Abstract

Until recently, discussions of semi-implantable or totally implantable middle ear hearing devices for treatment of sensorineural hearing loss have primarily centered around laboratory data and preliminary clinical trial data on small patient pools. The first implantable middle ear hearing device to be approved by the US Food and Drug Administration (August 31, 2000) and to receive the European Union authorization to affix the CE Mark (March 1998) was the Vibrant Soundbridge manufactured by Symphonix® Devices, Inc. in San Jose, California. At the end of March 2002, nearly 1,000 devices had been implanted worldwide. The primary developer of the device and co-founder of Symphonix Devices, Inc. is Geoff Ball. He founded the company in 1994 and for the next three years the company took the core technology, the Floating Mass Transducer® (FMT™), and developed the semi-implantable Vibrant Soundbridge.
The Soundbridge is intended for adults with a moderate-to-severe sensorineural hearing loss who have had previous experience with acoustic hearing aids. Figure 1 contains the targeted audiometric range of hearing loss for ensuring a positive outcome with the device. The device is electromagnetic and operates by transforming externally generated sound into direct, mechanical vibrations of the ossicular chain, specifically the incus.
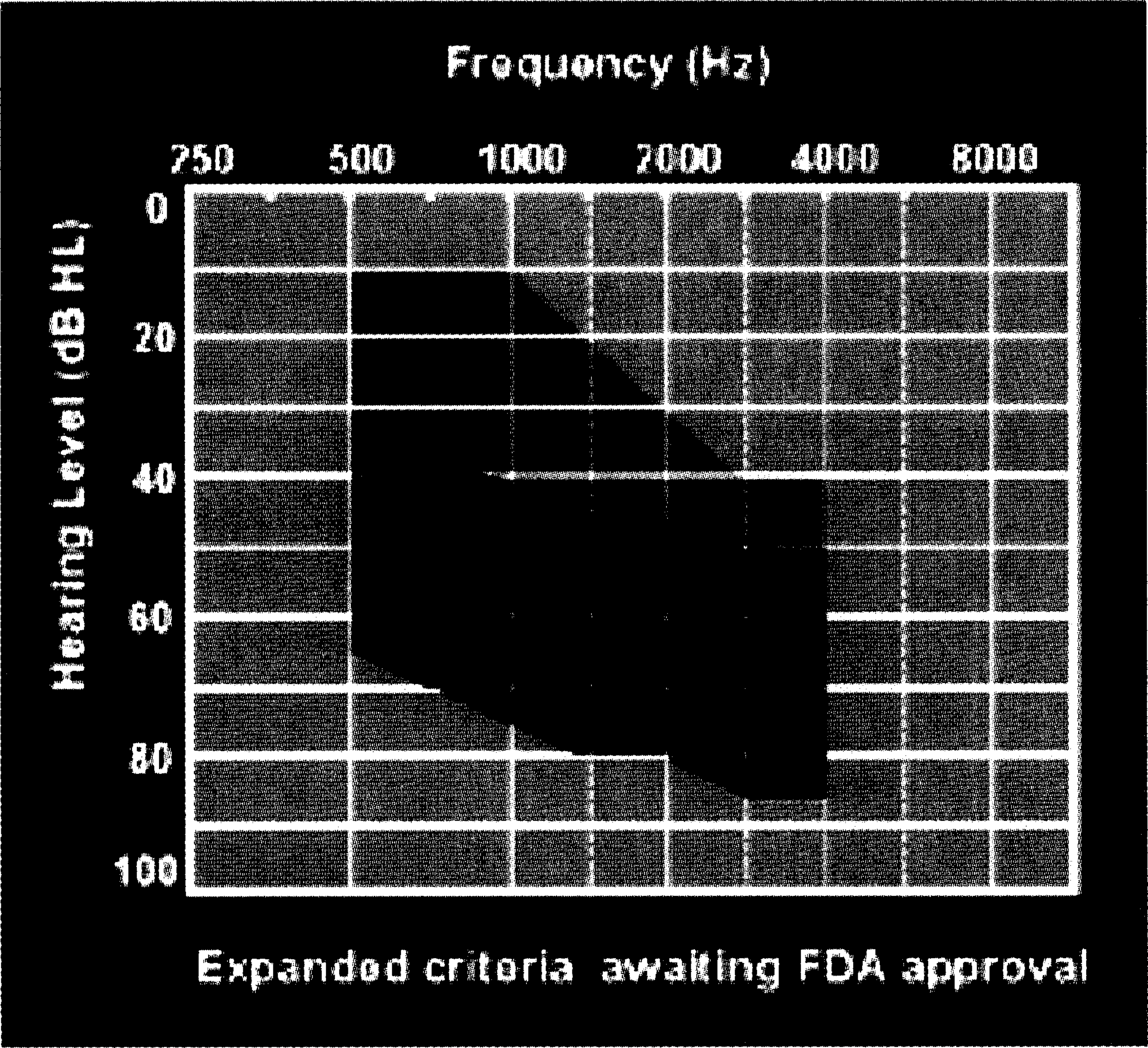
Graph shows the targeted audiometric range of hearing loss for ensuring a positive outcome with the device.
The Vibrant Soundbridge consists of two primary components: the Vibrating Ossicular Prosthesis™ (VORP™) and the Audio Processor™ (AP) that, when coupled, provide electromagnetic, direct-drive amplification. The VORP (Figure 2A) is surgically implanted under the skin posterior-superior to the pinna at a 45° angle to the ear canal. The AP contains an omnidirectional, electret microphone; a programmable, digital signal processor; modulator circuit, a magnet, and is powered by a single, standard 675 zinc air battery.

Magnetic attraction between the implanted magnet in the VORP and the magnet in the AP holds the AP securely to the scalp (Figure 2B). Hair can be worn over the AP, which makes it more cosmetically appealing.
The mode of operation consists of a signal being picked up by the AP's microphone, which is transmitted via an amplitude-modulated carrier across the skin to the internal receiver in the VORP. The signal is then sent through the demodulator via the conductor link to the FMT, which is attached onto the long process of the incus via a titanium attachment clip (Figure 2C).
The FMT of the Vibrant Soundbridge has been specifically designed to avoid mass loading the ossicular chain (Kartush and Tos, 1995). The tiny transducer (2.2 mm by 1.5 mm), as seen in the enlargement in Figure 2C, has a single, reversible attachment point and weighs around 25 mg. Because it is formed around the long process of the incus with a specialized “forming forcep,” the FMT does not permanently alter the ossicular chain or the middle ear space; therefore, this surgery is uniquely considered reversible. Patient safety from overstimulation of the FMT by excessively loud environmental stimuli is ensured by the action of the demodulator which separates the active signal from the carrier signal and contains circuitry to limit the maximum power that can be transmitted to the FMT.
The implanted transducer is composed of two coils wound around a hermetically sealed titanium alloy bobbin. The coil wire is 50 gauge, polyamide coated gold. The windings have a secondary layer of mechanical protection provided by a thick, conformal coating of silicone epoxy. Hermeticity is achieved by laser welding the bobbin seams. The bobbin contains a permanent magnet and two spring elements. The transducer requires a single attachment point. Previous conceptual devices suggested at least two points of attachment and meticulous surgical skill in aligning the components.
Actuation is achieved through inertial transfer of vibratory energy rather than a displacement force acting between two points (Dietz et al., 1997). During surgical implantation, the FMT is positioned such that the primary axis of the bobbin is aligned with the natural motion of the ossicular chain. From 1.5 to 6 kHz, the transducer driven with 1mA of current generates the equivalent of 100 dB SPL. The in vitro response of the transducer-driven ear is congruent with the sound-driven response.
Previous investigators have reported on the unique design of the electromagnetic FMT (Dietz et al., 1997) and the bench testing and in vivo testing which demonstrated that the FMT, when placed appropriately on the long process of the incus with its axis of motion parallel to the axis of the lenticular process, exhibited motion mimicking that of the ossicular chain. The use of “direct-drive” stimulation of the ossicular chain has been studied (Goode et al., 1995; Fredrickson et al., 1995; Hough et al., 1987; and Kartush and Tos, 1995) for its purported advantages to patients. These studies have indicated improvements in patient-perceived sound quality and signal distortion, and patient satisfaction with direct-drive technology.
Although the surgical procedure to implant the VORP has elements in common with other otologic procedures, such as implantation of cochlear prostheses, notable device-specific differences do exist. The attachment of the Floating Mass Transducer to the long process of the incus is particularly unique to the implantation of the VORP. Electric current passed through the coil produces vibrations of the magnet, which is capable of producing sound pressure level equivalents greater than 115 dB SPL when properly positioned on the incus (Tjellstrom et al., 1997; Snik and Cremers, 2000).
Test Methods and Clinical Results
Early results with the Vibrant Soundbridge looked at its safety profile as well as limited performance measures. These have been reported (Snik et al., 1998; Lenarz et al., 1998; Snik and Cremers, 1999; Fisch et al., 2001; Fraysee et al., 2001; and Snik et al., 2001) as a function of the European clinical trial (1997–98) and with subsequent commercially implanted patients. Test methods varied, but the majority of researchers consistently reported on residual hearing, adverse events, functional gain, patient self-assessments, and in some cases, speech recognition. The inclusion criteria for the patients varied, with some sites reporting results on mild-to-severe, as well as moderate-to-severe, sensorineural hearing loss. Study results consistently supported the stability in residual hearing with statistically significant changes seldom seen.
The US clinical trial began with Phase I in 1997 and culminated with Phase III in August 2000 with the 100th patient. The data from the five Phase I patients and first 53 Phase III patients were used to gain approval of the Vibrant Soundbridge Premarket Approval Application. The clinical trial was a prospective, single-subject, repeated measures study in which each patient served as his/her own control. Single subject designs, as opposed to more traditional group designs, explicitly recognize the heterogeneity and variability within the target population. All patients were current binaural users of appropriately fit acoustic amplification as determined by compliance with the prescriptive formula, NALR-1. Approximately 35% (18 of the 53) of the patients in this study were wearing digital or digitally programmable hearing aids. Patients were required to have bilateral, symmetrical sensorineural hearing loss within the range displayed in Figure 1. Additionally, previous otologic history was required to be “unremarkable,” with no significant measured fluctuations (15dB) in thresholds in the most recent two years. Further exclusion criteria included subjects with retrocochlear or central auditory disorders, and those with physical, psychological, or emotional disorders that would interfere with their ability to undergo testing or surgery. Further, patients had to exhibit word recognition ability at the implant ear exceeding 50% under earphones for stimuli presented at 30 dB SPL or MCL. Demographically, patients ranged in age from 28 to 86 years. Gender was evenly distributed at 50.9% (women) and 49.1% (men), while mean duration of hearing loss was 21 years. All patients had three months or more experience with acoustic hearing aids.
The patients were implanted at one of ten implanting centers in the United States. After implantation, the patients were able to continue using their existing acoustic hearing aid at the implant ear until healing had occurred (approximately eight weeks postsurgery). At that time, patients were fit with the external AP and the audiologist performed the appropriate programming. Initial Soundbridge test measures were made at activation, followed by retests four weeks later and, finally, three months postactivation. Patients were first evaluated with a WDRC, analog AP, Model 302, and then a subset of 50 were fit with a three-channel, digital circuit AP, the Model 304. The digital device represented an upgrade to the analog unit and offered the patients the same increased programming flexibility as seen in acoustic devices.
In an effort to reduce site-to-site test variability, standardized test methods and identical materials were used as part of a uniform clinical protocol. The patients were evaluated using a comprehensive test battery of both psychometric and audiometric tools. Measures included air-conduction/bone-conduction, immittance, functional gain, speech discrimination in quiet and noise, and patient self-assessments.
Residual hearing was the primary measure to determine “safety” of the procedure, with the occurrence of any adverse events as secondary indicators. In the original pivotal population of 53 patients implanted with the Soundbridge, 51 (96%) showed a shift in pure tone average (PTA) of less than 10 dB at the three-month postactivation interval. One subject experienced a shift in PTA of 12 dB, while a second patient had a shift of 18 dB. The mean difference in thresholds across the frequency range was less than 5 dB and was not found to be statistically significant. The mean change in SRT was 3 dB, consistent with the 2.7 dB mean shift in PTA. For the larger population of 100 patients, residual hearing results were consistent with those found in the initial 53 and were stable through one year, postoperatively (Figure 3).

A comparison of mean residual hearing thresholds.
Clinical immittance indicated 98% of subjects had Type A or Type As tympanometry results, postoperatively. One subject experienced negative pressure (Type C) at the final test interval but was also affected at the nonimplant ear. This was attributed to an upper respiratory condition that resolved over time. Loudness discomfort levels (LDLs) and most comfortable level (MCLs) suprathreshold measures were also compared to preoperative baseline. An analysis of group distribution (two-sided, paired t-test; P = 0.74 for MCL and P = 0.08 for LDL) revealed no significant differences. The type and incidence of adverse events with implantation of the Soundbridge were consistent with that of other similar, well-established otologic procedures (mastoidectomies and cochlear implants) and resulted in a very acceptable risk-to-benefit profile.
Functional gain was calculated at the implant ear with the contralateral ear occluded. Initial results with the 53 pivotal trial patients were obtained with the analog Model 302 AP (Vibrant P). With that device, mean increases in functional gain (compared to the presurgery appropriately fit hearing aid) were significantly increased (P < 0.01, Wilcoxon signed rank test and two-sided, paired t-test) at each measured frequency except 500 Hz. The introduction of the Model 304, three-channel digital AP (Vibrant D) at the end of the clinical trial revealed further increases in functional gain for a 50-patient subset of the original population. Mean increases in functional gain (Figure 4) were significant at all frequencies (P < 0.001, Wilcoxon signed rank test; two-sided, paired t-test). By 12 months postsurgery, all 100 patients were using the digital AP.

Mean functional gain of hearing aid compared to Vibrant D at 12 months (n = 54).
For laboratory measures of speech recognition with the device, in quiet, the Northwestern University (NU-6) Word Lists were used and for measures in noise, the Speech Perception Noise Test—Revised (SPIN-R). Soundbridge-aided word recognition in quiet (72%) using the NU-6 word lists indicated no significant difference from the presurgery (76%) hearing aid condition (P = 0.12, Wilcoxon signed rank test). There were many subjects whose word recognition scores were “good” before implantation, which resulted in a “ceiling effect.” With the SPIN-R, calculations were made for low-predictability (LP), high-predictability (HP) and composite scores. For comparison purposes, the LP scores were analyzed (Kalikow et al., 1977) due to their similarity to word recognition test items (monosyllabic words presented in a carrier phrase). While HP scores provide syntactic and semantic cues to the identity of the final word in the test, LP scores reflect basic word recognition skills in noise. Patients with the analog, as well as with the digital AP, failed to show any significant mean changes in LP scores when compared to their acoustic hearing aid, even though the subjects as a group demonstrated a mean increase of 6%.
Patient self-assessments included the Profile of Hearing Aid Performance (PHAP) (Cox and Gilmore, 1990), the Hearing Device Satisfaction Scale (HDSS) and the Soundbridge Hearing Aid Comparison Questionnaire (SHACQ). The PHAP was used to assess subject-perceived improvements in everyday and challenging listening situations. The construct of the inventory uses seven subscales that contain multiple descriptions of listening situations. Those subscales include Familiar Talkers, Ease of Communication, Reverberation, Reduced Cues, Background Noise, Aversiveness, and Distortion of Sounds. Patients were asked to fill out the questionnaire prior to implantation, again six months later after implantation, (with a three-month acclimitization period with the Vibrant P), and then several months later after fitting and six week's use with the digital AP (Vibrant D). The improvement in PHAP scores with the Vibrant P and the Vibrant D was significant (P = 0.001, Wilcoxon signed rank tests) compared to the aided presurgery condition. Figure 5 depicts the PHAP scores for the presurgery, hearing aid condition and with the Vibrant D. Individual subject analyses looked at significant improvements for a subject in each of the PHAP subscales. This was determined by estimating the critical difference values calculated from standard error values reported by Cox and Gilmore, 1990.

Graph demonstrates the improvement in Profile of Hearing Aid Performance scores with the Vibrant P and the Vibrant D compared to hearing aid.
The HDSS was developed by Symphonix to obtain information on the satisfaction level of the subjects with their Soundbridge as well as their hearing aid in variety of listening situations. As with the PHAP, individual and group results indicated significantly increased levels of satisfaction with the Soundbridge compared to their hearing aid for a wide range of listening situations and sound quality issues. Specifically, of the subjects completing the question regarding “Overall Sound Quality”, 94% (44) improved their satisfaction rating with the Soundbridge and as did 88% on the query regarding “Sound Quality of Own Voice.” As for feedback, of the 32 (60%) patients reporting it with their hearing aids, only one reported it with the Soundbridge and it was programmed “out.” Fit and comfort was also a category in which all of 11 previously dissatisfied hearing aid patients were 100% satisfied with the Soundbridge.
Patients wearing digital or digitally programmable acoustic devices presurgi-cally (n = 18/53) were extracted as a group and their results analyzed for PHAP and functional gain. Those patients experienced statistically significant improvements between hearing aid and Soundbridge for all seven subscales of the PHAP, and they experienced increased gain (statistically significant) for functional gain measures of 1000, 1500, 2000, and 4000 Hz. No difference was apparent between the perceived satisfaction levels with the Soundbridge in patients who presurgi-cally wore digital/digitally programmable or analog devices.
Future Directions
Since the Vibrant Soundbridge was approved in August 2000, the AP has been “upgraded” twice. Model 404, the most recent upgrade to the digital AP, has shown increased flexibility in programming. This most recent eight-channel, six-band (compression) digital AP also comes in a variety of colors (gray, light brown, and dark brown) and is programmed using the CONNEXX™ platform with a customized database called Symfit. All of the new APs are designed to be compatible with the implanted model 502 VORP, thus ensuring patients the opportunity for continual improvements in hearing and increased device options. Current development for the semi-implantable device involves a slimmer profile, directional microphone options, and multi-programming switches. The first generation, totally implantable Soundbridge is completing verification testing at this time and will soon be ready for clinical trials.
Implantable hearing technology has been proven to be safe, effective, and a highly desirable option for conventional, acoustic hearing aid users. The Vibrant Soundbridge has been on the forefront of development of this technology. Long-term results, 24-, 36-, and 48-month data is being acquired and will be the subject of future publications.
