Abstract
Objective:
No clear answer has yet been attained as to the influence of prenatal exposure to infection on autism spectrum disorder (ASD) and attention-deficit/hyperactivity disorder (ADHD), either alone or as co-occurring issues. The current study examined links between hospital-recorded and maternal-reported prenatal infections and ASD, ADHD, and co-occurring ASD and ADHD.
Methods:
Participants were n = 15,462 children and mother pairs from the Millennium Cohort Study (MCS), a population-representative UK sample.
Results:
Findings show associations between maternal-reported infections and ASD, and some evidence of links with ADHD and co-occurring ASD and ADHD. Hospital-recorded infections were not found to be associated with ASD, ADHD, or their co-occurrence. Agreement between hospital-recorded and maternal-reported infections was low, which may explain the discrepant findings.
Conclusion:
Prenatal maternal infections may be associated with increased odds of ASD and ADHD. Findings point to the importance of drawing on multiple sources of information when ascertaining prenatal infection status.
Previous research has suggested that prenatal maternal infections may contribute to increased odds of neurodevelopmental conditions such as autism spectrum disorder (ASD; Jiang et al., 2016) and schizophrenia (Khandaker et al., 2013). Few studies have, however, examined links with ADHD. Further, no studies, to our knowledge, have examined the relation between prenatal infection and co-occurring ASD and ADHD. In the current study, we seek to address this gap using data from a UK-representative cohort study. We use these data to evaluate the links between prenatal infection and two of the most common early neurodevelopmental disorders ASD and ADHD, as individual and co-occurring conditions.
ASD is characterized by difficulties in social communication as well as restricted, repetitive patterns of behavior, activities or interests (American Psychiatric Association, 2013), with an estimated global prevalence of around 1% to 2% (Lyall et al., 2017). ADHD comprises symptoms of inattention and/or hyperactivity and impulsivity (American Psychiatric Association, 2013) and is estimated to be more common than ASD, with a prevalence of 5% to 7% (Polanczyk et al., 2007; Thomas et al., 2015). ASD and ADHD are both heterogeneous conditions and frequently co-occur (Faraone et al., 2015; Masi et al., 2017). A recent meta-analysis (Lai et al., 2019) found that 28% of those with ASD also had ADHD. Symptom-level studies have further reported that 40% to 70% of individuals with ASD have ADHD symptoms, while around 15% to 25% of individuals with ADHD show symptoms of ASD (Antshel et al., 2016). The DSM-5 is the first edition to allow ADHD to be diagnosed in conjunction with ASD (American Psychiatric Association, 2013). As a result, it was challenging to study the risk factors for co-occurring ASD and ADHD prior to the publication of DSM-5 in 2013.
Considerable attention has been paid to the possible link between prenatal maternal infection and individual neurodevelopmental outcomes, such as ASD (Jiang et al., 2016) and schizophrenia (Khandaker et al., 2013). It has been suggested that infections requiring admission to hospital have stronger links to offspring ASD compared to other, milder infections (Jiang et al., 2016). The link between prenatal maternal infections and ASD is, however, not without controversy. For example, Croen et al. (2019) found that only infection accompanied by fever was associated with ASD, suggesting that this link may be due to the immune response to the infection rather than the infectious agent itself. A small number of studies have explored the relationship between prenatal maternal infections and ADHD, which has only recently been formally recognized as a neurodevelopmental disorder (American Psychiatric Association, 2013). These studies have yielded mixed results. Mann and McDermott (2011) found that prenatal genitourinary infections, gathered from records from physicians’ offices and hospitals, were associated with increased odds of ADHD. Dreier et al. (2016), utilizing self-reported exposure data, found increased odds of ADHD in relation to fever in gestational weeks 9 to 12 and genitourinary infections in weeks 33 to 36. A more recent study by Ginsberg et al. (2019) found associations between prenatal maternal infections and ADHD after controlling for various confounds and covariates. However, the associations were no longer statistically significant in sibling comparisons, which help adjust for additional confounds that are not easily accounted for in typical approaches to estimating the association between prenatal exposures and offspring outcomes. No study, to our knowledge, has studied links between prenatal maternal infections and co-occurring ASD and ADHD.
Previous studies have operationalized and measured prenatal infections in various ways. Two commonly used methods are patient self-report and hospital inpatient records. It can be argued that maternal-reported infections capture a broader range of infections than inpatient records, as less severe infections, treated at the level of general practice or in outpatient clinics, will not appear in inpatient records, but may still be reported by the women themselves. However, there are several well-known limits to self-reported data, compounded in the cases where the self-reports are retrospective. This is suboptimal from the perspective of obtaining as accurate-as-possible determinations of whether or not an infection is present. Most studies rely on a single source of information regarding prenatal infection, typically either maternal self-reports or hospital records.
The current study uses data from both inpatient hospital records and retrospective maternal self-reports to assess links between exposure to prenatal infections and offspring ASD, ADHD and co-occurring ASD and ADHD in a population-representative UK sample. Our aims were: (1) to examine whether previous results on links between hospital-recorded prenatal maternal infections and ASD would replicate in a representative UK cohort study when relevant confounders are taken into account; (2) to explore whether maternal-reported infections would be associated with increased odds of ASD, as the literature on these associations is not clear; (3) to add to the literature on links between prenatal infections and ADHD as well as co-occurring ASD and ADHD. We hypothesized that prenatal infections would be associated with increased odds of ASD, ADHD, and their co-occurrence irrespective of the source of information (maternal reports vs. hospital records) but that the association would be attenuated when adjusting for a range of relevant confounders.
Method
Cohort
Participants were the n = 15,462 children and mother pairs from the UK Millennium Cohort Study (MCS; Connelly & Platt, 2014) who had data available on both prenatal infections (maternal-reported or hospital-recorded) and children’s ASD or ADHD between the ages 5 and 14. The MCS is a longitudinal birth cohort study following over 18,000 families in the UK since the year 2000. At the time of this study, seven sweeps of data had been collected; when the children were the ages of 9 months, 3, 5, 7, 11, 14, and 17 years. The cohort was drawn from and stratified via the stratification of electoral wards in each of the four countries of the UK; England, Scotland, Wales, and Northern Ireland, providing information on families’ socioeconomic status. Families living in disadvantaged circumstances, as well as those of ethnic minorities, were overrepresented in the sample. As a consequence of these recruitment efforts, the MCS sample is representative of the UK population, in particular in terms of socioeconomic status, when appropriate design weights are used. For further information on sampling procedures, see Plewis (2007) and for more information on the cohort, see Connelly and Platt (2014) and documentation on the MCS website (https://cls.ucl.ac.uk/cls-studies/millennium-cohort-study/). The data are available for download at no cost from the UK Data Service (https://ukdataservice.ac.uk/). The MCS obtained ethical approval and informed consent for all data collection sweeps (Shepherd & Gilbert, 2019).
At the time of the second sweep of the MCS, just under 700 new families were recruited. We excluded these families from our sample as data on infections and many of the covariates used in analyses were collected in the first sweep, when the children were around 9 months of age, whereas the new families answered these questions when the children were around 3 years of age; a much longer retrospective recall period. Further, families with more than one child taking part in the MCS were excluded to ensure independence of observations regarding parent characteristics and home environment (Figure 1).
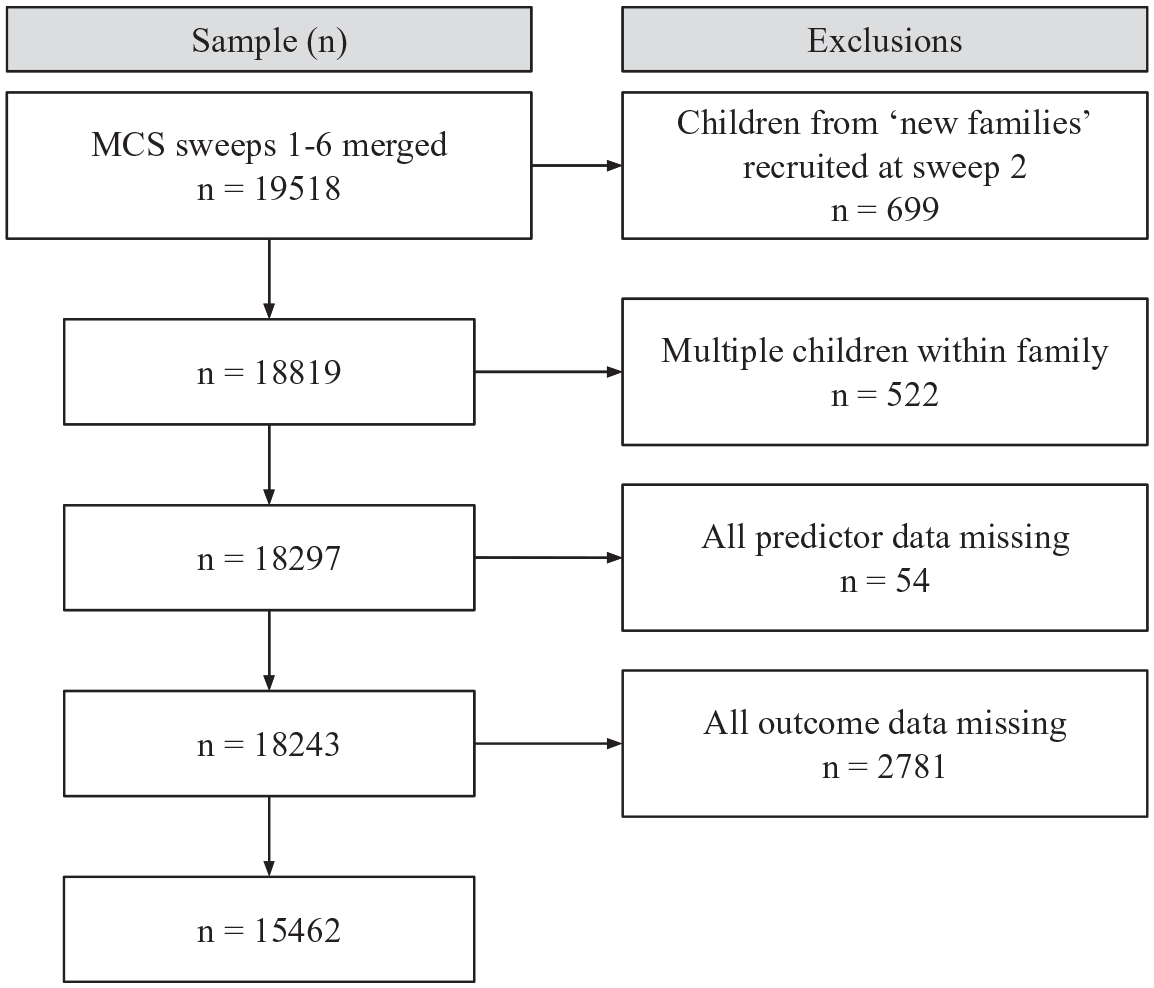
Sample exclusions.
Measures
Data on infections diagnosed in hospital during pregnancy were gathered from linked maternity inpatient records. The diagnostic information made available were three-digit codes using the International Classification of Diseases, 10th revision (ICD-10; World Health Organisation, 2011). For the purposes of this study, all recorded ICD-10 codes in the data were examined and those representing infections or inflammatory illness were filtered out to use as an indication of hospital-recorded infection. For a list of the ICD-10 codes included in the study, and their respective frequencies, see Supplemental Table S1.
Information on maternal-reported infections during pregnancy was gathered from interviews completed at the first sweep of the MCS, which took place around 9 months (range: 8.1–12.7 months) after birth of the cohort child. Mothers were asked: “Did you have any illnesses or other problems during your pregnancy that required medical attention or treatment?” If mothers responded positively to this question, they were asked: “What illnesses or problems did you have?” The data released had already been coded into categories, two of which represented infections; (1) Urinary infection and (2) Other/Non-trivial infections (National Centre for Social Research, 2003). For the purposes of this study, if either infection code was present, this was coded as “infection,” if another illness was reported this was coded as “other illnesses” and negative responses to the initial question of whether any illness had been experienced were coded as “no illness.” The “other illness” category included issues relating to foetal health, such as “too much fluid around the baby,” “suspected slow growth of the baby,” “foetal heart slow, faint, inaudible, foetal distress,” as well as maternal illnesses. The most frequently reported non-infectious illnesses were “bleeding or threatened miscarriage in early pregnancy,” “raised blood pressure, eclampsia/preeclampsia, or toxaemia,” “persistent vomiting,” “bleeding in later pregnancy,” and “anemia.”
Information on whether a child had ever had ASD and/or ADHD was gathered from parental interviews at the third, fourth, fifth, and sixth sweeps of the study, when the children were around the ages of 5, 7, 11, and 14, respectively. The main respondent, which in the majority of cases was the child’s mother, was asked at each sweep: “Has a doctor or other health professional ever told you that [Cohort member’s name] had any of the following problems? Autism, Asperger’s Syndrome or other Autistic Spectrum Disorder?” The same question was asked for ADHD; “Has a doctor or other health professional ever told you that [Cohort member’s name] had Attention Deficit and Hyperactivity Disorder (ADHD)?” For the purposes of this study, two variables were created, one representing each disorder. If the respondent responded positively to the question at any of the four time points, the child was coded as having the disorder. We chose to include all those who had at any point responded positively to the question in order to capture those who may have “grown out” of their condition according to clinical criteria but might still have had limiting symptoms. However, sensitivity analyses were carried out, where children were coded as not having ASD/ADHD if their parent at one point said that they had the condition but at a later sweep of the MCS said that they did not. Lastly, a variable for co-occurring ASD and ADHD was created from the ASD and ADHD variables, representing those who were reported to have both ASD and ADHD at some point between the ages of 5 and 14, regardless of at which sweep each condition was reported. In sensitivity analyses, only the children where both conditions were reported at the same data collection sweep were coded as having co-occurring ASD and ADHD.
A number of potential confounders and covariates were selected before conducting analyses. The selection was based on a literature search of previous studies on links between prenatal immune activation and neurodevelopmental outcomes, and then the individual associations. The variables selected were: child’s sex (Arnett et al., 2015; Ferri et al., 2018), maternal age (Mikkelsen et al., 2016; Wu et al., 2017), maternal education, area-based deprivation (Minh et al., 2017; Mortensen et al., 2011), maternal smoking during pregnancy (Langley et al., 2005), and maternal history of psychiatric illness (Daniels et al., 2008; López Seco et al., 2015). Child’s gestational age and birthweight were also considered as covariates, but not included in primary analyses as these factors may act as mediators in the relationship between prenatal infections and neurodevelopmental outcomes such as ASD and ADHD (Atladottir et al., 2010; Lampi et al., 2012; Sciberras et al., 2017). However, as views may differ on this, sensitivity analyses were carried out which additionally adjusted for gestational age and birthweight.
Data on all confounds and covariates were gathered from parental interviews at the first sweep of data collection (around 9 months after the birth of the cohort child). Child sex was provided as male/female and maternal age as a continuous variable of whole years. Maternal education was classified as National Vocational Qualification (NVQ) levels. For the current study, we categorized maternal education as NVQ level 3 (which equates to upper secondary education) or lower. Area-based deprivation was assessed using the Index of Multiple Deprivation (IMD; Noble et al., 2006). Maternal smoking during pregnancy was coded as yes/no, as was maternal history of psychiatric illness. In the case of the latter, mothers were asked whether they had any longstanding illness, responses to which were provided as ICD-10 codes. For the purposes of the current study, we filtered out codes which represented psychiatric disorders as indications of psychiatric illness. Further information can be found in questionnaire documentation (National Centre for Social Research, 2003).
Statistical Analyses
The data were analyzed using R (R Core Team, 2018) and RStudio (RStudio Team, 2017). Data were restructured and merged to allow for longitudinal analysis (script: https://github.com/Lydia-G-S/Millennium-Cohort-Study-Data-Restructuring-in-R). We used the survey package (Lumley, 2019) in order to adjust for clustering (electoral wards), stratification (socioeconomic status and ethnicity), and unequal selection probabilities and attrition bias, using weights. The survey package further allowed us to include a finite population correction factor, in order to adjust for sampling from a finite population without replacement. Adjustments were carried out in line with guidance published by the Centre for Longitudinal Studies (Ketende & Jones, 2011).
Logistic regressions with robust estimators were used to examine whether prenatal infections were associated with increased odds of ASD and/or ADHD. First, we fit unadjusted models of associations between prenatal maternal infections (separately for any infection, hospital-recorded infection, and maternal-reported infection) and ASD, ADHD and co-occurring ASD and ADHD. We then fit the same set of models while adjusting for potential confounds and covariates. Missing data were dealt with using attrition weights. Results are presented using Odds Ratios (OR) and corresponding 95% Confidence Intervals (CI).
Results
Results from raw descriptive analyses showed that 1,346 (8.7%) mothers had an infection during pregnancy, either maternal-reported or hospital recorded. Of those who responded to the question on self-reported illness (n = 15,459), 1,151 (7.4%) reported having an infection in pregnancy. Of those who had linked hospital admission data (n = 11,954), 253 (2.1%) were reported as having an infection. In the overall sample, 3.7% of the children were reported as having ASD at some point between the ages 5 and 14, 3.2% were reported to have ADHD, and 1.2% were reported to have both ASD and ADHD. Table 1 shows descriptive statistics of study covariates by infection status (any infection vs. no infection).
Descriptive Statistics for Study Variables.
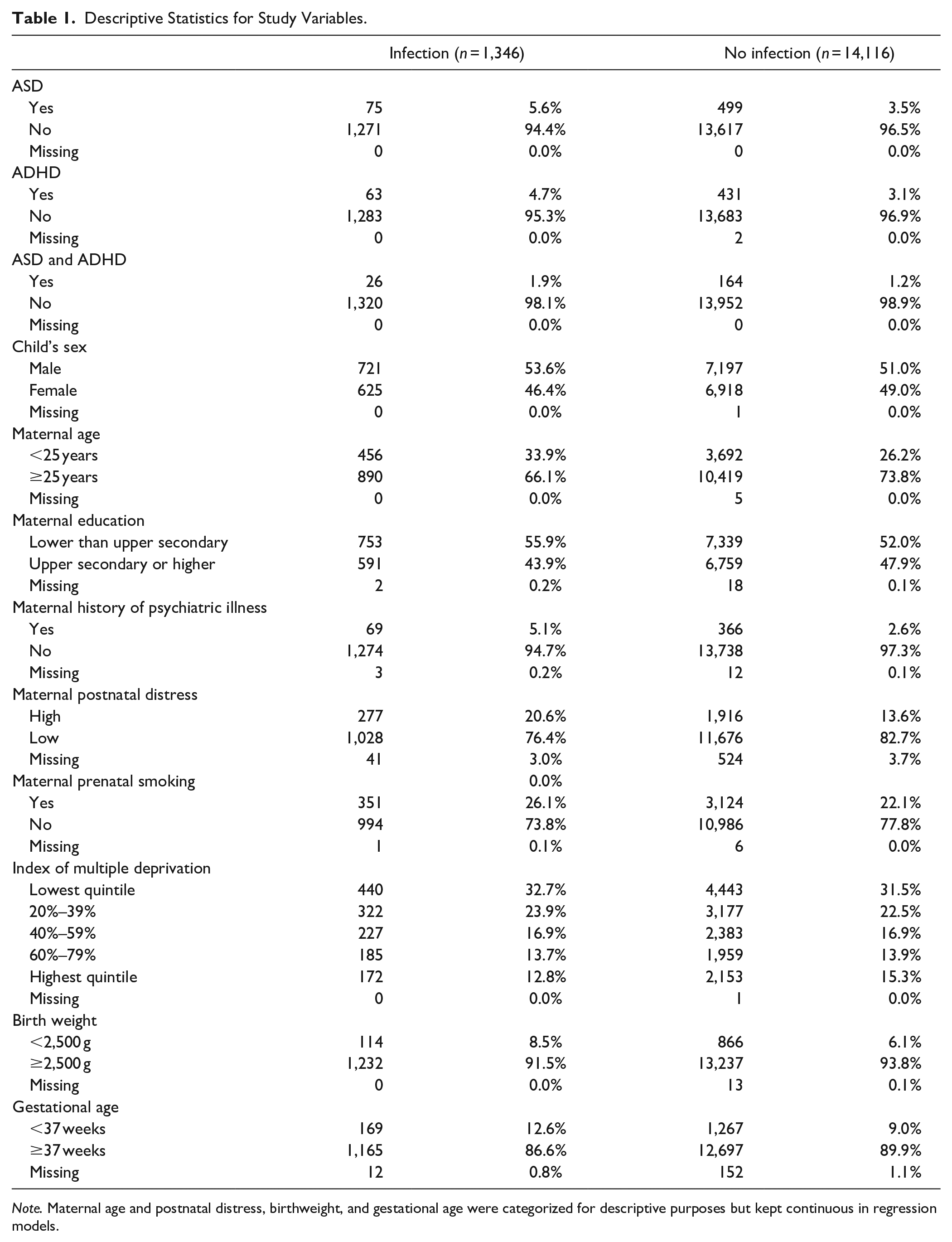
Note. Maternal age and postnatal distress, birthweight, and gestational age were categorized for descriptive purposes but kept continuous in regression models.
Any Infection
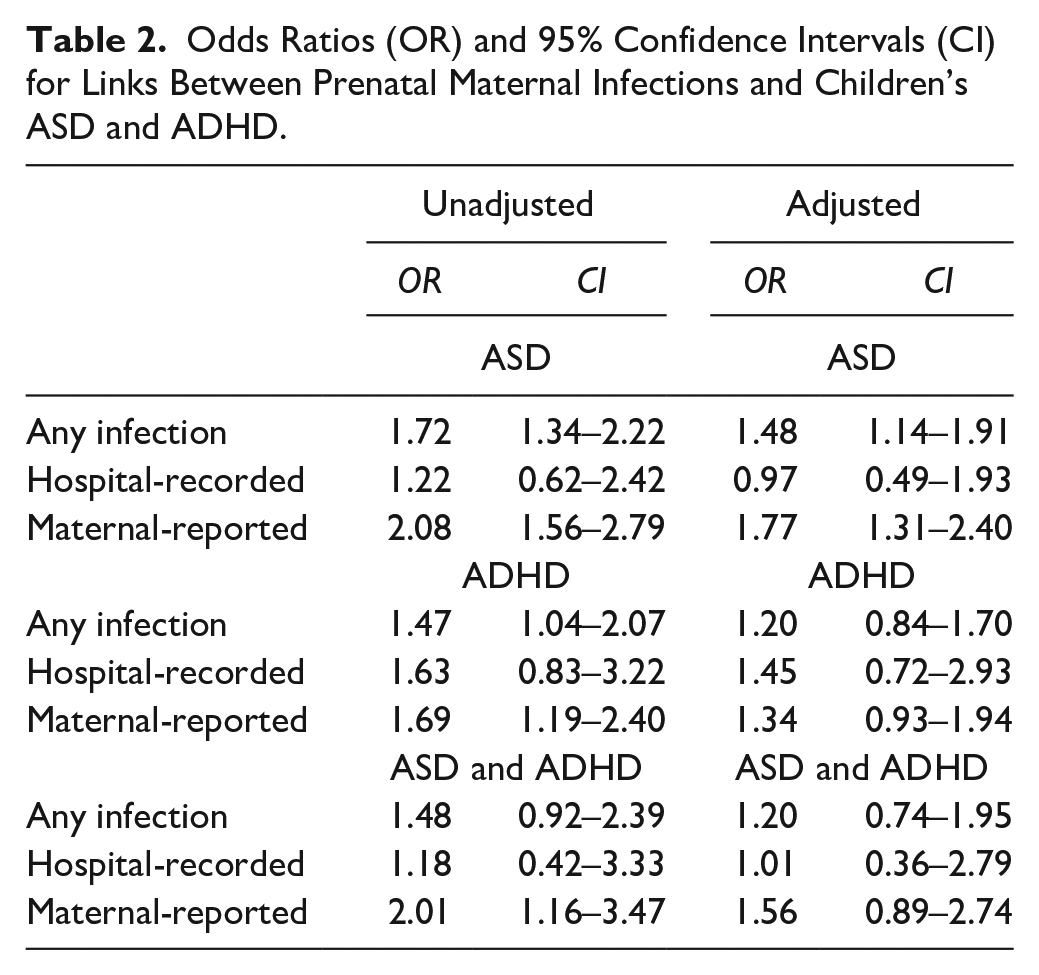
Results from unadjusted logistic models showed that the occurrence of any prenatal infection, either hospital recorded or maternal-reported, was associated with increased odds of both ASD, OR = 1.72 (CI = 1.34–2.22) and ADHD, OR = 1.47 (CI = 1.04–2.07), but not co-occurring ASD and ADHD, OR = 1.48 (CI = 0.92–2.39). When adjusting for potential confounds and covariates, only links with ASD were found, OR = 1.48 (CI = 1.14–1.91; Table 2).
Odds Ratios (OR) and 95% Confidence Intervals (CI) for Links Between Prenatal Maternal Infections and Children’s ASD and ADHD.
Hospital-Recorded Infection
Results from unadjusted logistic models showed that the occurrence of a hospital-recorded prenatal infection was not associated with the odds of ASD, OR = 1.22 (CI = 0.62–2.42), ADHD, OR = 1.63 (CI = 0.83–3.22), or co-occurring ASD and ADHD, OR = 1.18 (CI = 0.42–3.33). Adjusted models also showed no associations (Table 2).
Maternal-Reported Infection
Results from unadjusted logistic models showed that the occurrence of a maternal-reported prenatal infection was associated with increased odds of both ASD, OR = 2.08 (CI = 1.56–2.79) and ADHD, OR = 1.69 (CI = 1.19–2.40), as well as co-occurring ASD and ADHD, OR = 2.01 (CI = 1.16–3.47). When adjusting for potential confounds and covariates, results only showed links with ASD, OR = 1.77 (CI = 1.31–2.40; Table 2).
Sensitivity Analyses
When a more restrictive coding method for ASD and ADHD was used (see Measures), 3.4% were reported to have ASD, 2.6% ADHD, and 0.3% both ASD and ADHD. Using these variables, results from adjusted logistic regressions showed that maternal-reported prenatal infections were associated with increased odds of ASD, OR = 1.60 (CI = 1.15–2.22). In contrast to the findings of the primary analyses, adjusted models also showed links between maternal-reported infections and ADHD, OR = 1.51 (CI = 1.00–2.27) and co-occurring ASD and ADHD, OR = 3.35 (CI = 1.24–9.06). No links were found between hospital-recorded infections and any of the outcomes in neither unadjusted nor adjusted models (Table 3).
Sensitivity Analyses. Odds Ratios (OR) and 95% Confidence Intervals (CI) for Links between Prenatal Maternal Infections and Children’s ASD and ADHD.
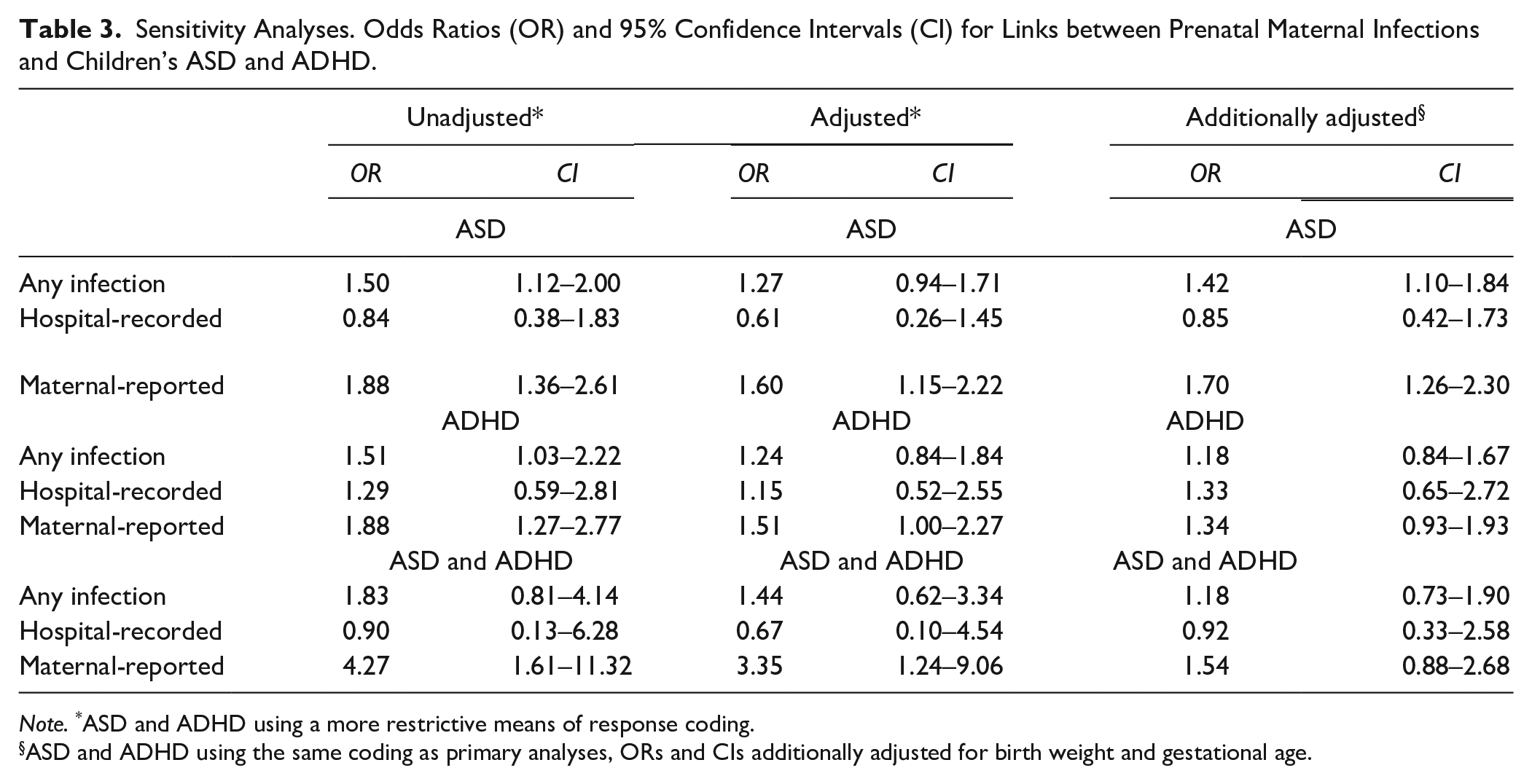
Note. *ASD and ADHD using a more restrictive means of response coding.
ASD and ADHD using the same coding as primary analyses, ORs and CIs additionally adjusted for birth weight and gestational age.
The latter sensitivity analysis, in which we additionally adjusted for birth weight and gestational age, showed the same pattern of results as the primary analyses. Adjusted models found increased odds of ASD to be associated with any infection, OR = 1.42 (CI = 1.10–1.84) and maternal-reported infections OR = 1.70 (CI = 1.26–2.30). No associations were found with hospital-recorded infections (Table 3).
Discussion
The aims of this study were to examine links between prenatal maternal infections, both hospital-recorded and maternal-reported, and children’s ASD, ADHD, as well as co-occurring ASD and ADHD. Results showed that maternal-reported infections were associated with increased odds of ASD, after adjusting for potential confounds and covariates. Primary analyses showed no associations between maternal-reported infections and ADHD or co-occurring ASD and ADHD after adjustment. However, sensitivity analyses, where more restrictive versions of the coding of ADHD and co-occurring ASD and ADHD were used, showed links between maternal-reported infections and increased odds of children’s ADHD as well as co-occurring ASD and ADHD. Hospital-recorded infections were not found to be associated with any of the study outcomes, neither in unadjusted nor adjusted models, indicating a clear lack of association. The results on links between any infection, regardless of whether the infection was self-reported or hospital-recorded, and children’s outcomes were largely in line with those where self-reported infections were used as the predictor. It thus seems that those findings were driven by the self-reported variable.
Our findings on links with ASD somewhat contradict evidence from previous studies. The meta-analysis of Jiang et al. (2016) reported that exposure to any prenatal infection was associated with increased odds of ASD (OR = 1.13), and a stronger association (OR = 1.30) when including only infections requiring hospitalization. This is the opposite to what the current study found. However, some original studies have reported links between particular types of hospital-recorded infections and ASD (e.g., Atladottir et al., 2010; Fang et al., 2015; Zerbo et al., 2015), and some only at specific trimesters (e.g., Atladottir et al., 2010; Fang et al., 2015). This suggests that the association between prenatal infection and ASD is not general but may be limited to particular infective agents and/or depend on timing. The literature on links between maternal-reported prenatal infections and ASD is more mixed. For example, Hornig et al. (2017) found links between maternal-reported fever and increased odds of ASD, though only in the second trimester, again, suggesting a timing-specific effect. Atladottir et al. (2012) found no association after adjusting for other factors, which included additional paternal factors that the current study did not include. Unfortunately, we were unable to examine links between specific types and timings of prenatal maternal infections and children’s neurodevelopmental outcomes in the current study due to methodological limitations. No information was provided on timing of exposure to infections in the MCS. Further, the self-reported data only distinguished between urinary tract infections and other infections and we determined that our study would have been underpowered to examine associations with different hospital-recorded infections separately.
ASD is a heterogeneous condition, with differences in severity of autistic symptoms, intellectual functioning, and comorbidities. Children with severe autism with intellectual disability have shown more signs of cortical abnormalities in neuroimaging studies (Lenroot & Yeung, 2013), in addition to being more likely than those with milder ASD to have epilepsy (Tuchman, 2017), compared to children on the less severe end of the spectrum. Rates of ASD diagnoses have risen in the past decades, with more children with mild symptoms, sometimes referred to as “high functioning,” receiving a diagnosis than before (Atladottir et al., 2015; Boyle et al., 2011). It could be argued that links between prenatal infections and children’s ASD are stronger for more severe cases of ASD, and that the milder cases included in the current definition may thus have attenuated these potential links. Further research on links between prenatal infections and children’s ASD, where severity of ASD, and/or the presence of intellectual disabilities is considered, may be warranted.
Primary analyses showed no association between prenatal infections, either hospital-recorded or self-reported, and children’s odds of ADHD. These findings contradict those of Mann and McDermott (2011) who found a significant association between prenatal infections and ADHD, although these were specific to genitourinary infections. Our results also contradict the findings of Ginsberg et al. (2019) who found significant links between hospital-recorded prenatal infections and ADHD, after adjusting for a number of covariates. However, this association was attenuated to non-significance in sibling comparisons, suggesting that the previously observed association may have been due to unmeasured familial factors. Our findings support those of Dreier et al. (2016) who found no association between any maternal-reported infection at any point during pregnancy and ADHD. However, they reported associations with fever in gestational weeks 9 to 12 as well as genitourinary infections in weeks 33 to 36, suggesting agent- and timing-specific effects, similar to the findings of some studies on links between prenatal infection and ASD.
Sensitivity analyses, where children whose parent at one point said that they had ADHD but at a later point said that they did not were coded as not having the condition, however, found increased odds of ADHD to be associated with maternal-reported infections. This suggests that the association between prenatal infections and ADHD may only hold for severe or persistent ADHD (Caye et al., 2016). However, the confidence interval for this result (CI = 1.00–2.27) suggests that these findings should be interpreted with caution.
A similar pattern of results emerged for co-occurring ASD and ADHD as for standalone ADHD. Primary analyses showed no association between prenatal maternal infections, hospital-recorded or maternal-reported, and co-occurring ASD and ADHD. However, in sensitivity analyses, where only those children who were reported to have both conditions at the same data collection sweep were coded as having co-occurring ASD and ADHD, links were found with maternal-reported infections. Thus, as suggested for ADHD, it could be argued that links between prenatal infections and children’s co-occurring ASD and ADHD may only exist for more severe cases. Ours is the first study, that we are aware of, to attempt to assess this association. However, a replication using definitive clinical diagnosis of co-occurring ASD and ADHD is warranted.
Studies on animal models suggest that it may rather be the body’s immune response to the infection rather than the infectious agent itself that is associated with neurodevelopmental issues in offspring, via mechanisms such as altered neurotransmitter levels (Bitanihirwe et al., 2010; Goeden et al., 2016; Romero et al., 2008), structural changes in the cortical and limbic brain regions (Samuelsson et al., 2006; Wischhof et al., 2015), and altered gene expression (Oskvig et al., 2012). Further, studies have suggested that immune system dysfunction in general may be associated with increased odds of both ASD (Siniscalco et al., 2018) and ADHD (Dunn et al., 2019). Links between prenatal maternal infections and children’s ASD have been considerably more researched than links with ADHD, and even less evidence exists on links with co-occurring ASD and ADHD. The results from primary analyses in the current study suggest that ASD may be etiologically dissociable from ADHD and co-occurring ASD and ADHD. However, sensitivity analyses suggest that these differences may be attenuated when the neurodevelopmental issues are more severe, though this remains speculative. The study of Oerlemans et al. (2016) attempted to tease apart unique and shared pre- and perinatal risk factors for ASD and ADHD. Interestingly, they found that most pre- and perinatal factors associated with ASD and ADHD were unique to each condition. Further, they found that prenatal maternal infections were uniquely associated with increased odds of ASD and not ADHD. In addition, they suggested that pre- and perinatal risk factors may have a more important role in the etiology of ASD compared to ADHD and that the high co-occurrence of ASD and ADHD is not likely to be explained by shared pre- and perinatal factors. Further research, especially on ADHD, is required to better understand the potential inflammation-related etiological differences, or similarities, between ASD and ADHD, both as standalone and co-occurring conditions.
Co-occurrence between ASD and ADHD in the sample was considerable, with around 30% of those reported having ASD also reported to have ADHD, at some point between the ages of 5 and 14, and 40% of those with ADHD also reported as having ASD. These rates somewhat contradict previous findings which suggest a higher proportion of children with ASD having ADHD symptoms compared to those with ADHD having ASD symptoms (Antshel et al., 2016). However, when using a more stringent definition of co-occurrence, where only children with reports of both conditions at the same data sweep were coded as having co-occurring ASD and ADHD, the rate of co-occurrence in the sample dropped from 1.2% to 0.3%. When we examined co-occurrence separately for each sweep prior to the DSM-5, which allowed ASD and ADHD to be diagnosed simultaneously, we found that a little over 20% of those with ASD at ages 5, 7, and 11 were also reported as having ADHD at the same sweep, compared to 18% the last sweep (age 14). It thus seems that simultaneous diagnoses were being carried out before the DSM-5 was published.
The rates of ASD reported in the study are higher, whereas rates of ADHD are lower, compared to best current estimates of global population rates (Lyall et al., 2017; Polanczyk et al., 2015; Thomas et al., 2015). Russell et al. (2014) have previously discussed the rates of ASD and ADHD in the MCS sample. They argued that among possible explanations for higher-than-expected rates of ASD could be increased awareness of the condition in the UK, compared to other countries, as well as less stigma around the ASD label. Rates of ADHD, on the other hand, may be lower than elsewhere because of more apprehension about that condition in the UK. They further argued that until diagnostic classifications allowed simultaneous diagnosis of ASD and ADHD, it seems that clinicians in the UK were more likely to diagnose children who showed hyperactive symptoms, in addition to social difficulties, with ASD rather than ADHD.
The different findings for associations with hospital-recorded and maternal-reported infections are noteworthy. We found that the agreement between hospital-recorded and maternal-reported infections was low (weighted Cohen’s kappa = 0.07). Of the 253 women in the study sample with a hospital-recorded infection, only 58 (23%) self-reported an infection. This result is consistent with a previous study (Atladottir et al., 2012) which showed low agreement where the self-report measure was an open-ended question. They found that agreement between maternal and hospital reports was fairly good (65%–77%) when women were specifically asked about exposure to particular infections (e.g., “Have you had cystitis?”). However, the agreement was low (6%–7%) on information gathered from an open-ended question (“Have you had other infections?”). The low agreement rates in the current study may, therefore, be due to the fact that individual infections were not itemized. In addition, the data on hospital-recorded infections included all infections that were diagnosed during a hospital admission, so a distinction between primary diagnoses and others was not made. In fact, only 33 of the 253 indications of hospital-recorded infections were from primary diagnoses. Mothers may thus not have remembered, or even been aware of, the infection. Rates of maternal-reported infections were considerably higher than hospital-recorded infections (7.5% vs. 2.1%, respectively), suggesting that the self-report measure may have captured a broader range of infections, including milder infections that were not associated with a hospital admission. However, previous studies have found rates of maternal-reported prenatal infections to be up to 60% (Collier et al., 2009), which is considerably higher than in the current study. This may, again, be attributed to the open-ended means by which women were asked about prenatal illness in the interviews, as well as the length of the recall period. Unfortunately, the MCS did not contain outpatient records, which would have allowed for a more objective assessment of milder infections than those recorded during an admission to hospital. Low agreement between hospital-recorded and self-reported infections may also help explain some of the discrepancies in the literature on links between prenatal infections and neurodevelopmental outcomes, as it seems that the method of assessing exposure to infections may affect the type or severity of infections that are identified.
It is worth noting that over 20% of mothers who provided self-reported information on infections did not have linked hospital admission data. This may have introduced bias to the findings. Consent to data linkage in the MCS has been previously studied (Tate et al., 2005) and the results suggested that, for example, mothers with higher degrees (above bachelor level) or no educational qualifications, those from minority ethnic groups and lone parents were less likely than others to agree to linkage. However, non-response weights were embedded in the complex survey design used in the current study, which should alleviate effects of non-response bias.
The study had several other limitations which must be considered. Even though the overall MCS sample is large, with over 18,000 families taking part in the initial sweep, the numbers for key measures in the current study were fairly low. Of those children whose mothers had a hospital-recoded prenatal infection, only 11 had ASD and 11 had ADHD. Of those whose mothers self-reported a prenatal infection, 66 had ASD and 56 had ADHD. Thus, our models would not be powered to detect very small associations.
As outlined above, we did not attempt to replicate subgroup findings focusing on particular types of infections or specific trimesters, due to the size of the sample and nature of the data. As well as considering the timing of exposure, future studies should examine the potential links between different types of infections on children’s outcomes. Evidence suggests that potential effects of prenatal infections on children’s neurodevelopmental outcomes may differ by type of infection, for example, bacterial versus viral infections (Jiang et al., 2016). This might be partly explained by differences in the immune response which different infectious agents elicit and whether or not infections cross the placental barrier. The COVID-19 pandemic has made better understanding these differences all the more important, with significant implications for issue such as vaccine prioritization and possible shielding for expectant mothers and the adaptation of antenatal care during viral pandemic conditions. The vast data collection efforts sparked by the COVID-19 pandemic provide an opportunity to advance understanding in this area.
Another factor to consider is the retrospective nature of the items on maternal-reported infection. The interviews where these data were gathered took place around 9 months after birth. Some mothers may thus not have recalled whether they had had an infection or not. The MCS further relied on parent-reports of ASD and ADHD status. As a consequence, it is unknown whether all the identified cases were a result of a clinical diagnostic process. It could be argued that those who have experienced an infection during pregnancy may be more alert toward their children’s symptoms of neurodevelopmental issues and therefore be more likely to seek medical advice for such symptoms and their child potentially receiving a diagnosis. However, it remains unclear how aware the public is of the potential links between prenatal maternal infections, especially those associated with mild illness, and children’s neurodevelopmental conditions. This is an interesting topic for future research.
Finally, we did not have access to biomarkers of infection that could help us determine whether our findings were also reflected in the assumed mechanisms by which prenatal infection affects neurodevelopment, as mentioned previously. However, studies with human participants examining relationships between various immune markers, such as C-reactive protein and inflammatory cytokines have not yielded definitive findings (for a review, see Zhang et al., 2018).
A strength of the current study lies in the sampling and recruitment efforts of the MCS. The MCS sample is unique with regards to demographic factors and groups of people which researchers in the UK often find difficult to reach were oversampled. Having a population-representative sample, such as this one, is important within this area of research. For example, socioeconomic status has been shown to influence maternal inflammatory processes. Gilman et al. (2017) found that socioeconomic disadvantage was linked to lower levels of the pro-inflammatory cytokine interleukin-8 during late pregnancy which, in turn, was linked to neural abnormalities in infants. Further, the study of Russell et al. (2016), using data from the MCS, found that ADHD in children was associated with socioeconomic disadvantage including poverty, living in social housing, low maternal education, and low household income. This shows how important it is for study samples to have adequate heterogeneity in order to produce externally valid findings.
Conclusion
The current study presents evidence for links between maternal-reported prenatal infections and ASD and ADHD, although associations with the latter were only found where the definition of ADHD status was restricted to those children who may have had more severe and/or persistent symptoms. Similarly, links with co-occurring ASD and ADHD were only found when the definition of co-occurrence was restricted to those children who were reported as having both conditions at the same data collection sweep. No associations were found between hospital-recorded infections and children’s ASD, ADHD, or co-occurring ASD and ADHD. This discrepancy between findings relating to hospital-recorded and maternal-reported infections may be due to the low observed agreement between these measures, suggesting that they may have identified distinct types of infections. Our findings contribute to an extensive literature on hospital-recorded infections and ASD, add to the scarcer body of evidence on maternal-reported infections and ASD and supply new evidence to a small but growing literature on links between prenatal infections and ADHD. Our study is the first that we know of to examine links between prenatal infections and co-occurring ASD and ADHD. Future studies should focus on replicating analyses using larger data sets, which allow for sub-group analyses focusing on different types of infection as well as timing of exposure. Further, studies should utilize sibling samples to account for the heritability of ASD and ADHD, and to help control for within-family confounding factors.
Supplemental Material
sj-docx-1-jad-10.1177_10870547211015422 – Supplemental material for Prenatal Maternal Infections and Children’s Neurodevelopment in the UK Millennium Cohort Study: A Focus on ASD and ADHD
Supplemental material, sj-docx-1-jad-10.1177_10870547211015422 for Prenatal Maternal Infections and Children’s Neurodevelopment in the UK Millennium Cohort Study: A Focus on ASD and ADHD by Hildigunnur Anna Hall, Lydia Gabriela Speyer, Aja Louise Murray and Bonnie Auyeung in Journal of Attention Disorders
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors have been supported by the following bodies throughout the course of this project: UK Economic and Social Research Council via the Scottish Graduate School of Social Sciences (ES/R500938/1) (HAH); University of Edinburgh Principal’s Career Development Scholarship (LGS); European Union’s Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie grant agreement No.813546, and the UK Economic and Social Research Council (ES/N018877/1) (BA). ALM has no funding to declare for this project.
Supplemental Material
Supplemental material for this article is available online.
Author Biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
