Abstract
Objective:
The effectiveness of behavioural interventions to improve sleep in children with ADHD has been demonstrated, although most treatments are conducted in-person, with limited evidence for paediatric telehealth approaches. Study 1 is a randomised controlled trial evaluating the ADHD Sleeping Sound© intervention to compare outcomes between in-person and telehealth modalities. Study 2 examined whether baseline sleep problems and treatment response were predicted by baseline symptom presentation, attachment security, and family functioning.
Method:
Children aged 5 to 12 years (65.4% male) with ADHD received either in-person (n = 40) or telehealth (n = 38) treatment. Subjective (Children’s Sleep Habits Questionnaire) and objective (actigraphy) sleep variables were assessed at baseline, post-intervention (1 week), and follow-up (3 months).
Results:
As expected, subjectively measured sleep disturbance significantly improved post-treatment, with no significant difference in rate of change between in-person and telehealth modalities. For Study 2, baseline measures comprised the Conners CBRS, Attachment Insecurity Screening Inventory, Security Scale, and Family Assessment Device. Latent growth curve modelling analysed data via intention-to-treat (primary) and per protocol (secondary) methods. Higher attachment insecurity and lower family functioning were significantly associated with greater (subjective) baseline sleep problems. Additionally, attachment security predicted changes in sleep duration over time, highlighting the importance of considering relational family factors when designing sleep interventions.
Conclusion:
Telehealth ADHD sleep interventions represent a promising approach to enhancing health equity and access for families experiencing geographical and/or structural barriers to in-person treatment.
Trial Registration
ANZCTR, ACTRN12621001681842. Registered 9 December 2021 – https://anzctr.org.au/ACTRN12621001681842.aspx
Keywords
Introduction
ADHD is characterised by high levels of hyperactivity, impulsivity, and/or inattention across settings that affects approximately 6% to 8% of children worldwide (Faraone et al., 2021; Salari et al., 2023). Prior research has established an association between ADHD and disrupted sleep (Corkum et al., 1999). For example, children (5–11 years) with ADHD are nine times more likely to be diagnosed with a sleep disorder than those without ADHD (Ahlberg et al., 2023). Commonly reported sleep problems include delayed sleep onset, bedtime resistance, co-sleeping, night awakenings, and parasomnias (Deshpande et al., 2022). The relationship between ADHD and sleep has been described as complex and bidirectional, due to overlapping symptoms and cognitive impairments (Hvolby, 2015; Weiss et al., 2015). Sleep difficulties may exacerbate ADHD symptomatology, and both sleep problems and ADHD can independently impact neuropsychological functioning, with a cumulative impact noted (Schneider et al., 2016; Weiss et al., 2015). The presence of co-occurring conditions in individuals with ADHD can also increase the complexity of sleep disturbance and compound functional outcomes (Cortese et al., 2009).
The effectiveness of behavioural sleep interventions has been established (for reviews, see Cortese et al., 2024; Malkani et al., 2022; Nikles et al., 2020). These interventions typically involve active parent/caregiver involvement to implement strategies to improve their child’s sleep. Interventions for school-aged children, such as the Sleeping Sound with ADHD© program (hereafter referred to as ‘Sleeping Sound’), involve psychoeducation plus techniques aimed at managing sleep-related behaviours (Sciberras et al., 2011, 2020). Randomised controlled trials (RCTs) examining the effectiveness of ADHD-sleep behavioural interventions have found immediate and sustained improvements (Mehri et al., 2020; Shokravi et al., 2016). One Australian RCT involving 244 school-aged children reported significant improvement in sleep disturbance using Sleeping Sound at 3, 6, and 12-month follow-up compared to controls, with additional benefits in behaviour, daily functioning, and quality of life (Hiscock et al., 2015).
Despite their effectiveness, in-person interventions can be associated with accessibility challenges related to cost, transport, and time, and they can be difficult for families living in rural/ remote communities to access (Spencer et al., 2020). In more recent years, in part due to technological advances and the COVID-19 pandemic, there has been an uptake of electronic communication healthcare treatments, including online telehealth/videoconferencing options (Taylor et al., 2021). Indeed, meta-analytic data has indicated that telehealth approaches are more effective in treating chronic insomnia in adults than in-person methods (Sharafkhaneh et al., 2022). However, whilst telehealth has been shown to be an effective method for treating ADHD symptoms (Myers et al., 2015; Xie et al., 2013), few paediatric ADHD-sleep interventions have been offered remotely.
One program, Better Nights/Better Days, offered a remote modality whereby parents were educated on sleep techniques via a treatment manual and weekly telephone coaching, and although positive results were demonstrated via their parallel group RCT (i.e., 41.7% improvement on the Children’s Sleep Habits Questionnaire), the sample size of participants with ADHD was small (n = 22) and excluded individuals with co-occurring conditions (Corkum et al., 2016). Roberts et al. (2019) evaluated a parent-based sleep education program delivered in-person and online in a cohort of children with autism, with 48% of the sample having co-occurring ADHD. Whilst subgroup analyses related to diagnoses were not conducted, the intervention demonstrated significant improvements in parent-reported sleep outcomes, particularly in younger children and those in the online delivery group.
The acceptability of telehealth sleep interventions for children with other neurodevelopmental conditions has more recently been explored (Tan-MacNeill et al., 2023). Johnson et al. (2023) found improvements in sleep disturbances for autistic children aged 2 to 7 years following a telehealth-delivered parent training RCT intervention. Furthermore, a pilot study explored the use of an online cognitive-behavioural therapy for insomnia in a small group of school-aged autistic children and demonstrated immediate improvements in sleep outcomes sustained at 1-month follow-up (McCrae et al., 2021). These studies provide promising evidence for the use of telehealth-delivered sleep treatment for children with neurodevelopmental disorders.
Past research indicates that children with ADHD commonly experience other neurodevelopmental and/or psychiatric conditions, such as autism, anxiety, and depression (Cuffe et al., 2020; Gruber, 2009), with approximately 60% to 80% of children experiencing at least one additional condition (Ghanizadeh et al., 2008; Larson et al., 2011). The presence of co-occurring conditions can exacerbate functional impacts (Armstrong et al., 2015; Booster et al., 2012; Cuffe et al., 2020), such as contributing to school problems, low social competence, and reduced family communication (Larson et al., 2011). An increased burden of comorbid conditions in individuals with ADHD is also associated with lower levels of functioning and may worsen sleep disturbance (Kamara & Beauchaine, 2020).
Sleep problems are not unique to those with ADHD and are often reported by those with co-existing neurodevelopmental and mental health conditions (Gregory & Sadeh, 2016). For example, sleep problems in autism and fetal alcohol spectrum disorder are highly prevalent, with research often noting problems with sleep initiation and maintenance (Hanlon-Dearman et al., 2018; Kotagal & Broomall, 2012; Krakowiak et al., 2008). Numerous reviews have examined the association between childhood mental health disorders and sleep (e.g., Accardo et al., 2012; Belmon et al., 2019; Ivanenko et al., 2004). Additionally, the underlying behavioural profile of children with externalising behaviours is thought to be associated with sleep problems (Bayes & Bullock, 2019); with a reported association between oppositional defiant behaviours and sleep onset delay, bedtime resistance, and daytime sleepiness (Iwadare et al., 2015). An Australian study established that mood disturbance and externalising behaviours were associated with subjective sleep disturbance in adolescents, regardless of the presence of ADHD (Loram et al., 2023).
However, sleep problems may be more severe in children with ADHD who have other comorbidities compared to those with ADHD alone (Lycett et al., 2015; Virring et al., 2017). For example, children with ADHD and mood disturbance report higher sleep problem scores (Mayes et al., 2009), and children with ADHD and anxiety demonstrated significantly higher parent-reported scores of sleep onset latency, bedtime resistance, and night wakings (Accardo et al., 2012). Research also indicates that impacts on functional outcomes and sleep disturbance may heighten with a stepwise increase in comorbid disorders (Larson et al., 2011).
Other possible factors complicate the interrelationship between sleep and ADHD. That is, families of children with ADHD typically experience higher parenting stress, poorer functioning, and more conflict compared to controls (Cussen et al., 2012; Dekkers et al., 2022; Moen et al., 2016). Children with ADHD are also more likely to have an insecure attachment pattern due to the association between adverse family environments and their symptoms (Bhide et al., 2024; Dekkers et al., 2021, 2022; Storebø et al., 2016). Research has highlighted an association between poor family outcomes and sleep problems in children with ADHD. Sung et al. (2008) identified an association between moderate to severe sleep problems and poorer family quality of life, caregiver mental health, and caregiver work attendance. Lycett et al. (2014, 2016) reported that poorer caregiver mental health was a risk factor for children experiencing transient sleep problems measured over 12 months and that poorer family quality of life, caregiver mental health, and primary caregiver work difficulties were associated with transient and persistent sleep problems.
The establishment of attachment patterns may also overlap with the developmental progression of sleep regulation through infancy and early childhood (Anders, 1994; Benoit et al., 1992). Various co-sleeping studies have demonstrated that maternal presence and a positive infant-child relationship support infant sleep architecture and promote rapid eye movement (REM) sleep (McKenna et al., 1993, 2007). Therefore, sleep in children appears to be inherently related to attachment security, given the caregiver’s role in the sleep process (Adams et al., 2014). Research exploring the attachment–sleep relationship in children also includes studies assessing family functioning, emotional insecurity, and facets of marital instability. Lower levels of attachment security and higher levels of emotional insecurity have been associated with poorer sleep via both objective and subjective measures across early and middle childhood (Kelly & El-Sheikh, 2013; Troxel et al., 2013). Keller and El-Sheikh (2011) provided evidence that children’s emotional insecurity towards parental marital functioning was associated with increased sleep latency in girls, higher prevalence of sleep-wake problems in boys, and increased sleepiness overall. However, it does not appear that past research has explored the attachment–sleep relationship in children with ADHD.
Our primary research objective was to explore three research questions across two studies. Study 1 assessed the effectiveness of an established ADHD behavioural sleep intervention and whether the in-person and telehealth delivery modalities demonstrated similar effectiveness in children aged 5 to 12 years. 1 It was hypothesised that regardless of delivery modality, children’s sleep outcomes would improve following the Sleeping Sound intervention. Secondly, it was hypothesised that the rate of change in sleep outcomes following the intervention would be similar across in-person and telehealth modalities. Finally, Study 2 explored a third research question as to whether baseline sleep problems and rate of change following intervention were predicted by symptom presentation, attachment security, and family functioning measured at baseline. It was hypothesised that (a) greater symptom presentation complexity, (b) lower attachment security, and (c) poorer global family functioning would predict more severe sleep problems prior to the intervention. In addition, we hypothesised that (d) lower symptom presentation complexity, (e) better attachment security, and (f) greater family functioning would predict a greater improvement in sleep problems following the sleep intervention.
Study 1
Method
Design/Ethics
The trial protocol for this study has been published (Malkani et al., 2023) and was registered with ANZCTR, ACTRN12621001681842 on 9 December 2021 (submitted 4 November 2021) – https://anzctr.org.au/ACTRN12621001681842.aspx
Recruitment and baseline data collection occurred between October 2021 and November 2022, and the intervention was delivered between October 2021 and March 2023. Children and their primary caregiver(s) received the intervention in person (standard care) or via telehealth (videoconferencing). The University of Western Australia Ethics Committee approved the study (Ref: 2021/ET000637).
Recruitment
Advertisements were used to recruit parents/caregivers of children aged 5 to 12 years with ADHD and sleep problems and circulated to relevant health services and ADHD support groups in Western Australia. Children were required to have been formally diagnosed with ADHD by a medical specialist, have moderate to severe sleep problems as reported by their parent, caregiver, or legal guardian, and experience one or more of the following by parent description: delayed sleep phase (i.e., shift in sleep-wake cycle resulting in a later bedtime and wake time); insomnia and/or night-time waking (i.e., marked difficulty initiating and/or maintaining sleep); sleep onset association (i.e., falling asleep is associated with an object or person); limit setting (resistance or refusal at bedtime); and night-time anxiety (specific night time fears and/or worrying when attempting to sleep).
Children were excluded if they had a diagnosis of obstructive sleep apnoea or narcolepsy, were receiving specialist sleep treatment, or had been diagnosed with an intellectual disability. Participants were permitted to use melatonin as a sleep aid, and details of usage were recorded. Families deemed eligible after screening were invited to attend an introductory 1-hr meeting, which included completion of consent (parent) and assent (child) forms and baseline measures, as well as instructions for how to use the actigraphy device. The intervention was free, and enrolled families were offered $10 as remuneration for participation.
Randomisation
To determine treatment modality, a randomisation matrix was generated by the first author prior to commencement of participant enrolment using covariate adaptive randomisation in a block size of four. Covariates of age (5–8 or 9–12 years old), sex at birth, and presence of at least one comorbid disorder (yes or no) were used in a binary format. Participants were entered into the matrix, and a non-populated version of the matrix was retained to verify that the randomisation procedure was implemented as planned.
Intervention
Sleeping Sound consists of two 50-min consultations for the child participant and their caregiver(s), plus a brief follow-up telephone call, each held 2 weeks apart (see Sciberras et al., 2010). The consultations were conducted by Sleeping Sound-trained psychologists (N = 16) under supervision from an experienced clinical psychologist. During the first consultation, the child’s sleep problems were assessed by the clinician, who ascertained the family’s treatment goals. The clinician provided psychoeducation about normal sleep, sleep cycles, and healthy sleep practices and collaboratively formulated an individualised behaviour management plan. Handouts on psychoeducation and recommended intervention strategies were provided. Parents were asked to complete a paper-based sleep diary for daily record keeping. The second consultation and follow-up call were used for progress review, intervention reinforcement, provision of alternative intervention strategies (if required), and troubleshooting.
Treatment Fidelity
Clinicians participated in weekly supervision sessions with author (XX), an experienced clinical psychologist, to monitor adherence to the Sleeping Sound intervention, which included viewing recorded sessions. Clinicians completed a standardised consultation form for each participant, which included information regarding intervention adherence.
Measures
Demographic (e.g., sex, child age, and parent age) and medical (e.g., medical diagnoses and sleep problems) data were collected at baseline. Subjective (parent-reported sleep questionnaire – CSHQ) and objective (actigraphy) data were collected. Sleep outcomes were measured at three-time points: baseline, post-intervention (1-week post-intervention), and follow-up (3 months post-intervention, see Figure 1). Once randomised, reasonable efforts to maintain contact and follow the child for the entire study duration were undertaken; instances of non-adherence to the outcome measurement schedule were documented.
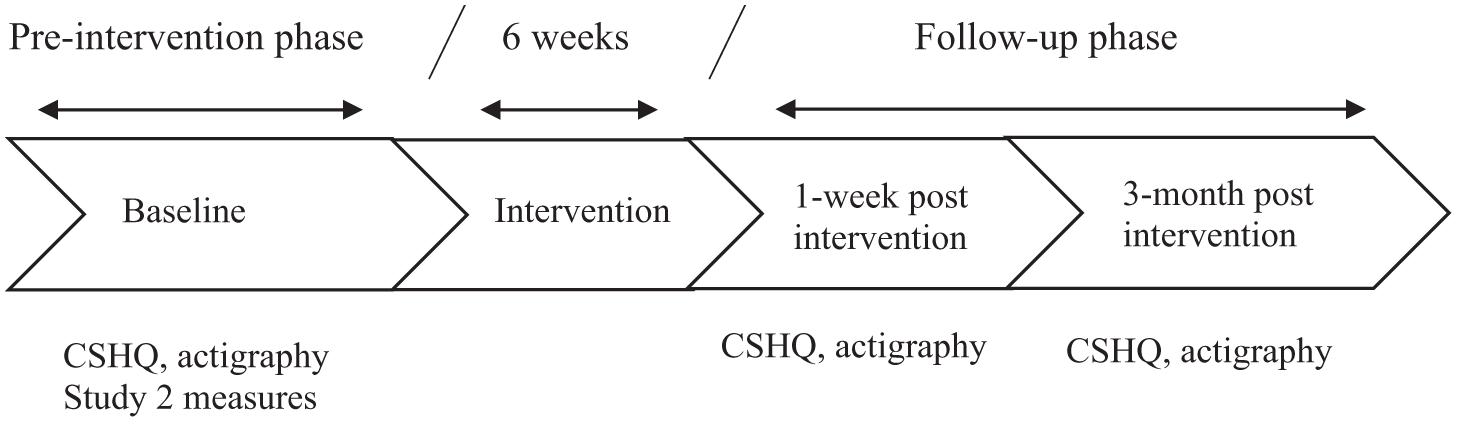
Timeline of assessment schedule.
Primary Outcome: Children’s Sleep Habits Questionnaire (CSHQ)
The CSHQ (Owens et al., 2000) is a reliable (α = .79), 33-item parent-report questionnaire of sleep initiation and maintenance problems in school-aged children. Items were rated on a 3-point Likert scale from ‘rarely’ to ‘usually’, with total sleep disturbance scores ranging from 33 to 99. A total sleep disturbance score over 41 (clinical cut-off) indicates a paediatric sleep disorder.
Secondary Outcome: Actigraphy
The ActiGraph wGT3X-BT (ActiGraph Corporation), worn on the child’s non-dominant wrist, assessed the parameters of sleep onset latency (SOL), total sleep time (TST), and sleep efficiency (SE). Sleep data were collected in 30-s epochs and processed using ActiLife software. The Sadeh algorithm (validated for paediatric populations; Sadeh et al., 1989; Meltzer et al., 2012) was applied to distinguish sleep from wake states, with sensitivity thresholds set according to established recommendations. Sleep onset latency was operationalised as the number of minutes between reported bedtime and the first sustained sleep epoch (≥10 consecutive minutes of sleep). Total sleep time was defined as the sum of all epochs scored as sleep between sleep onset and the final morning awakening. Sleep efficiency was calculated as the percentage of time spent asleep relative to total time in bed (TST ÷ time in bed × 100). This approach is consistent with previous validation studies of actigraphy in paediatric populations (Acebo et al., 1999; Cole et al., 1992; Meltzer et al., 2012). Data were collected for seven consecutive nights to allow for potential participant or equipment errors and more reliable recordings (Acebo et al., 1999; Liang et al., 2023). Children were not required to wear the ActiGraph continuously due to anticipated co-occurring sensory sensitivity and/or resistance to wearing the device (Ankrah et al., 2023). Therefore, participants were directed to wear the device each afternoon (4–6 pm) until the following morning (at least 15 min after they got out of bed).
Data were scored using the ActiLife software and a validated sleep algorithm (Sadeh et al., 1989). Movement patterns were analysed in 60-s epochs to differentiate between sleep and wake periods. Parents were asked to note in the sleep diary if the child experienced any injuries or illnesses during the period the device was worn. Nights were classed as unusable, and therefore not scored, if: (a) the device was not worn (9.5%); (b) sleep diary information was missing or not returned (8.4%); (c) the device was put on after the reported bedtime (1.4%); or (d) device failure (1.4%). Collection periods with <5 nights of usable data were not used within the analysis.
Sample Size
G*Power 3.1 (Faul et al., 2009) was used to determine the sample size required to identify an effect of treatment, regardless of treatment modality. The analysis used 3-month follow-up CSHQ outcome data from Hiscock et al. (2015). Twenty-six children per treatment arm were required to detect a large treatment effect (0.8), with 80% power and a two-tailed significance level of .05. As Hiscock et al. (2015) reported a 28.3% loss to follow-up at their 3-month assessment, 30% attrition over the study duration was incorporated. Therefore, after allowing for attrition, the final sample size needed was 68 children, 34 in each treatment arm.
Statistical Analysis
IBM SPSS Statistics for Macintosh, Version 29.0, was used to perform data cleaning and descriptive analyses. Modelling methods deviated from the protocol as, during preliminary examination of the outcome data, a non-linear trajectory was identified; therefore, this required alternative Structural Equation Modelling (SEM) methods. As such, latent growth curve modelling (LGCM) was performed in Mplus Version 8 (Muthén & Muthén, 1998–2017). The principal analysis was completed on an intention-to-treat basis for all sleep variables. A supplementary, per protocol analysis was limited to participants with complete data across all three time points.
Assessment of the occurrence and pattern of missingness was performed on the CSHQ item data. Items were found to be missing completely at random based on Little’s MCAR test at each assessment time point. 2 As such, imputation was conducted using the Expectation–Maximisation (EM) algorithm. Once item imputation was complete, the CSHQ total score was computed and rounded following standard rounding conventions.
Latent Growth Curve Modelling
Overall model fit was assessed using multiple fit indices: chi-square value and degrees of freedom; Akaike Information Criterion (AIC); Root-Mean-Square Error of Approximation (RMSEA); Comparative Fit Index (CFI); Tucker–Lewis Index (TLI); and Standardised Root-Mean-Squared Residual (SRMR). Recommended guidelines for good fit indicators were: non-significant chi-square value; RMSEA <0.06; CFI and TLI = 0.95; SRMR <0.08; and a smaller AIC was favoured (Akaike, 1974; Hu & Bentler, 1999). As the data were normally distributed, Maximum Likelihood Estimation (ML) 3 was used for the analyses as it handles missing outcome data (Muthén & Muthén, 1998–2017). Time was modelled as 0 (T0, baseline), 1 (T1, post-intervention), and 2 (T2, follow-up).
Growth modelling was performed to assess sleep scores at baseline and the direction and rate of change of sleep scores over time. Growth trajectories were modelled for the outcome variables CSHQ total score and actigraphically measured TST, SOL, and SE. Various growth functional forms for trajectories were examined, including intercept-only, linear, and freely-estimated 4 growth trends. Following the identification of a suitable growth model for the sleep outcomes, the impact of intervention delivery modality was assessed to determine if the models were statistically different from each other. Multiple-group SEM was conducted to compare intercepts and slopes between the intervention delivery modalities (in-person = 0 and telehealth = 1). Within Model A, no parameters were constrained; subsequently, Model B constrained parameters (simultaneously constraining intercept and slope) to equality. If constraining the model significantly diminished model fit, then parameters across the groups differ significantly. Invariance was assessed using chi-squared difference tests for each set of models, whereby a non-significant difference indicates model invariance. However, given the small sample sizes and potentially reduced power when comparing models for two separate groups, a supplementary analysis was conducted to assess delivery modality as a binary predictor (in-person = 0 and telehealth = 1) of intercept and slope terms from a model combining both intervention groups. We required that both methods of analysis produced the same finding.
Study 2
Method
Design
Study 2 consisted of the same sample as Study 1. 5 Since no significant differences in intercept and slope were found between intervention delivery modalities, Study 2 used a combined-group approach for all analyses.
Participants
The sample consisted of 78 children aged 5 to 12 years. Inclusion/exclusion criteria and demographic data collected were the same as for Study 1.
Measures
Study 2 utilised the sleep outcome data collected in Study 1. At baseline, participants and their primary caregivers completed measures (described below) assessing symptom presentation and impairment within home settings, attachment security (parent- and child-report), and family functioning.
Conners Comprehensive Behaviour Rating Scale–Parent (CBRS–P)
The CBRS–P (Conners, 2010). is a 203-item parent-report measure that assesses children for a range of emotions, behaviours, and academic difficulties using a Diagnostic and Statistical Manual of Mental Disorders (fifth ed; DSM-5; American Psychiatric Association, 2013) diagnostic framework. Items were rated on a 4-point scale from ‘not true at all’ to ‘very much true’. For this study, the CBRS-P captured two variables: (a) a symptom presentation complexity index (‘CBRS–P’) and (b) impact of symptom severity on home life (‘Home Impairment’). If the CBRS-P Symptom Count scale was ‘probably met’ and T scores were over 65, the disorder was coded as ‘yes’. For each participant, the number of CBRS-P disorders coded as ‘yes’ was totalled to create a CBRS-P symptom presentation complexity index. Home Impairment scale scores were classified on a 4-point scale from ‘never’ to ‘very often’.
Attachment Insecurity Screening Inventory (AISI)
The AISI is a parent-report measure of the parent-child attachment relationship in children aged 2 to 5 years (Wissink et al., 2016) and 6 to 12 years (Spruit et al., 2018). Both versions of the AISI consist of 20 items rated on a 6-point Likert scale from ‘never’ to ‘always’. Item scores were summed to represent a total score of attachment insecurity ranging from 20 to 120. Wissink et al. (2016) reported a total attachment insecurity (sub)clinical cut-off score of 46, with higher scores indicating greater attachment insecurity. The AISI has demonstrated good reliability and validity across age groups. In middle childhood, the AISI shows convergence with parent-reported socio-emotional functioning and attachment-related difficulties (Spruit et al., 2018). Factor-analytic studies also support a three-dimensional structure (avoidant, ambivalent, and disorganised) consistent with attachment theory, reinforcing its validity as a screening tool (Spruit et al., 2018).
McMaster Family Assessment Device General Functioning subscale (FAD-GF)
The FAD (parent report) assesses an individual’s perceptions of their family based on the McMaster Model of Family Functioning (Epstein et al., 1983; Miller et al., 1985). Within the FAD, the 12-item General Functioning subscale (FAD–GF) is widely used as a validated, short-form to assess global family functioning that, for example, incorporates levels of emotional support, warmth, and responsiveness, as well as communication, problem-solving, and appropriate behaviour regulation (Byles et al., 1988). The FAD-GF has excellent psychometric properties and high intercorrelations with the six FAD dimensions, and it is relevant to the Australian context (Boterhoven de Haan et al., 2015). FAD–GF items were rated on a 4-point Likert scale ranging from ‘strongly disagree’ to ‘strongly agree’, including six items describing healthy family functioning and six items regarding unhealthy family functioning. A mean score is calculated, ranging from 1 (healthy functioning) to 4 (unhealthy functioning), whereby higher scores indicate poorer family functioning.
Security Scale (SS)
Kerns’ Security Scale (SS) is a 15-item self-report measure designed to assess parental attachment security in middle childhood (8 to 12 years; Kerns et al., 1996) with robust psychometric properties (Brumariu et al., 2018). The items were administered using Harter’s (1982) format, in which children are presented with a statement that describes two types of children, for example, ‘Some kids find it easy to trust their [mum/dad], BUT Other kids are not sure if they can trust their [mum/dad]’. Children were instructed to identify which child they were most like and subsequently indicate whether this is ‘sort of true’ or ‘really true’ for them, scored on a 4-point scale. When completing the measure, children were instructed to think of their primary caregiver (Zajac et al., 2020). Item scores were averaged to produce a continuous measure of perceived attachment security (from 1 to 4), with higher scores indicating greater attachment security.
Statistical Analysis
Data cleaning and descriptive analyses were performed using SPSS 29.0 (IBM Corp, 2022). The EM algorithm was used to impute items within the SS after assessment using Little’s MCAR test
Study 1
Results
Sample Characteristics
Of the 206 families who contacted the researchers, 92 were deemed eligible following screening, and 78 consented to participate (see Figure 2) exceeding the minimum sample size of 68 estimated by the power analysis. This modest oversampling was intentional to safeguard against higher-than-expected attrition and ensure adequate statistical power was maintained. Child participants aged from 5 to 12 years old (M = 8.79 years, SD = 1.90) were allocated across both trial arms. See Table 1 for baseline characteristics. Total number of parent-reported co-occurring diagnoses ranged from 0 to 3, with most children having one additional diagnosis (44.9%); 9.0% had two diagnoses, and 5.1% had three diagnoses. The most commonly reported co-occurring diagnoses across both treatment groups were anxiety (28.2%), autism (12.8%), specific learning disorders (12.8%), and ‘other’ conditions (12.8%, e.g., tic disorder and language disorder).
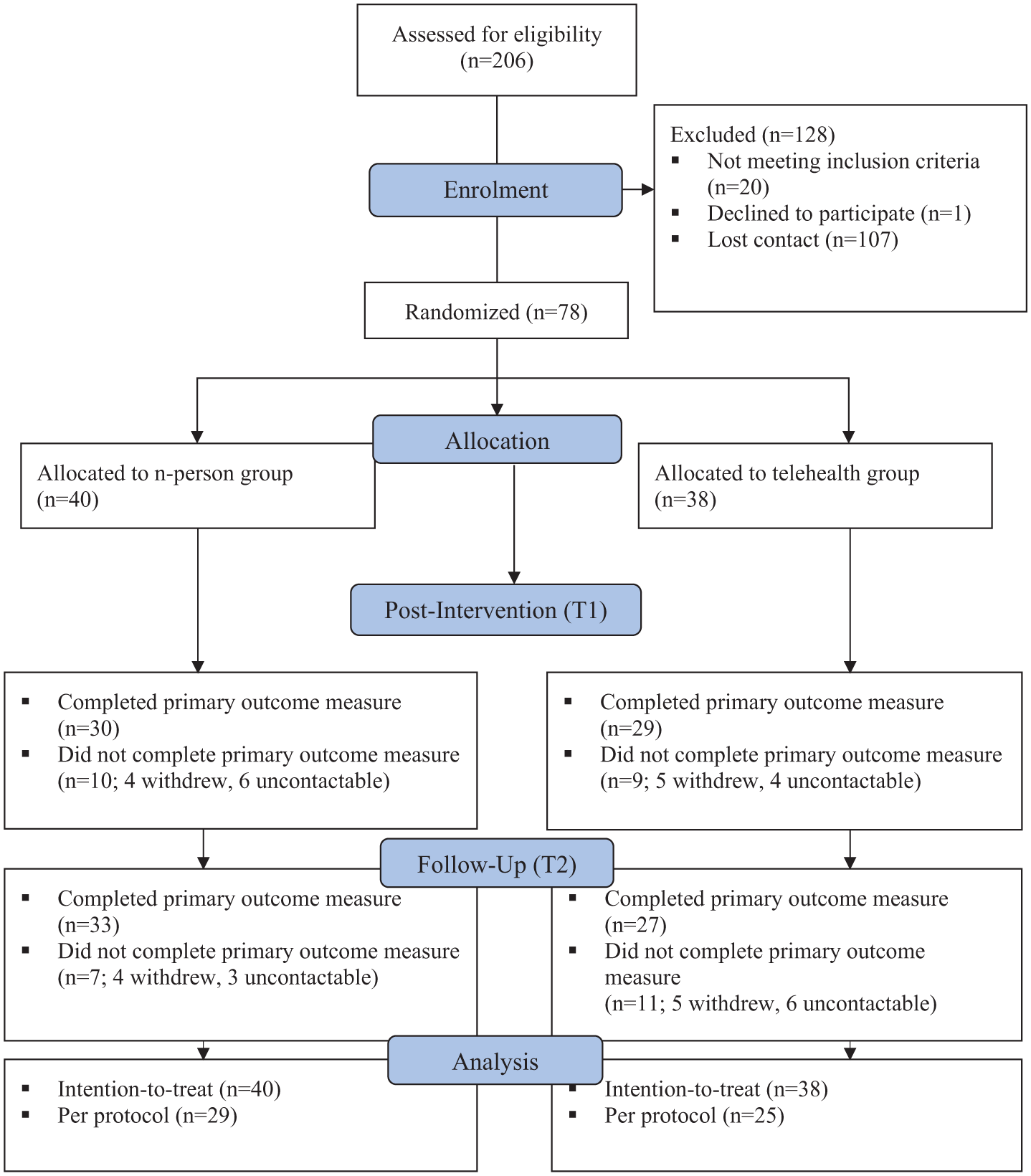
CONSORT flowchart of participants.
Baseline Characteristics of Participants.
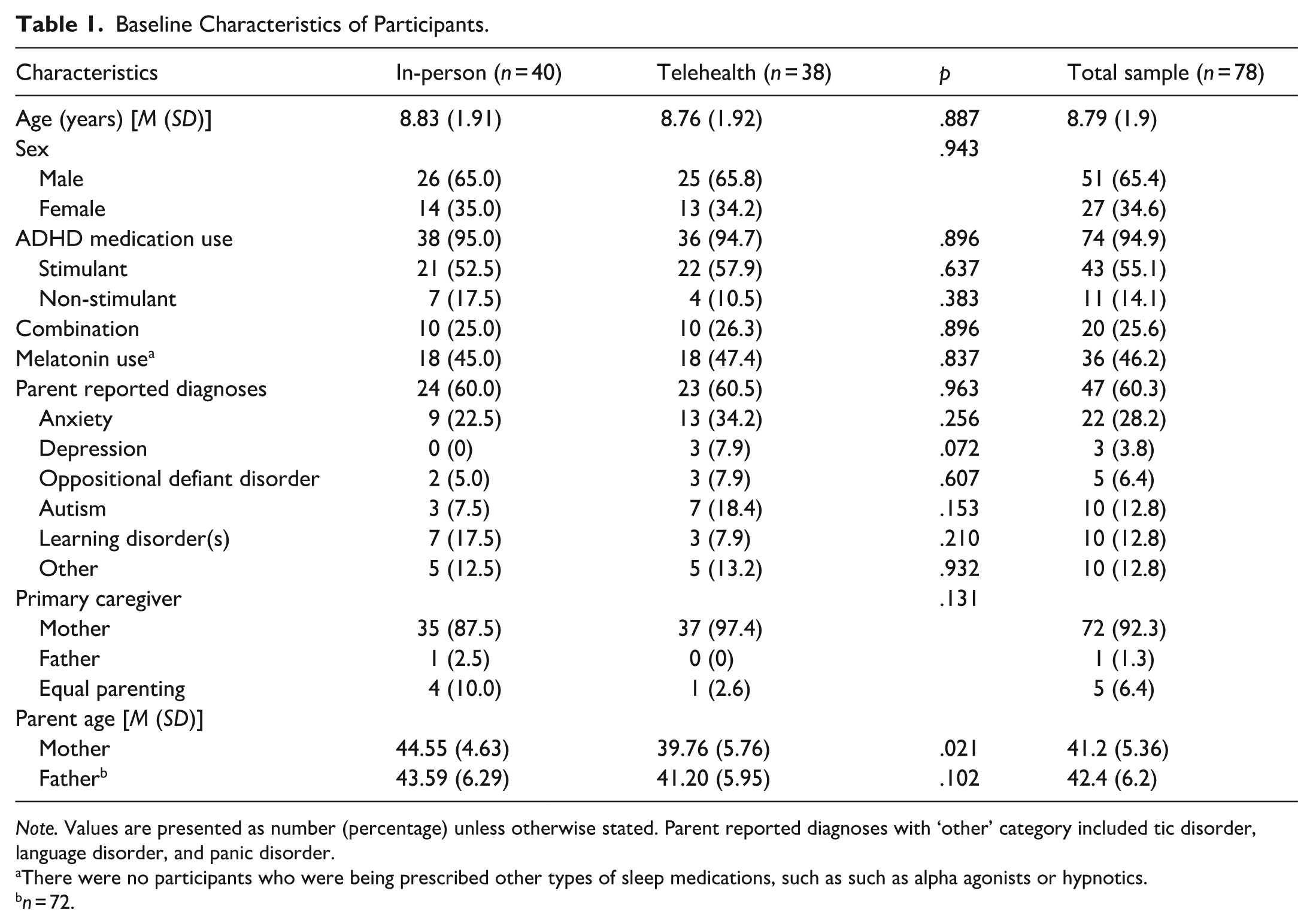
Note. Values are presented as number (percentage) unless otherwise stated. Parent reported diagnoses with ‘other’ category included tic disorder, language disorder, and panic disorder.
There were no participants who were being prescribed other types of sleep medications, such as such as alpha agonists or hypnotics.
n = 72.
Most children were prescribed either stimulant medication, non-stimulant medication, or a combination of both. Primary caregivers were more likely to be mothers (92.3%). Additionally, mothers in the telehealth delivery group were younger than mothers in the in-person group. There were no other demographic differences between groups. The means and standard deviations of all outcomes for both delivery modalities are presented in Table 2. Actigraphy data were collected for 75 participants, and complete adherence (per protocol), that is, usable data for three time points, was available for 31 participants (41.3%; in-person [n = 13], telehealth [n = 18]).
Outcome Scores by Group and Time.
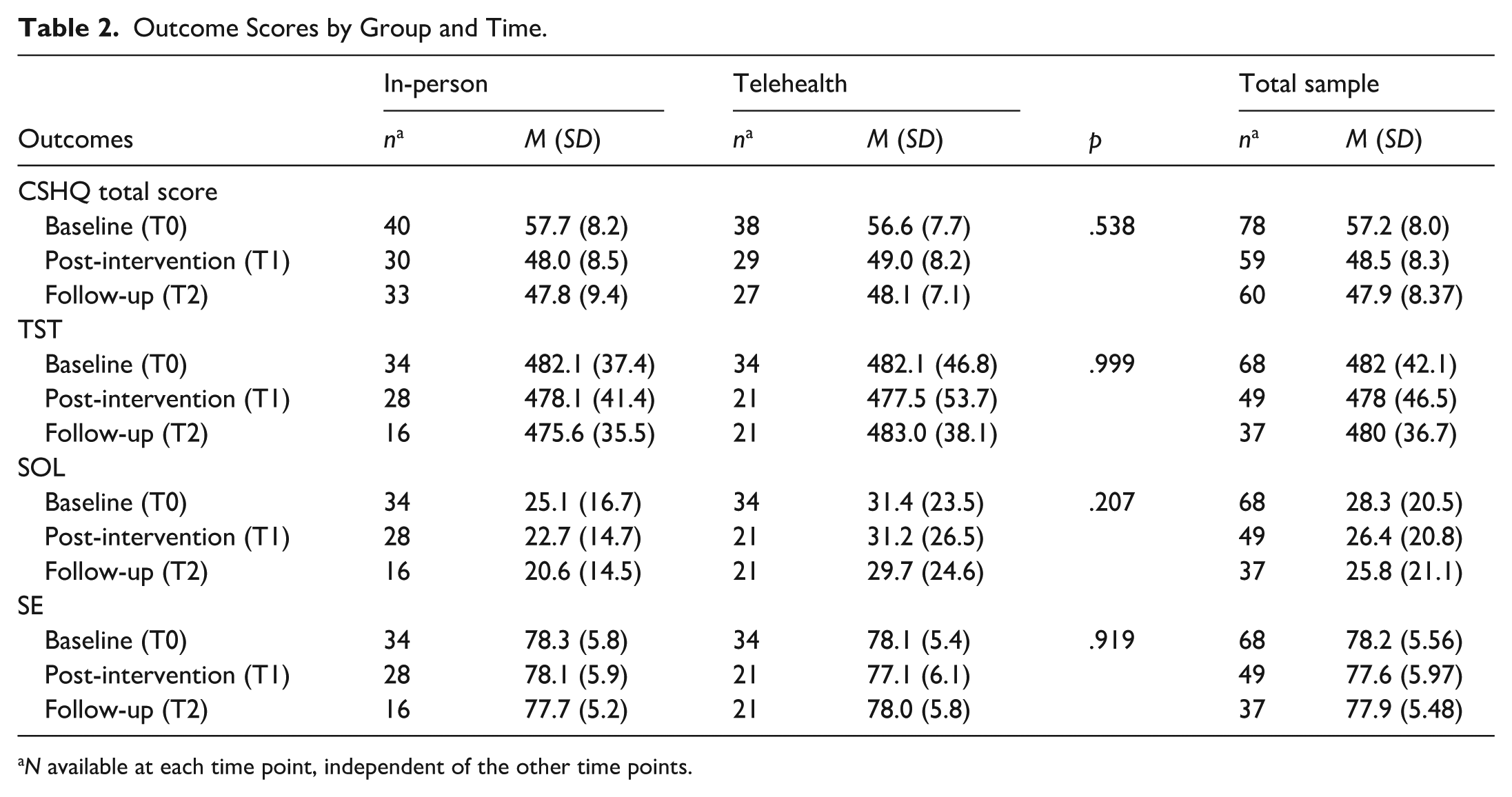
N available at each time point, independent of the other time points.
Programme Fidelity
Overall, 72 families (92.3%) received the first consultation (mean duration = 62 min, range: 30–90 min), 66 families (84.6%) received the second consultation (mean duration = 51 min, range: 20–75 min), and 61 families (78.2%) received the telephone call (mean duration = 25 min, range: 5–60 min). Per the consultation forms, children experienced an average of 1.8 sleep-related diagnoses and received 2 management strategies. Sleep-related diagnoses 6 were anxiety (n = 32), bedtime resistance (n = 17), sleep onset association (n = 19), insomnia (n = 9), delayed sleep onset (n = 23), restless leg syndrome (n = 2), nightmares (n = 7), night terrors (n = 3), sleep walking (n = 2), early morning waking (n = 7), night waking (n = 2), co-sleeping (n = 1), and diagnosis not reported (n = 3). Comorbidity of sleep-related diagnoses was noted, with participants reporting between one and five diagnoses: 1 (n = 32), 2 (n = 25), 3 (n = 7), 4 (n = 4), and 5 (n = 1).
Intention to Treat Analyses
Model 1: CSHQ Total Scores
A freely-estimated growth trajectory with an estimated slope factor for T2 (time = 1.16) was identified as the best-fitting single growth model for CSHQ total scores (Table 3; Model 1) based on fit indices and interpretability. Supplemental Table S1 outlines the different forms of growth trends that were considered. No significant relationship was found between baseline CSHQ total score (intercept term) and the reduction (slope term) in reported sleep disturbance over time following intervention (r = −.06, p = .982).
Best-Fitting Single Growth Curve Models for CSHQ Total Scores, TST, SOL, and SE.
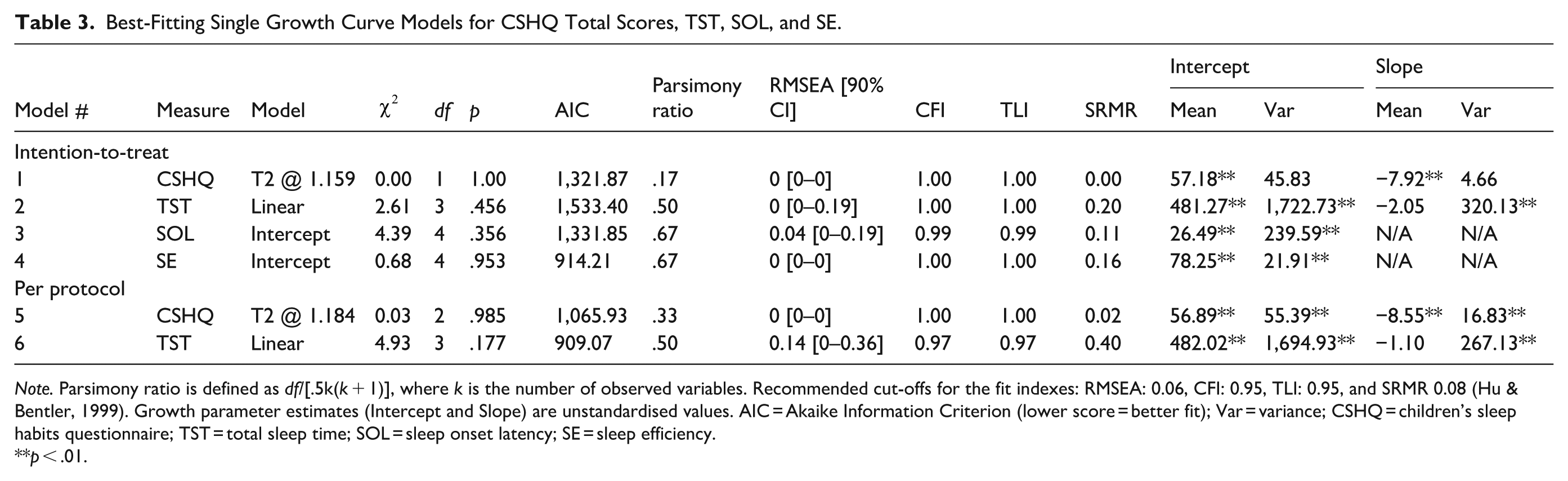
Note. Parsimony ratio is defined as df/[.5k(k + 1)], where k is the number of observed variables. Recommended cut-offs for the fit indexes: RMSEA: 0.06, CFI: 0.95, TLI: 0.95, and SRMR 0.08 (Hu & Bentler, 1999). Growth parameter estimates (Intercept and Slope) are unstandardised values. AIC = Akaike Information Criterion (lower score = better fit); Var = variance; CSHQ = children’s sleep habits questionnaire; TST = total sleep time; SOL = sleep onset latency; SE = sleep efficiency.
p < .01.
At baseline, the estimated mean CSHQ score was 57.18, which is indicative of sleep disturbance above the clinical threshold. A significant reduction in sleep problems was observed, representing an average decrease in CSHQ total scores of 7.92 points from baseline to post-intervention (T1). During the follow-up period (T1–T2), the rate of improvement slowed considerably, with an estimated additional reduction of 1.26 points, representing 15.9% of the initial rate of change. This resulted in a total estimated reduction of 9.17 7 points from baseline to follow-up. This pattern is reflected in the mean scores presented in Table 2, indicating that the most substantial gains occurred during the active intervention phase. These results suggest that there was a significant decline in the severity of subjectively reported sleep disturbance over time across groups.
Model 2: TST Scores
A linear model (with the variance of T0 and T2 fixed at 0 due to small negative residual variances) was selected as the best-fitting single-growth model for TST (Table 3, Model 2). See Supplemental Table S2 for the different forms of growth trends that were considered. Participants who had more severe parent-reported sleep disturbance at baseline had a greater decrease in their total sleep over time (r = −.58, p < .001). The estimated average TST scores at baseline varied significantly between participants. The linear slope term identified was non-significant; however, significant variability between participants was found.
Models 3 and 4: SOL and SE Scores
Intercept-only models were the best fitting single growth models for SOL and SE (Table 3, Models 3 and 4, respectively). See Supplemental Table S3 (SOL) and Supplemental Table S4 (SE) for the different forms of growth trends that were considered. At baseline, the estimated averages of SOL and SE were approximately 26 min and 78%, respectively. Both varied significantly between participants.
Per Protocol Analyses
Per protocol LGCM analysis was conducted for CSHQ total scores and TST scores only. Additional modelling was not completed for SOL and SE scores as intercept-only models were selected within the intention-to-treat analysis.
Model 5: CSHQ Total Scores
A freely estimated growth trajectory with an estimated slope factor for T2 (time = 1.184; variance of T0 fixed at 0) was selected as the best-fitting single growth model for per protocol CSHQ total scores (Table 3, Model 5; see Supplemental Table S5). A significant relationship was found between baseline CSHQ total scores (intercept term) and the decrease (slope term) in reported sleep disturbance over time following intervention (r = −.52, p < .001). At baseline, the estimated average CSHQ total score was 56.89 points (intercept), which varied significantly between participants. The estimated slope term identified a significant reduction of 8.55 points between baseline and post-intervention, which also varied significantly between participants. Overall, from baseline to follow-up, a decrease of 10.12 points 8 was found; consistent with the estimated improvement for the entire sample. Similar to the intention-to-treat growth curve, a shallower slope of change was found from post-intervention to follow-up.
Model 6: TST Scores
A linear model (with the variance of T0 and T2 fixed at 0 due to small negative residual variances) was identified as the best-fitting single growth model for per protocol TST scores (Table 3, Model 6; see Supplemental Table S6). A significant relationship was found between baseline TST scores (intercept term) and the decrease (slope) in total sleep time (r = −.54, p < .001). The estimated average TST score at baseline was 482.02 min, and a non-significant linear slope showed a 1.10-min decrease over time per visit. Both baseline and rate of change estimates demonstrated significant variability between participants, suggesting individual differences in the degree of change in TST with intervention.
Effect of Intervention Delivery Modality
To compare the trajectories (slope) of sleep outcome changes between participants who received the intervention in-person and via telehealth, a multiple-group LGCM was performed on the best-fitting single growth curve models where significant slope variance was identified: Model 5 (CSHQ, per protocol); Model 2 (TST, intention-to-treat); and Model 6 (TST, per protocol). Although the multiple-group LGCM was planned for the CSHQ intention-to-treat analysis, this was not completed, as no significant slope variance was found. Table 4 outlines goodness of fit statistics and growth parameter estimates for the models.
Multiple-Group Analysis Comparing In-person and Telehealth Intervention Delivery.
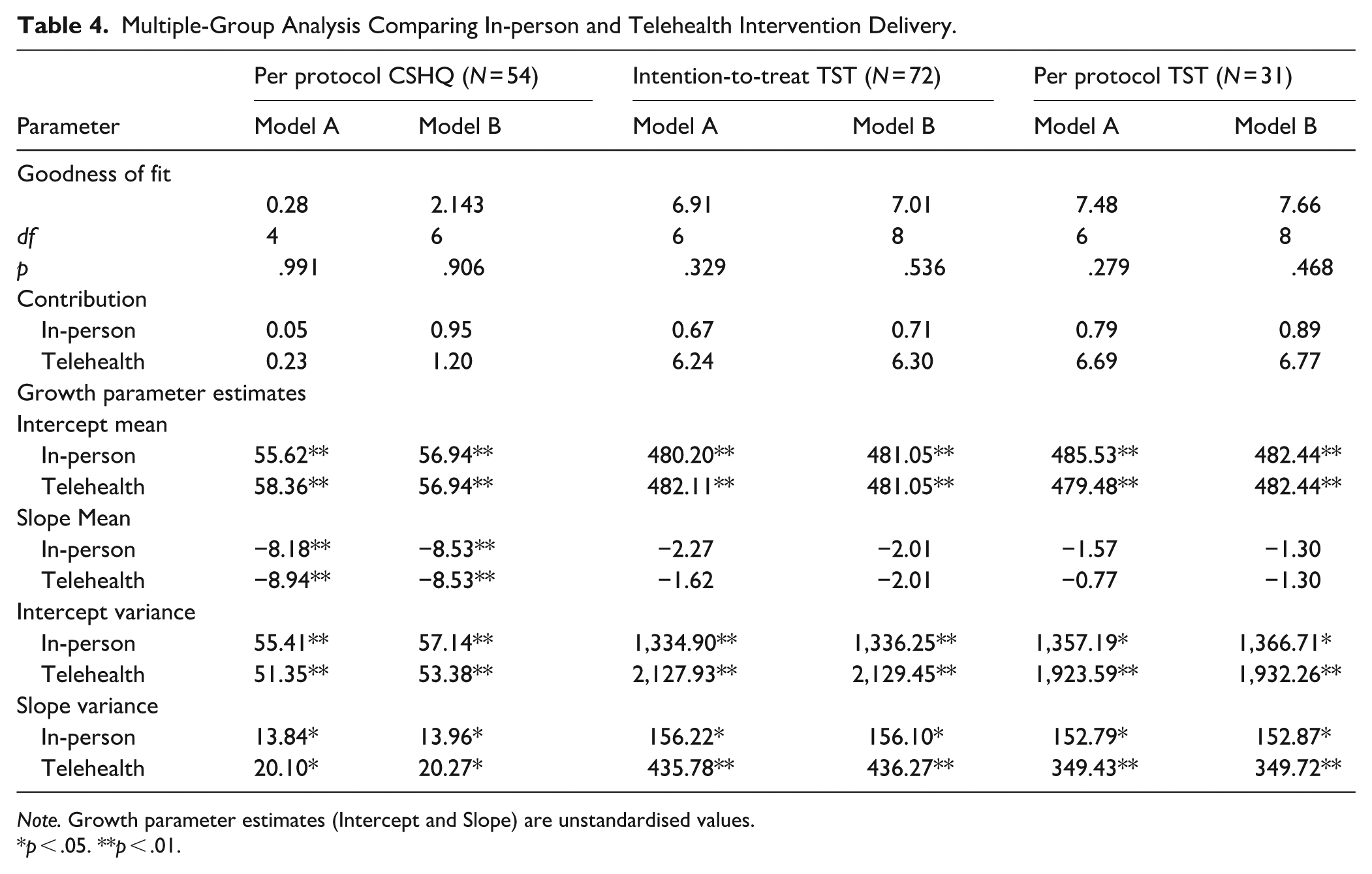
Note. Growth parameter estimates (Intercept and Slope) are unstandardised values.
p < .05. **p < .01.
CSHQ Total Scores
Figure 3(A) illustrates the estimated growth trajectories for both groups. Models A and B were not statistically significantly different
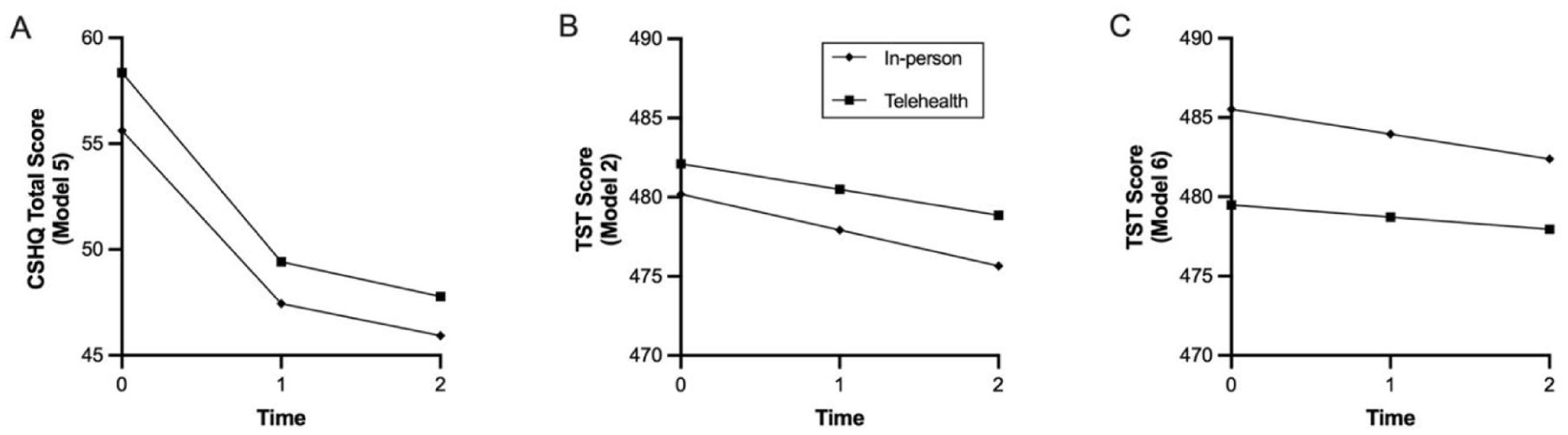
Between-group estimated growth trajectories. Panel A: Growth trajectories for the per protocol CSHQ total scores (Model 5). Panel B: Growth trajectories for the intention-to-treat TST scores (Model 2). Panel C: Growth trajectories for the per protocol TST scores (Model 6).
TST Scores
Within the intention-to-treat TST model, despite an apparent difference in slope means between participants who received the intervention in person and those who received the intervention via telehealth, when intercepts and slopes were constrained to be the same, this did not affect fit (Models A and B were not statistically different; (2) = 1.10, p = .951), indicating no significant difference in the rate of change between intervention delivery modalities (Figure 3(B)). This finding was confirmed when delivery modality was regressed onto Model 2 (slope = 0.03, p = .864), with no significant effect on the rate of change found.
A similar finding was identified within the per protocol TST model, whereby an apparently higher slope mean for the in-person group compared to the telehealth group was not significant. Again, Models A and B were not statistically different χ2 ((2) = 0.18, p = .914), indicating no significant difference in the rate of change between intervention delivery modalities (Figure 3(C)). Furthermore, no effect of delivery modality on change over time was found when regressed onto Model 6 (slope = 0.02, p = .892).
Reliable Change Index (RCI)
Reliable change analyses were conducted for per protocol participants between T0 and T2. Per the Reliable Change Index (RCI, Jacobson & Traux, 1991) scores greater than 1.96 reflect positive reliable change, scores lower than −1.96 indicate negative reliable change, and scores between −1.96 and 1.96 were classified as showing no reliable change. Within our per protocol sample, 44.4% (n = 24) of participants reliably improved and 55.6% n = 30) demonstrated no reliable change, noting no participants showed reliable deterioration.
Study 2
Results
Descriptive Statistics
Rating scale outcomes are presented in Table 5. Participants mostly met the criteria for three symptom scales on the CBRS-P, with their symptoms being rated as seriously affecting their home life ‘occasionally’ (24.0%), ‘often’ (38.7%), or ‘very often’ (30.7%). Only 6.7% of parent respondents rated symptoms as having no functional impairment within home settings. On the CBRS-P Symptom scales, externalising symptoms were common, with oppositional defiant symptoms reported in 54.7% and conduct disorder symptoms in 22.7%. Internalising symptoms were also endorsed, particularly separation anxiety (54.7%), depression (49.3%), and generalised anxiety (36%). Symptom ratings consistent with other conditions, such as obsessive-compulsive disorder (30.7%), social anxiety (21.3%), and autism (12%) were reported at lower frequencies.
Outcome Statistics for Baseline Measures.
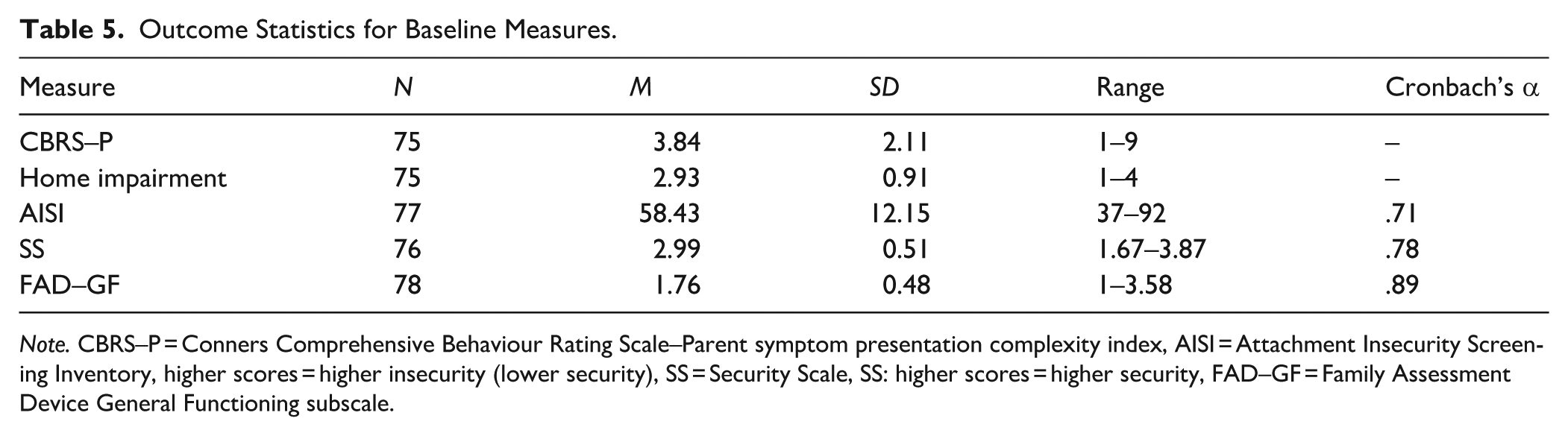
Note. CBRS–P = Conners Comprehensive Behaviour Rating Scale–Parent symptom presentation complexity index, AISI = Attachment Insecurity Screening Inventory, higher scores = higher insecurity (lower security), SS = Security Scale, SS: higher scores = higher security, FAD–GF = Family Assessment Device General Functioning subscale.
Mean total attachment insecurity (AISI) was above the (sub)clinical cut-off, indicating that, by parent report, participants demonstrated high levels of insecure attachment. By contrast, the mean SS score signalled strong perceived attachment security. The FAD–GF participant scores were moderately skewed (0.80), representing a tendency towards healthy family functioning.
Predictors of Sleep Outcome
For the intention-to-treat CSHQ model, participants with greater attachment insecurity (higher AISI) and greater family dysfunction, respectively (higher FAD–GF scores), had higher levels of sleep disturbance at baseline (Table 6, Model 5a). The variance in baseline sleep disturbance was explained more by the FAD–GF score than the AISI score (estimate = 4.16 vs. 0.16).
Models of Sleep Outcomes Predicted by Age, Sex, Co-occurring Conditions, Attachment (In)Security, and Family Functioning.
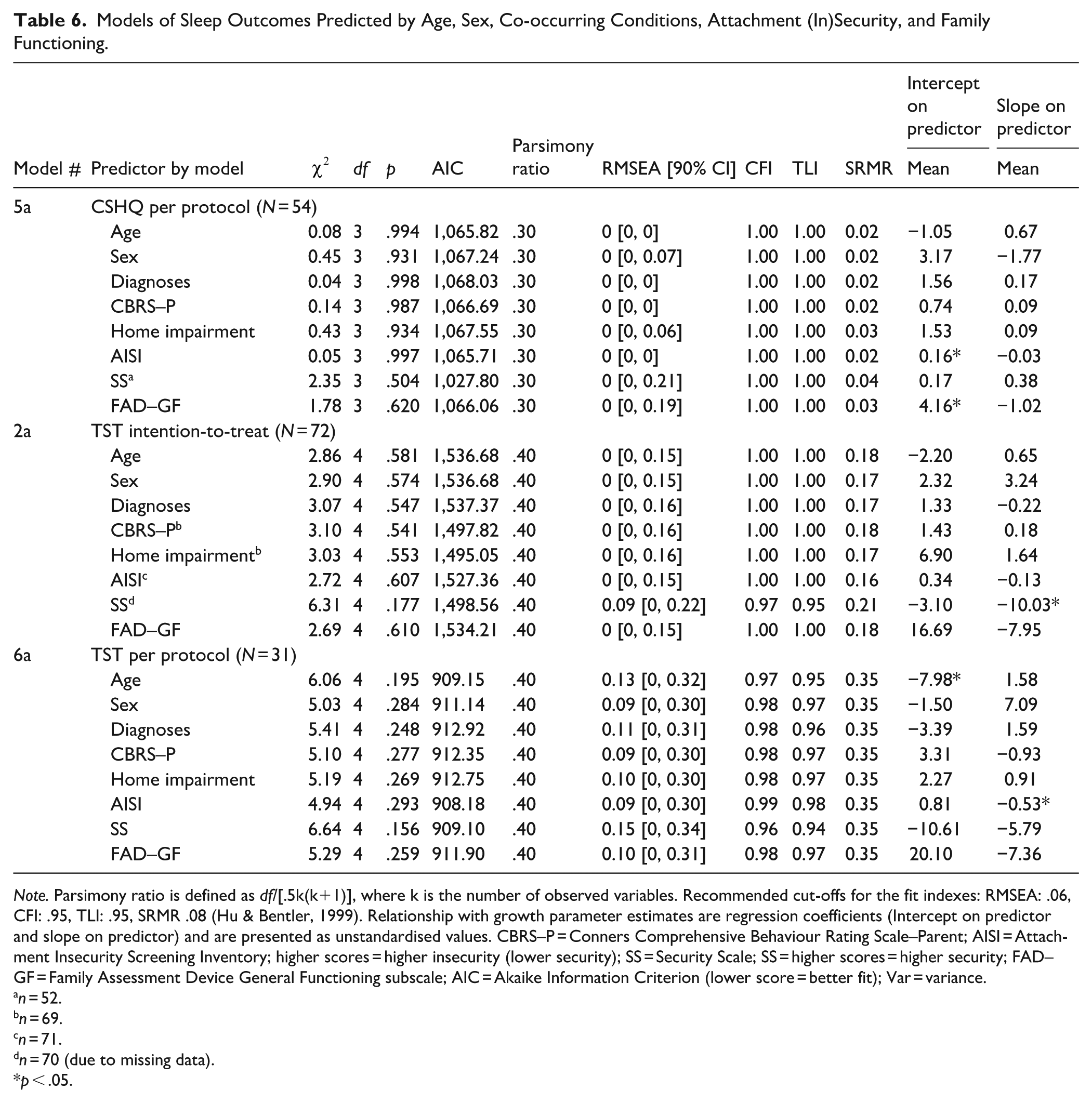
Note. Parsimony ratio is defined as df/[.5k(k+1)], where k is the number of observed variables. Recommended cut-offs for the fit indexes: RMSEA: .06, CFI: .95, TLI: .95, SRMR .08 (Hu & Bentler, 1999). Relationship with growth parameter estimates are regression coefficients (Intercept on predictor and slope on predictor) and are presented as unstandardised values. CBRS–P = Conners Comprehensive Behaviour Rating Scale–Parent; AISI = Attachment Insecurity Screening Inventory; higher scores = higher insecurity (lower security); SS = Security Scale; SS = higher scores = higher security; FAD–GF = Family Assessment Device General Functioning subscale; AIC = Akaike Information Criterion (lower score = better fit); Var = variance.
n = 52.
n = 69.
n = 71.
n = 70 (due to missing data).
p < .05.
However, no variables significantly predicted the rate of change in sleep disturbance on the CSHQ over time. Within the intention-to-treat TST model, children who reported greater attachment security (SS scores) at baseline demonstrated a steeper reduction in TST over time (Table 6, Model 2a). Whereas within the per protocol TST model (Table 6, Model 6a), younger participants slept for longer at baseline, and higher parent-reported attachment insecurity (AISI scores, i.e., poorer attachment) were associated with a significantly faster decrease in TST over time.
Discussion
Study 1 implemented an RCT sleep intervention for children aged 5 to 12 years with ADHD and compared outcomes between in-person and telehealth treatment modalities. Support was found for the first hypothesis that children’s subjective sleep outcomes would improve following intervention. At baseline, children across both treatment modalities had total sleep disturbance scores above the clinical cut-off of 41. Following intervention, a reduction of approximately 9 points occurred, with a greater reduction shortly after intervention completion compared to the 3-month follow-up. Although the improvement in parent-reported sleep disturbance indicated a significant reduction in the severity of sleep initiation and/or maintenance problems, total scores were still above the CSHQ clinical cut-off at the final follow-up. Additionally, within our per protocol sample RCI analyses the results suggested that individual changes varied such that nearly half of the participants experienced reliable improvement, while the remaining participants did not experience reliable change.
This finding is in keeping with past research, which has reported CSHQ total scores above the clinical cut-off following intervention despite a significant reduction in sleep problem severity (Shokravi et al., 2016). In fact, both Hiscock et al. (2015) and Corkum et al. (2016) reported a similar CSHQ reduction (approximately 8 points) at their 2- and 3-month follow-up, respectively. This consistency with past research provides insight into the moderate but meaningful improvement that may be seen following a brief sleep intervention. The results also suggest that families may require follow-up booster sessions to sustain gains, particularly to accommodate potential changes in children’s sleep habits due to developmental maturation. Alternatively, variability in treatment response may reflect moderating factors or limitations in the sensitivity of the CSHQ for detecting clinically meaningful change in children with ADHD.
Regarding objective sleep outcomes measured by actigraphy, maintenance of sleep efficiency was consistent with previous Sleeping Sound research; though Hiscock et al. (2015) found an increase in total sleep time in their subsample of 54 children, reporting a small effect. Corkum et al. (2016), in their Better Nights/Better Days RCT, reported that their online intervention group exhibited improved sleep onset latency at follow-up, although sleep duration remained unchanged. The difference in actigraphy results with our findings may be due to challenges with actigraphy adherence encountered in our study. Nonetheless, it is not uncommon for researchers to report a discrepancy between subjective and objective (actigraphy) outcomes in ADHD sleep interventions. For example, Roberts et al. (2019) found that objective sleep improvements were less apparent via actigraphy in their sample of children with both autism and ADHD.
Secondly, support was found for the hypothesis that telehealth would perform similarly to in-person delivery in terms of treatment response. This is in keeping with past research supporting the use of telehealth interventions for ameliorating childhood insomnia in children without ADHD (McLay et al., 2020). Thus, our results suggest that, regardless of delivery modality, Sleeping Sound helps to reduce sleep disturbance in school-aged children. This is an important finding, particularly as telehealth-delivered interventions offer flexible accessibility of treatment for those with limited access to in-person services. While there is limited research to date investigating the utility of online ADHD sleep interventions (Gabarron et al., 2025), a UK team is developing a digital parent-guided behavioural intervention for chronic insomnia in children with ADHD (the DISCA study) to improve sleep and other outcomes related to child behaviour and parental well-being, although results are not yet available (Hornsey et al., 2023). However, our findings are in keeping with a similar Australian RCT which evaluated the telehealth delivery of a brief behavioural Sleeping Sound intervention in children 5 to 12 years with autism, who reported a significant group by time difference in child sleep also using the Children’s Sleep Habits Questionnaire (Lewis et al., 2024).
Contrary to expectation, the presence of greater symptom complexity did not predict baseline sleep problem severity or treatment outcome despite a substantial burden of externalising and internalising symptoms within our sample. This finding suggests that the presence of additional symptom complexity in children with ADHD may not result in a cumulative negative effect on sleep problems, contrasting with past research reporting more severe sleep problems in children with ADHD and co-existing conditions (Bondopadhyay et al., 2022; Dimakos et al., 2024). Nonetheless, our findings do indicate that the Sleeping Sound intervention is an effective treatment for children with ADHD who present with functionally significant co-existing symptoms of oppositional behaviour, anxiety, depression, and autism.
Our results are in keeping with Hvolby et al.’s (2008) report that co-occurring oppositional defiance disorder and anxiety did not aggravate sleep problems in children with ADHD. Bond et al. (2024) reported no significant differences in CSHQ scores between children with ADHD and those who had additional comorbid conditions. Sciberras et al. (2020) also did not find in their RCT that the presence of co-occurring conditions moderated sleep treatment outcomes in Australian children (5–13 years old) with ADHD.
However, whilst the CBRS parent ratings provide insight into the social, emotional, and behavioural challenges exhibited by children with ADHD in our community sample, the symptom scales do not represent formal medical diagnoses, potentially accounting for why our hypotheses were not supported. Parent report symptomatology may be affected by subjective perceptions, and the limited range of co-occurring conditions within the sample may have reduced the variability needed to detect meaningful effects.
Conversely, partial support was found for our hypothesis that children with greater attachment insecurity and poorer family functioning had higher levels of subjective sleep disturbance at baseline. This finding is consistent with past research that reported better child–mother security was associated with decreased daytime sleepiness in children and fewer subjective sleep problems (Keller & El-Sheikh, 2011). Our findings underscore the importance of family factors on sleep quality and suggest that ADHD sleep treatments should consider environmental influences and incorporate treatment goals that incorporate a biopsychosocial framework to improve child-parent attachment and clinical outcomes.
Secondly, limited support was found for the hypothesis that children with lower symptom complexity, stronger attachment security, and healthier family functioning would demonstrate a greater treatment response. Whilst there were no identified predictors for the rate of change of total sleep disturbance scores, greater child-reported attachment security and lower parent-reported attachment security (i.e., greater insecurity) predicted a steeper decline in total sleep over time. One possible reason for this finding is that securely attached children are more likely to experience consistent emotional regulation and parental responsiveness, which may facilitate the consolidation of more efficient sleep routines (Perpétuo et al., 2021). In this context, reductions in TST may reflect increased sleep efficiency (less time in bed awake, fewer night-time awakenings) rather than a detrimental decrease in restorative sleep. Secure attachment has been linked to greater adaptability to new behavioural routines and improved coping with transitional stressors (Adams et al., 2014), both of which could support the goals of Sleeping Sound. Another explanation for this could be that attachment security may affect how receptive children are to behavioural interventions, given the Sleeping Sound focus on reducing caregiver involvement during the night (Åslund et al., 2018; Whittall et al., 2021). Parental withdrawal at night may be interpreted negatively by the child and result in an attachment rupture, impacting treatment outcomes. Indeed, Hornstra et al. (2022) compared consequent versus antecedent-based techniques as moderating factors to behavioural parent training effectiveness for children with ADHD and found consequent-based techniques to be less effective for those more securely attached to their caregivers but effective for more insecurely attached children. Parents of children who report more secure attachment may be more accommodating if their child shows resistance to the intervention strategies at the cost of adhering to the treatment protocol (Berliner et al., 2020). Conversely, parents who report a poorer attachment relationship may not be as responsive to the needs of their child and/or may experience greater difficulty implementing sleep strategies, subsequently impacting treatment effectiveness (Koehn & Kerns, 2018). Interestingly, the value of the TST rate of change was greater for the child-report compared to the parent-report measure. This finding may suggest that the child’s perception of the attachment relationship quality, rather than the parent’s perception, may be more central to understanding the relationship between parent-child attachment and sleep (Molina & Musich, 2016).
Although preliminary, Study 2’s findings suggest an influence of parent-child attachment and family functioning on sleep problem severity in children with ADHD. Moreover, the findings may indicate an interaction between intervention techniques and parent-child attachment, particularly from the child’s perspective, which could contribute to worsening sleep problems. However, further investigation is needed to explore the possible influence of parenting behaviours (e.g., responsiveness, support of the child’s autonomy, and behavioural control) in relation to parent-child attachment on sleep outcomes.
Nonetheless, to adapt Sleeping Sound for families with lower attachment security or less stable caregiving contexts, we propose integrating attachment-focussed components such as emotion-coaching and bedtime relational rituals, increasing parental sensitivity to distress at night, and explicitly teaching caregivers how to buffer separation anxiety at bedtime. In addition, providing guided bedtime routines and consistency training, along with psychoeducation on supporting children’s emotional regulation, could help anchor sleep behaviour change within a more responsive caregiving framework – potentially increasing children’s capacity to adopt the behavioural sleep strategies. This approach aligns with a trauma-informed and attachment-informed online sleep intervention used in a foster-care population, which emphasises relational regulation as a foundation for sleep behaviour change (Alfano et al., 2023).
Strengths, Limitations, and Future Directions
To the best of our knowledge, this is the first RCT to compare a telehealth intervention to an in-person modality within a paediatric ADHD population. Treatment groups were well matched on demographic and clinical variables at baseline. The study employed both subjective and objective measures of sleep, enabling a more comprehensive assessment of treatment outcomes. A consistent between-group reduction in total sleep disturbance was found across both intention-to-treat and per-protocol analyses. These results suggest that telehealth performed as well as standard in-person care, which is valuable for those experiencing geographical and/or structural barriers to treatment. Additionally, our findings provide supporting evidence for the effectiveness of Sleeping Sound for children with ADHD and are in keeping with similar research, demonstrating improvement in subjectively rated sleep disturbance at 3 months post-intervention (Hiscock et al., 2015).
Although most children with ADHD in our study presented with at least one co-existing condition, some studies examining behavioural sleep interventions have excluded participants with co-existing conditions (e.g., Mehri et al., 2020; Shokravi et al., 2016). Whilst our results suggested that the complexity of symptom presentation may not influence the severity of sleep problems or response to intervention, they did support the broader generalisability of this sleep intervention across children with ADHD with varying levels of comorbidity.
However, several study limitations merit consideration. It is possible that higher-functioning families self-selected to participate in the study. Sample characteristics were notable for a higher proportion of maternal respondents (92%) and male participants (65%), also potentially limiting generalisability. Although no sex differences were observed in the current study, further research is warranted, given evidence that females with ADHD have a significantly higher risk for any sleep disorder compared with males (Ahlberg et al., 2023). It is also noteworthy that this study was conducted during the COVID-19 pandemic, with fluctuations in public health restrictions and community transmission. These contextual factors may have influenced participant engagement, retention, and sleep patterns due to illness, isolation requirements and increased child and/or parent stress. Indeed, complete actigraphy adherence was low, with data loss impacted by lost devices, illness, and refusal to wear the device. Sensory sensitivities may have contributed to non-adherence, especially as 12% of the sample had a co-occurring diagnosis of autism. Whilst actigraphy data were collected for 96% of the sample, usable data across all three time points were available for only 41%. It is difficult to determine whether this level of attrition is typical, given the limited reporting of actigraphy adherence rates for paediatric ADHD cohorts in the published literature. Furthermore, to reduce reporting bias, we supplemented subjective parent-reported outcomes with objective sleep indices (actigraphy). However, we acknowledge that because parents both delivered the intervention and reported outcomes, the lack of independent assessment may have introduced bias and should be considered when interpreting the results. Future trials should place greater emphasis on objective measures to strengthen the study design.
Due to the nature of the study design, families could not be blinded to their delivery modality status, thus potentially introducing expectancy bias and/or affecting compliance. Prior to randomisation, some families expressed a preference for telehealth as they did not live within the metropolitan area or were unwilling to travel. Conversely, some parents preferred in-person treatment due to technological barriers, privacy concerns, and therapeutic reasons. Consequently, it may be beneficial for future research to replicate this study as a pragmatic trial, allowing families to select their intervention delivery modality to maximise engagement.
Finally, no significant predictors were found to explain the inter-individual variability within the reduction in total sleep disturbance over time, possibly due to limited sample size and/or selection of individual factors. Future research should consider cross-sectional and longitudinal assessment of factors relevant to ADHD as predictors of treatment response. Factors could be considered at the individual (e.g., other symptoms, temperament, circadian rhythm/light exposure, melatonin use, and exercise), family (e.g., attachment, parenting behaviours, caregiver mental health, and family functioning), and environmental (e.g., socioeconomic status) level (Becker, 2020; Bronfenbrenner, 1979; Meltzer, 2017). Exploration of these dimensions and how they relate to the trajectory of sleep problems in children with and without ADHD with different sleep profiles can help us better understand the risk factors associated with poor sleep and poorer treatment response to inform theoretical models, as well as assessment and intervention methods (Becker, 2020; Williamson et al., 2019).
Conclusion
Given the prevalence of sleep disturbance in paediatric ADHD, effective and accessible treatment is important. The current study extends the literature on behavioural sleep interventions by providing supporting evidence for a telehealth-delivered version of Sleeping Sound. Telehealth promotes the accessibility of psychological services for families, therefore improving barriers to health equity. Furthermore, preliminary evidence was found for the potential relevance to sleep of parent attachment quality in children with ADHD. In the hopes of supporting more effective sleep interventions and improving functional outcomes, future research exploring the associations between sleep disturbance, attachment insecurity, and family-level factors is recommended.
Supplemental Material
sj-docx-1-jad-10.1177_10870547251393071 – Supplemental material for Does Delivery Mode Matter? A Randomised Controlled Trial of the Sleeping Sound© Intervention: Predicting Sleep Outcomes in Children With ADHD
Supplemental material, sj-docx-1-jad-10.1177_10870547251393071 for Does Delivery Mode Matter? A Randomised Controlled Trial of the Sleeping Sound© Intervention: Predicting Sleep Outcomes in Children With ADHD by Maya K. Malkani, Andrew M. C. Sheridan, Alison J. Crichton, Romola S. Bucks and Carmela F. Pestell in Journal of Attention Disorders
Footnotes
Acknowledgements
We thank the families and clinicians who took part in this study for their valuable contribution. The authors would also like to thank Associate Professor Emma Sciberras and Professor Harriet Hiscock for the use of the Sleeping Sound© ADHD intervention program within this trial, as well as their helpful advice and collaboration.
Ethical Considerations
The University of Western Australia Human Research Ethics Committee (2021/ET000637) approved the study. Written informed consent was obtained from all participants and/or their legal guardians for participation in the study and for the publication of these findings.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data supporting this study’s findings are not publicly available due to confidentiality but are available from the corresponding author upon reasonable request.
Supplemental Material
Supplemental material for this article is available online.
Notes
Author Biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
