Abstract
Atrial fibrillation (AF) frequently occurs concurrently with heart failure (HF). The two conditions can exacerbate each other, resulting in higher morbidity and mortality. In our analysis, we evaluated mortality trends related to AF in individuals with underlying HF. Cross-sectional analyses were performed using publicly available data from the Center for Disease Control and Prevention database to compare AF-related age-adjusted mortality rates across age, gender, racial/ethnic, and geographic subgroups. Mortality trends were evaluated by fitting log-linear regression models followed by calculation of the average annual percentage change (AAPC) using the Monte Carlo permutation test. We identified a total of 55,917 deaths within the United States from AF with comorbid HF between 1999 and 2020. Males, older adults, White populations, and non-metropolitan regions had higher age-adjusted mortality compared to females, younger adults, Black populations, and metropolitan regions, respectively. The AAPC among younger adults was significantly higher compared to older adults. Our results demonstrate existing disparities among age, gender, racial, and geographic subgroups related to AF mortality among individuals with comorbid HF. Although decreased overall mortality was observed within younger populations compared to older populations, the prominent AAPC seen in younger populations warrants further investigation. Detection of AF among younger adults with comorbid HF should prompt the intensification of preventative and treatment measures.
Introduction
Atrial fibrillation (AF) is the most common arrhythmia in the United States (US), often coexisting with heart failure (HF). These conditions work in tandem, precipitating one another, leading to increased morbidity and mortality. Population-based exploratory analyses reveal existing disparities and warrant future targeted research, promoting healthcare equity. Therefore, we sought to quantify mortality trends from AF in individuals with comorbid HF.
Methods
This study uses data from the CDC Wide-Ranging Online Data for Epidemiologic Research database to capture all death certificate information through the National Vital Statistics System. 1 The underlying cause of death is defined by the disease process that led directly or initiated the sequence of events to death, and the multiple causes of death are defined as the diseases that contributed to death. All diagnoses are based on the International Classification of Diseases, Tenth Revision (ICD10). All mortality data that included atrial fibrillation/flutter (ICD10 code: I48) as the underlying cause of death in patients with HF (ICD10 code: I50) as the multiple causes of death were queried from 1999 to 2020. Age below 35 and above 85 years were excluded given the likelihood of congenital heart disease and poor diagnostic reporting reliability, respectively. 2 Data were quantified as age-adjusted mortality rate (AAMR) per 100,000 population, average annual percentage change (AAPC), and respective 95% confidence intervals (CIs). AAMR was adjusted to the US population in the year 2000 using the direct method which entails applying age-specific rates to a standardized age distribution within a population of interest. This approach effectively eliminates disparities in observed rates that arise due to variations in the age distribution among different populations. Subgroup analyses were completed using age, gender, race, and geographic information. To examine yearly mortality patterns, we employed Joinpoint Regression (National Cancer Institute) to determine noteworthy shifts in age-adjusted mortality. Using log-linear regression models, this regression analysis can detect joinpoints, or points of inflection, in the mortality trends. To calculate the annual percentage change, we used the Monte Carlo permutation test, and we then determined the AAPC by computing weighted averages of the annual percentage change. Data visualization was completed using Stata (StataCorp LLC, College Station, TX 77845, USA). Institutional review board approval was not required as the data collected were publicly available and de-identified.
Results
There were a total of 55,917 AF-related deaths in the United States in individuals with comorbid HF between the years 1999 and 2020. All inflection points are depicted in Supplemental Table S1. AAMR increased from 0.61 (95% CI, 0.58–0.65) in 1999 to 2.73 (95% CI, 2.67–2.81) in 2020 with an AAPC of 8.3% (95% CI, 6.8–9.9, p < 0.001). AAMR was higher in males (1.78 (95% CI, 1.76–1.80)) compared to females (1.48 (95% CI, 1.46–1.50)), but AAPC was similar (+9.2% (95% CI, 7.7–10.8, p < 0.001) and +7.5% (95% CI, 5.9–9.1, p < 0.001), respectively). AAMR was greater in older (65–84 years) adults (6.61 (95% CI, 6.55–6.66)) compared to younger (35–64 years) adults (0.15 (95% CI, 0.14–0.15)). AAPC was significantly higher among younger adults (14.1% (95% CI, 12.4–15.7, p < 0.001)) compared to older adults (+7.9% (95% CI, 6.3–9.5, p < 0.001); Figure 1(a) and (b)). AAMR was greater in White adults (1.67 (95% CI, 1.66–1.69)) compared to Black adults (1.46 (95% CI, 1.43–1.51)); however, AAPC was similar (7.5% (95% CI, 5.3–9.7, p < 0.001) and 7.8% (95% CI, 6.9–8.7, p < 0.001), respectively; Figure 2(a)). Black male and White male adults had a similar AAMR (1.80 (95% CI, 1.73–1.87) and 1.81 (95% CI, 1.79–1.83), respectively). White female adults had a higher AAMR compared to Black female adults (1.51 (95% CI, 1.50–1.53) and 1.23 (95% CI 1.18–1.28), respectively; Figure 2(b)). Non-metropolitan (1.91 (95% CI, 1.87–1.94)) regions had a higher AAMR compared to metropolitan regions (1.53 (95% CI, 1.52–1.55)) with a similar AAPC (+8.1% (95% CI, 5.0–11.3, p < 0.001) and +8.2% (95% CI, 6.5–9.8, p < 0.001), respectively). AAMR was similar in the West (1.67 (95% CI, 1.64–1.70)), Midwest (1.67 (95% CI, 1.64–1.70)), and South regions (1.65 (95% CI, 1.63–1.67)), but was the lowest in the Northeast (1.37 (95% CI, 1.35–1.40)) region. The AAPC among these four regions were statistically similar (+8.4% (95% CI, 3.7–13.3, p < 0.001), +7.5% (95% CI, 5.0–10.1, p < 0.001), +7.7% (95% CI, 6.9–8.5, p < 0.001), and +6.5% (95% CI, 5.1–7.9, p < 0.001), respectively).
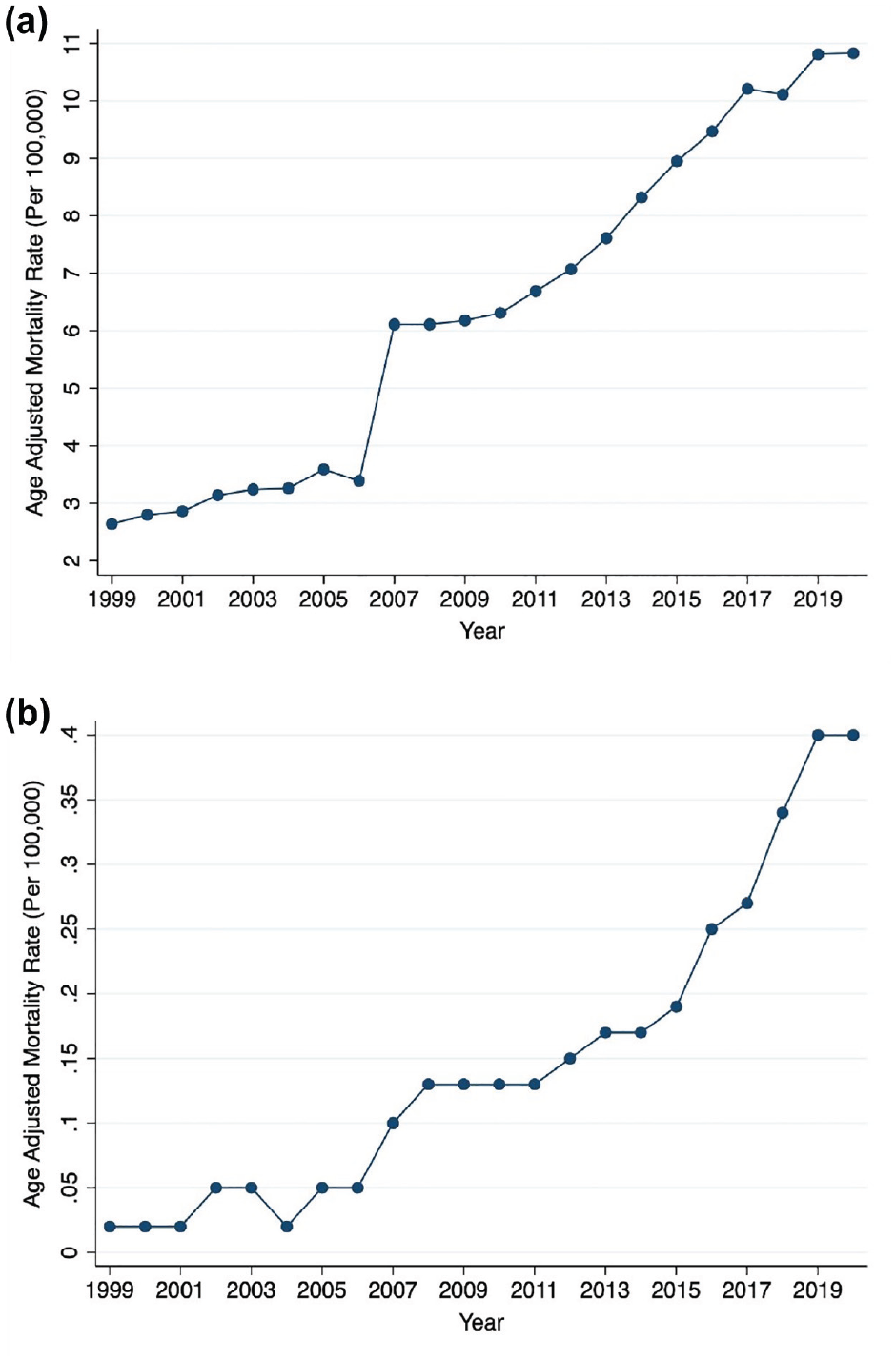
Yearly mortality trends from 1999 to 2020 by age groups: (a) older adults (65–84 years) and (b) younger adults (35–64 years).
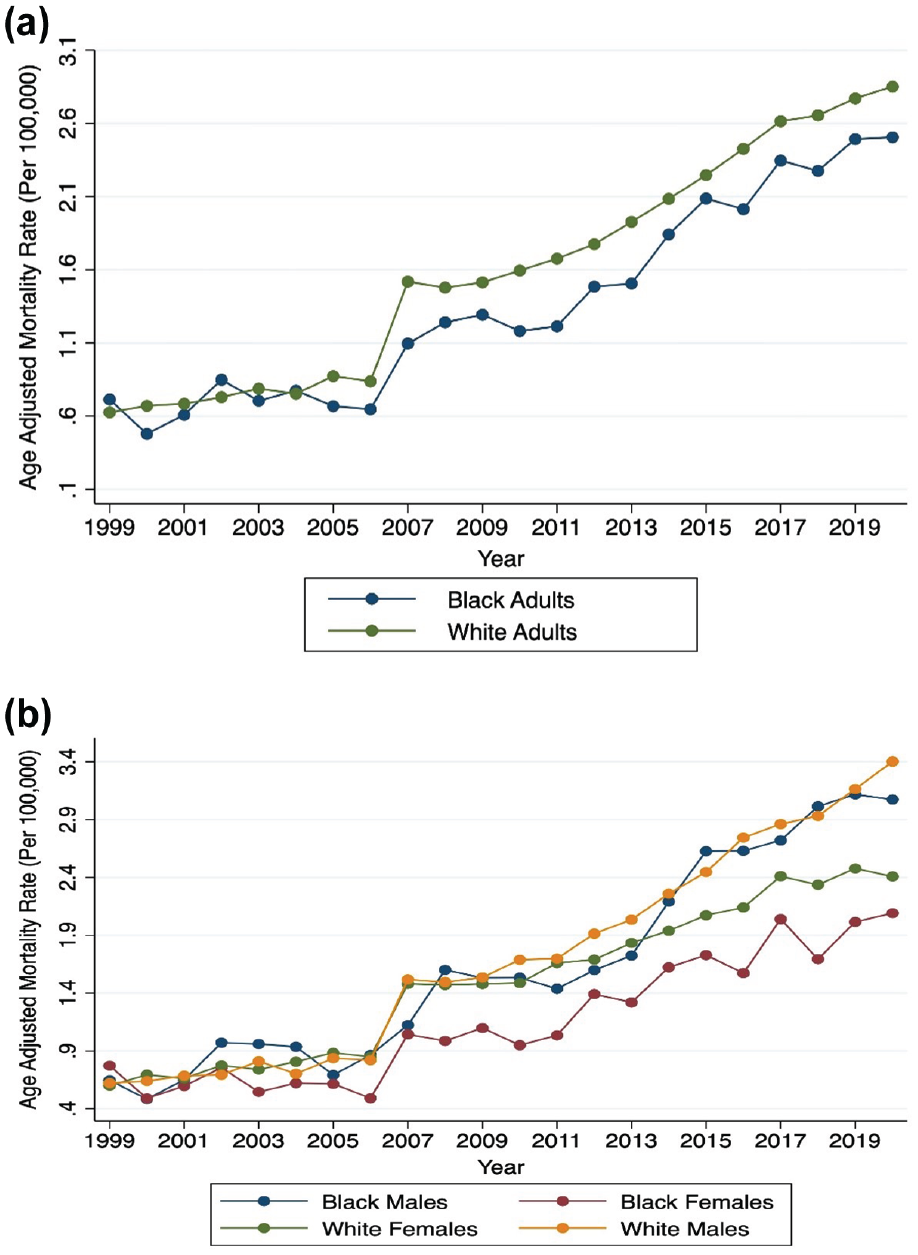
Yearly mortality trends from 1999 to 2020 by (a) race (Black and White adults) and (b) race and gender (Black and White adults, males and females).
Discussion
Our analysis identified that (1) overall AF-related AAMR in patients with HF increased from 1999 to 2020, (2) it disproportionately affected males, White adults, and in non-metropolitan regions, (3) and the increase in mortality among younger adults was almost double the rate of acceleration compared to older adults.
The extent to which AF contributes directly to mortality is unclear, but AF could be the primary cause of decompensation in a patient with HF especially when it leads to rapid ventricular rates and loss of atrial contraction. With an increasing prevalence of AF coupled with the increased aging of the population and associated risk factors, an increase in AF-related mortality is likely to be seen. 3 Mortality was higher among males compared to females in our results, consistent with previous studies that identified an overall greater prevalence of AF and HF in males. 4 These findings are likely attributable to the prevalence of cardiovascular disease in males and sex-specific susceptibility to atrial arrhythmias. 5 Cumulative AAMR was higher among older populations; however, accelerating trends in mortality were more pronounced in younger adults. This is consistent with a previous analysis that identified a prominent increase in mortality among younger adults related to AF without pre-specified HF. 6 An increase in midlife cardiovascular mortality and associated risk factors has been previously identified, congruent with our analysis.7,8 Detecting AF among younger populations and its prognostic implications remain less well understood. As screening modalities become increasingly utilized, further investigation into triaging those at greater risk of mortality is warranted.
Although we have identified greater mortality rates among White adults compared to Black adults, previous studies have been contradictory. One study revealed higher mortality among Black populations, likely a result of decreased anticoagulation use and subsequent strokes. 9 Another study revealed similar mortality rates among Black and White populations. 10 These findings may be confounded by racial differences in the sensitivity of methods to detect AF. 11 Further research is warranted to identify, if any, racial differences in mortality related to AF with comorbid HF. Mortality variability among geographic subgroups is likely impacted by social determinants of health and access to healthcare providers. 12
We have discovered a notable turning point in our analysis, occurring around the years 2007 to 2009, which is evident in the cumulative population as well as among various subpopulations. These findings align with previous studies examining hospitalizations and mortality related to cardiovascular diseases and HF. Specifically, there was a significant increase in the occurrence of HF and hospitalizations among younger adults during this period. 13 This surge can be attributed to multiple factors, such as changes in the reporting of mortality data, the expanded utilization of electronic health records for HF diagnosis, the implementation of programs aimed at reducing readmissions, and a rise in risk factors associated with cardiovascular diseases. 14
Limitations
Limitations to our analysis include the use of death certificates to identify causes of mortality, which may be subjected to inaccuracies. In addition, covariates such as characterization of AF vs flutter, hemodynamic status, rate vs rhythm control utility, AF burden, and anticoagulation were not available in this database.
Conclusion
In conclusion, we identified an increasing rate of AF-related AAMR in patients with HF and significant disparities among subgroups. As detection of AF among younger adults becomes common, comorbid HF should warrant concern for risk factor modification and intensification of care.
Supplemental Material
sj-docx-1-imj-10.1177_10815589231208478 – Supplemental material for Atrial fibrillation mortality trends in individuals with heart failure
Supplemental material, sj-docx-1-imj-10.1177_10815589231208478 for Atrial fibrillation mortality trends in individuals with heart failure by Ramzi Ibrahim, Vikram J Singh, Simar J Singh, Ayman Hussein and Justin Z Lee in Journal of Investigative Medicine
Footnotes
Authors’ note
All authors take responsibility for all aspects of the reliability and freedom from bias of the data presented and their discussed interpretation
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval was not required given the data is publicly available and de-identified.
Data availability
All data are publicly available in the CDC WONDER Database.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
