Abstract
In recent years, telemonitoring solutions have gained prominence for monitoring patients at home. However, patient and clinician enthusiasm, as well as adherence rates, have been inconsistent. This article presents a primary care evaluation of an autonomous foot-measuring home device, designed to monitor physiological changes in medium to high-risk chronic heart failure patients at risk of decompensation. The Heartfelt device garnered positive reception among the 13 participating patients. Remarkably, in 77% of cases (10 patients), the device captured and transmitted patient data on more than half of the days each month, while only 8% (one patient) managed to provide equivalent daily measurements using weighing scales. These results indicate that the Heartfelt device holds promise as an effective telemonitoring solution for patients who face adherence challenges with traditional telemonitoring systems, and self-management checks more generally. By adopting this technology, healthcare providers may be better equipped to proactively manage chronic heart failure patients and improve overall patient outcomes.
Introduction
Heart failure is a complex syndrome characterized by the heart’s inability to pump blood at a rate sufficient to meet the body’s metabolic demands. This growing global health issue, with high mortality and morbidity rates, impacts over 60 million people worldwide, particularly among the elderly population. 1 Approximately 50% of patients with heart failure may succumb to the condition within 5 years. 2
In the United States (US) and Europe, patients diagnosed with heart failure constitute 1% to 3% of the adult population, making these regions the most prevalent for the condition. 3 A US-based study estimated that the number of individuals living with heart failure could surge from 6.5 million in 2012 to 8 million by 2030, with an accumulative healthcare expenditure of $70 billion.4,5 In the United Kingdom (UK) alone, over 900,000 patients are affected by this disease, 6 which is a concerning statistic. Concurrently, heart failure medical costs account for 1% to 2% of Europe and the USA’s annual healthcare budgets. 7 As a result, the global financial burden of heart failure amounts to approximately $108 billion per year, primarily due to hospitalizations. 8 Furthermore, a systematic review suggests that outpatient outcomes are generally better than those for hospitalized patients, 9 indicating that early identification of heart failure decompensation can be beneficial to the patient. Consequently, preventing emergency hospital admissions is a pressing challenge that must be prioritized in heart failure management.
The need for increased utilization of telemonitoring (TM) in routine heart failure management has been highlighted by the growing pressure on healthcare systems and the aging population. 10 Remote patient monitoring platforms offer a convenient and accessible way to provide continuous care, including diagnosis and treatment, 11 while also reducing the burden on healthcare systems and professionals. Studies conducted earlier have established the effectiveness of non-invasive TM in reducing mortality risks and unplanned hospitalizations, as well as enhancing patients’ quality of life.12,13 Furthermore, the demand for healthcare services is expected to increase as the population ages. 10 There has been a shift toward virtual healthcare, with the accelerated adoption of TM devices during the COVID-19 pandemic. 14 Many countries, particularly in Europe, have incorporated these platforms into their clinical practice for heart failure management, resulting in encouraging outcomes.13 -16
Currently, a variety of TM systems are either in use or under development. 11 Some of the most prominent and promising devices include CardioMEMS Heart Failure System, HeartPOD (Abbott), HeartLogic (Boston Scientific), V-LAP (Vectorious Medical Technologies), Medtronic OptiVol Fluid Status Monitoring and CareLink Network (OptiLink Heart Failure), and Chronicle IHM (Medtronic).14 -20 However, many of these TM technologies are invasive and require patient adherence, typically for the transfer of patient-initiated sensor data to an internet-connected device. 17 Previous studies have reported high adherence rates, but it should be noted that these studies typically exclude patients who are non-adherent with TM or disinclined to participate due to onerous trial protocols. Berry and colleagues suggest that future innovations should consider this excluded patient group and address the issue of “low compliance.” 21
In this evaluation, we assess an innovative TM device (Heartfelt HF-3, Heartfelt Technologies Ltd, UK), which may be promising for patients who are typically disengaged with technology. The device does not require patient adherence for data collection and is non-invasive, making it suitable for frail patients or those ineligible for implantable devices. The Heartfelt device employs 3D imaging technology and AI to precisely measure foot volumes from captured images, compared to the gold standard of water displacement. 22 Subsequently, it automatically identifies patients at increased risk of readmission (as indicated by a rise in peripheral edema 23 ) for the clinical team to evaluate and determine the appropriate action(s). Approximately half of the affected patients regularly experience peripheral edema as an early warning sign of worsening or decompensation of heart failure.24 -27 This symptom is more prevalent in patients with right-sided heart failure, who are more likely to be hospitalized and have limited treatment options.28,29
The purpose of this evaluation is to investigate the experiences of patients and primary care physicians in Hull (UK) with the use of Heartfelt devices outside of a clinical trial, with a focus on data availability and acceptance. The aim is to contribute to the existing body of knowledge on TM solutions for patients with heart failure and to identify potential areas for improvement in the implementation of such technology, with a particular focus on patients who are at high risk of hospital re-admissions due to adherence issues.
Materials and Methods
This evaluation was carried out as part of a service evaluation, with devices funded through post-market monitoring funds provided by the manufacturer. Participants were recruited from a general practitioner (GP) practice in Hull, UK, between June 2020 and July 2021. In accordance with the World Medical Association Declaration of Helsinki, national ethical approval was not required for this evaluation. The plans for this service evaluation were reviewed and approved by senior team members of the practice, including GPs and the practice manager. The clinical team members (including authors) declare no conflicts of interest in relation to the evaluation.
Patient Recruitment
The clinical team identified patients based on the following criteria:
Patients with heart failure considered to be at high risk of decompensation and hospital readmission by their GP (this is significant due to the high costs associated with avoidable readmissions)
Patients who have previously presented with peripheral edema associated with heart failure decompensation (this is important since the device evaluated measures changes in foot volumes)
Patients without double amputations, continuous extensive bandages on both feet, and those not bed-bound (these are contraindications for the device under evaluation).
A total of 14 patients were approached to participate in the service evaluation, which was anticipated to last 6 months. Patients were provided with information about the evaluation, and out of the 14 patients approached, 13 participants enrolled after completing the consent process.
Procedure
Patients who agreed to participate in the evaluation were visited by an installer who set up 2 devices in their homes: the Heartfelt device (Heartfelt Technologies Ltd.); and Bluetooth-connected weighing scales (Xiaomi Smart Scales, Anhui Huami Information Technologies). The Heartfelt device is connected to household AC power, while the Xiaomi scales are battery-operated (new batteries were supplied at the start of the evaluation). The Heartfelt device does not depend on the weighing scales for operation, or vice versa, however, the Heartfelt device was configured to operate as the Bluetooth-to-internet transmission channel for the weighing scales.
Weight data were automatically transmitted to the Heartfelt device via Bluetooth when patients stepped on the scales. Both sets of data, with associated collection timestamps, were then transferred through the internet, with 4G dongles provided if Wi-Fi was unavailable or insufficient. Since daily weight monitoring is the standard recommendation in the UK for home monitoring of heart failure, 30 the weighing data was used for comparison of usage and data collection against the Heartfelt device.
Patients received standard heart failure monitoring education and lifestyle advice from the GP or practice nurse, in accordance with the British Heart Foundation recommendations. 31 However, patients were advised not to rely solely on the scales or Heartfelt data transmission, and to promptly report any health issues (including significant weight gain) to their primary care doctor without delay. Significant increases in weight and foot volume measurements led to a call to the patient or carer, in line with standard practice in TM. At the end of the six-month period, participants were given the option to continue using the scales and device and extend the evaluation period.
Heartfelt Device
The Heartfelt device (Heartfelt Technologies Ltd.) is installed in the patient’s home in an area where the patient frequently has bare feet, often the bedroom or living room. The device operates by automatically capturing infra-red images of the exposed lower legs when the patient enters its field of view during their daily/nightly routine, without requiring them to specifically stand in front of the device. Relevant foot volume data is then measured and transmitted to a centralized monitoring service. This entire process occurs automatically, eliminating the need for direct patient or clinician involvement.
The data collection and processing for the Heartfelt device are fully automated, necessitating no action from patients other than ensuring the device is powered on and continuing with their usual routines. 32 As only 3D images of the barefoot and lower leg are transferred to the cloud for further processing and interpretation, patients experienced minimal privacy concerns.
Data Collection and Outcomes
Patients provided consent for the use of their age, sex, date of heart failure diagnosis, comorbidities, medication changes during the evaluation, date and length of hospital admission in the 24 months prior to enrollment and during the evaluation, relevant information from GP, nurse, and hospital appointments during the evaluation, and their postcode to report on deprivation levels of their residential area in this report. The authors obtained the Index of Multiple Deprivation, Health & Disability Rank, and the IDAOPI score from the patient’s postcode using the Ministry of Housing, Community, and Local Government web tool. 33
The first outcome of this service evaluation was patient acceptance of the Heartfelt device, determined by the number of patients using the equipment during the service evaluation period and feedback provided through questionnaires.
The second outcome was the number of days with available Heartfelt and/or weight data, representing the reliability of data collection for this group of patients. This figure was calculated and expressed as a percentage of days with data, ranging from 0% to 100%. A valid day was defined as follows:
As part of the setup, patients were encouraged to step on the scale to establish connectivity. Therefore, weight readings on the first day were excluded as they were not spontaneously taken of the patient’s own accord. Weight data was manually reviewed to exclude measurements from other household members (despite requesting that only the patient steps on the scales, several other people used the scales).
Heartfelt data were considered present for the day if the quality and quantity of the data collected on the patient that day were sufficient to generate at least one volume reading. At the initial setup, the patient is typically asked to stand near the device to help it recognize the patient’s feet. That data is not collected spontaneously as part of routine activities and is therefore discarded for the purpose of this data acquisition analysis. The data was reviewed to ensure that the Heartfelt device did not erroneously include non-patient volume readings.
Furthermore, the threshold value for effective TM frequency is a minimum of 16 days per month, as recommended by Medicare in the US. 34 We also assessed how many patients met this threshold to examine the effectiveness of both methods.
The evaluation noted the frequency of measurements from December to February (inclusive) and compared it to the period June to August (inclusive) to evaluate the device usability in winter seasons when patients typically wear socks or slippers, which could pose an issue since the Heartfelt device excludes data when footwear is worn. This evaluation was performed for each 12 complete months of data, with 7 patients providing a total of 9 such periods. To ensure that weight data was not underreported, patients were asked if they used different scales during the evaluation, and none reported doing so.
During the evaluation, patients were asked to provide their opinions on the following questions:
Are you comfortable with the device installed in your bedroom by your bed? (Options for answers: Yes, No, if no, details could be added).
Do you think that the data collected by the Heartfelt device is more or less reliable than weighing? (Options for answers: Yes, Unsure, No, details could be added).
Do you have any concerns about the device? (Options for answers: Yes, Maybe, No, details could be added).
Starting from June 2022, we also contacted patients with a Likert scale question. This question was presented to patients who were still using the device and those who were no longer taking part in the evaluation:
How likely would you be to recommend this device to a friend if they had heart failure? (5-point Likert scale)
Data Analysis
Quantitative data obtained from surveys were analyzed using Microsoft Excel (sections 3.1, 3.2). Responses to open-ended questions were reviewed by both authors to categorize them into predefined classifications, and “other” answers are presented as quotes in the article. The results of the 5-point Likert scale were not pooled and presented in full (details are shown in Table 2).
Python and the NumPy package were employed to analyze the mean and median number of days per week or month per patient for recorded volume and weight measurements (section 3.3). In calculating the average days-per-week of data collection for winter and summer (Table 1), only periods of 12 consecutive complete calendar months were included for each patient. Some patients contributed two such 12-month periods to the analysis.
The Percentage of Days With Patient Data.
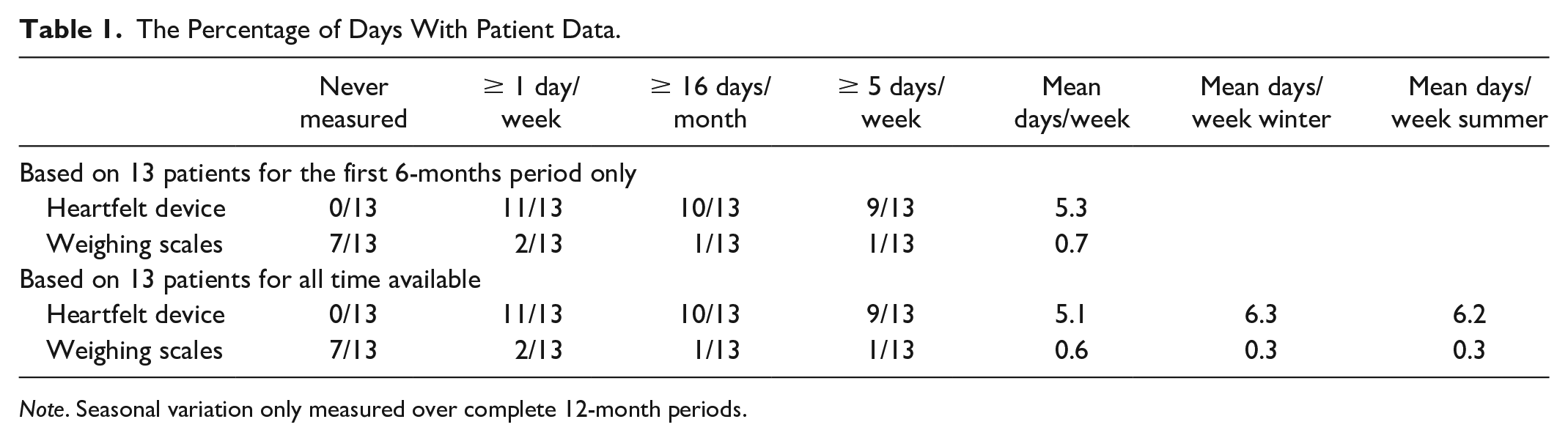
Note. Seasonal variation only measured over complete 12-month periods.
Results
Patients’ Characteristics
Between June 2020 and July 2021, we enrolled 13 patients diagnosed with heart failure and a history of peripheral edema (median age 74.4 years [71.9, 82.9], 2 patients (15%) were women, 9 patients (69%) also had diabetes, and 6 patients (46%) had chronic kidney disease; one patient was blind, and one had lymphedema). These patients were approached as they were considered at high risk of hospital admission. There were 2 hospital admissions (2 different patients) during the evaluation period, both of which were heart failure related. All participants lived in Kingston upon Hull, the fourth most deprived area in the UK, 33 and details of individual Health and Disability Deciles they live in are presented in Figure 1.
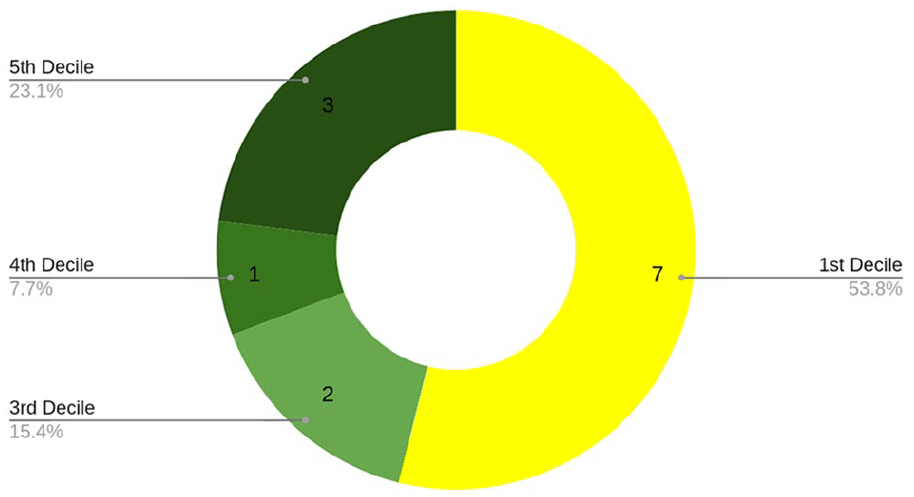
Number of participants in each Health and Disability Decile
At the installation time, 9 patients (69%) had Wi-Fi, the remainder being provided with 4G broadband at installation. Overall, 100% of patients’ homes had sufficient connectivity for data to be transmitted reliably from the devices.
Patient Acceptance
Out of 14 patients initially contacted, 13 (93%) signed written informed consent and agreed to take part in the evaluation. Notably, the reason given for not taking part was feeling uncomfortable with people coming to the home during the COVID-19 pandemic to install the device.
Although this evaluation was initially for 6 months, several participants requested to keep the devices and extend the evaluation. The results below are therefore presented in 2 sets: data collected during the first 6 months period, or data collected over all the time available. Three patients dropped out before the end of the 6-month period, as seen in Figure 2. Over the observation period of 15.3 months on average [6.8 months, 24.7 months], at the time of writing, 10 patients had the device for more than 6 months, and 7 were still using it in September 2022.
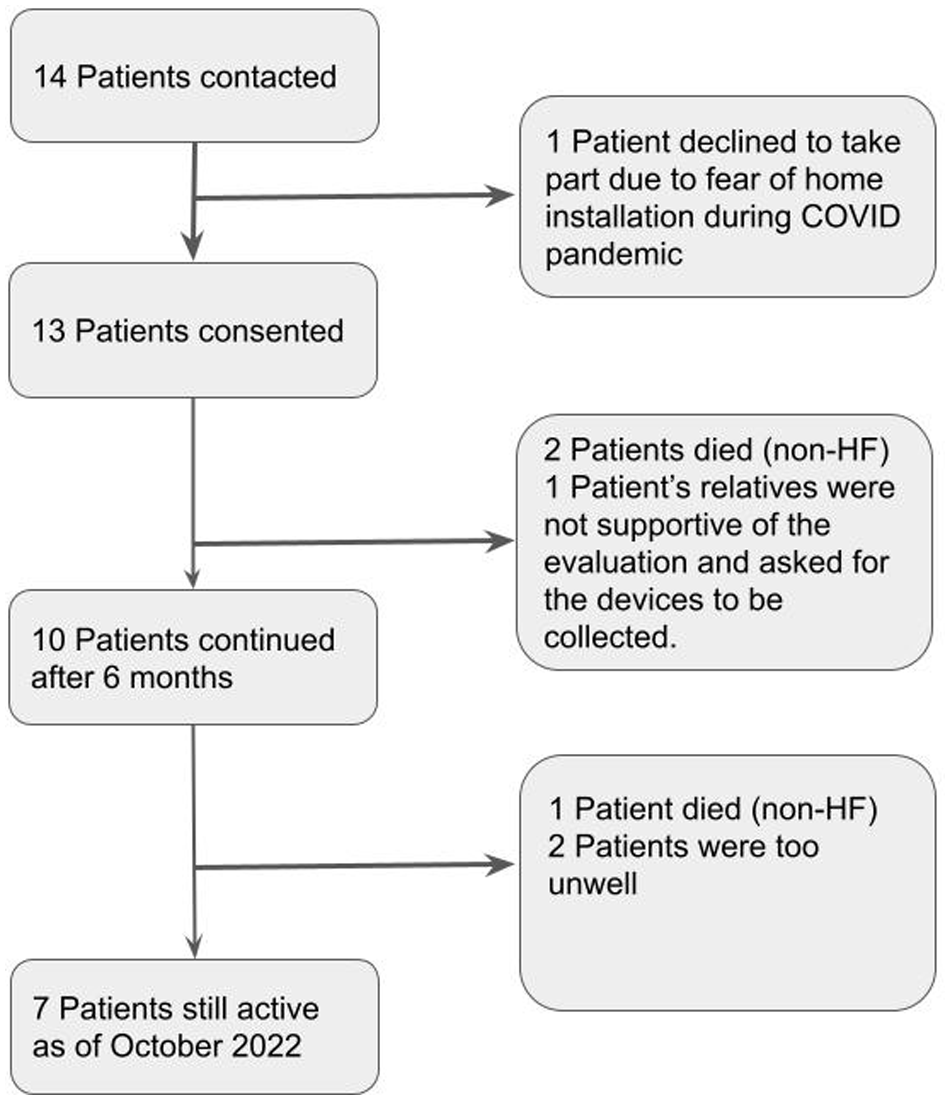
Consort diagram.
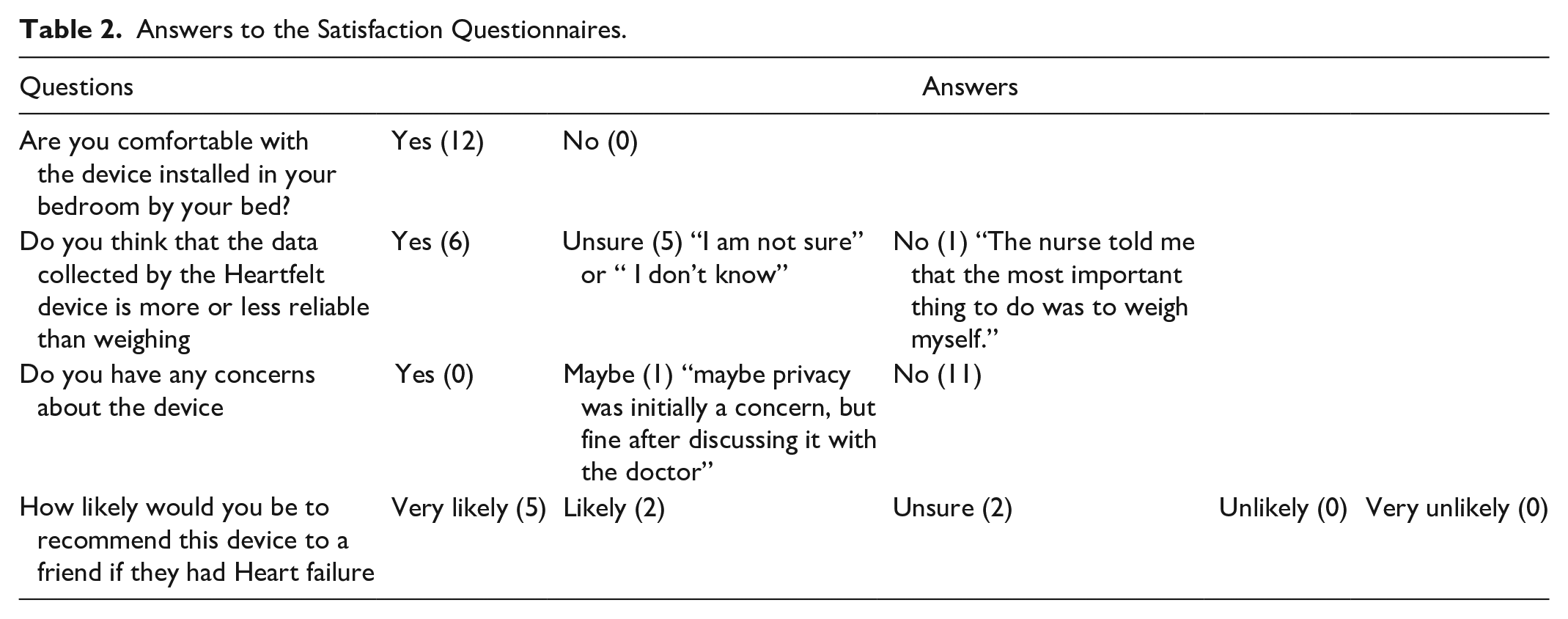
Of the 13 evaluation participants, 12 provided some answers to satisfaction evaluation questionnaires, and 9 did this more than once during the evaluation period; these are presented in Table 2. Twelve participants reported being comfortable with the idea of the Heartfelt device being installed in their room by their bed. Half of the participants felt that the Heartfelt device would be more reliable than weighing scales, 5 patients were unsure, and one thought that the scales would be more reliable. Eleven participants reported having no concerns about the device, and one reported that “maybe privacy was initially a concern, but fine after discussing it with the doctor.” Five participants stated that they were very likely to recommend the Heartfelt device to a friend who had heart failure, 2 were likely, and 2 were unsure. For those who completed questionnaires more than once during the study period, the ratings did not change.
Answers to the Satisfaction Questionnaires.
Reliability of Data Collection
Although regular weighing is a standard recommendation given to heart failure patients, 30 this evaluation recorded that only 8 patients (61%) stepped on the scales during the 6-month evaluation period (excluding the first day, under direction of the person installing the scales). Meanwhile, all 13 participants had data from the Heartfelt device, and this continued to be the case after the 6-month period for those asking to extend the evaluation.
As shown in Table 1, in the majority of cases (10 patients, 77%), the Heartfelt device was able to capture and transmit patient data on more than half of the days. In comparison, the equivalent amount of data for weighing scales was only present for one patient (8%).
One concern that we had when starting the evaluation was the effect of indoor footwear, which may be more common in winter and may interfere with the Heartfelt device’s readings. As seen in Table 1, for those patients who have used the device for more than 12 months (allowing for the analysis of the difference between winter and summer months), we observed no difference in scale usage or Heartfelt data acquisition rate between cooler and warmer months.
Another concern is the rate of decline in device usage. Over the 6-month period, we have not reported a reduction in the rate of data acquisition for the Heartfelt device, nor did we notice this over the longer time period, as seen in Table 1.
Discussion
These results represent the first real-world application of this TM device used by patients and their primary care clinicians. The novel TM device evaluated was acceptable to patients, it allowed an improved level of remote collection of physiological data, and satisfaction ratings were good. The results show sufficient physiological data being collected by the device to allow the safe monitoring of patients who did not show a high rate of adherence with established self-management recommendations such as daily weight checks.
This evaluation was conducted in Kingston upon Hull, a highly deprived area of the UK, where patients often have multiple comorbidities and challenges with the social determinants of care. This patient group represents one of the most challenging patient groups to include in a TM study, and overwhelming numbers of study appointments and activities have been identified as barriers to the inclusion of underserved groups by the NIHR-INCLUDE project. 35 For these reasons, we have chosen to keep our evaluation very light from the participant’s perspective. Higher incidences of HF are experienced in patients from disadvantaged socio-economic backgrounds, 36 and these patients experience higher rates of mortality from heart failure than other groups. 36 The authors wish to highlight the feasibility of engaging these patients in evaluations, as this experience could be replicated in clinical trials with a light protocol.
Of the 14 introduced patients, 13 patients consented to this evaluation, resulting in a high enrollment rate of 93%. It suggested that the device is suitable for most patients, including those not meeting the criteria to use other heart failure devices. In addition, the response of patients about the device in terms of privacy is positive. Privacy and security have been reported in the literature as some of the barriers to the adoption of digital healthcare tools 37 ; this may have been part of the concerns that relatives of one participant had (the participant dropped out of the evaluation early).
The data acquisition rate for these patients was high, particularly considering the fact that these patients were not selected based on their likely adherence level. The passive nature of this device may help explain this sustained high rate of data acquisition, as previously reported. 32
Given the heart failure sub-population that is most regularly readmitted to the hospital, it was important to ensure that this evaluation enrolled representative patients. Indeed, many heart failure studies have strict criteria, with some excluding patients living with dementia or general adherence issues. And yet, these are the patients disproportionately represented in heart failure hospital readmissions. 38 In addition, poor adherence to medications of cognitively impaired patients could indicate that they are less likely to comply with the instructions of TM systems 21 ; therefore, those could be perceived as less effective for this patient group. Another challenge for TM technologies is the decrease in compliance linked to the increasing number of required actions from patients. 21 Based on these findings, patients, especially older ones, who have poor adherence to medications would also struggle to use a proactive monitoring regime over a long time. With the Heartfelt device, patients do not need to change their routines because the device passively records data when it captures images of the patient’s lower legs. Hence, the interaction-free feature may be the reason for the high rate of data acquisition of the device reported in this evaluation.
We note anecdotally that those patients who chose to extend the evaluation past the initial 6 months were the more stable patients. Those who are no longer using the device have either passed away (3), have decided to stop as too unwell (1), have relatives who are not supportive of the patient taking part in the evaluation (1), or because they were planning building work on their house (1). It was also interesting to note that the patient who felt that weight was more reliable than the Heartfelt measurements was a patient who did not use the scales at all during the evaluation.
From the clinician experience, the authors suggest that getting data on half of the days would be a significant improvement for patients who were, until now, not engaging with self-management recommendations and rarely provided proactive reports of weight changes or symptom changes. Additionally, the threshold of 16 days for each month set by Medicare in the US to provide reimbursement for medical physicians reviewing data coming from the devices in the remote patient monitoring program focusing on elderly patients seems to be universally accepted amongst professionals. 34 Although the weighing scale is the standard care for heart failure monitoring, the number of patients who achieved this 16-day minimum requirement using the heartfelt device was substantially higher than those using scales (10/13 patients vs 1/13 patients). This is very promising for the long-term monitoring of non-adherent patients with heart failure.
Interestingly, the clinical team also had concerns over the ability of the device to capture data of the lower leg in winter when patients tend to wear thick socks, slippers, etc. Our analysis revealed that even in the coldest months in this part of the UK (November to April), the frequency of measurements of the heart failure device is still superior to those of the weighing scale. This finding also indicates that the usability of the device is not negatively affected by environmental factors.
To apply this technology in the primary care setting, we would need to ensure that the overall cost of the device and the time taken by clinicians to review data is cost-effective. This was not the purpose of this evaluation, as the device is not yet widely available in the NHS. However, the promising results of this study suggest that the Heartfelt device may be a valuable addition to current heart failure monitoring methods in the future, particularly for patients who struggle with adherence to self-management recommendations. Further research, including cost-effectiveness analyzes, larger trials, and the examination of clinical outcomes, would be necessary to fully understand the potential impact of this device on patient care and healthcare systems.
Limitations of the Evaluation
Our evaluation and findings come with several limitations. Firstly, this study was a service evaluation rather than a randomized clinical trial. While this offers a real-life experience of a UK-based primary care practice utilizing the technology without the support of research administrators, it also limits the generalizability of the results. Moreover, the recruitment of participants occurred during the COVID-19 lockdown, which may have contributed to the high uptake, as patients could perceive it as a means of being monitored during a period when face-to-face appointments were challenging to secure within the NHS.
We only had one patient with a high level of static edema during the evaluation (caused by lymphedema), which is insufficient to draw a meaningful conclusion about the device’s effectiveness in such cases. Further assessment with more patients in similar situations is required to determine whether this should be considered a contraindication for the device.
The evaluation was not designed to assess the accuracy of the measurements obtained from the devices (weight or foot volumes), as both were CE-marked and expected to perform as certified. Furthermore, the validity of the alerts was not evaluated in this study. Although the clinical team did not feel that the number of alerts was excessive, we recommend future evaluation of this aspect in an observational clinical trial with a larger patient population. Participants were aware that we were evaluating the potential of this new device, which may have influenced their behavior. However, since the Heartfelt device is passive, it is difficult for patients to affect the quantity of data collected. We observed that one patient weighed themselves during the evaluation, despite not previously engaging with self-management recommendations. This change could be attributed to the enthusiasm for participating in an evaluation or concerns related to the COVID-19 pandemic when patients with chronic conditions were constantly reminded of their increased risk of death.
Lastly, the study unintentionally recruited a majority of male participants. While no data suggest that the device would be less suitable or effective for women, future evaluations should be more conscious of this biological sex imbalance, particularly as right-sided heart failure seems most amenable to monitoring via peripheral edema, and is known to skew toward a female patient population.
Conclusion
In conclusion, the COVID-19 pandemic has underscored the necessity for innovative and effective TM solutions in primary care practices, particularly in the management of heart failure patients at home. The Heartfelt device could be a practical and user-friendly solution, enabling clinical teams to monitor patients proactively while overcoming the challenges posed by reduced face-to-face appointments.
By providing continuous data through its entirely passive operation, the Heartfelt device can facilitate early detection of heart failure decompensation, allowing appropriate adjustments in medication(s). Furthermore, the technology has the potential to optimize patient care by tailoring community nurse visits according to individual patient needs, ultimately reducing the burden on healthcare systems and improving patient outcomes.
The results of this evaluation demonstrate the feasibility and promise of the Heartfelt device in a UK context, known for its world-class heart failure community care. It is important to consider the potential impact of this technology on a global scale, particularly in countries with varying levels of community care and healthcare infrastructure. Future studies should not only focus on exploring the long-term efficacy (reduction in morbidity and mortality) and data acquisition rates of the Heartfelt device but also assess its cost-effectiveness, integration with existing healthcare systems, and potential to reduce health disparities. Given that HF patients have an average life expectancy of 5 years after diagnosis, TM solutions such as this need to demonstrate benefits to patients and healthcare systems over extended periods.
By addressing these areas of inquiry and building on the positive findings of this service evaluation, the Heartfelt device may prove to be a transformative solution in the management of HF patients who previously were unable to perform self-management checks such as daily weighing, ultimately contributing to a more proactive, personalized, and efficient healthcare system.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
