Abstract
Obesity occurs with a low-grade inflammation as a result of the imbalance between energy intake and expenditure. This physiopathological status is accompanied by alterations in the gut microbiota. Living microorganisms called “probiotics” can affect the gut microbiota and can heal this imbalance, thus obesity. The aim of this study is to evaluate the changes in the gut microbiota in high-fat-diet-induced rats and to investigate whether any difference may be observed with the supplementation of
Keywords
Introduction
The human gastrointestinal tract is home to over 100 trillion bacteria. Gut has an anaerobic environment; obligate anaerobes, including
Obesity is based on energy imbalance, and it is characterized by chronic “low-grade” inflammation. This inflammation is associated with some changes in intestinal epithelial functions and microbiota composition.
7
Living microorganisms that affect beneficially the host by improving the balance of gut microbiota are called “Probiotics.” Recently, studies have pointed to the relationship between the body weight, fatness, IR, and gut microbiota, and many studies have showed the healing effects of the probiotics on obesity.8,9
Significance Statement
There are some studies showing that the gut microbiota is associated with obesity-related disorders.
This study showed the change of gut microbiota both with high-fat diet and with the supplementation of
Studies to be conducted with these observed species may provide clearer results regarding their relationships with the probiotic use against different nutritional regimens.
Materials and methods
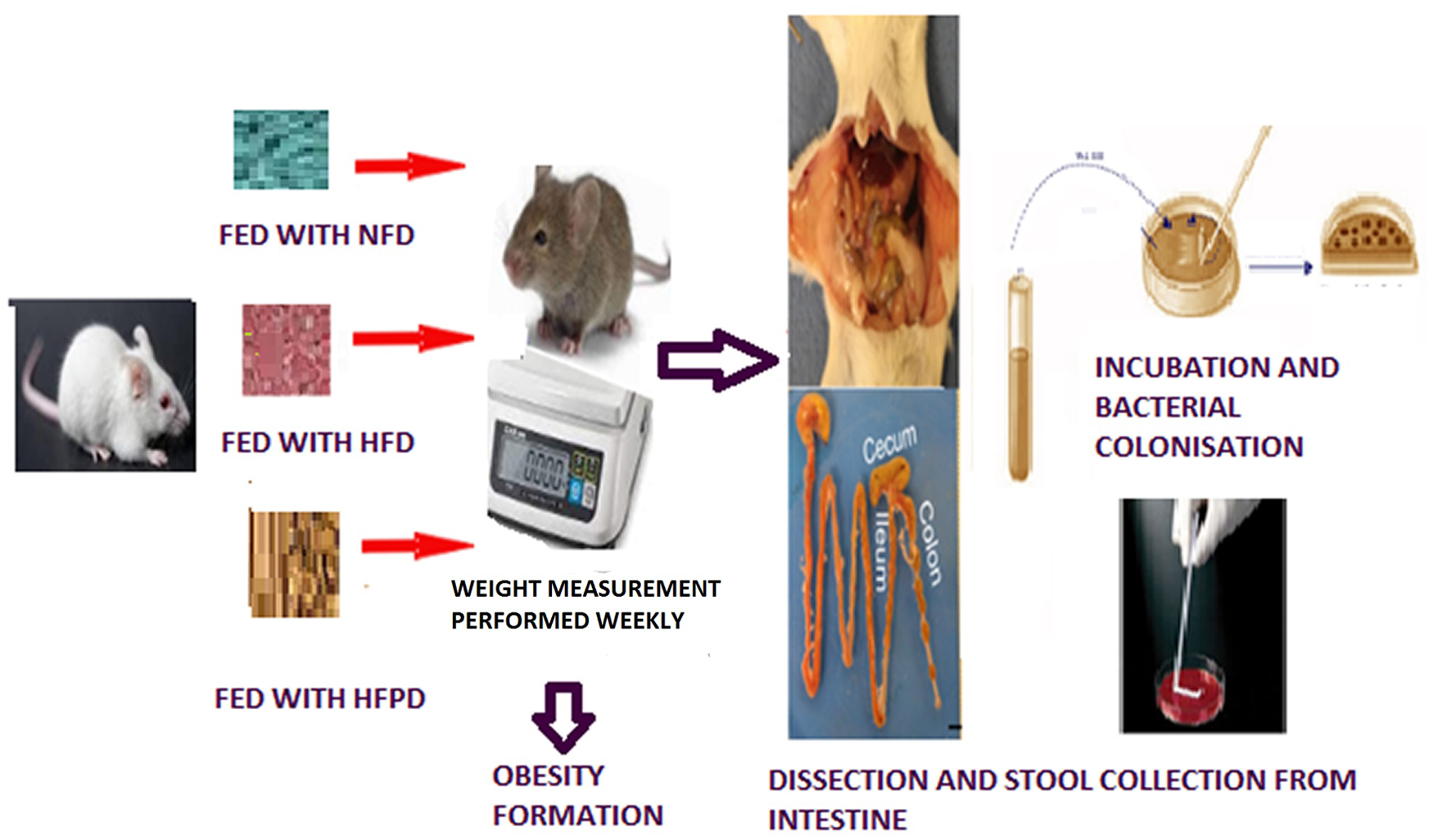
Animals and dietary intervention
This study was performed in Animal Experiments Laboratory of Medicine Faculty of University Ordu, Ordu, Turkey. Twenty-week-old male Sprague–Dawley rats weighing 250–300 g (
The probiotic containing
The rats were weighed in a tared plastic container with a lid. For weight measurement, an AND FZ 5000İ digital precision scale (AND, A&D COMPANY, LIMITED, Japan) was used. Rat weight was recorded on a weekly basis. The Lee index was used in order to calculate the obesity index. 13 At the end of the 8-week treatment period, after the fasting time of 6 h, all rats were sacrificed by cervical dislocation under deep anesthesia with intraperitoneal of ketamine 90 mg/kg (Ketalar; Eczacıbaşı, Istanbul, Turkey) and xylazine hydrochloride 3 mg/kg (Rompun; Bayer, Leverkusen, Germany) frequently used for anesthesia in rats. 14 The sigmoid column removed and was sent for examination of the intestinal flora bacteria.
Biochemical analysis
Blood samples were collected by entering from the bifurcation aorta with a 10 cc injector, and 4–6 cc of blood was taken from each rat. Blood samples were collected in anticoagulant-free biochemical tubes and transferred to appropriate laboratory parameter tubes. Glucose measurements were made by colorimetric method and insulin measurements were made by ELISA (cat no. 201-11-0708-48t; SUNRED, Shanghai, China). The homeostasis model assessment–IR (HOMA–IR) was calculated for the measurement of IR from fasting glucose and insulin.
Isolation and characterization of bacteria
Rats were fed on their respective diets for 8 weeks. Moreover, on the 0th, 3rd, 6th, and 8th weeks of the feeding period, feces were collected from each rat. Fecal samples taken from rats were brought to the microbiology laboratory under sterile conditions in half an hour. Immediately following collection and weight determination, stool samples were dissolved in filter-sterilized phosphate buffer. (Brine (PBS) solution (per liter dH2O: NaCl, 8.60 g; Na2HPO4, 0.86 g; KH2PO4, 0.40 g, pH 7.2), 0.02% peptone from fortified meat (Roth, cat no. 2366), and 0.05% L-cysteine Tenfold serial dilutions were made and prepared for each sample. Bacterial suspensions (100 µl) were spread on Mueller–Hinton Agar 103872 (Merck Microbiologycal Frankfurt Germany) and CASO (Trypic Soy) agar using sterile glass rods. Counts were determined after 6 days of incubation. Only plates containing 20–200 colonies/plate were obtained. All single colony morphology species observed after 6 days of growth were streaked on to Trypic Soy agar to ensure purity. Culture purity was examined by observing cell morphology after gram staining and colony morphology. Cryostocks (100 µl) bacterial suspensions were stored at −80 °C after mixing equal volumes of Tris-buffered aqueous solution (60 mM) containing 40% glycerol. 15 After gram and spore staining, 130 bacterial colonies were reduced to 50 colonies under the microscope and sent for analysis. Microbial characterization was performed using the automated system VITEK® 2 (BioMérieux, Craponne, France). In order to transfer a sufficient number of bacterial samples, they were previously passaged into blood agar and EMB agar (GBL, Biology Laboratory, Istanbul, Turkey) medium 24–48 h before using the applicator stick. Colonies of the pure culture were suspended and loaded into the device. The test data from the unknown organism were compared with the database of the device, defined according to their proximity to each of the database taxa and Eu cast criteria. 16
Statistical analysis
All statistical analysis was performed using SPSS 26 program (Sosa) IBM. Obtained data were presented as the mean with either standard deviation or standard error of the mean. The comparison between the groups were made using Tukey test, one-way analysis of variance. Differences were considered statistically significant at
Ethic statement
The animal experiments were conducted with the approval of the Animal Experiments Local Ethics Committee of University Ordu (February 16, 2021, no. 82678388). All methods of this study were performed in accordance with the relevant guidelines and regulations of this committee.
Results
Bacterial flora
It was determined that the feces of rats fed with an HFD contained
Gut microbiota analysis of rats with different feeding regimes.
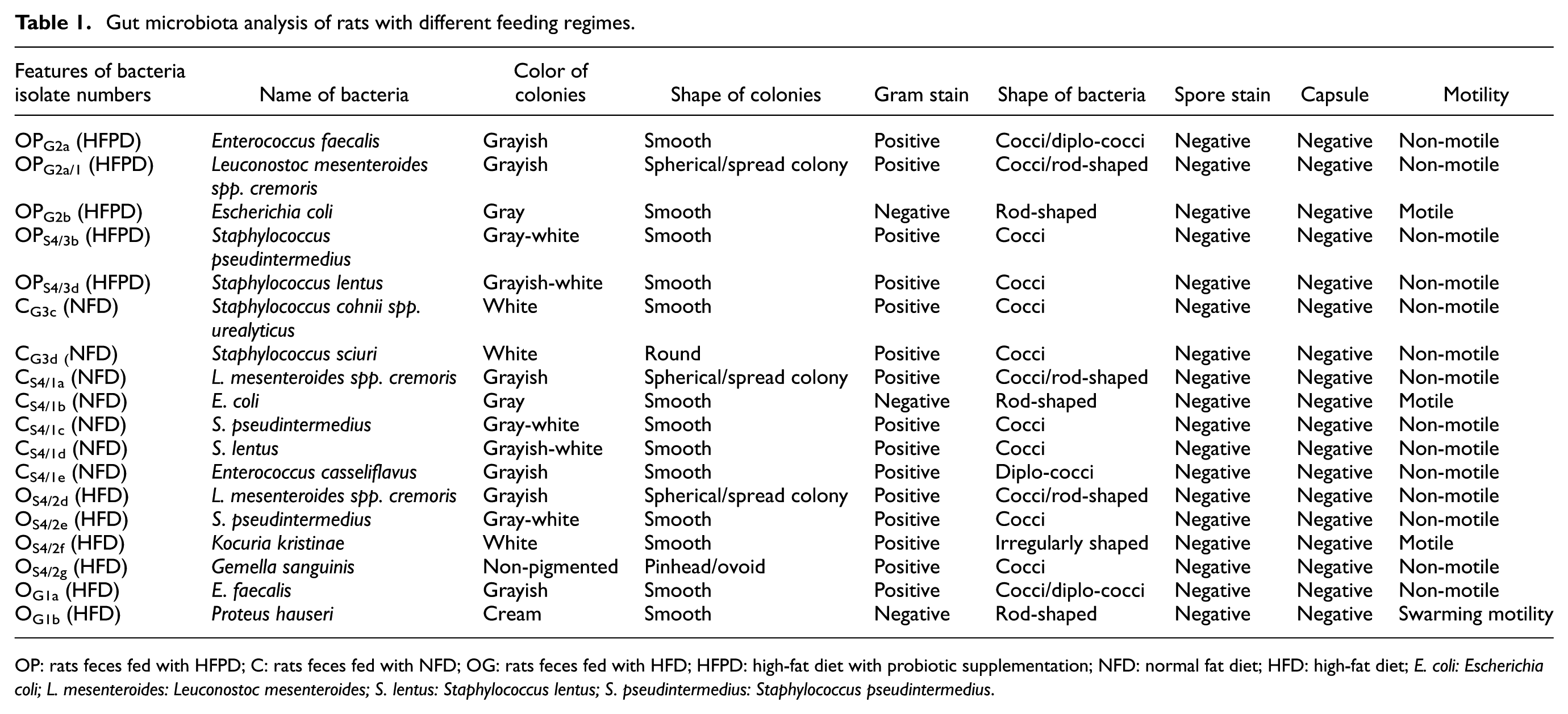
OP: rats feces fed with HFPD; C: rats feces fed with NFD; OG: rats feces fed with HFD; HFPD: high-fat diet with probiotic supplementation; NFD: normal fat diet; HFD: high-fat diet;
To determine the microbial flora, we analyzed 130 bacterial isolates, and we isolated 50 pure bacterial cultures determined by macroscopic and microscopic observations as gram staining, spore staining, and colony morphology. We tested 50 pure bacterial cultures on the VITEK® 2 (bioMérieux) system for bacterial species identification. Our VITEK® 2 results revealed that 11 of the 50 bacterial isolates were different species. It was observed that
Biochemical identification of the isolates by VITEK® 2 identification system.

Results of dietary–bacterial interaction according the groups.
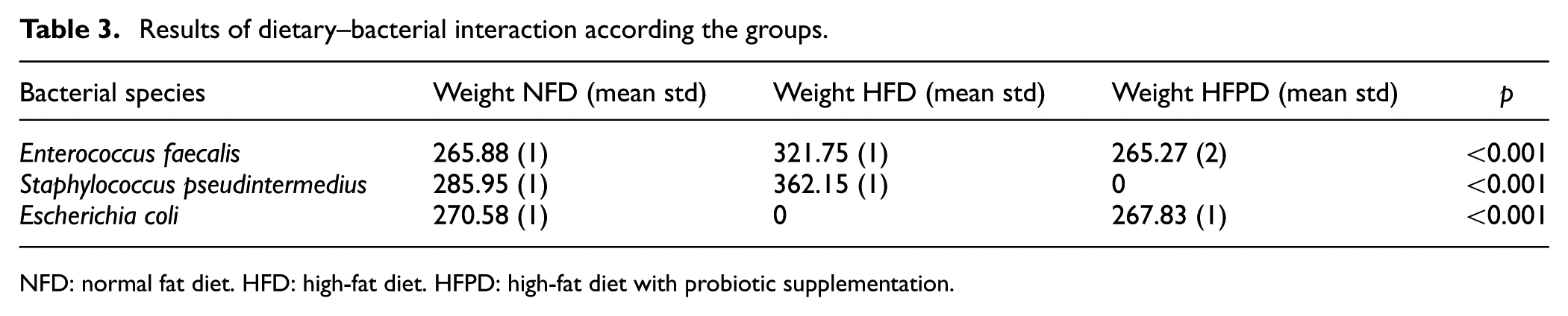
NFD: normal fat diet. HFD: high-fat diet. HFPD: high-fat diet with probiotic supplementation.
Body weight
Weight measurements were taken by animal laboratory technician throughout the study. From the beginning of the study, NFD, HFD, and HFPD groups showed a noticeable body weight gain over the 2.5-week period (time 1). In particular, the HFD group approached the obese criteria with 303.27 g. NFD and HFPD groups were very close to each other, on average 279.5 and 269 g (Figure 1(a)). A significant difference was found between the weight of HFD group compared to those of NFD and HFPD groups (
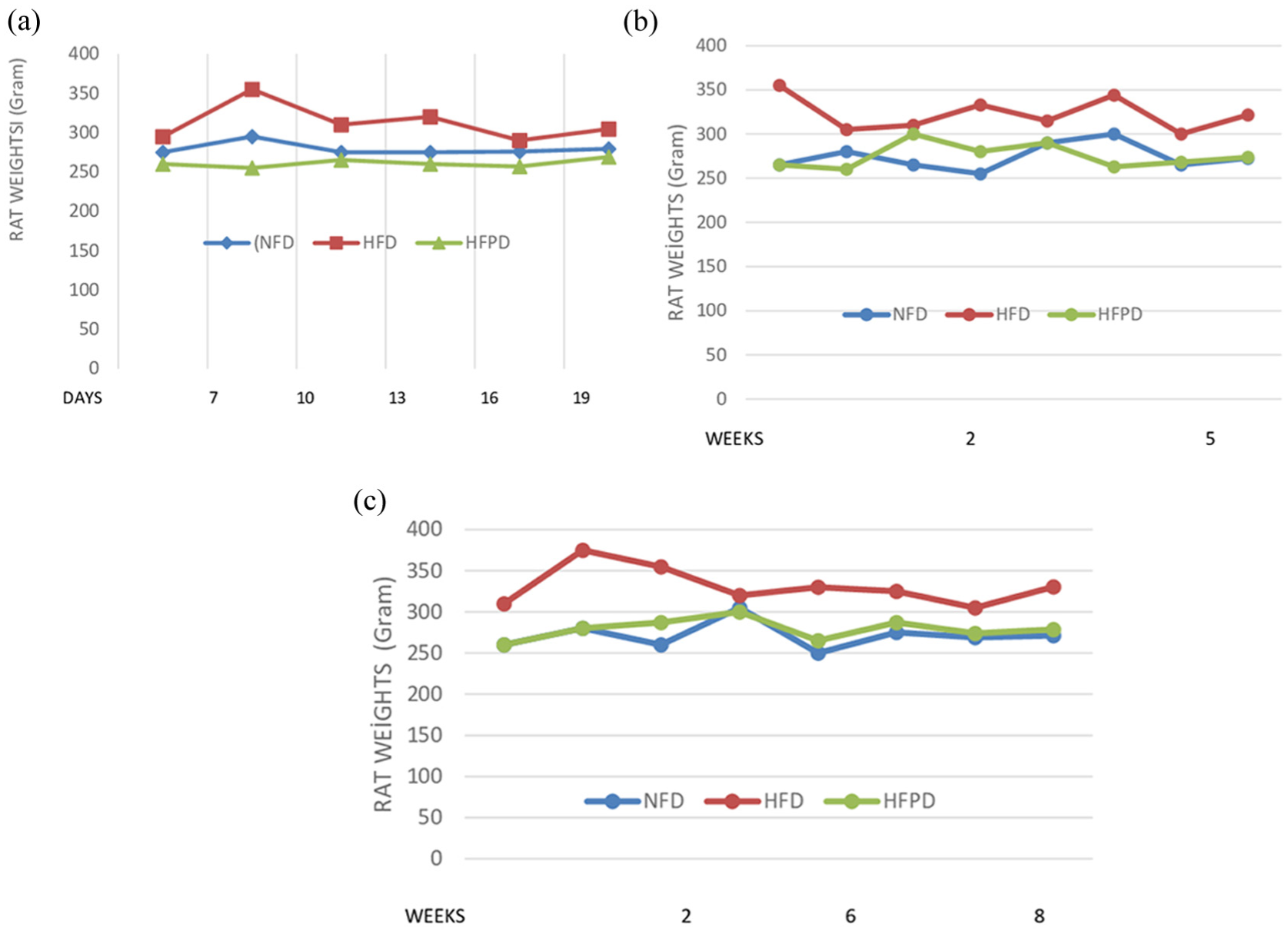
(a) The weight changes of the groups at 2.5 weeks of the study, (b) the weight changes of the groups at 5 weeks of the study, and (c) the weight changes of the groups at 8 weeks of the study.
Insulin and glucose levels
At the end of the study, the mean glucose and insulin levels were found to be 186.4 mg/dL and 4.3 IU, respectively, in the HFD group. However, these values were 129.4 mg/dL and 2.22 IU, respectively, in the NFD group and 139.1 mg/dL and 2.27 IU, respectively, in the HFPD group. HOMA–IR values were found as 0.7, 2, and 0.8 for the NFD, HFD, and HFPD groups, respectively. A significant difference was found between the groups in terms of glucose and insulin values (
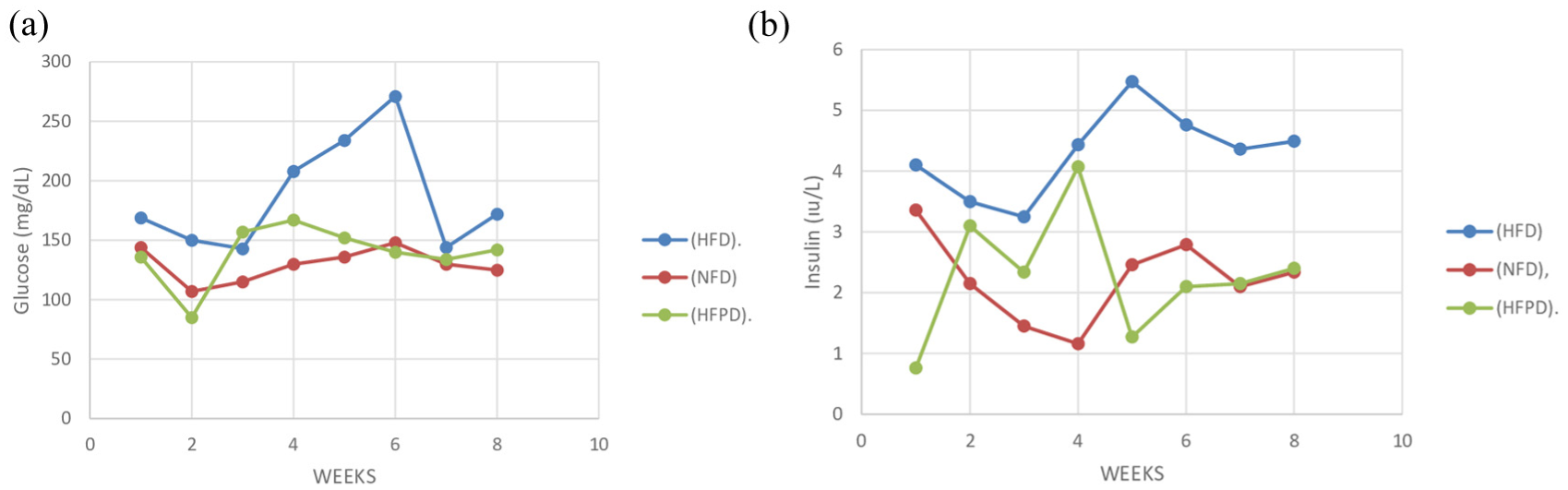
(a) Changes in glucose levels according to the groups and (b) changes in insulin levels according to the groups.
Discussion
The prevalence of obesity is increasing worldwide, primarily due to overeating and high-calorie food consumption. The relationships between the gut microbiota and the obesity are mentioned in many studies.
In our study, different nutritional regimes caused some changes in gut microbiota. Among these, it was noteworthy that
HFD significantly increases bile acid production and secretion from the gall bladder. The bile acid emulsifies luminal fat and exposes micelles to lipase-mediated digestion and thus damage the mucosal intestinal barrier and gut microbiota. 25 In addition, luminal dietary metabolites can engage with endogenous ligands, which may differentially regulate intestinal metabolic processes through various nuclear receptor signaling, thus fine-tuning intestinal immune response. 26 Diet-induced gut dysbiosis leads to an increase in mucin-degrading bacteria, weakens the intestinal firewall, allows toxic or undesired substances to penetrate the mucosal and submucosal layers and pass into the bloodstream, resulting in some mucosal immune responses. 27 Through this process, microbiota possibly modulates the gut immune response linked to both local inflammation and some metabolic diseases.
Many studies mention the association of the gut microbiota with some inflammatory diseases. The gut microbiota ferments carbohydrates, proteins, and some fats in the nutrients, thereby providing the host’s energy needs and producing some metabolites.
28
Gut microbiome produces tryptophan metabolites, lipid metabolites, and short-chain fatty acids (SCFAs). Tryptophan metabolites influence immune system cells, SCFAs induce GLP-1, and enteroendocrine cells release peptide tyrosine tyrosine. These mechanisms contribute to regulate the balance between energy intake and expenditure.
29
The intestinal epithelium barrier prevents the passage of toxins and food antigens and protects the host from harmful substances, which activate the immune system. Local and systemic inflammation developed in obesity damage the protective effects of the gut microbiota and intestinal epithelium.
30
Microbial metabolites and components of cell wall, such as lipopolysaccharide (LPS), are the main responsible agents of the host–gut microbiota interaction. The failure of the intestinal epithelial membrane and increased gut permeability lead to bacterial fragments, such as LPS, diffusing through the gut into the bloodstream. This metabolic disorder is called “metabolic endotoxemia.”31,32 LPS levels were found to be higher in people with obesity than in healthy individuals.
33
The
In this study, the probiotic
Conclusion
The studies on the identification of beneficial strains associated with obesity will enable the identification of candidate strains. These findings can identify a targetable subset of the microbiota in treating people with metabolic inflammation. To determine both preventive and therapeutic strategies, precise microbiological targets must be identified and their potential clinical effects revealed.
Footnotes
Ethical considerations
The animal experiments were conducted with the approval of the Animal Experiments Local Ethics Committee of University Ordu (February 16, 2021, no. 82678388). All methods of this study were performed in accordance with the relevant guidelines and regulations of this committee.
Author contributions
Conceptualization: Ömer Ertürk. Data curation: Özlem Özdemir. Formal analysis: Selim Görgün. Funding acquisition: Özlem Özdemir and Ömer Ertürk. Investigation: Özlem Özdemir, Ömer Ertürk, and Selim Görgün. Methodology: Ömer Ertürk and Selim Görgün. Software: Özlem Özdemir. Validation: Ömer Ertürk. Visualization: Ömer Ertürk. Writing – original draft: Özlem Özdemir. Writing – review and editing: Özlem Özdemir and Ömer Ertürk.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by Ordu University Scientific Project Coordination Department (ODUBAP, project no. D-2204, April 19, 2022).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data generated in the present study are included in this manuscript. The corresponding author can be contacted for further information on the data.
