Abstract
Several monoclonal antibodies (McAbs) have been developed that show high sensitivity and specificity to gastric cancer and colorectal cancer. However, few of the antigens recognized by these antibodies have been identified. The authors now report the selection of anti-idiotype (anti-id) antibodies of MGb1 McAb against gastric cancer and MC5 McAb against colorectal cancer using phage-displayed single-chain variable fragment (ScFv) libraries. After purification, the anti-id antibodies were approximately 30 kd and could be recognized by MGb1/MC5 McAb. Anti-id antibodies significantly blocked the binding of MGb1 and MC5 to gastric cancer/colorectal cancer cells, respectively, suggesting that the antibodies were specific to MGb1 and MC5. Antibodies against gastric and colorectal cancer could be detected in mice at 6 weeks after immunization with the anti-id antibodies. At week 8, antibody titers reached 1:400. The anti-id antibodies may be useful as novel reagents for developing vaccines against gastric cancer and colorectal cancer.
Keywords
Introduction
I
Materials and Methods
Construction of the single-chain variable fragment phage display libraries
MGb-1 and MC5 McAb, preserved at the Institute of Digestive Disease, the Fourth Military Medical University, Xi’an China, were conjugated with KLH and complete Freund’s adjuvant (Sigma, St. Louis, MO) and were used to immunize Balb/C mice on days 0 and 7. Mice were boosted with the same reagents on days 14, 28, 42, and 56. Spleens from mice with high titers of antibody against corresponding McAb were used for mRNA extraction with the RNA isolation kit (Sigma).
The purified mRNA was reversely transcribed into cDNA using random primers. The VH (IgG variable heavy chain) and VL (IgG variable light chain) DNA fragments were separately amplified by PCR (30 cycles: 94 °C × 1 min, 55 °C × 2 min, 72 °C × 2 min). The PCR products were purified with agarose gel and then mixed with linker primers at an equimolar ratio and assembled into ScFv DNA by PCR (7 cycles: 94 °C × 1 min, 63 °C × 4 min). Afterward, the ScFv DNA was amplified, with primers incorporating the SfiI site at the 5′ end and a NotI site at the 3′ end. The ScFv DNA was then ligated into phagemid vector pCANTAB5E (Amersham Biosciences, Piscataway, NJ) after digestion with SfiI and NotI. The recombinants were electrotransformed into Escherichia coli TG1 and incubated for 1 h at 37 °C in 2× YT medium containing ampicillin and glucose (2× YT-AG medium). Bacteria were infected with M13K07 helper phage for 1 h at 37 °C with shaking. After centrifugation, the bacteria were gently resuspended in 2× YT medium containing ampicillin and kanamycin (2× YT-AK medium) and incubated overnight at 37 °C with shaking. The supernatant containing the recombinant phage was harvested by centrifugation, and the phages were precipitated with PEG8000 and NaCl and resuspended in 2× YT medium, filtered through a 0.45-µm filter and stored at 4 °C.
Biopanning of the phage ScFv libraries with MGb1 and MC5 antibodies
The filtered phages were incubated with blocking buffer for 15 min and then added to culture flasks coated with MGb1/MC5 McAb and incubated for 2 h at 37 °C. The supernatant was removed and the flasks were washed with phosphate-buffered saline (PBS) 3 times. Then E. coli TG1 at the log phase was added to the flasks and incubated at 37 °C for 1 h. Ampicillin, glucose, and M13K07 helper phage were added and incubated for 1 h. The bacteria were harvested and resuspended with 2× YT2AK medium and incubated overnight. Phages in the supernatant were then harvested and filtered as described above and underwent the next round of panning. After 4 rounds of panning, the infected bacteria were plated onto SOBAG agar plates and grown overnight.
Selection of the phages expressing the anti-MGb1/MC5 ScFv by enzyme-linked immunosorbent assay
Colonies from the plates were picked and inoculated into individual tubes. Supernatant from each tube was screened with 96-well plates coated with either MGb1 or MC5. Murine IgG was used as negative control. Horseradish peroxidase (HRP)–conjugated anti-M13K07 phage monoclonal antibody was used as the secondary antibody. Tetramethylbenzidine (TMB) substrate was used for visualization.
Soluble expression, purification, and characterization of the anti-MGb1/MC5 ScFv
Phages from positive colonies identified by enzyme-linked immunosorbent assay (ELISA) were used to infect E. coli HB2151. Periplasmic proteins were prepared and examined by dot blotting. The ScFv fragments were then purified with the RPAS purification kit (Amersham Biosciences) and characterized by sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE) and Western blot.
Inhibition of MGb1/MC5 McAb binding to gastric/colorectal cancer cells by anti-idiotype antibodies
We added 1 × 105 KATO III cells (gastric cancer cell line) or SW480 cells (colorectal cancer cell line) to a 96-well plate after washing with PBS and then centrifuged them at 1200 rpm for 15 min. Then, 0.25% glutaraldehyde diluted with PBS was used to fix the cells for 12 min. Cells were then washed 3 times by PBS and then blocked with 3% bovine serum albumin (BSA). MGb1 McAb/MC5 McAb (final concentration of 40 µg/mL) were mixed with purified corresponding ScFv proteins at different concentrations and then added to each well. After incubation at room temperature for 1 h, the cells were washed with PBS buffer containing 0.1% Tween-20. HRP-conjugated goat antimouse antibody was used as the secondary antibody. TMB substrate was used for visualization. Purified ScFv proteins from nonscreened phages were used as negative controls.
Immunization of mice with purified ScFv and detection of antibody titer by cell ELISA
Balb/c mice were immunized with purified ScFv proteins of MGb1 or MG7 conjugated with complete Freund’s adjuvant by intraperitoneal injection, and mice immunized with only complete Freund’s adjuvant were set as the negative control group (10 mice in each group). Mice were boosted at day 14. Murine sera were prepared at weeks 5, 6, 7, and 8 and diluted 1:100. KATO III and SW480 cell plates were prepared and blocked as described above in the inhibition assay. The diluted sera were added to the wells and incubated at room temperature for 2 h. After washing 3 times with PBS, HRP-labeled goat antimouse antibody was then added and incubated at room temperature for 1 h. To detect peroxidase activity, 200 µL of ortho-phylenediamine (OPD) substrate solution was added to each well and incubated for 10 min at room temperature followed by addition of 50 µL 3 M HCl to stop the reaction. The plates were read at 450 nm on a multiwell plate reader. To determine the titer of the antibodies, murine sera at week 8 were diluted 1:100, 1:200, 1:400, and 1:800 and underwent cell ELISA assay as described above. To further verify the specificity of the antibodies induced by anti-id proteins, another cell ELISA assay was performed using the GES-1 cell, an immortalized human gastric epithelial cell line that does not express the MC5 or MGb1 antigen. For all ELISA assays, triplicate wells were used for each serum sample from the mice.
Results
Selection of anti-MGb1/MC5 McAb antibodies
After 4 rounds of panning, 132 colonies positive for MGb1 and 134 colonies positive for MC5 were identified by ELISA. Twenty-five MGb1-positive clones and 21 MC5-positive clones that showed the highest specificity to corresponding antibodies and did not respond to the normal murine IgG (as suggested by the ELISA assay; data not shown) underwent enzyme digestion with SfiI and NotI. Insertion of the ScFv fragments into the phage was confirmed in all the clones, which was further verified by sequencing.
Protein purification and characterization
Twenty-five MGb1-positive clones and 21 MC5-positive clones were induced to express soluble ScFv. The periplasmic proteins were prepared and tested for affinity to MGb1 McAb or MC5 McAb by dot blotting ( Fig. 1 ). Clones that showed the best affinities for MGb1 (A3, A4, A5) and MC5 (C7, D6, D8) were selected to express ScFv proteins. The periplasmic proteins were then purified. By SDS-PAGE, the ScFv proteins were approximately 30 kd in size (see Fig. 2 ). Western blot assay suggested that the purified SvFv proteins could be recognized by MGb1 or MC5 monoclonal antibody but not by normal murine IgG1 ( Fig. 3 ).
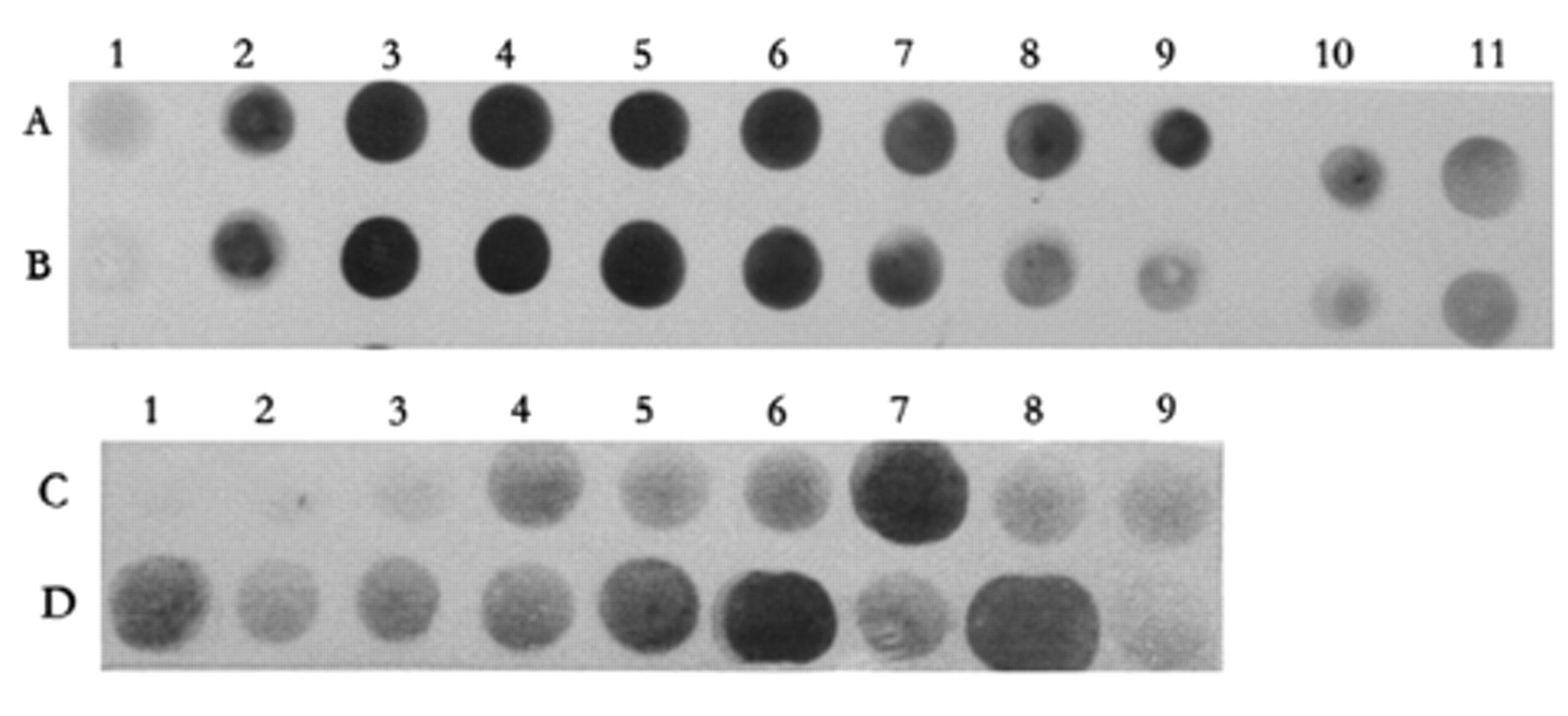
Typical pictures of testing phages binding to MGb1 and MC5 by dot blotting. A1-A11 and B2-B11 were MGb1 anti-idiotype phage-displayed clones selected by biopanning, and B1 was the negative control. C2-D9 was an MC5 anti-idiotype phage-displayed clone selected by biopanning, with C1 being the negative control.
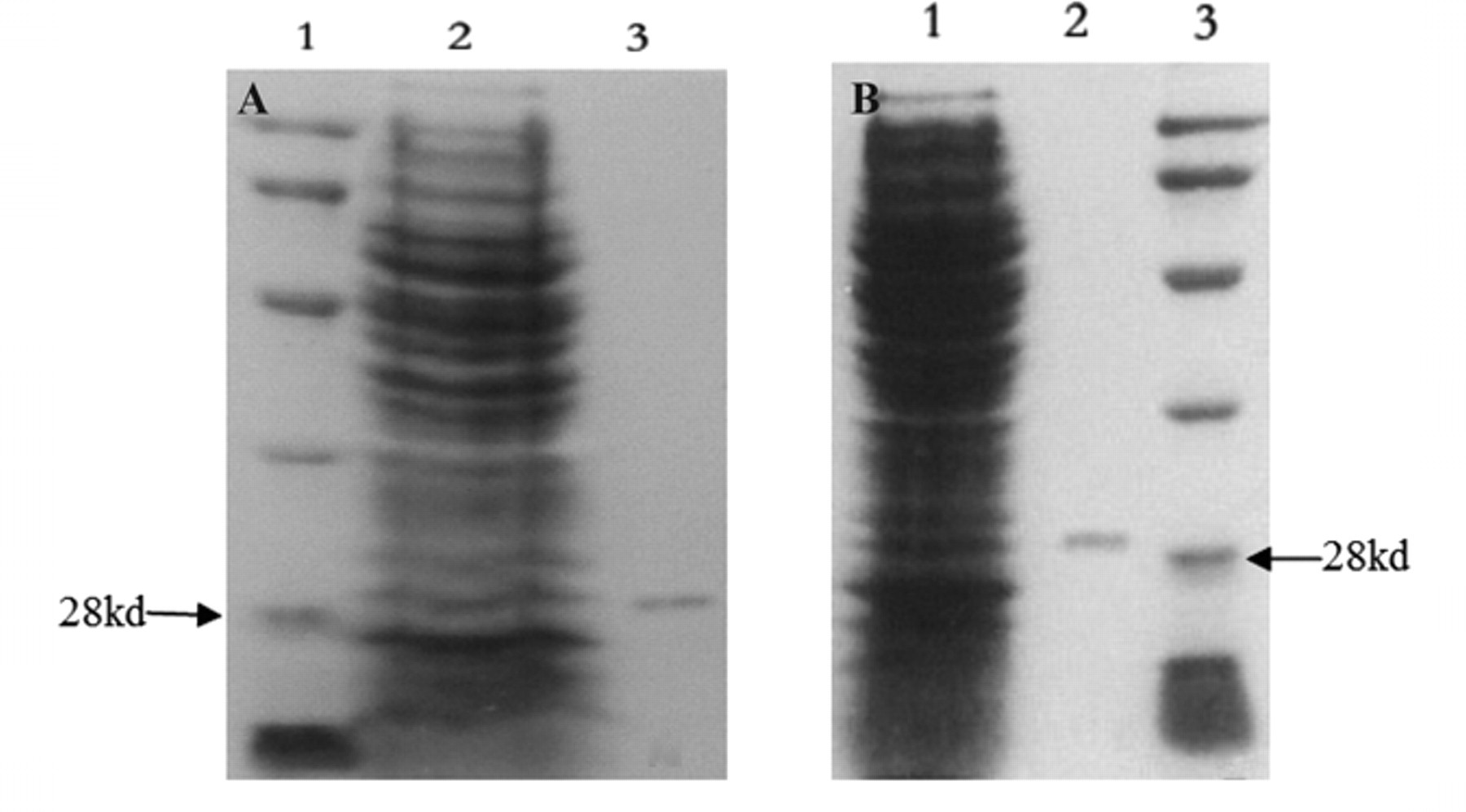
SDS-PAGE examination of the purified ScFv proteins. Escherichia coli HB2151 was infected with phages positive for MGb1 or MC5 and induced to express soluble ScFv proteins. Periplasmic proteins were prepared and loaded onto sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE). (
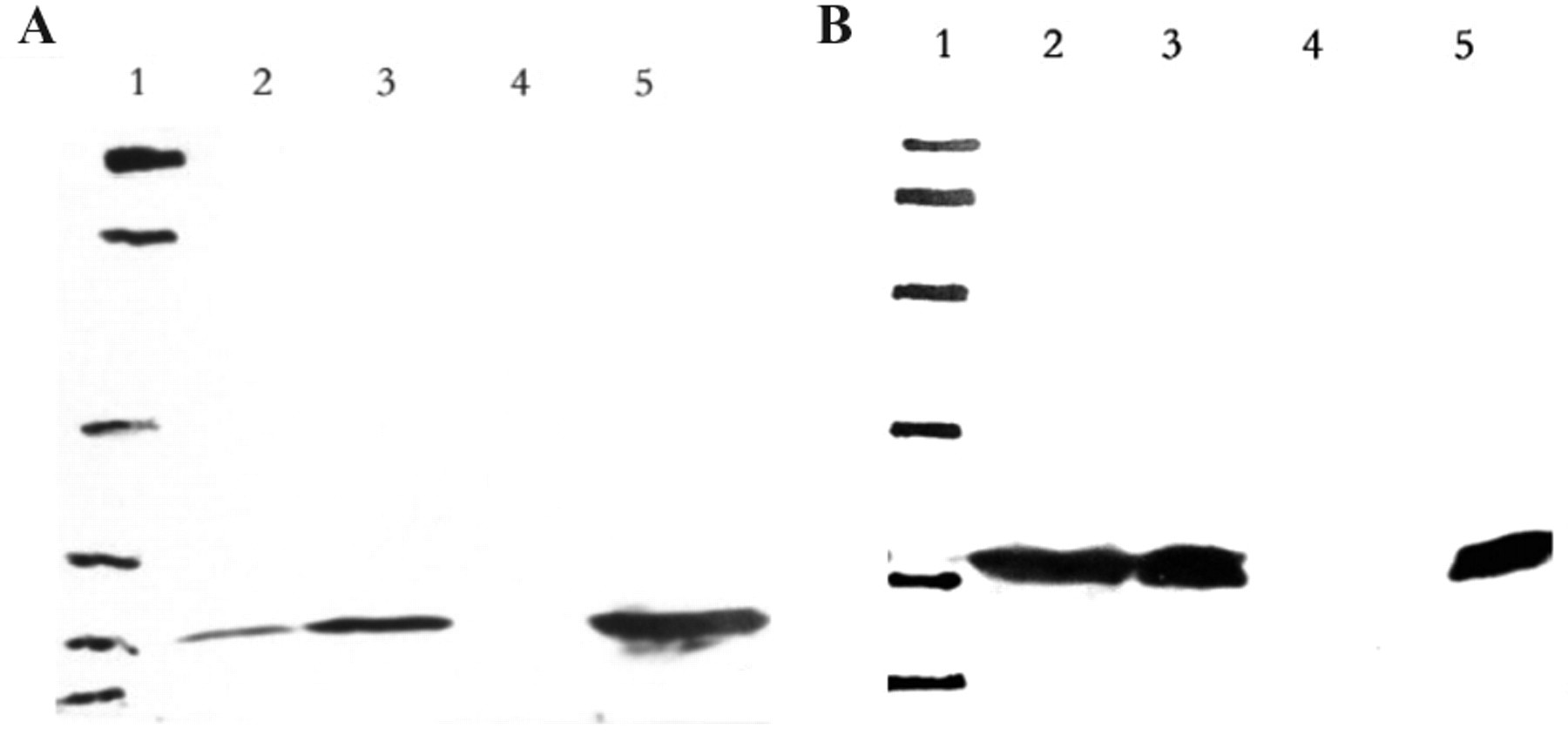
Western blot detection of the purified anti-idiotype antibodies. Purified ScFv proteins were detected by MGb1/MC5 McAbs by Western blot, and IgG1 murine negative control (which is negative for human antigens) was used as negative control. (
Inhibition of MGb1/MC5 McAb binding to gastric/colorectal cancer cells by anti-idiotype antibodies
To further test the specificity of the purified ScFv proteins, cell ELISA competition assays were performed. As shown in Figure 4 , the anti-idiotype antibodies significantly blocked the binding of MGb1/MC5 to gastric/colorectal cancer cells but had no effect on other McAbs. These findings suggested that the anti-idiotype antibodies were specific for MGb1 and MC5.
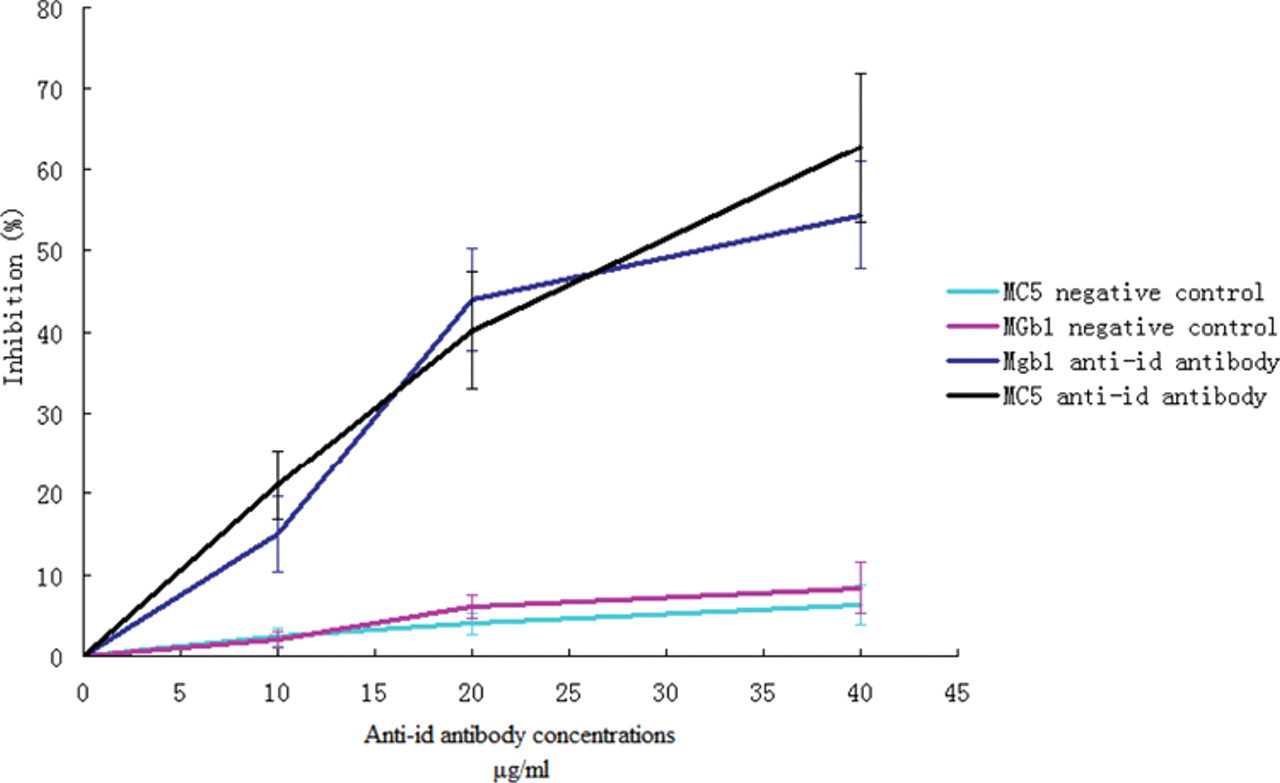
Inhibition of McAbs binding to cancer cells by anti-idiotype antibodies. ScFv proteins from A5 for MGb1 and C7 for MC5 were used in the cell enzyme-linked immunosorbent assay (ELISA) competition assay. Proteins isolated from phages that were not screened were used for gastric cancer and colorectal cancer assays. Inhibition ratio was calculated as follows: Inhibition ratio = (OD control – OD test)/OD control × 100%.
Immunization of mice and detection of the antibody titer
To detect the immunogenicity of the purified anti-idiotype antibodies, we immunized mice with purified ScFv proteins. Murine sera were prepared at different time points, and cell ELISA assays were performed to detect the antibody titers. As shown in Figure 5 , antibody against gastric/colorectal cancer cells increased as time prolonged. At week 8, antibodies against gastric cancer/colorectal cancer could be detected at 1:400 dilution ( Fig. 6 ). The result of cell ELISA using GES-1 cells is presented in Figure 7 . No reaction with GES-1 cells was detected for sera from either immunized or nonimmunized mice.
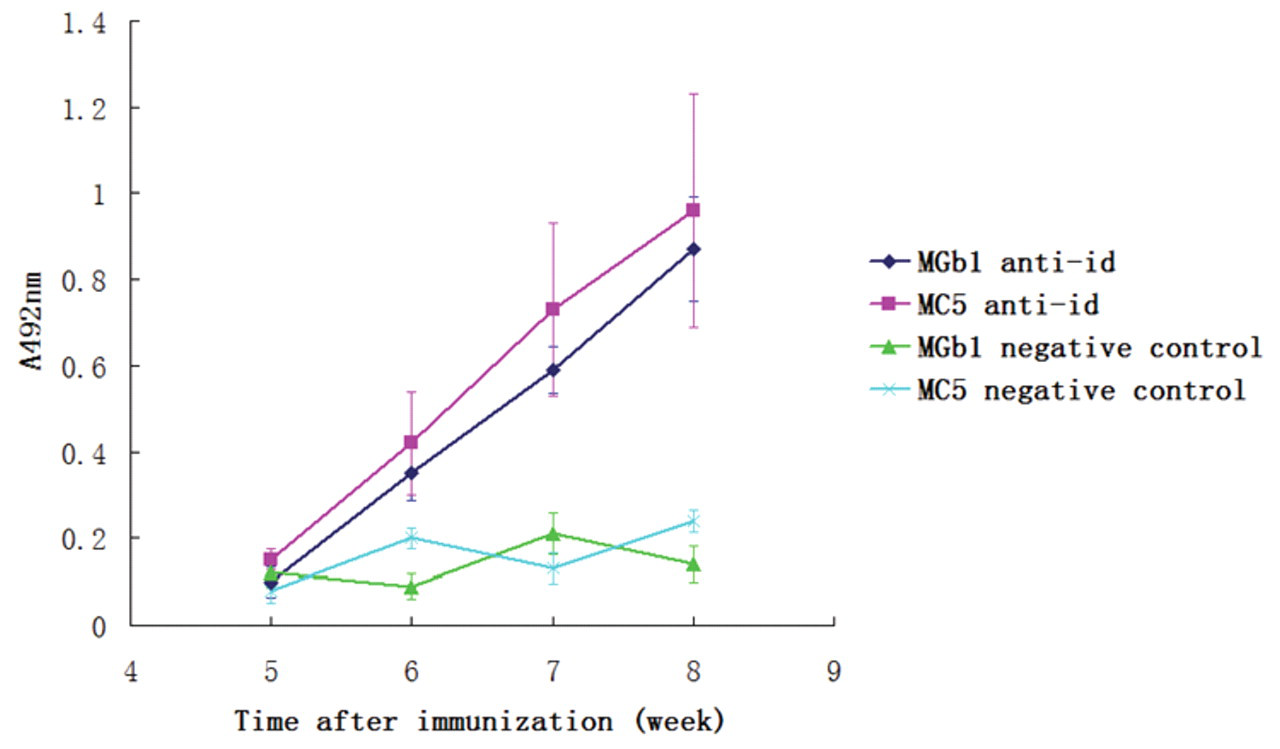
Antibody against cancer cells detected by cell enzyme-linked immunosorbent assay (ELISA) at different time points after immunization with anti-idiotype antibodies. Antibodies against gastric/colorectal cancer cells were detected 6 weeks after immunization, and the antibody titers increased with time. No reaction with the cancer cells was detected in mice immunized with only complete Fraud’s adjuvant (negative control).
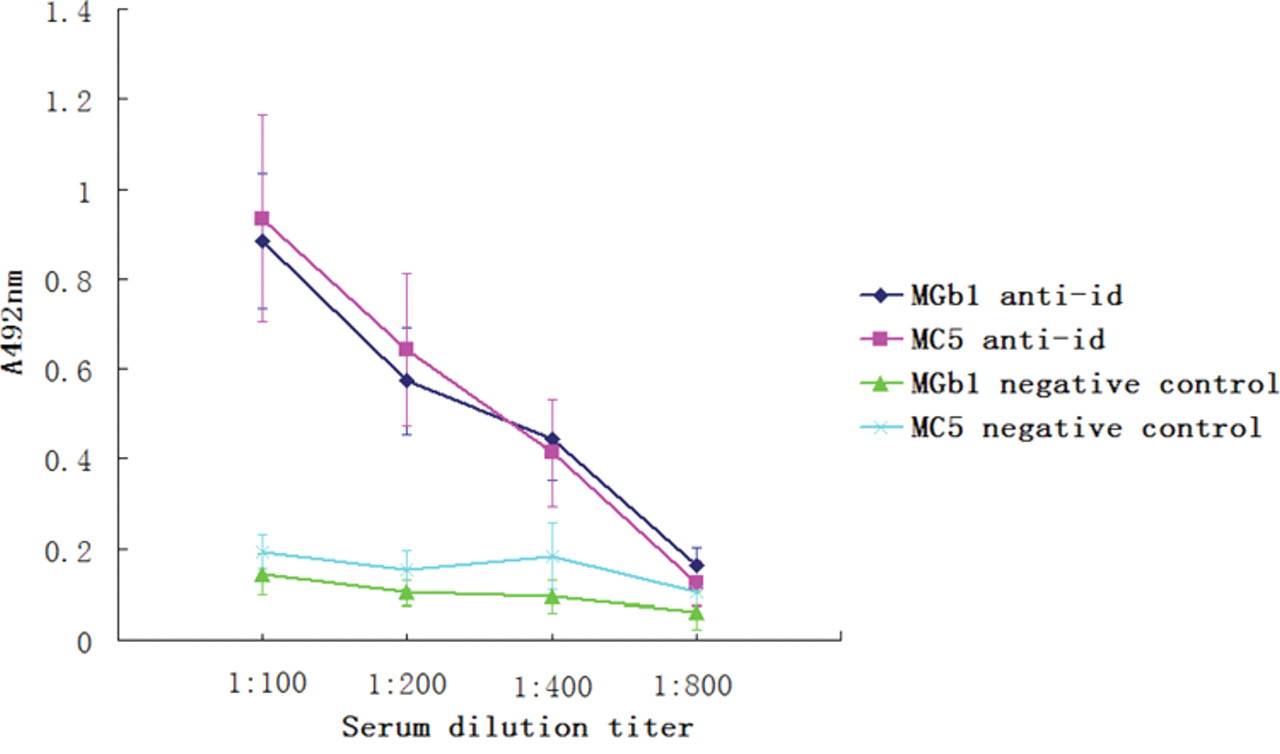
Antibody titers detected at week 8 after immunization with anti-idiotype antibodies. Serum from mice after immunization with anti-idiotype antibodies was used in a cell enzyme-linked immunosorbent assay (ELISA) assay at different dilutions. Reaction with gastric/colorectal cancer cells was detected even when the serum was 1:400 diluted.
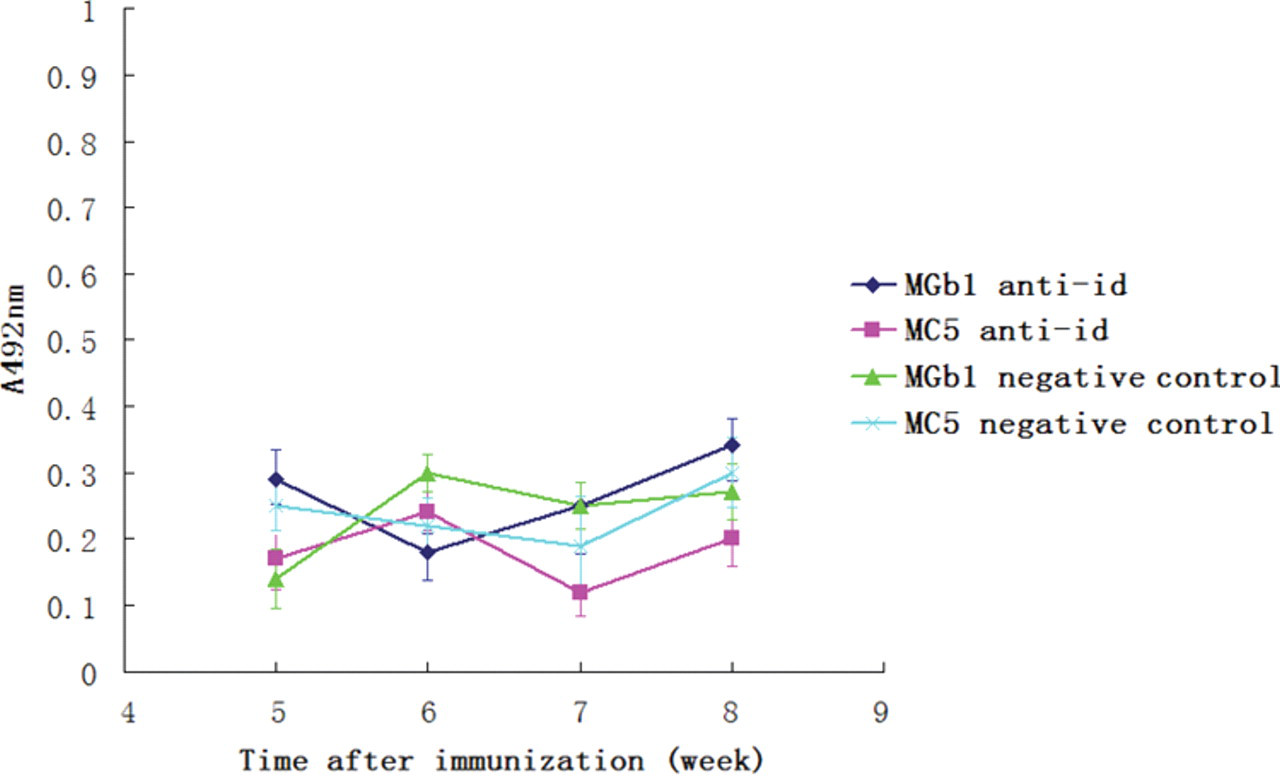
Cell enzyme-linked immunosorbent assay (ELISA) using cells (GES-1) with no expression MC5 or MGb1 antigen. No reaction with GES-1 was detected for sera from immunized mice (with MGb1 anti-idiotype or MC5 anti-idiotype proteins) or control mice (immunized with only complete Freund’s adjuvant). Note: the cell ELISA assays were done on different plates and presented in the same figure, so there are 2 negative controls in this figure.
Discussion
Gastric cancer and colorectal cancer are major malignancies of the digestive system. Several gastric cancer and colorectal cancer vaccines have been developed and are in clinical use. Major targets of gastric cancer vaccines are patient-specific cytotoxic T lymphocyte (CTL) peptides isolated from the draining lymph nodes of the patients, gastrin, tumor markers such as carcinoembryonic antigen (CEA), and other cancer antigens such as MAGE. And for colorectal cancer, CEA, CD55, GA733 antigen, CTL peptide, ras peptides, and p53 were the major molecules used to develop vaccines. 11-20 Among these targets, cancer CTL peptides were mostly patient specific and difficult to isolate and identify. Tumor markers such as p53, CEA, and gastrin had moderate or poor specificity and sensitivity for gastric cancer and colorectal cancer. MC5 and MGb1 were monoclonal antibodies developed in our institute by immunizing mice with colorectal/gastric cancer cells. By immunohistochemical staining, it was found that 99.2% of the colorectal cancer specimens were MC5 positive and 78% of gastric cancer specimens were MGb1 positive, 1,3 suggesting that antigens recognized by these antibodies were abundant in either cancer type. However, these antigens have not been identified. MGb-1 and MG7 are other MG series antibodies against gastric cancer developed in our institute. Human 37-kDa laminin receptor precursor was identified as an MGr1 antigen, which was found be associated with multidrug resistance of gastric cancer. Inhibiting MGr1-Ag expression significantly sensitized gastric cancer cells to chemotherapeutic drugs. 21-23 For MG7, approximately 90% of gastric cancer cells expressed MG7-Ag. 24-26 Vaccines based on MG7 mimicry peptide induced significant anticancer immune response. 7-9
To improve the efficacy of gastric cancer vaccines and provide novel candidate molecules for colorectal cancer vaccine, we constructed the phage-displayed murine ScFv libraries from mice immunized with MGb1 McAb or MC5 McAb. By screening the library, anti-id antibodies against MGb1 and MC5 were identified. These anti-id antibodies could inhibit the binding of the McAb to gastric or colorectal cancer cells, suggesting that the anti-id antibodies were specific for the original McAbs. Immunization of mice with anti-id antibodies induced antibodies against gastric cancer or colorectal cancer but not against cells that do not express MGb1 or MC5 antigen, which indicated that these ScFv proteins were both immunogenic and specific. Given that almost all colorectal cancer cells express MC5 and that 78% of gastric cancer cells express MGb1-Ag, the MC5 and MGb1 anti-id antibodies would serve as good candidates for a colorectal and gastric cancer vaccine. Although MGr1 antigen was present in only 18% of gastric cancer cells, its role in multidrug resistance of gastric cancer cells indicated that MGr1 antigen-based vaccines may provide additional benefit by sensitizing cancer cells to chemotherapeutic drugs. Including multiple epitopes such as MG7-Ag, MGr1-Ag, and MGb1 anti-id antibody in gastric cancer vaccine development seems to be a reasonable strategy, and it would be very interesting to test if the MGr1 anti-id antibody vaccines can affect the drug sensitivity of gastric cancer. More work is needed to clarify whether vaccines based on MC5 anti-id antibodies would provide a protective effect on colorectal cancer and whether multiepitope gastric cancer vaccines, including MG7-Ag, MGb1 anti-id antibodies, and MGr1-Ag, would be more effective than single-epitope vaccines. Research on this aspect is under way in our institute.
