Abstract
Cisplatin use is often limited by its ototoxic side effects, which can lead to irreversible hearing loss. Preventing cisplatin-induced ototoxicity is crucial to improve patient outcomes. Fluoxetine and fluvoxamine, both selective serotonin reuptake inhibitors antidepressants, inhibit the NLR pyrin domain-containing protein 3 inflammasome, a potential therapeutic target for preventing ototoxicity. However, human studies have not evaluated if these antidepressants may protect against cisplatin-induced ototoxicity. The object of this study is to assess the association between fluoxetine or fluvoxamine use and incidence of hearing loss or tinnitus in a large cohort of patients receiving cisplatin chemotherapy. We use a retrospective cohort study within the U.S. Department of Veterans Affairs healthcare system. Adult patients with cancer who received cisplatin chemotherapy between 2000 and 2023 are included. Incidence of ototoxicity, defined by international classification of diseases revision 9-CM or international classification of diseases revision 10-CM diagnoses of hearing loss or tinnitus is compared between concurrent use of fluoxetine or fluvoxamine and cisplatin alone. A total of 20,552 patients were included. Of those, 489 received cisplatin and fluoxetine or fluvoxamine. After propensity score adjustment, the hazard of ototoxicity was lower in the group receiving fluoxetine or fluvoxamine compared to the group receiving cisplatin alone (HR = 0.62, 95% CI = (0.41–0.94)). Fluoxetine or fluvoxamine use may be associated with a reduced risk of cisplatin-induced ototoxicity. Randomized clinical trials are needed to confirm these findings and establish the efficacy of the medications in ototoxicity prevention. Further research is also warranted to investigate the potential mechanisms underlying this protective effect.
Introduction
Cisplatin is a platinum-based chemotherapeutic utilized to treat solid tumor malignancies. While efficacious, cisplatin is ototoxic, damaging the inner ear, resulting in tinnitus and irreversible hearing loss. Cisplatin-induced ototoxicity affects 36% of adult patients to 60% of pediatric patients.1–3 Clinical guidance recommends audiology assessments to monitor ototoxicity and to prevent subjective hearing loss, 4 although these assessments are underutilized. 5 Sodium thiosulfate is U.S. FDA approved to prevent cisplatin ototoxicity in pediatric patients. 6 No drugs are approved for preventing cisplatin ototoxicity in adults. Therefore, new therapies are needed that could potentially limit the ototoxicity of cisplatin.
Cisplatin is known for its ototoxic side effects and can lead to irreversible hearing loss.
There are no current therapies available for adults to protect against cisplatin ototoxicity.
The NLR pyrin domain-containing protein 3 (NLRP3) inflammasome is a potential therapeutic target for preventing ototoxicity.
Fluoxetine and fluvoxamine have been demonstrated to bind and inhibit NLRP3 activation.
Patients using fluoxetine or fluvoxamine with cisplatin exhibited a lower hazard of tinnitus/hearing loss compared to those receiving cisplatin alone.
Other selective serotonin reuptake inhibitors antidepressants, which do not inhibit the NLRP3 inflammasome, are not associated with tinnitus or hearing loss following cisplatin administration.
Fluoxetine or fluvoxamine use was not associated with a statistically significant increase in mortality.
Randomized clinical trials would be needed to confirm the protective effect of fluoxetine and fluvoxamine on ototoxicity.
The results provide preliminary evidence for the NLRP3 inflammasome as a therapeutic target for ototoxicity.
Cisplatin enters the cochlea through various molecular transport mechanisms and is not rapidly cleared compared to other organs including the liver and kidney. 7 The mechanism of the ototoxicity of cisplatin involves generation of reactive oxygen species that overwhelms the cells antioxidant capability resulting in apoptosis of hair cells and subsequently hearing loss. Inner ear structures were previously thought to be “immuno-privileged” due to barriers blocking immune cells. 8 However, recent research suggests a critical role of inflammation in cisplatin-induced ototoxicity.9–11 The NLR pyrin domain-containing protein 3 (NLRP3) inflammasome is a key regulator of the innate immune system and is implicated in the pathogenesis of various inflammatory diseases. The NLRP3 inflammasome is implicated in noise-induced hearing loss following acoustic trauma 12 and a key mediator of cisplatin induced ototoxicity. 13 An increased expression of NLRP3, ASC, and caspase-1 is found in cisplatin-treated marginal cells (MCs) and the down regulation of NLRP3 reduced the number of damaged MCs, 13 suggesting that cisplatin activates the NLRP3 inflammasome subsequently contributing to hair cell death.
Fluoxetine and fluvoxamine, both selective serotonin reuptake inhibitors (SSRI) indicated for the treatment of depression, bind to the NLRP3 inflammasome and inhibit its activation. 14 As NLRP3 inflammasome inhibitors, we hypothesize that fluoxetine or fluvoxamine could reduce cisplatin ototoxicity. We test this hypothesis using real-world data analyses of a national cohort of U.S. veterans undergoing cisplatin treatment.
Materials and methods
Data source
The drug-disease association study was conducted using data from the U.S. Department of Veterans Affairs. Study data were extracted from the VA Informatics and Computing Infrastructure, which includes infusion, inpatient, outpatient data (coded with international classification of diseases (ICD) revision 9-CM, revision 10-CM), and pharmacy claims. The study was conducted in compliance with U.S. Department of Veterans Affairs requirements, received Institutional Review Board, and Research and Development approval. Strengthening the Reporting of Observational studies in Epidemiology guidelines was followed for this study.
Cohort creation and outcome
The cohort included patients receiving intravenous (IV) cisplatin administration. All cisplatin administrations were extracted from the VA IV medication database. Administrations from the year 2000, with valid start and stop times, were included. Patients who had no cisplatin dosage information for any administrations were excluded. Cohort data were organized longitudinally, with patient-cisplatin administration as the unit of analysis. Patients were followed up from first cisplatin administration to first incidence of death, ototoxicity, next cisplatin administration, November 1, 2023, or 180 days after administration.
Ototoxicity was categorized as a composite of incident diagnosis of tinnitus or hearing loss. All diagnosis codes used are listed in Supplemental Table S1. Patients with diagnoses for either tinnitus or hearing loss prior to the first cisplatin administration were excluded.
Exposure and covariate data
The primary exposure of interest was treatment with fluoxetine or fluvoxamine, both SSRI antidepressants. Patients were considered exposed if they had a supply of either drug at the time of the cisplatin administration. Exposure was coded dynamically for each cisplatin administration. All fluoxetine and fluvoxamine prescriptions were extracted from VA outpatient pharmacy dispensing data.
Covariates that potentially influence risk of cisplatin ototoxicity were included. Cumulative cisplatin dosage was calculated as the cumulative sum of the number of milligrams administered with each cisplatin administration. Radiation therapy can increase ototoxicity risk. This was captured longitudinally by the number of cumulative days with radiation therapy. Radiation therapy was extracted using current procedural terminology (CPT) codes (Supplemental Table S1). Patients with head and neck cancers have higher risks of otologic complications. Patients with head and neck cancers were extracted from VA oncology data using primary tumor sites including gum, hypopharynx, larynx, lip, salivary gland, mouth, nasopharynx, oropharynx, palate, pyriform sinus, submandibular gland, tongue, tonsilliar fossa, and tonsillar pillar. Patients with head and neck cancer were flagged if they appeared in the oncology data within 6 months prior to the first cisplatin administration.
Other data included demographics, age, race, gender, and clinical characteristics such as body mass index (BMI) and, as a measure of comorbidity burden, the Charlson comorbidity index. Patients who smoked were flagged using the VA health factors data, which reports results of screening questions. Health factors indicating the current use of tobacco were extracted using text search and subsequently flagged as smokers.
Statistical analysis
Patients’ characteristics are displayed comparing patients who had any fluoxetine or fluvoxamine exposure and those who never had fluoxetine or fluvoxamine exposure during cisplatin administrations. Means and standard deviations are presented for continuous variables. N values and percentages are presented for categorical variables. T-tests and chi-square tests along with the mean standardized difference are used to compare similarities of the cohorts. To assess the association of fluoxetine or fluvoxamine and ototoxicity, incidence rates are calculated using Poisson regression, along with corresponding 95% confidence intervals (CIs) and a Poisson regression-based p-value. Cox proportional hazards models are fit to estimate the risk of ototoxicity. Because treatment was not randomized, propensity score analysis was used to minimize bias. Propensity score adjustment was used including the propensity score as a covariate in the Cox model. To flexibly model the relationship between propensity score and outcome, we used natural spline with three knots. Further, we fit a marginal structural model, using stabilized propensity score weights. All time-dependent and time-fixed covariates were included in the propensity score model. Cox models are all fit using cluster (patient) robust standard errors.
Prior research showed that while fluoxetine and fluvoxamine can both bind and inhibit NLRP3 inflammasome activation, other SSRI medications had no such effect. 14 Therefore, exposure to any other SSRI medications including sertraline, paroxetine, citalopram, escitalopram, vortioxetine was tested as well. Sertraline, one of the most common SSRIs utilized, was tested separately. Cox models were used to examine associations between other SSRI use and ototoxicity.
Mortality analysis
As a secondary outcome, the effect of fluoxetine or fluvoxamine exposure on mortality was assessed. Cohort data were extracted separately from the primary analysis, and patients were followed up from their first cisplatin administration to subsequent cisplatin administrations, death, November 1, 2023, or a maximum of 5 years after their last cisplatin administration. Like the primary analysis, data were structured longitudinally, with the unit of analysis being patient-cisplatin administration. Fluoxetine or fluvoxamine exposure was coded time-dependently, indicating if patients had a supply of either medication on the cisplatin administration day. Cox models and propensity score analysis from the primary analysis were replicated for the mortality analysis. As an additional analysis, effects between sertraline, other non-fluoxetine or fluvoxamine SSRIs, and mortality were assessed.
Results
Ototoxicity
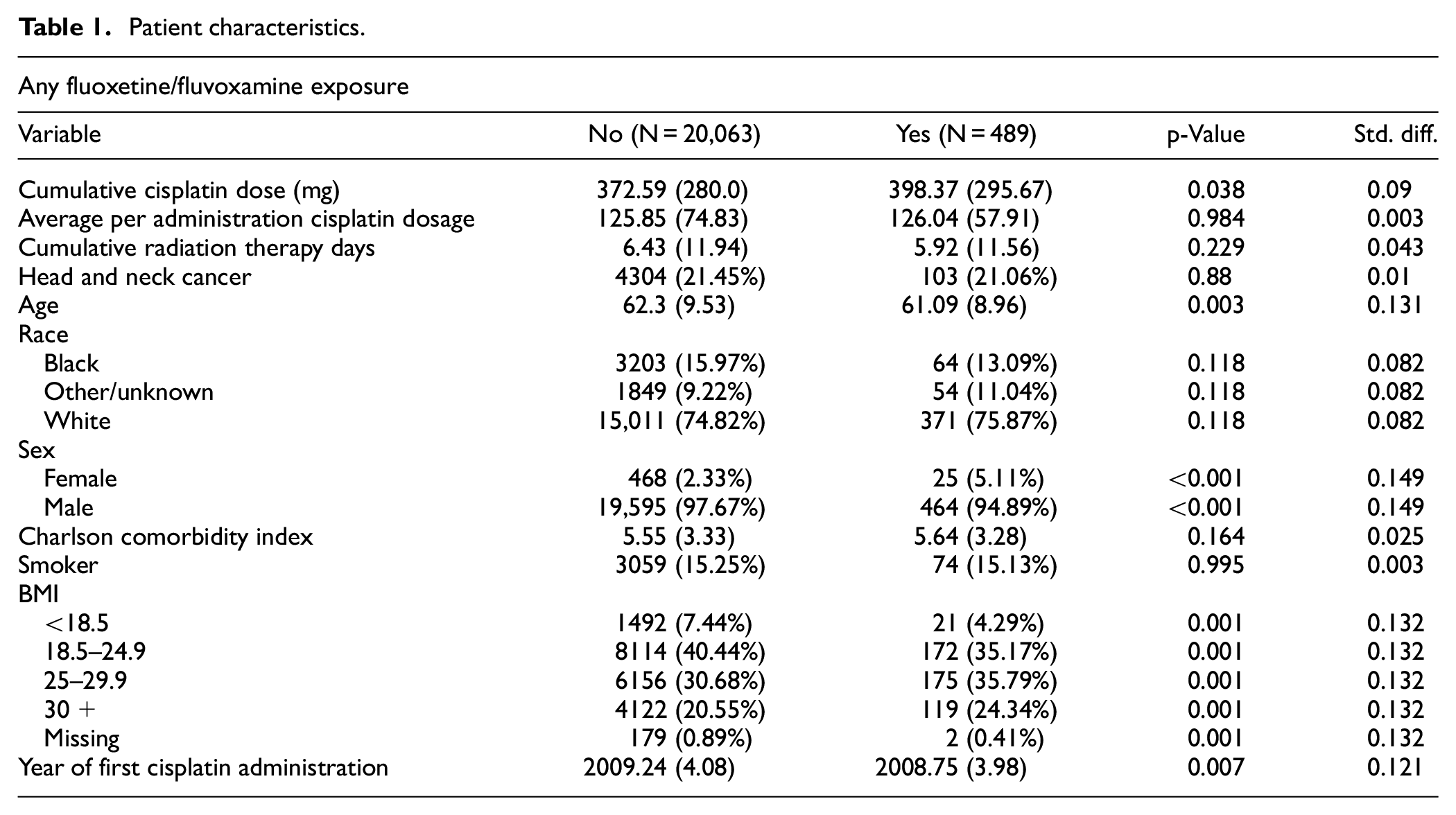
A total of 20,552 patients treated with cisplatin were included. Table 1 displays patient characteristics comparing patients exposed to either fluoxetine or fluvoxamine during cisplatin administration compared to unexposed patients. On average, patients with exposure had 6% higher total cumulative cisplatin dosages compared to those without exposure (372 mg vs 398 mg, p = 0.04). The average per administration cisplatin dosage was not statistically different between cohorts (125.8 mg vs 126 mg, p = 1.0). No statistically significant differences were observed in the total cumulative radiation therapy days or the incidence of head and neck cancer. Several variables showed notable differences. Patients exposed to fluoxetine/fluvoxamine were slightly younger, with a mean age of 61 years compared to 62.11 years in the unexposed group (p = 0.003). Furthermore, a significantly higher proportion of females (5.2%) were observed in the exposed group compared to the non-exposed (2.3%; p < 0.001). The exposed group also had higher frequencies in the BMI categories of 25–29.9 and 30+ (p = 0.001).
Patient characteristics.
Incidence rates of ototoxicity per 1000 person-years for patients with and without fluoxetine/fluvoxamine exposure are displayed in Table 2. Fluoxetine/fluvoxamine exposure had a lower per 1000 person-years incidence rate (117.7, 95% CI = (78.2–177.1)) compared to no exposure (190.6, 95% CI = (182.4–199.1)).
Ototoxicity incidence rate per 1000 person-years.
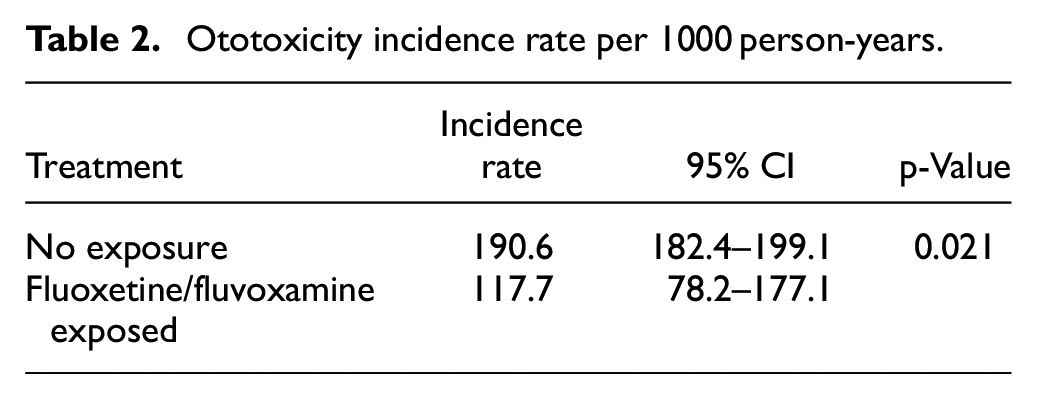
Table 3 presents the results from time-dependent Cox models assessing the risk of ototoxicity. Fluoxetine/fluvoxamine exposure was associated with a reduced hazard of ototoxicity compared to non-exposure (HR = 0.62). Higher cumulative cisplatin exposure was linked to a significant increase in ototoxicity risk in the original sample and marginal structural modeling estimates. Similarly, increased cumulative radiation therapy days were associated with higher ototoxicity hazards in both original sample models and the marginal structural models estimates. Head and neck cancer consistently elevated ototoxicity risk in all models. In the marginal structural model, head and neck cancers were associated with a 75% increased risk of ototoxicity (HR = 1.75, 95% CI = (1.59–1.92)). Compared to black patients, white patients had a consistently higher risk of ototoxicity in all models.
Cox model results: ototoxicity.
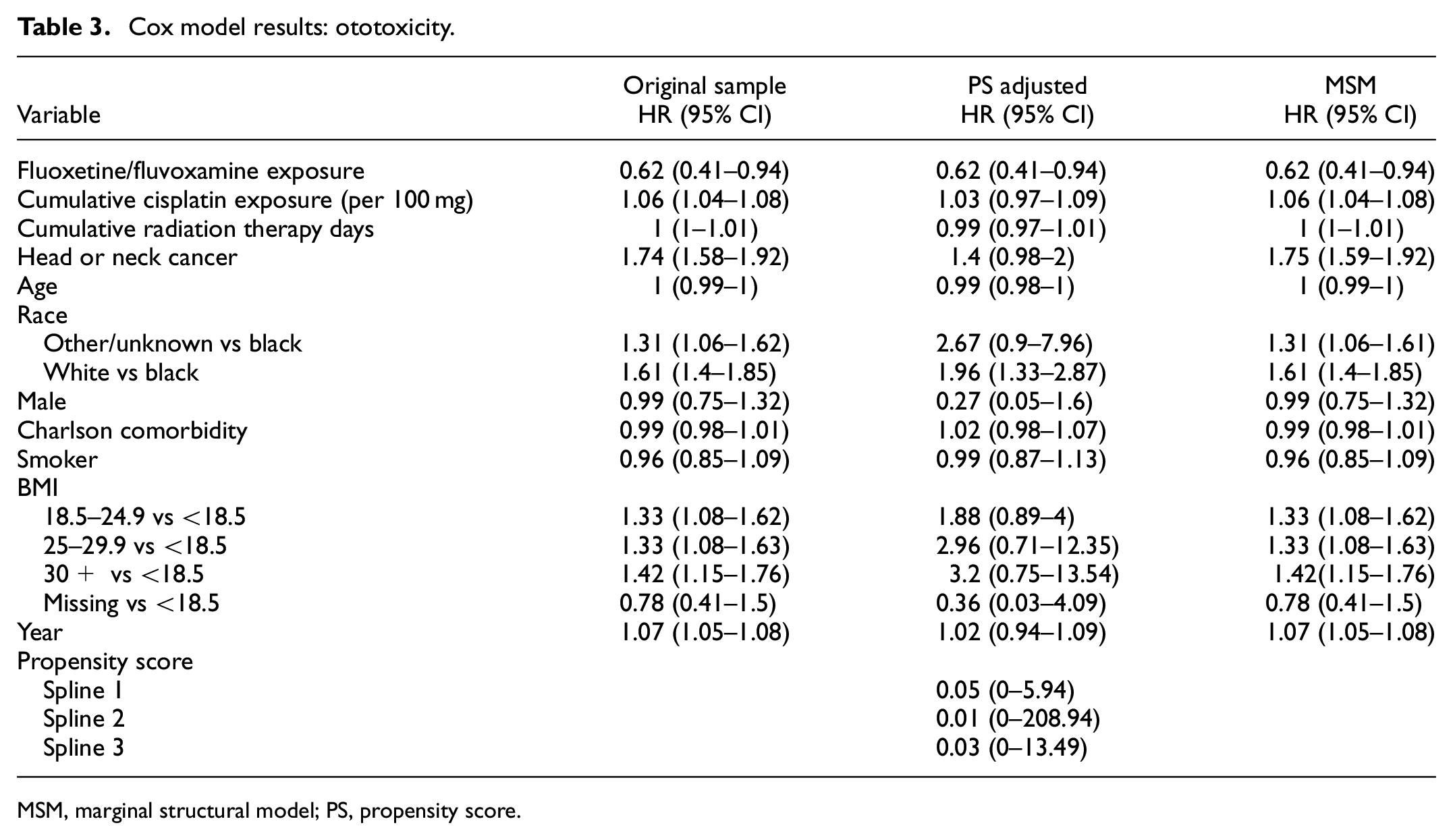
MSM, marginal structural model; PS, propensity score.
To examine if the observed relationship between fluoxetine or fluvoxamine is the result of a general antidepressant effect rather than being medication specific, models were fit assessing non-fluoxetine or fluvoxamine SSRIs on ototoxicity. Cox model results are displayed in Table 4. The results find no significant association between sertraline exposure and ototoxicity. Moreover, exposure to any non-fluoxetine or fluvoxamine SSRIs demonstrated no significant association with ototoxicity.
Cox model: SSRI, Sertraline exposure and ototoxicity.
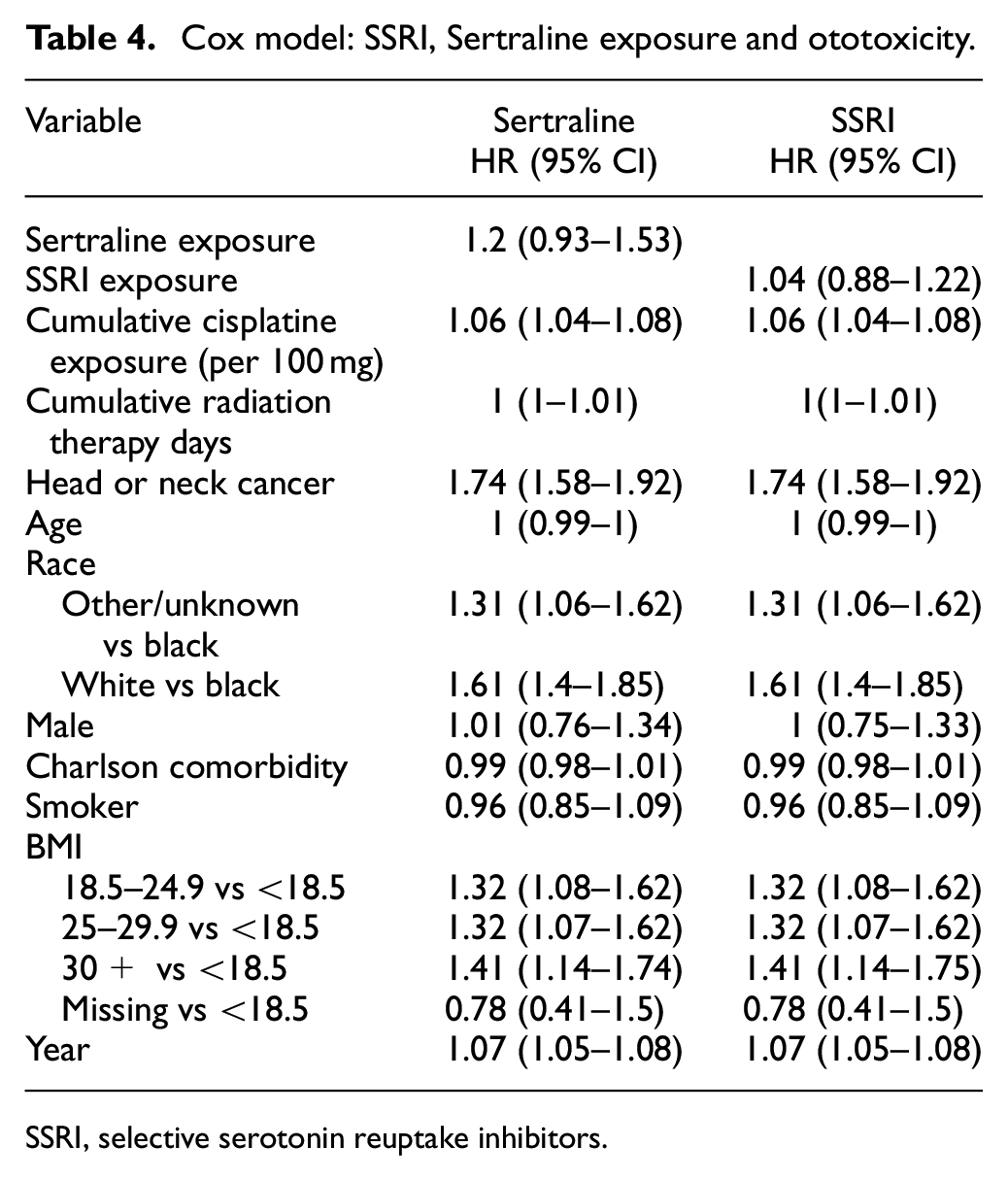
SSRI, selective serotonin reuptake inhibitors.
Mortality
Overall, 29,479 patients, of whom 776 had exposure to fluoxetine/fluvoxamine during cisplatin administration, are included in the mortality analysis. Supplemental Table S2 displays patient characteristics. Patients with exposure demonstrate a slightly higher total cumulative cisplatin dose compared to unexposed counterparts (417.2 mg vs 387.8 mg, p = 0.019). However, no significant differences were noted in average per administration cisplatin dosage, or number of radiation days between exposed and never exposed groups. The prevalence of head and neck cancers was similar in both groups, approximately 22%. There were differences in the demographic profiles between groups. Fluoxetine/fluvoxamine-exposed patients were slightly younger, on average (61.9 vs 63.2, p < 0.001), less likely to be black (11% vs 14.3%, p = 0.03) and more likely to be female (4% vs 2%, p < 0.001).
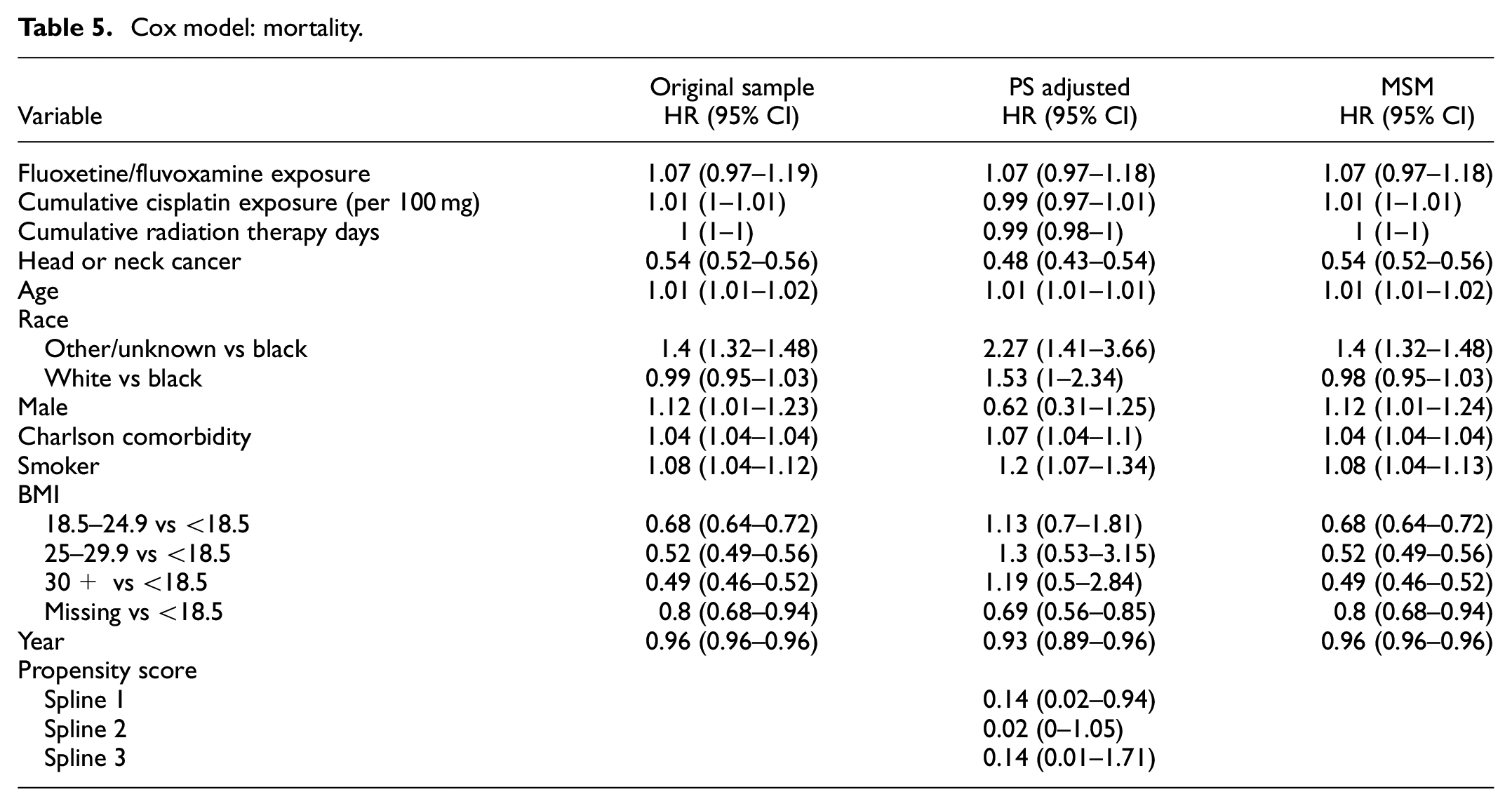
Hazard ratios and associated CIs from the multivariable Cox models are presented in Table 5. Fluoxetine/fluvoxamine exposure was not associated with a statistically significant difference in mortality compared to non-exposure across all models. Point estimates revealed a hazard ratio of 1.07 indicating an increased risk of mortality, however, CIs cannot rule out a protective association as well. Variables associated with a higher risk of mortality included older age and smoking status. Supplemental Table S3 displays the Cox model results for both sertraline and other non-SSRI antidepressants. No statistically significant relationship between sertraline, or other non-SSRI antidepressants, and mortality was found.
Cox model: mortality.
Discussion
Cisplatin, a potent chemotherapeutic agent, is an essential component in treating various solid tumor malignancies. Its use is limited by the development of ototoxicity, which can manifest as tinnitus and irreversible hearing loss. While guidelines recommend audiology assessments 4 to monitor ototoxicity, underutilization persists. 5 Sodium thiosulfate remains the sole U.S. FDA-approved preventive measure in pediatric patients, 6 leaving an unmet need for effective interventions, particularly in adults. Fluoxetine and fluvoxamine, both SSRI have been shown to bind to and inhibit the NLRP3 inflammasome. 14 Prior research has indicated downregulation of the NLRP3 inflammasome resulted in less cell death among MCs of the stria vascularis, an important trafficker of cisplatin in the cochlea. Given preclinical findings regarding fluoxetine/fluvoxamine, the NLRP3 inflammasome, and its potential for to prevent cell death in the cochlea, this study aimed to investigate any association between fluoxetine or fluoxetine and hearing loss or tinnitus after cisplatin. Our findings shed light on complex interplay of factors contributing to cisplatin ototoxicity and provide insight for future studies.
The analysis encompassed a large cohort of 20,552 patients undergoing cisplatin treatment. The primary outcome of the study, ototoxicity, was assessed using a composite of incident diagnoses of tinnitus or hearing loss. Incidence rates indicated a lower rate of ototoxicity among patients with fluoxetine exposure, providing evidence of potential protective effects. Cox models, adjusted for important factors related to cisplatin ototoxicity, further substantiated this finding, demonstrating a reduced hazard of ototoxicity associated with fluoxetine or fluvoxamine exposure in the all models (original sample, propensity score-adjusted analysis, and marginal structural model). Other SSRIs including sertraline showed no such association, suggesting specificity of the effect to fluoxetine/fluvoxamine. Moreover, treatments used in conjunction with cisplatin should not adversely impact cisplatin’s effectiveness. Secondary analysis showed that fluoxetine/fluvoxamine had no statistically significant effect on mortality. Importantly, however, point estimates suggest a higher 5-year mortality risk even though the result was not statistically significant.
This study was conducted to evaluate the possibility of targeting the NLRP3 inflammasome to prevent ototoxicity. While our findings are promising, they warrant further research and validation prior to clinical use. Randomized clinical trials would be needed to determine a cause-and-effect relationship and examine ototoxicity and mortality risks when cisplatin and fluoxetine or fluvoxamine are administered.
Study limitations need to be considered. As a real-world drug-association study, audiology data were not available; the outcome relies on diagnostic coding of tinnitus or hearing loss in medical claims data. Moreover, as a retrospective study, treatment assignment was not randomized, and residual confounding factors may remain. That said, we used propensity score analysis, including time-dependent propensity scores via covariate adjustment and marginal structural model to minimize the potential bias.
We undertook other analysis to examine if the observed associations were due to a systematic bias toward patients with an antidepressant. To explore whether the observed effects are specific to fluoxetine or fluvoxamine or are a class-wide effect of SSRIs, we conducted a secondary analysis using exposure of any non-fluoxetine/fluvoxamine SSRIs as well as sertraline, one of the most common SSRIs. Point estimates for the other SSRIs indicated an opposite effect, patients were more at risk of ototoxicity, but this was not statistically significant. Results for sertraline exposure were similar, a point estimate indicating a higher, but non-statistically significant, risk of ototoxicity. These findings suggest that the protective effects observed with fluoxetine or fluvoxamine may not be due to indication bias, emphasizing the importance of considering specific mechanisms, notably NLRP3 inflammasome inhibition. Further mechanistic studies examining the potential for NLRP3 inflammasome inhibition as a therapeutic target for cisplatin ototoxicity are warranted.
In conclusion, cisplatin-induced ototoxicity is a significant concern in cancer treatment, affecting a substantial proportion of patients. This study highlights the potential of fluoxetine or fluvoxamine as protective agents against cisplatin-induced ototoxicity.
Supplemental Material
sj-docx-1-imj-10.1177_10815589241247796 – Supplemental material for Fluoxetine, fluvoxamine, and hearing loss or tinnitus after cisplatin treatment: A retrospective cohort study
Supplemental material, sj-docx-1-imj-10.1177_10815589241247796 for Fluoxetine, fluvoxamine, and hearing loss or tinnitus after cisplatin treatment: A retrospective cohort study by Joseph Magagnoli, Tammy H. Cummings, James W. Hardin, S. Scott Sutton and Jayakrishna Ambati in Journal of Investigative Medicine
Footnotes
Acknowledgements
No funding agency had a role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to the manuscript for publication. The content of this article is solely the responsibility of the authors and does not necessarily represent the official views of the U.S. Department of Veterans Affairs, nor does mention of trade names, commercial products, or organizations imply endorsement by the U.S. government. This paper represents original research conducted using data from the U.S. Department of Veterans Affairs and is the result of work supported with resources and the use of facilities at the Dorn Research Institute, Columbia VA Health Care System, Columbia, South Carolina. We thank Charles L. Bennett, MD, PhD, MPP, Professor, College of Pharmacy, University of South Carolina, for discussions and editing of the manuscript. Dr Bennett did not receive financial compensation for this contribution.
Author contributions
J.M. had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis; Concept and design: J.M., S.S.S., and J.A.; Acquisition, analysis, or interpretation of data: J.M., T.H.C., J.W.H., S.S.S., and J.A.; Drafting of the manuscript: J.M., S.S.S., and J.A.; Critical revision of the manuscript for important intellectual content: T.H.C. and J.W.H.; Statistical analysis: J.M.; and Obtained funding: S.S.S. and J.A.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Sutton has received research grants from Boehringer-Ingelheim, Gilead Sciences, Alexion Pharmaceuticals, and United Therapeutics, all for projects unrelated to study. J.A. is a co-founder of DiceRx, iVeena Holdings, iVeena Delivery Systems and Inflammasome Therapeutics, and, unrelated to this work, he has been a board member for Theragen and consultant for Abbvie/Allergan, Boehringer-Ingelheim, Janssen, Olix Pharmaceuticals, Retinal Solutions, and Saksin LifeSciences. The other authors declare no competing interests.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: S.S.S., J.M., and T.C. are supported by National Institutes of Health (NIH) grant R01DA054992 and the South Carolina Center for Rural and Primary Healthcare unrelated to this study. J.A. discloses support from the UVA Strategic Investment Fund and NIH grants (R01EY029799, R01EY031039, R01AG078892, R01AG082108) and the DuPont Guerry, III, Professorship.
Data availability
Analyses of the Veterans Health Administration Database were performed using data within the U.S. Department of Veterans Affairs secure research environment and the VA Informatics and Computing Infrastructure (VINCI). The completeness, utility, accuracy, validity, and access methods are described on the VA website: ![]() .
.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
