Abstract
Bone marrow mesenchymal stem cells (MSCs) promote nerve growth and functional recovery in animal models of spinal cord injury (SCI) to varying levels. The authors have tested high-content screening to examine the effects of MSC-conditioned medium (MSC-CM) on neurite outgrowth from the human neuroblastoma cell line SH-SY5Y and from explants of chick dorsal root ganglia (DRG). These analyses were compared to previously published methods that involved hand-tracing individual neurites. Both methods demonstrated that MSC-CM promoted neurite outgrowth. Each showed the proportion of SH-SY5Y cells with neurites increased by ~200% in MSC-CM within 48 h, and the number of neurites/SH-SY5Y cells was significantly increased in MSC-CM compared with control medium. For high-content screening, the analysis was performed within minutes, testing multiple samples of MSC-CM and in each case measuring >15,000 SH-SY5Y cells. In contrast, the manual measurement of neurite outgrowth from >200 SH-SY5Y cells in a single sample of MSC-CM took at least 1 h. High-content analysis provided additional measures of increased neurite branching in MSC-CM compared with control medium. MSC-CM was also found to stimulate neurite outgrowth in DRG explants using either method. The application of the high-content analysis was less well optimized for measuring neurite outgrowth from DRG explants than from SH-SY5Y cells.
Keywords
Introduction
M
The reported mechanisms responsible for the functional improvements noted in animal SCI models following MSC transplantation vary considerably. However, MSCs are known to synthesize and secrete factors that promote neuronal survival and axonal outgrowth. 3,4 The extent to which such soluble neurotrophic factors are synthesized by MSCs and the effects that MSC transplantation has on functional improvement in SCI vary considerably between bone marrow donors. 4,5 Indeed, the extent of functional improvement after MSC transplantation may be attributed, in part, to the soluble neurotrophic factors synthesized by the transplanted MSC grafts. Hence, a preclinical method to determine the capacity of human MSCs to synthesize neurotrophic factors would provide a useful tool in screening for optimized MSC transplantation therapies. For this purpose, we have tested the capacity of human MSC-conditioned medium (MSC-CM) to stimulate neurite outgrowth in vitro, comparing high-content and previously published manual analysis. 6-8
Materials and Methods
MSC-conditioned medium
Following ethical approval and informed consent, bone marrow was harvested from the iliac crest of 4 donors. MSCs were culture expanded from these aspirates and serum-free MSC-CM generated as described previously. 8
Preparing neuronal cultures for analysis
SH-SY5Y cells
SH-SY5Y cells were seeded in serum-free Dulbecco’s modified Eagle’s medium (DMEM)/ITS-X medium in 96-well tissue culture plates (Sarstedt, Hannover, Germany) at a density of 5000 cells per well. Plates were incubated at 37 °C for 24 h to permit cell adherence and then fed with MSC-CM or fresh serum-free nonconditioned (control) medium in triplicate. Following a 48-h incubation, digitized images were captured using phase contrast microscopy for manual neurite outgrowth analysis.
Chick dorsal root ganglia (DRG) explants
DRG explants were seeded into 24-well culture plates that had been coated with type I collagen as described, 8 which contained either MSC-CM or control medium. Following a 48-h incubation, digitized images of cultures were captured using phase contrast microscopy for manual neurite outgrowth analysis.
Neurofilament 200-kD immunostaining
SH-SY5Y cells and DRG explants were fixed by gently adding an equal volume of 20% (v/v) formalin to the culture medium for 10 min. Wells were extensively washed with immunobuffer (0.05% bovine serum albumen and 0.1% Triton X-100) twice for 10 min. Plates were blocked (in immunobuffer containing 10% goat serum; Sigma-Aldrich, St. Louis, MO) prior to incubation with antibodies specific for neurofilament 200 kD (1:200; clone NE14, Sigma-Aldrich). Immunopositivity was revealed using an antimouse Alexa Fluor 488 antibody (1:100; Invitrogen Life Technologies, Carlsbad, CA), counterstaining for nuclei with Hoechst 33342 (Invitrogen Life Technologies).
High-content screening
The Cellomics Arrayscan platform and neuronal profiler algorithm (Cellomics Ltd, Pittsburgh, PA) was used to quantitate neurite area coverage, neurite length, and neurite branching. Multiple fields of view were taken in each well, and each field of view was captured in 2 fluorescence channels. Channel 1 captured Hoechst nuclear staining, whereas channel 2 captured neurofilament 200-kD immunostaining. The main parameter settings of the algorithm were optimized for SH-SY5Y and DRG explant cultures; these parameters are outlined in Table 1 , and there derivation is detailed below. The algorithm operates by identifying nuclei based on their size. For SH-SY5Y, cell nuclei were generally larger than in the DRG explant cultures. As such, an increased nuclear segmentation value was used for SH-SY5Y cultures compared to DRG explant cultures. In both, the nuclei were clearly defined by Hoechst staining and therefore required no nuclear smoothing (blurring). A rejection threshold was added so that objects under a certain size, such as nuclear debris or dead cells with blebbing nuclei, were excluded from analysis. A maximum threshold was also used for nuclear area such that large objects over a certain size, representing cell clumps or debris, were excluded from the analysis.
Main Parameter Settings of the Algorithm That Have Been Optimized for SH-SY5Y and Dorsal Root Ganglia (DRG) Explant Cultures
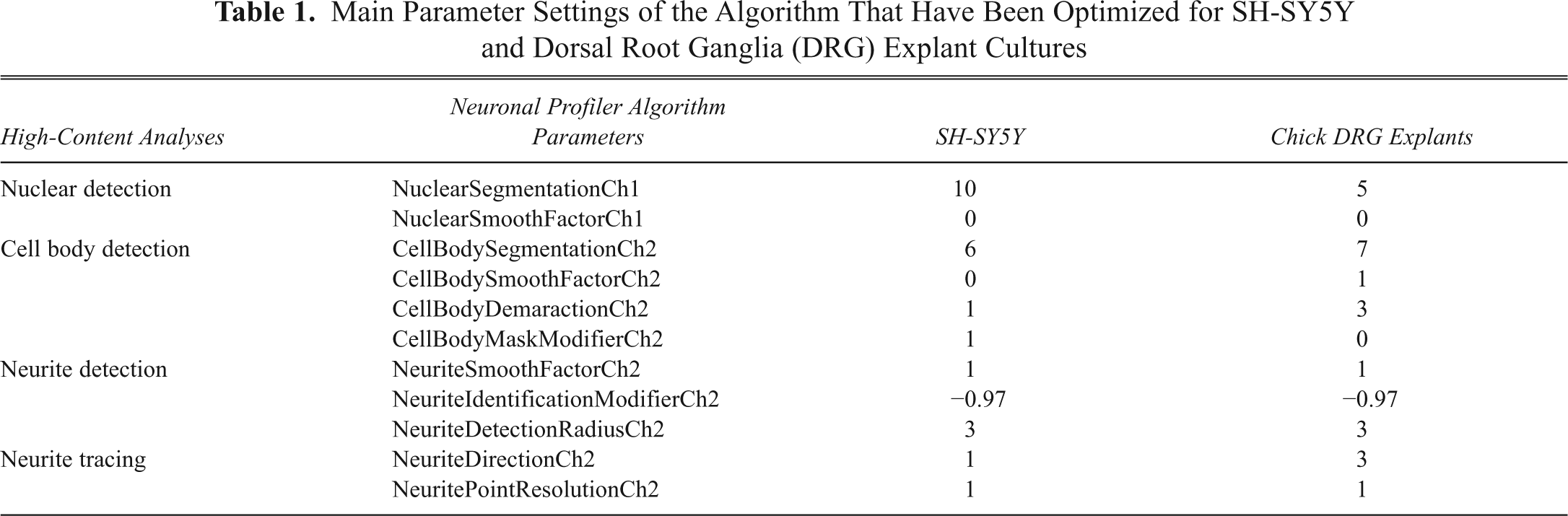
Upon identification of valid nuclei in channel 1, the associated channel 2 neurofilament staining was analyzed. Cell body segmentation and smoothing parameters were optimized similarly to nuclear detection. For cell body detection, the algorithm measured changes in fluorescence over a continuous signal so that intensity peaks and troughs were used to separate cell bodies and associate them to their respective nuclei. Boundaries between cell bodies and neurites were controlled by the cell body demarcation setting. In this setting, a low value would create large cell bodies that may encompass neurites, whereas high values would result in smaller cell bodies where the outer regions of these cells are interpreted as neurites. This setting therefore required precise optimization to differentiate between cell bodies and neurites for each neuronal culture. The cell body mask modifier was used to alter the cell body area so that it was larger than that calculated using the cell body demarcation setting for SH-SY5Y cultures. In SH-SY5Y cultures, tiny projections that may or may not have been true neurites were then in effect swallowed by the enhanced cell body mask. Minimum and maximum size rejection thresholds were also added so that cell clumps or debris would be excluded from the analysis.
For neurite detection, the “box” method was employed. This is different from the detection of nuclei and cell bodies due to the relatively dim staining of neurites in comparison. The box detection method involves replacing each pixel value in an image with the mean value of that pixel and its neighboring pixels, thus computing the mean value of the pixels in that region for amplification purposes. For neurite analysis, low levels of neurite smoothing were applied to the algorithm to ensure that adjacent neurites were not merged into single neurites. The neurite identification modifier was optimized for maximal sensitivity for neurites while rejecting any background “noise” that could be interpreted as neurites. The neurite detection radius was used to define the width of neurites; to avoid classifying multiple adjacent neurites as a single neurite, a low setting was applied to this parameter in both cultures.
Neurite tracing affects the ability of the algorithm to untangle crossed neurites and assign them to their correct cell bodies. Neurites were traced from their cell bodies to their tips, and any branch points were added as they were detected. The neurite direction parameter was used to specify the number of pixels required to determine changes in neurite direction at branch points. For the cell line SH-SY5Y (where neurites were generally shorter), a lower value was required for this parameter compared to DRG explant cultures where longer neurites required larger values. The neurite point resolution setting represents the minimum number of pixels between each adjacent branch point. This parameter was set to its lowest in each culture in order for the algorithm to sensitively determine whether 2 branch points originated from the same neurite or if there was crossover between neighboring neurites. The neurite algorithm mask traced neurites according to these parameter settings and was colored pink, light blue, and green for each cell detected on a cyclical basis to clarify their origin.
Manual analysis of neuronal cultures
As for the high-content analysis, neurite outgrowth was quantitated in all cultures after 48 h in MSC-CM or control medium. Cultures were monitored by phase contrast microscopy (Nikon Eclipse TS100). For SH-SY5Y cells, previously described methods 7 were adapted for analysis using IP Lab software (Version 3.6, Becton Dickinson Biosciences, Franklin Lakes, NJ). Briefly, 5 digitized images were collected with a Hamamatsu digital camera (C4742-95) from each well by moving dishes in an identical pattern. Neurites were defined as a cell process ≥10 µm long; these neurites were manually traced, and their lengths were automatically measured from pixels/µm calibrations using the IP Lab software. The mean length of those neurites was then calculated per cell. The digitized images were also used to determine the percentage of SH-SY5Y cells with neurites (scoring >200 cells/well). Furthermore, the number of neurites per cell was scored but only for the subpopulation of SH-SY5Y cells that had at least 1 neurite.
For wells seeded with DRG explants, phase contrast images were taken of the entire explant, and the neurite outgrowth was measured following methods previously described. 8 Briefly, neurites with an identifiable growth cone were traced back along the neurite length toward the explant until a clear path could not be distinguished from surrounding networks. At this point, a straight line was drawn back to the nearest outer edge of the DRG explant. These paths were automatically measured from pixels/µm calibrations using IP Lab. The mean length of those neurites was then calculated per DRG explant.
Statistical analysis
The Mann-Whitney U test was used to assess significant differences at the 95% confidence level between the quantity, area, length, and number of branches for neurites assessed using high-content analysis versus manual analysis and between each of these parameters when neuronal cultures are grown in MSC-CM versus control medium.
Results and Discussion
Human MSCs have been reported to increase neurite outgrowth from human neuroblastoma SH-SY5Y cells in co-culture. In these co-cultures, MSCs were grown in tissue culture inserts, and as such, the observed neurotrophic activity was attributed to their synthesis of soluble neurotrophic factors. 5 In this study, using both high-content and manual analysis, we have similarly demonstrated that MSC-CM stimulated neurite outgrowth from SH-SY5Y cells compared to cells grown in control medium. For high-content analysis, a set of images captured from channels 1 and 2 was generated and analyzed after the application of an optimized neuronal profiler algorithm mask. High-content image sets for the SH-SY5Y neuronal analysis in MSC-CM compared with control medium are shown in Figure 1A . Images for manual analysis of the same cultures and their hand-traced neurite overlays are shown in Figure 1B .

High-content analysis versus manual analysis of SH-SY5Y stimulated by mesenchymal stem cell–conditioned medium (MSC-CM). (
Treatment with MSC-CM was shown to increase the percentage of SH-SY5Y cells that extended neurites by ~200% when examined using either the high-content neuronal profiler algorithm or using manual analysis. There was no significant difference in the increase in the percentage of SH-SY5Y cells that had neurites when quantitated using either method (
Fig. 2A
). The high-content analysis demonstrated that culturing SH-SY5Y cells in MSC-CM also significantly increased the mean number of neurites per cell (
Fig. 2B
). In contrast, although the same trend was evident, there was no significant difference in the mean number of neurites per cell in MSC-CM versus control medium when quantitated using the manual method (
Fig. 2C
). Of those SH-SY5Y cells that had formed neurites, there was no significant difference in the mean length of those neurites when cultures were grown in either MSC-CM or control medium, as determined by both methods of analysis (
Fig. 2D
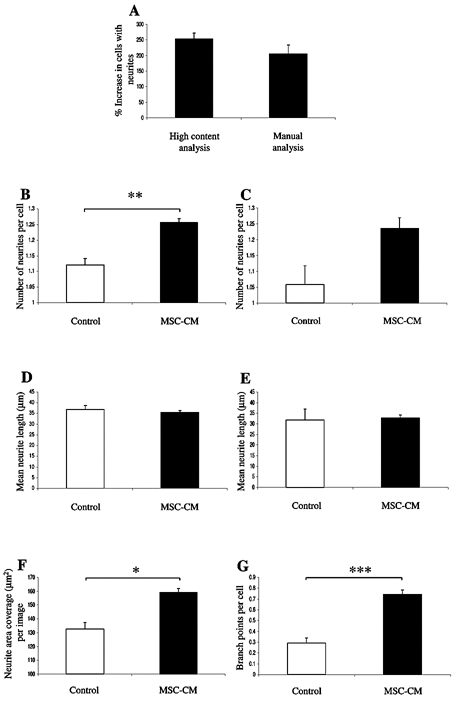
Quantitation of neurite outgrowth from SH-SY5Y cells stimulated by mesenchymal stem cell–conditioned medium (MSC-CM) using high-content or manual analyses. (
MSC-CM has also been previously shown to increase neurite outgrowth from DRG explants,
4,8
where a number of neurotrophic agents in the MSC-CM were identified.
4
Similarly, in this study, MSC-CM also stimulated neurite outgrowth from DRG explants compared to DRG explants, which were cultured in control medium. Representative high-content image sets for DRG explant analysis in MSC-CM versus control medium are shown in
Figure 3A
. Representative images for manual analysis of the same cultures and their hand-traced neurite overlays are shown in
Figure 3B
. Application of the neuronal profiler algorithm demonstrated that there was a significant increase in total neurite area coverage per DRG explant and in the total number of neurite branch points per DRG explant in MSC-CM compared to DRG explants in control medium (
Fig. 3C
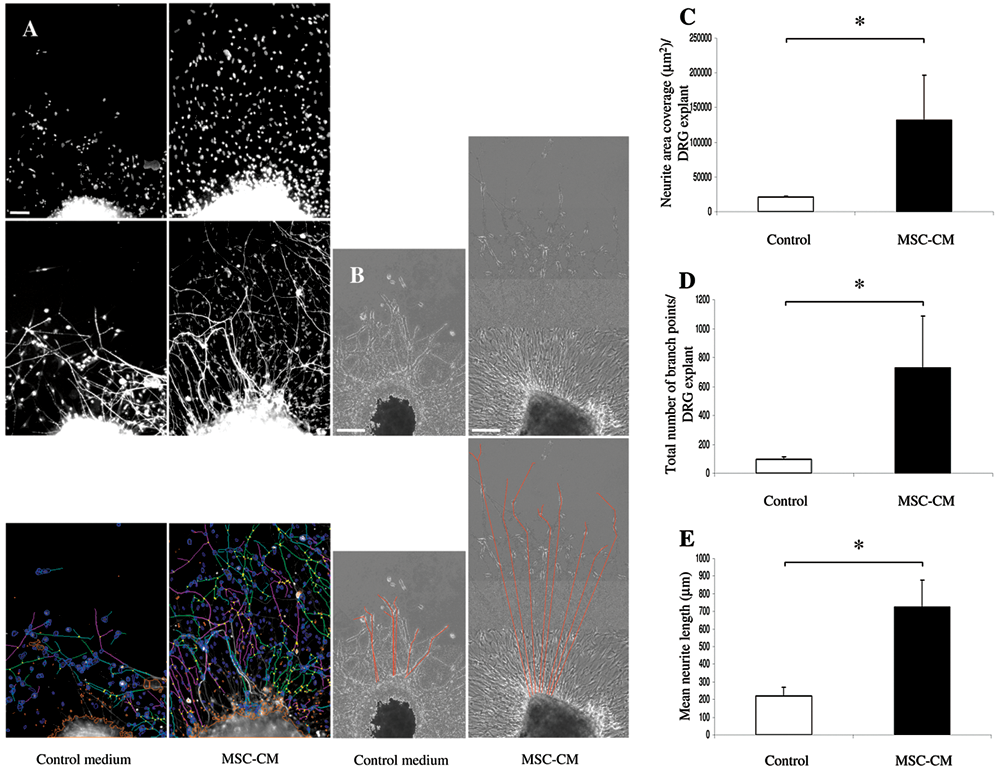
High-content analysis versus manual analysis of chick dorsal root ganglia (DRG) explant cultures stimulated by mesenchymal stem cell–conditioned medium (MSC-CM). (
The amount of BDNF, VEGF, Il-6, SCF, and SDF1a present in MSC-CM has been shown to vary considerably between MSC donors and also within donor MSC subpopulations. 4,5 Furthermore, the degree of functional recovery in SCI models following MSC transplantation also varies. 4 In this study, we have identified a high-throughput means for testing the capacity of MSC-CM to stimulate neurite outgrowth. The potential of this high-content analysis may have application to the development of rapid preclinical screening techniques for optimized MSC transplantation for the treatment of SCI.
Footnotes
Acknowledgements
This study was funded by the Biotechnology and Biological Sciences Research Council (BBSRC).
