Abstract
In this retrospective cohort study, we investigated the prognostic value of sarcopenia evaluated by Computed Tomography (CT)-based indices for adverse hospitalization outcomes in patients with acute infections. We analyzed data from 225 patients admitted to the hospital for acute infections between 2019 and 2020. Patients who had undergone an abdominal CT scan either up to 1 month before or within the first 3 days of hospitalization were included. CT image analysis was used to evaluate skeletal muscle mass (by skeletal muscle index (SMI)) and muscle quality (by psoas muscle density, pMD). Low pMD was associated with higher in-hospital mortality (31% vs 11.4% p < 0.001) as well as higher longer-term mortality rates (p = 0.008 for 30 days and <0.001 for 90- and 1-year mortality). Low pMD remained an independent poor prognostic factor after controlling for confounders, with an adjusted odds ratio (aOR) of 2.74, (95% CI 1.33–5.67, p = 0.006) for 1-year mortality, and aOR of 2.61, (95% CI 1.23–5.55) for a prolonged hospital stay. Low SMI was associated with adverse outcomes, although this association was not independent after controlling for confounders. Notably, patients with both low SMI and pMD exhibited the poorest hospitalization outcomes: aOR for 1-year mortality 5.015 (95% CI 1.767–14.23, p = 0.002), and prolonged length of stay aOR 3.197, (95% CI 1.159–8.821, p = 0.025). CT-based muscle indices serve as independent prognostic factors in medical patients admitted with acute infection. Incorporating radiological assessments of sarcopenia into routine care for hospitalized patients with acute infection may enable risk stratification and early intervention in reversible conditions.
Introduction
In the 2018 revision by the European Working Group on Sarcopenia in Older People (EWGSOP), sarcopenia was redefined into three distinct levels of dysfunction. “Probable sarcopenia” was defined as a decrease in muscle strength, “definite sarcopenia” was defined as low muscle strength and low muscle quality or quantity, and “severe sarcopenia” determined if there is an additional decline in physical performance. 1
Imaging-based body composition parameters, such as muscle area or muscle density have diverse effects on patients’ outcomes in different clinical scenarios.
Loss of muscle area, represented by low skeletal muscle index (SMI), is associated with perioperative complications, 2 decreased survival of oncological patients,3,4 and adverse outcomes in chronic infection and acute intensive care unit admissions.5,6 While some studies have found adverse effects of low SMI on hospitalization outcomes in patients with aspiration pneumonia 7 or severe sepsis, 8 the data regarding the influence of skeletal muscle indices and body composition determinants in patients hospitalized for acute infection is less robust.
Psoas muscle density (pMD) derived from muscle attenuation on Computed Tomography (CT) and measured in Hounsfield units (HU), serves as a marker for muscle quality, reflecting myosteatosis. Previous research has demonstrated associations between decreased pMD and adverse outcomes, such as increased mortality in patients undergoing dialysis, 9 or suffering from chronic lung disease, and adverse outcomes after surgery10,11 One study evaluated myosteatosis through thoracic CT scans as a predictor for increased disease severity in Coronavirus Disease 2019 (COVID-19) patients. 12
Sarcopenia is related to adverse short and long-term outcomes
CT-based calculations of muscle area and muscle density are part of Sarcopenia evaluation
The influence of skeletal muscle indices and body composition determinants on outcomes of patients hospitalized for acute infection is not well known.
We found that low muscle quality, as indicated by psoas muscle density, is an independent poor prognostic factor for hospitalization outcomes.
The integration of radiological assessment for sarcopenia into the routine care of hospitalized patients with acute infection has the potential to enhance risk stratification and facilitate early intervention in reversible conditions.
Infectious diseases are a leading cause of hospitalization and mortality among elderly patients, 13 with a rising trend in admissions in the last decades. 14 Given this context, the investigation and modification of major prognostic factors affecting hospitalization outcomes is paramount.
The integration of radiological assessment for sarcopenia into the routine care of hospitalized patients with acute infection has the potential to enhance risk stratification and facilitate early intervention in reversible conditions.
Methods
The study protocol was approved by the local institutional review board of our institution. Data were collected from existing patient records, obviating the need for participants’ informed consent.
Patients’ selection
In this retrospective cohort study, consecutive patients admitted to the hospital for the treatment of acute infection between 2019 and 2020 were recruited. Infectious syndromes encompass pneumonia, gastroenteritis, urinary tract infection, cellulitis, and sepsis according to the International Classification of Disease (ICD-9) definitions. Patients were eligible for inclusion if they had undergone an abdominal CT with intravenous contrast 1 month prior to or during the first 3 days of hospitalization. Exclusion criteria encompassed patients with no information regarding height or weight or degraded quality of CT scans.
Patients’ characteristics and variables
Demographic data included age, sex, body mass index (BMI-calculated by dividing patients’ weight in kilograms divided by height in meters squared), medical history, including comorbidities, and hospitalization characteristics such as acute decompensated heart or lung disease were retrieved from computerized medical records. Laboratory tests, conducted upon admission and including hemoglobin levels, albumin, glucose levels, and creatinine levels were documented. The need for intensive care or invasive ventilation was noted. Charlson’s comorbidity index was calculated. 15 Hospitalization outcomes comprised short and long-term mortality, length of stay (LOS, considered prolonged if exceeding 1 week), re-hospitalization within 30 days post-discharge, and functional deterioration determined by the need for rehabilitation/long-term facility care or a decrease in independence according to the discharge letter.
Computed tomography image analysis
Skeletal cross-sectional area and attenuation were quantified using axial CT image at level of the third lumbar vertebra (L3) as illustrated in Figure 1.
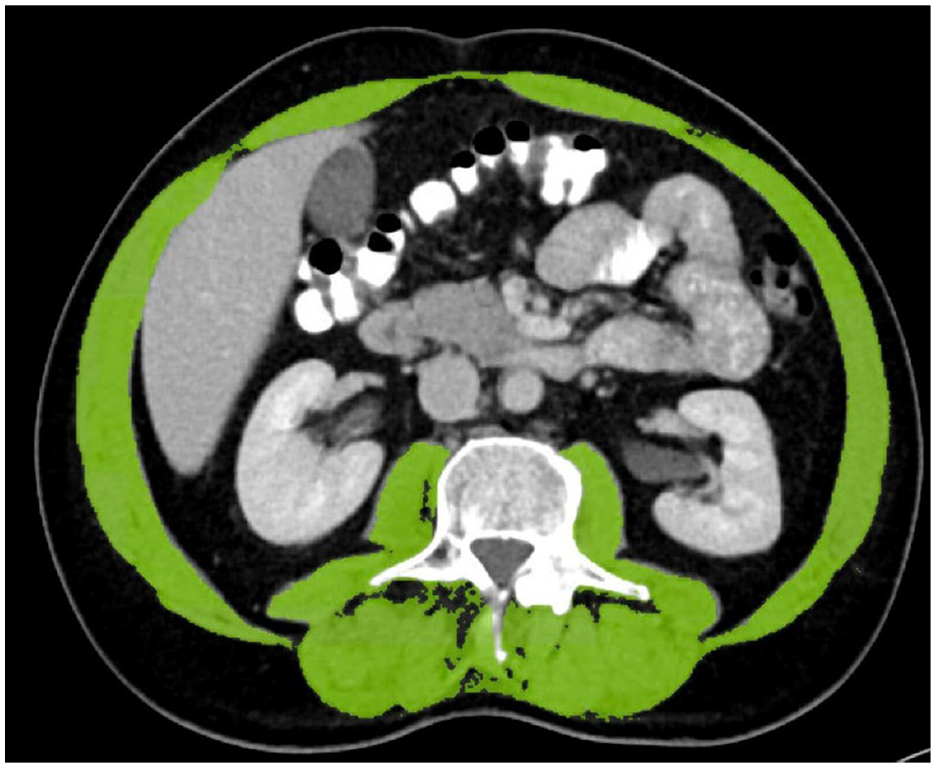
Identification of skeletal muscles (area presented in color) by software at the level of third lumbar vertebra (Singo. via Work Station, Version 8.6, Siemens, Germany).
These CT scans were part of routine clinical care and were retrospectively identified in the local picture archiving and communicating system (PACS; Sectra, Sweden). Only CT scans obtained after IV-contrast injection were analyzed. The measurements were subsequently carried out on an axial plane located at the center of the third lumbar vertebra using semiautomatic measurements by Singo.via Work Station (Version 8.6, Siemens, Germany). Skeletal muscle tissue was identified \ using HU thresholds defined as −29 to +150. Skeletal muscle area (in squared centimeters) was corrected for patients’ height (in squared meters) and multiplied on slice thickness of 3 mm accomplishing volumetric measurement. All measurements were performed by an experienced radiologist. To evaluate intra-reader reliability, measurements were repeated in 15 randomly selected CT scans by a second radiologist.
Muscle mass (evaluated by SMI) and muscle quality (evaluated by pMD) were measured by CT-derived indices. Previously published cutoff values are inconsistent. We considered SMI values below 40.9 cm2/m2 for women and 52.9 cm2/m2 for men as indicative of low muscle quantity. Myosteatosis, or low pMD, remains less well-defined in literature. For our study, we considered values below 40HU as low pMD.16,17 Sarcopenic obesity was defined as patients with BMI levels exceeding 30 and either low pMD or low SMI.
Statistics
All analyses were performed by using SPSS software (IBM SPSS statistics for Windows, Ver.25.0, IBM Corp. Armonk, NY, USA, 2017). All statistical tests were two-sided, and p < 0.05 was considered statistically significant. Categorical data are expressed as frequencies and percentages and compared among groups using Chi-square tests and Fisher’s exact test. Continuous variables were evaluated for normal distribution using histogram and Q-Q plot and expressed as the mean and standard deviation when normally distributed or median and interquartile range (IQR) when not. Continuous variables were compared using the Mann–Whitney test or independent sample t-test.
The cohort was stratified into three groups based on the combination of SMI and pMD. Patients with low SMI and pMD constituted the “low muscle mass and quality group,” those with high SMI and pMD formed the “good muscle mass and quality group,” while the remaining patients were categorized as the “mixed indices” group. Multivariable logistic regression was employed to examine the associations between body composition indices and mortality, functional deterioration, LOS, and readmission while controlling for confounders. Variables included in the regression included age, sex, and Charlson’s comorbidity index.
Results
There were 472 patients eligible for the study, 182 patients did not have BMI measurements and 65 underwent scans without contrast media or had degraded quality of CT scans. Finally, body composition parameters were calculated for 225 patients hospitalized in our institution between January 2019 and December 2020.
Muscle quantity—SMI
A comparison between 122 patients with low SMI and 103 patients with normal/high SMI revealed significant differences. Patients with lower SMI levels were notably older (78 vs 66 years p < 0.001), presented with more complex comorbidities (Charlson comorbidity index 6 (3–7) vs 3 (0–6), p < 0.001), and exhibited lower BMI levels (24 kg/m2 vs 26 kg/m2 p < 0.001). These patients also had a higher prevalence of disability (29.5% vs 14.6%, p = 0.004) and a higher frequency of glucocorticoid use prior to their admission (13.1% vs 2.9%, p = 0.006). Pneumonia was the primary reason for hospitalization among patients with lower muscle quantity (43.4% vs 28.2% p = 0.018). Upon admission, they had lower hemoglobin (p < 0.001) and Albumin (p = 0.013) levels. Tables 1 and 2 present the general and hospitalization characteristics, respectively.
General characteristics of hospitalized patients with acute infection, divided by skeletal muscle index a and psoas muscle density.
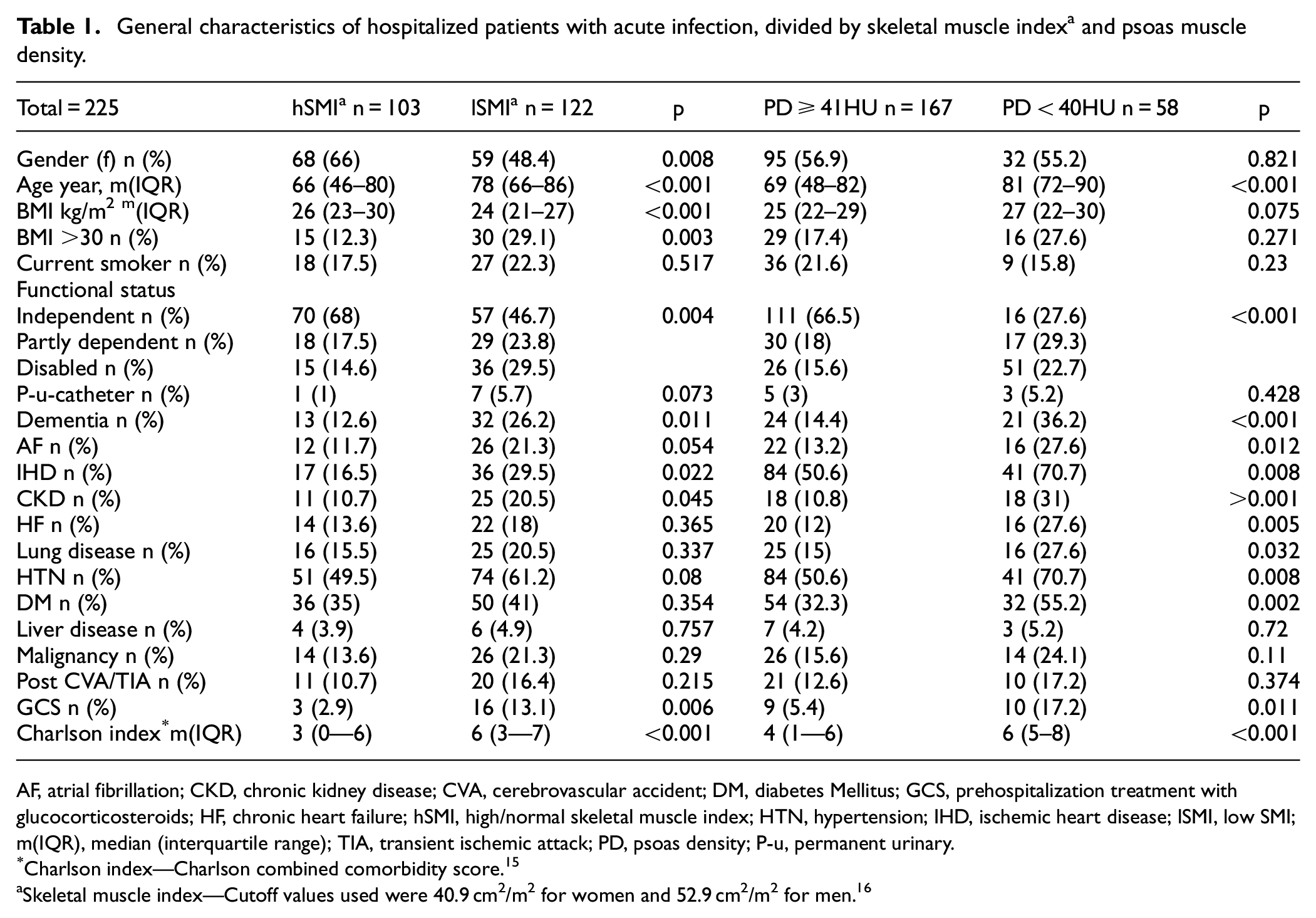
AF, atrial fibrillation; CKD, chronic kidney disease; CVA, cerebrovascular accident; DM, diabetes Mellitus; GCS, prehospitalization treatment with glucocorticosteroids; HF, chronic heart failure; hSMI, high/normal skeletal muscle index; HTN, hypertension; IHD, ischemic heart disease; lSMI, low SMI; m(IQR), median (interquartile range); TIA, transient ischemic attack; PD, psoas density; P-u, permanent urinary.
Charlson index—Charlson combined comorbidity score. 15
Skeletal muscle index—Cutoff values used were 40.9 cm2/m2 for women and 52.9 cm2/m2 for men. 16
Hospitalization characteristics of patients admitted with acute infection, divided by skeletal muscle index a and psoas muscle density.
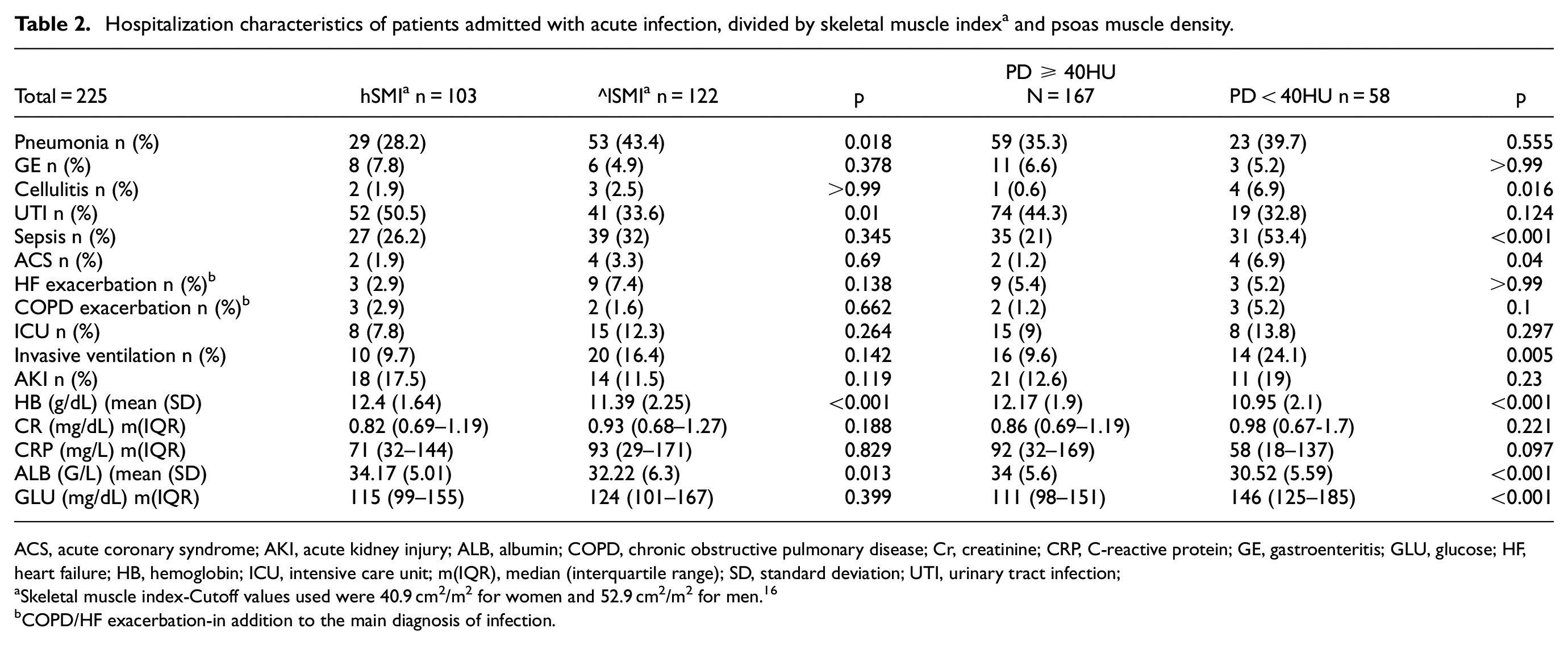
ACS, acute coronary syndrome; AKI, acute kidney injury; ALB, albumin; COPD, chronic obstructive pulmonary disease; Cr, creatinine; CRP, C-reactive protein; GE, gastroenteritis; GLU, glucose; HF, heart failure; HB, hemoglobin; ICU, intensive care unit; m(IQR), median (interquartile range); SD, standard deviation; UTI, urinary tract infection;
Skeletal muscle index-Cutoff values used were 40.9 cm2/m2 for women and 52.9 cm2/m2 for men. 16
COPD/HF exacerbation-in addition to the main diagnosis of infection.
Mortality rates were notably higher among patients with low SMI, both for 90 days (28.7% vs 15.5%, p = 0.019), and 1 year (41.8 vs 18.4% p < 0.001). Among those who survived the index hospitalization, low muscle quantity was also associated with other adverse hospitalization outcomes including functional deterioration (35% vs 16.8% p = 0.002), readmission (22.1% vs 11.2% p = 0.049), and prolonged hospital stay (LOS >7days- 59% vs 37.9%, p = 0.002). However, SMI was not independently associated with adverse hospitalization outcomes after controlling for confounders (Table 3).
Hospitalization outcomes of patients admitted with acute infection according to skeletal muscle index a and psoas density.
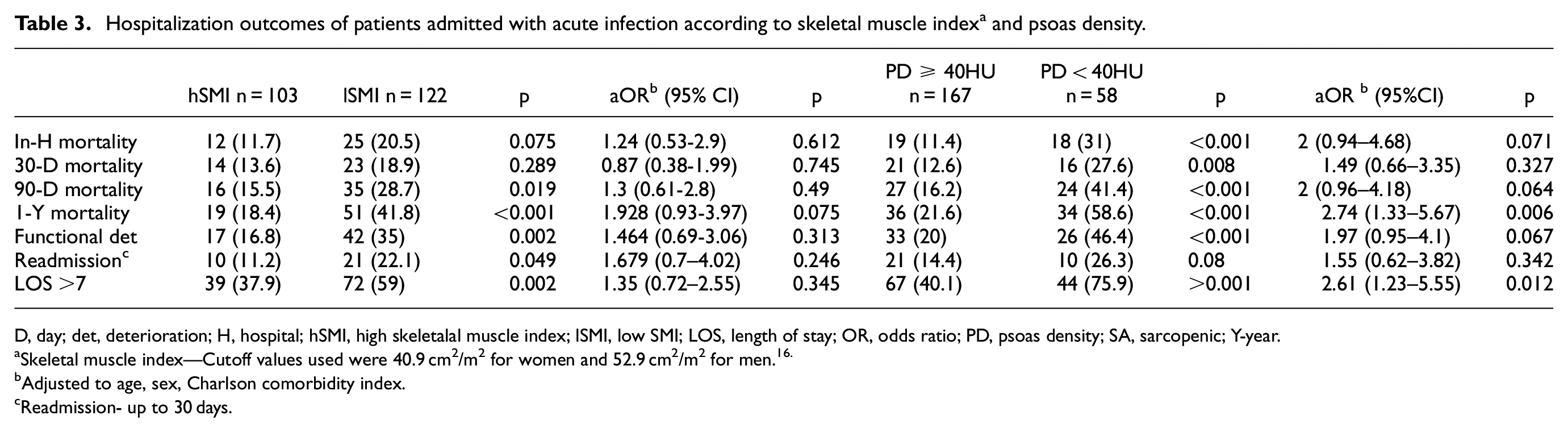
D, day; det, deterioration; H, hospital; hSMI, high skeletalal muscle index; lSMI, low SMI; LOS, length of stay; OR, odds ratio; PD, psoas density; SA, sarcopenic; Y-year.
Skeletal muscle index—Cutoff values used were 40.9 cm2/m2 for women and 52.9 cm2/m2 for men.16.
Adjusted to age, sex, Charlson comorbidity index.
Readmission- up to 30 days.
Muscle quality—psoas density
Comparison between 58 patients with low and 167 patients with normal pMD revealed significant differences. Patients with low pMD were notably older, with a mean age of 81 years compared to 69 years in the normal pMD group (p < 0.001), were more frequently disabled (22.7% vs 15.6%, p < 0.001), and had higher comorbidity index scores (6 (5–8) vs 4 (1–6) p < 0.001; Table 1). Hospitalization was more complex for patients with lower muscle quality, as evidenced by higher rates of sepsis (p < 0.001), acute coronary syndrome (p = 0.04), and invasive ventilation (p = 0.005). Patients with lower pMD presented with lower hemoglobin levels (p < 0.001), albumin levels (p < 0.001), and higher glucose levels (p < 0.001) upon admission (Table 2). Low muscle quality was associated with adverse hospitalization outcomes, including in-hospital mortality (31% vs 11.4% p < 0.001). Longer-term mortality rates were also higher in patients with low muscle quality (p = 0.008 for 30 days and <0.001 for 90- and 1-year mortality). Low pMD remained an independent poor prognostic factor after controlling for confounders, with an adjusted odds ratio (aOR) of 2.74, 95% CI 1.33–5.67, p = 0.006 for 1-year mortality, and aOR of 2.61, 95% CI 1.23–5.55 for prolonged hospital stay (LOS > 1 week). Hospitalization outcomes are presented in Table 3.
Muscle quality and quantity
The cohort was divided into three groups based on the combination of muscle mass and quality indices: patients with low muscle mass and quality indices (39 patients), mixed muscle mass and quality indices (102 patients), and good muscle mass and quality indices (84 patients). In-hospital mortality rates were 9.5% in the good muscle mass and quality group, 14.7% in the mixed group, and 35.9% among the patients with low muscle mass and quality group (p < 0.001). Similar trends were observed for 30, 90, and 1-year mortality rates (p = 0.051, <0.001, <0.001, respectively). Prolonged LOS was significantly more common among patients with low muscle mass and quality (p < 0.001), and they also had higher rates of readmission (p = 0.034) and functional deterioration (p < 0.001). Low muscle mass and quality were independently associated with a higher risk of 1-year mortality (aOR 5.015, 95% CI 1.767–14.23, p = 0.002), and prolonged LOS (aOR 3.197, 95% CI 1.159–8.821, p = 0.025), with a trend toward functional deterioration (aOR 2.735, 95% CI 0.994–7.53, p = 0.051; Figure 2).
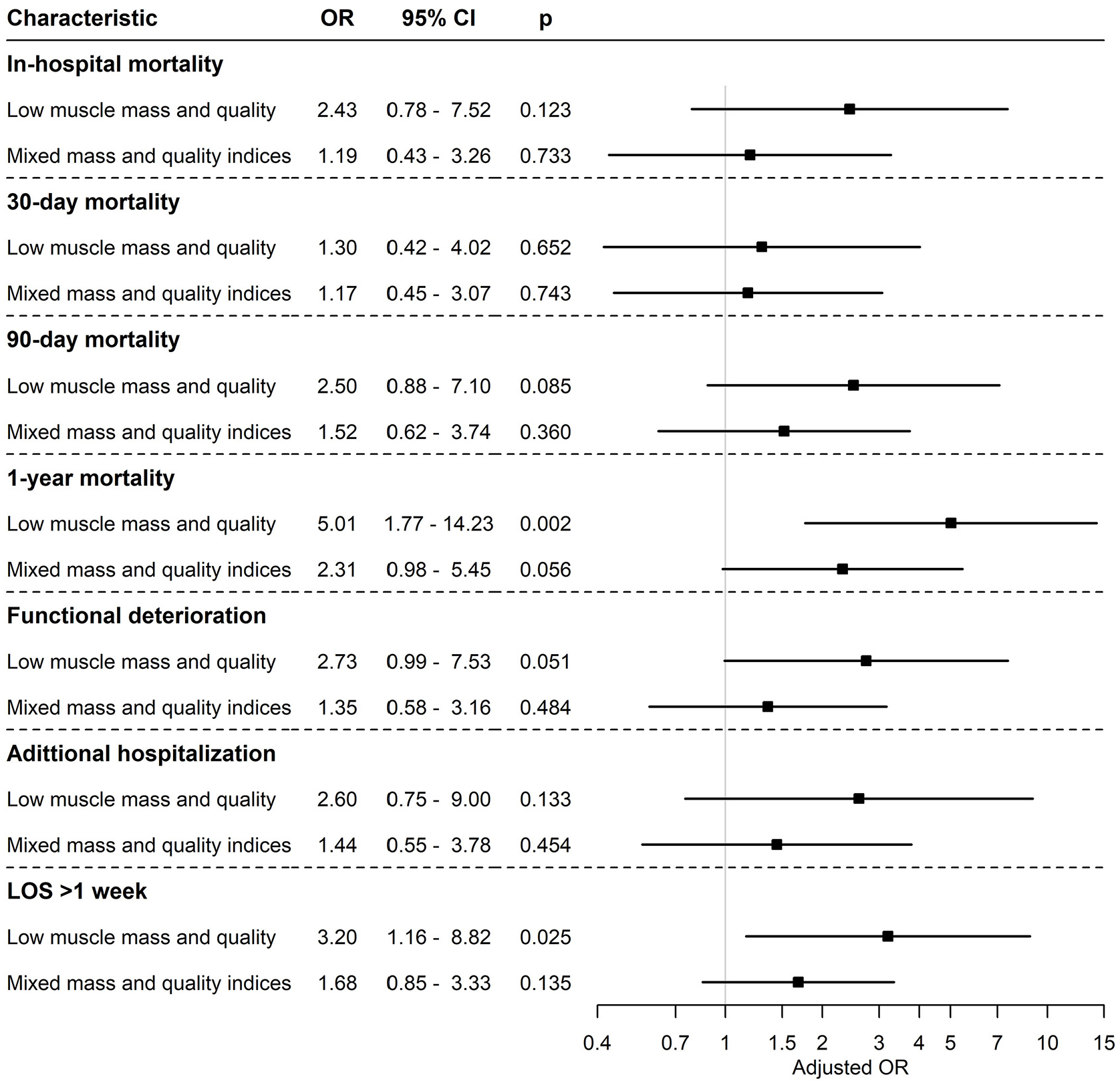
Primary outcomes of patients according to muscle mass indices: The odd ratios and confidence intervals measured by the logistic regression model are shown, with adjustment for age, sex, and Charlson’s comorbidity index.
Sarcopenic obesity as defined by either pMD or SMI, showed no significant differences in outcomes compared to non-sarcopenic obesity, although the small sample size may have impacted our results (data not shown).
Discussion
Sarcopenia, characterized by muscle loss, is associated with various health concerns, including falls and fractures, cardiac and respiratory diseases, lower quality of life, loss of independence, and mobility disorders. 1 Sarcopenia prevalence varies, ranging from 10% to 16% of the elderly worldwide and 15%–30% among the long-term care population. 18
Hospitalization rates due to infectious diseases have been on the rise, particularly among the elderly. 19 Numerous poor prognostic factors are known to affect hospitalization outcomes among patients with acute infection among medical patients, such as comorbidities, lactate levels, and platelet count. 20
In our study, we enrolled 225 medical patients hospitalized for acute infection, aiming to assess CT-based body composition indices as predictors of adverse hospitalization outcomes. We found that low muscle quality, or myosteatosis, as indicated by pMD, is an independent poor prognostic factor for hospitalization outcomes including mortality and prolonged LOS. While low muscle quantity, indicated by SMI, was associated with adverse hospitalization outcomes, this association lost statistical significance after controlling for confounders. SMI may serve as a surrogate marker for adverse hospitalization outcomes. Notably, the patients with low muscle mass and quality exhibited the poorest hospitalization outcomes, including prolonged LOS, functional deterioration, and 1-year mortality.
Previous research regarding pMD and SMI has predominantly focused on oncologic or surgical patient populations, 21 with limited attention to medical patients such as those with COVID-19 12 or sepsis. 8 The role of sarcopenia in patients with acute infection has remained underexplored. Given the significance of host response managing infections, a muscle-associated anti-inflammatory effect mediated by the release of muscle-derived peptides (myokines) in both local and peripheral hormone-like fashion has been suggested. 22 Our findings demonstrate that poor muscle indices have both in-hospital (prolonged LOS and functional deterioration) and long-term (mortality) adverse effects on patients’ outcomes. This effect may be particularly pronounced in patients with low muscle quality indices. Based on the proposed myokines theory, fatty muscle infiltration can hinder proper myokine release, resulting in reduced anti-inflammatory reaction and diminished host response to infection.
Grip strength and chair stand tests are commonly employed to measure skeletal muscle strength while physical performance is assessed through gait speed. However, these tests are not always practical among elderly hospitalized patients, especially during the initial days of hospitalization. Identifying sarcopenic patients in the context of acute hospitalization provides an opportunity for intervention, which is particularly crucial as elderly hospitalized patients face the risk of developing acute, admission-related sarcopenia. 23
Accumulated clinical knowledge regarding the radiologic assessment of sarcopenia has made imaging modalities effective tools for the evaluation of sarcopenia.24,25 Skeletal muscle CT attenuation as a measure of muscle fatty infiltration evaluation is easy to obtain from CT scans and offers reproducibility. However, the absence of widely accepted cut-offs and definitions for CT-based indices of sarcopenia hampers generalization of our findings. Consequently, further research is encouraged to establish universally accepted thresholds for imaging-based indices.
Treatment of sarcopenia relies primarily on a combined intervention of nutrition and exercise that may improve strength and walking speed in the long term (beyond 3 months). 26 Early mobilization (within 24 h of admission) interventions have been shown to improve outcomes in a variety of patient populations, including elderly adults with pneumonia. Barriers to early mobilization exist for both the patients (pain, fear of falling, weakness) and the medical staff (being understaffed, lack of time, or fear of self-injury). 27 Strategies to overcome these barriers are currently under development. Early lifestyle interventions such as physical and nutritional recommendations during hospitalization may mitigate the progression of acute sarcopenia and improve the long-term outcomes of patients with chronic sarcopenia. 28
Study limitations
Our retrospective observational single-center study has inherent limitations. The selection of patients undergoing CT scans at or close to hospitalization might introduce recruitment bias. Although, CT scans are commonly used in the emergency room and at the onset of hospitalization. 29
Variability in CT scan techniques may affect SMI and pMD measurements, although we minimized imprecisions by analyzing CT scans with the same technical parameters and only those obtained after IV-contrast injections. 30
Psoas density as a single muscle measurement may not fully predict whole-body muscle quality, as a local decrease in muscle quality can appear in patients with spinal disease leading to selective psoas atrophy. Finally, the lack of accepted cut-offs and definitions for CT-based indices for sarcopenia complicates the generalization of our findings.
Conclusions
Our study suggests that the combination of low muscle mass and muscle quality as evaluated through CT scans, independently predict poor prognostic outcomes for medical patients hospitalized with acute infection.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
