Abstract
Dysregulated cholesterol metabolism represents an increasingly recognized feature of autism spectrum disorder (ASD). Children with fetal valproate syndrome caused by prenatal exposure to valproic acid (VPA), an anti-epileptic and mood-stabilizing drug, have a higher incidence of developing ASD. However, the role of VPA in cholesterol homeostasis in neurons and microglial cells remains unclear. Therefore, we examined the effect of VPA exposure on regulation of cholesterol homeostasis in the human microglial clone 3 (HMC3) cell line and the human neuroblastoma cell line SH-SY5Y. HMC3 and SH-SY5Y cells were each incubated in increasing concentrations of VPA, followed by quantification of mRNA and protein expression of cholesterol transporters and cholesterol metabolizing enzymes. Cholesterol efflux was evaluated using colorimetric assays. We found that VPA treatment in HMC3 cells significantly reduced ABCA1 mRNA, but increased ABCG1 and CD36 mRNA levels in a dose-dependent manner. However, ABCA1 and ABCG1 protein levels were reduced by VPA in HMC3. Furthermore, similar experiments in SH-SY5Y cells showed increased mRNA levels for ABCA1, ABCG1, CD36, and 27-hydroxylase with VPA treatment. VPA exposure significantly reduced protein levels of ABCA1 in a dose-dependent manner, but increased the ABCG1 protein level at the highest dose in SH-SY5Y cells. In addition, VPA treatment significantly increased cholesterol efflux in SH-SY5Y, but had no impact on efflux in HMC3. VPA differentially controls the expression of ABCA1 and ABCG1, but regulation at the transcriptional and translational levels are not consistent and changes in the expression of these genes do not correlate with cholesterol efflux in vitro.
Autism spectrum disorder (ASD) is a developmental disorder of the nervous system that is usually diagnosed in early childhood. Cholesterol plays a major role in neural development. Deficiency in cholesterol is associated with structural disorders of neurons and may contribute to development of ASD. Valproic acid (VPA) is widely used as an antiepileptic drug. Maternal use of VPA during pregnancy has been associated with increased risk of development of ASD in children.
VPA treatment reduces plasma cholesterol levels, but effect of VPA treatment on cholesterol metabolism in cell types found in the brain is still unclear. Our study found that VPA treatment altered cholesterol transport and metabolizing enzymes in two key cell types, microglia, and neurons that compose the developing brain.
There is much literature affirming the role of VPA in the development of ASD, but the mechanism of VPA involvement in ASD is still unknown. Our study suggests that VPA exposure causes hypocholesterolemic effects in neurons by promoting cholesterol efflux. Together, our results showed that VPA impairs cholesterol homeostasis in human neurons. Further studies are necessary for a better understanding of the involvement of cholesterol in the mechanisms by which VPA leads to ASD and expanded knowledge may translate into novel preventative therapies for this serious disorder.
Introduction
Autism spectrum disorder (ASD) is a neurodevelopmental condition with lifelong consequences and poorly understood pathophysiology. This behavioral disorder is characterized by impairments in social interaction, both verbal and nonverbal communication, as well as restricted interests and repetitive patterns of behavior. Many potential causes of ASD have been explored, including genetic, infectious, and environmental factors. 1 Recent studies have shown that a low level of cholesterol in maternal or child plasma is associated with ASD.2–5 Furthermore, children with inborn errors in cholesterol metabolism have behavioral abnormalities that are consistent with those observed in autism. These findings underscore essential role of cholesterol in neuroactive steroid production, myelin sheath formation, and normal brain development. The brain contains 23% of all sterol content in the body despite being only 2% of body weight, and most of this is distributed between the plasma membranes of the neurons, the glia, where it is used in the myelin sheath surrounding the axons, and lipid rafts that regulate the release of neurotransmitters into the synaptic cleft. Therefore, deficiencies in cholesterol may lead to defective myelination and synapse formation during the development that result in ASD. 6
Numerous studies have demonstrated that in utero exposure to the anticonvulsant and mood-stabilizing drug valproic acid (VPA), causes autism-like neural and behavioral deficits in humans and rodents. While the mechanisms by which VPA leads to these deficits are unclear, exposure to VPA has been demonstrated to reduce plasma cholesterol levels in both humans and animal models. However, VPA induces autistic-like behaviors by disrupting cholesterol metabolism in neurons is unknown. In light of the importance of cholesterol as a link between VPA and ASD, we report here the results of a series of studies to evaluate the effects of VPA on the expression of cholesterol transporters and cholesterol metabolizing enzymes in cultured human neuronal and microglial cell lines in vitro. This is a first step toward mitigating ASD-promoting effects of cholesterol dysregulation as an approach to autism prevention and treatment.
Material and methods
Cell culture
SH-SY5Y cells
SH-SY5Y human neuroblastoma cells were seeded at a density of 500,000 cells/mL in 60 mm culture plates in DMEM/F-12 medium supplemented with 10% fetal calf serum (FCS), 5% PenStrep, and 5% GlutaMAX and grown at 37°C in 5% CO2. Three days later, cells had formed neurites and were treated with VPA at 0, 250, 1000, and 5000 µM for 24 h.
HMC3
HMC3 human microglial cells were seeded at a density of 400,000 cells/mL in 60 mm culture plates in DMEM medium supplemented with 10% fetal calf serum (FCS), 5% PenStrep, and 5% GlutaMAX and grown at 37°C in 5% CO2. Three days later, cells were treated with VPA at 0, 250, 1000, and 5000 µM for 24 h or 48 h.
Real-time RT-PCR
Total RNA was isolated after 24 h of VPA treatment with the Trizol reagent and dissolved in nuclease-free water. The quantity of total RNA from each condition was measured by absorption at 260 and 280 nm wavelengths by ultraviolet spectrophotometry. cDNA was copied from 1 µg of total RNA using Murine Leukemia Virus reverse transcriptase primed with oligo dT. Equal amounts of cDNA were taken from each reverse transcription reaction mixture for real-time PCR amplification using the FastStart SYBR Green Reagents Kit according to the manufacturer’s instructions on the Roche Light Cycler 480 (Roche Applied Science, Indianapolis, IN, USA). The level of mRNA for the genes of interest (ABCA1 ABCG1, CD36, and 27-hydroxylase) was calculated in relation to the amplification level for the housekeeping gene GAPDH; the threshold cycle values were determined using the instruments software.
Protein isolation and western blotting
Protein was collected from whole cell lysates after 48 h of VPA treatment using radioimmunoprecipitation assay (RIPA) lysis buffer (98% PBS, 1% Igepal, 0.5% sodium deoxycholate, 0.1% sodium dodecyl sulfate (SDS)), supplemented with 10 μl per ml of protease inhibitor cocktail (Sigma). Protein content was measured in triplicate using the BCA Protein Assay Kit by absorption at 562 nm (Pierce Biotechnology Inc., Rockford, IL, USA). Whole cell lysate protein extracts were separated and analyzed by 10% SDS-polyacrylamide gel electrophoresis (SDS-PAGE). About 8.3 µg of each sample were loaded per gel lane and transferred to polyvinylidene fluoride membranes.
The blots were subsequently blocked with 5% NFDM in TBST for 1 h 20 min at room temperature and incubated in primary antibodies at 4°C overnight. For immunoblot analysis, proteins were probed with Abcam primary antibodies against ABCA1 (ab66217, mouse monoclonal) and ABCG1 (ab52617, rabbit monoclonal). Antibodies were diluted at 1:1000 in 5% NFDM in TBST, respectively. A 1:1000 dilution of beta actin (Cell Signaling Technologies, CST3700, mouse monoclonal) in 5% NFDM in TBST was used as a control. Bound antibodies were visualized with 1:2000 dilution of their respective horseradish peroxidase-conjugated secondary antibodies prepared in 1% NFDM in TBST. The immunoreactive protein was detected using ECL western blotting detection reagents (ThermoFisher Scientific™ SuperSignal™ West Pico PLUS Chemiluminescent Substrate and ThermoFisher Scientific™ SuperSignal™ West Femto Maximum Sensitivity Substrate) and the Bio-Rad ChemiDoc Touch Imaging System. Protein expression was normalized with respect to expression of beta actin and quantified via densitometry with ImageJ computer software (Rasband WS, National Institutes of Health, Bethesda, MD, USA, 2018).
Cholesterol transport assays
Cholesterol efflux analysis was performed on SH-SY5Y and HMC3 cells plated in 96-well plates at 1 × 106 cells/mL in supplemented DMEM-F12 or DMEM medium, respectively, in the presence or absence of VPA at concentrations of 250 µM, 1000 µM, and 5000 µM for 24 h using the Amplex Red Cholesterol Assay Kit (Invitrogen, Waltham, MA, USA) according to the manufacturer’s protocol.
Reactions performed in the presence and absence of cholesterol esterase, total (TC) and free (FC) cholesterol were analyzed. Cholesterol esters (CEs) were estimated as the difference between TC and FC, and CE/FC ratio was calculated. Fluorescence was read at 585 nm, and cholesterol efflux was expressed as percentage of fluorescence in efflux medium to total fluorescence of the cells and medium combined.
Statistical analysis
Data were evaluated using the linear models or linear mixed models with R to evaluate the effects of treatment with GAPDH or β-actin as a co-variate. Differences between treatments were evaluated by contrasts and slopes transformed and expressed as 2−ΔΔCt or fold-difference from control. Data are shown as means and standard errors for a minimum of three separate experiments. Differences where p < 0.05 are considered statistically significant.
Results
Effect of VPA exposure on expression of cholesterol transport protein in HMC3 microglia cells
In HMC3, VPA exposure for 24 h caused a concentration-dependent significant decrease in ABCA1 mRNA and an increase in ABCG1 (80-fold at the highest dose; 5000 µM) mRNA levels. VPA treatment also increased mRNA levels of CD36 in a dose-dependent manner (Figure 1(a)–(c)). Protein levels of both ABCA1 and ABCG1 were reduced after 48 h of VPA incubation (Figure (d) and (e)).
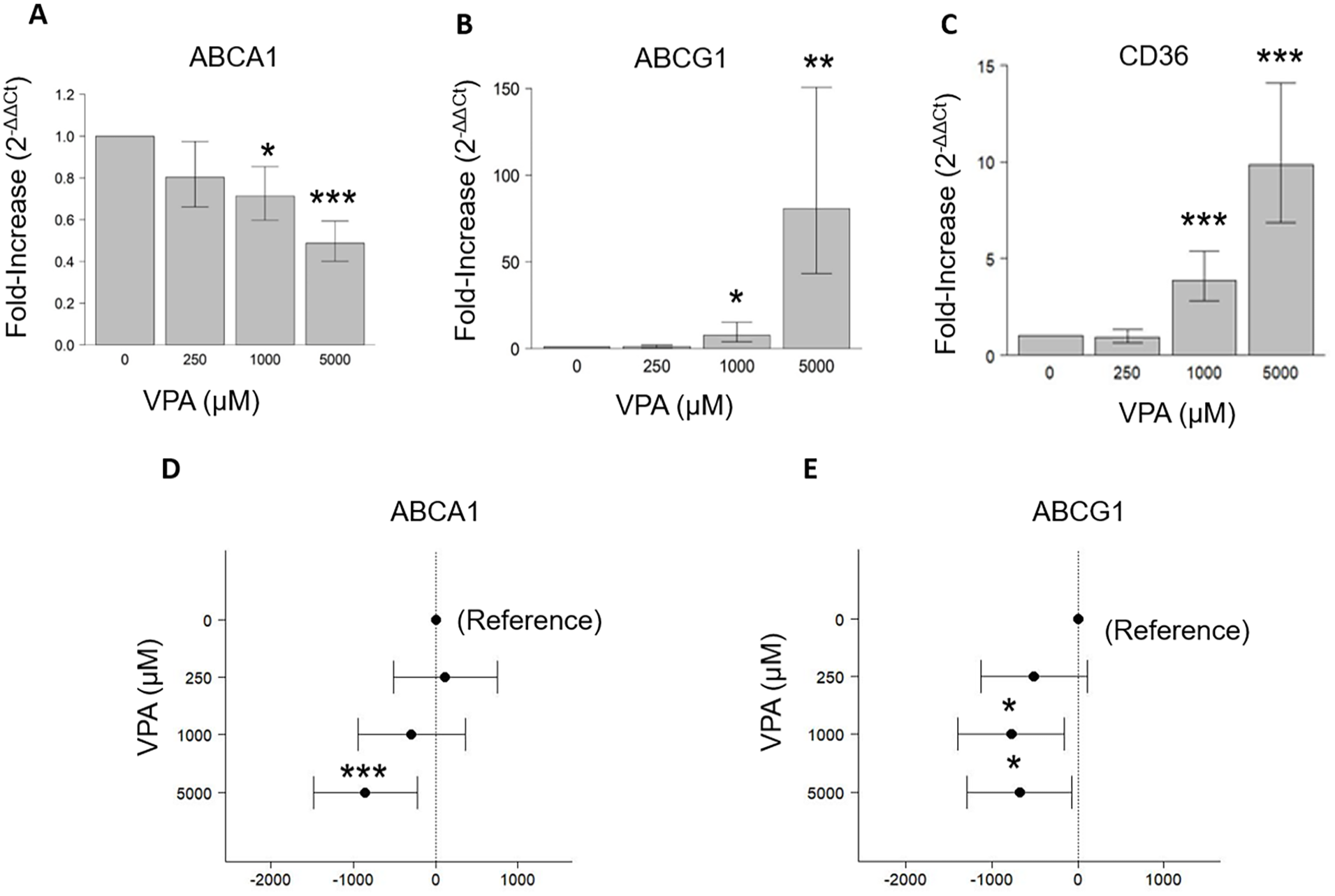
Effects of VPA on expression of cholesterol transporters in HMC3 glial cells. (a–c) Quantitative Real-time PCR analysis of ABCA1, ABCG1, and CD36 in HMC3 glial cells. GAPDH was used as an internal control. (d and e) Western blot analysis of ABCA1 and ABCG1 in HMC3 glial cells. β-Actin was used as an internal control. The data represent difference from Control (±95% CI) and verified by three independent experiments (***p < 0.001, **p < 0.01, *p < 0.05).
Changes to cholesterol transport proteins induced by VPA exposure in SH-SY5Y neurons
We replicated the same sets of experimental conditions in SH-SY5Y neuronal cells and found that exposure to VPA for 24 h significantly increased the mRNA levels of ABCA1, ABCG1, CD36, and 27-hydroxylase in a dose-dependent manner (Figure 2(a)–(d)). Western blotting, however, showed that VPA treatment for 48 h significantly reduced ABCA1 protein in a dose-dependent manner, but increased ABCG1 at the highest dose, that is, 5000 µM (Figure 2(e) and (f)).
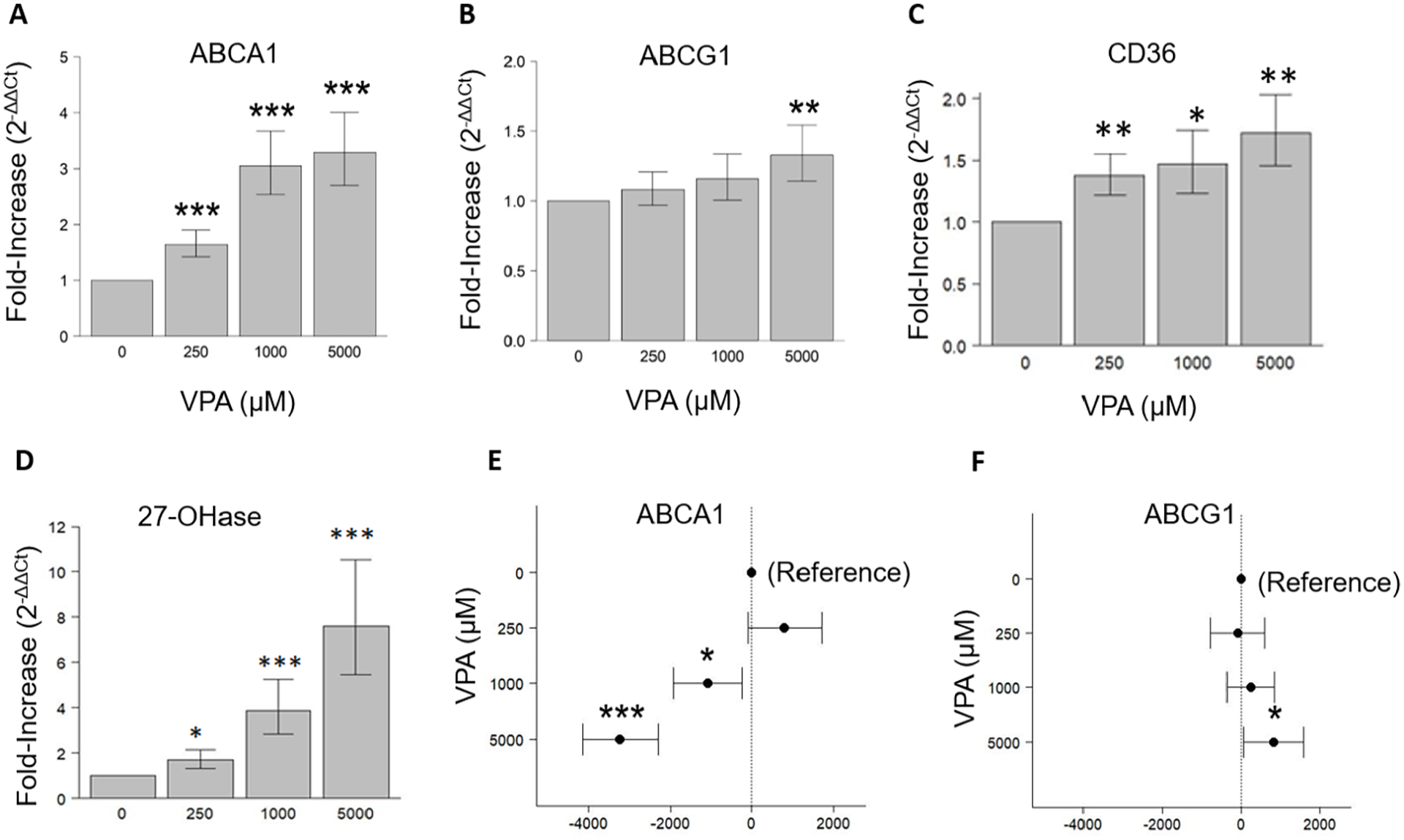
Effect of VPA on expression of cholesterol transporters in SH-SY5Y neurons. (a–d) Quantitative real-time PCR analysis of ABCA1, ABCG1, CD36, and 27-hydroxylase in SH-SY5Y cells. GAPDH was used as an internal control. (e and f) Western blot analysis of ABCA1 and ABCG1 in SH-SY5Y cells. β-Actin was used as an internal control. The data represent difference from Control (±95% CI) and verified by three independent experiments (***p < 0.001, **p < 0.01, *p < 0.05).
Cholesterol transport assays
No effect of VPA on cholesterol efflux was detected in HMC3 microglia cells; however, VPA treatment for 24 h significantly increased cholesterol efflux in SH-SY5Y cells at 250 and 1000 μM (Figure 3(a) and (b)).
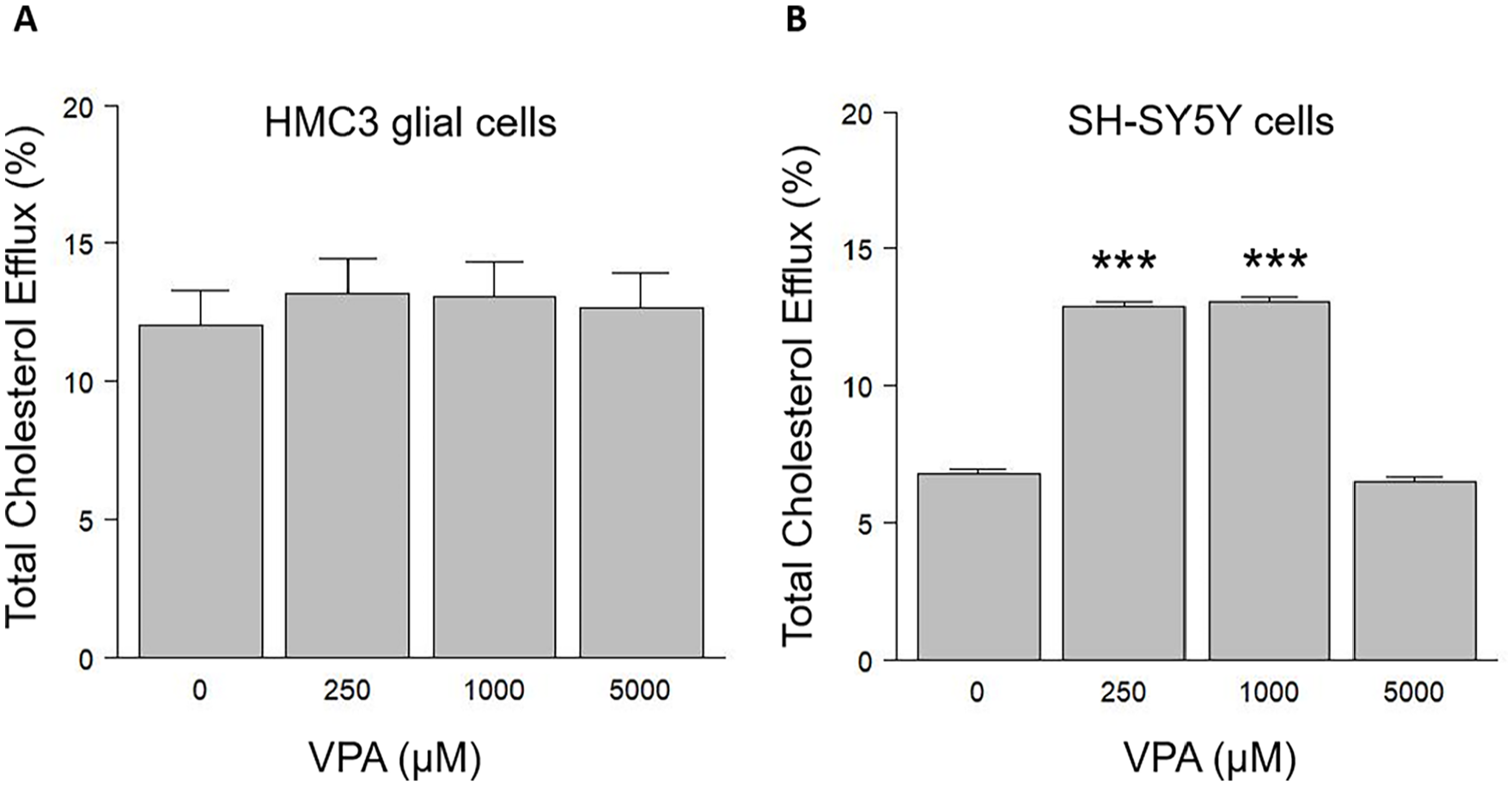
Effects of VPA on total cholesterol efflux. Cholesterol efflux was measured in (a) HMC3 glial cells, (b) SH-SY5Y cells. The results were verified by three independent experiments (***p < 0.001).
Discussion
Recent studies have suggested that low levels of low-density lipoprotein-cholesterol in maternal plasma at birth increases the risk of the development of autism. 7 Previous studies in children have also demonstrated an association between hypocholesterolemia and autism.3,5 Children born with inborn errors in cholesterol metabolism (e.g., Smith–Lemli–Opitz disorder) frequently have autistic-like behaviors.8–10 Intrauterine exposure to VPA is associated with increased risk of autism.11–16 Experimental studies with rats and mice have confirmed that VPA exposure during pregnancy results in autistic-like behaviors in the offspring.11–15 VPA has become a well-accepted animal model for studying the neurological changes associated with the condition that include hypomyelination of neurons and changes in cholesterol homeostasis in the brain. 17 Therefore, we examined the impact of VPA on cholesterol transporters and metabolizing enzymes in human neurons as well as microglia whose dysfunction/activation has also been associated with autism.18–21
We found that VPA reduced mRNA levels for ABCA1 but increased mRNA levels for ABCG1 and CD36 in a dose-dependent manner in microglia (HMC3) cells. ABCA1 and ABCG1 are both mediators of cholesterol transfer out of the cell, a process known as reverse cholesterol transport. 22 Although the reduction in ABCA1 would suggest an 80-fold increase in ABCG1, which showed a net efflux of cholesterol from HMC3 cells. However, Western blotting analysis revealed that VPA reduced protein levels slightly for both of these cholesterol transporters. Furthermore, cholesterol efflux assays detected no effect of these changes in gene and protein expression on cholesterol efflux from the microglial cells. This suggests that the observed effects of VPA on ABCA1 and ABCG1 expression are regulated at both the transcriptional as well as the translational level, but that they have no net effect on cholesterol outflow from these cells or, alternatively, some other genes may compensate for the effects of VPA in order to maintain cholesterol levels in microglia.
In the SH-SY5Y neurons, VPA increased the expression of ABCA1 mRNA, but reduced protein levels of ABCA1 in a dose-dependent manner, suggesting that VPA suppressed ABCA1 expression at the translational level, but induced it at the transcriptional level. Both mRNA and protein levels for ABCG1 increased at the highest dose (500 µM) of VPA. VPA exposure also increases mRNA levels of CD36 and 27-hydroxylase in a dose-dependent manner. Cholesterol efflux was significantly increased at 250 and 1000 µM of VPA, indicating that cholesterol efflux did not correlate with ABCG1 levels and might be regulated by the CD36 scavenger receptor and other genes.
Despite many studies demonstrating that VPA alters cholesterol levels in the brain, this is the first study, to our knowledge, where the effects of VPA on cholesterol efflux and expression of cholesterol transport proteins and metabolizing enzymes has been assessed in human neurons and microglia. Our finding that VPA reduced ABCA1 protein levels in microglia and neurons is similar to a previous report that used small cell lung carcinoma cells, except that VPA reduced both mRNA and protein levels of ABCA1. 23 Another study with HepG2 human hepatoma cells suggested that VPA significantly increased ABCG1 mRNA expression that was similar to our finding for both neurons and microglial cells. 24
The function of CD36, a well-known fatty acid translocase, is highly variable in different tissues and includes long-chain fatty acid metabolism, lipid uptake, and cholesterol efflux. 25 In the current study, VPA exposure increased CD36 expression in microglia and neurons, which is consistent with one of the previous studies of increased CD36 expression after VPA treatment in HepG2 cells, LO2 cells, and livers of C57B/6J mice (28366540, 33007364). However, the role of CD36 in VPA-induced cholesterol efflux in microglia and neurons is not fully elucidated and requires additional experiments to create a complete picture.
Apart from cholesterol transport proteins, we also found the increased mRNA level of 27-hydroxylase (CYP27A1), a cholesterol metabolizing enzyme, after VPA treatment in neurons. 27-hydroxylase converts cholesterol into 27-hydoxycholesterol, which can exit the cell easily and also enhances cholesterol efflux by upregulating cholesterol transporters, thus reducing cellular cholesterol accumulation. 26 The increased levels of 27-hydroxylase after VPA treatment in neurons is very well correlated with increased total cholesterol efflux. Furthermore, 27-hydroxycholesterol is a neurotoxic molecule.27,28
Strengths of our study include the use of well-characterized human cell lines to study the processes involved in ASD and the use of a well-established method for inducing the condition in animal models. Our findings are limited by factors inherent to in vitro studies. We are unable to determine if the observed effects of VPA on cholesterol transport and metabolism lead to reductions in myelination and synaptogenesis in the developing brain. We are also unable to evaluate how the observed changes may affect behavior. Some of these limitations may be overcome by novel cell culture models that allow myelination to be studied in vitro and in vivo. Imaging studies of infants may also be useful in this regard.
As we move forward, we also consider the close relationship between cholesterol metabolism and the production of amyloid in the brain, which has shown itself to be a critical aspect of Alzheimer’s disease.29,30 Derangements in amyloid processing may also be involved in ASD.31–34 Interestingly, VPA has been found to inhibit γ-secretase cleavage of amyloid precursor protein, reducing brain amyloid load and VPA hypocholesterolemic effects could be a mechanism contributing to its favorable impact on ASD via amyloid reduction. 35 This requires further exploration as most studies of this relationship have focused on cognitive impairment in older population and not on developmental effects on amyloid.
Conclusion
In this study, we have shown that VPA has a dramatic effect on cholesterol transport genes in two key cell types, microglia and neurons, that compose the developing brain. The net impact of the changes observed in these cholesterol-related genes would be outflow and metabolism. Furthermore, enhanced 27-hydroxylase activity produces an oxysterol metabolite with neurotoxic effects that include downregulating synaptic proteins and decreasing neurite number and length. Together, our results suggest that VPA impairs brain cholesterol homeostasis. A better understanding of the involvement of cholesterol in the mechanisms by which VPA leads to ASD may translate into innovative preventative pharmacotherapies for this serious disorder.
Footnotes
Acknowledgements
We thank Ms. Lynn Drucker, Mr. Edmonds Bafford, and Mr. Robert Buescher.
Author contributions
Conceptualization: M.R.P., A.B.R., A.P., and J.D.L.; Experiments and Methodology: M.R.P., A.B.R., H.A.R., S.A., and Y.A.; Data analysis: L.J.K. and A.S.; writing original draft: M.R.P., A.B.R., and J.B.; writing and editing: A.B.R., T.W., and A.S.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Alzheimer’s Foundation of America Award AWD00004772 and by The Herb and Evelyn Abrams Family Amyloid Research Fund. (A.B.R.), and also supported by NIH grants AG066512 and AG060882 (T.W.).
Ethical approval
Not applicable.
