEA, VEGF, and CA 19-9 of early stage cancers (UICC stages I and II) with late-stage cancers (UICC stages III and IV), no significant differences could be observed, reflecting the screening potential of all four markers also for early stage tumors.
There was no correlation between any of the 12 tested markers and tumor localization, age, or sex. However, higher levels in one cytokine often correlated with higher levels in other markers (
Screening performance of biomarkers IL-8, VEGF, CA 19-9, and CEA
To determine the screening potential of IL-8, CEA, VEGF, and CA 19-9, the areas under the ROC curves and the sensitivities at a specificity of about 95% were computed ( Table 4 ). For all four markers, the AUCs were statistically different from chance (p < 0.05). The largest AUC was observed for IL-8 (0.737), followed by CEA (0.677), VEGF (0.611), and CA 19-9 (0.597) ( Figure 3 ). Sensitivities at a specificity of 95% were highest for CEA and IL-8 (both 28%), followed by CA 19-9 (19%) and VEGF (10%). Stage-dependent AUCs showed no differences between biomarkers’ performance. Marker combinations as assessed by logistic regression analyses with correction for overoptimistic performance did not show an advantage compared to IL-8 assessment alone (optimism-corrected performance: AUC = 0.695). However, a marker combination showed superior performance if compared to single analysis of CEA, VEGF, and/or CA 19-9.
Area Under the Receiver Operating Characteristic Curve for All 14 Analyzed Biomarkers
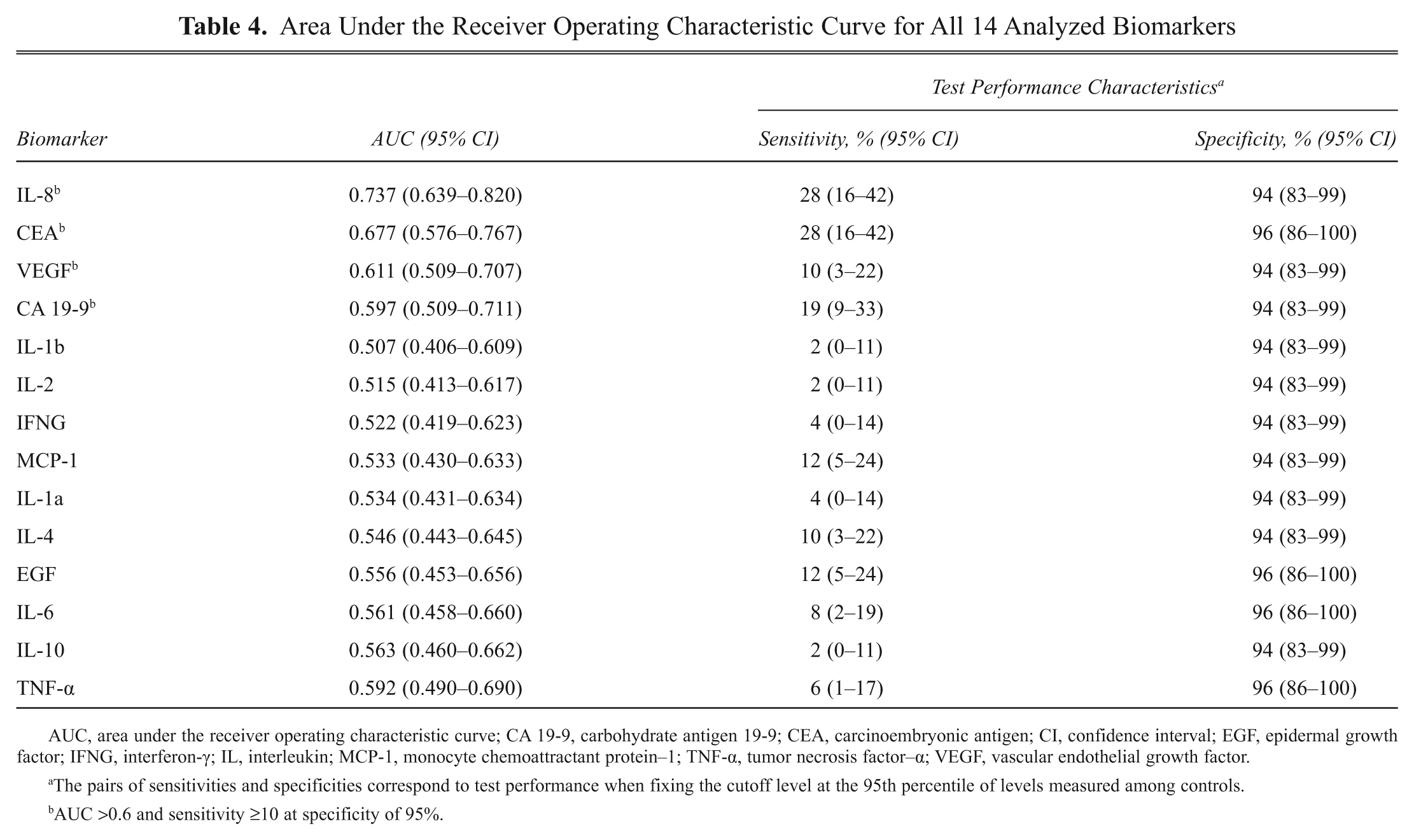
AUC, area under the receiver operating characteristic curve; CA 19-9, carbohydrate antigen 19-9; CEA, carcinoembryonic antigen; CI, confidence interval; EGF, epidermal growth factor; IFNG, interferon-γ; IL, interleukin; MCP-1, monocyte chemoattractant protein–1; TNF-α, tumor necrosis factor–α; VEGF, vascular endothelial growth factor.
The pairs of sensitivities and specificities correspond to test performance when fixing the cutoff level at the 95th percentile of levels measured among controls.
AUC >0.6 and sensitivity ≥10 at specificity of 95%.
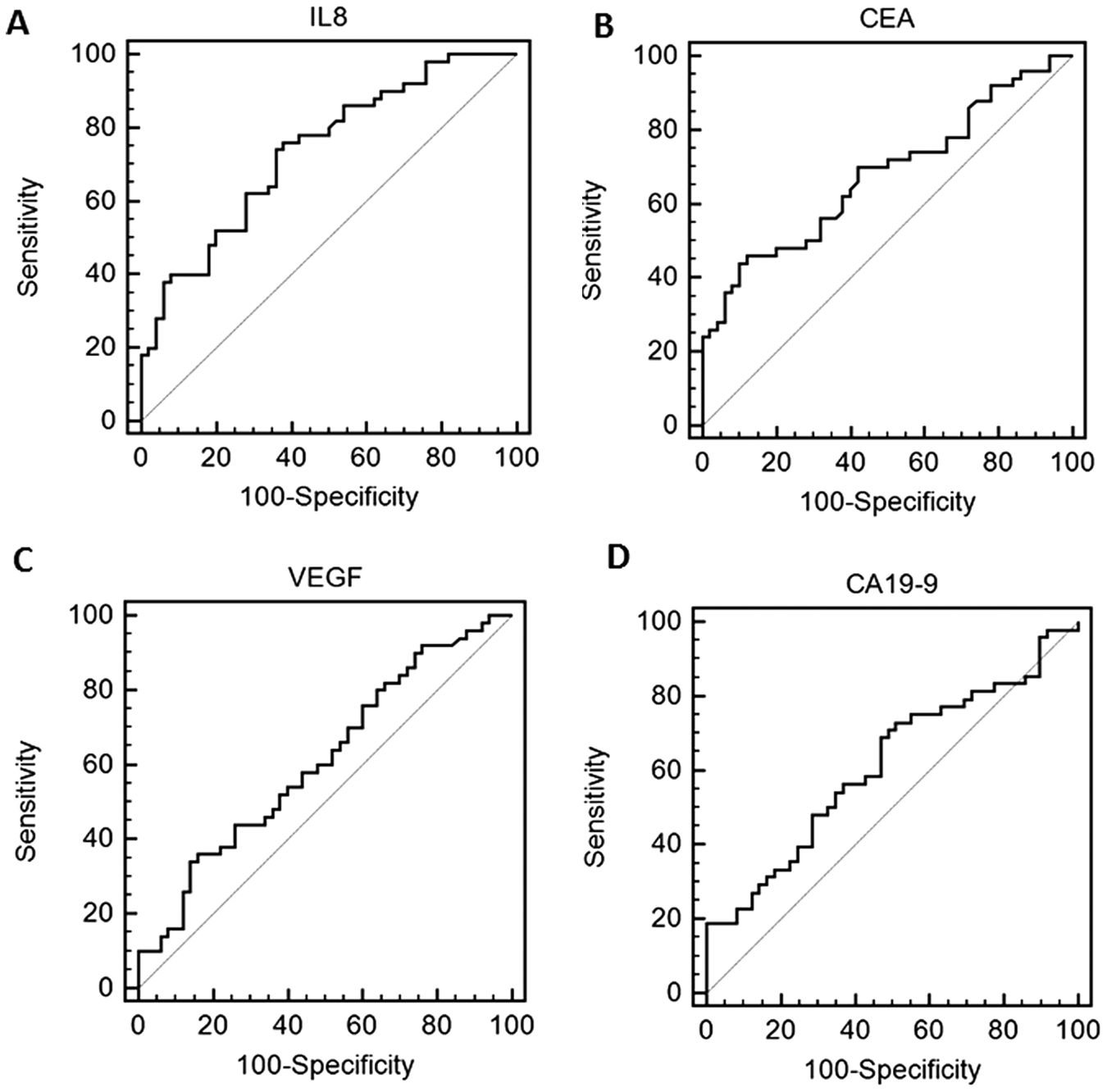
Receiver operating characteristic curve regarding the discrimination between colon cancer and controls for (
Discussion
This is the first study to report the simultaneous assessment of 12 serum cytokines using a multiplex biochip array in comparison to CEA and CA 19-9 to identify clinically applicable serum markers for colon cancer screening in a highly standardized serum sample cohort of colon cancer patients and controls. In our analysis, only IL-8 and CEA showed a significant higher concentration in cancer patient serum in comparison to serum of healthy individuals, whereas IL-8, CEA, VEGF, and CA 19-9 showed diagnostic potential using area under the ROC curve analyses.
IL-8 is a well-known proinflammatory cytokine and important chemoattractant factor for leukocytes. Moreover, it was shown that IL-8 contributes to cancer progression through its potential functions as a motility-stimulating, mitogenic, and angiogenic factor. 25 IL-8 protein levels have been reported to be elevated not only in serum but also in tissue of colon cancer patients: Malicki et al. 17 showed significantly stronger expression of IL-8 in tumor tissue compared to normal adjacent colon mucosa. This observation is in agreement with the study by Rubie et al., 26 who also showed that IL-8 mRNA and protein expression was significantly higher in colorectal tumor tissue when compared to neighboring tissue. The expression of IL-8 in tumor tissue significantly correlated with tumor size, depth of infiltration and liver metastasis, and tumor stage.26,27 Ueda et al. 28 reported that serum IL-6 levels correlated closely with serum IL-8 and CA 19-9 levels. Our results show a similar correlation between IL-6 and IL-8 serum levels but did not find a correlation with CA 19-9.
Similar to IL-8, VEGF is also a pleiotropic cytokine triggering inflammatory, immunoresponse, and cancerogenic effects. VEGF is also a key regulator of angiogenesis and functions as an endothelial cell mitogen. 29 Several studies have demonstrated the importance of angiogenesis during cancer development. In addition, inhibition of angiogenesis in cancer treatment using a VEGF-targeting monoclonal antibody (bevacizumab) seems to improve the outcome of patients with metastatic colorectal cancer. 30
Preoperative serum CEA and CA 19-9 levels are routinely used as markers regarding prognosis, disease-free survival, and therapy response.10,11 The markers are commonly accepted as prognostic, independent predictors to indicate patients with high risk for colon cancer recurrence and/or metastases during postoperative follow-up. 31 However, because of their insufficient sensitivity and specificity at early disease stages (approximately 10%–60%, depending on tumor stage), they are not currently considered as screening markers for clinical routine use.
Interestingly, many of the markers analyzed here had been investigated individually in previous studies. For instance, Berghella et al.
32
analyzed the peripheral blood of 105 colorectal cancer patients using commercially available ELISA kits showing diagnostic and prognostic indices for disease progression of IL-4 and IL-8. Galizia et al.
33
found IL-10 and IL-6 serum levels to be elevated in 30 cancer patients with respect to controls. Chung and Chang,
34
Belluco et al.,
35
and Kinoshita et al.
36
reported also significantly higher IL-6 level in patients with colorectal cancer than in normal controls. Similar results were reported for IL-10, TNF-α, MCP-1, and IFNG.
37
Our study could not confirm these earlier reports (
Having identified IL-8, VEGF, CEA, and CA 19-9 as potential screening markers in serum, we also tried to account for any potential biases of our results. However, we could not find any major influencing factors in terms of sample processing/preservation and/or clinical characteristics. Even though age was significantly higher in cancer patients than in controls (p = 0.03; Table 1 ), no correlation between age and any of the serum marker levels could be detected. To test the overall validity of the multiplex chip assay, IL-8 and VEGF results between the cytokine multiplex array biochip and the commercially available ELISA assay (R&D Systems, Minneapolis, MN) were previously compared using 40 human serum samples. A good correlation was observed for both VEGF and IL-8, with r values >0.95 (unpublished data). A number of independent studies and publications have been completed showing the validity of the Randox Evidence Cytokine products.38-49
The biochip array technology represents a highly standardized technique for cytokine and/ or cancer research. Important and interesting at this point is the simultaneous determination of multiple analytes in a single patient sample, whereas ELISAs are limited to single analyte determinations per sample. Because of this, the economics of consumables and most notably sample volume requirements are much lower for the biochip assay (100 µL total for assaying 12 analytes simultaneously, equal to 8.3 µL per analyte), which is an essential advantage concerning precious clinical samples. In addition, simultaneous detection in a single sample provides valuable information relating to each cytokine under test and possible associations between cytokines in each sample. Besides this, the throughput by a single operator is much higher for the multiplex assay than for ELISAs: In our setup, 84 patient samples can be processed for 12 analytes within 4 h under highly standardized conditions. Automatic software and calibration updates reduce the need for operator input. Furthermore, a scale-up of the multiplex assay to high-throughput is easily possible by using semi- or full-automation Evidence Multiplex Analyzers, which have the capacity to perform a test output in excess of 1200 samples for 12 analytes simultaneously per hour. Thus, this approach should become of increasing interest when considering limited sample volumes, costs, high-throughput, and reproducibility. 50
Although known to be elevated in different solid malignancies, our results suggest that the assessment of IL-8, CEA, VEGF, and possibly CA 19-9 serum levels could be useful also for colorectal cancer screening with the potential of also detecting early stage tumors. The value of these and additional promising markers on a multiplex array format relative to existing screening tools should be validated in prospective trials.
Footnotes
Acknowledgements
We thank Dr. Britta Fritzsche and Timo Gemoll, M.Sc., for advice and Elke Gheribi and Regina Kaatz for assistance with clinical sampling. Grant support: The consortium “Colorectal Cancer Screening Chip” was generously supported by the German Federal Ministry of Education and Research (BMBF) within the Molecular Diagnostics funding scheme (grants 01ES0720, 01ES0721, 01ES0722, and 01ES0723). The study was performed based on the serum collection of the biomaterial bank “ColonBiomics” being part of the Surgical Center for Translational Oncology–Lübeck (SCTO-L) and the North German Tumorbank of Colorectal Cancer network, the latter being generously supported by the German Cancer Aid Foundation (Dt. Krebshilfe e.V., grant 108446).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
