Abstract
To explore the apoptotic and antitumor activities of metallo-salens, the authors have synthesized several Fe(III)-salen and salphen complexes and analyzed their effects on human cancer and noncancer cells. Their results demonstrated that Fe(III)-salen and salphen complexes affect cell viability and induce nuclear fragmentation and apoptosis in breast cancer (MCF7) cells. The IC50 values for the active metallo-salen complexes ranged between 0.3 and 22 µM in MCF7 cells. Biochemically active Fe(III)-salen and salphen complexes induced caspase-3/7 activation and release of cytochrome c from the mitochondria to cytosol, suggesting the involvement of the mitochondrial pathway of apoptosis. Comparison of IC50 values toward 3 different cell lines demonstrated that selected Fe(III)-salen complexes induce tumor cell-selective apoptosis in cultured cells. Overall, the studies demonstrated that Fe(III)-salen and salphen complexes induced efficient apoptosis in cultured human cells. The nature of the substituents and the bridging spacer between diamino groups play critical roles in determining the apoptotic activities of Fe(III)-salen and salphen complexes.
Introduction
T
Metal complexes of salen (bis(salicylidine)ethylenediamine) ligands have been known for a long time. Metallo-salens have been extensively used as catalysts for organic epoxidation. 7 Biological chemists have exploited the oxidative nature of metallo-salens for developing novel chemical nucleases that modify nucleic acids. 8-12 For example, Mn(III)-salen induces DNA cleavage in vitro in the presence of cooxidant magnesium monoperoxyphthalate. 13 Fe(II)-salen and Cu(II)-salen complexes produce free radicals in the presence of reducing agent and cleave DNA in a sequence-neutral fashion. 14 Nickel(II)-salen complexes induce DNA damage/cross-linking under an oxidative environment. 15 Cobalt(II)-salen complexes activate natural oxygen in aqueous solution that induce oxidative DNA damage in vitro. 16 Various metallo-salen complexes are also implicated in hydrolytic cleavage of DNA and RNA. 17,18 Notably, most of these biological studies were focused on analyzing the DNA cleavage/interaction properties of metallo-salens in vitro.
Recently, we demonstrated that Fe(III)-salen not only damages DNA in vitro but also induces efficient apoptosis in vivo. 19,20 We also showed that Fe(III)-salnaphen complexes with more potent DNA cleavage activities induce less efficient apoptosis in cultured human cells. 19 Gust and colleagues 21 showed that diaryl-cobalt salen complexes affect cell viability likely via nonoxidative DNA damage mode of action. Lange et al. 22 demonstrated that Fe(II)-salphen induces selective cytotoxicity toward platinum-resistant ovarian cancer cells. Mn(III)-salen complexes are shown to exhibit synthetic superoxide dismutase (SOD) and catalase activities and therefore are considered synthetic SOD mimics. 12,23 Mn(III)-salen complexes also display cytoprotective features in fibroblast cultures via hydrogen peroxide scavenging. 23 Recently, we demonstrated that Mn(III)-salen complexes induce caspase activation and tumor-selective apoptosis in cultured human cells. 24 The cellular interaction and apoptotic activity of the metallo-salen complexes appeared to be highly dependent on the nature of central metal-ion and substituents present on the salen moiety. 19,20,24
Herein, to understand more detail on the structure and cytotoxicity relationship, we synthesized 14 different Fe(III)-salen and salphen complexes with varying substituents and bridging groups and analyzed their biochemical activities toward different cultured human cancer and nonmalignant cells. Our results demonstrated that several Fe(III)-salen complexes induce caspase-3/7 activation and apoptosis with a nanomolar range of IC50. Selected Fe(III)-salen complexes also induced tumor-selective apoptosis. Importantly, cytotoxicity of the Fe(III)-salen and salphen complexes is highly dependent on the nature and position of the substituents.
Experimental Procedure
Reagents and chemicals
All reagents for organic synthesis and buffers were purchased from Sigma-Aldrich (St. Louis, MO) unless otherwise noted. Tissue culture medium, Dulbecco’s modified Eagle’s medium (DMEM), fetal bovine serum (FBS), penicillin, streptomycin, and antiactin antibody were purchased from Sigma-Aldrich. Ferric chloride (anhydrous) was purchased from Spectrum Chemical Manufacturing Corporation (Gardena, CA). Anti–cytochrome c (monoclonal) antibody was purchased from Upstate Biotech (Billerica, MA), and FITC-conjugated antimouse secondary antibody was obtained from Jackson ImmunoResearch Laboratory (West Grove, PA). DAPI (4′, 6-diamidino-2-phenylindole) was obtained from Invitrogen (Carlsbad, CA). MTT (3-(4, 5-dimethylthiazolyl-2)-2, 5-diphenyltetrazolium bromide) was obtained from Tokyo Chemical Industry Co. (Tokyo, Japan). MCF7, MCF10, and CCL228 cell lines were obtained from American Type Culture Collection (ATCC, Manassas, VA).
Synthesis of Fe(III)-salen and salphen complexes (compounds 1-14)
Fe(III)-salen and salphen complexes were synthesized and characterized following a general procedure as described previously. 20,25 In brief, salen and salphen ligands were synthesized by mixing 2 equivalents of salicylaldehyde (or derivatives) with 1 equivalent of ethylenediamine (or ortho-phenylenediamine) in methanol at room temperature that resulted in yellow- or orange-colored precipitate of respective salen and salphen ligands. Fe(III)-salen and salphen complexes were synthesized by mixing respective ligands with equivalent amounts of anhydrous Fe(III) chloride in methanol followed by heating at 60°C with continuous stirring for 30 min. This resulted in a dark brown/black solution that was cooled down to room temperature. In most cases, the metal complexes were precipitated out upon cooling the reaction mixtures. In some cases, the metal complexes were precipitated by adding diethyl ether into the cold reaction mixtures. The products were isolated, recrystallized from methanol, and characterized by mass spectral (electrospray ionization–mass spectrometry [ESI-MS]) analysis and/or elemental analysis.
Fe(III)-salen chloride and Fe(III)-salphen chloride (compounds 1 and 8)
These 2 compounds were synthesized as described by us previously. 19
3, 3′-Dihydroxysalen Fe(III) chloride (compound 2)
Two equivalents of 2, 3-dihydroxybenzaldehyde were mixed with 1 equivalent of ethylenediamine in methanol that resulted in orange precipitate of 3, 3′-dihydroxysalen that was complexed with 1 equivalent of Fe(III) chloride to obtain compound 2. Observed m/z value (compound 2): 354.20 (M+, -Cl). CHN analysis for compound 2: calculated (for C16H14N2O4FeCl. 1.5 H2O): C 46.13%, H 4.11%, N 6.72%; observed: C 46.32%, H 4.22%, N 6.85%.
4, 4′-Dihydroxysalen Fe(III) chloride (compound 3)
Two equivalents of 2, 4-dihydroxybenzaldehyde were mixed with 1 equivalent of ethylenediamine in methanol that resulted in light orange precipitate of 4, 4′-dihydroxysalen. This ligand was complexed with 1 equivalent of Fe(III) chloride to obtain compound 3. Observed m/z value (compound 3): 354.20 (M+, -Cl). Calculated (for C16H14N2O4FeCl. 0.5 H2O): C 48.21%, H 3.78%, N 7.03%; observed: C 48.11%, H 4.08%, N 6.98%.
5, 5′-Dihydroxysalen Fe(III) chloride (compound 4)
Two equivalents of 2, 5-dihydroxybenzaldehyde were mixed with 1 equivalent of ethylenediamine in methanol that resulted in reddish brown precipitate of 5, 5′-dihydroxysalen. This ligand was complexed with 1 equivalent of Fe(III) chloride to obtain compound 4. Observed m/z value (compound 4): 354.20 (M+, -Cl). CHN analysis: calculated (for C16H14N2O4FeCl. 2.4 H2O): C 44.39%, H 4.38%, N 6.47%; observed: C 44.16%, H 4.33%, N 6.62%.
3, 3′-Dimethoxysalen Fe(III) chloride (5)
Two equivalents of 2-hydroxy-3-methoxybenzaldehyde were mixed with 1 equivalent of ethylenediamine in methanol that resulted in bright yellow precipitate of 3, 3′-dimethoxysalen ligand. This ligand was complexed with 1 equivalent of Fe(III) chloride to obtain compound 5. Observed m/z value (compound 5): 382.20 (M+, -Cl). CHN analysis: calculated (for C18H18N2O4FeCl): C 51.89%, H 4.35%, N 6.72%; observed: C 52.15%, H 4.58%, N 6.93%.
4, 4′-Dimethoxysalen Fe(III) chloride (6)
Two equivalents of 2-hydroxy-4-methoxybenzaldehyde were mixed with 1 equivalent of ethylenediamine in methanol that resulted in orange brown precipitate of 4, 4′-dimethoxysalen ligand. This ligand was complexed with 1 equivalent of Fe(III) chloride to obtain compound 6. Observed m/z value (compound 6): 382.20 (M+, -Cl). CHN analysis: calculated (for C18H18N2O4FeCl. 1.3 H2O): C 49.02%, H 4.71%, N 6.35%; observed: C 48.64%, H 4.46%, N 6.81%.
5, 5′-Dimethoxysalen Fe(III) chloride (7)
Two equivalents of 2-hydroxy-5-methoxybenzaldehyde were mixed with 1 equivalent of ethylenediamine in methanol that resulted in bright yellow precipitate of 5, 5′-dimethoxysalen. This ligand was complexed with 1 equivalent of Fe(III) chloride to obtain compound 7. Observed m/z value (compound 7): 382.20 (M+, -Cl). CHN analysis: calculated (for C18H18N2O4FeCl. 0.2 H2O): C 51.32%, H 4.40%, N 6.65%; observed: C 51.06%, H 4.53%, N 6.88%.
3, 3′-Dihydroxysalphen Fe(III) chloride (9)
Two equivalents of 2, 3-dihydroxybenzaldehyde were mixed with 1 equivalent of o-phenylenediamine in methanol that resulted in red-colored precipitate of 3, 3′-dihydroxysalphen. This ligand was complexed with 1 equivalent of Fe(III) chloride to obtain compound 9. CHN analysis: calculated (for C20H14N2O4FeCl. 2.7 H2O): C 49.40%, H 4.02%, N 5.76%; observed: C 49.08%, H 3.63%, N 5.76%.
4, 4′-Dihydroxysalphen Fe(III) chloride (compound 10)
Two equivalents of 2, 4-dihydroxybenzaldehyde were mixed with 1 equivalent of o-phenylenediamine in methanol that resulted in dark orange red precipitate of 4, 4′-dihydroxysalen. This ligand was complexed with 1 equivalent of Fe(III) chloride separately to obtain compound 10. CHN analysis: calculated (for C20H14N2O4FeCl. 1.8 H2O): C 51.17%, H 3.78%, N 5.97%; observed: C 51.53%, H 4.28%, N 5.57%.
5, 5′-Dihydroxysalphen Fe(III) chloride (compound 11)
Two equivalents of 2, 5-dihydroxybenzaldehyde were mixed with 1 equivalent of o-phenylenediamine in methanol that resulted in dark reddish brown precipitate of 5, 5′-dihydroxysalphen. This ligand was complexed with 1 equivalent of Fe(III) chloride separately to obtain compounds 11. CHN analysis: calculated (for C20H14N2O4FeCl. 1.7 H2O): C 51.30%, H 3.75%, N 5.98%; observed: C 51.28%, H 3.77%, N 5.62%.
3, 3′-Dimethoxysalphen Fe(III) chloride (compound 12)
Two equivalents of 2-hydroxy-3-methoxybenzaldehyde were mixed with 1 equivalent of o-phenylenediamine in methanol that resulted in bright orange red precipitate of 3, 3′-dimethoxysalen ligand. This ligand was complexed with 1 equivalent of Fe(III) chloride to obtain compound 12. CHN analysis: calculated (for C22H18N2O4FeCl. 3 H2O): C 50.89%, H 4.66%, N 5.39%; observed: C 50.63%, H 4.0%, N 5.28%.
4, 4′-Dimethoxysalphen Fe(III) chloride (compound 13)
Two equivalents of 2-hydroxy-4-methoxybenzaldehyde were mixed with 1 equivalent of o-phenylenediamine in methanol that resulted in bright orange precipitate of 4, 4′-dimethoxysalphen ligand. This ligand was complexed with 1 equivalent of Fe(III) chloride to obtain compound 13. CHN analysis: calculated (for C22H18N2O4FeCl): C 56.74%, H 3.89%, N 6.01%; observed: C 56.62%, H 4.04%, N 6.02%.
5, 5′-Dimethoxysalphen Fe(III) chloride (compound 14)
Two equivalents of 2-hydroxy-5-methoxybenzaldehyde were mixed with 1 equivalent of o-phenylenediamine in methanol that resulted in light red precipitate of 5, 5′-dimethoxysalen. This ligand was complexed with 1 equivalent of Fe(III) chloride to obtain compound 14. CHN analysis: calculated (for C22H18N2O4FeCl): C 56.74%, H 3.89%, N 6.01%; observed: C 56.2%, H 4.08%, N 5.93%.
Cell viability assay and IC50 measurements
Human breast cancer (MCF7), colon cancer (CCL228), and breast epithelial (MCF10) cells were grown and maintained in DMEM that was supplemented with 10% heat-inactivated FBS, 2 mM L-glutamine, and penicillin/streptomycin (100 units and 0.1 mg/mL) as described previously. 26 The cytotoxicity of Fe(III)-salen and salphen complexes was determined by using the MTT assay as described previously. 24 In brief, approximately 10,000 cells were seeded into each well of a 96-well microplate and incubated 24 h in 150 µL DMEM. An additional 50 µL DMEM containing the required amount of each metallo-salen to obtain 0.05 to 60 µM final concentration (in 200-µL final volume) in 5 replicate wells was added. Control wells were treated with equivalent amount of DMSO. After 96 h of incubation, 20 µL of MTT (5 mg/mL) was added into each well, and cell viability was assayed by measuring the formazan absorption at 560 nm as described previously. 27 The absorbance (at 560 nm) values were plotted against concentration of metallo-salens to determine the IC50. The concentration of the metal complexes at which the conversion of MTT to formazan by viable cells is reduced by 50% compared to control cells is defined as the IC50. The experiments were repeated twice with 5 replicates each time.
Nuclear staining and cytochrome c immunostaining
MCF7 cells (grown on a cover slip) were incubated with 100 µM Fe(III)-salen or salphen complexes for 24 h. Control cells were treated with an equivalent amount of DMSO. Cells were fixed with 4% formaldehyde, washed twice with phosphate-buffered saline (PBS), permeabilized with 0.2% Triton X-100, washed again, and incubated with DAPI (5 µg per slide) for 10 min as described previously. 24 DAPI-stained cells were mounted on a microscope slide and visualized under a fluorescence microscope. For the immunostaining with anti–cytochrome c, MCF7 cells (on cover slip) were treated with 100 µM Fe(III)-salen and salphen complexes separately for varying time periods and subjected to immunostaining with anti–cytochrome c antibody as described by us previously. 24
Caspase-3/7 activity assay
For the caspase-3/7 activation assay, CCL228 cells were treated with 100 µM Fe(III)-salen and salphen complexes for 0, 8, 16, and 24 h; lysed; and centrifuged (2500 g for 10 min at 4°C). The supernatant was diluted (to 1 µg/µL of protein). Caspase-3/7 activity was assessed by using the SensoLyte Homogeneous AMC Caspase-3/7 Assay Kit (AnaSpec, Fremont, CA) as described previously. 24 Briefly, 150 µL of extract was mixed with 50 µL of assay buffer containing caspase-3/7 substrate and incubated for 15 min at 37°C, and then fluorescence intensity (ex: 350; em: 440) was measured at every 10-min interval (up to 2 h). The concentration of activated caspase-3/7 was calculated by using the calibration curve and finally expressed relative to untreated control cells.
Results and Discussion
Cytotoxicity of Fe(III)-salen and salphen complexes
To investigate the structure-activity relationship and analyze the biochemical activities of metallo-salen complexes, we synthesized 14 different Fe(III)-salen and salphen complexes by introducing different functional groups and bridging spacers in the salen ligand (compounds 1-14;
Fig. 1
).
24
Each metal-complex was characterized by mass spectrometry and/or elemental analysis, and the results are consistent with the proposed structures. Initially, we determined the cytotoxicity of different Fe(III)-salen and salphen complexes toward a breast cancer cell line, MCF7. In brief, MCF7 cells were incubated with varying concentrations of the complexes separately for 96 h and then subjected to the MTT assay. The percent of viable cells (relative to the control) were plotted as a function of concentration of the Fe(III)-salen and salphen complexes (
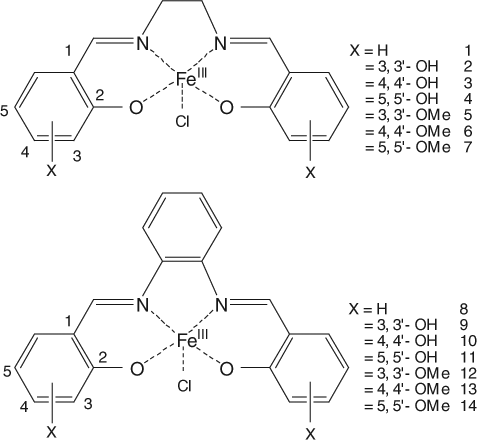
Structures of the Fe(III)-salen (1-7) and salphen (8-14) complexes.
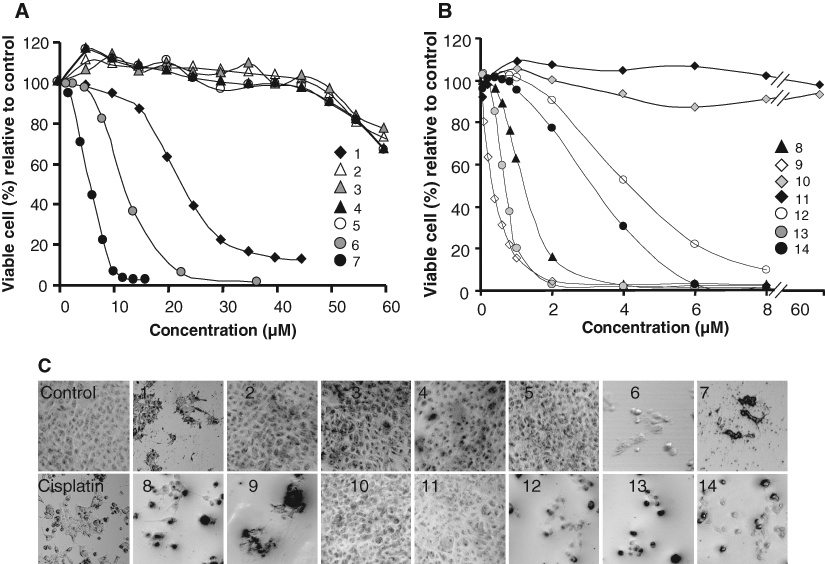
Cytotoxicity measurements of Fe(III)-salen and Fe(III)-salphen complexes. Curves 1 to 14 in
IC50 Values of Fe(III)-Salen and Salphen Complexes toward Different Cancer and Noncancer Cells
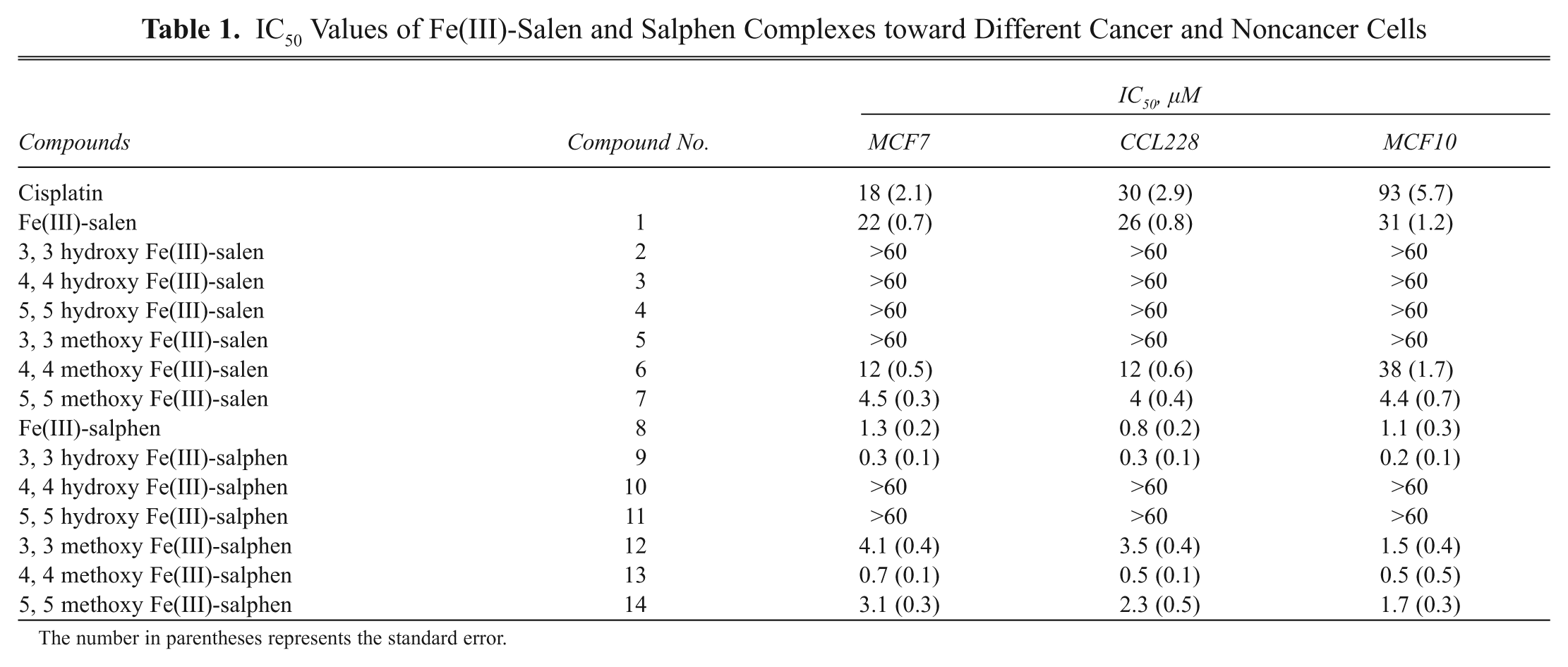
The number in parentheses represents the standard error.
Our results demonstrated that treatment with several Fe(III)-salen and salphen complexes affected cell viability efficiently toward MCF7 cells ( Fig. 2 and Table 1 ). For example, among the salen derivatives, treatment with Fe(III)-salen and 4, 4′- and 5, 5′-dimethoxy substituted Fe(III)-salen complexes (compounds 1, 6, and 7, respectively) induced efficient cell death with IC50 values of 22, 12, and 4.5 µM, respectively, for MCF7 cells ( Fig. 2A and Table 1 ). In contrast, 3, 3′-dimethoxy Fe(III)-salen (compound 5) and any of the hydroxy-substituted Fe(III)-salen derivatives (compounds 2-4) did not exhibit a significant effect on cell viability (IC50 > 60 µM; Fig. 2A and Table 1 ). In the case of Fe(III)-salphen complexes, all the methoxy-substituted derivatives (compounds 12-14) as well as 3, 3′-dihydroxy Fe(III)-salphen (compound 9) were efficient in inducing cell death on MCF7 cells (IC50s are 4.1, 0.7, 3.1, and 0.3 µM, respectively; Fig. 2B and Table 1 ). Notably, 4, 4′- and 5, 5′-dihydroxy-substituted Fe(III)-salphen complexes (compounds 10 and 11) did not have any effect on cell viability (IC50 > 60 µM; Fig. 2B and Table 1 ). Interestingly, among the effective compounds, the IC50 values for 3, 3′-dihydroxy Fe(III)-salphen (compounds 9) and 4, 4′-dimethoxy-Fe(III)-salphen (compound 13) were in the nanomolar range (330 and 680 nM, respectively) toward MCF7 cells ( Table 1 ).
Microscopic observation showed that treatment with biochemically active Fe(III)-salen and salphen complexes (compounds 1, 6-9, and 12-14) induced distinct morphological changes (such as clumping, rounding up, and generation of cell debris) in MCF7 cells ( Fig. 2C ). The cells treated with noneffective compounds (compounds 2-5, 10, and 11) were grown as healthy as control cells ( Fig. 2C ). These observations further demonstrated that compounds 1, 6-9, and 12-14 induced efficient cell death in MCF7 cells. Notably, as seen under microscope, treatment with cisplatin also induced efficient cell death in MCF7 cells. The IC50 value of cisplatin toward MCF7 cells was determined to be 18 µM ( Table 1 ), and these data are comparable with the reported IC50 value (13 µM) of cisplatin toward MCF7 cells. 27,28
Overall, cytotoxicity analysis and microscopic observations demonstrated that several Fe(III)-salen and salphen complexes are highly effective in inducing cell death in MCF7 cells. The nature of substituents (hydroxy vs. methoxy) and the bridging spacers (ethylenediamine vs. orthophenelynediamine) play critical roles in determining the cytotoxicity of Fe(III)-salen and salphen complexes. The changes in the ethylenediamine bridge to o-phenylenediamine increased the cytotoxicity of Fe(III)-salen complexes. For example, unmodified Fe(III)-salen (compound 1) has an IC50 of 22 µM, whereas corresponding Fe(III)-salphen (compound 8) has an IC50 of 1.3 µM toward MCF7 cells ( Table 1 ). 3, 3′-Dihydroxy Fe(III)-salen (compound 2) is inactive (IC50 > 60 µM), whereas corresponding 3, 3′-dihydroxy Fe(III)-salphen (compound 9) is highly active (IC50: 0.3 µM; MCF7, Table 1 ). Similarly, 3, 3′-dimethoxy Fe(III)-salen (compound 5) is inactive (IC50 > 60 µM), whereas corresponding 3, 3′-dimethoxy Fe(III)-salphen (compound 12) is highly active (IC50: 4.1 µM; MCF7, Table 1 ). Notably, this trend of cytotoxicity does not follow for several other hydroxy and methoxy salen derivatives, suggesting that additional factors may be contributing toward the cytotoxicity of Fe(III)-salen and salphen complexes ( Table 1 ). In general, methoxy-substituted Fe(III)-salen and salphen were more biochemically active than corresponding hydroxy-substituted derivatives with some exceptions ( Table 1 ). For example, 4, 4′- and 5, 5′-dimethoxy-substituted Fe(III)-salen and salphen complexes were more active than corresponding hydroxy-substituted derivatives ( Table 1 ). Although analyses of the cytotoxicity data suggest the existence of some degree of structure-activity relationship, these cannot be generalized for a wide range of compounds. The ultimate cytotoxicity of metal complexes depends on diverse factors such as mode of cellular interaction, transport/cellular uptake potential, intrinsic structure, and reactivity of the compounds. 14,21
Cytotoxicity of Fe(III)-salen and salphen complexes toward different cancer and normal cell lines
To further explore cell selectivity and antitumor potential, we analyzed the IC50 values of Fe(III)-salen and salphen complexes toward 3 different cell lines: MCF7 (breast cancer), CCL228 (colon cancer), and MCF10 (nonmalignant breast epithelial cells). Our results demonstrated that most of the Fe(III)-salen and salphen complexes killed both malignant (MCF7 and CCL228) and nonmalignant (MCF10) cells efficiently ( Table 1 ). Interestingly, compounds 1 (Fe(III)-salen) and 6 (4, 4’-dimethoxy Fe(III)-salen) showed some preference in killing cancer cells over nonmalignant cell. 4, 4’-Dimethoxy Fe(III)-salen killed breast and colon cancer cells about 3-fold more efficiently than nonmalignant breast epithelial cells ( Table 1 ). These data suggest that compounds 1 and 6 have potential toward novel antitumor application.
Herein, we examined only 3 different cell lines. However, for complete evaluation of the antitumor activities, these studies need to be extended further toward various other types of cell lines. Furthermore, as several Fe(III)-salphen complexes (such as compounds 9 and 13) have a nanomolar range of IC50 values and many effective antitumor drugs have nanomolar IC50 values, these compounds have potential toward novel antitumor application. Notably, IC50 value for cisplatin in MCF7 cells is measured to be ~18 µM, which suggests that Fe(III)-salen and salphen complexes are comparable or even more effective in inducing cell death in vitro.
Fe(III)-salen and salphen complexes induce nuclear fragmentation
To examine the nature of cell death induced by various Fe(III)-salen and salphen complexes, we analyzed their effects on nuclear integrity by using DAPI staining. 29 We treated MCF7 cells with 100 µM of Fe(III)-salen and salphen complexes separately for 24 h, stained them with DAPI, and visualized them under a fluorescence microscope. Interestingly, in agreement with cell viability data (in Fig. 2 ), all the biochemically active Fe(III)-salen and salphen complexes (compounds 1, 6-9, and 12-14) showed significant effects on nuclear morphology and resulted in nuclear condensation (intense DAPI staining) and fragmentation in most of the cell population ( Fig. 3 ). The single-cell expanded image is shown in Supplementary Figure S1. The effects were more severe in some cases in comparison to others, likely because of their difference in efficiencies. The treatment with cisplatin also induced similar nuclear morphological changes, as seen in the case of Fe(III)-salen and salphen complexes ( Fig. 3 ). The change in nuclear morphology, nuclear condensation, and fragmentation indicates that Fe(III)-salen and salphen complexes induced apoptotic cell death in MCF7 cells.
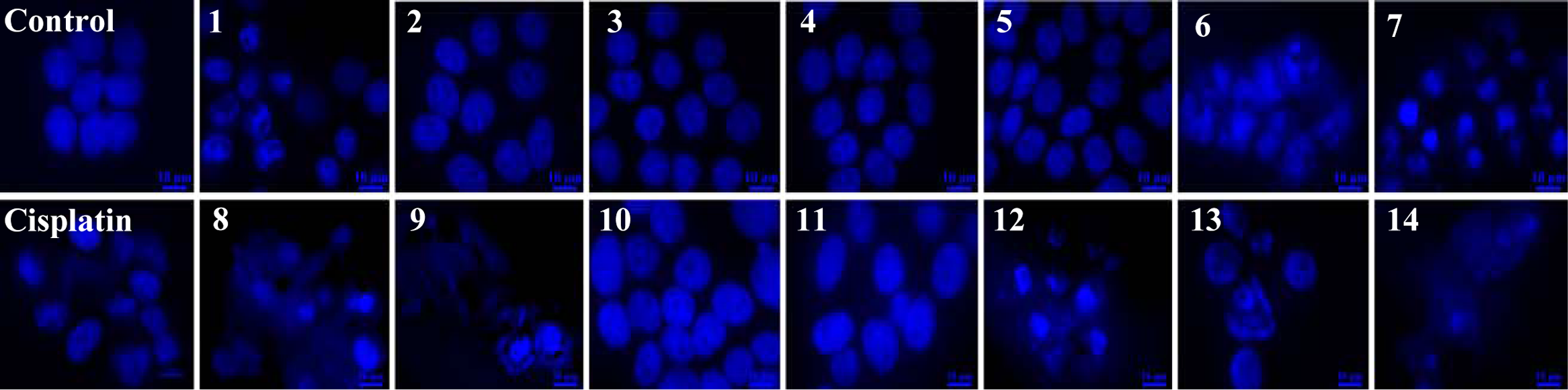
Effect of Fe(III)-salen derivatives on nuclear integrity (DAPI staining). MCF7 cells, grown on a cover glass in a 60-mm culture plate for 24 h, were treated alone or with 100 µM Fe(III)-salen and salphen complexes separately for 24 h, fixed with formaldehyde, and stained with DAPI, followed by visualization under a fluorescence microscope. Control panel indicates cells that were treated with DMSO. Panels 1 to 14 represent cells treated with compounds 1 to 14, respectively. Cisplatin was used as a positive control.
Fe(III)-salen and salphen complexes induce release of cytochrome c from the mitochondria to cytosol
To further understand the pathway by which Fe(III)-salen derivatives induced apoptosis in MCF7 cells, we performed immunostaining with cytochrome c antibody before and after treatment with Fe(III)-salen derivatives. 30 The cytochrome c immunostained cells were visualized under a fluorescence microscope, and the corresponding cell nucleus was visualized by DAPI staining ( Fig. 4 ). As expected, cytochrome c is localized inside the mitochondria in control untreated cells, as evidenced by the speckles outside the nucleus ( Fig. 4 ). However, treatment with biochemically active Fe(III)-salen and salphen complexes (compounds 1, 6-9, and 12-14) resulted in release of cytochrome c from mitochondria to the cytosol, as evidenced by its spreading throughout the cell in the majority of cell populations. In contrast, inactive compounds (compounds 2-5, 10, and 11) showed no significant effect on cytochrome c translocation. The single-cell expanded images of cytochrome c immunostaining are shown in Supplementary Figure S2. Notably, cytochrome c is a mitochondrial enzyme that is closely associated with metabolic pathways and stress response. It is well known that cytochrome c is normally present inside the mitochondria. However, upon induction of apoptosis, mitochondrial membrane potential is perturbed, which results in release of cytochrome c from the mitochondria to cytosol, which ultimately leads to caspase-9 activation followed by caspase-3/7 activation and apoptotic cell death. 31 Thus, the release of cytochrome c from the mitochondria to cytosol upon treatment with various biochemically active Fe(III)-salen and salphen complexes suggested that they induced apoptosis in MCF7 cells via the mitochondrial pathway.
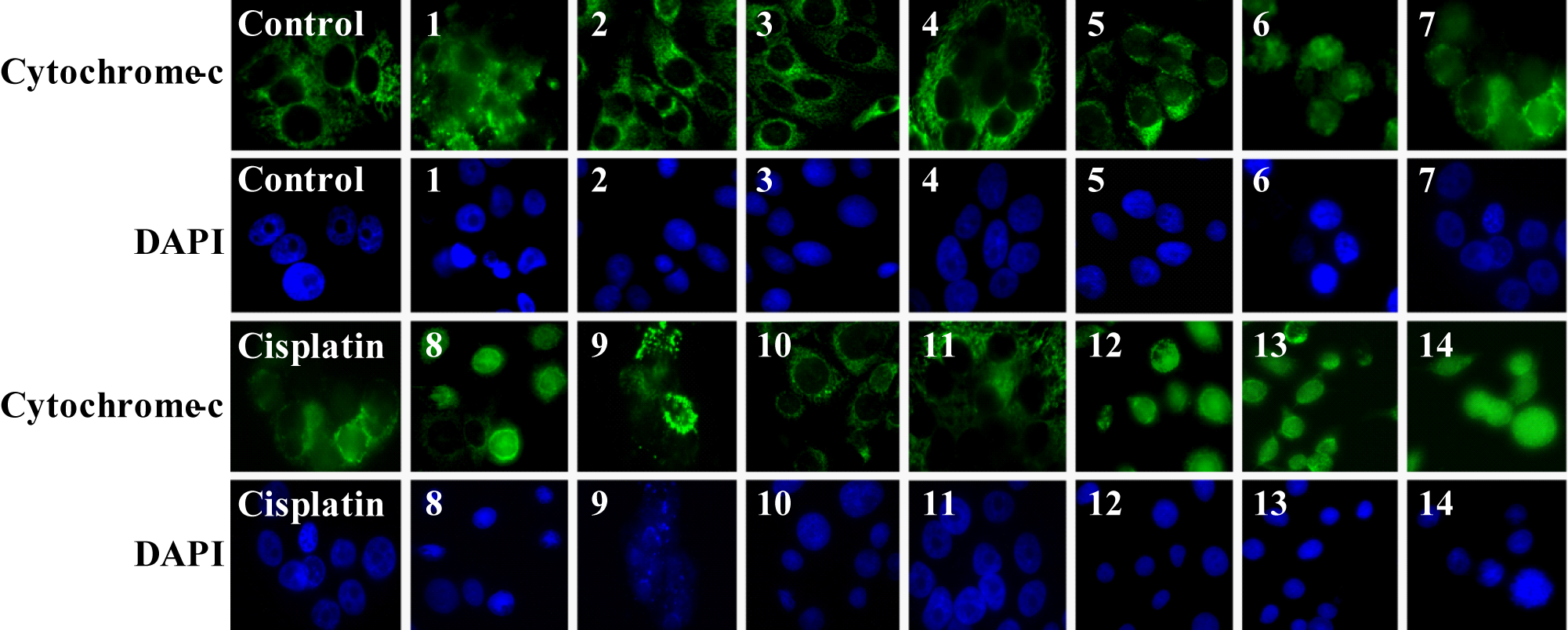
Cytochrome c translocation induced by Fe(III)-salen and salphen complexes. MCF7 cells that were treated with 100 µM metallo-salen complexes were immunostained with anti–cytochrome c and FITC-conjugated secondary antibodies and visualized under a fluorescence microscope. The corresponding cell nucleus was visualized by DAPI staining. Control panels: cell treated with DMSO; panels 1 to 14 represent cells that were treated with compounds 1 to 14, respectively. Cisplatin was used as a positive control.
Casapse-3/7 activation by Fe(III)-salen and salphen complexes
Caspase activation is central to all apoptotic cell death. 31 In the mitochondria-initiated pathway of apoptosis, caspase activation is triggered by the formation of a multimeric Apaf-1/cytochrome c complex that results in activation of procaspase-9. 31 Activated caspase-9 then cleaves and activates downstream caspases such as caspase-3, -6, and -7 that ultimately activates endonucleases, resulting in nuclear fragmentation and apoptosis. Herein, to further confirm that Fe(III)-salen and salphen complexes induced apoptosis in cultured cells, we analyzed the caspase activation (specifically caspase-3/7) in Fe(III)-salen- and salphen-treated cells. Notably, we performed most of our cytotoxicity and biochemical studies using MCF7 cells. However, it is well known that MCF7 cells lack caspase-3. 32 Therefore, we analyzed the caspase-3/7 activities in colon cancer cells (CCL228) and correlated their cytotoxicity values in the same cell line. In brief, CCL228 cells were treated with 100 µM of Fe(III)-salen and salphen complexes (compounds 1-14) separately for varying time periods (0, 8, 16, and 24 h) and then subjected them to caspase-3/7 activation assay using a commercial caspase-3/7 assay kit (Sensolyte; AnaSpec). The caspase-3/7 activities of the cells treated with active Fe(III)-salen and salphen complexes increased with time with a maxima at ~16 h and then declined likely due to cell death (data not shown). The relative caspase-3/7 activation by different active Fe(III)-salen and salphen complexes at a given time point (at 16 h posttreatment) was plotted ( Fig. 5 ). Our results demonstrated that compounds (compounds 2-5, 10, and 11) with low cytotoxicity toward CCL228 cells (IC50s: over 60 µM; Table 1 ) did not activate caspase-3/7 significantly (data not shown). However, active compounds (such as compounds 1, 6-9, and 12-14; IC50s: 26, 12, 4, 0.8, 0.3, 3.5, 0.5, and 2.3 µM, respectively, toward CCL228 cells; Table 1 ) showed about 2- to 10-fold caspase-3/7 activation in comparison to the control untreated cells ( Fig. 5 ). The Fe(III)-salphen complexes (compounds 9 and 13) with nanomolar ranges of IC50 (0.3 and 0.5 µM) induced the highest caspase-3/7 activation (about 11- and 9-fold, respectively). Overall, the caspase-3/7 activities correlated very well with their cytotoxicity values (correlation coefficient, 0.83). These observations suggest that Fe(III)-salen and salphen complexes induced apoptosis in CCL228 cells via caspase activation.
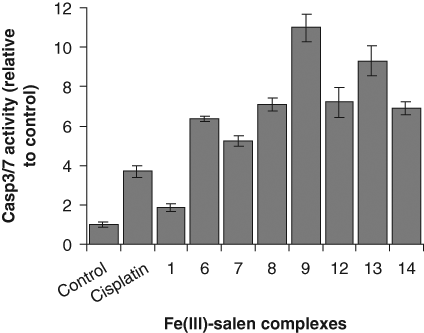
Fe(III)-salen and salphen complexes activate caspase-3/7. CCL228 (human colon cancer) cells were treated with 100 µM Fe(III)-salen and salphen complexes for 16 h and subjected to caspase-3/7 activation assay. The relative caspase-3/7 activity (compared to the untreated control) was plotted for different active Fe(III)-salen and salphen complexes. Bars indicate standard error.
For a comparative study in a different cell line (nonmalignant MCF10), we analyzed the effects of a less active compound (such as compound 6; 4, 4-methoxy Fe(III)-salen, IC50: 38 µM toward MCF10) and a highly active compound (such as compound 9; 3, 3’-dihydroxy Fe(III)-salphen, IC50: 0.2 µM) on nuclear integrity, cytochrome c release, and caspase-3/7 activation. As expected, compound 9 shows more severe effects on MCF10 cells in comparison to compound 6 ( Fig. 6 ). These results are consistent with observation in MCF7 cells.
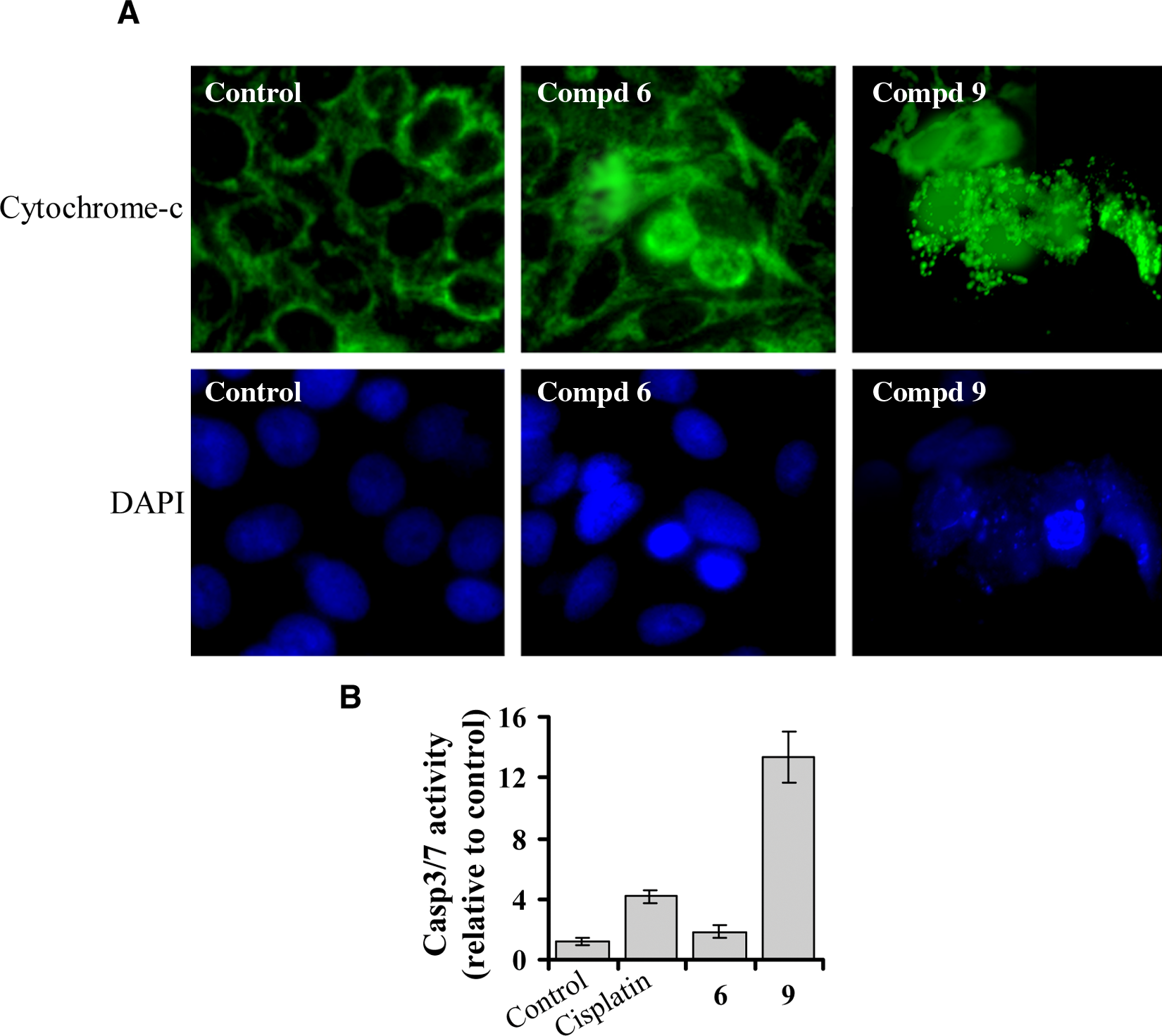
Comparison of apoptotic activity of compound 6 (4, 4′-dimethoxysalen Fe(III) chloride) and compound 9 (3, 3′-dihydroxysalphen Fe(III) chloride) toward nonmalignant breast epithelial cells (MCF10). (
Conclusion
Overall, our studies demonstrated that Fe(III)-salen and salphen complexes induce apoptosis in cultured human cells via the mitochondrial pathway and caspase activation. IC50 values for the biochemically active Fe(III)-salen and salphen complexes ranged from 0.3 to 22 µM. The nanomolar IC50 values for the selective Fe(III)-salphen complexes suggest that these compounds are very effective in killing cells. Although a detailed mechanism still remains elusive, our studies demonstrated that the nature of the substituents, the bridging spacers, and the central metal ion play key roles in determining the apoptotic activities and efficiencies of the metallo-salen derivatives. The changes in the ethylenediamine bridge to o-phenylene diamine increased apoptotic activities of Fe(III)-salen complexes. In general, methoxy-substituted Fe(III)-salen and salphen complexes were more biochemically active than corresponding hydroxy-substituted derivatives (with some exceptions) likely because of easy permeation through the cell membrane. Selected Fe(III)-salen derivatives induced preferential apoptosis in breast and colon cancer cells over normal nonmalignant breast epithelial cells, indicating their potential toward novel antitumor application.
Interestingly, however, when we compare the level of caspase-3/7 activation for compounds 6 (4, 4-methoxy Fe(III)-salen) and 9 (3, 3′-dihydroxy Fe(III)-salphen) with reference to cisplatin toward CCL228 and MCF10 cells lines, it is obvious (see Figs. 5 and 6B ) that compound 6 shows more caspase-3/7 activation than cisplatin in the malignant CCL228 cells but less caspase-3/7 activation than cisplatin in the nonmalignant MCF10 cells. In contrast, compound 9 shows similar activity with respect to cisplatin (about a 3-fold increase in activity) in both cell lines ( Figs. 5 and 6B ). These observations suggest that compound 6 might be a safer compound and may provide a better lead structure for exploring chemotherapeutic application than compound 9. Although our studies indicate potentials of metallo-salen complexes toward novel antitumor application, further studies are needed for detailed evaluation of these compounds toward various other types of cancer and normal cells. The mechanism of action of metallo-salen complexes, their biological targets, and mode of entry into the cell still remain elusive.
Footnotes
Acknowledgements
We thank Saoni Mandal and other lab members for helpful discussion. This work was supported in parts by grants from ARP (00365-0009-2006) and the American Heart Association (0765160Y).
