Abstract
A chip-based automated patch-clamp technique provides an attractive biophysical tool to quantify solute permeation through membrane channels. Proteo–giant unilamellar vesicles (proteo-GUVs) were used to form a stable lipid bilayer across a micrometer-sized hole. Because of the small size and hence low capacitance of the bilayer, single-channel recordings were achieved with very low background noise. The latter allowed the characterization of the influx of 2 major classes of antibiotics—cephalosporins and fluoroquinolones—through the major Escherichia coli porins OmpF and OmpC. Analyzing the ion current fluctuations in the presence of antibiotics revealed transport properties that allowed the authors to determine the mode of permeation. The chip-based setup allows rapid solution exchange and efficient quantification of antibiotic permeation through bacterial porins on a single-molecule level.
Introduction
T
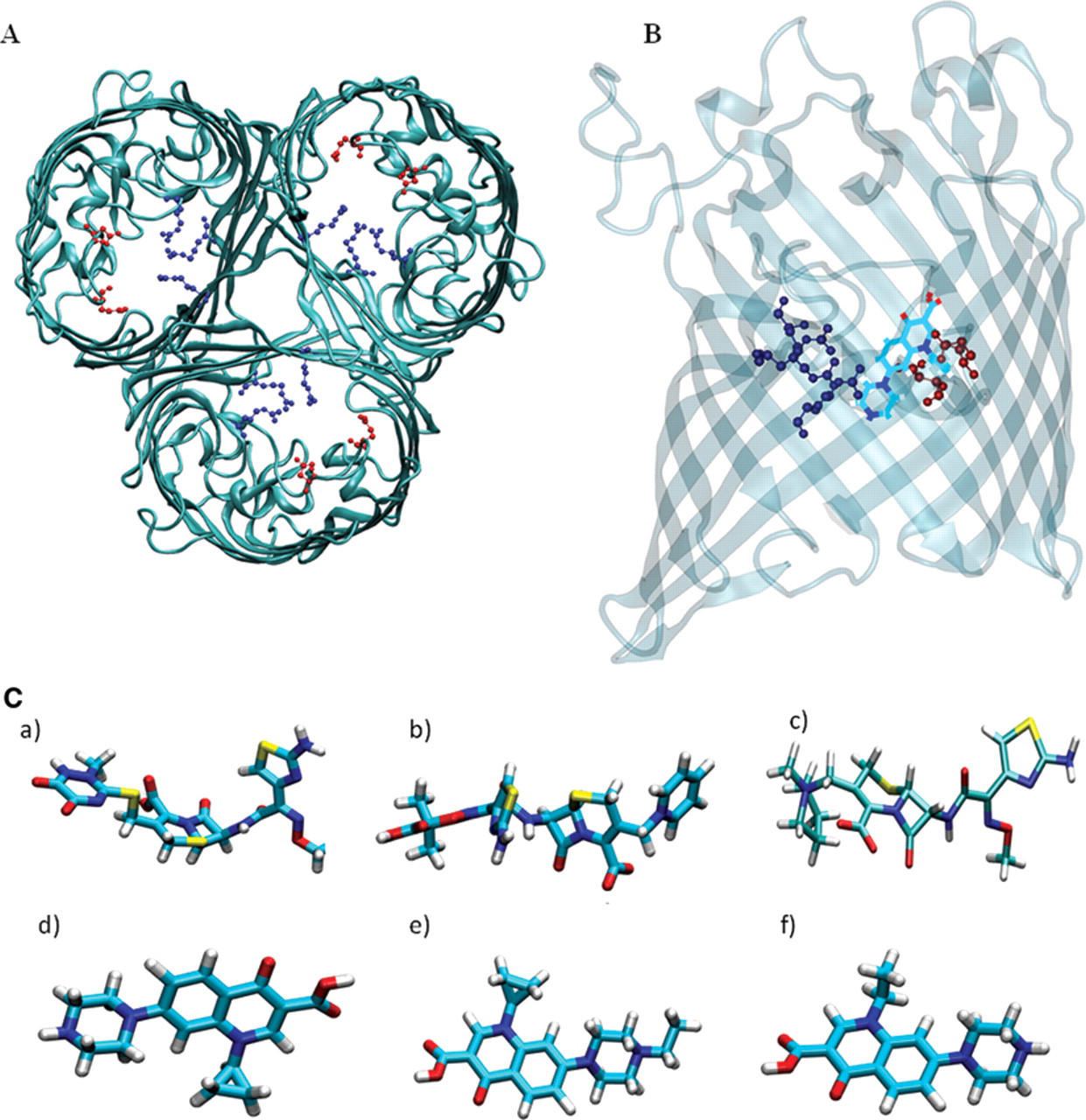
(
Electrophysiology has been a technique of choice to study the biophysical properties of porins. Purified porins can be reconstituted into planar lipid bilayers (black lipid membranes [BLM]). 6 In this technique, membrane channels are individually inserted into the bilayer, and insertion is followed by an increase in conductance. Permeation of molecules inside the channel causes fluctuations in the ion current, reflecting the molecular interactions with the channel wall. 5,7,8 Recent improvements in ion current fluctuation analysis on a single-channel level have given new insights on the interaction of solutes with the membrane channel. 9,10 Recently, a new method was introduced for the formation of lipid bilayers on a glass substrate with an approximately 1-µm size aperture. Stable lipid bilayers are formed by bursting a giant unilamellar vesicle (GUV) on the glass surface, 9-12 forming a free-standing portion above the hole. Previously, high-resolution alamethicin channel measurements have been achieved by inserting these ion channels in the already formed µ-bilayer. 10 Porins, on the other hand, can be directly reconstituted into GUVs before the formation of the lipid bilayer, resulting in patching and subsequent rupture of proteo-GUVs. Single-channel recordings in the presence of the antibiotics are possible by perfusing the chip with the relevant solution.
In this study, the translocation of antibiotics through E. coli OmpF and OmpC was investigated, and translocation rates were determined. For our studies, we selected 2 main classes of antibiotics used in human and veterinary medicine: fluoroquinolones and cephalosporins. The mechanism of action of the fluoroquinolones is based on inhibiting the catalytic activity of enzymes essential to the DNA replication, whereas cephalosporins inhibit the bacterial cell wall synthesis. 1 Cephalosporins tested include ceftriaxone and ceftazidime (negatively charged) and cefepime (zwitterionic compound; Fig. 1C ). Fluoroquinolones included were ciprofloxacin, norfloxacin, and enrofloxacin (all zwitterionic compounds; Fig. 1C ).
Materials and Methods
The following chemical reagents were used in this study: KCl, 2-(N-morpholino)ethanesulfonic acid (MES), n-pentane, hexadecane, norfloxacin, enrofloxacin, ceftriaxone, and ceftazidime (Sigma Aldrich, Buchs, Switzerland); ciprofloxacin, cholesterol, trichloromethane, and sorbitol (Applichem, Darmstadt, Germany); cefepime (Basilea Pharmaceutica Ltd., Basel, Switzerland); 1,2-diphytanoyl-sn-glycero-3-phosphatidylcholine (DPhPC; Avanti Polar Lipids, Alabaster, AL); and N-octylpolyoxyethylene (octyl-POE) (Bachem, Bubendorf, Switzerland). Bio-Beads were obtained from Bio-Rad (Bio-Rad, Munich, Germany). Double-distilled water was used to prepare solutions. Antibiotic stock solution of 5 mM was used for the measurements except enrofloxacin and ciprofloxacin (1 mM).
Preparation of vesicles, channel reconstitution, and formation of planar lipid bilayers
GUVs were prepared by the electroformation method 9,13 in an indium tin oxide (ITO)–coated glass chamber connected to the Nanion Vesicle Prep Pro setup (Nanion Technologies, Munich, Germany). The ITO layers on the 2 glass slides are electrically conductive and therefore served as electrodes. Lipid-containing solution, 5 mM DPhPC with 10% cholesterol dissolved in chloroform, was deposited on the ITO-coated glass surface for the vesicle preparation. The process of electroformation was controlled by the Vesicle Prep Pro setup, and all parameters (amplitude, frequency, duration, etc.) for the electroformation were programmed in the VesicleControl software (Nanion Technologies, Munich, Germany). Purified wild-type OmpF or OmpC (1.5 mg/mL) in 1% octyl-POE was reconstituted into GUVs as described previously. 9,10 The final concentration of porins was around 10 to 20 nM, and the final detergent concentration was around 0.001%. Large batches of proteo-GUVs with different concentrations of porins were used, and a protocol was developed to get only a single channel per bilayer patch. After incubating the GUVs with the porin solution, octyl-POE was removed using Bio-Beads. The mixture was incubated at 4 °C overnight, and then Bio-Beads were removed afterwards by centrifugation. The protein containing GUVs was used directly for planar lipid bilayer formation. When kept at 4 °C, the proteo-GUVs were used for a week successfully. For formation of a planar lipid bilayer containing the proteins, 2 to 3 µL of the proteoliposome solution was pipetted onto the microstructured glass chip containing an aperture of approximately 1 µm in diameter. Electrophysiology experiments were performed with the Port-a-Patch automated patch-clamp system (Nanion Technologies), using borosilicate glass chips. 9 Antibiotic titrations were performed in symmetric solutions of 1 M KCl, 20 mM MES (pH 6). Enrofloxacin measurements were done in 150 mM KCl, 10 mM MES (pH 6) due to solubility problems. Application of all solutions was achieved using an automated external and internal perfusion system. The perfusion system allowed the fast addition of the antibiotic to both sides of the lipid membrane. Single-channel current measurements were recorded using an Axopatch 200B amplifier (Axon Instruments, Foster City, CA) connected to Ag/AgCl electrodes in the Port-a-Patch system. The signal was filtered using a 4-pole low-pass Bessel filter at a frequency of 10 kHz and sampled at 50 kHz, acquired using a Digidata 1440A digitizer, and analyzed using the pClamp 10.0 software (Axon Instruments). Ion current blockage events were measured following the addition of antibiotics to the cis or trans side of the lipid membrane.
Results and Discussion
A stable planar lipid bilayer containing single trimeric OmpF or OmpC was formed by fusion of proteo-GUVs onto a microperforated glass chip as described previously. 9 After the formation of the lipid bilayer over the aperture of the glass surface, we perfused the lipid bilayer with the buffer solution to remove remaining proteo-GUVs to prevent multiple-channel insertions. In agreement with previous measurements on classical planar lipid bilayers, single trimeric OmpF in the bilayer showed channel conductance of 4.0 ± 0.3 nS, whereas OmpC showed single-channel conductance of 2.5 ± 0.3 nS, both in 1 M KCl (pH 6). The porin channels show voltage gating activity similar to classical BLM measurements on the new system. 9 At transmembrane potentials below 100 mV, the channels were open without any gating perturbations. The threshold potential (critical voltage) inducing closure of the channels was found to be ±150 mV for OmpF and ±300 mV for OmpC.
Permeation of antibiotics through OmpF
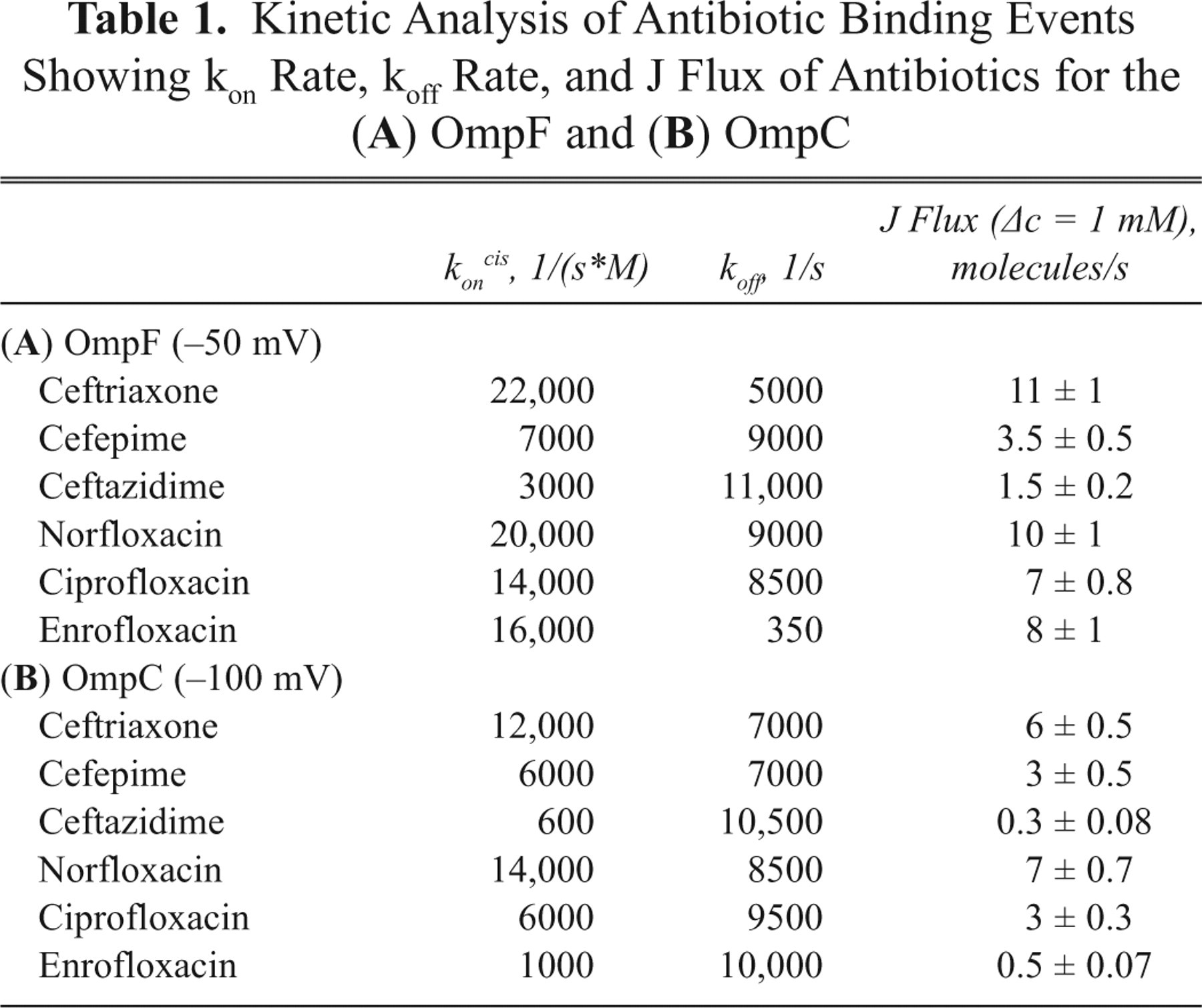
In a first series of measurements, we compared the permeation of fluoroquinolones and cephalosporins across the E. coli OmpF. Channel blockages were analyzed after adding antibiotic to the system. 5,7-9 In the absence of antibiotic, ion currents through the channels were stable without fluctuations. Addition of antibiotics caused fluctuations of the ion current, indicating strong interactions of the antibiotics with the channel. As shown in Figure 2 , at 5 mM cephalosporins, monomer blocking events of a single trimeric channel were clearly visible. Increasing the antibiotic concentration increased the frequency of the blockage events. Ceftriaxone strongly interacted with OmpF, resulting in complete monomer blockage in contrast to cefepime and ceftazidime, which produced partial monomer closures of a single trimeric channel ( Fig. 2A ). The residence time of the antibiotics was calculated by dwell time analysis, fitting a single exponential decay to the blocked time histograms. 7,8 The strength of the OmpF-cephalosporin interaction is in the following order: ceftriaxone > cefepime > ceftazidime. In the case of fluoroquinolones also, we observed strong ion current blockages reflecting antibiotic–channel interactions but with varying strength of interaction ( Fig. 2B ). Enrofloxacin interaction with the channel was significantly stronger than ciprofloxacin or norfloxacin (as measured in 150 mM KCl [pH 6] due to the drug’s insolubility). The average residence time of enrofloxacin was calculated to be 3.0 ± 0.4 ms at −50 mV and 1.5 ± 0.3 ms at +50 mV, whereas all other antibiotics showed residence times of 100 to 200 µs in OmpF. The number of binding events in the case of fluoroquinolones was strongly dependent on the polarity of the applied voltage, and we observed stronger binding events in the case of negative voltage than at positive voltage. This mechanism of voltage asymmetry is not completely understood. Antibiotic binding kinetics for the OmpF channel is summarized in Table 1A .
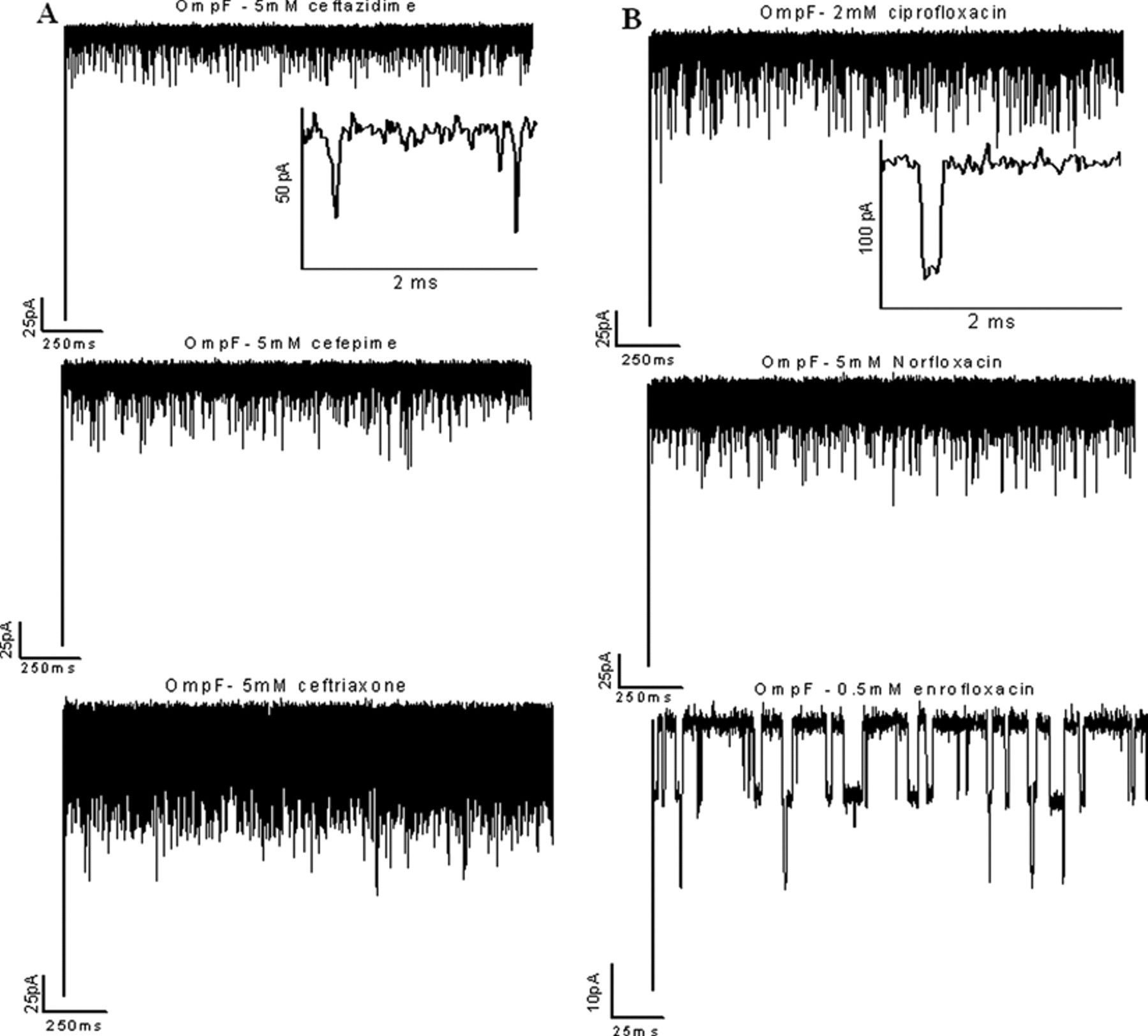
Typical tracks of ion conductance through single trimeric OmpF channels reconstituted into planar lipid membranes in the presence of (
Kinetic Analysis of Antibiotic Binding Events Showing kon Rate, koff Rate, and J Flux of Antibiotics for the (
Permeation of antibiotics through OmpC
In a second series of measurements, we investigated the influx of antibiotics through E. coli OmpC. Cefepime and ceftriaxone strongly interacted with the OmpC, resulting in lasting ionic current blockage, whereas in the case of ceftazidime, blockage events were short and less frequent, indicating a weak interaction of the antibiotic with the channel surface ( Fig. 3A ). Fluoroquinolones, ciprofloxacin, and norfloxacin also interacted with the OmpC channel, producing time-resolved interruptions in the ionic current, with the blockage events strongly concentrated and voltage dependent. In the case of the enrofloxacin, unlike other fluoroquinolones, we observed very short blockages and less frequent binding events ( Fig. 3B ). Our data clearly show that cefepime, ceftriaxone, norfloxacin, and ciprofloxacin strongly interact with an affinity site in the OmpC channel that facilitates their translocation through the porin. Kinetic analysis of our data showed considerable interaction of the antibiotics with OmpC, suggesting an internal affinity site. Kinetic parameters for the antibiotic binding to OmpC channel are summarized in Table 1B .
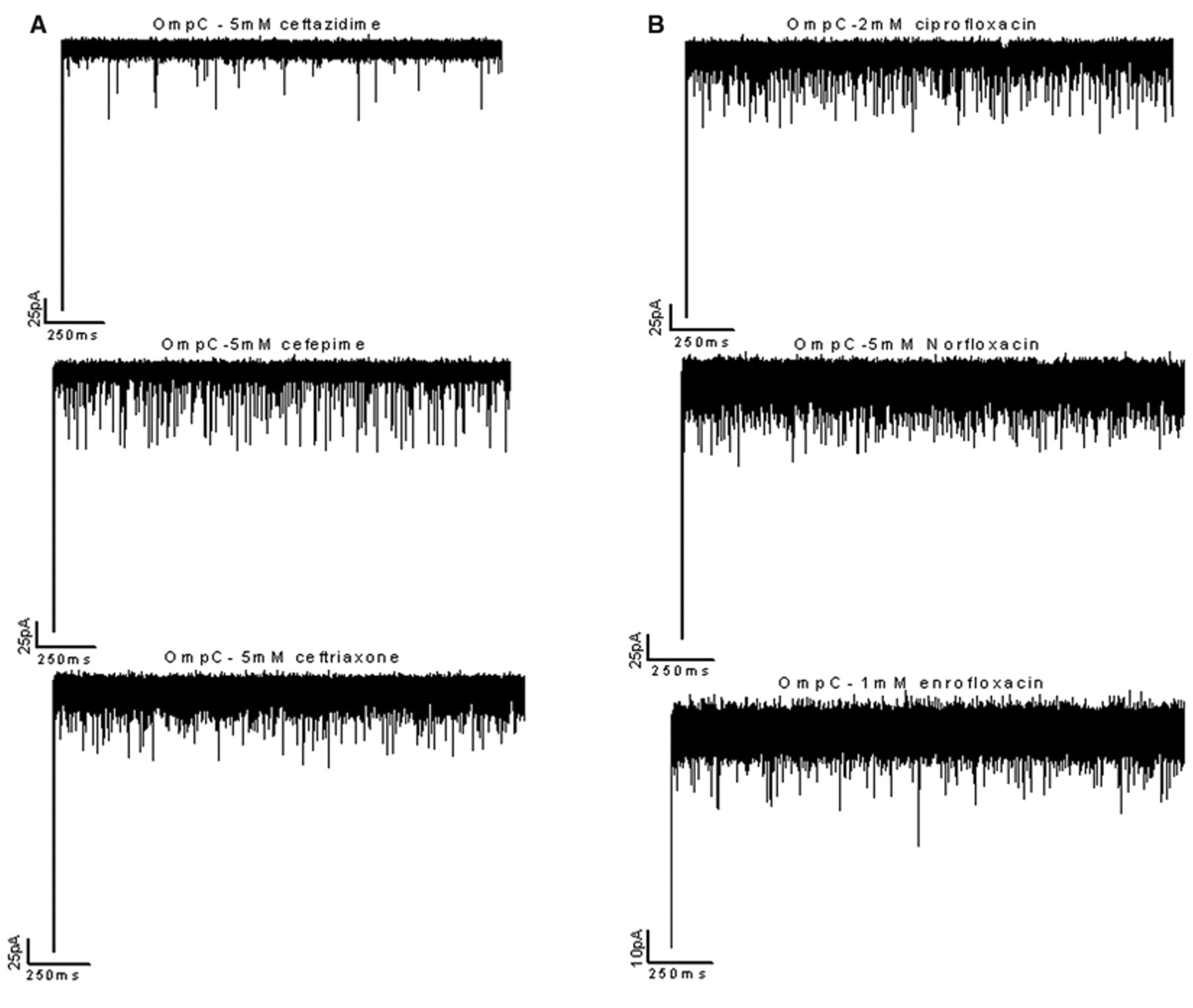
Typical tracks of ion conductance through single-trimeric OmpC channels reconstituted into planar lipid membranes in the presence of (
The on rate was calculated from the number of binding events per second, and the off rate was calculated from the residence time, as described previously. 5,7,8 The association rate constant kon gives the permeation rate of an antibiotic molecule from the cis (kon cis) or trans side (kon trans) to the affinity site in the channel. The dissociation rate constant koff describes the rate at which antibiotic molecules are released from the affinity site in the channel to the cis or trans side. The strong attractive interaction between the antibiotic and the channel facilitates drug permeation with high efficiency. 5,7,8,14 Assuming a simple symmetrical model, flux of the antibiotic through the channel is proportional to the kon rate ( Table 1 ). 2,8 All antibiotics tested translocated through the OmpF channel but with different efficiency. Our data showed that ceftriaxone, which is a negatively charged antibiotic, binds strongly and translocates through OmpF and OmpC more efficiently compared to zwitterionic compounds. We report for first time the biophysical evidence that supports the role of E. coli OmpC in the permeation of cephalosporins and fluoroquinolones. Compared to OmpF, the number of binding events is reduced in the case of OmpC for all 3 cephalosporins, which show that OmpF is more efficient in the translocation of antibiotics. Hydrophilic quinolone antibiotics such as ciprofloxacin and norfloxacin showed almost the same binding kinetics for both OmpF and OmpC. Enrofloxacin strongly binds with the OmpF channel with a residence time of 3 ms, whereas in the case of OmpC, the interaction strength is drastically reduced with a short residence time of 100 µs. The OmpC channel is smaller than that of OmpF, and earlier studies have shown that antibiotic molecules with bulky side chains were more restricted in their permeation through OmpC than through the wider OmpF channel. 1,2 The OmpC crystal structure will help further studies with molecular dynamics simulations to researchers understand the molecular mechanism. 4
In addition to single-channel analysis, we also performed power spectrum analysis on the current traces. Interaction of antibiotics with the channel is a double Markovian process and generates an excess ion current noise whose power spectra can be described by a single Lorentzian. Fitting measured power spectra to a single Lorentzian gives the relaxation time of the process. 5,7 Noise spectra obtained in the presence of effective antibiotics revealed excess noise compared to those in the absence of antibiotics (data not shown).
There are several advantages to using patch-clamp chips containing small micrometer-sized holes compared to the classical Montal-Mueller membrane of larger size. Despite the small area, the chip system allows successful high-resolution single-channel measurements, rapid perfusion of solutes, and continuous measurements over a long time. In addition, lipid bilayers formed on this system were stable up to 500 mV. Larger membrane areas create larger background noise, which represents a serious problem for resolving short binding events. Some antibiotics in particular translocate through the channel too fast for detection. For example, in the case of ceftazidime translocation through OmpF, we could not resolve all binding events when using the classical lipid bilayer technique (see Supplementary Figure 1 at hhtp://jbx.sagepub.com/supplemental). We have shown previously that temperature-dependent measurements can be used to study fast translocation. 8 However, using the patch-clamp chip, we clearly showed that very brief antibiotic blockage events through the channels can be resolved ( Fig. 2A ). Another advantage is the small volume. Some antibiotics used in our studies such as cefepime are available only in small quantities, and using the patch-clamp method, we substantially reduced the sample volume required as compared to traditional bilayer recording experiments. With the help of the perfusion system, we could rapidly screen different antibiotics in the same single-trimeric channel. The external and internal perfusion systems allow the addition of the antibiotic to both sides of the lipid membrane and the study of asymmetric and symmetric permeation of antibiotics. Developing this higher throughput screening possibility for antibiotics could place planar lipid bilayers as a first step in screening new antibiotics on a larger scale. We successfully employed this technique to characterize antibiotic translocation through the porin isolated from other pathogenic bacteria such as Enterobacter aerogenes and Providencia stuartii that are more clinically relevant. Results obtained from our system can be combined with several other techniques such as molecular dynamics simulations, fluorescence measurements, and microbiological assays. 2 Combining all these techniques allows us to understand the molecular basis of antibiotic translocation through the outer membrane of bacteria. Our study of antibiotic translocation at the single-molecule level gives information on the uptake kinetics of antibiotics through the porin channels, which in turn would help researchers to design novel antibiotics against pathogenic bacteria.
Footnotes
Acknowledgements
We thank Basilea Pharmaceutica Ltd. (Switzerland) for the donation of cephalosporins and Tivadar Mach, Soroosh Pezeshki, and Catalin Chimerel for stimulating discussions.
This study was supported by EU-grant MRTN-CT-2005-019335 (Translocation).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
