Abstract
Patients with heart failure with reduced ejection fraction (HFrEF) are at risk for chronic kidney disease (CKD). Elevated levels of circulating biomarkers soluble urokinase plasminogen activator receptor (suPAR), galectin-3, soluble suppression of tumorigenicity 2 (ST2), and N-terminal prohormone B-type natriuretic peptide (NT-proBNP) are associated with CKD progression and mortality. The predictive value of these biomarkers in a population with HFrEF and kidney disease is relatively unknown. We sought to determine whether these biomarkers were associated with longitudinal trajectory of estimated glomerular filtration rate (eGFR) in HFrEF and assess their association with mortality using a joint model to account for competing risks of ventricular assist device (VAD) implantation and heart transplantation. We included participants from the Registry Evaluation of Vital Information for Ventricular Assist Devices in Ambulatory Life with repeated eGFR measures over 2 years. Of 309 participants, mean age was 59 years, median eGFR 60 ml/min/1.73 m2, 45 participants died, 33 received VAD, and 25 received orthotopic heart transplantation. Higher baseline serum standardized suPAR (β coefficient = −0.36 √(ml/min/1.73 m2), 95% confidence interval (−0.48 to −0.24), p < 0.001), standardized galectin-3 (−0.14 √(ml/min/1.73 m2) (−0.27 to −0.02), p = 0.02), and log NT-proBNP (−0.23 √(ml/min/1.73 m2) (−0.31 to −0.15), p < 0.001) were associated with eGFR decline. ST2 and log NT-proBNP were associated with mortality. Higher baseline suPAR, galectin-3, and NT-proBNP are associated with eGFR decline in patients with HFrEF. Only ST2 and NT-proBNP are associated with greater mortality after controlling for other factors including change in eGFR. These biomarkers may provide prognostic value for kidney disease progression in HFrEF and inform candidacy for advanced heart failure therapies.
What is already known on this topic?
Kidney disease is a common comorbid condition in heart failure and is associated with adverse outcomes.
What this study adds?
Baseline soluble urokinase plasminogen activator receptor (suPAR) and galectin-3 were associated with a decline in estimated glomerular filtration rate (eGFR) over 2 years after controlling for competing events of death, ventricular assist device implantation, and heart transplantation, while soluble suppression of tumorigenicity 2 (ST2) was not in an advanced heart failure with reduced ejection fraction (HFrEF) population.
After controlling for longitudinal change in eGFR, only baseline ST2 and N-terminal prohormone B-type natriuretic peptide (NT-proBNP) were associated with mortality.
Baseline eGFR was no longer associated with mortality after controlling for NT-proBNP.
How this study might affect research, practice, or policy?
Use of these biomarkers in clinical practice could provide prognostic utility with regards to changes in kidney function and mortality and help candidacy for advanced heart failure therapies.
Introduction
Kidney disease is a common comorbid condition among patients with heart failure. Chronic kidney disease (CKD) may impact approximately 42% of individuals with heart failure, and acute kidney injury is a common occurrence among patients admitted for acute decompensated heart failure.1,2 Kidney disease is associated with adverse clinical outcomes such as a greater risk for mortality and heart failure rehospitalization in patients with heart failure. 3 It is because of this association with adverse outcomes that kidney disease impacts treatment decisions, particularly among patients with advanced heart failure with reduced ejection fraction (HFrEF). For example, patients with advanced kidney disease undergoing heart transplant evaluation may or may not need simultaneous kidney transplantation depending on whether their kidney disease is felt to be reversible. 4 Similarly, patients with advanced kidney disease felt to be irreversible may not be candidates for advanced heart therapies such as durable left ventricular assist devices (LVADs). 5
However, determining decline of kidney function in this clinical setting is not straightforward. There is an unmet need to establish biomarkers that are predictive of future kidney disease progression and decline in estimated glomerular filtration rate (eGFR) to guide clinical decision-making. Soluble urokinase plasminogen activator receptor (suPAR) and galectin-3 are associated with CKD progression in patients without heart failure, but their association with CKD progression among patients with advanced HFrEF is unknown.6,7 Soluble suppression of tumorigenicity 2 (ST2) may also be associated with CKD progression in general CKD cohorts, though observations from prior studies have been conflicting.7,8 Higher suPAR, galectin-3, ST2, and N-terminal prohormone B-type natriuretic peptide (NT-proBNP) have all been associated with a greater risk for mortality in heart failure, but whether levels are influenced by decreased clearance of these biomarkers from the blood due to a lower glomerular filtration rate (GFR) in the setting of kidney disease has also not been established in prior studies.9–11
The primary aim of this study was to determine whether baseline circulating levels of suPAR, galectin-3, and ST2 are associated with a decline in eGFR over time in patients with advanced HFrEF from the Registry Evaluation of Vital Information for Ventricular Assist Devices in Ambulatory Life (REVIVAL). The secondary aim of this study was to evaluate whether these three biomarkers and NT-proBNP were associated with mortality after controlling for longitudinal change in eGFR. We hypothesized that higher baseline levels of these biomarkers would each be independently associated with decline in eGFR over time, using the 2021 CKD Epidemiology Collaboration (CKD-EPI) equation, and a greater risk of mortality during follow-up, even after controlling for eGFR.
Methods
Participants and setting
For this study, we used de-identified data from REVIVAL obtained from the National Heart, Lung, and Blood Institute’s Biologic Specimen and Data Repository Information Coordinating Center. Details about the study design and patient population have been previously published. 12 Briefly, 400 participants with advanced chronic systolic heart failure from 21 different centers in the United States were enrolled in this prospective observational cohort study between July 2015 and June 2016. Enrolled participants were followed for 2 years or until occurrence of a study endpoint with the primary aim to better understand clinical trajectory of these patients and who may benefit from ventricular assist device (VAD) therapy. All enrolled participants provided written informed consent. The REVIVAL protocol was approved by institutional review boards overseeing the study’s data coordinating center and at all participating institutions.
Study visits occurred at enrollment (Baseline A) and at the following time points post-enrollment: 2 (Baseline B), 6, 12, 18, and 24 months. Events including hospitalizations, implantation of temporary or durable mechanical circulatory support, heart transplantation, and death were captured at each visit. Demographics, medical history, New York Heart Association (NYHA) class, Interagency Registry for Mechanically Assisted Circulatory Support profile, VAD treatment preference, and routine laboratory measures were collected at each visit.
Laboratory variables
Biomarkers, including suPAR, galectin-3, ST2, and NT-proBNP were collected at the Baseline B visit. The present study included all patients who had serum suPAR, galectin-3, ST2, and NT-proBNP measured at Baseline B visit and repeated eGFR measurements. Ten milliliters of blood were collected for serum biomarker analysis and sent to the Biomarker Core Lab as described in the REVIVAL Manual of Procedures. suPAR, galectin-3, and ST2 were measured using an enzyme-linked immunosorbent assay.
Outcome measures
The prespecified primary outcome was longitudinal change in eGFR over time of follow-up. eGFR was calculated from serum creatinine using the 2021 CKD-EPI equation. 13 Participants were censored at the time of the competing event of death, heart transplantation, or durable VAD implantation, or were censored at the end of the follow-up period—whichever came first. The secondary outcome was all-cause mortality.
Statistical analysis
Patients were stratified into groups characterized by whether they were alive or reached an outcome during follow-up (heart transplantation, durable VAD, or death). Categorical data were described as proportions, and continuous data were presented as means ± standard deviation (SD) or median (25th, 75th percentile). To compare categorical variables between patients with different events, Fisher’s exact test was used if expected N < 5; otherwise, Chi-square test was used. For continuous variables, Welch’s one-way analysis of variance was applied to normally distributed data, and the Kruskal–Wallis test was applied to not normally distributed data.
Alive patients were censored at 750 days. The cumulative incidence of death was estimated by the Kaplan–Meier product-limit method and compared between patients with different levels of eGFR (<45, 45–59, and ≥60 ml/min/1.73 m2). Statistical comparisons of incident death across tertiles of biomarkers were determined using the log-rank test. Unadjusted and adjusted Cox proportional hazards regression models were constructed to evaluate associations of baseline square root transformed eGFR and biomarker (suPAR, galectin-3, ST2, and log transformed NT-proBNP) levels with mortality. The proportional hazards assumption was assessed using Schoenfeld residuals. Adjusted models controlled for age, sex, race, history of diabetes mellitus, NYHA Functional Classification (Class I and II vs Class III and IV), and NT-proBNP.
For analysis of joint models, biomarkers were standardized to be centered around 0 with SD of 1. Joint models with competing risks were built with a linear mixed effects submodel to assess the association between baseline biomarkers and changes in eGFR (square root transformed) over time as the longitudinal process and a Cox proportional hazards submodel for death, VAD implantation, and heart transplant to account for censoring. In the longitudinal submodel, a linear mixed effects model with random intercept and random slope was implemented. Multivariable analysis controlled for age, sex, race, diabetes, NYHA Functional Classification, and log NT-proBNP. The Cox proportional hazards submodel used B-splines to approximate the log baseline hazard and the adaptive Gauss–Hermite integration rule. The effect of covariates was shown using unadjusted and adjusted β coefficients with 95% confidence intervals (CIs) for linear regression analysis and adjusted hazard ratio (HR) with 95% CIs for survival analysis.
All analyses were performed using R software, version 4.0. 14 The JM package was used for joint model analysis. Two-sided p < 0.05 was considered statistically significant.
Results
Baseline characteristics
Of the 400 patients enrolled in REVIVAL, 309 had baseline serum biomarker and repeated eGFR measurements and were included for analysis. Baseline characteristics stratified for patients who were alive (and did not have a competing event), died, or received a heart transplant or VAD by the end of the observation period are summarized in Table 1. Those who died were older as compared with those who had no competing event (heart transplantation or VAD implantation). Among all groups, the study population was predominantly male and predominantly white. Those who received heart transplantation or VAD had a higher proportion with NYHA class III and IV symptoms vs NYHA class I and II symptoms as compared with those who died or had no events. Baseline eGFR was lowest among those who died and highest among those who received heart transplantation.
Baseline characteristics by competing event.
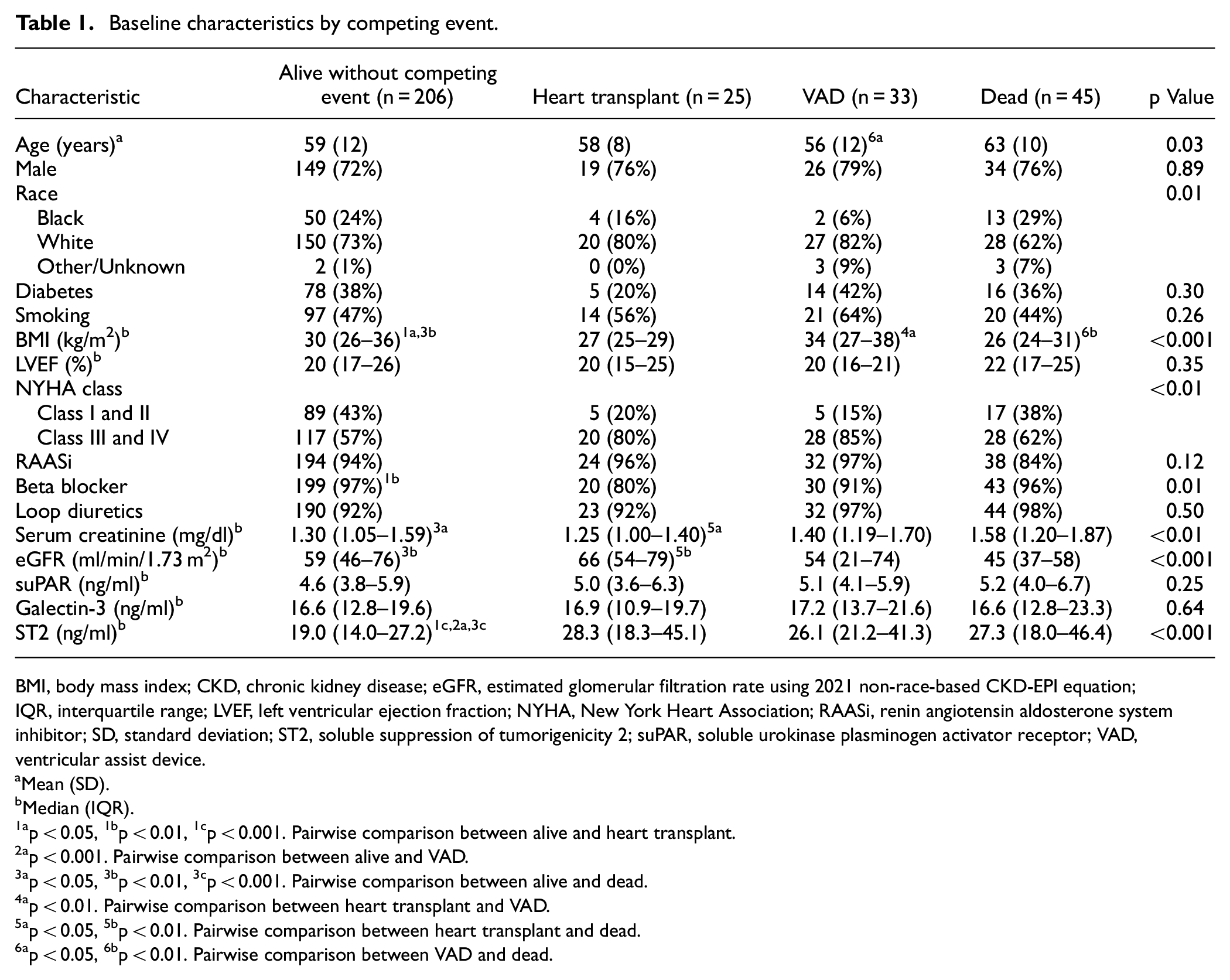
BMI, body mass index; CKD, chronic kidney disease; eGFR, estimated glomerular filtration rate using 2021 non-race-based CKD-EPI equation; IQR, interquartile range; LVEF, left ventricular ejection fraction; NYHA, New York Heart Association; RAASi, renin angiotensin aldosterone system inhibitor; SD, standard deviation; ST2, soluble suppression of tumorigenicity 2; suPAR, soluble urokinase plasminogen activator receptor; VAD, ventricular assist device.
Mean (SD).
Median (IQR).
p < 0.05, 1bp < 0.01, 1cp < 0.001. Pairwise comparison between alive and heart transplant.
p < 0.001. Pairwise comparison between alive and VAD.
p < 0.05, 3bp < 0.01, 3cp < 0.001. Pairwise comparison between alive and dead.
p < 0.01. Pairwise comparison between heart transplant and VAD.
p < 0.05, 5bp < 0.01. Pairwise comparison between heart transplant and dead.
p < 0.05, 6bp < 0.01. Pairwise comparison between VAD and dead.
Biomarker levels at baseline grouped by competing event can be seen in Figure 1. Baseline suPAR and galectin-3 levels were no different among those who were alive, died, received VAD, or received heart transplantation (Figure 1(a) and (b)). ST2 was lower among those who were alive and did not reach a competing event as compared to those who died (p < 0.001), received a VAD (p < 0.001), or received a heart transplant (p = 0.02) (Figure 1(c)). Similarly, NT-proBNP was lower among those who were alive and did not reach a competing event as compared to those who died (p < 0.001), received a VAD (p < 0.001), or received a heart transplant (p = 0.007) (Figure 1(d)).
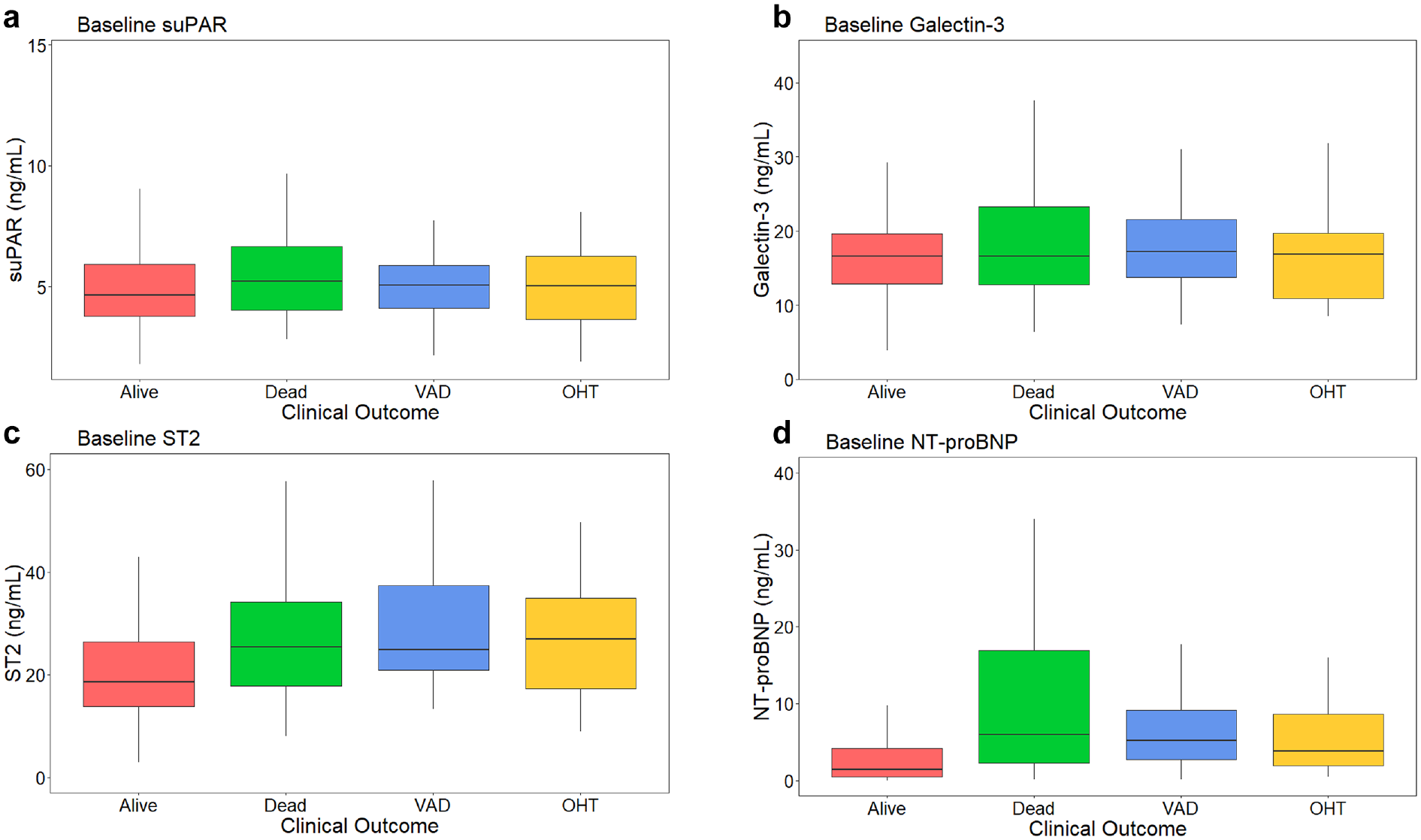
Baseline biomarker levels grouped by clinical outcome. Baseline serum levels of (a) suPAR, (b) Galectin-3, (c) ST2, and (d) NT-proBNP grouped by clinical outcome. Pairwise comparisons between groups were performed and significant values are as follows: for ST2 Alive vs Dead p < 0.001, Alive vs VAD p < 0.001, Alive vs OHT p = 0.02; for NT-proBNP Alive vs Dead p < 0.001, Alive vs VAD p < 0.001, Alive vs OHT p = 0.007. p Values were adjusted using the Holm’s adjustment method. Alive represents patients who were alive at the end of the observation period and did not receive a VAD or undergo heart transplantation.
Baseline biomarker levels stratified by baseline eGFR are shown in Figure 2. Patients with a baseline eGFR of 30–59 ml/min/1.73 m2 and those with baseline eGFR < 30 ml/min/1.73 m2 had significantly higher levels of suPAR at baseline as compared to patients with a baseline eGFR > 60 ml/min/1.73 m2 (both p < 0.001) (Figure 2(a)). Patients with baseline eGFR of 35–59 ml/min/1.73 m2 also had higher baseline levels of galectin-3 (p = 0.01) (Figure 2(b)) and ST2 (p < 0.001) as compared to those with baseline eGFR > 60 ml/min/1.73 m2 (Figure 2(c)). NT-proBNP levels were higher in those with a baseline eGFR of 30–59 ml/min/1.73 m2 (p < 0.001) and those with baseline eGFR < 30 ml/min/1.73 m2 (p = 0.005) compared to those with a baseline eGFR > 60 ml/min/1.73 m2 (Figure 2(d)).
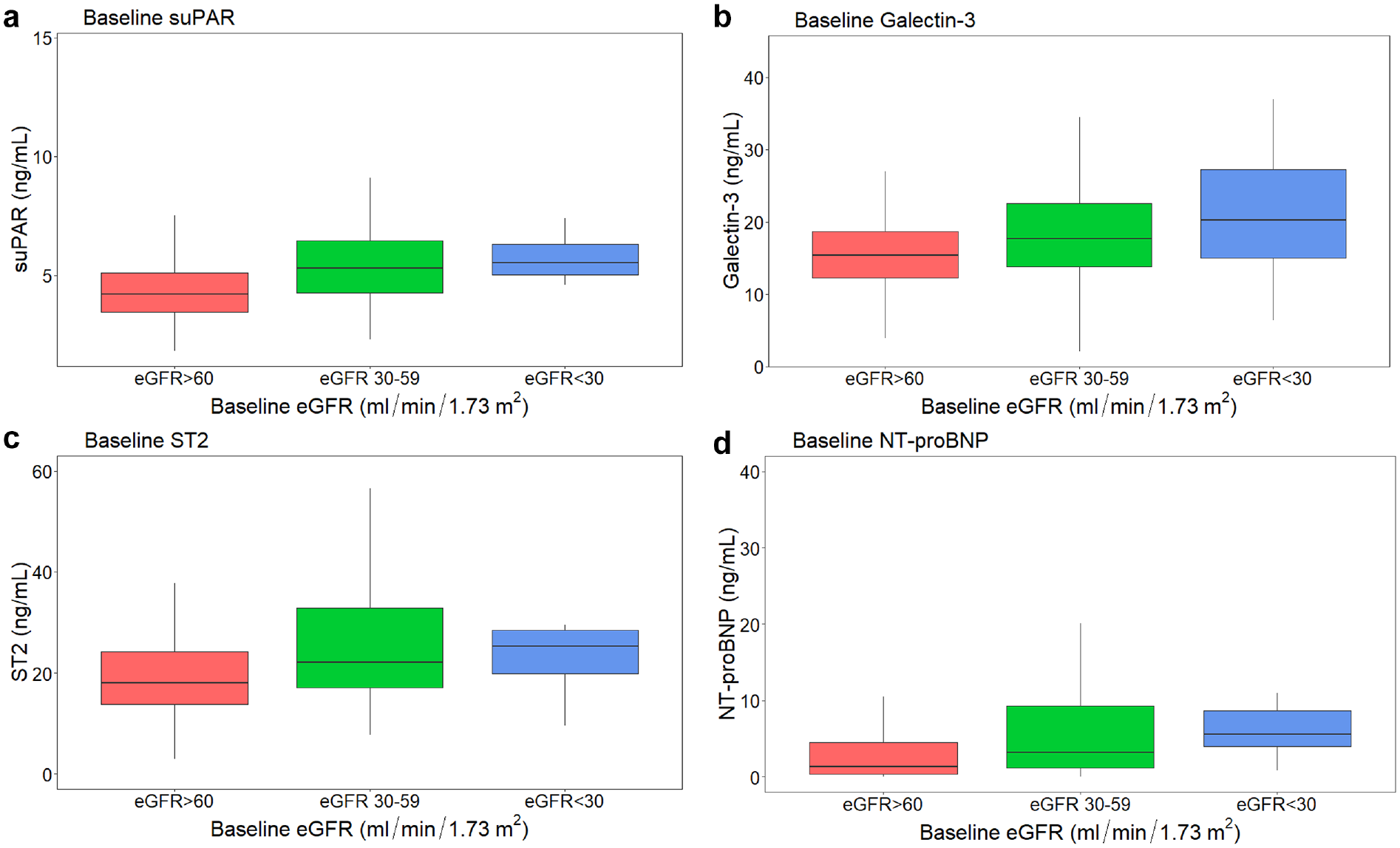
Baseline biomarker levels grouped by baseline eGFR. Baseline serum levels of (a) suPAR, (b) Galectin-3, (c) ST2, and (d) NT-proBNP grouped by baseline eGFR > 60 ml/min/1.73 m2 (n = 143), eGFR 30–59 ml/min/1.73 m2 (n = 155), and eGFR < 30 ml/min/1.73 m2 (n = 11). Pairwise comparisons between groups were performed and significant values are as follows: for suPAR eGFR > 60 ml/min/1.73 m2 vs eGFR 30–59 ml/min/1.73 m2 p < 0.001, eGFR > 60 ml/min/1.73 m2 vs eGFR < 30 ml/min/1.73 m2 p < 0.001; for Galectin-3 eGFR > 60 ml/min/1.73 m2 vs eGFR 30–59 ml/min/1.73 m2 p = 0.01; for ST2 eGFR > 60 ml/min/1.73 m2 vs eGFR 30–59 ml/min/1.73 m2 p < 0.001; for NT-proBNP eGFR > 60 ml/min/1.73 m2 vs eGFR 30–59 ml/min/1.73 m2 p < 0.001, eGFR > 60 ml/min/1.73 m2 vs eGFR < 30 ml/min/1.73 m2 p = 0.005. p Values were adjusted using the Holm’s adjustment method.
Outcome analyses
Kaplan–Meier survival analysis revealed that patients with the lowest ST2 level by tertile had greater survival as compared with those who had the highest ST2 level by tertile (log-rank p = 0.001) (Figure 3(c)). Similarly, patients with the lowest NT-proBNP level by tertile had greater survival as compared with those in both the middle tertile and highest tertile (log rank p < 0.001) (Figure 3(d)). There were no significant differences in survival by tertile for suPAR or galectin-3.
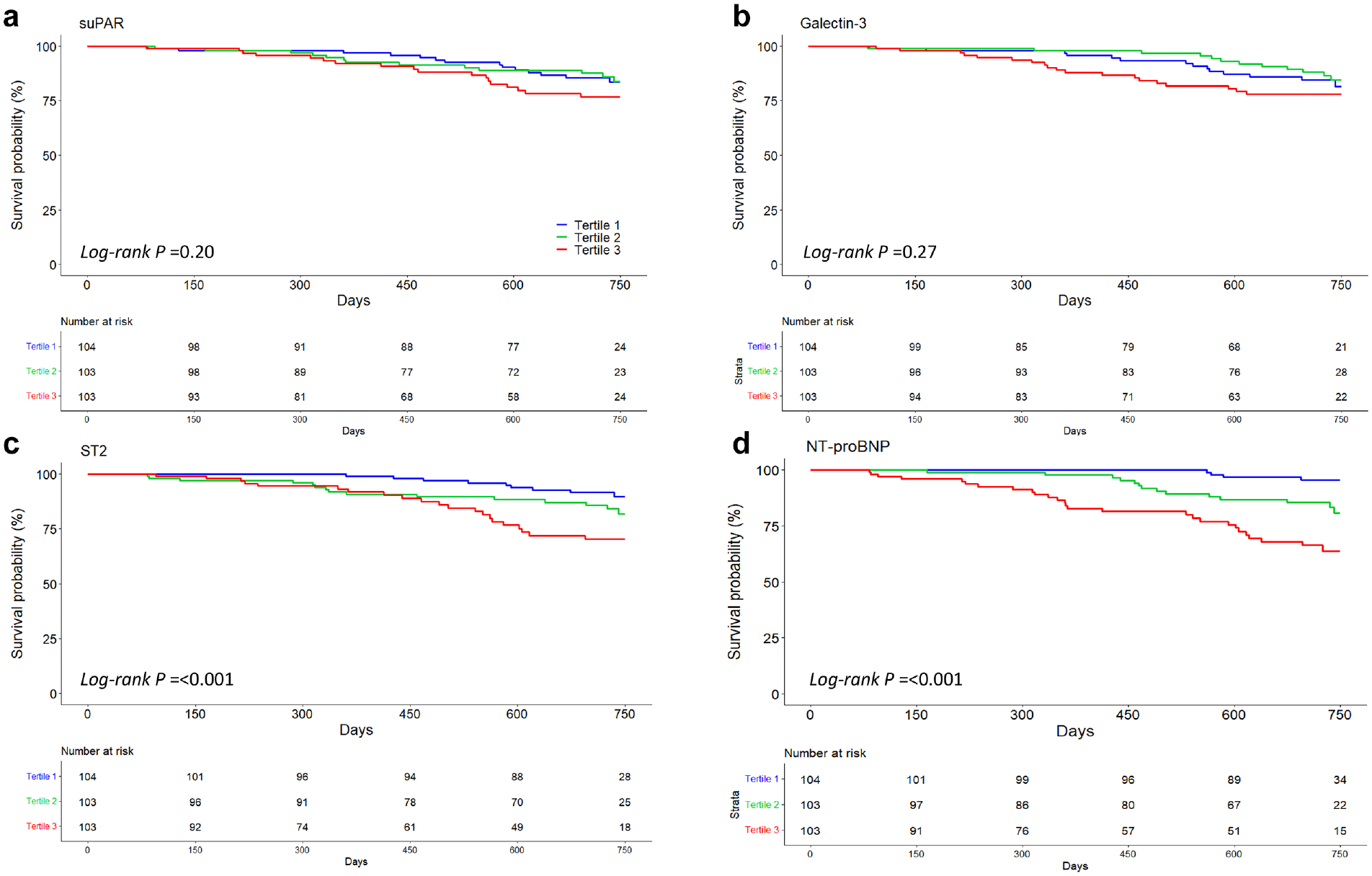
Survival according to baseline biomarker tertiles. Kaplan–Meier survival curves for (a) suPAR, (b) Galectin-3, (c) ST2, and (d) NT-proBNP with corresponding number at risk. p Values from log rank test for trend shown. Pairwise comparisons and p values adjusted by Holm’s method were performed for ST2: first tertile vs second tertile p = 0.15, first tertile vs third tertile p = 0.001, second tertile vs third tertile p = 0.15; and for NT-proBNP: first tertile vs second tertile p = 0.007, first tertile vs third tertile p < 0.001, second tertile vs third tertile p = 0.007.
Univariable Cox proportional hazard regression analysis, without controlling for longitudinal change in eGFR, showed that higher baseline eGFR was associated with lower risk of mortality, and higher suPAR, ST2, and NT-proBNP were associated with greater risk of mortality. suPAR was no longer associated with mortality after controlling for age, sex, diabetes, NYHA class, and race (Table 2). Baseline eGFR was no longer associated with mortality after controlling for NT-proBNP. Controlling for longitudinal change in eGFR using a joint model and standardizing the regression coefficients, ST2 was associated with a greater risk for mortality in univariable analysis (HR 1.52, 95% CI 1.26–1.84, p < 0.001) and multivariable analysis (HR 1.33, 95% CI 1.04–1.69, p = 0.02). Log NT-proBNP was also associated with a greater risk for mortality in univariable analysis (HR 1.62, 95% CI 1.28–2.07, p < 0.001) and multivariable analysis (HR 1.62, 95% CI 1.27–2.07, p < 0.001).
Association of biomarkers with all-cause death.
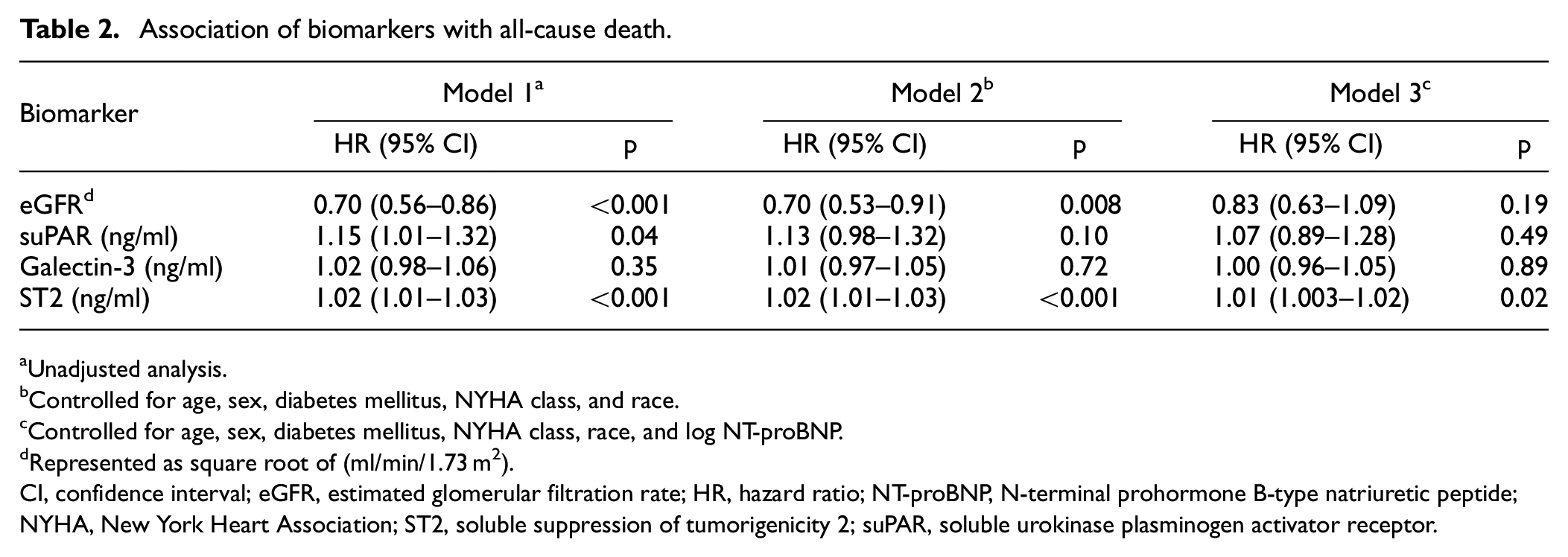
Unadjusted analysis.
Controlled for age, sex, diabetes mellitus, NYHA class, and race.
Controlled for age, sex, diabetes mellitus, NYHA class, race, and log NT-proBNP.
Represented as square root of (ml/min/1.73 m2).
CI, confidence interval; eGFR, estimated glomerular filtration rate; HR, hazard ratio; NT-proBNP, N-terminal prohormone B-type natriuretic peptide; NYHA, New York Heart Association; ST2, soluble suppression of tumorigenicity 2; suPAR, soluble urokinase plasminogen activator receptor.
There was no significant association between suPAR or galectin-3 and mortality in the joint model (Table 3). Controlling for the competing events of death, VAD, and heart transplantation using a joint model, higher levels of suPAR, galectin-3, ST2, and NT-proBNP were each associated with a decline in eGFR over the follow-up observation period on univariable analysis. After controlling for age, sex, diabetes mellitus, NYHA class, race, and log NT-proBNP, these associations remained significant only for suPAR and galectin-3: suPAR (β coefficient, −0.36 √(ml/min/1.73 m2), 95% CI (−0.48 to −0.24), p < 0.001), galectin-3 (β coefficient, −0.14 √(ml/min/1.73 m2), 95% CI (−0.27 to −0.02), p = 0.02). Similarly, these associations remained significant for log NT-proBNP after controlling for age, sex, diabetes mellitus, NYHA class, and race (β coefficient, −0.23 √(ml/min/1.73 m2), 95% CI (−0.31 to −0.15), p < 0.001) (Table 3). Nonstandardized regression coefficients for suPAR and galectin-3 controlling for age, sex, diabetes mellitus, NYHA class, race, and log NT-proBNP were as follows: suPAR (β coefficient, −0.21 √(ml/min/1.73 m2), 95% CI (−0.27 to −0.14), p < 0.001), galectin-3 (β coefficient, −0.02 √(ml/min/1.73 m2), 95% CI (−0.04 to −0.01), p = 0.02) (Supplemental Table).
Joint model point estimates using standardized regression coefficients.
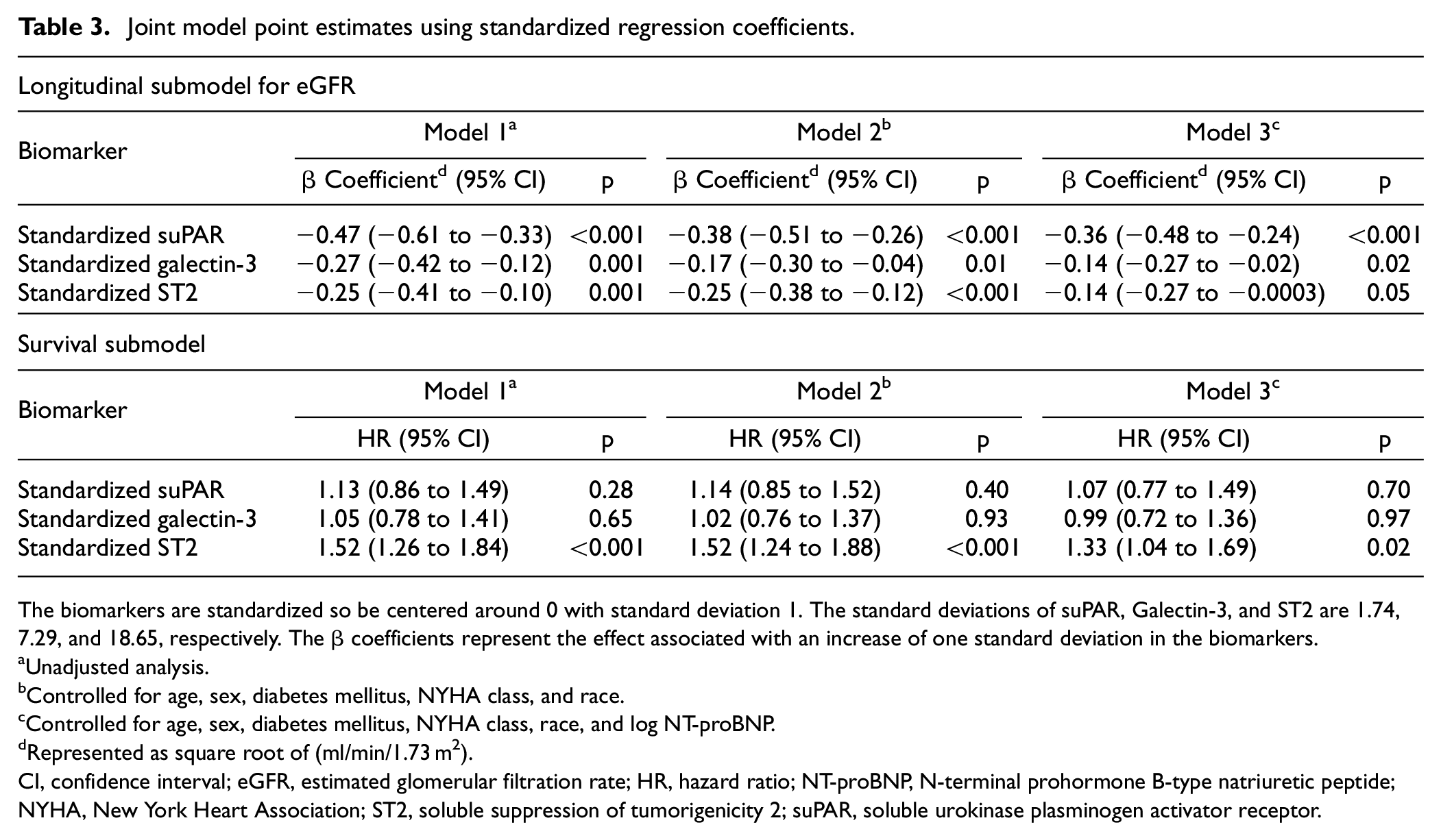
The biomarkers are standardized so be centered around 0 with standard deviation 1. The standard deviations of suPAR, Galectin-3, and ST2 are 1.74, 7.29, and 18.65, respectively. The β coefficients represent the effect associated with an increase of one standard deviation in the biomarkers.
Unadjusted analysis.
Controlled for age, sex, diabetes mellitus, NYHA class, and race.
Controlled for age, sex, diabetes mellitus, NYHA class, race, and log NT-proBNP.
Represented as square root of (ml/min/1.73 m2).
CI, confidence interval; eGFR, estimated glomerular filtration rate; HR, hazard ratio; NT-proBNP, N-terminal prohormone B-type natriuretic peptide; NYHA, New York Heart Association; ST2, soluble suppression of tumorigenicity 2; suPAR, soluble urokinase plasminogen activator receptor.
Discussion
In this longitudinal analysis of individuals with advanced HFrEF, we found that baseline serum levels of biomarkers suPAR, galectin-3, and NT-proBNP were all associated with a decline in eGFR over 2 years of follow-up, even when controlling for competing events of death, VAD, and heart transplantation. Importantly, previous studies examining this association had not accounted for the competing risks of VAD or heart transplantation. Additionally, we found that of these biomarkers, only ST2 and NT-proBNP were associated with all-cause mortality after controlling for longitudinal changes in eGFR, which to our knowledge, is a new finding.
Our study significantly adds to the current available literature on suPAR, ST2, and galectin-3 by evaluating these biomarkers as markers of kidney function decline in a novel population—patients with advanced HFrEF. suPAR and galectin-3 have been associated with kidney disease progression in the general population, whereas studies of ST2 have been conflicting.6,7,15 Neither have been studied in patients with HFrEF, to our knowledge. Galectin-3 has recently been linked to abnormal kidney function in heart failure, though the data are sparse and have not focused specifically on a population with advanced HFrEF.16–18 Our findings build on this growing area of research by demonstrating that all three of these biomarkers are associated with future declines in kidney function in the setting of advanced HFrEF. Future prospective studies examining longitudinal changes in these biomarkers and eGFR in patients with advanced HFrEF, particularly those receiving an LVAD or heart transplantation, are needed. This may shed light on potential mechanisms of cardiorenal interactions. These biomarkers could also be used to help guide candidacy for advanced heart failure therapies among HFrEF patients with concurrent kidney disease or discussion of prognosis.
We found that higher baseline suPAR levels were associated with future declines in eGFR but not mortality. suPAR is the soluble form of the anchor protein urokinase plasminogen activator receptor, which is present on immune cells, endothelial cells, and the kidney and is believed to be involved in acute inflammation and oxidative stress. 19 Previous studies have shown a relationship between suPAR and decline in eGFR in general CKD cohorts, but suPAR’s relationship with kidney function has not been studied in advanced HFrEF. A retrospective study of 3683 patients enrolled in the Emory Cardiovascular Biobank, a general CKD cohort, found that higher baseline suPAR levels were associated with an annual decrease in eGFR by 0.9 ml/min/1.73 m2. 6 In another general CKD cohort from China, higher suPAR was associated with a higher risk of incident end stage kidney disease (ESKD). 20 suPAR has also been associated with disease-specific CKD states, with higher levels reported in patients with diabetic kidney disease21,22 and focal segmental glomerulosclerosis, 23 suggesting that suPAR may be a useful biomarker for kidney disease. Beyond its role in the general CKD population, suPAR is a potentially useful biomarker specifically for heart failure associated kidney disease because of its role in inflammation and oxidative stress. Inflammation and oxidative stress may play a specific pathophysiologic role in cardiorenal syndrome by mediating structural and fibrotic changes in the kidney. 24 Further study of suPAR both in general CKD populations and in patients with heart failure-related kidney disease is warranted to better characterize its role in kidney disease progression.
Like suPAR, our study showed galectin-3 was associated with a decline in eGFR at 2 year follow-up but not with mortality. Galectin-3 belongs to the family of β-galactoside binding lectins. Galectins seem to be involved in a wide variety of processes with galectin-3 largely playing a role in inflammation via its effects on cell–cell adhesion, cell–matrix adhesion, and tissue fibrosis. 25 In particular, galectin-3 has been linked to both kidney and cardiac fibrosis.26–28 Higher galectin-3 levels have been associated with CKD. Two observational studies demonstrated that higher baseline galectin-3 levels were associated with CKD progression in multivariable analysis in general CKD cohorts.7,15 Galectin-3 was associated with the development of incident CKD over 16 year follow-up in the Atherosclerosis Risk in Communities Study, a population with neither pre-existing CKD nor heart failure. 29 Among patients with heart failure, with or without a preserved left ventricular ejection fraction, galectin-3 was shown to be associated with declines in eGFR. In a prospective study of 260 participants with chronic heart failure, higher baseline galectin-3 was associated with worsening kidney function, defined as increase in serum creatinine >0.3 mg/dl, increase in serum creatinine 25% above baseline, or decrease in eGFR 20% below baseline, at 1 year. 17 Our analysis extends these findings over a longer observation period and by controlling for competing events of VAD or heart transplantation.
ST2 was associated with mortality but not with a decline in eGFR in our study. ST2 is a member of the interleukin (IL)-1 receptor family and is involved with mediation of inflammatory factors and cytokines in various organ systems in the body. 30 ST2 may mediate atherosclerosis through its proinflammatory affects 31 and is also associated with hypertension. 32 Given ST2’s association with vascular disease and the vascularity of the kidneys, it stands to reason that ST2 could have prognostic value in CKD. However, prior studies have yielded mixed results. For example, a study of 352 participants found that log ST2 was associated with CKD progression, which they defined as >50% reduction in eGFR over follow-up or ESKD. 7 In contrast, an analysis of the Framingham Offspring Study, ST2 was not associated with incident CKD in multivariable analysis. 33 An analysis of 2763 participants enrolled in the Cardiovascular Heart Study without heart failure found no association between ST2 and incident CKD or a 30% decline in eGFR over follow-up. 34 Another study of 841 participants from two longitudinal cohorts found that doubling of ST2 was not associated with CKD progression, defined as progression to eGFR < 15 ml/min/1.73 m2 or development of ESKD, in multivariable analysis. 15 None of these studies were performed in a population, specifically, comprised of patients with HFrEF as in our study. Similar to the latter studies, ST2 was not associated with declines in eGFR in our study. The association of ST2 with eGFR was attenuated when NT-proBNP was added to the model. This may be because ST2 is predominantly a marker of myocardial stress rather than a broader biomarker of fibrosis beyond the heart. 35
suPAR, galectin-3, ST2, and NT-proBNP have all been studied as biomarkers of hard outcomes in heart failure. However, studies did not account for longitudinal changes in GFR, as in our study. NT-proBNP is known to be associated with increased risk of mortality and heart failure hospitalizations.36,37 suPAR has been associated with higher mortality among patients with acute decompensated heart failure and ambulatory patients with chronic heart failure.9,10 ST2 is believed to mitigate heart failure by blocking the cardioprotective effects of IL-33 leading to increased heart muscle cell apoptosis and fibrosis. 38 Higher levels of ST2 are associated with worse outcomes among patients with both acute decompensated and chronic heart failure, including increased risk of mortality, sudden cardiac death, and hospital readmissions for acute decompensated heart failure.39–41 Similarly, galectin-3 is also associated with poor outcomes among heart failure patients, including mortality, cardiovascular events, and hospitalization for acute decompensated heart failure.42–45
Our study extends these findings by controlling for longitudinal changes in eGFR. This becomes important because declines in eGFR are prevalent among patients with advanced heart failure, and these biomarkers are at least partially renally cleared. Therefore, the association of suPAR and galectin-3 with mortality in prior studies may be influenced by reduced kidney function, as suPAR and galectin-3 are both at least partially cleared by the kidneys, possibly leading to increased blood levels of suPAR and galectin-3 due to decreased kidney clearance among patients with advanced heart failure.46,47
It is also important to point out that when interpreting analyses of biomarkers and mortality, eGFR has historically been inversely associated with mortality, although we did not find this in our study after adjusting for NT-proBNP. 48 If reductions in eGFR are not accounted for, this could result in spurious confounding associations. For example, a study of 154 patients with heart failure with preserved ejection fraction (HFpEF) found a significant interaction between galectin-3 and eGFR with peak oxygen consumption. 49 Similarly, a study of 1540 patients admitted for acute decompensated heart failure found that when testing the association between galectin-3 and ST2 with mortality, there was a significant interaction between galectin-3 and eGFR. There was no interaction between ST2 and eGFR. 50 Our joint model analysis controls for changes in eGFR over time, thus mitigating possible confounding between biomarker levels and reductions in eGFR.
It is also important to point out that these biomarkers may have different prognostic utility depending on heart failure subtype or stage. The study participants in our analysis are comprised of a cohort with advanced heart failure. The majority of previous studies examining biomarker associations with mortality reflect a more heterogeneous population with regards to heart failure severity and include patients with both HFpEF and HFrEF of various stages.10,45,51,52 This may explain why we did not see associations between suPAR and galectin-3 and mortality. In a study of 1235 patients with heart failure examining the association between an array of biomarkers with the composite outcome of cardiovascular death or heart failure hospitalization across the various stages of heart failure, suPAR was associated with the primary composite outcome only among those with Stage C/D heart failure. 53 Galectin-3 was only associated with the composite outcome in patients with Stage A/B heart failure. Contrary to our findings, ST2 was not associated with the composite outcome in any stage of heart failure. A study of 592 patients admitted for acute decompensated heart failure and followed for 18 months with serial galectin-3 measurements found that although galectin-3 levels were no different between patients with HFpEF and HFrEF, galectin-3 was more predictive of mortality in patients with HFpEF. 45 Alternatively, given that we failed to see an association with suPAR or galectin-3 in survival analyses that both did and did not account for changes in eGFR, it may be that we were underpowered to detect these associations.
Our study has several strengths. First, unlike previously published studies, we utilized a joint model in our primary analysis. In doing so, we were able to control for the effects the competing events of death, VAD implantation, and heart transplantation could have had on longitudinal changes in eGFR. Similarly, in our survival analyses, we were able to control for the confounding effects that changes in eGFR may have had on mortality. We also implemented the 2021 CKD-EPI equation to estimate GFR at baseline and follow-up in our longitudinal analyses, which is now the standard or care and, to our knowledge, has not been used in previously published studies of these biomarkers in heart failure. The 2021 CKD-EPI equation removed the race coefficient due to increasing concerns that use of a race coefficient was contributing to systemic racism and healthcare disparities in medicine. 13 Lastly, this was a multicenter study that enrolled participants from 21 different centers so these data may be more generalizable than previous cohort studies reporting these associations.
Our study also has a few limitations. Only baseline measures of suPAR, galectin-3, and ST2 were available in the dataset, so we were not able to analyze whether these biomarker levels changed in participants over time or what effects those changes may have had on eGFR or mortality. Similarly, cystatin C was only available at baseline, so we were only able to ascertain longitudinal changes in eGFR using serum creatinine. Serum creatinine levels are influenced by muscle mass which may be decreased in patients with advanced heart failure leading to less accurate GFR estimates.54,55 In addition, proteinuria was not available in the database so could not be accounted for in the analysis. Finally, although participants were enrolled from different centers, the population was predominantly comprised of white men. While our results are generalizable at the population level, they may not be generalizable at the individual patient level and need to be validated in cohorts comprised of a larger proportion of women and diverse groups.
Conclusion
In a population with advanced HFrEF, higher serum levels of suPAR, galectin-3, and NT-proBNP at baseline were all associated with a decline in eGFR over 2 years, even after controlling for competing events of death, VAD, and heart transplantation. Controlling for longitudinal changes in kidney function decline, only ST2 and NT-proBNP were associated with mortality. Use of these biomarkers in clinical practice could provide prognostic utility and warrant investigation to establish whether they inform candidacy for advanced heart failure therapies.
Supplemental Material
sj-docx-1-imj-10.1177_10815589241249991 – Supplemental material for Association of suPAR, ST2, and galectin-3 with eGFR decline and mortality in patients with advanced heart failure with reduced ejection fraction
Supplemental material, sj-docx-1-imj-10.1177_10815589241249991 for Association of suPAR, ST2, and galectin-3 with eGFR decline and mortality in patients with advanced heart failure with reduced ejection fraction by Bethany Roehm, Meredith McAdams, Jonathan Gordon, Song Zhang, Pin Xu, Justin L Grodin and S Susan Hedayati in Journal of Investigative Medicine
Footnotes
Acknowledgements
This Manuscript was prepared using REVIVAL Research Materials obtained from the National Heart, Lung, and Blood Institute (NHLBI) Biologic Specimen and Data Repository Information Coordinating Center and does not necessarily reflect the opinions or views of the REVIVAL or the NHLBI.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: J. Grodin receives fees for scientific consulting from Pfizer, Eidos/BridgeBio, Astra-Zeneca, and Sarepta.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: B. Roehm is supported by the National Center for Advancing Translational Sciences of the National Institutes of Health (NIH) under Award Number KL2TR003981. M. McAdams was supported by training grant 5T32DK007257-38 from the National Institutes of Diabetes and Digestive and Kidney Diseases. S. Hedayati is supported by an R38 residency training grant from the National Heart, Lung, and Blood Institute (NHLBI), R38HL150214 and the Lina Obeid Chair in Biomedical Sciences at Stony Brook University, Renaissance School of Medicine. B. Roehm, S. Hedayati, and J. Grodin are also supported by the NHLBI under Award Number R21HL168496-01. Some of the statistical analysis was supported by grant UL1TR003163 awarded to University of Texas Southwestern Medical Center (UTSW). The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
