Abstract
This study intended to delineate the mechanism and functional role of integrin α2 (ITGA2) in non-small-cell lung cancer (NSCLC) cell immune escape. Bioinformatics analysis was utilized to analyze ITGA2 expression in NSCLC tissues, and correlations between ITGA2 expression and patient survival time, ITGA2 expression and programmed cell death ligand 1 (PD-L1; CD274) expression, and ITGA2 expression and CD8+ T-cell infiltration. Quantitative real-time polymerase chain reaction detected ITGA2 expression. Transmission electron microscopy was applied to examine the morphology of exosomes, and western blot measured CD9, CD63, and PD-L1 levels. CCK-8 measured cell viability. Cell toxicity experiment measured the killing effect of CD8+ T cells on cancer cells. Enzyme-linked immunosorbent assay assessed secretion levels of interleukin-2, interferon-gamma, tumor necrosis factor-alpha, and PD-L1 expression in exosomes. Immunohistochemistry detected ITGA2, CD8, and PD-L1 expression in patient tissue samples. ITGA2 was highly expressed in NSCLC, and Pearson correlation analysis showed a negative correlation of ITGA2 with CD8+ T-cell infiltration and a positive correlation of ITGA2 with PD-L1 expression. Cell experiments showed that silencing ITGA2 hindered NSCLC cell progression and increased levels of CD8+ T-cell secretory factors. Further mechanism studies found that ITGA2 reduced CD8+ T-cell-mediated antitumor immunity via the increase in PD-L1 expression. Clinical sample testing unveiled that ITGA2 was upregulated in NSCLC tissues. PD-L1 upregulation was seen in exosomes separated from patient blood, and correlation analysis showed a positive correlation of exosomal PD-L1 expression in blood with ITGA2 expression in tissues. This study displays a novel mechanism and role of ITGA2 in NSCLC immune escape, providing directions for the clinical therapy of NSCLC patients.
Non-small-cell lung cancer (NSCLC) is a serious health problem with high mortality and unfavorable prognosis.
Immunotherapy has good clinical efficacy in NSCLC, but immune escape hinders its effectiveness. Integrin α2 (ITGA2) has been demonstrated to be associated with the malignant progression of various cancers.
ITGA2 repressed CD8+ T-cell activity and promoted immune escape in NSCLC.
ITGA2 inhibited CD8+ T-cell activity by enhancing programmed cell death ligand 1 (PD-L1) expression in exosomes.
Clinical evidence showed that exosomal PD-L1 expression in the blood of NSCLC patients was positively correlated with ITGA2 expression in tissues.
Mechanistic investigations unraveled that ITGA2 overexpression promoted exosomal PD-L1 expression in NSCLC, repressed the immune response of T cells, and mediated immune escape in NSCLC.
A positive correlation of ITGA2 expression with PD-L1 expression was seen in NSCLC tissue samples.
We obtained tissue and blood samples from NSCLC patients to determine the clinical relevance of the results.
Introduction
Non-small-cell lung cancer (NSCLC) is the most common subtype of lung cancer cases. NSCLC patients are usually diagnosed in the late stages of the disease ascribable to the late onset of clinical symptoms and shortage of screening tools. 1 Currently, treatment advances in surgery, radiotherapy, chemotherapy, targeted therapy, and immunotherapy have brought an increase in NSCLC survival rates. 2 However, treatment deficiencies, high postoperative recurrence rates, dismal prognosis, and low patient survival rates make NSCLC treatment status pessimistic. 3 In addition, immune escape is the main obstacle to improving cure rates. 4 Immune escape of tumor cells can be induced by lack of co-stimulatory antigens on tumor cells, reduction of cytotoxic immune cells, increase in immune suppressor cells, and increase in immune inhibitory factors in the tumor microenvironment.5–7 Hence, the immune escape mechanism of NSCLC needs to be addressed to improve the clinical efficacy of NSCLC treatment.
Immune escape occurs when tumor cells increase their growth rate and spread to other parts of the body while avoiding detection and destruction by immune cells. 8 Cancer immunotherapy targeting the programmed cell death protein 1 (PD-1) and programmed cell death ligand 1 (PD-L1) pathways has opened the era of modern oncology, and drugs that block PD-1 or PD-L1 can promote endogenous antitumor immunity. 9 Immune checkpoint inhibitors targeting PD-1 and PD-L1 pathways achieve certain effects in NSCLC patients. 10 However, over 50% of treated patients have a poor response to immunotherapy. Previous reports mentioned that upregulated exosomal PD-L1 is a contributor to low immunotherapy response. 11 The molecular regulatory mechanisms affecting PD-L1 expression in NSCLC cell-derived exosomes are not yet clear. Hence, understanding molecular mechanisms that affect tumor immune escape is of high essence to improve patient treatment outcomes.
The role of integrin α2 (ITGA2), a subunit of the collagen receptor ITGA2/β-1, 12 in cancer is largely researched, such as ITGA2 overexpression promoting proliferation and invasion through FAK/AKT signaling in esophageal squamous cell carcinoma (ESCC). 13 ITGA2 overexpression upregulates PD-L1 level through STAT3 signaling, thus promoting proliferation and invasion of pancreatic cancer cells. 14 Chen et al. 15 unraveled that ITGA2 overexpression and low methylation of the promoter region promoted the invasive and migratory abilities of thyroid cancer cells. However, the regulatory effect of ITGA2 through exosomal PD-L1 on tumor immunity in NSCLC is still unclear. Therefore, we investigated the molecular mechanism by which ITGA2 modulated the antitumor immune function of T cells via exosomal PD-L1 and provided a reference for novel immunotherapy regimens for NSCLC patients.
Here, we analyzed ITGA2 and exosomal PD-L1 expression in NSCLC and the effects on the immune escape of NSCLC cells. We tested the correlation of exosomal PD-L1 of blood from NSCLC patients with ITGA2 expression in tumor tissues. In conclusion, we confirmed molecular mechanisms by which ITGA2 modulated the antitumor immune function of CD8+ T cells by enhancing exosomal PD-L1 expression, and our results lay a theoretical basis for immunotherapy in NSCLC patients.
Materials and methods
Patient selection
We recruited 20 patients with NSCLC who received anti-PD-L1 therapy at Xuzhou Central Hospital from June 30, 2021 to August 31, 2022. Tumor tissue and blood samples were collected from patients (including 12 cases of lung adenocarcinoma and 8 cases of lung squamous cell carcinoma). In addition, blood samples were obtained from 10 healthy donors at the hospital’s physical examination center. Lung tumor histology was classified by the 2015 WHO classification system. This study was approved by the local ethics committee of the hospital. Patients were informed about the necessary information regarding this study and provided written informed consent.
Bioinformatics analysis
NSCLC mRNA expression data were obtained from The Cancer Genome Atlas (TCGA) and normalized using the edgeR package. CD8+ T-cell immune infiltration data of TCGA samples were from TIMER. T-tests were used to perform a differential analysis of target genes. Pearson correlation analysis analyzed correlations of target genes with PD-L1 expression and CD8+ T-cell immune infiltration.
Cell culture
Human NSCLC (A549, H1299, and H460) cell lines and normal bronchial epithelial (BEAS-2B) cell lines were accessed from the BeNa Culture Collection (BNCC, Beijing, China). BEAS-2B cells were maintained in Dulbecco’s Modified Eagle Medium high-glucose (DMEM-H) (Thermo Fisher, Waltham, Massachusetts, USA) containing 10% fetal bovine serum (FBS, Vivacell, USA) and 1% penicillin/streptomycin (Thermo Fisher, Waltham, Massachusetts, USA); A549 cells were maintained in F12k medium (Thermo Fisher, Waltham, Massachusetts, USA) containing 10% FBS and 1% penicillin/streptomycin (Thermo Fisher, Waltham, Massachusetts, USA); H1299 and H460 cells were maintained in RPMI-1640 medium (Thermo Fisher, Waltham, Massachusetts, USA) containing 10% FBS and 1% penicillin/streptomycin (Thermo Fisher, Waltham, Massachusetts, USA). All cells were maintained in a 37°C incubator with 5% CO2. 16
Cell transfection
oe-NC and oe-ITGA2, si-ITGA2, and si-NC were synthesized by GenePharma (Shanghai, China). Lipofectamine 2000 (Invitrogen, Carlsbad, California, USA) was applied for transfection. Transfection efficiency was assessed 24 h post-transfection, and subsequent studies were conducted accordingly.
Quantitative real-time-PCR
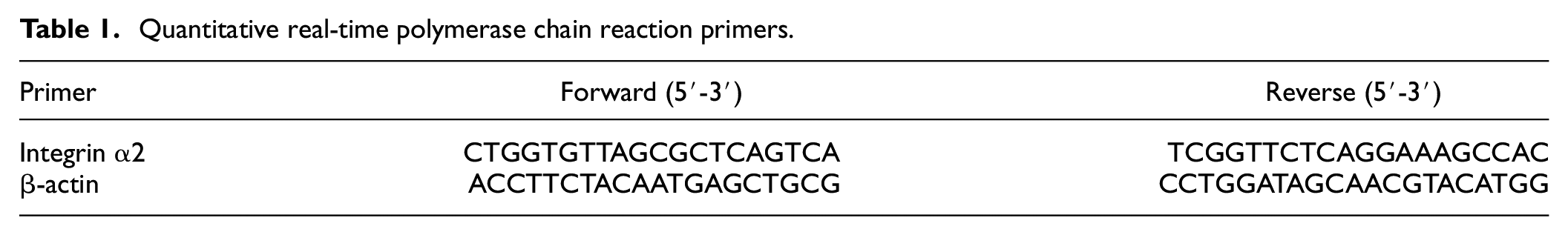
Total RNA was isolated from cultured NSCLC cells with Trizol (TaKaRa, Kyoto, Japan). cDNA was synthesized with the PrimeScript RT kit (TaKaRa, Kyoto, Japan) and stored at −20°C. Quantitative real-time polymerase chain reaction (qRT-PCR) was performed with SYBR Green PCR Master Mix (TaKaRa, Kyoto, Japan) on an ABI 7500 real-time PCR system (Applied Biosystems, Carlsbad, California, USA) to determine the expression of target genes and β-actin. Each analysis was performed in triplicate, and β-actin was an internal reference gene. Gene expression was calculated by the 2−△△CT method. The primers are shown in Table 1. 13
Quantitative real-time polymerase chain reaction primers.
Western blot analysis
Total proteins were obtained from cells with RIPA buffer (Thermo Fisher Scientific, Waltham, Massachusetts, USA). Protein concentration was assessed with a BCA protein assay kit (Thermo Fisher Scientific, Waltham, Massachusetts, USA). Protein lysates were separated by sodium dodecyl-sulfate polyacrylamide gel electrophoresis and then transferred to the polyvinylidene fluoride membrane. The membrane was incubated with primary antibodies rabbit anti-CD9 (1:1000, ab236630, Abcam, Cambridge, UK), CD63 (1:1000, ab134045, Abcam, Cambridge, UK), PD-L1 (1:1000, ab213524, Abcam, Cambridge, UK), and β-actin (1:10,000, ab124964, Abcam, Cambridge, UK) overnight, followed by adding secondary antibody goat anti-rabbit IgG H&L (HRP) (ab205718, 1:20,000, Abcam, Cambridge, UK) at room temperature for 1 h. Protein bands were analyzed after adding an enhanced chemiluminescence-based substrate (Thermo Fisher, Waltham, Massachusetts, USA). 17
CD8+ T-cell isolation and activation
CD8+ T cells were isolated from peripheral blood mononuclear cells (BNCC, Beijing, China) with CD8+ T Cell Isolation Kit (Novoabio, Beijing, China) and were seeded into a 24-well plate and cocultured with 2.5 μg/mL CD3 and 2 μg/mL CD28 antibodies for 48 h to stimulate the isolated CD8+ T cells and induce their activation.
Isolation of exosomes
After two times of phosphate-buffered saline (PBS) rinses, cells were grown in a serum-free RPMI-1640 medium. Following 48 h of incubation, the medium was centrifuged at 300g for 10 min, 2000g for 10 min, and 10,000g for 30 min at 4°C to completely remove cellular debris. The supernatant was centrifuged at 100,000g for 70 min at 4°C. The pellet was rinsed with PBS, centrifuged at high speed, and resuspended in PBS. Patients’ blood samples were thawed and centrifuged in the same way. The isolated exosomes were quantified with a protein assay kit (Bio-Rad, Hercules, California,USA) and stored at −80°C. 18
Coculture of isolated exosomes and activated CD8+ T cells
Exosomes derived from NSCLC cells were added to a 12-well plate and cocultured with pre-activated CD8+ T cells. The plate was then incubated for 24 h and used for subsequent experiments. 19
Coculture of tumor cells and CD8+ T cells
CD8+ T cells were resuspended in a cell culture medium (1 × 106 cells/ml). For all coculture systems, CD8+ T cells were added to a 96-well plate with tumor cells at a ratio of 10:1 (E:T). The 96-well plate was subject to centrifugation and incubation for 18 h.
Transmission electron microscopy
The isolated exosomes were suspended in PBS. The suspension was placed on copper grids immersed in a 0.125% Formvar chloroform solution and negatively stained with uranyl acetate. The images were photographed under transmission electron microscopy (TEM). 17
CCK-8 assay
Transfected cells (3000 cells/well) were plated in 96-well plates. Following 0, 24, 48, 72, and 96 h of culture, 10 μL of CCK-8 reagent (Dojindo, Kyushu, Japan) was added, and plates were kept away from light at 37°C for 2 h. The absorbance value at 450 nm was measured for each well with a microplate reader (Rayto, Shenzhen, China). The experiment was repeated three times.
Enzyme-linked immunosorbent assay
Interferon-gamma (IFN-γ), tumor necrosis factor-alpha (TNFα), interleukin-2 (IL-2), and PD-L1 levels were assessed with the enzyme-linked immunosorbent assay (ELISA) kit (Abcam, Cambridge, UK). Briefly, the supernatant from each coculture system was collected. Hundred microliters of enzyme conjugate was added to each sample well, and plates were maintained at 37°C for 30 min. Hundred microliters of the chromogenic substrate was added to each sample well, and plates were kept at 37°C for 20 min. Fifty microliters of stop solution was added to each sample well, and absorbance at 450 nm was measured using a microplate reader within 20 min. 20
CD8+ T-cell cytotoxicity assay
CD8+ T-cell cytotoxicity was assessed with a Lactate Dehydrogenase Cytotoxicity Detection Kit (Thermo Fisher, Waltham, Massachusetts, USA). The formula for calculating cytotoxicity was as follows: cytotoxicity = [1 − (ODcase − ODeffector cells)/ODtarget cells] × 100%. 20
Immunohistochemistry
Paraffin-embedded tumor samples were sectioned and deparaffinized, with antigen retrieval and treatment with 3% hydrogen peroxide to remove peroxidase activity. After sealing with 5% goat serum, the sections were incubated overnight at 4°C with ITGA2, CD8, PD-L1, and Ki67 antibodies (Abcam, Cambridge, UK). The sections were then maintained at 37°C for 1 h with goat anti-mouse IgG H&L secondary antibody (Abcam, Cambridge, UK). Finally, sections were rinsed with washing buffer (Beyotime, Shanghai, China) at room temperature and visualized with a DAB kit (Solarbio, DA1010, Beijing, China). The sections were observed under a microscope.
Statistical analysis
All data were processed on GraphPad Prism 8.0 (GraphPad, San Diego, California, USA). Differences between the two groups were assayed by t-tests, and one-way analysis of variance was employed for multiple comparisons. Pearson’s coefficient analyzed correlations. Each experiment was done at least three times and presented as mean ± SD. A difference with a p-value < 0.05 is considered significant.
Results
Expression of ITGA2 in NSCLC
To investigate ITGA2 expression in NSCLC, we conducted a bioinformatics analysis, which showed that ITGA2 was noticeably upregulated in NSCLC tissues but not in adjacent normal tissues (Figure 1(a)). ITGA2 level was significantly positively associated with the T stage in NSCLC patients (Figure 1(b–e)). Correlation analysis between ITGA2 level and survival time of NSCLC patients unveiled that increased ITGA2 was related to shorter overall survival time (Figure 1(f)). Subsequent bioinformatics analysis unveiled a positive correlation of ITGA2 with CD274 (PD-L1) (Figure 1(g)), and a negative correlation of ITGA2 level with CD8+ T-cell infiltration (Figure 1(h)). As revealed by qRT-PCR, ITGA2 was substantially upregulated in NSCLC cells compared to BEAS-2B cells. In NSCLC cells, ITGA2 was highly expressed in the A549 cell line and expressed at a lower level in the H460 cell line (Figure 1(i)). Therefore, we used these two cell lines for subsequent experiments. Thus, high ITGA2 expression was implicated in unfavorable prognosis in NSCLC patients.
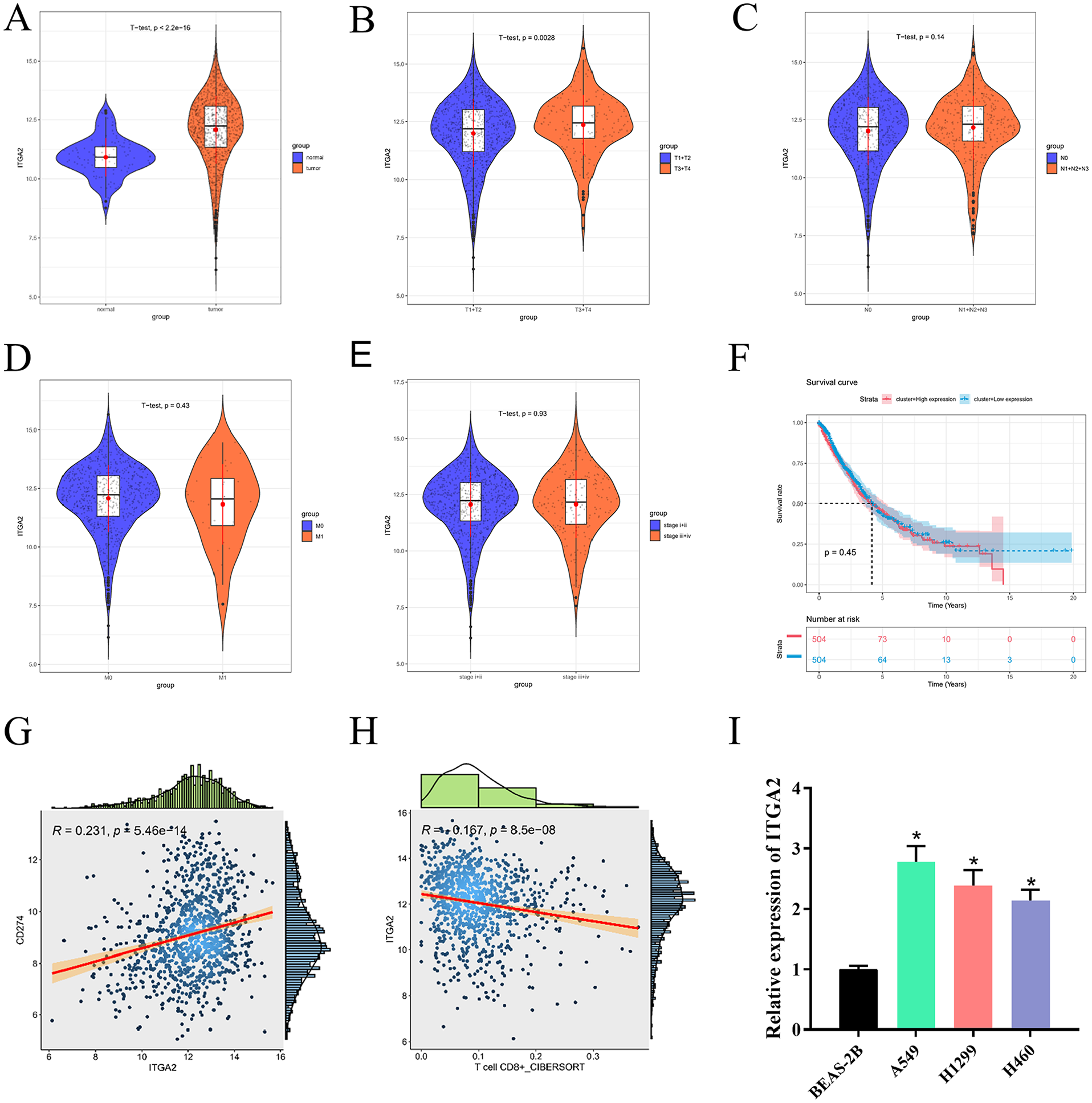
ITGA2 expression in NSCLC: (a) ITGA2 expression in NSCLC tissues compared to adjacent normal tissues, (b–e) correlation of ITGA2 expression with different stages of NSCLC tumors, (f) correlation of ITGA2 expression with survival time of NSCLC patients, (g) correlation of ITGA2 with CD274 (PD-L1), (h) correlation of ITGA2 expression with CD8+ T-cell infiltration, and (i) ITGA2 expression in NSCLC cells and normal lung epithelial cells.
ITGA2 upregulates exosomal PD-L1 and modulates T-cell activity
Overexpression of ITGA2 can enhance PD-L1 levels in pancreatic cancer cells by transcription, thereby promoting cancer cell invasion. 14 Exosomal PD-L1 induces immune dysfunction in cancer cells. 21 Therefore, it is speculated that ITGA2 in NSCLC can affect T-cell immune function by manipulating exosomal PD-L1 levels. In this study, stable transfections of oe-NC/oe-ITGA2 and si-ITGA2/si-NC were constructed in NSCLC cells, and qRT-PCR assessed ITGA2 expression in H460 and A549 cells that had undergone transfection treatment. ITGA2 expression in A549 cells was reduced when ITGA2 was silenced in A549 cells, while ITGA2 overexpression significantly increased ITGA2 expression in H460 cells, indicating successful transfection (Figure 2(a)). Cell viability was assayed by CCK-8. ITGA2 silencing decreased A549 cell viability, but ITGA2 overexpression stimulated H460 cell viability (Figure 2(b) and (c)). Exosomes were extracted and identified from the above treatment groups. TEM was implemented to observe the morphology of exosomes and vesicles with sizes between 50 and 100 nm, and similar morphological characteristics to exosomes were identified (Figure 2(d)). Western blot assessed exosome markers CD9 and CD63. ITGA2 silencing downregulated CD9 and CD63 in exosomes in A549 cells, while ITGA2 overexpression increased the expression of CD9 and CD63 in exosomes in H460 cells (Figure 2(e)), indicating that ITGA2 could affect the secretion of exosomes. Further western blot analysis confirmed that ITGA2 overexpression significantly increased exosomal PD-L1 level, while ITGA2 silencing significantly decreased exosomal PD-L1 level (Figure 2(f)). Coculturing of exosomes and CD8+ T cells showed that exosomes secreted from H460 cells overexpressing ITGA2 reduced the activity of CD8+ T cells, as demonstrated by CCK-8 assay. On the other hand, exosomes secreted from A549 cells with ITGA2 knockdown enhanced the activity of CD8+ T cells. These results indicate that ITGA2 plays a crucial role in regulating the immune activity of CD8+ T cells through exosome secretion (Figure 2(g) and (h)). Activated CD8+ T cells were then cocultured with NSCLC cells from different transfection groups. CD8+ T-cell killing ability was substantially reduced against NSCLC cells overexpressing ITGA2, while it was significantly increased against tumor cells with knocked down ITGA2 (Figure 2(i)). ELISA assayed the expression of cytokines (IFN-γ, IL-2, and TNF-α) secreted by cells in the supernatant of the coculture system. oe-ITGA2 decreased secretion levels of IFN-γ, IL-2, and TNF-α, while si-ITGA2 significantly increased their secretion levels (Figure 2(j)). Thus, ITGA2 upregulated exosomal PD-L1 expression and reduced T-cell activity.
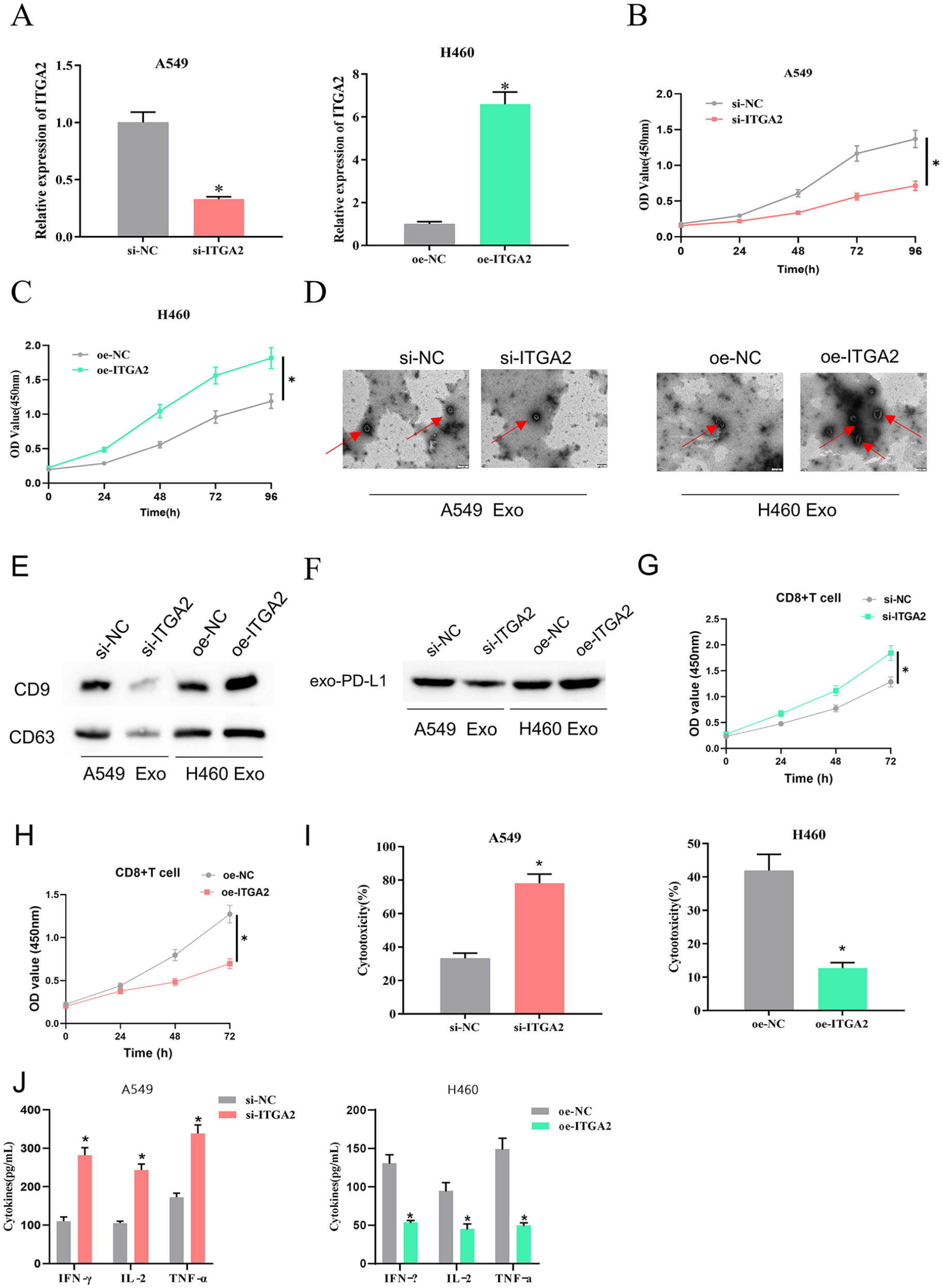
ITGA2 upregulates exosomal PD-L1 expression and modulates T-cell activity. (a) Expression of ITGA2 was determined by qRT-PCR, (b) and (c) cell viability was assessed using CCK-8, (d) exosome morphology was observed by TEM, (e) exosomal markers such as CD9 and CD63 were assayed through western blot, (f) expression of exosomal PD-L1 was measured by western blot after transfection, (g) and (h) CCK-8 detection of cell viability of CD8+ T cells cocultured with extracellular vesicles secreted by NSCLC cells, (i) lactate dehydrogenase cytotoxicity assay, and (j) secretion levels of IL-2, IFN-γ, and TNFα were tested through ELISA.
Investigation of correlation of ITGA2 expression with PD-L1 levels in samples from cancer patients
We recruited 20 NSCLC patients who were eligible for anti-PD-L1 therapy and collected tumor tissue and blood samples from these patients. We used immunohistochemistry to assess ITGA2, PD-L1, and CD8 levels in tumor tissues of NSCLC patients with high and low ITGA2 expression. We found that in NSCLC patients with high ITGA2 expression (we selected three patients with the highest ITGA2 expression for the presentation of the results), PD-L1 expression was generally higher in the tumor tissues, but CD8 expression was lower in the tumor tissues. By contrast, in NSCLC patients with low ITGA2 expression (we selected three patients with the lowest ITGA2 expression for the presentation of the results), PD-L1 expression presented a decreasing trend and CD8 expression showed an increasing trend (Figure 3(a)). Therefore, we further analyzed the correlation between ITGA2, PD-L1, and CD8 expression in all patients. ITGA2 expression in NSCLC tissue was notably negatively correlated with CD8 expression (Figure 3(b)) and markedly positively correlated with PD-L1 expression (Figure 3(c)). To determine exosomal PD-L1 levels in the blood of NSCLC patients, we first isolated and identified exosomes. The size of the vesicles from peripheral blood of NSCLC patients and healthy individuals was between 30 and 150 nm, and they had similar morphological characteristics to exosomes, as observed by TEM (Figure 3(d)). Subsequently, western blot detection showed that levels of exosome markers (CD9 and CD63) were higher in NSCLC patients, indicating that there were more exosomes in the blood of NSCLC patients (Figure 3(e)). ELISA detection presented that exosomal PD-L1 of the blood NSCLC was upregulated in NSCLC (Figure 3(f)). After our analysis, we found that ITGA2 expression in NSCLC tumor tissues was positively correlated with PD-L1 level in exosomes of the blood (Figure 3(g)).
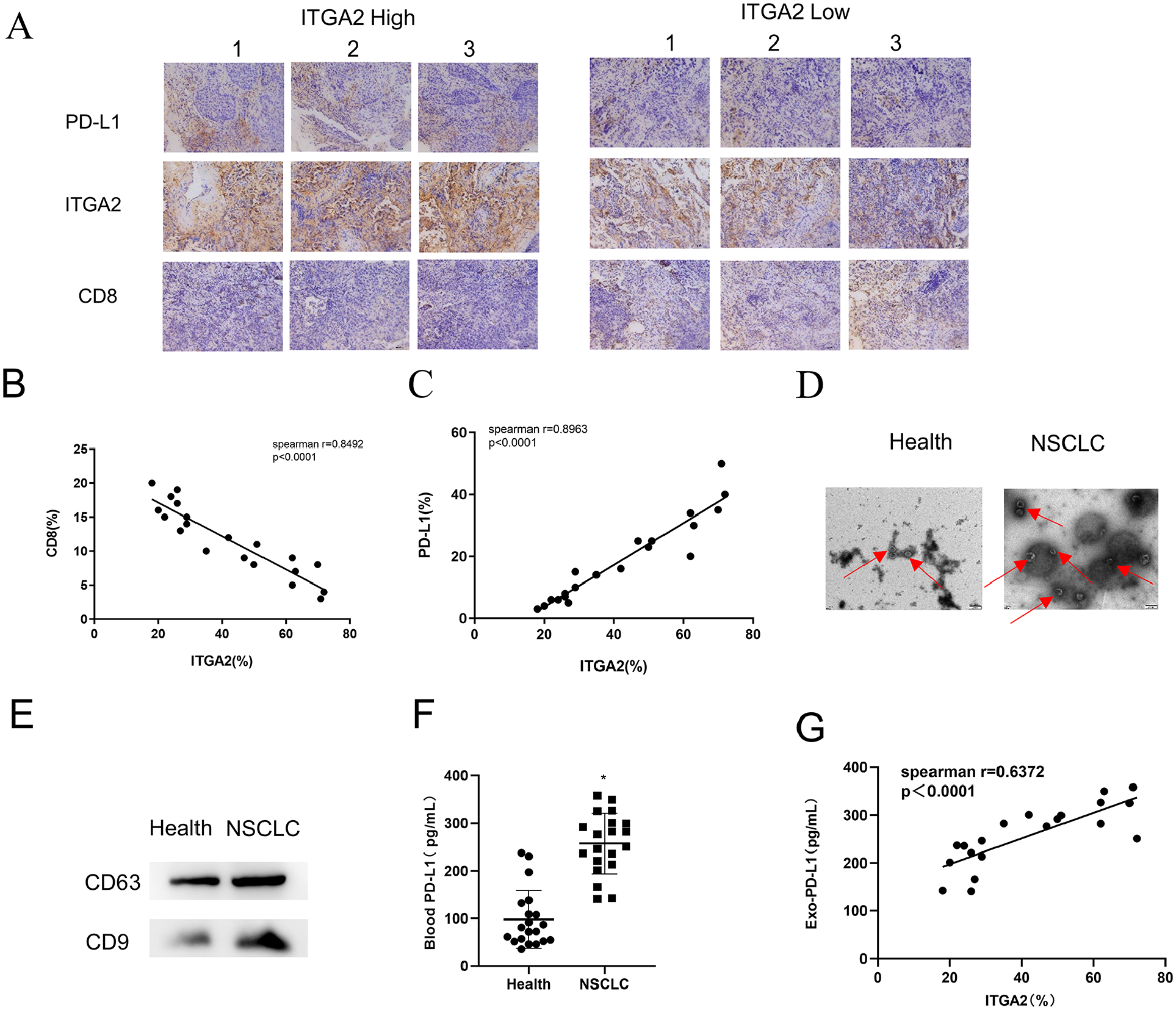
Investigation of the correlation between ITGA2 expression and PD-L1 levels in samples from cancer patients: (a) ITGA2, PD-L1, and CD8 expression in tumor tissues of NSCLC patients with high and low expression of ITGA2, as determined by immunohistochemistry analysis (selected the three patients with the highest and lowest expression levels for presentation, respectively), (b) correlation between ITGA2 and CD8 expression in NSCLC tissues, (c) correlation between ITGA2 and PD-L1 expression in NSCLC tissues, (d) detection of exosomes by TEM, (e) expression levels of exosome markers (CD9 and CD63) in NSCLC patients, as tested by western blot, (f) detection of PD-L1 expression level in exosomes, as determined by ELISA, (g) correlation between ITGA2 expression in NSCLC tumor tissues and exosomal PD-L1 expression level in the blood.
Discussion
Significant progress has been achieved in the screening, diagnosis, and treatment of NSCLC, resulting in improved patient outcomes. 22 However, the majority of advanced NSCLC patients still develop immune escape to current therapies, leading to eventual death. Abnormal expression of PD-L1 on tumor cells in NSCLC hinders antitumor immunity and triggers immune escape. 23 Molecular mechanisms underlying immune escape in NSCLC remain poorly understood. We tended to uncover the novel molecular mechanism of immune escape in NSCLC, providing potential targets for blocking NSCLC immune escape in clinical practice.
As revealed in this study, ITGA2 was upregulated in NSCLC presenting a negative correlation with CD8+ T-cell immune infiltration. Recent studies have also reported that ITGA2 plays an important part in modulating tumor progression. In ESCC, ITGA2 drives ESCC cell malignant phenotype through the FAK/AKT signaling pathway. 13 In gastric cancer, ITGA2 shows a noticeably increased level, and silencing ITGA2 can inhibit gastric cancer cell migration and induce apoptosis. 24 This study found that silencing ITGA2 could increase CD8+ T-cell activity, thus suppressing NSCLC immune escape. Therefore, we believe that ITGA2 is crucial in the treatment of NSCLC.
Exosomal PD-L1 level may be implicated in immune dysfunction and tumor malignant progression. Endoplasmic reticulum stress facilitates the release of exosomal PD-L1, promoting M2 macrophage polarization in head and neck cancer cells. 25 In NSCLC, exosomal PD-L1 induces immune escape and tumor growth by reducing the production of cytokines in CD8+ T cells. 18 Similarly, our study observed high exo-PD-L1 expression in NSCLC patients’ blood, and cell experiments demonstrated that ITGA2 could promote exosomal PD-L1 expression in NSCLC, thus hampering CD8+ T-cell activity. In conclusion, our data present a new molecular mechanism of ITGA2 mediating tumor immune escape in NSCLC, which may help to reveal new strategies and potential approaches for NSCLC immunotherapy.
Overall, a significant upregulation of ITGA2 and PD-L1 was seen in NSCLC. Mechanistic investigations unraveled that ITGA2 overexpression promoted exosomal PD-L1 expression in NSCLC, repressed the immune response of T cells, and mediated immune escape in NSCLC. In addition, a positive correlation of ITGA2 expression with PD-L1 expression was seen in NSCLC tissue samples. Hence, combined anti-PD-L1 and ITGA2-targeting therapy may improve the efficacy of NSCLC treatment. Our experiments were not limited to cellular investigations, as we obtained tissue and blood samples from NSCLC patients to determine the clinical relevance of the results. However, our study has not yet explored the upstream regulatory molecules of ITGA2, and deeper investigation is warranted to make a more precise and comprehensive explanation of ITGA2 function in NSCLC progression. Our results lay a fundamental basis for exploring novel therapeutic approaches to improve immunotherapy for NSCLC.
Footnotes
Author contribution
HJ and MM conceived of the study, participated in its design and interpretation, and helped to draft the manuscript. YM W and YY participated in the design and interpretation of the data and drafting/revising the manuscript. MJ Y and JH Z performed the statistical analysis and revised the manuscript critically. SA L, KL, and XB C participated in its design and interpretation and helped to revise the manuscript critically. All the authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Exploring the value of blood exosome derived PD-L1 in the diagnosis of stage IIIB-IV NSCLC patients and predicting drug response. Xuzhou Science and Technology Innovation Project, Project number: KC21167.
Ethics approval and consent to participate
The study was approved by the ethics committee of Xuzhou Central Hospital (XZXY-LK-20210922-028) and the Animal Ethics Committee of Xuzhou Medical University (202208W023).
Data availability statement
The data and materials in the current study are available from the corresponding author upon reasonable request.
