Abstract
The FK506-binding protein (FKBP5) plays significant roles in mediating stress responses by interacting with glucocorticoids, participating in adipogenesis, and influencing various cellular pathways throughout the body. In this review, we described the potential role of FKBP5 in the pathogenesis of two common chronic liver diseases, metabolic dysfunction-associated steatotic liver disease (MASLD), and alcohol-associated liver disease (ALD). We provided an overview of the FK-binding protein family and elucidated their roles in cellular stress responses, metabolic diseases, and adipogenesis. We explored how FKBP5 may mechanistically influence the pathogenesis of MASLD and ALD and provided insights for further investigation into the role of FKBP5 in these two diseases.
Introduction
Intracellular homeostasis plays a crucial role in regulating the stress response and maintains a dynamic equilibrium in the face of internal or external challenges.
1
These mechanisms involve a complex physiological response that encompasses multiple genetic factors and enables the sensing, integration, and adaptation to environmental changes.
1
FK506-binding protein 51 (FKBP51 or FKBP5), an important modulator of stress responses, critically contributes to the maintenance of metabolic homeostasis by playing pivotal roles in glucose and lipid metabolism.
2
Dysfunction in FKBP5 and their signaling pathways are known contributors to the development of metabolic diseases. Studies in humans have reported associations between FKBP5 and specific disorders, including major depressive disorder, asthma, obesity, and type 2 diabetes (T2D).3–5 The tissue distribution of the
MASLD and ALD are the most common causes of chronic liver diseases.10,11 An increase in the prevalence and incidence of both liver diseases contributed to a rise in the epidemic of obesity and alcohol consumption.12–14 MASLD is closely associated with obesity and the presence of metabolic syndromes in the absence of excessive alcohol use.15–17 A subset of patients with MASLD may develop a more advanced disease, metabolic dysfunction-associated steatohepatitis (MASH), a condition that can progress to advanced fibrosis and cirrhosis.18,19 ALD is an adverse consequence secondary to excessive alcohol use. ALD comprises a spectrum of histopathological changes in patients with excessive alcohol consumption, ranging from alcoholic steatosis, steatohepatitis, advanced fibrosis, and cirrhosis.20,21 Alcoholic steatosis occurs in most if not all patients who consume alcohol excessively; however, the progression of ALD to advanced stages such as alcoholic cirrhosis only develops in 15%–20% of excessive drinkers.22,23 In both MASLD and ALD, once the diseases progress to decompensated cirrhosis with complications of portal hypertension, 24 the overall prognosis and survival are poor. 25
While the specific pathways of FKBP5 in mediating the pathogenesis of MASLD and ALD are not fully elucidated, preliminary studies have indicated a potential association. In this review, we explored and described the involvement of FKBP5 in these liver diseases to gain insights into their complex mechanisms.
Overview of FKBP5 and its role in cellular stress responses
FKBP5 is encoded by the
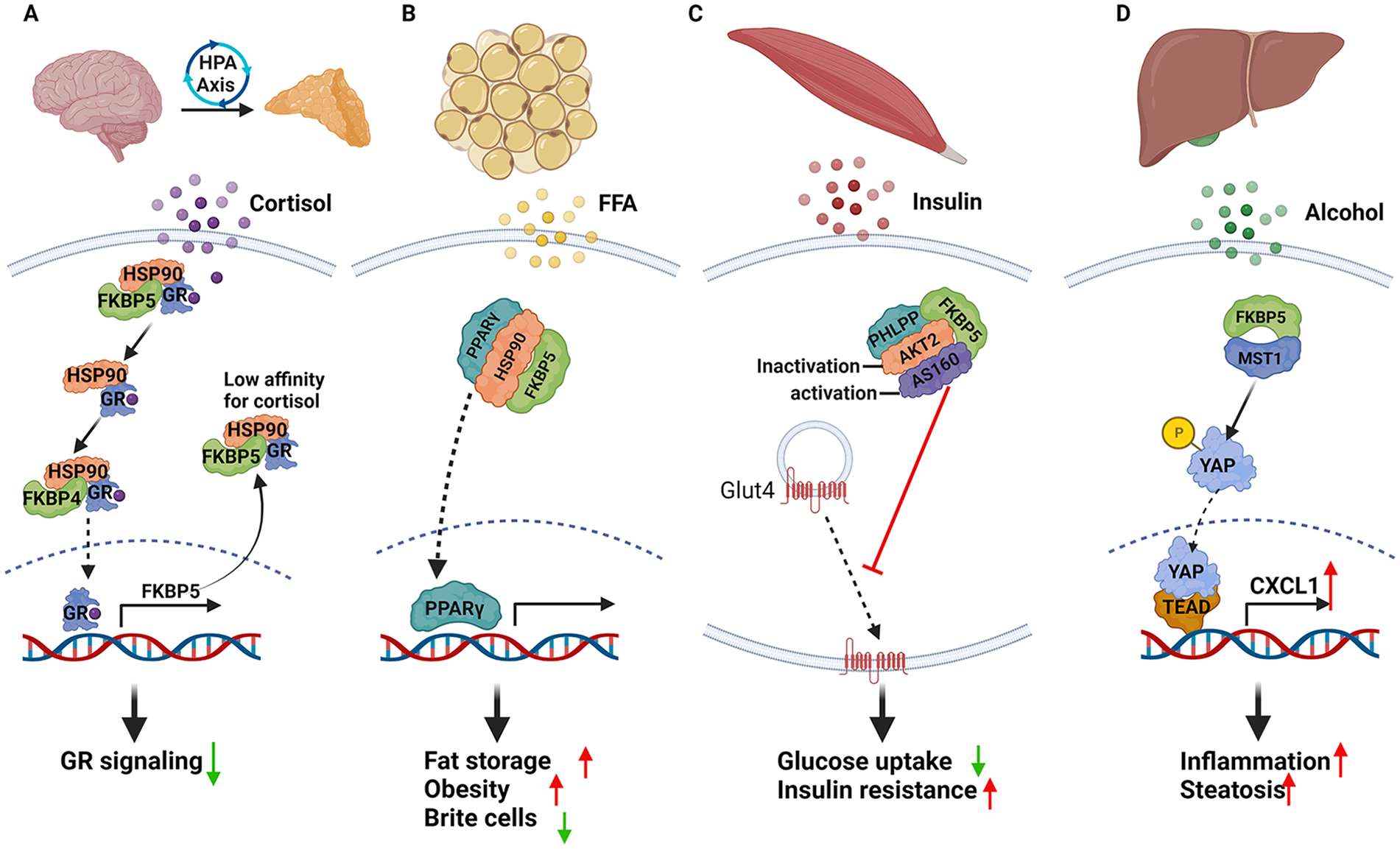
Schematic diagram of FKBP5 and its roles in mediating cellular function in different organs. (a) As a co-chaperone protein, FKBP5 can bind with other proteins to enact downstream signaling and mediate cellular function. The FKBP5 TPR domain interacts with the highly conserved HSP90 MEEDV motif3,34 and is further involved in regulating the GR and other SR signaling.3,5,35 Once formed, the FKBP5–HSP90–GR/SR complex exerts the effects on steroid hormone binding, nuclear translocation, and activation of the target genes. 5 (b) In adipose tissue, FKBP5 controls adipogenesis through its effect as a positive regulator of PPARγ, a key transcription factor in the adiopogenic process and as a negative regulator of GR through AKT and P38 MAP kinase. 6 (c) In skeletal muscle, FKBP5 regulates phosphorylation of AKT2 and its downstream AS160, a key signaling protein involved in insulin-stimulated glucose transport in skeletal muscle. The deficiency of FKBP5 improves insulin resistance and increases cellular glucose uptake. 7 (d) In alcoholic liver, the FKBP5 expression is upregulated by alcohol. FKBP5 interacts with MST1 affecting its ability to phosphorylate YAP, thus translocating YAP from the cytosol to the nucleus leading to TEAD1 activation. Activation of TEAD1 increases the expression of its target gene CXCL1, a neutrophil recruiter, causing hepatic inflammation. 36
FKBP5 and molecular signaling pathways in mediating cellular function
As a co-chaperone protein, FKBP5 can bind with other proteins to enact downstream signaling and mediate cellular function. 5 FKBP5 is known to be a co-chaperone of the HSP90 complex, a ubiquitous chaperone protein in most cells. 3 The FKBP5 TPR domain interacts and binds with the highly conserved HSP90 MEEDV motif.3,34 The binding between FKBP5 and HSP90 is essential in regulating the GR and other SR signaling, such as progesterone receptor (PR), androgen receptor (AR), and mineralocorticoid receptor (MR).3,5,35 Once formed, the FKBP5–HSP90–GR/SR complex exerts effects on steroid hormone binding, nuclear translocation, and activation of the target genes. 5 The affinity of the SR to bind to the TPR domain of FKBP5 and HSP90 is varied. 35 GR and PR display the most sensitive and apparent reaction to the TPR domain, whereas AR’s activity is moderately impaired by most cofactors. 35 Similar with its consequence on the GR, FKBP5 exerts an inhibitory effect on the PR and MR activities.5,37,38 Conversely, the AR is activated by the FKBP5 chaperone complex.39,40
FKBP5 can also bind and alter the phosphorylation state of its target proteins. Glycogen synthase kinase 3β (GSK3β), a serine–threonine kinase, regulates energy homeostasis and plays an important role in the pathogenesis of many diseases.41,42 The activity of the GSK3β depends on its phosphorylated state; the phosphorylation at serine 9 inhibits its enzymatic activity and activates the downstream signaling pathways. 34 The phosphorylation of GSK3β is tightly controlled by the upstream kinases such as AKT or protein kinase B or Cyclin-dependent kinase 5 (CDK5) and protein phosphatases such as protein phosphatase 1 and 2A.43–45 FKBP5, through its PPIase domain, binds with GSK3β and modulates the composition of the GSK3β’s complex by associating with the phosphatase PP2A and the CDK5. 41 As a result, FKBP5 increases the phosphorylation of GSK3β at the serine 9 site, inhibits the GSK3β activity, and activates the GSK3β’s downstream targets such as β-catenin and Tau,41,45 influencing various cellular processes.
FKBP5 negatively regulates all three isoforms of the serine/threonine protein kinase AKT (AKT1, AKT2, and AKT3). 7 The activity of AKT is regulated by its phosphorylation and FKBP5 acts as a scaffolding protein to modulate of AKT’s phosphorylated status. Specifically, FKBP5 functions to recruit protein phosphatase, pleckstrin homology (PH) domain, and leucine-rich repeat protein phosphatases and facilitate its dephosphorylation of AKT. 46 AKT is a critical regulator involved in insulin signaling and glucose metabolism,47,48 cell cycle,49,50 cell survival,51,52 glycogen synthesis, 43 and autophagy.53,54 By regulating the phosphorylation of the AKT, FKBP5, therefore, affects numerous cellular functions that are vital for cell growth, metabolism, and survival.
NF-κB, the nuclear factor kappa-light-chain-enhancer of activated B cells, is a protein complex with essential roles in controlling multiple cellular processes such as inflammation, proliferation, maturation, differentiation, survival, and apoptosis.55–57 It is ubiquitously found and acts as a stress response to external stimuli in multiple cell types. 58 NF-κB plays an important role in the immune response process 59 and is associated with inflammatory and metabolic diseases. 60 The NF-κB subunits, comprising the NF-κB complex, reside in the cytosol during an inactive state. 61 Upon activation during cellular stress, it leads to an interaction with the IκB kinase (IKK) complex resulting in the phosphorylation of IκB and its degradation. 61 The degradation leads to the nuclear translocation of the remaining NF-κB dimer, where it binds to the DNA consensus sequence and activates the transcription of its target genes. 61 FKBP5 was described as a regulator of NF-κB signaling. Several members of the IKK complex, IKKα, IKKβ, and IKKγ, have been identified to interact with FKBP5 involved in NF-κB signaling activation.62–65 FKBP5 could positively affect the formation and activation of the IKK complex and further promote NF-κB signaling.3,5,66
The Hippo pathway is an important regulator of cellular differentiation, proliferation, and homeostasis. 67 The pathway responds to cellular stress by the activation of several upstream serine/threonine kinases, such as MST1/2 and large tumor suppressor 1 and 2 (LATS1/2), leading to the phosphorylation of the protein targets Yes1 associated transcriptional regulator (YAP) and transcriptional coactivator with PDZ-binding motif (TAZ), which promotes their degradation and cytosolic retention and inhibits the activation of TEAD-mediated transcription. 67 The alterations of the Hippo signaling pathway are associated with various diseases and metabolic dysfunction.68,69 FKBP5 has been identified to interact with YAP upstream kinase, MST1, affecting its ability to phosphorylate YAP 36 (Figure 1). By inhibiting YAP phosphorylation, FKBP5 promotes YAP nuclear translocation and enhances TEAD activation through its role as TEAD transcriptional co-activator. 70
In summary, FKBP5 can bind to proteins in multiple pathways which regulate various cellular functions. The binding, in some instances, affects the post-translational modification (i.e., phosphorylation) of the protein targets and dysregulates the downstream cellular signaling pathways. The alterations of FKBP5, therefore, may lead to diverse consequences affecting cellular and tissue dysfunction.
FKBP5 and its function in adipose tissue
As diverse signaling pathways are involved in the regulation of homeostatic systems, FKBP5 plays a significant role in mediating the crosstalk between stress and metabolic disorders. Several studies suggested the important role of FKBP5 in regulating whole-body energy metabolism, based on its expression primarily in the metabolically active tissues, such as the liver, adipose tissue, small intestine, ovary, thymus, heart, and kidney.9,36,71,72 Adipose tissue serves as the energy storage and thermal regulation. It is also an endocrine organ with the ability to secret adipokines, a group of active molecules involved in the regulation of metabolic homeostasis, immune responses, and energy balance.73,74 The adipose tissue can be categorized based on its distribution, subcutaneous, and visceral adipose tissues. 75 Central rather than peripheral adiposity is a risk factor for metabolic diseases.75,76 The limited expandability of the adipose tissue in the subcutaneous area causes an unacceptable adipose cell expansion and insulin resistance.75,77 The expansion of adipose tissue by increasing adipogenesis distributes excess fat between newly differentiated adipocytes and decreases the number of inflammatory cytokines producing adipocytes. 75 Adipogenesis is a multi-step and tightly regulated process when the pluripotent mesenchymal stem cells (MSCs), upon stimulation, commit to the changes into the adipocyte lineage. 75 The initial phase is the conversion of the MSCs into pre-adipocytes, which then differentiate into mature adipocytes with specific functions, such as lipid storage, lipid synthesis, insulin sensitivity, and the secretion of adipokines. The defect in the process leads to adipose tissue dysfunction and insulin resistance. 75
One of FKBP5’s functions in mediating metabolic processes is its effect on adipocyte differentiation.
74
During the clonal expansion phase of adipocyte differentiation in 3T3-L1 cells, the expression of FKBP5 transcript and its protein level exhibit similar temporal kinetics.
78
Its expression is essential to cellular adipogenesis for the transformation of pre-adipocytes into mature, lipid-bearing cells.6,9 FKBP5 expression level increases during 3T3-L1 preadipocyte differentiation and it rapidly shuttles from mitochondria to the nucleus at the onset of adipogenesis, the process regulated by the second messenger cyclic adenosine monophosphate (cAMP) and protein kinase A (PKA) signaling pathways.
74
When the adipogenesis is activated, cAMP–PKA regulates the rapid nuclear translocation of FKBP5, which progressively increases its interaction with GR and restrains its transcriptional capacity.
74
Knockdown of
The effect of FKBP5 in human adipose tissue is studied to explore the effects of adipose tissue insulin resistance induced by GC.
72
Paired biopsies from abdominal subcutaneous and visceral adipose tissues were obtained from patients undergoing elective surgery.
72
The protein expression of FKBP5 is increased in a dose-dependent manner when both subcutaneous and visceral adipose tissues are treated with dexamethasone. Notably, the increase in FKBP5 protein was significantly higher in visceral adipose tissue treated with dexamethasone compared to subcutaneous tissue.
72
The change in
FKBP5 and its association with several metabolic diseases
DNA methylation is an epigenetic modification of DNA that is important for the normal regulation of transcription, embryonic development, genomic imprinting, genome stability, and chromatin structure. Aberrations in the machinery responsible for DNA methylation patterns and chromatin structure also contribute to human disease.
80
Methylation of the
As a stress response protein, FKBP5 appears to be a molecular mediator linking stress with metabolic phenotypes. 83 A comprehensive review of the role of FKBP5, stress, and metabolic disorders has been described elsewhere. 2 Such association would be predicted that individuals carrying a high percentage of CpG methylation in FKBP5 DNA are more susceptible to metabolic diseases when exposed to external stimuli or stresses. Single nucleotide polymorphisms (SNPs) are variations in the genome, and they are responsible for rendering susceptibility to specific diseases.22,84–86 Of interest, there is also an association between the SNPs in the FKBP5 region and metabolic phenotypes. The rs2817056 and rs2395635 are associated with T2D and impaired 2-h OGTT, respectively. In addition, three SNPs, rs1334894, rs9380525, and rs9368881, are associated with the level of high-density lipoprotein (HDL) -cholesterol, while six SNPs are linked to serum triglycerides (rs9394309, rs10947563, rs7763535, rs2395635, rs6912833, and rs4713904). 72 Another piece of evidence confirming the role of FKBP5 variants in metabolic regulation comes from studies investigating the association of the FKBP5 rs1360780 genotype on weight loss outcomes after bariatric surgery.87,88 Patients with the T allele of the rs1360780 polymorphism, the variant with an increase in FKBP5 responsivity, have approximately 20% less excess weight loss and 10% less total weight loss compared with those of the alternate C allele during the follow-up.83,88
In another study, the
FKBP5 and metabolic dysfunction-associated steatotic liver disease
A recent study discovered a dysregulation of
Recent genome-wide association studies (GWAS) investigating MASLD have not identified any significant signals related to FKBP5,95,96 suggesting that the gene may not play a prominent role in the genetic predisposition to MASLD. However, GWAS encompass numerous genetic loci: most disease-associated variants have small effect sizes. Identifying core genes directly related to the disease with clear biological relevance can be challenging. While GWAS studies may not provide strong support for the involvement of FKBP5 in MASLD, the findings from the mechanistic studies outlined above suggested its role in MASLD pathogenesis.
FKBP5 in alcohol drinking and alcohol-associated liver disease
Excessive alcohol use (EAU) is one of the most significant risk factors for health problems.
97
Genetic variants of FKBP5 are correlated with an increased risk of alcohol dependence and EAU.98–101 Moreover, FKBP5 is related to alcohol withdrawal severity. One study revealed that minor alleles of rs3800373 (G), rs9296158 (A), rs1360780 (T), and rs9470080 (T) in FKBP5 were significantly associated with lower Clinical Institute Withdrawal Assessment for Alcohol-Revised scores, whereas the minor alleles of rs3777747 (G) and rs9380524 (A) were associated with higher scores in 399 alcohol-dependent inpatients with alcohol consumption.
102
The haplotype-based analyses also showed an association with alcohol withdrawal severity.
102
ALD is a complex disorder that develops as a consequence of EAU. Recently, our group identified an FKBP5–YAP–TEAD1–CXCL1 axis in the pathogenesis of ALD.
36
An increase in the expression of FKBP5 at the transcript and the protein levels is also found in the liver of mice fed with alcohol and in patients with ALD.
36
The loss of FKBP5 protects against alcohol-induced hepatic steatosis and reduces hepatic neutrophil infiltration and the level of hepatic inflammatory cytokines.
36
In alcohol-fed mice, the upregulation of FKBP5 expression by alcohol is secondary to the downregulation of methylation level at its 5′ UTR promoter region.
36
An increase in FKBP5 expression leads to an induction of transcription factor TEA Domain Transcription Factor 1 (TEAD1) through the Hippo signaling pathway. FKBP5 interacts with mammalian Ste20-like kinase 1 (MST1) affecting its ability to phosphorylate YAP, thus translocating YAP from the cytosol to the nucleus leading to TEAD1 activation. Activation of TEAD1 increases the expression of its target gene C-X-C Motif Chemokine Ligand 1 (
Conclusion and future perspective
The expression of FKBP5 is widely distributed throughout various tissues in the human body. Alterations in FKBP5 expression can result in downstream effects and contribute to organ dysfunction. This review focuses on the role of FKBP5 in cellular stress response and its function as a chaperone protein in mediating molecular signaling pathways. We provided an overview of its involvement in several metabolic pathways and specifically addressed its implications in metabolism-associated diseases. The selective FKBP5 ligands SAFit1 and SAFit2 have been employed in several in vivo disease models as selective pharmacological inhibitors of FKBP5. 2 Chronic treatment with SAFit2 has similar effects to that of FKBP5 deletion on the restoration of metabolic function and improving glucose tolerance. 7 Further studies investigating the potential relevance of FKBP5 to metabolic diseases such as MASLD and ALD would be highly beneficial. There is still much to uncover regarding FKBP5’s involvement in the pathogenesis and progression of these diseases. Conducting additional investigations can provide valuable insights into the specific mechanisms by which FKBP5 contributes to metabolic dysfunction and organ damage in MASLD and ALD.
Footnotes
Roles of authors
JM and ZY searched for relevant articles and drafted the manuscript. NH, HG, and YJ provided critical comments on the draft, and ZY and SL reviewed and finalized the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: ZY is supported by NIH K01AA26385, R01AA030993, Indiana University Research Support Fund Grant (IU RSFG), the Ralph W. and Grace M. Showalter Research Trust and the Indiana University School of Medicine, and Indiana Institute for Medical Research (IIMR) and the Central Society for Clinical and Translational Research (CSCTR); JM is supported in part by NIH K99AA031067 and The Indiana Clinical and Translational Sciences Institute postdoctoral challenge grant, SL is supported in part by R01 AA025208, U01 AA026917, UH2/UH3 AA026903, R01AA030312, U01AA026817, VA Merit Award 1I01CX000361, and Dean’s Scholar in Medical Research, Indiana University School of Medicine.
