Abstract
Anemia affects a significant portion of the population, and zinc deficiency may contribute to its pathogenesis. This study examined the impact of zinc supplementation on hemoglobin and zinc levels across high-risk subgroups. We conducted a retrospective study of adults ≥18 years with chronic anemia and zinc deficiency who received daily zinc sulfate 220 mg. Hemoglobin and plasma zinc were recorded at 3, 6, and 12 months. Subgroups included patients stratified by chronic kidney disease (CKD), body mass index (BMI), proton pump inhibitor (PPI) use, age >70, poly-micronutrient deficiency, and malnutrition or prior bowel resection. Among 125 patients (median age 68), mean hemoglobin increased from 9.8 g/dL to 11.4 g/dL by 12 months (p = 0.000022), with corresponding increases in zinc levels. Anemia resolved in 42% of patients. Zinc deficiency resolved in 63.8%. Those with CKD, malnutrition, prior bariatric surgery, or PPI use showed significant hemoglobin improvement after zinc replacement and PPI discontinuation. No statistically significant differences in response were observed by BMI. Zinc supplementation improved hemoglobin across multiple high-risk groups, suggesting an independent role of zinc in erythropoiesis. Limitations include the retrospective design, concurrent treatment of comorbidities including hematologic malignancies, and non-uniform monitoring of inflammatory markers. Zinc deficiency is an independent and reversible contributor to anemia. Screening and supplementation will need to be studied in a prospective trial to clarify the clinical significance of zinc deficiency in chronic and otherwise unexplained anemia, particularly among those with CKD, advanced age, malnutrition, prior bowel resection, and PPI use.
Introduction
Based on data from the early 2020s, anemia is present in nearly 1 in 10 people over the age of 2 in the United States. 1 Roughly 12% of adults over 65 years living in the community have anemia. In those greater than 80 years of age, approximately 24% have anemia. 2 While iron deficiency remains the most common form of anemia worldwide, ongoing research shows the impact of key micronutrients in maintaining adequate hemoglobin. 3 Zinc is a key micronutrient that plays many diverse roles in the body, including protein creation, wound healing, and immune response. 4 It is the second most common transition metal in living organisms after iron and is involved as a cofactor for over 300 enzymes. 5 Zinc deficiency has also been linked to anemia through many roles, including the zinc-dependent enzyme, erythrocyte copper-zinc superoxide dismutase, which provides anti-oxidative protection of red blood cells. As shown in Figure 1, Zinc is involved in heme synthesis by acting as the catalyst for the alpha-aminolevulinic acid dehydratase enzyme. 6 Zinc appears to also be involved in red blood cell production through zinc finger proteins GATA-1 and Gfi-1B. 2 Zinc has been shown to redistribute after induced erythropoiesis, further supporting its critical role in red blood cell production. 7 Studies have shown that a zinc-deficient diet in rats and mice results in decreased erythrocyte production and decreased erythroid lineage cells. 8 Recent studies by Jeng and colleagues demonstrated that zinc supplementation in rats stimulates erythropoiesis. 8
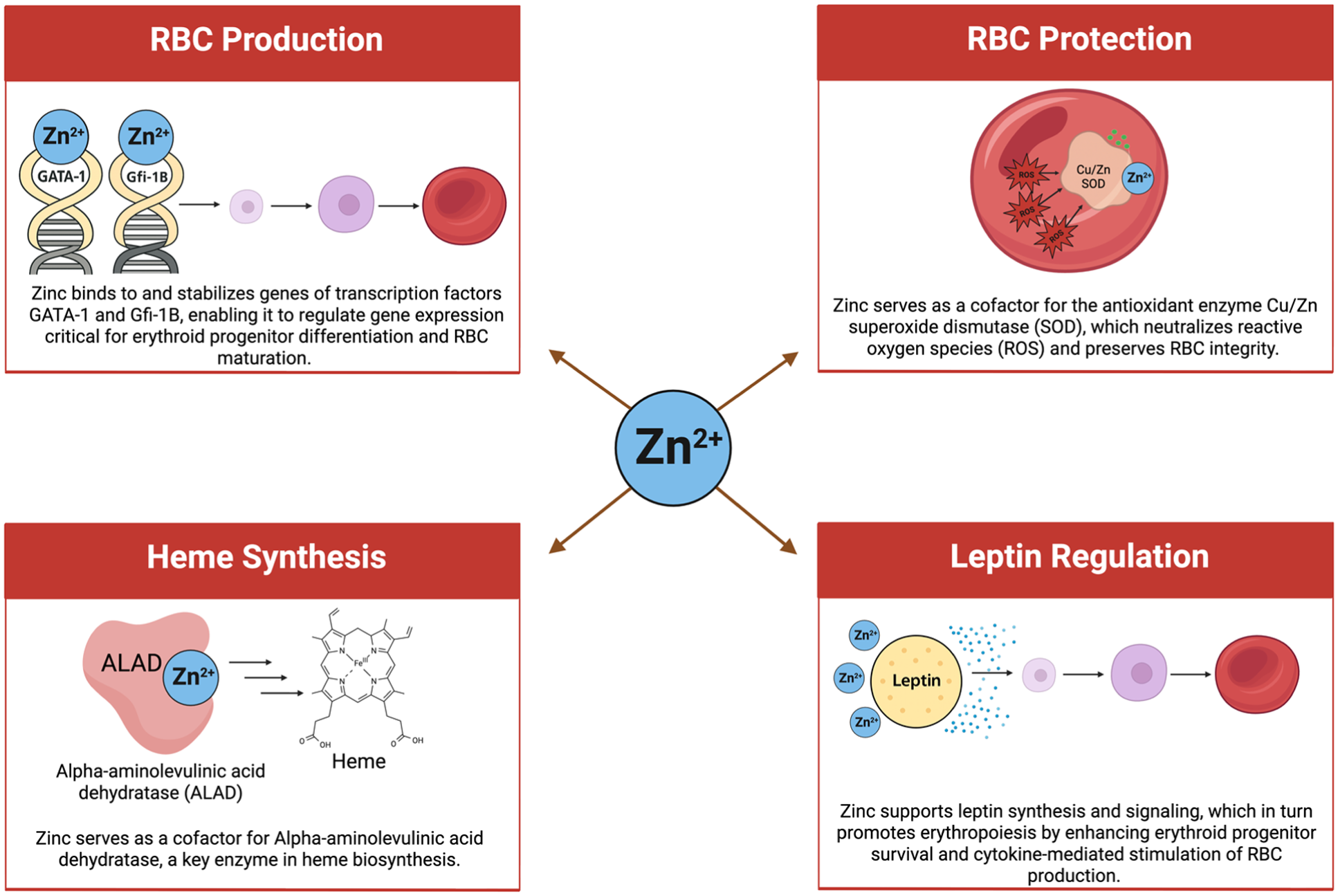
Roles of zinc in erythropoiesis. Created in BioRender.
Zinc is naturally found through dietary sources including meat and meat products, cereals, grains, milk and dairy products, fish, and vegetables. 10 Once ingested, zinc is primarily absorbed in the proximal small intestine. 4 Zinc cannot be stored in significant amounts in the body and therefore requires regular intake to maintain adequate levels. 11 It is estimated that approximately 17% of the world’s population is at risk of zinc deficiency. 12 Symptoms of zinc deficiency range based on the severity of the deficiency. Severely deficient states can cause alopecia, diarrhea, weight loss, increased susceptibility to infection, hypogonadism, and neurosensory disorders. In moderately deficient states, symptoms may include rough skin, poor appetite, mental lethargy, and delayed wound healing. 13 As we learn more about the many critical roles that zinc serves, it is crucial to understanding what risk factors predispose an individual to zinc deficiency.
Zinc deficiency has been linked to both malnutrition and obesity.14,15 Malnutrition is increasing in prevalence in the aging population and has multiple negative associations such as decreased immune function, anemia, and increased mortality. 16 Zinc deficiency may contribute to worsening malnutrition as it can result in decreased appetite through its interaction with leptin. 17 In one study, zinc deficiency was seen in 24% to 28% of people with obesity. 15 It is believed that zinc deficiency in obesity is due to a chronic inflammatory state, poor nutrition, and metabolic imbalances. 4 Many individuals with obesity will pursue some form of bariatric surgery; however, bariatric surgery is not without its risks. Those who have undergone bariatric surgery are at increased risk of zinc deficiency, likely secondary to decreased absorption.15,18 This is similarly seen in individuals with malabsorptive disorders such as ulcerative colitis, where absorption of micronutrients is impaired. 19 The decreased gastric acid that occurs after many bariatric surgeries also leads to impaired zinc solubilization and absorption. 4
A preliminary analysis of this data set focused on CKD patients with zinc deficiency showed that patients’ hemoglobin levels improved and their anemia resolved with zinc supplementation. 20
It is estimated that roughly 12% of adults over 65 years living in the community have anemia. In those greater than 80 years of age, approximately 24% have anemia. MacDonell et al. conducted a study of 285 adults greater than 65 years of age who reside in a care facility which showed that 72% had zinc deficiency, 32% had anemia, and <2% had low iron stores or iron deficiency. Those with zinc deficiency were five times more likely to have anemia compared to those with normal zinc levels. Interestingly, a univariate regression showed a statistically significant relationship between anemia and advanced age, taking gastric acid suppressing medications and zinc deficiency. 2
Along with the increasing prevalence of malnutrition as we age, many elderly individuals are on PPI medications or have undergone procedures such as vagotomies or gastric resections that lead to reduced gastric acid. This decrease can result in small bowel bacterial overgrowth which has been shown to lead to decreased weight and decreased intake of micronutrients.16,21 Proton pump inhibitors were the third most commonly prescribed medication class in 2009, and it has been shown that those on long-term PPI therapy have decreased zinc stores and worse response to zinc supplementation. 22
At this time, there is no standard recommendation for increased zinc intake, as healthy aging does not typically result in zinc deficiency. It is recommended to supplement micronutrients in the case of deficiency. 16 Zinc supplementation must be done cautiously as it has been linked to decreased copper when given at high doses. 23
Although zinc is known to be critical for many key cellular processes, its physiologic roles and states that predispose to deficiency are not well characterized. This retrospective study aims to explore the relationship between zinc deficiency and anemia, specifically focusing on populations with concurrent polynutrient deficiencies, obesity, malnutrition, prior bowel resection, advanced age, and those on proton pump inhibitor therapy.
Summary
What is already known about this subject?
Zinc plays a critical role in erythropoiesis.
Zinc deficiency is increasingly recognized as a contributor to anemia.
PPI use and gastrointestinal surgeries may exacerbate zinc deficiency.
What are the new findings?
Patients on PPIs should be closely monitored for micronutrient deficiencies including zinc.
Zinc supplementation appears to be effective across BMI categories.
How might it impact clinical practice in the foreseeable future?
Clinicians may check zinc levels in those with unexplained anemia, especially in high-risk groups.
Zinc supplementation may be tried as a low-risk, adjunctive therapy.
Clinicians may have increased scrutiny of long-term PPI use.
Methods
Inclusion criteria for all populations, unless otherwise stated, included individuals over the age of 18 with a diagnosis of chronic anemia (hemoglobin [HGB] <12 g/dL for women, HGB <13 g/dL for men for >3 months) identified to have zinc deficiency (plasma zinc, pZc < 65 μg/dL). Zinc Sulfate 220 (50)mg capsules were given daily. For each cohort, a retrospective analysis was completed including pertinent past medical history and lab data. HGB and pZc were recorded at 3, 6, and 12 months following initiation of micronutrient supplementation. Data was analyzed using ANOVA and CHI-squared.
Zinc deficiency and CKD stage 3 or above
Fifty-three patients met the inclusion criteria. The cutoff of GFR was <60 mL/Min/1.73 m2 for >3 months.
Zinc deficiency and BMI
One hundred and twenty-five patients met the inclusion criteria. These patients were further subdivided according to BMI, where BMI < 18 was considered underweight, 18–25 was normal, 25–30 was overweight, and > 30 was obese.
Zinc deficiency and PPI use
Forty-eight patients met the inclusion criteria and were on a PPI within 3 months of diagnosis of Zn deficiency. PPI medications were discontinued.
Zinc deficiency in the elderly
Fifty-nine patients over the age of 70 met the inclusion criteria of chronic anemia and zinc deficiency and were started on zinc supplementation.
Zinc deficiency with poly-micronutrient deficiencies
Seventy-five patients met the inclusion criteria and were found to have at least one other micronutrient deficiency at the time of Zn deficiency diagnosis. Micronutrient deficiencies included iron (ferritin <45 ng/mL and transferrin saturation <20%), copper (serum copper level <85 mcg/dL), B12 (serum B12 <400 pg/mL and methylmalonic acid >0.4 umol/L), and manganese (serum manganese <0.5 μg/dL). Those who had been receiving micronutrient replacement for iron deficiency, B12 deficiency, or folate deficiency within 3 months of zinc deficiency diagnosis were included.
Zinc deficiency with malnutrition or prior bowel resection
Twenty-three patients met the inclusion criteria and had a history of bowel resection, bariatric surgery, chronic alcohol use, or were diagnosed with protein-calorie malnutrition. Protein-calorie malnutrition was defined by a loss of more than 5% of body weight in 12 months or decreased oral intake of 50% of the recommended intake.
Results
Demographic and baseline characteristics
A total of 125 patients, aged 18–94 years, were included in the data analysis. Demographic and baseline characteristics are shown in Table 1. Zinc supplementation was started in all patients, and PPIs were discontinued.
Demographics and baseline characteristics of those with concurrent zinc deficiency and anemia.
Zinc deficiency and CKD stage 3 or above
This study included 53 patients with a median age of 72 years, 37.7% of whom were women. The number of patients with CKD 3 was 36, CKD 4 was 3, and CKD 5 or ESRD was 14. Baseline labs showed a mean HGB of 9.8 g/dL and pZc of 51.6 μg/dL. After starting zinc supplementation, mean HGB increased to 10.3 g/dL at 3 months (p = 0.07), 10.9 g/dL at 6 months (p =0.0026), and 10.8 g/dL at 12 months (p = 0.0039). Corresponding mean pZc values rose to 73.6 μg/dL at 3 months (p = 0.0000000007), 73.4 μg/dL at 6 months (p = 0.000000000083), and a mean of 69.3 μg/dL at 12 months (p = 0.0000000023). Overall, anemia resolution was reached in 33.9% of patients (18/53), and zinc deficiency resolved in 50% (8/16) as shown in Figure 2.
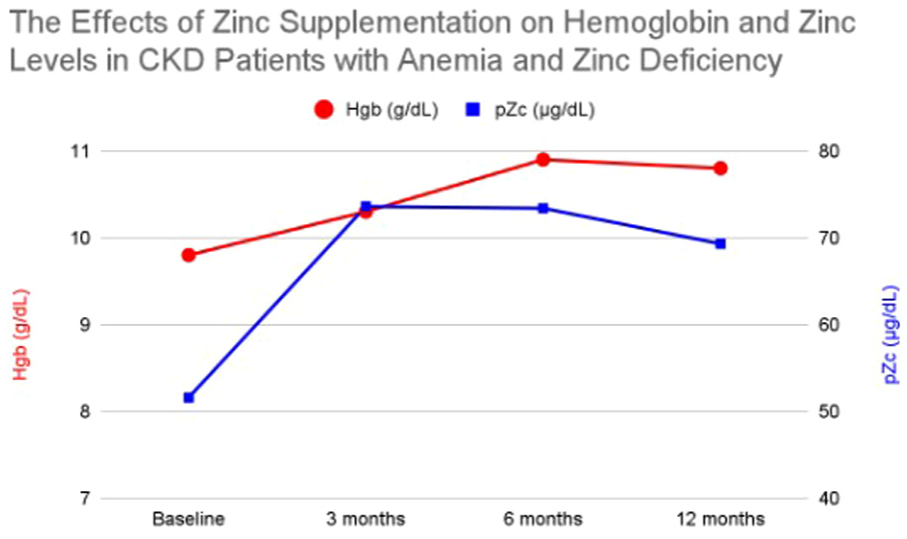
Average hemoglobin and plasma zinc levels in CKD patients with anemia and zinc deficiency at baseline and after 3, 6, and 12 months of zinc supplementation.
Zinc deficiency and BMI
This study included 125 patients with a median age of 68 years, 58.4% of whom were women. The mean BMI was 26.8 (range 13.3–58.0). Baseline labs showed a mean HGB of 9.8 g/dL and pZc of 52.5 μg/dL with no significant differences between BMI subgroups. HGB levels across BMI groups remained statistically non-significant at 3, 6, and 12 months following zinc supplementation (all p > 0.2). After starting zinc supplementation, mean HGB increased to 10.7 g/dL at 3 months (p = 0.000042), 11.3 g/dL at 6 months (p = 0.00000000066), and 11.4 g/dL at 12 months (p = 0.000022). Corresponding mean pZc values rose to 70.3 μg/dL at 3 months (p = 0.0000000000000000017), 74.7 μg/dL at 6 months (p = 0.000000000000091), and a median of 67.9 μg/dL at 12 months (p = 0.000000000000089). Overall, anemia resolution was reached in 42% of patients (53/125), and zinc deficiency resolved in 63.8% (62/84).
Zinc deficiency and PPI use
Prior to zinc replacement and PPI cessation, patients had an average HGB of 9.6 g/dL and mean pZc of 50.8 μg/dL. After 3 months of zinc supplementation, average HGB was 10.6 g/dL (p = 0.012) with a pZc of 67.9 μg/dL (p = 0.000000022). At 6 months, HGB reached 11.7 g/dL (p = 0.00000057) and pZc was 66.6 μg/dL (p = 0.0000049). At 12 months, HGB averaged 11.3 g/dL (p = 0.000041) and pZc was 61.5 μg/dL (p = 0.0015). Anemia resolution was reached in 23 of 48 patients (48%) as shown in Figure 3.
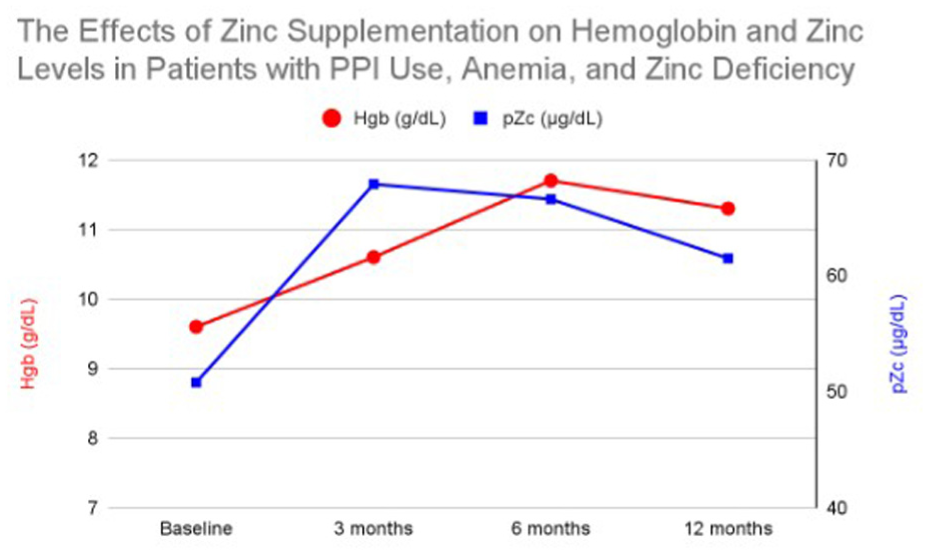
Average hemoglobin and plasma zinc levels in patients with PPI use, anemia, and zinc deficiency at baseline and after 3, 6, and 12 months of zinc supplementation.
Zinc deficiency in the elderly
Before zinc replacement, patients had an average HGB of 10.1 g/dL and a mean pZc of 52.8 μg/dL. After 3 months of zinc supplementation, HGB averaged 10.6 g/dL (p = 0.101) with a pZc of 70.5 μg/dL (p = 0.0000000000036). At 6 months, HGB rose to 11.06 g/dL (p = 0.0051) and pZc reached 80.3 μg/dL (p = 0.000000055). By 12 months, HGB averaged 11.01 g/dL (p = 0.018) and pZc was 70.8 μg/dL (p = 0.0000000000047). Anemia resolution was reached in 39% of patients (23/59), and zinc deficiency resolved in 69% (41/59) of patients as shown in Figure 4.
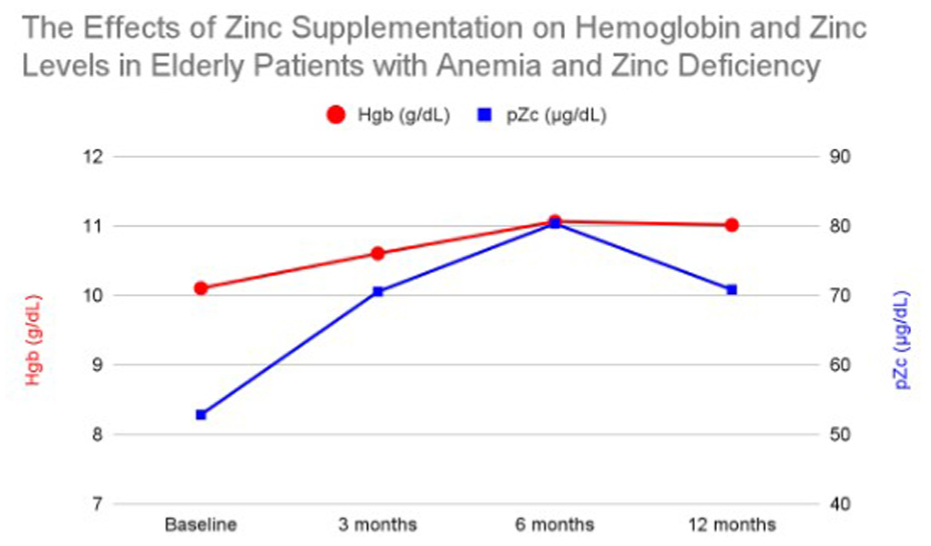
Average hemoglobin and plasma zinc levels in elderly patients with anemia and zinc deficiency at baseline and after 3, 6, and 12 months of zinc supplementation.
Zinc deficiency with poly-micronutrient deficiencies
Among 75 patients with poly-micronutrient deficiencies, iron deficiency was identified in 19 of 64 tested patients, with 35 receiving oral and 12 receiving IV iron. Copper deficiency was found in 13 of 63 patients, B12 deficiency in 7 of 34, and manganese and selenium deficiencies in 6 of 12 and 7 of 24 tested patients, respectively. No folate deficiency was detected among 36 tested individuals, though 6 patients were on folate supplementation prior to zinc deficiency diagnosis. Overall, 28 patients had two deficiencies, 6 had three, and one patient had four. At baseline, the mean HGB was 9.7 g/dL and pZc was 52.1 μg/dL. Following zinc replacement, mean HGB increased to 10.79 g/dL at 3 months (p = 0.00012) and pZc was 69.4 μg/dL (p = 0.000000000022). At 6 months, HGB reached 11.5 g/dL (p = 0.0000000059) with pZc of 75.0 μg/dL (p = 0.000000048). At 12 months, HGB was 11.6 g/dL (p = 0.0000000095) with pZc of 68.1 μg/dL (p = 0.000018). Anemia resolution was reached in 51% of patients (38/75).
Zinc deficiency with malnutrition or prior bowel resection
Twenty-three patients met the inclusion criteria, including 13 with a history of bowel resection or gastric bypass surgery, 12 with protein-calorie malnutrition, and 3 with chronic alcohol use disorder. At baseline, the mean HGB was 9.6 g/dL and pZc was 51.9 μg/dL. After 3 months of zinc replacement, mean HGB increased to 11.05 g/dL (p = 0.0052) and pZc reached 72.9 μg/dL (p = 0.0013). At 6 months, HGB rose to 12.0 g/dL (p = 0.000038) with pZc of 74.5 μg/dL (p = 0.0007). At 12 months, HGB reached 12.2 g/dL (p = 0.000018) with pZc of 70.4 μg/dL (p = 0.0015). Anemia resolution was reached in 47.8% of patients (11/23), and zinc deficiency resolution occurred in 75% of patients as shown in Figure 5.
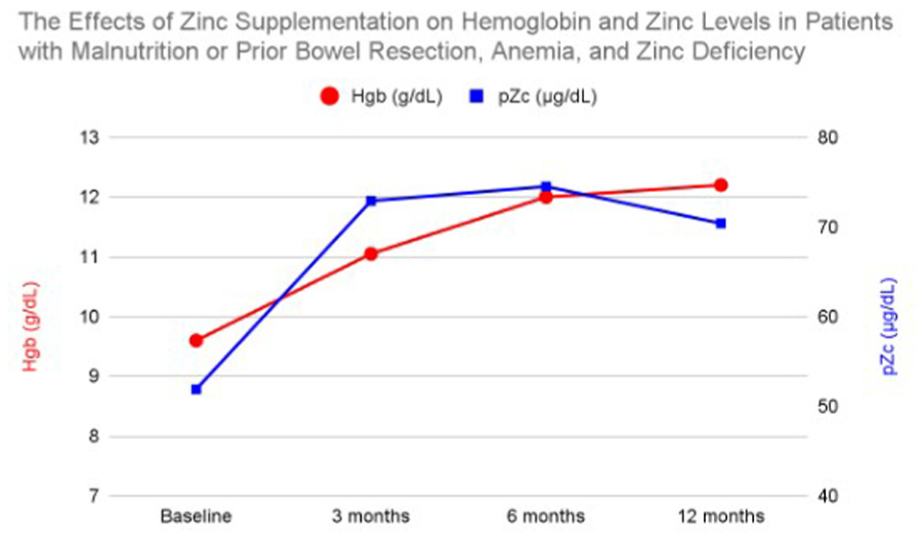
Average hemoglobin and plasma zinc levels in patients with malnutrition or prior bowel resection, anemia, and zinc deficiency at baseline and after 3, 6, and 12 months of zinc supplementation.
Discussion
Our study again demonstrates the link between zinc deficiency and anemia and its reversibility through zinc supplementation. In many high-risk subgroups, including those with CKD, advanced age, prior bowel resection, or malnutrition, zinc supplementation alone appeared to be sufficient in correcting zinc deficiency.
In our study, no clear relationship was seen between zinc deficiency and BMI. Likewise, BMI did not significantly influence HGB response to zinc supplementation which suggests zinc supplementation is effective regardless of BMI. Given the high prevalence of zinc deficiency in those pending bariatric surgery and those in the postoperative period, it is recommended to monitor closely for zinc deficiency in these populations. 24
Patients with zinc deficiency who had CKD were found to have a significant improvement in hemoglobin levels, with resolution of anemia in 34% of patients. As chronic anemia is prevalent in CKD, and often leads to the need for erythropoietin (EPO), zinc may have a role as an adjunct supplementation in order to resolve anemia and reduce dependence on EPO. Future investigation is needed to evaluate the use of zinc supplementation in CKD patients without deficiency.
Patients with zinc deficiency taking PPI medications showed notable improvement in hemoglobin after zinc supplementation and PPI discontinuation. This supports prior findings that gastric acid suppression with PPI therapy leads to decreased intra-luminal zinc absorption due to elevated pH. 25 This reinforces the need for cautious long-term use of PPI therapy and suggests the need for monitoring of micronutrients in chronic users.
In the elderly population, zinc supplementation led to significant anemia resolution. Notably, their hemoglobin increase was not statistically significant until month 6 of zinc supplementation suggesting that there may have been compounding factors in this population such as decreased absorption or polypharmacy. Couzy et al. conducted a study comparing zinc absorption in a young population of men (24–40 years) with an elderly population (70–83 years) and found no difference in zinc absorption between the groups. 26
Our study demonstrated a high co-occurrence of zinc deficiency with iron, copper, and B12 deficiencies. This suggests the need for a broader diagnostic workup in patients presenting with anemia of unclear etiology. For those with polynutrient deficiencies, supplementation of deficient micronutrients, including zinc, led to improved hemoglobin levels.
Limitations of our study include its retrospective design which limits control over confounding variables such as undocumented dietary changes or the presence of inflammation or chronic illness. It has been shown that when measuring zinc levels, inflammation should be taken into account as plasma zinc concentration is lowered in an inflammatory state. 14 Without accounting for markers of inflammation (CRP, ESR, IL-6), it is difficult to determine how much hemoglobin improvement is due to zinc supplementation or the resolution of an underlying inflammatory process. Given the variability in follow-up testing rates, there is also the possibility of selection bias. Because no racial/ethnic stratification or data on socioeconomic status was completed, there is limited generalizability.
Even with these limitations, we would recommend considering routine zinc screening in patients with CKD, chronic anemia, especially in elderly, malnourished, PPI users, and post-bariatric surgery patients. Additionally, zinc supplementation may serve as a low-risk adjunct in anemia management particularly for those with normocytic anemia without overt iron deficiency. Lastly, we would recommend that primary care and geriatric physicians frequently reevaluate chronic PPI use and monitor for potential micronutrient malabsorption.
Footnotes
Acknowledgements
We acknowledge patients and staff at the cancer center of Loma Linda University Medical Center for their support. We also appreciate the support of the Department of Medicine, the Internal Medicine Residency Program, and the office of GME.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
