Abstract
Although reports of familial clustering of hematologic malignancies have appeared for decades, the cause(s) of this uncommon occurrence is still not completely understood. Most modern investigations, however, support a genetic rather than an environmental exposure as a cause of this observation. Most pedigrees of families with familial hematologic malignancies demonstrate age of onset anticipation, with the disease diagnosed at an earlier age in successive generations. The cause of this phenomenon is clear in some familial neurologic disorders (trinucleotide repeat expansion) but not at all clear in familial hematologic malignancies. In preparation for molecular studies of familial clustering of hematologic malignancies, we have collected pedigrees on 738 families and have previously demonstrated anticipation in those with familial plasma cell myeloma, chronic lymphocytic leukemia, Hodgkin lymphoma or non-Hodgkin lymphoma (NHL). Here we present data on 36 families with both plasma cell myeloma and NHL in their pedigrees and demonstrate strong evidence for anticipation in these families. We encourage all health care personnel to ask patients multiple times about family medical history and carefully take note of family histories from individuals with uncommon illnesses and to refer families with clustering of such illnesses for further investigation.
Myeloma rarely occurs in families as does non-Hodgkin lymphoma.
Myeloma and non-Hodgkin lymphoma may occur in the same families, which suggests that the two B-cell malignancies may have a common genetic etiology.
Genetic studies of affected and unaffected members of such families need to be done and may reveal clues as to the basic nature of the two neoplasms.
Introduction
The study of familial hematologic malignancies may lead to the discovery of a gene or genes that cause such neoplasms or to a gene or genes that enhance susceptibility to an environmental agent. As previously reported, we have constructed pedigrees after collecting medical records including pathology reports and other material on 738 families with multiple hematologic malignancies. Many of the patients were referred by physicians and genetic counselors. Other patients were acquired through participation in internet patient chat rooms, and others were identified in our own practices. We 1 and others2–4 have previously reported families with multiple cases of multiple myeloma (MM). In our study, anticipation (i.e., diagnosis at an earlier age in succeeding generations) was clearly evident but this has not been the case in some other studies. 5 We have also reported on familial Hodgkin lymphoma (HL),6,7 non-Hodgkin lymphoma (NHL), 8 and chronic lymphocytic leukemia (CLL). 9 In addition, we have reported families with both MM and HL in their pedigrees 10 and have demonstrated anticipation in all of these studies. In the present study we investigated families with both plasma cell dyscrasias and NHL cases to determine whether anticipation could be demonstrated in them as well. 11
Methods
We assessed affected members of 36 families identified with NHL and a plasma cell dyscrasia for correct diagnosis, age at diagnosis, sex, generation of diagnosis, and anticipation. We included MM, monoclonal gammopathy of undetermined significance (MGUS), Waldenström’s macroglobulinemia (WM), and primary amyloidosis (AM). Anticipation was defined as a younger age at diagnosis than that of an affected individual in a previous generation.
Often it required repeated questioning of the propositus to determine whether or not other family members were in fact diagnosed with a hematologic malignancy, even when the propositus was a physician. Family history reporting by patients is often inaccurate.12–14 In the present study fewer than half of the afflicted individuals were accurately documented in medical records based on patient reporting and accurate data were only obtained upon repeated questioning by a research assistant who then acquired documentation, including physician records, pathology reports and pathology slides, which were reviewed by us.
The protocol for this study was approved initially by the local institutional review board and subsequently has been approved by the Western Investigational Review Board (IRB) (protocol # 20160881) for the past decade, and all members of each family studied gave written informed consent to participate, whether affected or not.
Results
We identified 36 families with individuals in our database (Supplemental Table 1) with NHL and one or more plasma cell dyscrasias (MM, MGUS, WM, or AM) in their pedigrees. Nineteen affected parent–child pairs were identified (Table 1). Those consisted of four mother–daughter pairs, three mother–son pairs, five father–son pairs and seven father–daughter pairs. The age at diagnosis was known for 14 of the parents (median, 65 years; range 45–90 years) and 17 of the children (median, 50 years; range 20–63 years). In each of the 13 pairs for which ages of both the parent and child were known, anticipation was evident and ranged from −4 to −60 years (median, −18 years). Six mothers had MM and one had NHL. Six of their children had NHL and one had MM. Two fathers had MM, seven had NHL, two had WM and one had amyloidosis. Five of their children had NHL, five had MM, one had WM and one had MGUS.
Parent–child pairs.
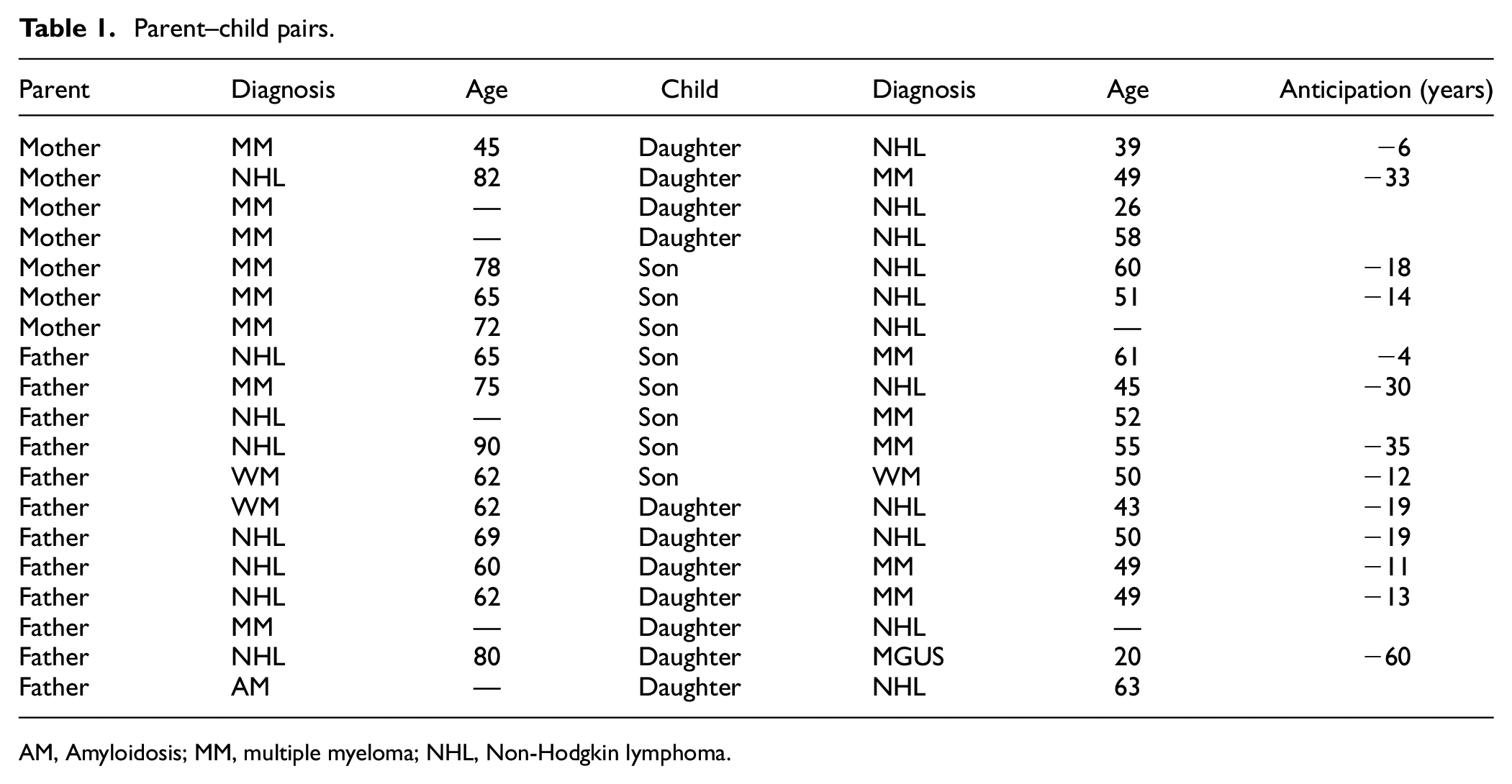
AM, Amyloidosis; MM, multiple myeloma; NHL, Non-Hodgkin lymphoma.
Twelve affected sibling pairs were found in the 36 families (Table 2). There were five brother–sister pairs (four MM–NHL pairs and one NHL–NHL pair), four brother–brother pairs (two MM–NHL pairs, one NHL–NHL pair and one WM–NHL pair) and three sister–sister pairs (one pair each of MM–NHL, NHL–WM and NHL–MGUS). In one brother–brother MM–NHL pair family, a third brother also had NHL, and in a brother–sister NHL–MM pair family, another brother had MM. In another brother–sister NHL–MM pair, the sister with MM was a half-sister and both sibs had the same father.
Affected sib pairs in the families.
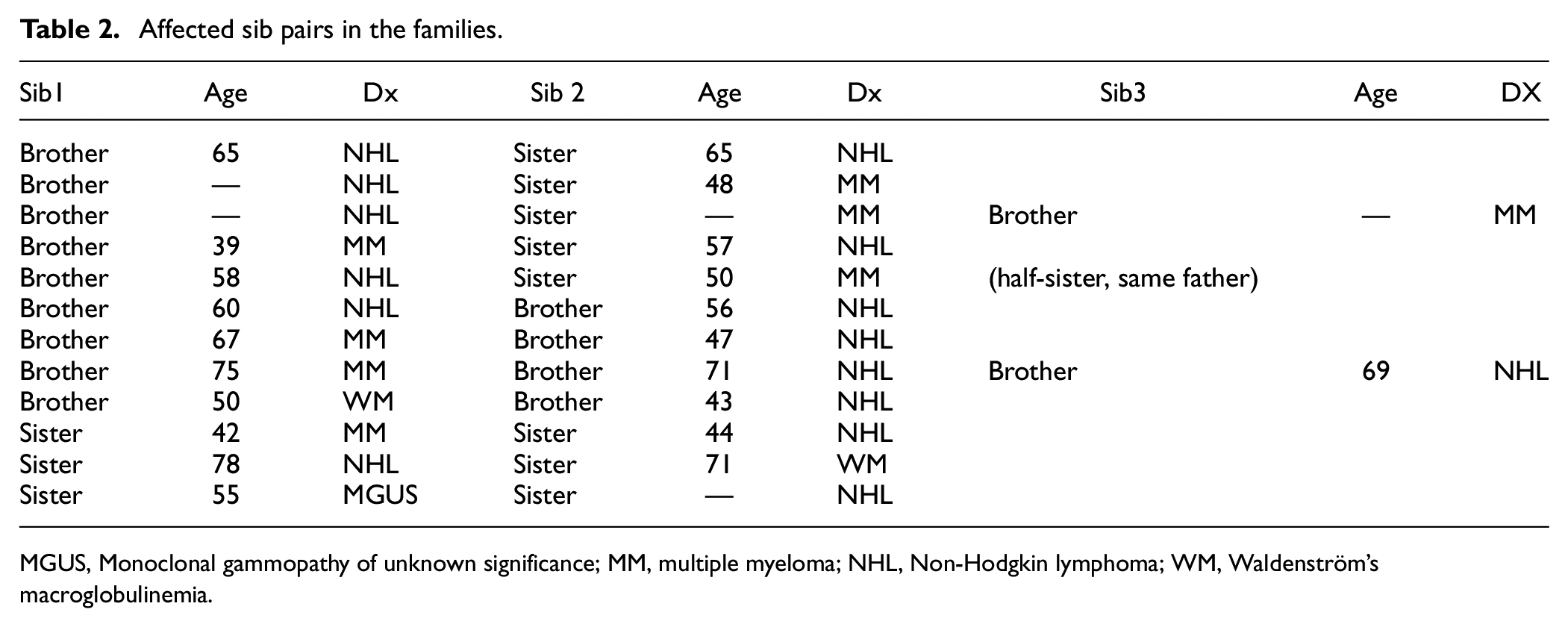
MGUS, Monoclonal gammopathy of unknown significance; MM, multiple myeloma; NHL, Non-Hodgkin lymphoma; WM, Waldenström’s macroglobulinemia.
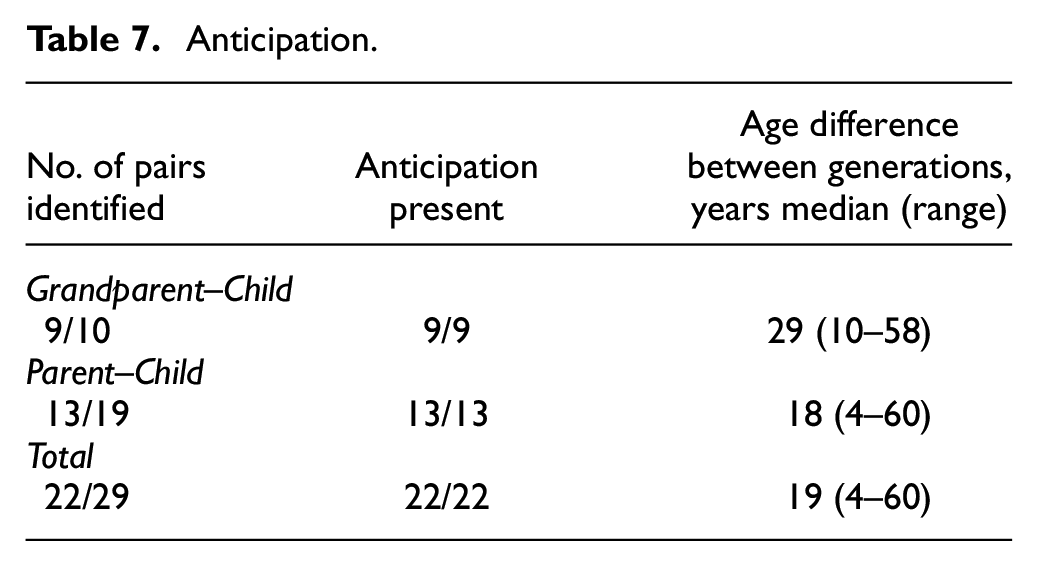
Ten affected grandparent–grandchild pairs were identified in these families (Table 3). Four were grandmother–grandson pairs in which all four grandmothers had MM. One of their grandsons also had MM and the other three had NHL. There were two grandmother–granddaughter pairs in which both grandmothers had MM as did one granddaughter. The other granddaughter had NHL. There were also three grandfather–granddaughter pairs and one grandfather–grandson pair. Two grandfathers with MM each had a granddaughter with NHL. Another grandfather had NHL and a granddaughter with MGUS. A fourth grandfather had MM and a grandson with NHL. Curiously, 9 of 10 grandparents in this family had MM and 7 of the 10 grandchildren had NHL. The age at diagnosis for 9 of 10 grandparents for whom data were available was a median of 78 years (range, 52–79 years) and that of 10 grandchildren was a median of 47 years (range, 20–60 years). Therefore, anticipation of −29 years median (range −10 to −58 years) is evident. Major anticipation was even noted in the two grandmother–grandchild pairs in which both individuals had MM (−29 and −35 years).
Grandparent–grandchild pairs.
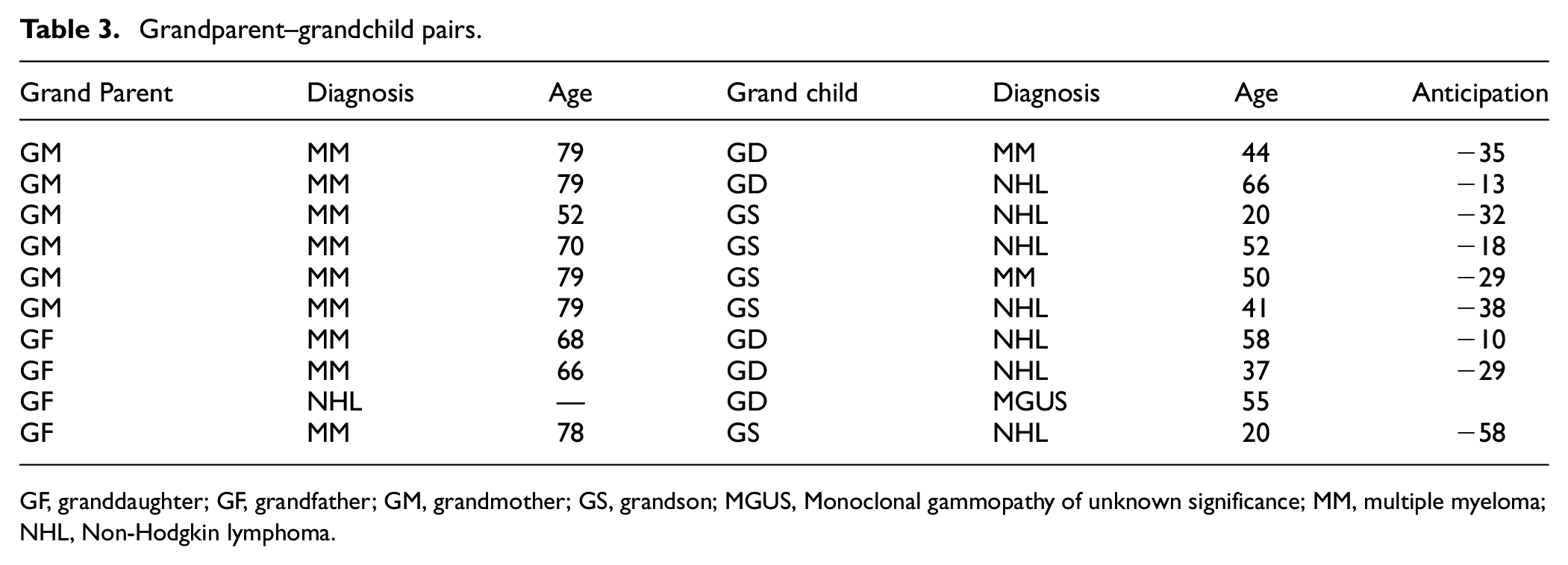
GF, granddaughter; GF, grandfather; GM, grandmother; GS, grandson; MGUS, Monoclonal gammopathy of unknown significance; MM, multiple myeloma; NHL, Non-Hodgkin lymphoma.
The diagnoses found in these families are summarized in Tables 4 and 5.
Generations affected.
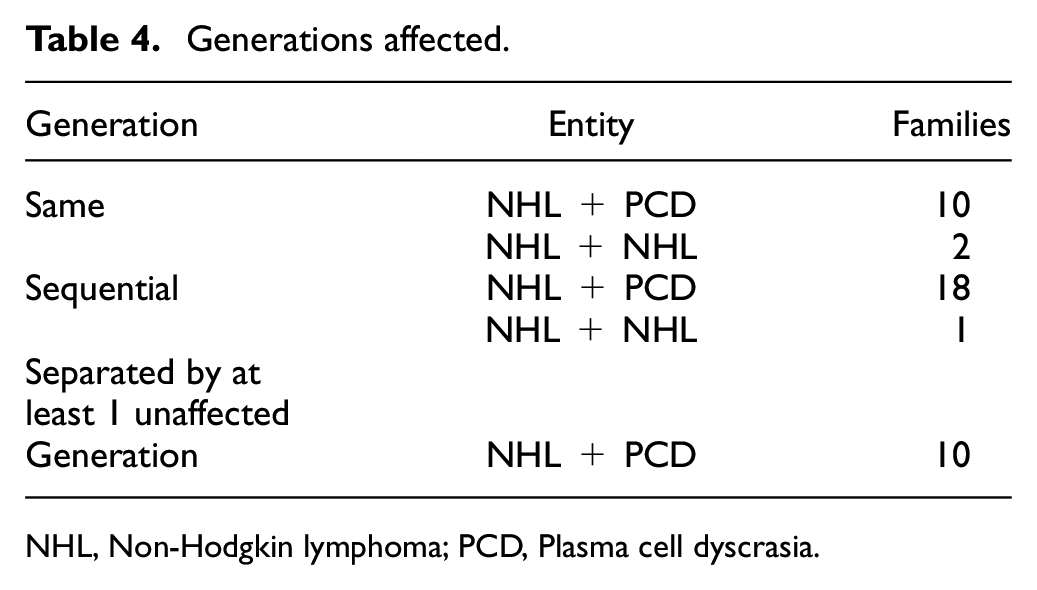
NHL, Non-Hodgkin lymphoma; PCD, Plasma cell dyscrasia.
Thirty-six families with plasma cell dyscrasias and non-Hodgkin lymphoma.

MGUS, Monoclonal gammopathy of unknown significance; MM, multiple myeloma; NHL, Non-Hodgkin lymphoma; WM, Waldenström’s macroglobulinemia.
The ages of many of the MM patients in these families is younger than might be expected (Table 6), which suggests that myeloma patients may have been present in earlier generations. The age at diagnosis of the known MM patients in these families is 63 years median, range 39–79 years and 18 of the 28 patients were <70 years of age. The median age at diagnosis for MM in general is 70 years. 15
Ages of patients in the families at diagnosis.
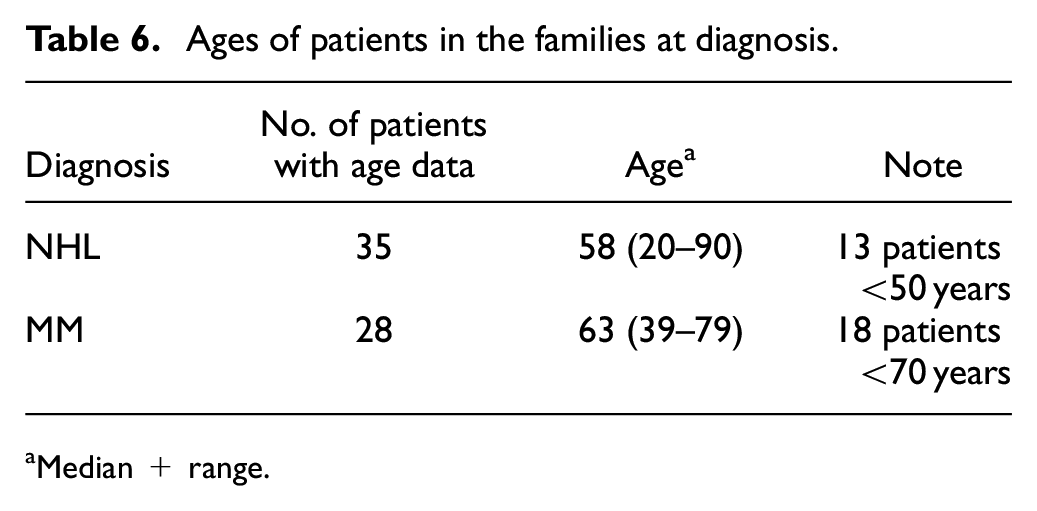
Median + range.
Discussion
Together with our previous studies of familial MM, 1 NHL, 8 HL6,7 and CLL, 9 the present study strengthens the argument that many if not all B-cell malignancies may have a common genetic etiology. Male transmission in the families in the present study was evident in 17 of the 29 multi-generational affected pairs identified, including the brother–half-sister pair in Table 2 with the same father. However, female transmission was evident in 13 affected pairs. Therefore, unlike families with MM1 and HL,6,7 we cannot confirm predominantly male transmission in the current study. Similarly, unlike familial HL, 7 it does not appear in this study that NHL-plasma dyscrasia siblings are almost always of the same sex since 5 of the 12 pairs were not.
Anticipation was observed in this study in all 22 affected pairs in which it could be assessed (Table 7). Anticipation has been ascribed to genetic factors or environmental exposure by various authors. Some have argued that a combination of genetic susceptibility to an environmental agent or exposure alone to such an agent is responsible for familial aggregation of diseases such as those studied here. 16 In other studies, however, others have concluded that environmental factors are likely not at play here. 17 We found no basis for implicating common environmental exposure in the families reported here. Individuals in these families have lived all over the USA in rural and urban areas.
Anticipation.
Anticipation in a number of familial neurologic disorders has been associated with expansion of unstable trinucleotide repeats passed down through succeeding generations,18,19 but this has been demonstrated not to be the case in familial leukemia. 20 At present, the molecular basis for anticipation in hematologic malignancies is unknown. It seems unlikely, however, that it is a statistical aberration based on presumed infertility 19 in patients with those neoplasms. A change in incidence over time of the diseases studied as an explanation for anticipation5,21 observed in this study also seems unlikely since the incidence of MM has remained stable for decades. 22
This is the largest study to date of families with MM and NHL in their pedigrees. However, others have reported that MM aggregates in families with NHL 21 and that risk for NHL is higher when a male relative has MM. 23 Chang et al. reported that having a first-degree relative with any hematopoietic malignancy was associated with an increased risk of NHL, and that a family history of MM was significantly associated with an increased risk of follicular NHL. 24 Lu et al. 25 also reported an increased incidence of NHL in families with a history of hematopoietic malignancy but did not comment specifically on families with myeloma patients, as have others. 26
This study suggests that familial MM and NHL may have genetic etiological features in common.
Other evidence that B-cell malignancies may share a common etiology includes the facts that there is an increased risk of CLL in first degree relatives of MM or MGUS patients. 27 Furthermore, MM and CLL have a common feature in that healthy members of MM families may have subclinical evidence of MM27 and healthy family members of CLL patients may have subclinical characteristics of CLL. 28 Further evidence of a common etiology is the observation that CLL may be converted into MM by the administration of an immune modulating agent. 29 While little is known about the etiology of these hematologic malignancies, progress in uncovering that knowledge is being made.30,31 Studies of families such as those presented here may facilitate that progress.
Conclusion
Families with both NHL and plasma cell dyscrasias in their pedigrees may be more common than previously recognized. In such families, anticipation as seen in other familial B-cell malignancies can be demonstrated. This finding suggests a genetic cause for these neoplasms in these families and a genetic relationship between NHL and plasma cell dyscrasias.
Supplemental Material
sj-docx-1-imj-10.1177_10815589231210516 – Supplemental material for Families with non-Hodgkin lymphoma and plasma cell dyscrasias in their pedigrees
Supplemental material, sj-docx-1-imj-10.1177_10815589231210516 for Families with non-Hodgkin lymphoma and plasma cell dyscrasias in their pedigrees by Peter H Wiernik and Janice P Dutcher in Journal of Investigative Medicine
Footnotes
Acknowledgements
Presented in part at the 2016 Eastern Division of the American Federation for Medical Research annual meeting. Supported in part by the Cancer Research Foundation of New York. We acknowledge the tireless efforts of our research associates, Patricia Marino, Kelly Brown, and Dona Wickramasinghe, who over more than a decade obtained and confirmed the clinical data collected for this paper.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
