Abstract
Objective:
In recent years, it has been emphasized that various growth factors that affect neurogenesis may lead to ADHD. In this study, we aimed to investigate the role of VEGF, IGF-1, and HIF-1α growth factors in the etiopathogenesis of ADHD.
Method:
Levels of VEGF, IGF-1, and HIF-1α were compared between 40 ADHD children and 40 healthy children, aged 7 to 13 years.
Result:
VEGF, IGF-1, and HIF-1α levels did not significantly differ between the groups. There was a negative correlation between serum VEGF levels and the parent-rated T-DSM-IV-S (AD) subscale. There was a positive correlation between serum IGF-1 levels and the parent-rated T-DSM-IV-S (AD) subscale, and SDQ (ES) subscale.
Conclusion:
Given our limitations and the fact that some of our findings differ from those of other studies, it is evident that this area requires additional research with larger samples.
Introduction
ADHD, one of the most prevalent neurodevelopmental disorders, is characterized by persistent and maladaptive inattention, hyperactivity, and impulsivity, and its prevalence in children and adolescents is estimated to be 5.9% to 7.1% worldwide (Munro et al., 1993; Willcutt, 2012). Studies on the etiology of ADHD indicate that the disorder is caused by interactions between genetic, neuronal, and environmental factors (Thapar & Cooper, 2016). However, the disorder’s heterogeneous clinical presentation encourages researchers to continue investigating its cause. Recently, with the development of neuroimaging techniques and the increase in knowledge about the development and functions of various brain regions, interest has shifted to the various factors that influence brain development in ADHD. In the scientific literature, the number of studies examining the appropriateness of using various growth factors influencing brain development as biomarkers in ADHD is rising.
During nerve development, many growth factors (GF) regulate complicated processes including neurogenesis, gliogenesis, cell migration, cell differentiation, and synapse formation (Costales & Kolevzon, 2016; Galvez-Contreras et al., 2017). Increasing evidence suggests that various growth factors enhance motor, cognitive, and sensory abilities (Galvez-Contreras et al., 2016). Vascular Endothelial Growth Factor (VEGF), a growth factor effective in angiogenesis, vasculogenesis, and vascular function, plays a crucial role in embryogenesis as well as prenatal and postnatal brain development (Jesmin, Togashi, Sakuma, et al., 2004; Takahashi & Shibuya, 2005). During the formation of the brain and neural tubes, neuronal cells produce VEGF, which controls vascularization (Ruiz de Almodovar et al., 2009). VEGF is a potential biomarker for predicting various neurodevelopmental disorders and their clinical severity due to its significant influence on neuronal development. The literature contains few studies examining the relationship between VEGF and ADHD. In two distinct animal experiments conducted by Jesmin et al., case groups exhibited significantly lower VEGF levels compared to controls (Jesmin, Togashi, Mowa, et al., 2004; Jesmin, Togashi, Sakuma, et al., 2004). Similarly, Yurteri et al. (2019) discovered that the serum VEGF levels of children with ADHD were lower than those of healthy children. In some studies, however, VEGF levels did not differ significantly between ADHD patients and controls (Torun et al., 2019).
Insulin-like growth factor-1 (IGF-1), a growth factor that mediates the effects of growth hormone and is effective in fetal development, childhood, and adolescence, is also thought to be associated with ADHD (Juul, 2003; Laron, 2001). IGF-1, like VEGF, plays a crucial role in several stages of central nervous system development (Russo et al., 2005). These effects on the central nervous system make IGF-1 an additional potential biomarker for predicting neurodevelopmental disorders and their severity. According to the findings of Wang et al., the ADHD group had lower IGF levels than the control group (Wang et al., 2023). In addition, a few studies have demonstrated that ADHD medication alters IGF-1 levels relative to the treatment baseline (Bereket et al., 2005; Mei et al., 2022; Sisley et al., 2013; Wang et al., 2022). Nevertheless, some studies did not find a significant difference in IGF-1 levels between people with ADHD and controls or after treatment for ADHD (Kim et al., 2020; Toren et al., 1997).
A transcription factor known as hypoxia-inducible factor 1α (HIF-1α) protects cells from damage caused by hypoxia (Semenza, 1998). The fact that HIF-1a activates the transcription of the VEGF gene, which is believed to play a role in neurodevelopmental processes, encourages investigation into its function in the etiopathogenesis of ADHD (Forsythe et al., 1996; Yurteri et al., 2019). It also regulates genes associated with angiogenesis, erythropoiesis, iron metabolism, glucose metabolism, cell proliferation, cell survival, and apoptosis, in addition to its contribution to VEGF effects (Semenza, 1998). It is further evidence that HIF-1a contributes to the pathogenesis of ADHD that these processes differ between persons with ADHD and healthy people (Gungor et al., 2021; Scassellati et al., 2020) . Although it has been shown that the development of ADHD may be linked to conditions such as hypoxia and ischemia, which are peripheral manifestations of HIF-1a, to our knowledge there is no study explicitly examining HIF-1a levels in ADHD patients (Miguel et al., 2015; Ohshima et al., 2016). Another way that HIF-1a is linked to ADHD is that proinflammatory cytokines like TNF-alpha and IL-1B, which cause HIF-1a to become active in normoxic conditions, also play a role in how ADHD develops (Jung et al., 2003; Koç et al., 2023; Zhou et al., 2003). All of these factors support the study of HIF-1a as a significant biomarker in the etiopathogenesis of ADHD.
Although a few studies have examined VEGF and IGF levels in individuals with ADHD, the number of studies investigating the relationship between these potential biomarkers and the clinical severity of the disease appears to be insufficient for drawing a general conclusion. In addition, although there are studies investigating HIF-1a and its associated effects in a variety of neurodevelopmental disorders, there is no study examining the relationship between HIF-1a and ADHD in the reviewed literature. Investigating these biochemical parameters in ADHD will help us better understand some etiopathogenetic pathways that have not yet been clearly elucidated due to the heterogeneous clinical presentation of the disorder.
In this study, we aim to compare VEGF, IGF-1, and HIF-1α levels between children with ADHD and healthy controls, as well as examine the relationship between these biochemical parameters and the severity of ADHD symptoms and the psychiatric comorbidities that frequently accompany ADHD. We hypothesize, based on previous research, that these biochemical parameters will differ between individuals with ADHD and healthy controls and correlate with the severity of the disorder.
Method
Participants
The ADHD group consisted of 40 children between the ages of 7 and 13 admitted to the Child and Adolescent Psychiatry outpatient clinic of the Faculty of Medicine at Suleyman Demirel University (SDU), who were newly diagnosed with ADHD and had not received any medical treatment previously. Exclusion criteria included the presence of a major physical or neurological illness (e.g., cardiovascular disorders, epilepsy, etc.), schizophrenia, bipolar disorder, autism spectrum disorder, tic disorder, substance abuse, head trauma, a history of taking psychotropic medications, a history of corticosteroid or immunomodulatory drug use in the last 6 months, or an intelligence quotient (IQ) score below 70 according to the Turkish version of the Wechsler Intelligence Scale for Children—Revised. (WISC-R) (Savasır & Şahin, 1995; Wechsler, 1974).
The control group consisted of unrelated 40 healthy children admitted to the outpatient clinics of the Medical Faculty at SDU, who volunteered along with their parents and were matched in terms of age, sex, socioeconomic status, sector, and ethnicity. For the control group that did not fulfill ADHD criteria and had no history of other mental, neurological, or metabolic diseases, the same exclusion criteria were performed. All of the study procedures were made by the Declaration of Helsinki and local laws and regulations. This study was approved by the local ethics committee. The parents who agreed to have their children take part were provided with a patient information sheet that outlined the research protocol; the parents also provided their written informed consent. Oral assent was also procured from all of the children.
Diagnostic and Symptomatic Assessment
The researchers used an interview form to collect data on the socio-demographic features of the participants. Each participant underwent a detailed diagnostic evaluation by a child/adolescent psychiatrist by using the Schedule for Affective Disorders and Schizophrenia for School-Aged Children, Present and Lifetime Version (K-SADS-PL) (Gökler et al., 2004; Kaufman et al., 1997), and ADHD diagnoses were made according to the DSM-5 criteria (Munro et al., 1993). Parents and teachers completed the Turgay DSM IV-Based Child and Adolescent Behavioral Disorders Screening and Rating Scale (T-DSM-IV-S) (Ercan et al., 2001; Turgay, 1994). Parents also completed the Strengths and Difficulties Questionnaire (SDQ) (Goodman et al., 2000; Güvenir et al., 2008). Children completed the Revised Child Anxiety and Depression Scales—Child Version (RCADS-CV) (Ebesutani et al., 2012; Gormez et al., 2017).
Blood Samples
The patients and controls fasted overnight. From 08.00 to 10.00 a.m, venous blood samples were taken from the antecubital vein to determine the VEGF, IGF-1, and HIF-1α serum/plasma concentrations. Venous blood samples of the patients included in the study were taken into biochemistry tubes/EDTA-containing tubes and centrifuged at 3,000 rpm for 10 min and the serum/plasma portion was obtained. The serum sample obtained was stored at −80°C until the working day. The levels of human HIF-1α, VEGF, and IGF-1 were determined by using commercial enzyme-linked immunosorbent assay (ELISA) kits [Elabscience, Wuhan, China; Catalog No: E-EL-H6066, E-EL-H0111, E-EL-H0086 respectively]. Standards, control serums, and samples were run in duplicate in each assay according to kit instructions. To determine the levels of mentioned parameters in patient samples, a standard concentration-optic density graphic was used. HIF-1α assay included 7-point calibrators as 62.5, 125, 250, 500, 1,000,2,000, 4,000 pg/ml. The sensitivity and detection range of the assay were 37.5 pg/ml and 62.5 to 4,000 pg/ml, respectively. VEGF assay included 7-point calibrators as 31.25, 62.5, 125, 250, 500, 1,000, and 2,000 pg/ml. The sensitivity and detection range of the assay were 18.75 pg/ml and 31.25 to 2,000 pg/ml, respectively. IGF-1 assay included 7-point calibrators as 1.56, 3.13, 6.25, 12.5, 25, 50, 100 ng/ml. The sensitivity and detection range of the assay were 0.94 ng/ml and 1.56 to 100 ng/ml, respectively.
Statistics
Statistical analyses were conducted by using the SPSS (Statistical Package for the Social Sciences) 26.0 statistical software. Variables were expressed either as a number (n) or as a mean ± standard deviation (SD). The Chi-square test was used to examine categorical variable group differences. Using Kolmogorov–Smirnov one-sample tests, the normal distribution of continuous variables in patients and controls was calculated. When the normality of the distribution of the variables was acceptable, the Student’s t-test or, in other cases, the Mann–Whitney U test was used to compare the variables. The Spearman rank correlation coefficient was used to evaluate correlations between continuous variables.
Results
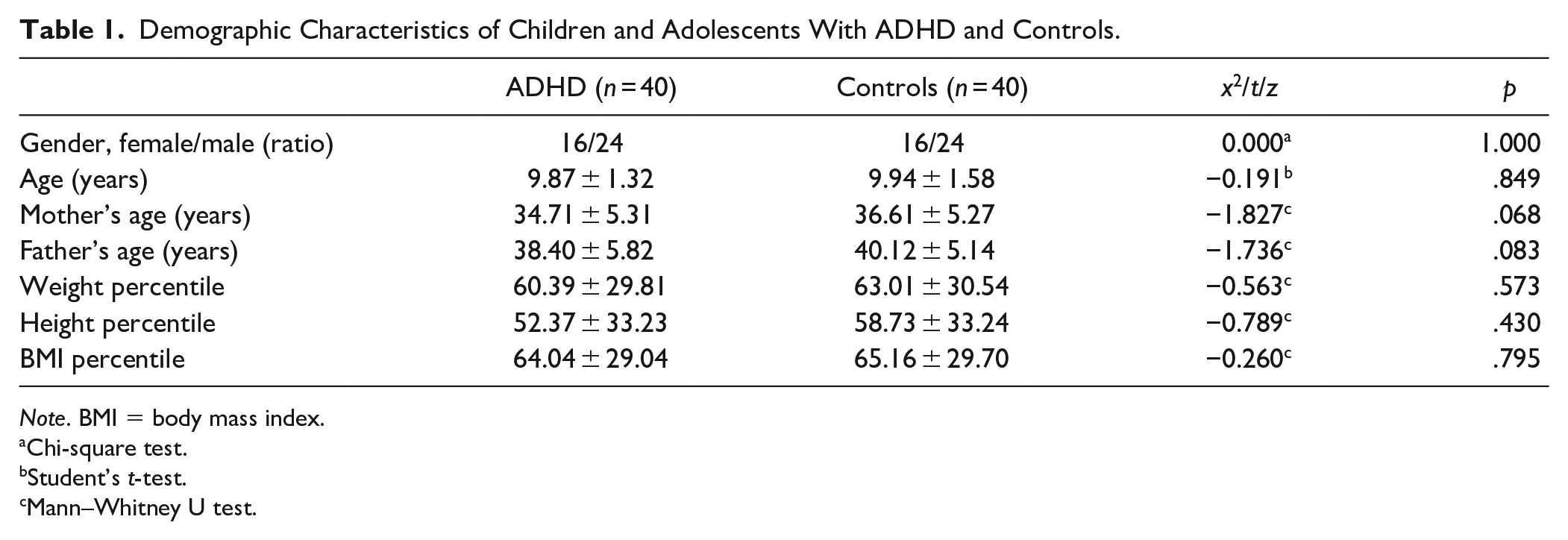
In the study, there were 40 children with ADHD (16 females and 24 boys), who had an average age of 9.87 ± 1.32 years, and 40 control kids (16 females and 24 males), whose average age was 9.94 ± 1.58 years (t = −0.191, p = .849). Age, sex, parents’ ages, weight percentile, height percentile, and BMI percentile were all statistically similar among the groups (Table 1).
Demographic Characteristics of Children and Adolescents With ADHD and Controls.
Note. BMI = body mass index.
Chi-square test.
Student’s t-test.
Mann–Whitney U test.
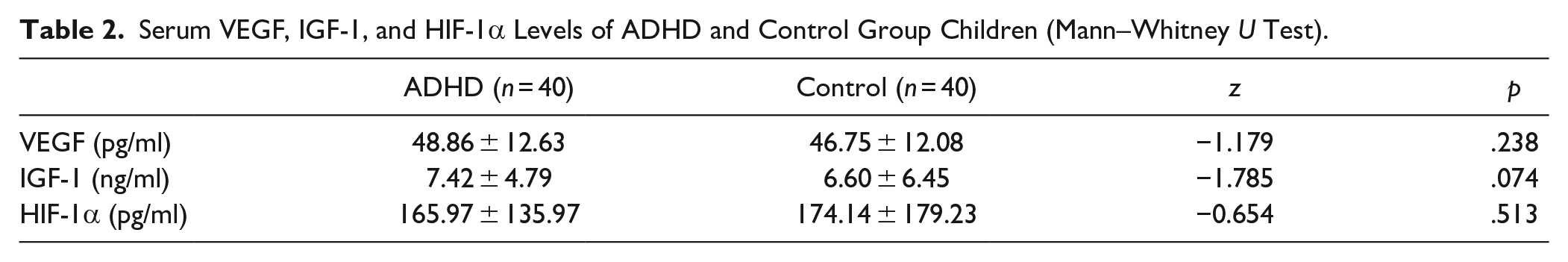
Mean serum VEGF levels were 48.86 ± 12.63 pg/ml in the ADHD group and 46.75 ± 12.08 pg/ml in the control group; mean serum IGF-1 levels were 7.42 ± 4.79 ng/ml in the ADHD group and 6.60 ± 6.45 ng/ml in the control group; and mean serum HIF-1α levels were 165.97 ± 135.97 pg/ml in the ADHD group and 174.14 ± 179.23 pg/ml in the control group. VEGF, IGF-1, and HIF-1α levels did not significantly differ between the groups (Table 2).
Serum VEGF, IGF-1, and HIF-1α Levels of ADHD and Control Group Children (Mann–Whitney U Test).
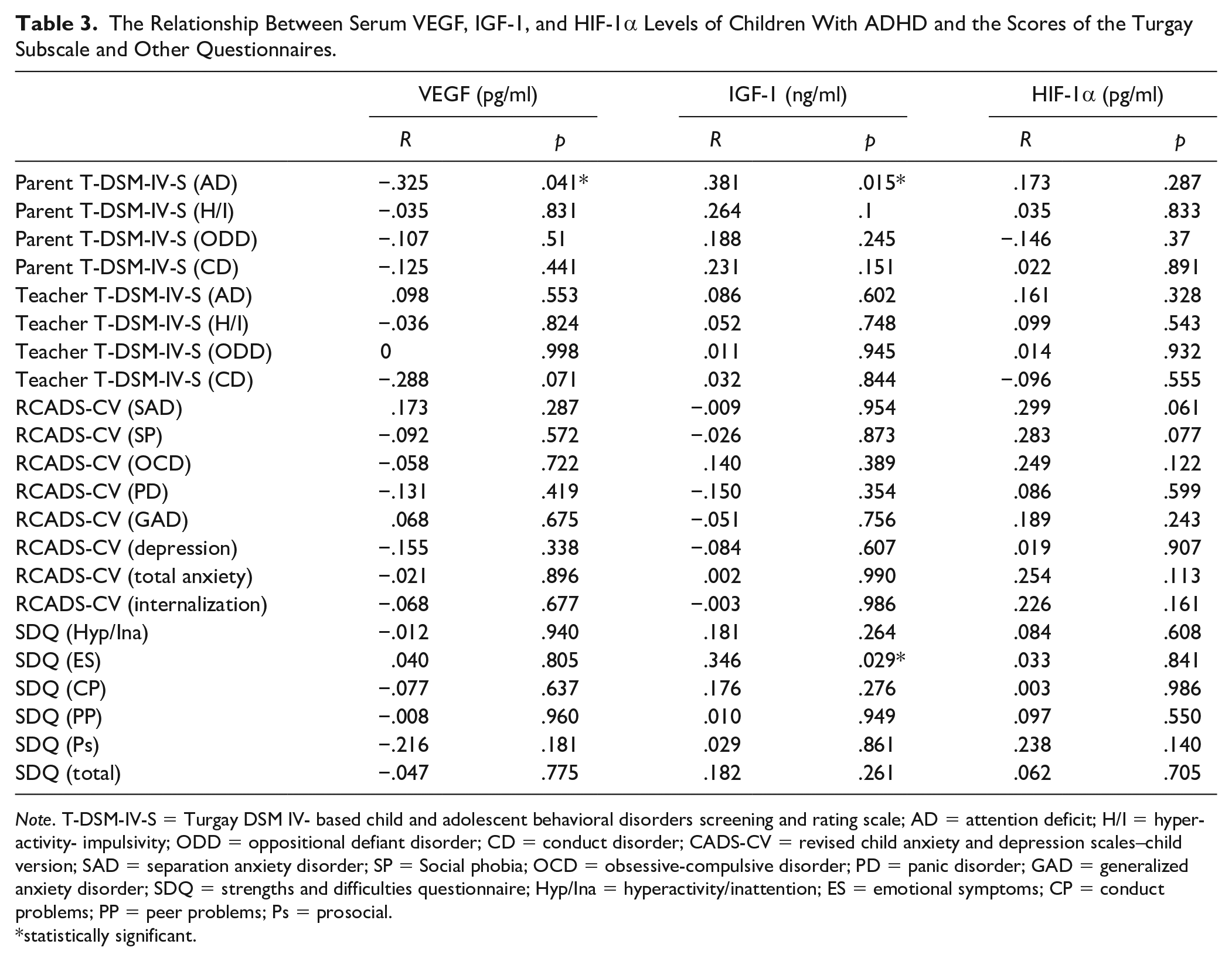
In addition, the associations between the parent- and teacher-rated T-DSM-IV-S, the total and subscale scores of the RCADS-CV, SDQ questionnaires, and the serum levels of VEGF, IGF-1, and HIF-1α in the ADHD patient group were investigated (Table 3).
The Relationship Between Serum VEGF, IGF-1, and HIF-1α Levels of Children With ADHD and the Scores of the Turgay Subscale and Other Questionnaires.
Note. T-DSM-IV-S = Turgay DSM IV- based child and adolescent behavioral disorders screening and rating scale; AD = attention deficit; H/I = hyperactivity- impulsivity; ODD = oppositional defiant disorder; CD = conduct disorder; CADS-CV = revised child anxiety and depression scales–child version; SAD = separation anxiety disorder; SP = Social phobia; OCD = obsessive-compulsive disorder; PD = panic disorder; GAD = generalized anxiety disorder; SDQ = strengths and difficulties questionnaire; Hyp/Ina = hyperactivity/inattention; ES = emotional symptoms; CP = conduct problems; PP = peer problems; Ps = prosocial.
statistically significant.
There was a statistically significant negative correlation between serum VEGF levels and the parent-rated T-DSM-IV-S attention deficit (AD) subscale (r = −.325, p = .041), and a significant positive correlation between serum IGF-1 levels and the parent-rated T-DSM-IV-S AD subscale (r = 0.381, p = 0.015). In addition, a positive correlation was seen between serum IGF-1 levels and the emotional symptoms subscale of the Strengths and Difficulties Questionnaire. There was no correlation between the total and subscale scores of the other questionnaires and serum levels of VEGF, IGF-1, or HIF-1α.
Discussion
In our investigation, we compared the serum concentrations of VEGF, IGF-1, and HIF-1α between ADHD patients and healthy controls. We also examined the association between these biochemical markers and the symptoms of ADHD and other psychiatric disorders that may be present in this patient population. Although a few research have examined VEGF and IGF-1 levels in ADHD, to the best of our knowledge, this is the first study to investigate HIF-1α levels in ADHD. Serum levels of VEGF, IGF-1, and HIF-1α did not differ significantly between patient and control groups. Moreover, there was a negative correlation between VEGF levels and the parent-rated T-DSM-IV-S attention deficit (AD) subscale, whereas there was a positive correlation between IGF-1 levels and the parent-rated T-DSM-IV-S attention deficit (AD) subscale and the SDQ emotional symptoms subscale.
In previous animal trials, VEGF levels were reported to be lower in the ADHD group compared to the control group, but Torun et al., similar to our study, discovered no difference between the patient group and controls in their investigation (Jesmin, Togashi, Mowa, et al., 2004; Jesmin, Togashi, Sakuma, et al., 2004; Torun et al., 2019). This may be because animal models do not fully depict the pathophysiology of human ADHD. In the study by Yurteri et al. (2019), the ADHD group had lower VEGF levels than the control group, contrary to our findings. In addition, we discovered a negative link between VEGF levels and the parent-rated T-DSM-IV-S attention deficit (AD) subscale when examining the correlation between VEGF levels and scores on the scales we employ. Yurteri et al. (2019) measured the severity of illness symptoms using the Du Paul ADHD Rating Scale and the Strengths and Difficulties Questionnaire—Parent Version and examined their link with VEGF. In contrast to our findings, they discovered a negative correlation between the hyperactive/inattentive and conduct SDQ subscales and serum VEGF levels. This difference may be attributable to the sample’s varied clinical features. For instance, the heterogeneous clinical presentation of ADHD, that is, which of the inattentive dominant subtype, hyperactivity dominant subtype, or combined subtype was more prevalent in the study group, may have influenced VEGF levels. The existence of studies demonstrating substantial differences in VEGF levels between these subtypes strengthens our argument (Yurteri et al., 2019). Some studies that examined VEGF levels in persons with ADHD also examined the effect of treatment on VEGF levels (Oakes et al., 2019). Due to the absence of an ADHD treatment group in our investigation, we were unable to analyze the effect of drug therapy on VEGF levels. This should be factored into future work plans.
The majority of studies examining IGF-1 levels in ADHD have focused on how ADHD medications alter IGF-1 levels. In investigations comparing serum IGF-1 levels between drug-naive ADHD patients and healthy controls, no difference was observed, which is generally consistent with our findings (Kim et al., 2020; Toren et al., 1997). In the study conducted by Wang et al., IGF levels were shown to be lower in the ADHD group than in the control group, which contradicts our findings (Wang et al., 2023). The small size of our study group may have contributed to this difference. Examining the correlation between IGF-1 levels and scores on the measures we use, we have identified a positive relationship between IGF-1 levels and the parent-rated T-DSM-IV-S attention deficit (AD) subscale and SDQ emotional symptoms subscale. Given the role of IGF-1 in neuronal development in regions of the brain believed to be associated with neurocognition and mood, the increase in IGF-1 observed in children with increased affective symptoms and attention problems may be a form of compensation (Das et al., 2015; Trejo et al., 2008). The absence of a study examining comparable correlations in the literature renders our hypothesis regarding the cause of this increase unsupported. Clearly, additional research is required to obtain more precise information in this domain.
Although HIF-1α levels have been studied in some neurodevelopmental disorders or other psychiatric disorders, to the best of our knowledge, this is the first study to assess HIF-1α levels in ADHD. For instance, Shibata et al. examined the expression levels of HIF-1α mRNA in patients with mood disorders and found that HIF-1α levels were higher during the depressive period compared to the control group (Shibata et al., 2013). In their study examining HIF-1α levels in ASD, Şimşek et al. (2021) found no significant difference in HIF-1α levels between the ASD group and the control group, but HIF-1α levels were associated with irritability and hyperactivity in the ASD group. While there was no significant difference in HIF-1α levels between the ADHD group and the control group, we have been unable to establish a correlation between HIF-1α levels and the severity of symptoms in the patient group.
Our study is the first in several respects, but it has several limitations. First, it should be noted that our study group is relatively small in comparison to similar studies conducted in the past. Another problem is that our study didn’t include a group of people who were taking medicine for ADHD. This made it harder for us to figure out how ADHD drugs affect relevant serum markers. It should also be noted that the ideal location for measuring the levels of these biomarkers to determine their impact on brain development is the central nervous system. Blood concentrations measured in the periphery may not accurately reflect true concentrations.
In this study, we have examined the levels of VEGF, IGF-1, and HIF-1α in ADHD. We have also looked at how the severity of disorder symptoms was linked to these serum markers. Given our limitations and the fact that some of our findings differ from those of other studies, it is evident that this area requires additional research with larger samples.
Footnotes
Acknowledgements
We thank to the patients and their family members who participated in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this study was provided by a grant from the Scientific Research Project Coordination Unit of Süleyman Demirel University (Project No: TTU-2019-7441).
